Mid-Term Effects of Forest Thinning on N Mineralization in a Semi-Arid Aleppo Pine Forest
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Silvicultural Treatments
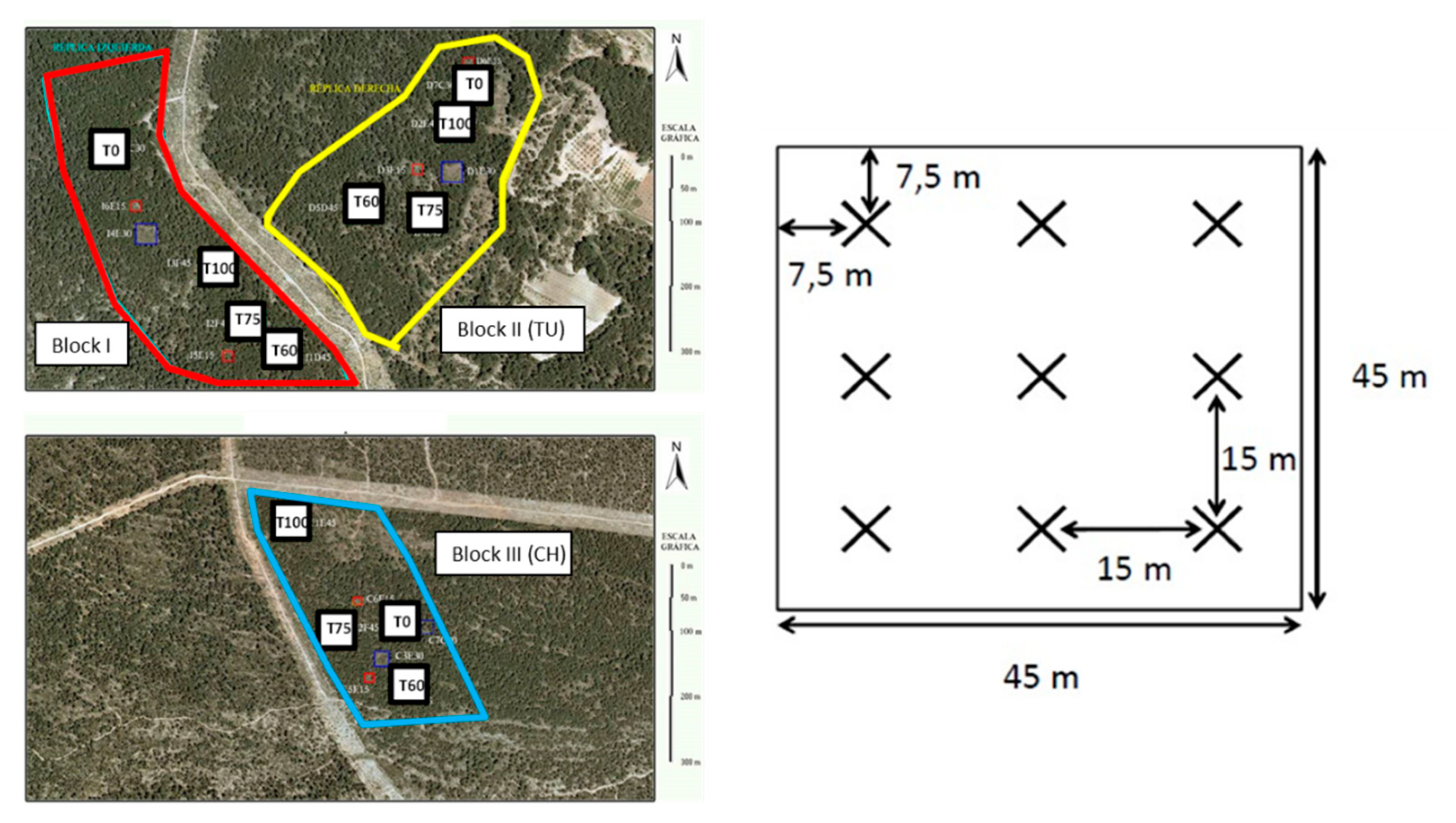
2.2. Experimental Design and Field Sampling
2.3. Chemical Analysis
2.4. Statistical Analysis
3. Results
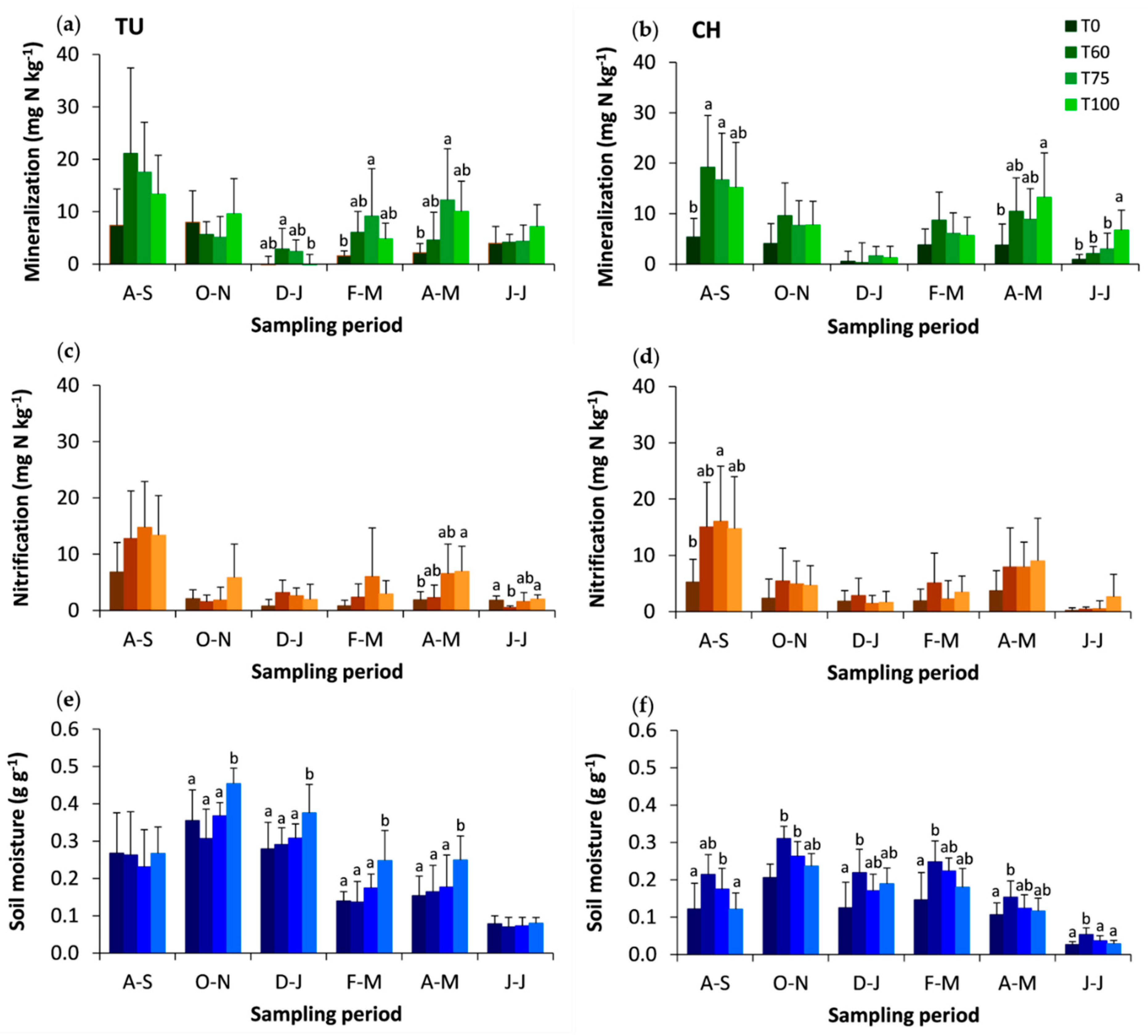
3.1. Effects of Experimental Thinning on N Dynamics, Net Mineralization, Nitrification and the Soil Total C and N
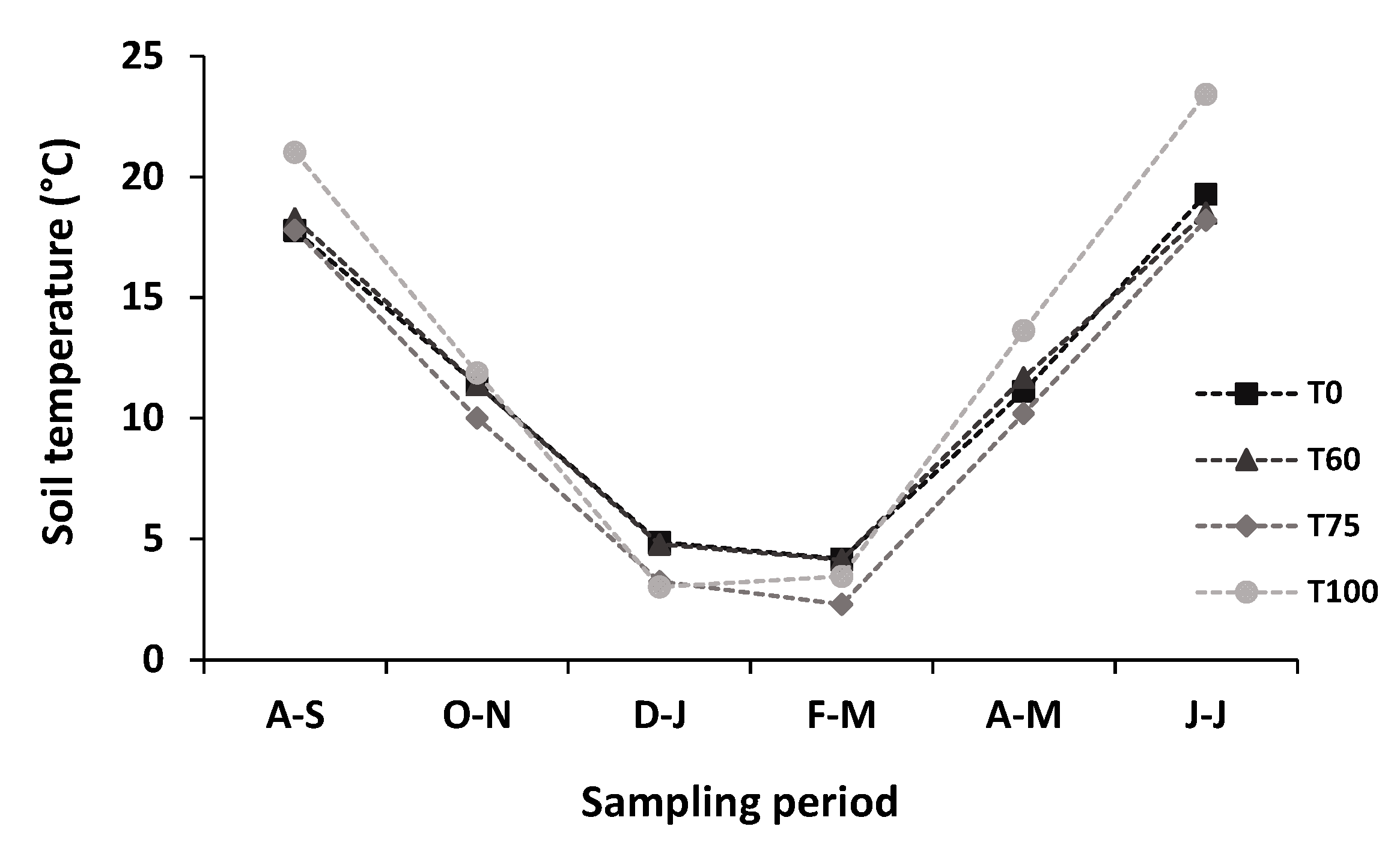
