Ants as Bioindicators of Riparian Ecological Health in Catalonian Rivers
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Design
2.2. Ant Sampling
2.3. Identification of River Typology
2.4. Assessment of the Pressure Gradient in Each River Type
2.5. Development of the Ant-Based Multimetric Index
2.6. Comparing the Ant-Based Index with a Traditional Physical and Structural-Based Index
3. Results
3.1. Ant Community
3.2. Functional and Compositional Metrics Response to Disturbance Gradient
3.3. Multimetric Ant Index for the Assessment of Ecological Health of Riparian Systems in the Upland and Lowland River Types of Catalonia
4. Discussion
4.1. Ant Community, Metrics and Response to Disturbance Gradient
4.2. Ant-Based MMI for Riparian Systems
4.3. Drawbacks and Strengths of the Method
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ward, J.V. The four-dimensional nature of lotic ecosystems. J. N. Am. Benthol. Soc. 1989, 8, 2–8. [Google Scholar] [CrossRef]
- Naiman, R.J.; Décamps, H. The Ecology of Interfaces: Riparian Zones. Annu. Rev. Ecol. Syst. 1997, 28, 621–658. [Google Scholar] [CrossRef]
- Riis, T.; Kelly-Quinn, M.; Aguiar, F.C.; Manolaki, P.; Bruno, D.; Bejarano, M.D.; Clerici, N.; Fernandes, M.R.; Franco, J.C.; Pettit, N.; et al. Global Overview of Ecosystem Services Provided by Riparian Vegetation. BioScience 2020, 70, 501–514. [Google Scholar] [CrossRef]
- Petts, G.E.; Amoros, C. Fluvial Hydrosystems; Chapman and Hall: London, UK, 1996; p. 322. [Google Scholar]
- Naiman, R.J.; Décamps, H.; Pollock, M. The role of riparian corridors in maintaining regional biodiversity. Ecol. Appl. 1993, 3, 209–212. [Google Scholar] [CrossRef]
- Corbacho, C.; Sánchez, J.M.; Costillo, E. Patterns of structural complexity and human disturbance of riparian vegetation in agricultural landscapes of Mediterranean area. Agric. Ecosyst. Environ. 2003, 95, 495–507. [Google Scholar] [CrossRef]
- Von Schiller, D.; Martí, E.; Riera, J.L.; Ribot, M.; Marks, J.C.; Sabater, F. Influence of land use on stream ecosystem function in a Mediterranean catchment. Freshw. Biol. 2008, 53, 2600–2612. [Google Scholar] [CrossRef]
- Fernandes, M.R.; Aguiar, F.C.; Ferreira, M.T. Assessing riparian vegetation structure and the influence of land use using landscape metrics and geostatistical tools. Landsc. Urban. Plan. 2011, 99, 166–177. [Google Scholar] [CrossRef]
- Aguiar, F.C.; Martins, M.J.; Silva, P.C.; Fernandes, M.R. Riverscapes downstream of hydropower dams: Effects of altered flows and historical land-use change. Landsc. Urban. Plan. 2016, 153, 83–98. [Google Scholar] [CrossRef]
- Karr, J.R.; Dudley, D.R. Ecological perspective on water quality goals. Environ. Manag. 1981, 5, 55–68. [Google Scholar] [CrossRef]
- Meyer, J.L. Stream health: Incorporating the human dimension to advance stream ecology. J. N. Am. Benthol. Soc. 1997, 16, 2. [Google Scholar] [CrossRef]
- Innis, S.A.; Naiman, R.J.; Elliott, S.R. Indicators and assessment methods for measuring the ecological integrity of semi-aquatic terrestrial environments. Hydrobiologia 2000, 422, 111–131. [Google Scholar] [CrossRef]
- Raven, P.J.; Fox, P.; Everard, M.; Holmes, N.T.H.; Dawson, F.H. River Habitat Survey: A new system for classifying rivers according to their habitat quality. In Freshwater Quality: Defining the Indefinable? Boon, P.J., Howell, D.L., Eds.; The Stationery Office: Edinburgh, UK, 1997; pp. 215–234. [Google Scholar]
- Munné, A.; Prat, N.; Solà, C.; Bonada, N.; Rieradevall, M. A simple field method for assessing the ecological quality of riparian habitat in rivers and streams: QBR index. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 147–163. [Google Scholar] [CrossRef]
- Del Tánago, M.G.; Jalón, D.G. Attributes for assessing the environmental quality of riparian zones. Limnetica 2006, 25, 389–402. [Google Scholar]
- Ollero, A.; Ballarín, D.; Díaz, E.; Mora, D.; Sánchez Fabre, M.; Acín, V.; Echev-Erría, M.T.; Granado, D.; Ibisate, A.L.; Sánchez Gil, L.; et al. Un índice hidrogeomorfológico (IHG) para la evaluación del estado ecológico de sistemas fluviales. Geographicalia 2007, 52, 113–141. [Google Scholar]
- Dziock, F.; Henle, K.; Foeckler, F.; Follner, K.; Scholz, M. Biological indicator systems in floodplains—A Review. Int. Rev. Hydrobiol. 2006, 91, 271–291. [Google Scholar] [CrossRef]
- Hölldobler, B.; Wilson, E.O. The Ants; Harvard University Press: Cambridge, UK, 1990; p. 732. [Google Scholar]
- Alonso, L.E.; Agosti, D. Biodiversity studies, monitoring and ants: An overview. In Standard Methods for Measuring and Monitoring Biodiversity; Agosti, D., Majer, J.D., Alonso, L.E., Schultz, T.R., Eds.; Smithsonian Institution Press: Washington, DC, USA; London, UK, 2000; p. 280. [Google Scholar]
