Carbon Sequestration in Mixed Deciduous Forests: The Influence of Tree Size and Species Composition Derived from Model Experiments
Abstract
1. Introduction
- Is it possible to reproduce daily carbon dynamics and carbon pools of a mixed deciduous forest in Germany with an individual-based forest model by integrating EC and inventory data?
- What is the contribution of different tree species to the overall productivity of the forest stand?
- What is the role of tree size for overall productivity of the forest, i.e., have a few larger trees a higher contribution to the productivity than many small trees?
2. Materials and Methods
2.1. Study Area
2.2. Inventory Data
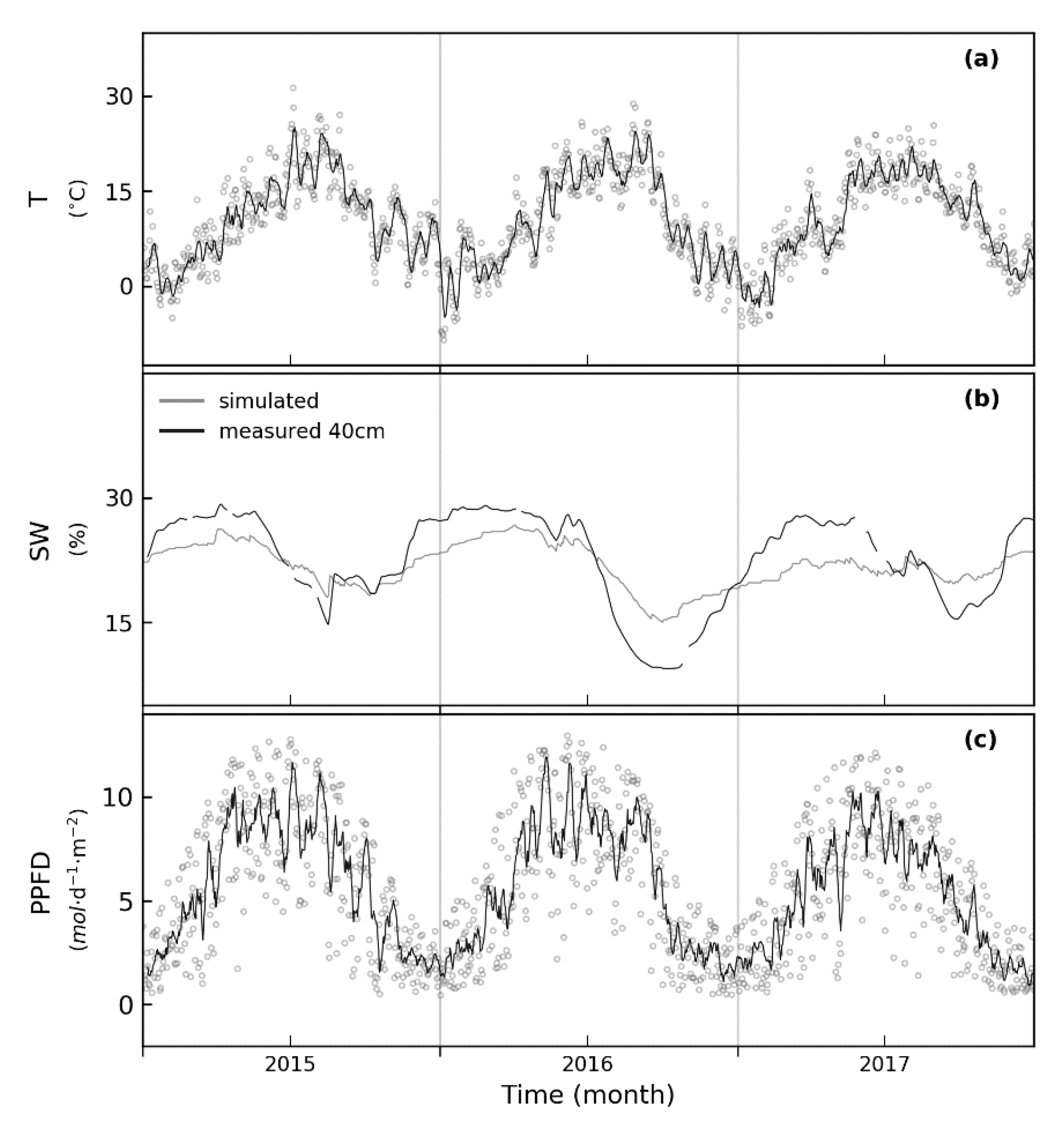
2.3. Environmental Data
2.4. Eddy Covariance Measurements
2.5. The Forest Model FORMIND
2.5.1. General Model Description
2.5.2. Model Setup
2.6. Tree Size Classes and Productivity Index
2.7. Virtual Experiment with Species Composition and Forest Structure
3. Results
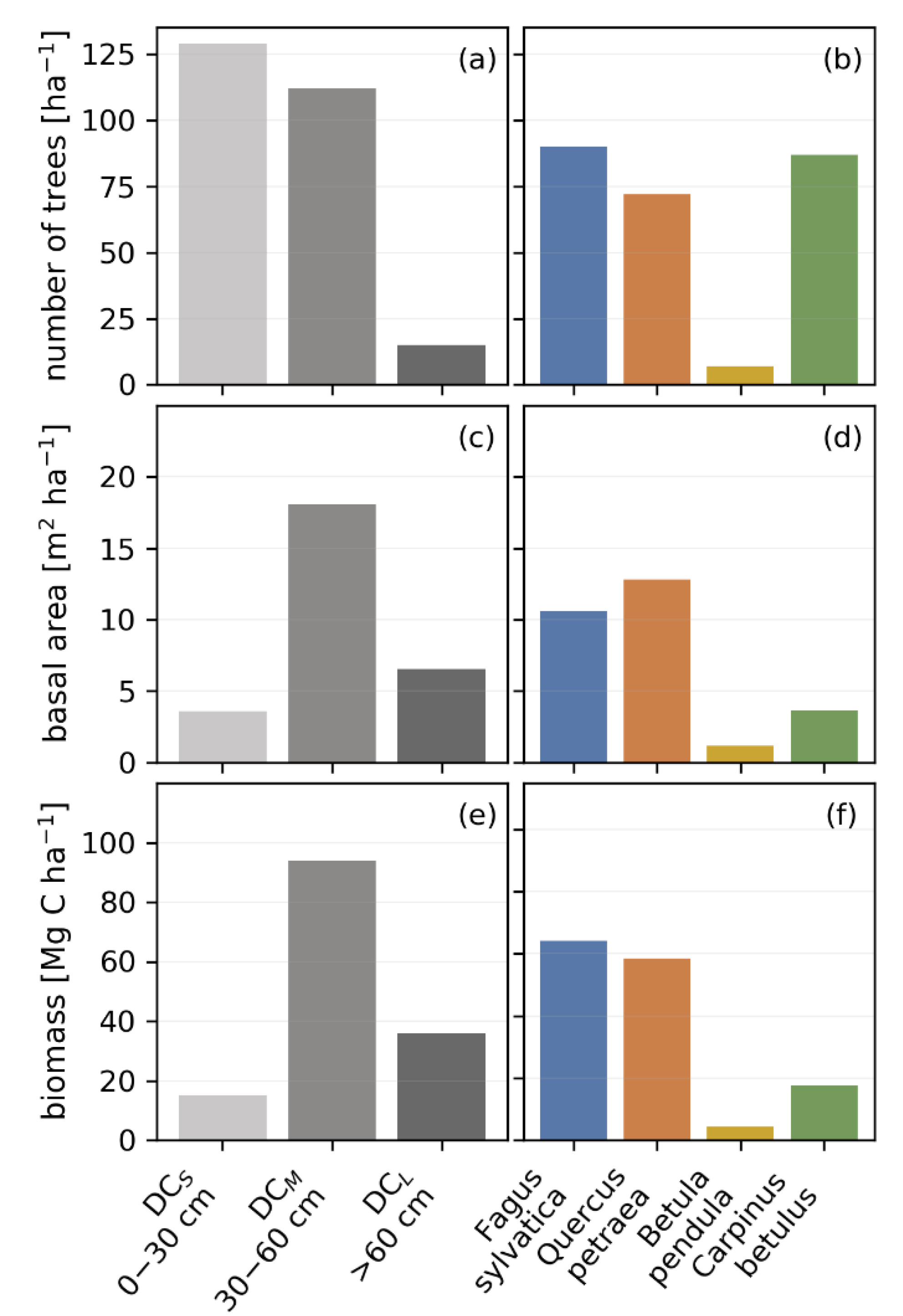
3.1. Biomass, Stem Size, Basal Area and Species Distribution Derived from Inventory Data
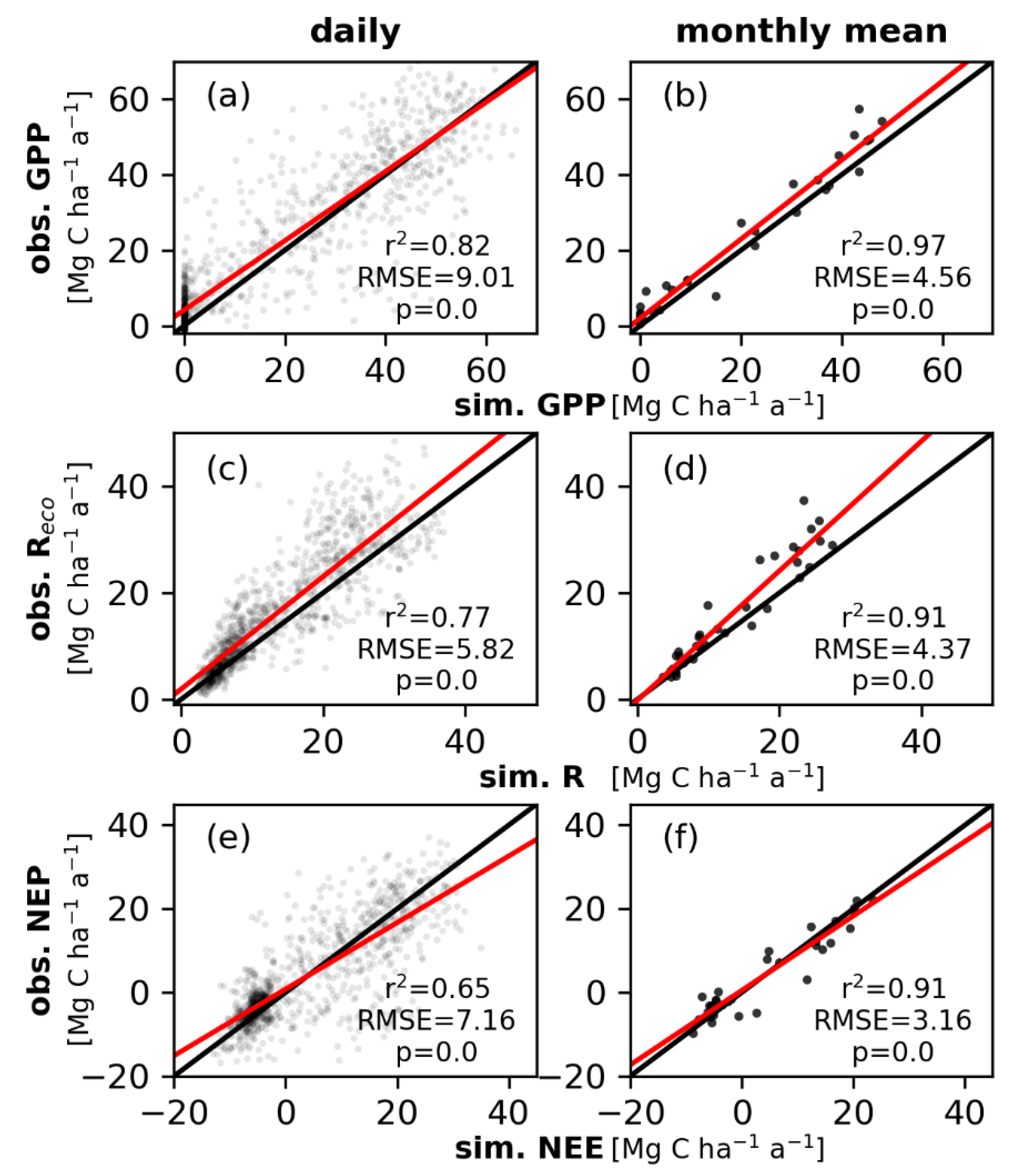
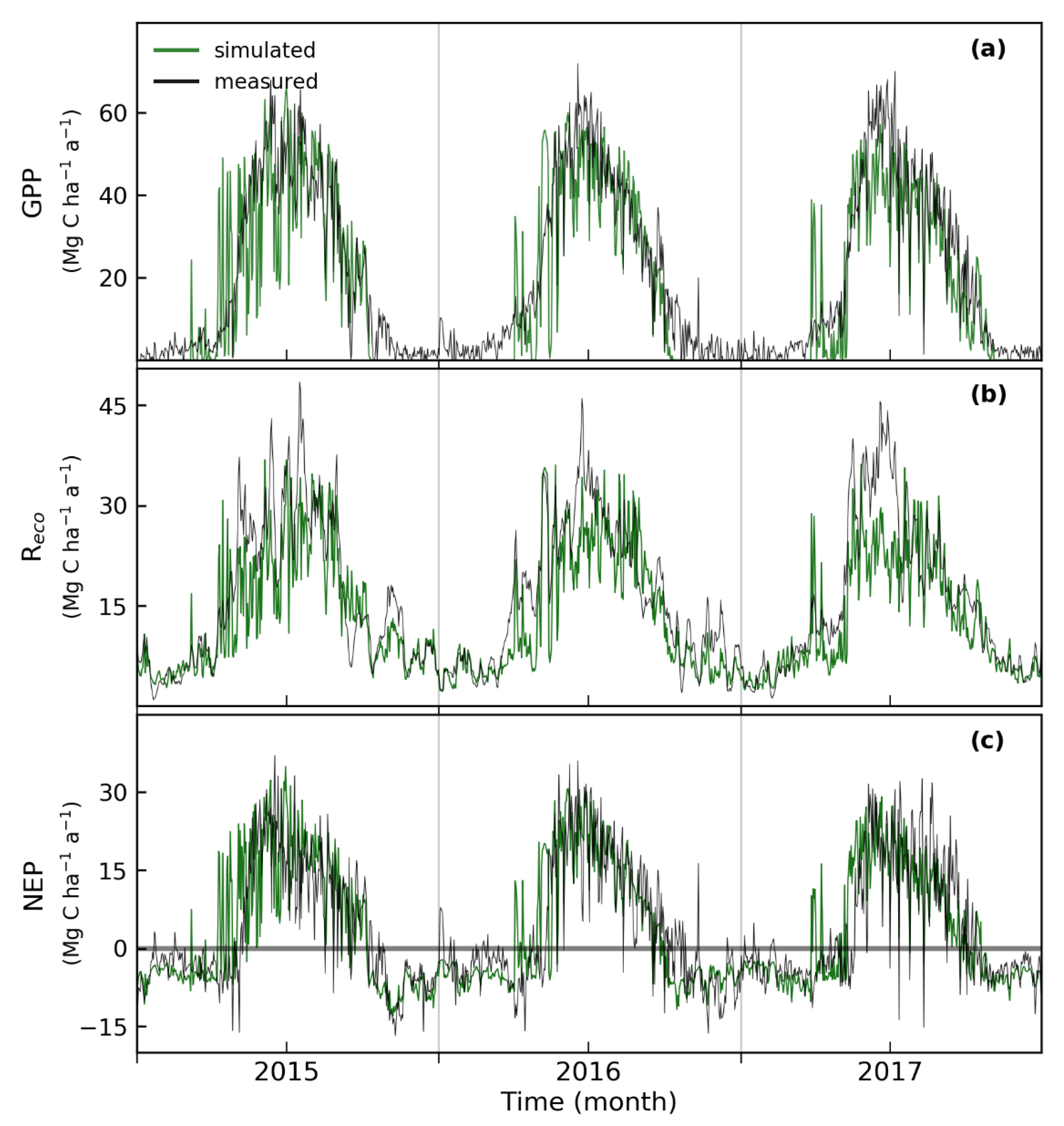
3.2. Simulated and Observed Daily Carbon Fluxes
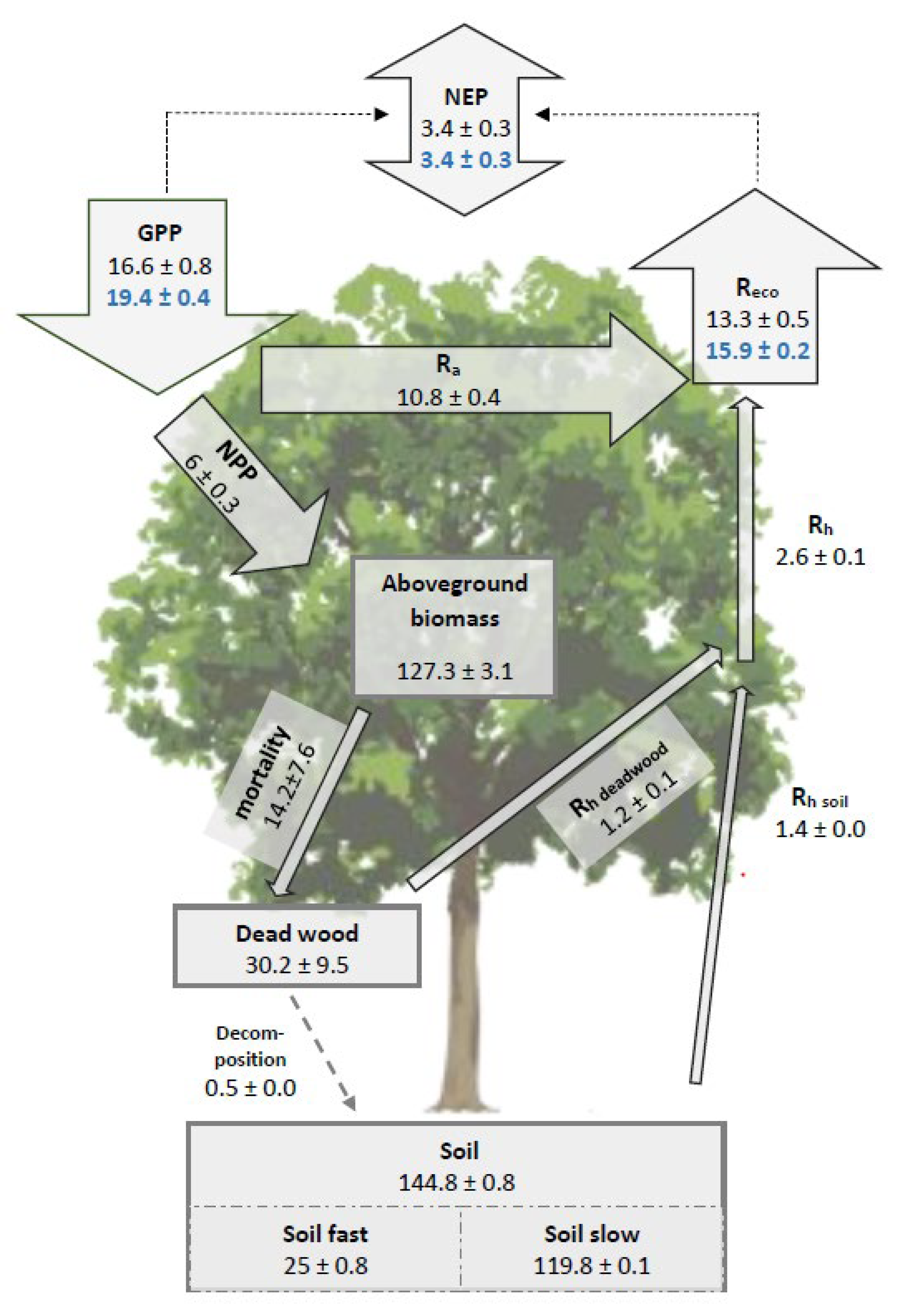
3.3. The Simulated Full Carbon Balance of a Temperate Mixed Deciduous Forest
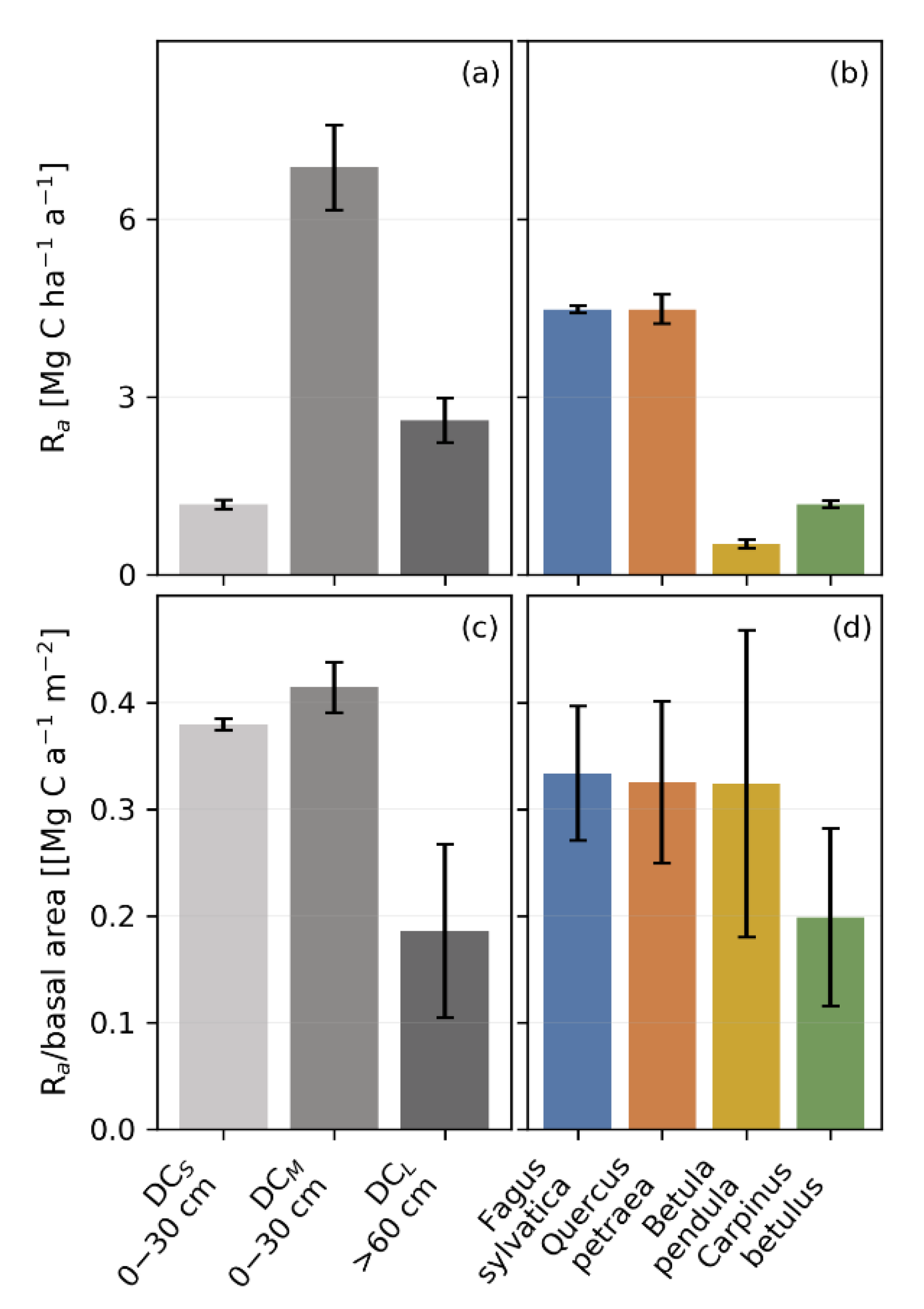
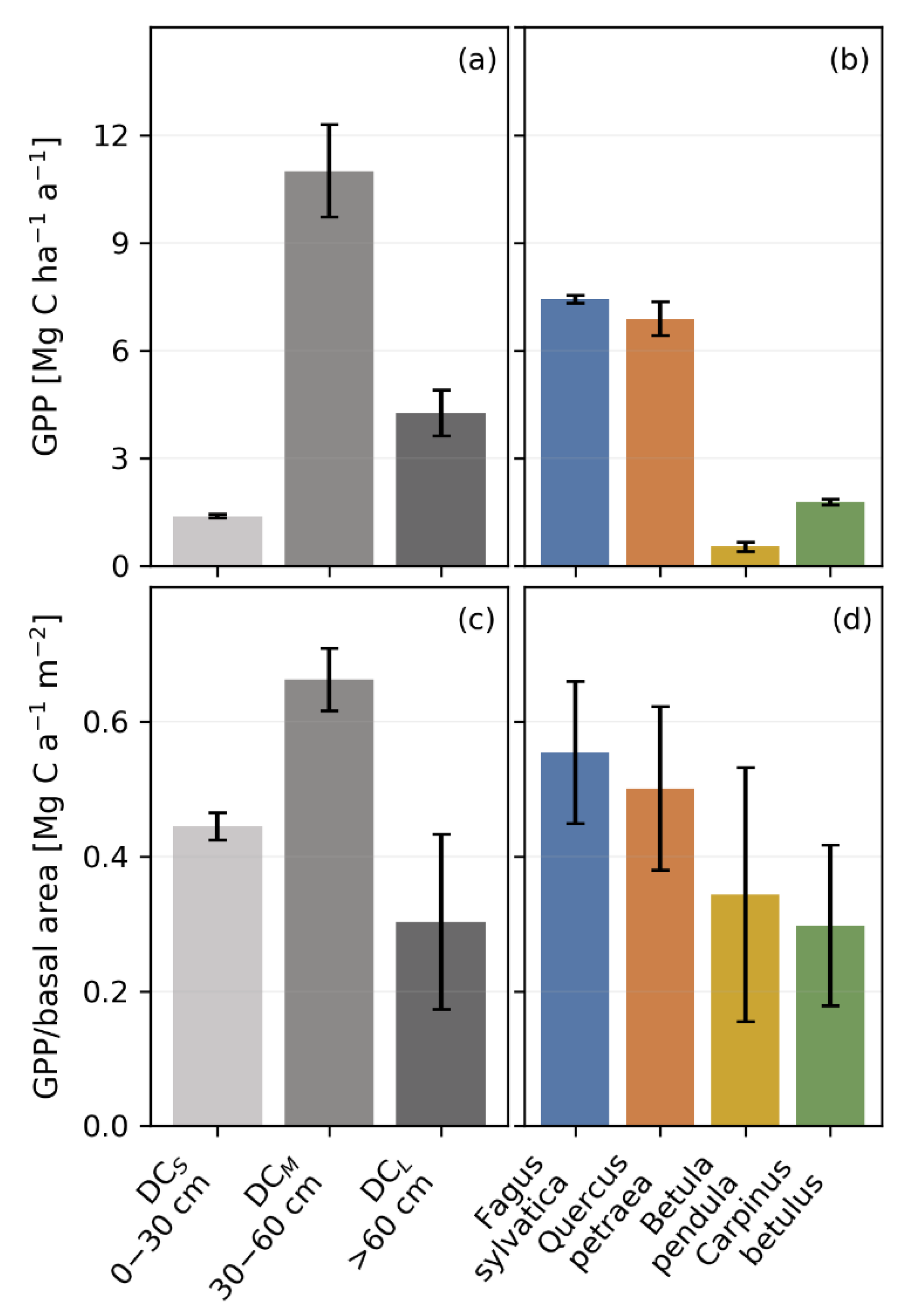
3.4. Productivity and Autotrophic Respiration across Tree Size Classes Derived from Model Simulation
3.5. Productivity and Autotrophic Respiration across Species Derived from Model Simulation
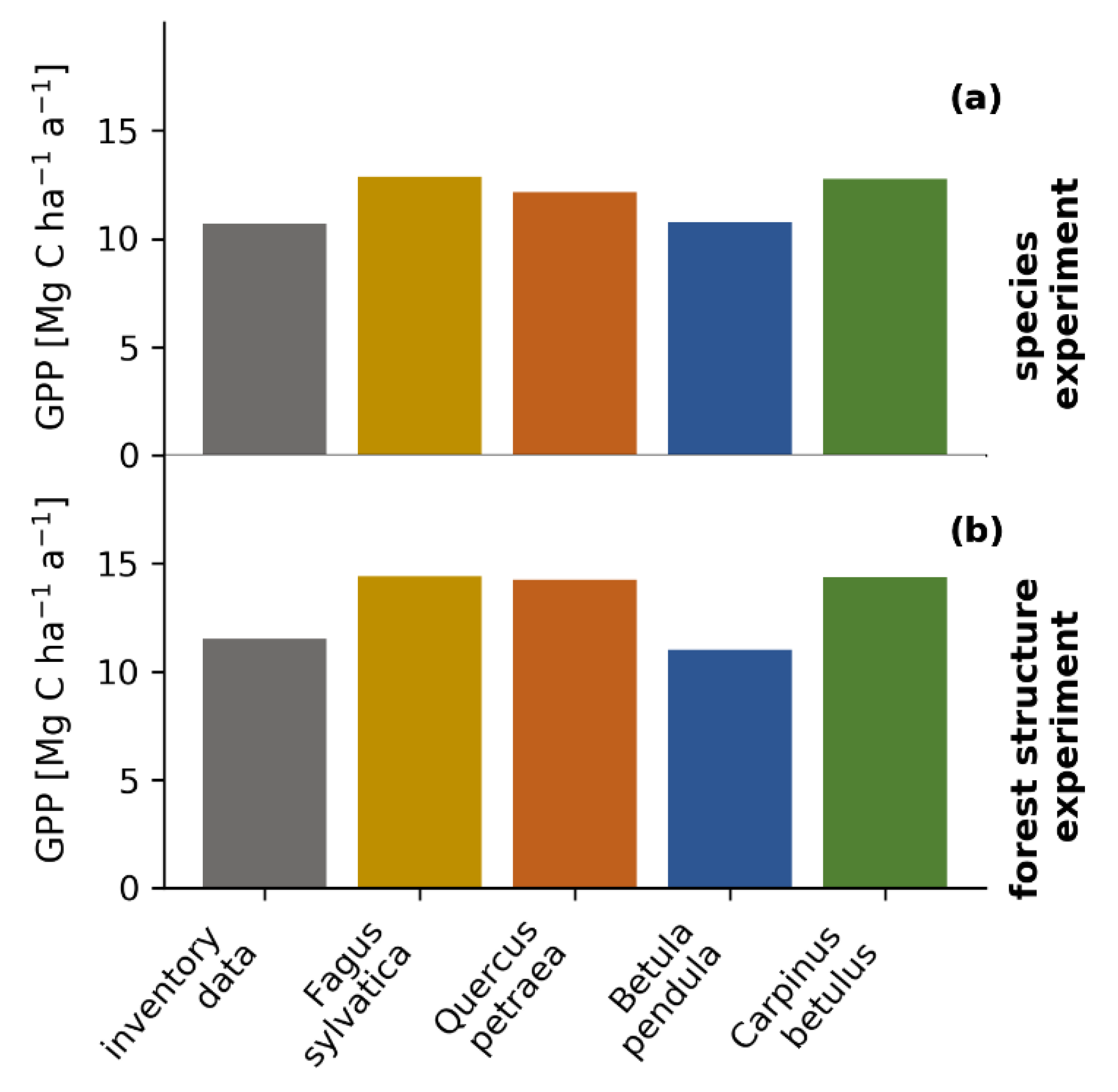
3.6. Virtual Experiments: The Influence of Species Composition and Forest Structure on Forest Productivity
4. Discussion
4.1. Simulated Daily Carbon Fluxes and Uncertainties
4.2. The Carbon Fluxes of Mixed Temperate Forests
4.3. The Impact of Tree Size and Forest Structure on Forest Productivity
4.4. Forest Productivity for Different Tree Species
4.5. The Benefit of Modelling Forest Carbon Fluxes
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Measured Variables at the EC Tower
| Variable Measured | Abbreviation | Unit | Instrument Type, Manufacturer |
|---|---|---|---|
| global radiation | SWD SWDR | W m−2 | CNR4, Kipp & Zonen, Delft, The Netherlands NR01, Hukseflux, Delft, The Netherlands |
| photosynthetic photon flux density | PPFD | µmol (photons) m−2 s−1 | LI-COR, Lincoln, NE, USA |
| air temperature | Tair | °C/K | HMP155, Vaisala, Helsinki, Finnland |
| precipitation | precip precip_back | mm | Thies 5.4032.35.008, Adolf Thies GmbH & Co. KG, Göttingen, Germany Thies 54000, Adolf Thies GmbH & Co. KG, Göttingen, Germany |
| soil water content | SM | Vol.% | CS616, 30cm length, Campbell Scientific, Shepshed, UK |
Appendix B
Appendix B.1. The Carbon Flux Module
Appendix B.2. Tree Photosynthesis
Appendix B.3. Tree Respiration
Appendix C. Model Parameter
Model Parameter Calibration