3.2. Effects of Experimental Thinning on Soil Temperature
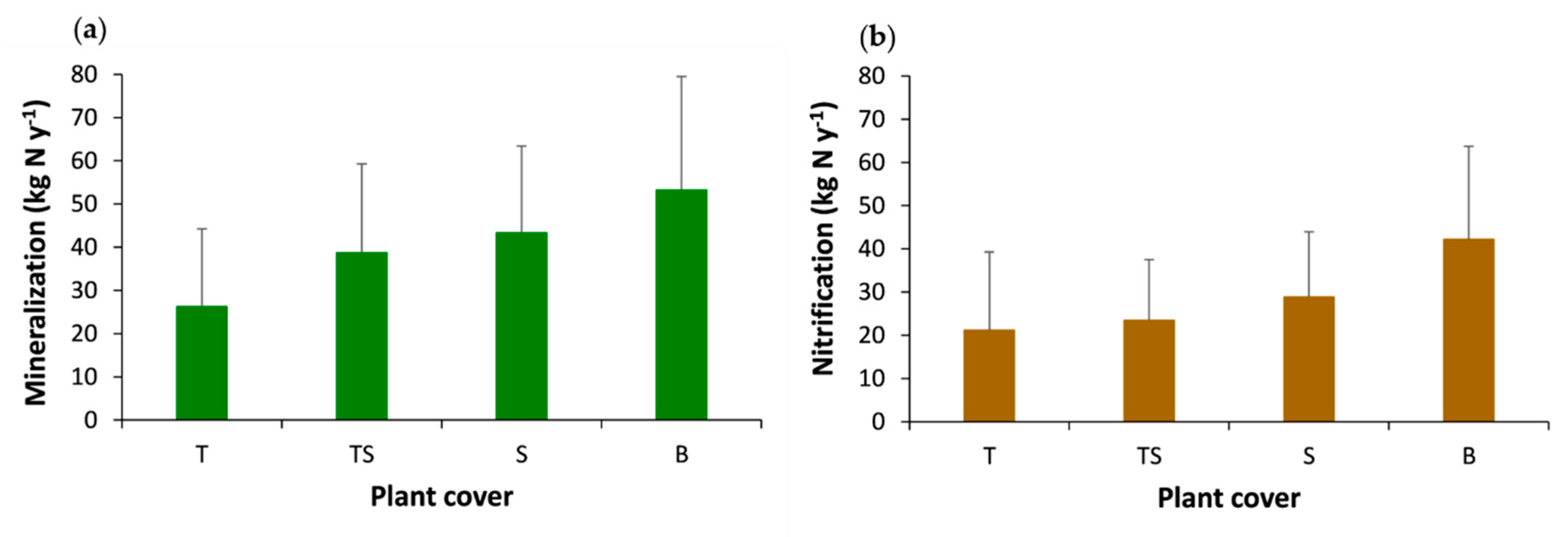
3.3. Effects of Tree Cover and Organic Horizon on the Net Mineralization and Nitrification Rates and on Mineral Nitrogen Inputs to Soil
3.4. Net Mineralization and Nitrification Rates in Disturbed and Undisturbed Samples
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Quézel, P. Taxonomy and biogeography of Mediterranean pines (Pinus halepensis and P. brutia). In Ecology, Biogeography and Management of Pinus halepensis and P. brutia Forest Ecosystems