- Underwood, E.C.; Fisher, B.L. The role of ants in conservation monitoring: If. when. and how. Biol. Conserv. 2006, 132, 166–182. [Google Scholar] [CrossRef]
- Ordóñez-Urbano, C.; Reyes-López, J.; Carpintero-Ortega, S. Estudio faunístico de los formícidos (Hymenoptera: Formicidae) asociados a los bosques de ribera en la provincia de Córdoba (España). Primeras aportaciones. Boletín Soc. Entomológica Aragonesa 2007, 40, 367–375. [Google Scholar]
- Glaser, F. Ants (Hymenoptera, Formicidae) in alpine floodplains—Ecological notes and conservation aspects. In Riverine Landscapes—Restoration—Flood Protection—Conservation. Proceedings of the Internationales LIFE-Symposium, Reutte-Breitenwang, Austria, 26–29 September 2005; Füreder, L., Sint, D., Vorauer, A., Eds.; Natur in Tirol. Naturkundliche Beiträge der Abteilung Umweltschutz: Reutte-Breitenwang, Austria, 2007; Volume 13, pp. 147–163. [Google Scholar]
- Dunn, R.R. Managing the tropical landscape: A comparison of the effects of logging and forest conversion to agriculture on ants, birds, and Lepidoptera. For. Ecol. Manag. 2004, 191, 215–224. [Google Scholar] [CrossRef]
- Crist, T.O. Biodiversity, species interactions, and functional roles of ants (Hymenoptera, Formicidae) in fragmented landscapes: A review. Myrmecol. News 2009, 12, 3–13. [Google Scholar]
- Andersen, A.N. Responses of ant communities to disturbance: Five principles for understanding the disturbance dynamics of a globally dominant faunal group. J. Anim. Ecol. 2019, 88, 1–13. [Google Scholar] [CrossRef]
- Jiménez-Carmona, F.; Heredia-Arévalo, A.M.; Reyes-López, J.L. Ants (Hymenoptera: Formicidae) as an indicator group of human environmental impact in the riparian forests of the Guadalquivir river (Andalusia, Spain). Ecol. Indic. 2020, 118, 106–762. [Google Scholar] [CrossRef]
- Folgarait, P.J. Ant biodiversity and its relationship to ecosystem functioning: A review. Biodivers. Conserv. 1998, 7, 1221–1244. [Google Scholar] [CrossRef]
- Paetzold, A.; Sabo, J.; Sadler, J.P.; Findlay, S.E.G.; Tockner, K. Aquatic–Terrestrial Subsidies along River Corridors. In Hydroecology and Ecohydrology: Past, Present and Future; Wood, P.J., Hannah, D.M., Sadler, J.P., Eds.; Wiley & Son, Lda.: Hoboken, NJ, USA, 2008; pp. 57–73. [Google Scholar]
- Del Toro, I.; Ribbons, R.R.; Pelini, S.L. The little things that run the world revisited: A reviwe of ant-mediated ecosystem services and disservices (Hymenoptera: Formicidae. Myrmecol. News 2012, 17, 133–146. [Google Scholar]
- Lawton, J.H.; Bifnell, D.E.; Bolton, B.; Blowmers, G.F.; Eggleton, P.; Hammond, P.M.; Hodda, M.; Holt, R.D.; Larsen, T.B.; Mawdsley, N.A.; et al. Biodiversity inventories, indicator taxa and effects of habitat modification in tropical forest. Nature 1998, 391, 72–76. [Google Scholar] [CrossRef]
- Leal, I.R.; Bieber, A.G.D.; Tabarelli, M.; Andersen, A.N. Biodiversity surrogacy: Indicator taxa as predictors of total species richness in Brazilian Atlantic forest and Caatinga. Biodivers. Conserv. 2010, 19, 3347–3360. [Google Scholar] [CrossRef]
- Jiménez-Carmona, F.; Carpinter, S.; Reyes-López, J.L. Ants (Hymenoptera: Formicidae) as surrogates for epigeic arthropods in Northen Andalusian ‘dehesas’ (Spain). Sociobiology 2020, 67, 201–212. [Google Scholar] [CrossRef]
- Majer, J.D. Ants as bioindicators of minesite rehabilitation land use and land conservation. Environ. Manag. 1983, 7, 375–383. [Google Scholar] [CrossRef]
- Andersen, A.N.; Hoffmann, B.; Müller, W.J.; Griffiths, A. Using ants as bioindicators in land management: Simplifying assessment of ant community responses. J. Appl. Ecol. 2002, 39, 8–17. [Google Scholar] [CrossRef]
- De Bruyn, L.A.L. Ants as bioindicators of soil function in rural environments. Agric. Ecosyst. Environ. 1999, 74, 1–3. [Google Scholar]
- Segat, J.C.; Vasconcellos, R.L.V.; Silva, D.P.; Baretta, D.; Cardoso, E.J.B.N. Ants as indicators of soil quality in an on-going recovery of riparian forests. For. Ecol. Manag. 2017, 404, 338–343. [Google Scholar] [CrossRef]
- Tiede, Y.; Schlautmann, J.; Donoso, D.A.; Wallis, C.I.B.; Bendix, J.; Brandl, R.; Farwig, N. Ants as indicators of environmental change and ecosystem processes. Ecol. Indic. 2017, 83, 527–537. [Google Scholar] [CrossRef]
- Peck, S.L.; Mcquaid, B.; Campbell, C.L. Using ant species (Hymenoptera: Formicidae) as a biological indicator of agroecosystem condition. Environ. Entomol. 1998, 27, 1102–1110. [Google Scholar] [CrossRef]