| Parameter | Fagus sylvatica | Quercus petraea | Betula pendula | Carpinus betulus | |
|---|---|---|---|---|---|
| Productivity | |||||
| pmax | max. photoproducitvity of leaf (µmol (CO2) s−1 m−2) | 15.768 | 20.244 | 22.672 | 15.768 |
| α | slope of light response curve (µmol (CO2) µmol(photons)−1) | 0.1288 | 0.0736 | 0.0728 | 0.1288 |
| Temperature | |||||
| Topt | optimal temperature for photosynthesis (°C) | 20.8 | |||
| Tsig | width of new temperature curve (°C) | 10.1 | |||
| Q10 | constant for temperature-dependent respiration | 2.12 | |||
| Tref | Reference temperature (°C) | 18.4 | |||
| Water | |||||
| SWpwp | permanent wilting point (vol-%) | 7.8 | |||
| SWfc | field capacity (vol-%) | 25.8 | |||
| ks | Fully saturated conductivity ms−1 | 0.0000061 | |||
| kl | Interception contant | 0.1 | |||
| ps | Pore size distribution | 0.105 | |||
| por | Porosity of the soil (vol-%) | 50.1 | |||
| SWInit | Initial soil water content (vol-%) | 22.24 | |||
| Θr | Residual soil water content (vol-%) | 1.5 | |||
| Establishment | |||||
| Nseeds | Number of global seeds per ha−1 a−1 (estimated) | 18 | 12 | 10 | 18 |
| Parameter | Fagus sylvatica | Quercus | Betula spp. | Carpinus betulus | |
|---|---|---|---|---|---|
| Biomass | |||||
| b1 | biomass calculation [72] | 1.202 | 1.151 | 1.091 | 1.202 |
| b2 | 5.727 | 5.187 | 6.394 | 5.727 | |
| d1 | Growth curve [72] | 4.70 × 10−3 | 7.06 × 10−3 | 3.74 × 10−3 | 4.70 × 10−3 |
| d2 | 1.252 | 0.703 | 1.445 | 1.252 | |
| d3 | 1.39 | 1.184 | 1.145 | 1.39 | |
| Geometry | |||||
| l0 | LAI-dbh relation [73] | 6.1 | 5.4 | 5.3 | 6.1 |
| l1 | 0 | 0 | 0 | 0 | |
| h0 | Height-dbh relation [72] | 1.916 | 1.879 | 1.711 | 1.916 |
| h1 | 61.036 | 45.341 | 51.488 | 61.036 | |
| c1 | Crown-dbh relation [72] | 0.155 | 0.173 | 0.207 | 0.155 |
| c2 | 0.125 | 0.054 | 1.760 | 0.125 | |
| c3 | 0.066 | 0.066 | 0.277 | 0.066 | |
| f0 | Form factor-dbh relation | 0.571 | 0.631 | 0.499 | 0.571 |
| f1 | 0.181 | 0.227 | 0.097 | 0.181 | |
| Mortality | |||||
| m0 | max. mortality at establishment | 0.00890 | 0.00657 | 0.04841 | 0.0890 |
| m1 | slope of mortality | −0.761 | −0.950 | −0.210 | −0.761 |
| r2 | [72] | 0.001 | 0.002 | 0.018 | 0.001 |
| Light and Establishment | |||||
| k | Light extinction factor | 0.7 | 0.7 | 0.7 | 0.7 |
| m | Transmission coefficient of leaves [74] | 0.1 | 0.1 | 0.1 | 0.1 |
| Imin | Min. light intensity% to establish [30] | 0.3 | 0.3 | 0.3 | 0.3 |
Appendix D. Autotrophic Respiration across Tree Size Classes and Species
Appendix E. Further Discussion: Inventory Data
References
- Fawzy, S.; Osman, A.I.; Doran, J.; Rooney, D.W. Strategies for mitigation of climate change: A review. Environ. Chem. Lett. 2020, 18, 2069–2094. [Google Scholar] [CrossRef]
- Bonan, G.B. Forests and Climate Change: Forcings, Feedbacks, and the Climate Benefits of Forests. Science 2008, 320, 1444–1449. [Google Scholar] [CrossRef]
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A Large and Persistent Carbon Sink in the World’s Forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef]