in the Mediterranean Basin; Backhuys Publishers: Leiden, The Nederlands, 2000; pp. 1–12. [Google Scholar]
- De Luis, M.; Čufar, K.; Di Filippo, A.; Novak, K.; Papadopoulos, A.; Piovesan, G.; Rathgeber, C.B.K.; Raventós, J.; Saz, M.A.; Smith, K.T. Plasticity in dendroclimatic response across the distribution range of Aleppo pine (Pinus halepensis). PLoS ONE 2013, 8, e83550. [Google Scholar] [CrossRef]
- Maestre, F.T.; Cortina, J. Are Pinus halepensis plantations useful as a restoration tool in semiarid Mediterranean areas? For. Ecol. Manag. 2004, 198, 303–317. [Google Scholar] [CrossRef]
- Chirino, E.; Bonet, A.; Bellot, J.; Sánchez, J.R. Effects of 30-year-old Aleppo pine plantations on runoff, soil erosion, and plant diversity in a semi-arid landscape in south eastern Spain. Catena 2006, 65, 19–29. [Google Scholar] [CrossRef]
- DECRETO 58/2013, de 3 de mayo, del Consell, por el que se Aprueba el Plan de Acción Territorial Forestal de la Comunitat Valenciana. [2013/4617]; (DOGV núm. 7019 de 08.05.2013); Generalitat Valenciana: Valencia, Spain, 2013.