- Andersen, A.N.; Sparling, G.P. Ants as indicators of restoration success: Relationship with soil microbial biomass in the Australian seasonal tropics. Restor. Ecol. 1997, 5, 109–114. [Google Scholar] [CrossRef]
- Lawes, M.J.; Moore, A.M.; Andersen, A.N.; Preece, N.D.; Franklin, D.C. Ants as ecological indicators of rainforest restoration: Community convergence and the development of an Ant Forest Indicator Index in the Australian wet tropics. Ecol. Evol. 2017, 7, 8442–8455. [Google Scholar] [CrossRef]
- King, J.R.; Andersen, A.N.; Cutter, A.D. Ants as bioindicators of habitat disturbance: Validation of the functional group model for Australia’s humid tropics. Biodivers. Conserv. 1998, 7, 1627–1638. [Google Scholar] [CrossRef]
- Ives, C.D.; Hose, G.C.; Nipperess, D.A.; Taylor, M.P. Environmental and landscape factors influencing ant and plant diversity in a suburban riparian corridors. Landsc. Urban. Plan. 2011, 103, 372–382. [Google Scholar] [CrossRef]
- Hering, D.; Feld, C.K.; Moog, O.; Ofenbock, T. Cookbook for the development of a multimetric index for biological condition of aquatic ecosystems: Experiences from the European AQEM and STAR projects and related initiatives. In The Ecological Status of European Rivers: Evaluation and Intercalibration of Assessment Methods; Furse, M.T., Hering, D., Brabec, K., Buffagni, A., Sandin, L., Verdonschot, P.F.M., Eds.; Hydrobiologia; Springer: Dordrecht, The Netherlands, 2006; Volume 566, pp. 311–324. [Google Scholar]
- Van Oosterhout, M.P.; van der Velde, G. An advanced index of biotic integrity for use in tropical shallow lowland streams in Costa Rica: Fish assemblages as indicators of stream ecosystem health. Ecol. Indic. 2015, 48, 687–698. [Google Scholar] [CrossRef]
- Umetsu, C.A.; Aguiar, F.C.; Ferreira, M.T.; Cancian, L.F.; Camargo, A.F.M. Addressing bioassessment of tropical rivers using macrophytes: The case of Itanhaém Basin, São Paulo, Brazil. Aquat. Bot. 2018, 150, 53–63. [Google Scholar] [CrossRef]
- Ruaro, R.; Gubiani, E.A.; Hughes, R.M.; Mormul, R.P. Global trends and challenges in multimetric indices of biological condition. Ecol. Indic. 2020, 110, 105862. [Google Scholar] [CrossRef]
- Karr, J.R. Assessment of biotic integrity using fish communities. Fisheries 1981, 6, 21–27. [Google Scholar] [CrossRef]
- Bryce, S.A.; Hughes, R.M.; Kaufmann, P.R. Development of a bird integrity index: Using bird assemblages as indicators of riparian condition. Environ. Manag. 2002, 30, 294–310. [Google Scholar] [CrossRef] [PubMed]
- Golfieri, B.; Hardersen, S.; Maiolini, B.; Surian, N. Odonates as indicators of the ecological integrity of the river corridor: Development and application of the Odonate River Index (ORI) in northern Italy. Ecol. Indic. 2016, 61, 234–247. [Google Scholar] [CrossRef]
- Nelson, S.M.; Andersen, D.C. An assessment of riparian environmental quality by using butterflies and disturbance susceptibility scores. Southwest. Nat. 1994, 39, 137–142. [Google Scholar] [CrossRef]
- Murphy, K.J.; Castella, E.; Clément, B.; Hills, J.M.; Obrdlik, P.; Pulford, I.D.; Schneider, E.; Speight, M.C.D. Biotic indicators of riverine wetland ecosystem functioning. In Global Wetlands: Old World and New; Mitsch, W.J., Ed.; Elsevier: Amsterdam, The Netherlands, 1994; pp. 659–682. [Google Scholar]
- Gollan, J.R.; Reid, C.A.M.; Barnes, P.B.; Wilkie, L. The ratio of exotic-to-native dung beetles can indicate habitat quality in riparian restoration. Insect Conserv. Divers. 2011, 4, 123–131. [Google Scholar] [CrossRef]
- Andersen, A.N. A Classification of Australian Ant Communities, Based on Functional Groups Which Parallel Plant Life-Forms in Relation to Stress and Disturbance. J. Biogeogr. 1995, 22, 15–29. [Google Scholar] [CrossRef]
- Andersen, A.N. Using Ants as bioindicators: Multiscale Issues in Ant Community Ecology. Conserv. Ecol. 1997, 1, 8. [Google Scholar] [CrossRef]
- Roig, X.; Espadaler, X. Propuesta de grupos funcionales de hormigas para la Península Ibérica y Baleares, y su uso como bioindicadores. Iberomyrmex 2010, 2, 28–29. [Google Scholar]
- Philpott, S.M.; Perfecto, I.; Armbrecht, I.; Parr, C.L. Ant diversity and function in disturbed and changing habitats. In Ant Ecology; Lach, L., Parr, C.L., Abbott, K., Eds.; Oxford University Press: Oxford, UK, 2010; pp. 137–157. [Google Scholar]
- Hoffmann, B.D.; Andersen, A.N. Responses of ants to disturbance in Australia, with particular reference to functional groups. Austral. Ecol. 2003, 28, 444–464. [Google Scholar] [CrossRef]
- Meteocat. Meteorological Service of Catalonia. Available online: www.meteo.cat (accessed on 12 April 2019).