- Bösch, M.; Elsasser, P.; Franz, K.; Lorenz, M.; Moning, C.; Olschewski, R.; Rödl, A.; Schneider, H.; Schröppel, B.; Weller, P. Forest ecosystem services in rural areas of Germany: Insights from the national TEEB study. Ecosyst. Serv. 2018, 31, 77–83. [Google Scholar] [CrossRef]
- Wellbrock, N.; Grüneberg, E.; Riedel, T.; Polley, H. Carbon stocks in tree biomass and soils of German forests. Cent. Eur. For. J. 2017, 63, 105–112. [Google Scholar] [CrossRef]
- Friedlingstein, P.; Jones, M.W.; O’Sullivan, M.; Andrew, R.M.; Hauck, J.; Peters, G.P.; Peters, W.; Pongratz, J.; Sitch, S.; Le Quéré, C.; et al. Global Carbon Budget 2019. Earth Syst. Sci. Data 2019, 11, 1783–1838. [Google Scholar] [CrossRef]
- Harris, N.L.; Gibbs, D.A.; Baccini, A.; Birdsey, R.A.; de Bruin, S.; Farina, M.; Fatoyinbo, L.; Hansen, M.C.; Herold, M.; Houghton, R.A.; et al. Global maps of twenty-first century forest carbon fluxes. Nat. Clim. Chang. 2021. [Google Scholar] [CrossRef]
- Liu, S. Quantifying the spatial details of carbon sequestration potential and performance. In Carbon Sequestration and Its Role in the Global Carbon Cycle; Mcpherson, B.J., Sundquist, E.T., Eds.; American Geophysical Union: Washington, DC, USA, 2009; Volume 183, pp. 117–128. [Google Scholar]
- Luyssaert, S.; Inglima, I.; Jung, M.; Richardson, A.D.; Reichstein, M.; Papale, D.; Piao, S.L.; Schulze, E.D.; Wingate, L.; Matteucci, G.; et al. CO2 balance of boreal, temperate, and tropical forests derived from a global database. Glob. Chang. Biol. 2007, 13, 2509–2537. [Google Scholar] [CrossRef]
- Shanin, V.; Komarov, A.; Mäkipä, R. Tree species composition affects productivity and carbon dynamics of different site types in boreal forests. Eur. J. For. Res. 2014, 133, 273–286. [Google Scholar] [CrossRef][Green Version]
- Ryan, M.G.; Binkley, D.; Fownes, J.H. Age-Related Decline in Forest Productivity: Pattern and Process. Adv. Ecol. Res. 1997, 27, 213–262. [Google Scholar] [CrossRef]
- Keith, H.; Lindenmayer, D.; MacKey, B.; Blair, D.; Carter, L.; McBurney, L.; Okada, S.; Konishi-Nagano, T. Managing temperate forests for carbon storage: Impacts of logging versus forest protection on carbon stocks. Ecosphere 2014, 5. [Google Scholar] [CrossRef]
- Mund, M.; Schulze, E.D. Impacts of Forest Management on the Carbon Budget of European Beech (Fagus sylvatica) Forests. Available online: https://www.researchgate.net/publication/42088941_Impacts_of_forest_management_on_the_carbon_budget_of_European_Beech_Fagus_sylvatica_forests (accessed on 19 February 2021).
- Vande Walle, I.; Mussche, S.; Samson, R.; Lust, N.; Lemeur, R. The above- and belowground carbon pools of two mixed deciduous forest stands located in East-Flanders (Belgium). Ann. For. Sci. 2001, 58, 507–517. [Google Scholar] [CrossRef]
- Teets, A.; Fraver, S.; Hollinger, D.Y.; Weiskittel, A.R.; Seymour, R.S.; Richardson, A.D. Linking annual tree growth with eddy-flux measures of net ecosystem productivity across twenty years of observation in a mixed conifer forest. Agric. For. Meteorol. 2018, 249, 479–487. [Google Scholar] [CrossRef]