- Tapias, R.; Climent, J.; Pardos, J.A.; Gil, L. Life histories of Mediterranean pines. Plant Ecol. 2004, 171, 53–68. [Google Scholar] [CrossRef]
- Scarascia-Mugnozza, G.; Oswald, H.; Piussi, P.; Radoglou, K. Forests of the Mediterranean region: Gaps in knowledge and research needs. For. Ecol. Manag. 2000, 132, 97–109. [Google Scholar] [CrossRef]
- Fabbio, G.; Merlo, M.; Tosi, V. Silvicultural management in maintaining biodiversity and resistance of forests in Europe—The Mediterranean region. J. Environ. Manag. 2003, 67, 67–76. [Google Scholar] [CrossRef]
- Morris, D.M.; Kimmins, J.P.; Duckert, D.R. The use of soil organic matter as a criterion of the relative sustainability of forest management alternatives: A modelling approach using FORECAST. For. Ecol. Manag. 1997, 94, 61–78. [Google Scholar] [CrossRef]
- Kimmins, J. Forest Ecology—A Foundation for Sustainable Forest Management and Environmental Ethics in Forestry, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2004. [Google Scholar]
- Blanco, J.A.; Zavala, M.A.; Imbert, J.B.; Castillo, F.J. Sustainability of forest management practices: Evaluation through a simulation model of nutrient cycling. For. Ecol. Manag. 2005, 213, 209–228. [Google Scholar] [CrossRef]
- Prescott, C.E. The influence of the forest canopy on nutrient cycling. Tree Physiol. 2002, 22, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Lado-Monserrat, L.; Lidón, A.; Bautista, I. Erratum to: Litterfall, litter decomposition and associated nutrient fluxes in Pinus halepensis: Influence of tree removal intensity in a Mediterranean forest. Eur. J. For. Res. 2016, 135, 203–214. [Google Scholar] [CrossRef]
- Bravo-Oviedo, A.; Ruiz-Peinado, R.; Onrubia, R.; del Río, M. Thinning alters the early-decomposition rate and nutrient immobilization-release pattern of foliar litter in Mediterranean oak-pine mixed stands. For. Ecol. Manag. 2017, 391, 309–320. [Google Scholar] [CrossRef]
- Bueis, T.; Bravo, F.; Pando, V.; Turrión, M.B. Local basal area affects needle litterfall, nutrient concentration, and nutrient release during decomposition in Pinus halepensis Mill. plantations in Spain. Ann. For. Sci. 2018, 75, 21. [Google Scholar] [CrossRef]
- Zhang, X.; Guan, D.; Li, W.; Sun, D.; Jin, C.; Yuan, F.; Wang, A.; Wu, J. The effects of forest thinning on soil carbon stocks and dynamics: A meta-analysis. For. Ecol. Manag. 2018, 429, 36–43. [Google Scholar] [CrossRef]
- Cortina, J.; Vallejo, V.R. Effects of clearfelling on forest floor accumulation and litter decomposition in a radiata pine plantation. For. Ecol. Manag. 1994, 70, 299–310. [Google Scholar] [CrossRef]
- Frazer, D.W.; McColl, J.G.; Powers, R.F. Soil Nitrogen Mineralization in a Clearcutting Chronosequence in a Northern California Conifer Forest. Soil Sci. Soc. Am. J. 1990, 54, 1145–1152. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Gosz, J.R.; Grier, C.C.; Melillo, J.M.; Reiners, W.A.; Todd, R.L. Nitrate losses from disturbed ecosystems. Science 1979, 204, 469–474. [Google Scholar] [CrossRef]
- Valdecantos, A.; Cortina, J.; Vallejo, V.R. Nutrient status and field performance of tree seedlings planted in Mediterranean degraded areas. Ann. For. Sci. 2006, 63, 249–256. [Google Scholar] [CrossRef]