- Folch, R. La vegetació dels Països Catalans, 2nd ed.; Corrected and Extended; Ketres: Barcelona, Spain, 1986; p. 541. [Google Scholar]
- IDESCAT—Institut d’Estadística de Catalunya. Available online: https://www.idescat.cat/?lang=es (accessed on 12 April 2019).
- Retana, J.; Cerdá, X. Patterns of diversity and composition of Mediterranean ground ant communities tracking spatial and temporal variability in the thermal environment. Oecologia 2000, 123, 436–444. [Google Scholar] [CrossRef]
- Gómez, C.; Casellas, D.; Oliveras, J.; Bas, J.M. Structure of ground-foraging ant assemblages in relation to land-use change in the northwestern Mediterranean region. Biodivers. Conserv. 2003, 12, 2135–2146. [Google Scholar] [CrossRef]
- Angulo, E.; Boulay, R.; Ruano, R.; Tinaut, A.; Cerdá, X. Anthropogenic impacts in protected areas: Assessing the efficiency of conservation efforts using Mediterranean ant communities. PeerJ 2016, 4, e2773. [Google Scholar] [CrossRef]
- Majer, J.D. The use of pitfall traps for sampling ants—A critique. Mem. Mus. Vic. 1997, 56, 323–329. [Google Scholar] [CrossRef]
- Parr, C.L.; Chown, S.L. Inventory and Bioindicator Sampling: Testing Pitfall and Winkler Methods with Ants in a South African Savanna. J. Insect Conserv. 2001, 5, 27–36. [Google Scholar] [CrossRef]
- Tista, M.; Fiedler, K. How to evaluate and reduce sampling effort for ants. J. Insect Conserv. 2011, 15, 547–559. [Google Scholar] [CrossRef]
- Collingwood, C.; Prince, A. A guide to ants of continental Portugal (Hymenoptera: Formicidae). Bol. Da Soc. Port. De Entomol. 1998, 5, 1–49. [Google Scholar]
- Gómez, K.; Espadaler, X. Hormigas Ibéricas. 2007. Available online: http://www.hormigas.org (accessed on 19 September 2018).
- Galkowski, C.L.C.; Wegnez, R.B.P. Guía De Campo De Las Hormigas De Europa Occidental; Edición Española; Omega: Barcelona, Spain, 2017; p. 415. [Google Scholar]
- MacQueen, J.B. Some Methods for classification and Analysis of Multivariate Observations. In Proceedings of the 5th Berkeley Symposium on Mathematical Statistics and Probability, Berkeley, CA, USA, 21 June–18 July 1965; University of California Press: Berkeley, CA, USA, 1967; Volume 1, pp. 281–297. [Google Scholar]
- Rivers and Catchments of Europe—Catchment Characterisation Model (CCM). European Commission, Joint Research Centre (JRC) [Dataset]; OOPEC: Luxembourg, Luxembourg, 1 June 2007; Available online: http://data.europa.eu/89h/fe1878e8-7541-4c66-8453-afdae7469221 (accessed on 1 December 2019).
- Horton, R.E. Erosional development of streams and their drainage basins hydro-physical approach to quantitative morphology. Geol. Soc. Am. Bull. 1945, 56, 275–370. [Google Scholar] [CrossRef]
- Strahler, A.N. Hypsometric (area-altitude) analysis of erosional topology. Geol. Soc. Am. Bull. 1972, 63, 1117–1142. [Google Scholar] [CrossRef]
- Nagel, D.E.; Buffington, J.M.; Parkes, S.L.; Wenger, S.; Goode, J.R. A landscape Scale Valley Confinement Algorithm: Delineating Unconfined Valley Bottoms for Geomorphic, Aquatic, and Riparian Applications; Gen. Tech., Rep. RMRSGTR-321; U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2014; 42p.