- Baldocchi, D. Measuring fluxes of trace gases and energy between ecosystems and the atmosphere-the state and future of the eddy covariance method. Glob. Chang. Biol. 2014, 20, 3600–3609. [Google Scholar] [CrossRef]
- Schmid, H.P. Source areas for scalars and scalar fluxes. Bound.-Layer Meteorol. 1994, 67, 293–318. [Google Scholar] [CrossRef]
- Medvigy, D.; Wofsy, S.C.; Munger, J.W.; Hollinger, D.Y.; Moorcroft, P.R. Mechanistic scaling of ecosystem function and dynamics in space and time: Ecosystem Demography model version 2. J. Geophys. Res. Biogeosci. 2009, 114, 1–21. [Google Scholar] [CrossRef]
- Baldocchi, D.; Falge, E.; Gu, L.; Olson, R.; Hollinger, D.; Running, S.; Anthoni, P.; Bernhofer, C.; Davis, K.; Evans, R.; et al. FLUXNET: A New Tool to Study the Temporal and Spatial Variability of Ecosystem-Scale Carbon Dioxide, Water Vapor, and Energy Flux Densities. Bull. Am. Meteorol. Soc. 2001, 82, 2415–2434. [Google Scholar] [CrossRef]
- Falge, E.; Baldocchi, D.; Tenhunen, J.; Aubinet, M.; Bakwin, P.; Berbigier, P.; Bernhofer, C.; Burba, G.; Clement, R.; Davis, K.J.; et al. Seasonality of ecosystem respiration and gross primary production as derived from FLUXNET measurements. Agric. For. Meteorol. 2002, 113, 53–74. [Google Scholar] [CrossRef]
- Reichstein, M.; Falge, E.; Baldocchi, D.; Papale, D.; Aubinet, M.; Berbigier, P.; Bernhofer, C.; Buchmann, N.; Gilmanov, T.; Granier, A.; et al. On the separation of net ecosystem exchange into assimilation and ecosystem respiration: Review and improved algorithm. Glob. Chang. Biol. 2005, 11, 1424–1439. [Google Scholar] [CrossRef]
- Grassi, G.; House, J.; Dentener, F.; Federici, S.; Den Elzen, M.; Penman, J. The key role of forests in meeting climate targets requires science for credible mitigation. Nat. Clim. Chang. 2017, 7, 220–226. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Trugman, A.T.; Badgley, G.; Anderson, C.M.; Bartuska, A.; Ciais, P.; Cullenward, D.; Field, C.B.; Freeman, J.; Goetz, S.J.; et al. Climate-driven risks to the climate mitigation potential of forests. Science 2020, 368, eaaz7005. [Google Scholar] [CrossRef]
- Ceccherini, G.; Duveiller, G.; Grassi, G.; Lemoine, G.; Avitabile, V.; Pilli, R.; Cescatti, A. Abrupt increase in harvested forest area over Europe after 2015. Nature 2020, 583, 72–77. [Google Scholar] [CrossRef]
- Shugart, H.H.; Wang, B.; Fischer, R.; Ma, J.; Fang, J.; Yan, X.; Huth, A.; Armstrong, A.H. Gap models and their individual-based relatives in the assessment of the consequences of global change. Environ. Res. Lett. 2018, 13, 033001. [Google Scholar] [CrossRef]
- Bugmann, H. A review of forest gap models. Clim. Chang. 2001, 51, 259–305. [Google Scholar] [CrossRef]
- Pretzsch, H.; Forrester, D.I.; Rötzer, T. Representation of species mixing in forest growth models: A review and perspective. Ecol. Modell. 2015, 313, 276–292. [Google Scholar] [CrossRef]