- Yoon, T.K.; Noh, N.J.; Chung, H.; Yang, A.R.; Son, Y. Soil nitrogen transformations and availability in upland pine and bottomland alder forests. Forests 2015, 6, 2941–2958. [Google Scholar] [CrossRef]
- Fisher, R.F.; Binkley, D. Ecology and Management of Forest Soils; Wiley: New York, NY, USA, 2000; ISBN 0-471-19426-3. [Google Scholar]
- Prescott, C.E. Effects of clearcutting and alternative silvicultural systems on rates of decomposition and nitrogen mineralization in a coastal montane coniferous forest. For. Ecol. Manag. 1997, 95, 253–260. [Google Scholar] [CrossRef]
- Kim, C. Soil Nitrogen Mineralization at Various Levels of Canopy Cover in Red Pine (Pinus resinosa) Plantations. J. For. Res. 1998, 3, 85–89. [Google Scholar] [CrossRef]
- Paavolainen, L.; Smolander, A. Nitrification and denitrification in soil from a clear-cut Norway spruce (Picea abies) stand. Soil Biol. Biochem. 1998, 30, 775–781. [Google Scholar] [CrossRef]
- Burns, D.A.; Murdoch, P.S. Effects of a clearcut on the net rates of nitrification and N mineralization in a northern hardwood forest, Catskill Mountains, New York, USA. Biogeochemistry 2005, 72, 123–146. [Google Scholar] [CrossRef]
- Jerabkova, L.; Prescott, C.E.; Titus, B.D.; Hope, G.D.; Walters, M.B. A meta-analysis of the effects of Clearcut and variable-retention harvesting on soil nitrogen fluxes in boreal and temperate forests. Can. J. For. Res. 2011, 41, 1852–1870. [Google Scholar] [CrossRef]
- Rapp, M. Nitrogen status and mineralization in natural and disturbed mediterranean forests and coppices. Plant Soil 1990, 128, 21–30. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Melillo, J.M. Nitrate losses from disturbed forests: Patterns and mechanisms. For. Sci. 1979, 25, 605–619. [Google Scholar] [CrossRef]
- Ritter, E.; Starr, M.; Vesterdal, L. Losses of nitrate from gaps of different sizes in a managed beech (Fagus sylvatica) forest. Can. J. For. Res. 2005, 35, 308–319. [Google Scholar] [CrossRef]
- Futter, M.N.; Ring, E.; Högbom, L.; Entenmann, S.; Bishop, K.H. Consequences of nitrate leaching following stem-only harvesting of Swedish forests are dependent on spatial scale. Environ. Pollut. 2010, 158, 3552–3559. [Google Scholar] [CrossRef] [PubMed]
- Matson, P.A.; Vitousek, P.M. Nitrogen mineralization and nitrification potentials following clearcutting in the Hoosier National Forest, Indiana. For. Sci. 1981, 27, 781–971. [Google Scholar] [CrossRef]
- Edmonds, R.L.; McColl, J.G. Effects of forest management on soil nitrogen in Pinus radiata stands in the Australian Capital Territory. For. Ecol. Manag. 1989, 29, 199–212. [Google Scholar] [CrossRef]
- Smethurst, P.J.; Nambiar, E.K.S. Distribution of carbon and nutrients and fluxes of mineral nitrogen after clear-felling a Pinus radiata plantation. Can. J. For. Res. 1990, 20, 1490–1497. [Google Scholar] [CrossRef]