- Johansen, K.; Phinn, S. Mapping structural parameters and species composition of riparian vegetation using IKONOS and Landsat ETM+ Data in Australian Tropical Savannahs. Photogramm. Eng. Remote Sens. 2006, 72, 71–80. [Google Scholar] [CrossRef]
- Clerici, N.; Weissteiner, C.J.; Paracchini, M.L.; Boschetti, L.; Baraldi, A.; Strobl, P. Pan-European distribution modelling of stream riparian zones based on multi-source Earth Observation data. Ecol. Indic. 2013, 24, 211–223. [Google Scholar] [CrossRef]
- European Commission. Directive 2000/60/EC of the European Parliament and of the council of 23rd October 2000 establishing a framework for community action in the field of water policy. In Official Journal of the European Communities; L327/1; European Commission: Brussels, Belgium, 2000. [Google Scholar]
- Arnan, X.; Cerdá, X.; Retana, J. Distinctive life traits and distribution along environmental gradients of dominant and subordinate Mediterranean ant species. Oecologia 2012, 170, 489–500. [Google Scholar] [CrossRef]
- Parr, C.L.; Dunn, R.R.; Sanders, N.J.; Weiser, M.D.; Photakis, M.; Bishop, T.R.; Fitzpatrick, M.C.; Arnan, X.; Baccaro, F.; Brandão, C.R.; et al. GlobalAnts: A new database on the geography of ant traits (Hymenoptera: Formicidae). Insect Conserv. Divers. 2007, 10, 5–20. [Google Scholar] [CrossRef]
- Torossian, C. Recherches sur la biologie et l’éthologie de Dolichoderus quadripunctatus (Hym. Form. Dolichoderidae). I. Étude des populations dans leur milieu naturel. Insectes Sociaux 1967, 14, 102–122. [Google Scholar]
- Gibb, H.; Sanders, N.J.; Dunn, R.R.; Arnan, X.; Baccaro, F.; Bishop, T.R.; Chick, L.; Donoso, D.; Fayle, T.M.; Glasier, J.; et al. The Global Ants Database. 2015. Available online: http://globalants.org/ (accessed on 2 June 2020).
- Retana, J.; Cerdá, X.; Alsina, A.; Bosch, J. Field observations of the ant Camponotus sylvaticus: Diet and activity patterns. Acta Oecologica 1988, 9, 101–109. [Google Scholar]
- Gómez, C.; Abril, S. Selective logging in public pine forests of the central Iberia Peninsula: Effects of the recovery process on ant assemblages. For. Ecol. Manag. 2011, 262, 1061–1066. [Google Scholar] [CrossRef]
- Arnan, X.; Cerdá, X.; Retana, J. Ant functional responses along environmental gradients. J. Anim. Ecol. 2014, 83, 1398–1408. [Google Scholar] [CrossRef] [PubMed]
- Czechowski, W. Around-nest “cemeteries” of Myrmica schencki Em. (Hymenoptera: Formicidae) their origin and a possible significance. Pol. J. Ecol. 2008, 56, 359–363. [Google Scholar]
- Cros, S.; Cerdá, X.; Retana, J. Spatial and temporal variations in the activity patterns of Mediterranean ant communities. Ecoscience 1997, 4, 269–278. [Google Scholar] [CrossRef]
- Cerda, X.; Retana, J.; Cros, S. Critical thermal limits in Mediterranean ant species: Trade-off between mortality risk and foraging performance. Funct. Ecol. 1998, 12, 45–55. [Google Scholar] [CrossRef]
- Pekas, A.; Tena, A.; Aguilar, A.; Garcia-Marí, F. Spatio-temporal patterns and interactions with honeydew-producing Hemiptera of ants in a Mediterranean citrus orchard. Agric. For. Entomol. 2011, 13, 89–97. [Google Scholar] [CrossRef]