- Wollschläger, U.; Attinger, S.; Borchardt, D.; Brauns, M.; Cuntz, M.; Dietrich, P.; Fleckenstein, J.H.; Friese, K.; Friesen, J.; Harpke, A.; et al. The Bode hydrological observatory: A platform for integrated, interdisciplinary hydro-ecological research within the TERENO Harz/Central German Lowland Observatory. Environ. Earth Sci. 2017, 76, 29. [Google Scholar] [CrossRef]
- Paulick, S.; Dislich, C.; Homeier, J.; Fischer, R.; Huth, A. The carbon fluxes in different successional stages: Modelling the dynamics of tropical montane forests in South Ecuador. For. Ecosyst. 2017, 4, 5. [Google Scholar] [CrossRef]
- Bohn, F.J.; Frank, K.; Huth, A. Of climate and its resulting tree growth: Simulating the productivity of temperate forests. Ecol. Modell. 2014, 278, 9–17. [Google Scholar] [CrossRef]
- Maidment, D. Handbook of Hydrology; McGrawHill Inc.: New York, NY, USA, 1993; Volume 141, ISBN 9780070397323. [Google Scholar]
- Zacharias, S.; Bogena, H.; Samaniego, L.; Mauder, M.; Fuß, R.; Pütz, T.; Frenzel, M.; Schwank, M.; Baessler, C.; Butterbach-Bahl, K.; et al. A Network of Terrestrial Environmental Observatories in Germany. Vadose Zone J. 2011, 10, 955–973. [Google Scholar] [CrossRef]
- Aubinet, M.; Grelle, A.; Ibrom, A.; Rannik, U.; Moncrieff, J.; Foken, T.; Kowalski, A.S.; Martin, P.H.; Berbigier, P.; Bernhofer, C.; et al. Estimates of the Annual Net Carbon and Water Exchange of Forests: The EUROFLUX Methodology. Adv. Ecol. Res. 2000, 30, 113–175. [Google Scholar] [CrossRef]
- Rebmann, C.; Aubinet, M.; Schmid, H.; Arriga, N.; Aurela, M.; Burba, G.; Clement, R.; De Ligne, A.; Fratini, G.; Gielen, B.; et al. ICOS eddy covariance flux-station site setup: A review. Int. Agrophys. 2018, 32, 471–494. [Google Scholar] [CrossRef]
- Fratini, G.; Mauder, M. Towards a consistent eddy-covariance processing: An intercomparison of EddyPro and TK3. Atmos. Meas. Tech. 2014, 7, 2273–2281. [Google Scholar] [CrossRef]
- Burba, G.; Anderson, D. Introduction to the Eddy Covariance Method: General Guidelines and Conventional Workflow; LI-COR Biosciences: Lincoln, NE, USA, 2007. [Google Scholar] [CrossRef]
- Wutzler, T.; Lucas-Moffat, A.; Migliavacca, M.; Knauer, J.; Sickel, K.; Šigut, L.; Menzer, O.; Reichstein, M. Basic and extensible post-processing of eddy covariance flux data with REddyProc. Biogeosciences 2018, 15, 5015–5030. [Google Scholar] [CrossRef]
- Moritz, S.; Bartz-Beielstein, T. Imputets: Time series missing value imputation in R. R J. 2017, 9, 207–218. [Google Scholar] [CrossRef]
- Chapin, F.S.; Woodwell, G.M.; Randerson, J.T.; Rastetter, E.B.; Lovett, G.M.; Baldocchi, D.D.; Clark, D.A.; Harmon, M.E.; Schimel, D.S.; Valentini, R.; et al. Reconciling Carbon-cycle Concepts, Terminology, and Methods. Ecosystems 2006, 9, 1041–1050. [Google Scholar] [CrossRef]
- Fischer, R.; Bohn, F.; Dantas de Paula, M.; Dislich, C.; Groeneveld, J.; Gutiérrez, A.G.; Kazmierczak, M.; Knapp, N.; Lehmann, S.; Paulick, S.; et al. Lessons learned from applying a forest gap model to understand ecosystem and carbon dynamics of complex tropical forests. Ecol. Modell. 2016, 326, 124–133. [Google Scholar] [CrossRef]