- Aber, J.D.; Melillo, J.M.; Nadelhoffer, K.J.; Pastor, J.; Boone, R.D. Factors controlling nitrogen cycling and nitrogen saturation in northern temperate forest ecosystems. Ecol. Appl. 1991, 1, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Idol, T.W.; Pope, P.E.; Ponder, F. N mineralization, nitrification, and N uptake across a 100-year chronosequence of upland hardwood forests. For. Ecol. Manag. 2003, 176, 509–518. [Google Scholar] [CrossRef]
- Kranabetter, J.M.; Coates, K.D. Ten-year postharvest effects of silviculture systems on soil-resource availability and conifer nutrition in a northern temperate forest. Can. J. For. Res. 2004, 34, 800–809. [Google Scholar] [CrossRef]
- Barg, A.K.; Edmonds, R.L. Influence of partial cutting on site microclimate, soil nitrogen dynamics, and microbial biomass in Douglas-fir stands in western Washington. Can. J. For. Res. 1999, 29, 705–713. [Google Scholar] [CrossRef]
- Grenon, F.; Bradley, R.L.; Joanisse, G.; Titus, B.D.; Prescott, C.E. Mineral N availability for conifer growth following clearcutting: Responsive versus non-responsive ecosystems. For. Ecol. Manag. 2004, 188, 305–316. [Google Scholar] [CrossRef]
- Kim, C.; Sharik, T.L.; Jurgensen, M.F. Canopy cover effects on soil nitrogen mineralization in northern red oak (Quercus rubra) stands in northern Lower Michigan. For. Ecol. Manag. 1995, 76, 21–28. [Google Scholar] [CrossRef]
- Carlyle, J.C. Nutrient management in a Pinus radiata plantation after thinning: The effect of thinning and residues on nutrient distribution, mineral nitrogen fluxes, and extractable phosphorus. Can. J. For. Res. 1995, 25, 1278–1291. [Google Scholar] [CrossRef]
- Thibodeau, L.; Raymond, P.; Camiré, C.; Munson, A.D. Impact of precommercial thinning in balsam fir stands on soil nitrogen dynamics, microbial biomass, decomposition, and foliar nutrition. Can. J. For. Res. 2000, 30, 229–238. [Google Scholar] [CrossRef]
- Grady, K.C.; Hart, S.C. Influences of thinning, prescribed burning, and wildfire on soil processes and properties in southwestern ponderosa pine forests: A retrospective study. For. Ecol. Manag. 2006, 234, 123–135. [Google Scholar] [CrossRef]
- Escrig, A.; Pérez, R.; Estruch, V.; Galiana, F. Evolución de la estructura y diversidad de la vegetación en un tratamiento selvícola experimental sobre pinar de carrasco en Tuéjar y Chelva (Valencia). In Proceedings of the IV Congreso Forestal Español, Zaragoza, Spain, 26–30 September 2005. [Google Scholar]
- Redding, T.E.; Hope, G.D.; Schmidt, M.G.; Fortin, M.J. Analytical methods for defining stand-clearcut edge effects demonstrated for N mineralization. Can. J. For. Res. 2004, 34, 1018–1024. [Google Scholar] [CrossRef]
- IGME. Mapa Geológico de España. E. 1:50.000 Chelva (666); Instituto Geológico y Minero de España: Madrid, Spain, 1973. [Google Scholar]
- GVA. Mapa de Suelos de la Comunidad Valenciana. Chelva (666). Proyecto LUCDEME (Icona); Centro de Investigaciones sobre Desertificación y Conselleria d’Agricultura i Mig Ambient; Generalitat Valenciana: Valencia, Spain, 1995. [Google Scholar]
- Rivas-Martínez, S. Memoria del Mapa de Series de Vegetación de España 1:400.000; Ministerio de Agricultura, Pesca y Alimentación, ICONA: Madrid, Spain, 1987. [Google Scholar]