- Ruiz, E.; Hoz-Martínez, M.; Martínez, M.D.; Hernández, J.M. Morphological study of the stridulatory organ in two species of Crematogaster genus: Crematogaster scutellaris (Olivier 1792) and Crematogaster auberti (Emery 1869) (Hymenoptera: Formicidae). Ann. De La Société Entomol. De Fr. 2006, 42, 99–105. [Google Scholar] [CrossRef]
- Van Sickle, J. Correlated metrics yield multimetric indices with inferior performance. Trans. Am. Fish. Soc. 2010, 139, 1802–1817. [Google Scholar] [CrossRef]
- Suárez, M.L.; Vidal-Abarca, M.R.; del Sánchez-Montoya, M.; Alba-Tercedor, J.P.; álvarez, M.; Avilés, J.; Bonada, N.; Casas, J.; Jáimez-Cuéllar, P.; Munneé, A.; et al. Las riberas de los ríos mediterráneos y su calidad: El uso del índice QBR. Limnetica 2002, 21, 135–148. [Google Scholar]
- Colwell, S.R.; Hix, D.M. Adaptation of the QBR index for use in riparian forests of central Ohio. In Proceedings,16th Central Hardwood Forest Conference; Jacobs, D.F., Michler, C.H., Eds.; West Lafayette, In Gen. Tech. Rep. NRS-P-24; US Department of Agriculture, Forest Service, Northern Research Station: Newtown Square, PA, USA, 2008. [Google Scholar]
- Kazoglou, Y.; Fotiadis, G.; Koutseri, I.; Vrahnakis, M. Assessment of Structural Components of Riparian Forest Vegetation of the Prespa Basin with the Means of the QBR Index; Balwois: Ohrid, Republic of Macedonia, 2010. [Google Scholar]
- Siromba, M.G.; Mesa, L.M. A method for assessing the ecological quality of riparian forests in subtropical Andean streams: QBR index. Ecol. Indic. 2012, 20, 324–331. [Google Scholar] [CrossRef]
- Andersen, A.N.; Fisher, A.; Hoffmann, B.D.; Read, J.L.; Richards, R. Use of terrestrial invertebrates for biodiversity monitoring in Australian rangelands, with particular reference to ants. Austral. Ecol. 2004, 29, 87–92. [Google Scholar] [CrossRef]
- Andersen, A.N.; Majer, J.D. Ants show the way down under: Invertebrates as bioindicators in land management. Frontier in Ecol. Environ. 2004, 2, 291–298. [Google Scholar] [CrossRef]
- Arnan, X.; Rodrigo, A.; Retana, J. Post-fire recovery of Mediterranean ground ant communities follows vegetation and dryness gradients. J. Biogeogr. 2006, 33, 1246–1258. [Google Scholar] [CrossRef]
- Rodrigo, A.; Retana, J. Post-fire recovery of ant communities in Submediterranean Pinus nigra forests. Ecography 2006, 29, 231–239. [Google Scholar] [CrossRef]
- Bishop, T.R.; Robertson, M.; Gibb, H.; van Rensburg, B.J.; Braschler, B.; Chown, S.L.; Foord, S.H.; Munyai, T.C.; Okey, I.; Tshivhandekano, P.G.; et al. Ant assemblages have darker and larger members in cold environments. Glob. Ecol. Biogeogr. 2006, 25, 1489–1499. [Google Scholar] [CrossRef]
- Margalef, R. Ecología; Omega: Barcelona, Spain, 1980; p. 951. [Google Scholar]
- Wetterer, J.K.; Espadaler, X.; Wetterer, A.L.; Cabral, S.G.M. Native and exotic ants of the Azores (Hymenoptera: Formicidae). Sociobiology 2004, 44, 265–297. [Google Scholar]
- Hoffmann, B.D. Using ants for rangeland monitoring: Global patterns in the responses of ant communities to grazing. Ecol. Indic. 2010, 10, 105–111. [Google Scholar] [CrossRef]