- Rödig, E.; Huth, A.; Bohn, F.; Rebmann, C.; Cuntz, M. Estimating the carbon fluxes of forests with an individual-based forest model. For. Ecosyst. 2017, 4, 4. [Google Scholar] [CrossRef]
- Grüneberg, E.; Schöning, I.; Riek, W.; Ziche, D.; Evers, J. Carbon Stocks and Carbon Stock Changes in German Forest Soils; Springer: Cham, Switzerland, 2019; pp. 167–198. [Google Scholar]
- Campioli, M.; Malhi, Y.; Vicca, S.; Luyssaert, S.; Papale, D.; Peñuelas, J.; Reichstein, M.; Migliavacca, M.; Arain, M.A.; Janssens, I.A. Evaluating the convergence between eddy-covariance and biometric methods for assessing carbon budgets of forests. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Anderson-Teixeira, K.J.; Herrmann, V.; Banbury Morgan, R.; Bond-Lamberty, B.; Cook-Patton, S.C.; Ferson, A.E.; Muller-Landau, H.; Wang, M.M.H. Carbon cycling in mature and regrowth forests globally. Environ. Res. Lett. 2021. [Google Scholar] [CrossRef]
- Luyssaert, S.; Ciais, P.; Piao, S.L.; Schulze, E.D.; Jung, M.; Zaehle, S.; Schelhaas, M.J.; Reichstein, M.; Churkina, G.; Papale, D.; et al. The European carbon balance. Part 3: Forests. Glob. Chang. Biol. 2010, 16, 1429–1450. [Google Scholar] [CrossRef]
- Anthoni, P.M.; Knohl, A.; Rebmann, C.; Freibauer, A.; Mund, M.; Ziegler, W.; Kolle, O.; Schulze, E.-D. Forest and agricultural land-use-dependent CO2 exchange in Thuringia, Germany. Glob. Chang. Biol. 2004, 10, 2005–2019. [Google Scholar] [CrossRef]
- Prescher, A.K.; Grünwald, T.; Bernhofer, C. Land use regulates carbon budgets in eastern Germany: From NEE to NBP. Agric. For. Meteorol. 2010, 150, 1016–1025. [Google Scholar] [CrossRef]
- Bravo-Oviedo, A.; Pretzsch, H.; Ammer, C.; Andenmatten, E.; Barbati, A.; Barreiro, S.; Brang, P.; Bravo, F.; Coll, L.; Corona, P.; et al. European mixed forests: Definition and research perspectives. For. Syst. 2014, 23, 518–533. [Google Scholar] [CrossRef]
- Pretzsch, H.; Bielak, K.; Block, J.; Bruchwald, A.; Dieler, J.; Ehrhart, H.P.; Kohnle, U.; Nagel, J.; Spellmann, H.; Zasada, M.; et al. Productivity of mixed versus pure stands off oak (Quercus petraea (Matt.) Liebl. and Quercus robur L.) and European beech (Fagus sylvatica L.) along an ecological gradient. Eur. J. For. Res. 2013, 132, 263–280. [Google Scholar] [CrossRef]
- Chave, J.; Muller-Landau, H.C.; Baker, T.R.; Easdale, T.A.; Steege, H.; Webb, C.O. Regional and phylogenetic variation of wood density across 2456 neotropical tree species. Ecol. Appl. 2006, 16, 2356–2367. [Google Scholar] [CrossRef]
- Caspersen, J.P.; Vanderwel, M.C.; Cole, W.G.; Purves, D.W. How Stand Productivity Results from Size- and Competition-Dependent Growth and Mortality. PLoS ONE 2011, 6, e28660. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Luyssaert, S.; Richardson, A.D.; Kutsch, W.; Janssens, I.A. Steeper declines in forest photosynthesis than respiration explain age-driven decreases in forest growth. Proc. Natl. Acad. Sci. USA 2014, 111, 8856–8860. [Google Scholar] [CrossRef]