- Hevia, V.; Ortega, J.; Azcárate, F.M.; López, C.A.; González, J.A. Exploring the effect of soil management intensity on taxonomic and functional diversity of ants in Mediterranean olive groves. Agric. For. Entomol. 2019, 21, 109–118. [Google Scholar] [CrossRef]
- Sanders, N.J.; Gotelli, N.J.; Heller, N.E.; Gordon, D.M. Community disassembly by an invasive species. PNAS 2003, 100, 2474–2477. [Google Scholar] [CrossRef] [PubMed]
- Perfecto, I. Indirect and direct effects in a tropical agroecosystem: The maize-pest-ant system in Nicaragua. Ecology 1990, 71, 2125–2134. [Google Scholar] [CrossRef]
- Pereira, J.L.; Silva, A.A.; Picanco, M.C.; Barros, E.C.; Jakelaitis, A. Effects of herbicide and insecticide interaction on soil entomofauna under maize crop. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2005, 40, 45–54. [Google Scholar] [CrossRef]
- Ives, C.D.; Hose, G.C.; Nipperess, D.A.; Taylor, M.P. The influence of riparian corridor width on ant and plant assemblages in northern Sydney, Australia. Urban. Ecosyst. 2011, 14, 1–16. [Google Scholar] [CrossRef]
- Ives, C.D.; Taylor, M.P.; Nipperess, D.A.; Hose, G.C. Effect of catchment urbanization on ant diversity in remnant riparian corridors. Landsc. Urban. Plan. 2013, 110, 155–163. [Google Scholar] [CrossRef]
- Ellison, A.M.; Record, S.; Arguello, A.; Gotelli, N.J. Rapid inventory of the ant assemblage in a temperate hardwood forest: Species composition and assessment of sampling methods. Environ. Entomol. 2007, 36, 766–775. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Ellison, A.M.; Dunn, R.R.; Sander, N.J. Counting ants (Hymenoptera: Formicidae): Biodiversity sampling and statistical analysis for myrmecologists. Myrmecol. News 2011, 15, 13–19. [Google Scholar]
- Garcia-Martínez, M.A.; Valenzuela-González, J.E.; Escobar-Sarria, F.; López-Barrera, F.; Castaño-Meneses, G. The surrounding landscape influences the diversity of leaf-litter ants in riparian cloud forest remnants. PLoS ONE 2017, 12, e0172464. [Google Scholar] [CrossRef]
- Tagwireyi, P.; Sullivan, S.M.P. Riverine landscape patches influence trophic dynamics or riparian ants. River Res. Appl. 2016, 32, 1721–1729. [Google Scholar] [CrossRef]
- Barredo, J.I.; Caudullo, G.; Dosio, A. Mediterranean habitat loss under future climate conditions: Assessing impatcs in the Natura 2000 protected area network. Appl. Geogr. 2016, 75, 83–92. [Google Scholar] [CrossRef]
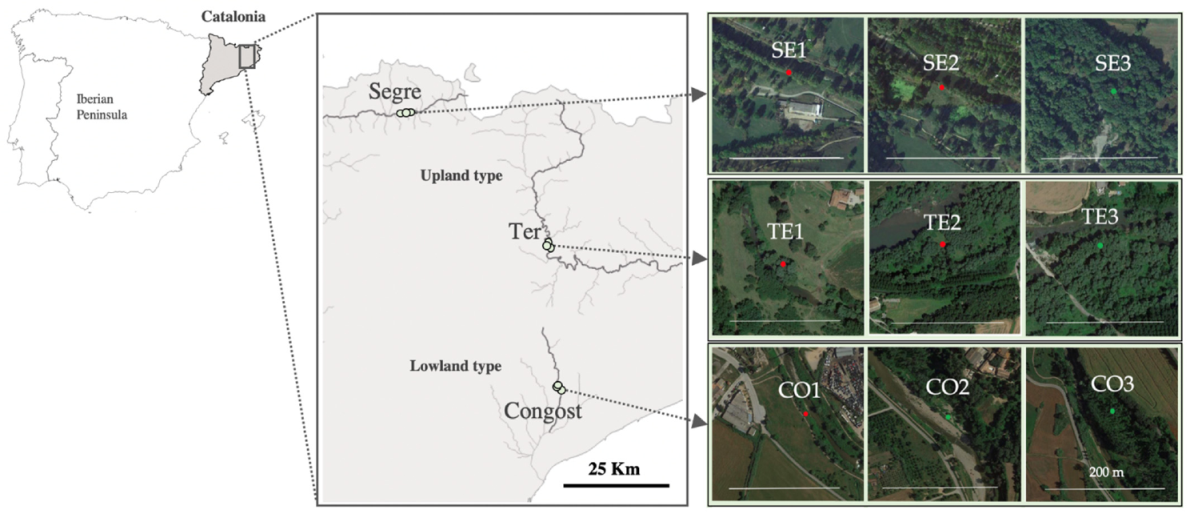



| Variables | Upland Type (N = 6) | Lowland Type (N = 3) |
|---|---|---|
| Average ± SD | Average ± SD | |
| Strahler number | 5 | 4 |
| Altitude (m) | 733 ± 284.10 | 206 ± 1.73 |
| Average annual air temperature (°C) | 10.35 ± 1.92 | 14.90 |
| Average annual rainfall (mm) | 579 ± 92.02 | 607 |
| Upstream drainage basin (km2) | 931.33 ± 142.62 | 166 |
| Valley confinement (mean 7 × 7) | −805.81 ± 313.46 | −727.12 ± 124.20 |
| Mann–Whitney U Test | |||
|---|---|---|---|
| Group | Significance p < 0.05 | Mean Rank | |
| Ant Metrics | Disturbed | Less Disturbed | |
| Upland River Type | |||
| Observed species richness | 0.028 | 24.42 | 33.67 |
| Closed-habitat species | <0.001 | 21.71 | 39.08 |
| Larger ants | 0.033 | 24.99 | 32.53 |
| Cryptics | 0.001 | 25.00 | 32.50 |
| Opportunists | 0.032 | 29.50 | 23.50 |
| Tetramorium caespitum foraging activity | 0.032 | 29.50 | 23.50 |
| Lowland River Type | |||
| Ant foraging activity | 0.011 | 19.39 | 11.31 |
| Seed harvesters | 0.023 | 18.89 | 11.56 |
| Aphaenogaster senilis foraging activity | <0.001 | 22.39 | 9.81 |
| Ant-Based MMI [Ant-Based MMI = n-Σ si; I = 1, ..., n] | Scores (si) | |||
|---|---|---|---|---|
| Core Metrics (n) | 1 (Poor) | 3 (Fair) | 5 (Good) | |
| [Boundaries Based on the Average of Disturbed and Less Disturbed] | ||||
| Upland Type | Observed species richness | ≤1.25 | 1.25–1.67 | ≥1.67 |
| Closed-habitat species | ≤1.89 | 1.89–5.56 | ≥5.56 | |
| Larger ants | ≤3.03 | 3.03–4.29 | ≥4.29 | |
| Cryptics | ≤0 | 0–1.26 | ≥1.26 | |
| Opportunists | ≥1.14 | 0–1.14 | ≤0 | |
| Tetramorium caespitum foraging activity | ≥0.04 | 0–0.04 | ≤0 | |
| Lowland Type | ||||
| Ant foraging activity | ≥9.67 | 6.06–9.67 | ≤6.06 | |
| Seed harvesters | ≥5.56 | 2.53–5.56 | ≤2.53 | |
| Aphaenogaster senilis foraging activity | ≥0.37 | 0.04–0.37 | ≤0.04 | |
| Upland Type | Lowland Type | |||
| Ant-Based MMI Range (absolute values) | 0–24 | 0–12 | ||
| Median of Reference (Less disturbed) Sites (md) | 18 | 12 | ||
| Thresholds of Ecological Classes (EQR) | ||||
| Excellent | EQR ≥ 0.67 | EQR ≥ 0.75 | ||
| Good | 0.48 ≥ EQR < 0.67 | 0.53 ≥ EQR < 0.75 | ||
| Moderate | 0.29 ≥ EQR < 0.48 | 0.31 ≥ EQR < 0.53 | ||
| Poor | 0.10 ≥ EQR < 0.29 | 0.09 ≥ EQR < 0.31 | ||
| Bad | EQR < 0.10 | EQR < 0.09 | ||
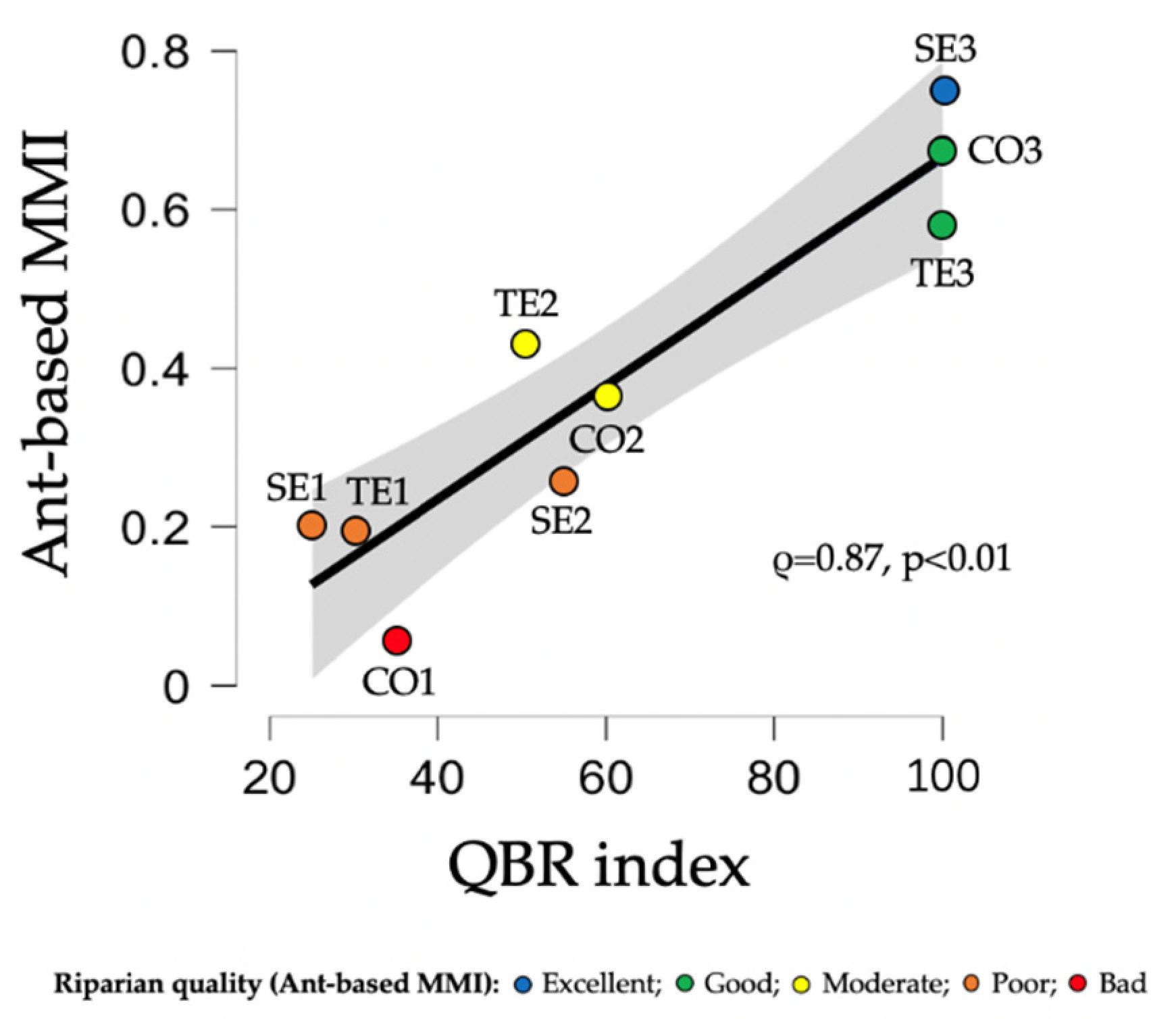
| River Type | Disturbance Group | River | Site | QBR | Ant-based MMI | ||
|---|---|---|---|---|---|---|---|
| Value | Quality Classes | Value | Quality Classes | ||||
| Upland | Disturbed | Ter | TE1 | 30 | Poor | 0.19 | Poor |
| TE2 | 50 | Poor | 0.43 | Moderate | |||
| Segre | SE1 | 25 | Bad | 0.20 | Poor | ||
| SE2 | 55 | Moderate | 0.26 | Poor | |||
| Less disturbed | Ter | TE3 | 100 | Excellent | 0.58 | Good | |
| Segre | SE3 | 100 | Excellent | 0.75 | Excellent | ||
| Lowland | Disturbed | Congost | CO1 | 35 | Poor | 0.06 | Bad |
| Less disturbed | CO2 | 60 | Moderate | 0.36 | Moderate | ||
| CO3 | 100 | Excellent | 0.68 | Good | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zina, V.; Ordeix, M.; Franco, J.C.; Ferreira, M.T.; Fernandes, M.R. Ants as Bioindicators of Riparian Ecological Health in Catalonian Rivers. Forests 2021, 12, 625. https://doi.org/10.3390/f12050625
Zina V, Ordeix M, Franco JC, Ferreira MT, Fernandes MR. Ants as Bioindicators of Riparian Ecological Health in Catalonian Rivers. Forests. 2021; 12(5):625. https://doi.org/10.3390/f12050625
Chicago/Turabian StyleZina, Vera, Marc Ordeix, José Carlos Franco, Maria Teresa Ferreira, and Maria Rosário Fernandes. 2021. "Ants as Bioindicators of Riparian Ecological Health in Catalonian Rivers" Forests 12, no. 5: 625. https://doi.org/10.3390/f12050625
APA StyleZina, V., Ordeix, M., Franco, J. C., Ferreira, M. T., & Fernandes, M. R. (2021). Ants as Bioindicators of Riparian Ecological Health in Catalonian Rivers. Forests, 12(5), 625. https://doi.org/10.3390/f12050625