- Magnani, F.; Mencuccini, M.; Grace, J. Age-related decline in stand productivity: The role of structural acclimation under hydraulic constraints. Plant Cell Environ. 2000, 23, 251–263. [Google Scholar] [CrossRef]
- Ryan, M.G.; Yoder, B.J. Hydraulic Limits to Tree Height and Tree Growth. Bioscience 1997, 47, 235–242. [Google Scholar] [CrossRef]
- Yang, S.; Tyree, M.T. Hydraulic resistance in Acer saccharum shoots and its influence on leaf water potential and transpiration. Tree Physiol. 1993, 12, 231–242. [Google Scholar] [CrossRef]
- Mencuccini, M.; Grace, J. Developmental patterns of above-ground hydraulic conductance in a Scots pine (Pinus sylvestris L.) age sequence. Plant Cell Environ. 1996, 19, 939–948. [Google Scholar] [CrossRef]
- Schäfer, K.V.R.; Oren, R.; Tenhunen, J.D. The effect of tree height on crown level stomatal conductance. Plant Cell Environ. 2000, 23, 365–375. [Google Scholar] [CrossRef]
- Delzon, S.; Sartore, M.; Burlett, R.; Dewar, R.; Loustau, D. Hydraulic responses to height growth in maritime pine trees. Plant Cell Environ. 2004, 27, 1077–1087. [Google Scholar] [CrossRef]
- Zaehle, S. Effect of Height on Tree Hydraulic Conductance Incompletely Compensated by Xylem Tapering. Funct. Ecol. 2005, 19, 359–364. [Google Scholar] [CrossRef]
- Stephenson, N.L.; Das, A.J.; Condit, R.; Russo, S.E.; Baker, P.J.; Beckman, N.G.; Coomes, D.A.; Lines, E.R.; Morris, W.K.; Rüger, N.; et al. Rate of tree carbon accumulation increases continuously with tree size. Nature 2014, 507, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Köstler, J.N.; Brückner, E.; Bibelriether, H. Die Wurzeln der Waldbäume; Parey: Berlin, Germany, 1968; Volume 120. [Google Scholar]
- Kutschera, L.; Lichtenegger, E. Wurzelatlas Mitteleuropäischer Waldbäume und Sträucher; Leopold Stocker Verlag: Graz, Austria, 2002. [Google Scholar]
- Paquette, A.; Messier, C. The effect of biodiversity on tree productivity: From temperate to boreal forests. Glob. Ecol. Biogeogr. 2011, 20, 170–180. [Google Scholar] [CrossRef]
- Jacob, M.; Leuschner, C.; Thomas, F.M. Productivity of temperate broad-leaved forest stands differing in tree species diversity. Ann. For. Sci. 2010, 67. [Google Scholar] [CrossRef]
- Fischer, R.; Rödig, E.; Huth, A. Consequences of a Reduced Number of Plant Functional Types for the Simulation of Forest Productivity. Forests 2018, 9, 460. [Google Scholar] [CrossRef]
- Sakschewski, B.; von Bloh, W.; Boit, A.; Poorter, L.; Peña-Claros, M.; Heinke, J.; Joshi, J.; Thonicke, K. Resilience of Amazon forests emerges from plant trait diversity. Nat. Clim. Chang. 2016, 6, 1032–1036. [Google Scholar] [CrossRef]
- Forrester, D.I. A stand-level light interception model for horizontally and vertically heterogeneous canopies. Ecol. Modell. 2014, 276, 14–22. [Google Scholar] [CrossRef]
- Rödig, E.; Knapp, N.; Fischer, R.; Bohn, F.J.; Dubayah, R.; Tang, H.; Huth, A. From small-scale forest structure to Amazon-wide carbon estimates. Nat. Commun. 2019, 10, 5088. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, A.W.; Bradford, J.B.; Fraver, S.; Palik, B.J. Forest management for mitigation and adaptation to climate change: Insights from long-term silviculture experiments. For. Ecol. Manag. 2011, 262, 803–816. [Google Scholar] [CrossRef]
- Burkhart, H.E.; Temesgen, H. Forest observational studies: Data sources for analysing forest structure and dynamics. For. Ecol. Manag. 2014, 316, 1–148. [Google Scholar] [CrossRef]
- Fischer, R.; Armstrong, A.; Shugart, H.H.; Huth, A. Simulating the impacts of reduced rainfall on carbon stocks and net ecosystem exchange in a tropical forest. Environ. Model. Softw. 2014, 52, 200–206. [Google Scholar] [CrossRef]
- Schober, R. Ertragstafeln Wichtiger Baumarten bei Verschiedener Durchforstung, 4th ed.; Sauerländer: Frankfurt am Main, Germany, 1995. [Google Scholar]
- Breuer, L.; Eckhardt, K.; Frede, H.G. Plant parameter values for models in temperate climates. Ecol. Modell. 2003, 169, 237–293. [Google Scholar] [CrossRef]
- Larcher, W. Ökophysiologie der Pflanzen: Leben, Leistung und Streßbewältigung der Pflanzen in ihrer Umwelt; UTB: Stuttgart, Germany, 2001; Volume 42, ISBN 3-8252-8074-8. [Google Scholar]
| Location: Hohes Holz (DE-HoH) | 52°08′ N 11°22′ E | |
|---|---|---|
| Inventory 2018 | Inventory data | |
| Biomass (tree carbon content) (Mg C ha−1) | 145 | |
| Basal area (m2 ha−1) | 28.25 | |
| Mean stem diameter (dbh) (cm) | 32.4 | |
| Average stand height (crown tops) (m) | 23.5 | |
| Stand density (ha−1) | 260 | |
| Stand Age (a) * | 91 | |
| Climatological means 2015–2017 | Climate data | |
| Mean daytime temperature (°C) | 10.4 | |
| Mean PPFD (µmol m−2s−1) | 559.9 | |
| Annual precipitation sum (mm a−1) | 516.8 | |
| Carbon flux means 2015–2017 | Eddy Covariance estimates | |
| GPP annual mean (Mg C ha−1 a−1) | 19.4 | |
| Reco annual mean (Mg C ha−1 a−1) | 15.9 | |
| NEP annual mean (Mg C ha−1 a−1) | 3.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holtmann, A.; Huth, A.; Pohl, F.; Rebmann, C.; Fischer, R. Carbon Sequestration in Mixed Deciduous Forests: The Influence of Tree Size and Species Composition Derived from Model Experiments. Forests 2021, 12, 726. https://doi.org/10.3390/f12060726
Holtmann A, Huth A, Pohl F, Rebmann C, Fischer R. Carbon Sequestration in Mixed Deciduous Forests: The Influence of Tree Size and Species Composition Derived from Model Experiments. Forests. 2021; 12(6):726. https://doi.org/10.3390/f12060726
Chicago/Turabian StyleHoltmann, Anne, Andreas Huth, Felix Pohl, Corinna Rebmann, and Rico Fischer. 2021. "Carbon Sequestration in Mixed Deciduous Forests: The Influence of Tree Size and Species Composition Derived from Model Experiments" Forests 12, no. 6: 726. https://doi.org/10.3390/f12060726
APA StyleHoltmann, A., Huth, A., Pohl, F., Rebmann, C., & Fischer, R. (2021). Carbon Sequestration in Mixed Deciduous Forests: The Influence of Tree Size and Species Composition Derived from Model Experiments. Forests, 12(6), 726. https://doi.org/10.3390/f12060726