- DiStefano, J.F.; Gholz, H.L. A proposed use of ion exchange resins to measure nitrogen mineralization and nitrification in intact soil cores. Commun. Soil Sci. Plant Anal. 1986, 17, 989–998. [Google Scholar] [CrossRef]
- Köhler, S.; Jungkunst, H.F.; Gutzler, C.; Herrera, R.; Gerold, G. Atmospheric ionic deposition in tropical sites of central sulawesi determined by ion exchange resin collectors and bulk water collector. Water Air. Soil Pollut. 2012, 223, 4485–4494. [Google Scholar] [CrossRef] [PubMed]
- Rutigliano, F.A.; D’Ascoli, R.; Virzo De Santo, A. Soil microbial metabolism and nutrient status in a Mediterranean area as affected by plant cover. Soil Biol. Biochem. 2004, 36, 1719–1729. [Google Scholar] [CrossRef]
- Raz-Yaseef, N.; Rotenberg, E.; Yakir, D. Effects of spatial variations in soil evaporation caused by tree shading on water flux partitioning in a semi-arid pine forest. Agric. For. Meteorol. 2010, 150, 454–462. [Google Scholar] [CrossRef]
- Prévosto, B.; Monnier, Y.; Ripert, C.; Fernandez, C. Can we use shelterwoods in Mediterranean pine forests to promote oak seedling development? For. Ecol. Manag. 2011, 262, 1426–1433. [Google Scholar] [CrossRef]
- Balisky, A.C.; Burton, P.J. Root-zone soil temperature variation associated with microsite characteristics in high-elevation forest openings in the interior of British Columbia. Agric. For. Meteorol. 1995, 77, 31–54. [Google Scholar] [CrossRef]
- Devine, W.D.; Harrington, C.A. Influence of harvest residues and vegetation on microsite soil and air temperatures in a young conifer plantation. Agric. For. Meteorol. 2007, 145, 125–138. [Google Scholar] [CrossRef]
- Varanda Andrade, J.A.; Gonçalves de Abreu, F.M.; Valeriano Madeira, M.A. Influence of litter layer removal on the soil thermal regime of a pine forest in a Mediterranean climate. Rev. Bras. Ciência Solo 2010, 34, 1481–1490. [Google Scholar] [CrossRef]
- Godefroid, S.; Rucquoij, S.; Koedam, N. Spatial variability of summer microclimates and plant species response along transects within clearcuts in a beech forest. Plant Ecol. 2006, 185, 107–121. [Google Scholar] [CrossRef]
- Maestre, F.T.; Cortina, J.; Bautista, S.; Bellot, J. Does Pinus halepensis facilitate the establishment of shrubs in Mediterranean semi-arid afforestations? For. Ecol. Manag. 2003, 176, 147–160. [Google Scholar] [CrossRef]
- Galiana, F.; Palacio, A.; Lidón, A.; Martínez-Campos, F.M.; Tortajada, L.; Bautista, I.; Lull, C. Evolución de la estructura de la vegetación y la regeneración del pinar en un tratamiento selvícola experimental en Tuéjar (Valencia). In Proceedings of the 7° Congreso Forestal Español, Plasencia, Spain, 26–30 June 2017; pp. 1–5. [Google Scholar]
- Moberg, D.P.; Johnson, R.L.; Sullivan, D.M. Comparison of Disturbed and Undisturbed Soil Core Methods to Estimate Nitrogen-Mineralization Rates in Manured Agricultural Soils. Commun. Soil Sci. Plant Anal. 2013, 44, 1722–1732. [Google Scholar] [CrossRef]
- Parker, S.S.; Schimel, J.P. Soil nitrogen availability and transformations differ between the summer and the growing season in a California grassland. Appl. Soil Ecol. 2011, 48, 185–192. [Google Scholar] [CrossRef]
- Lado-Monserrat, L.; Lull, C.; Bautista, I.; Lidón, A.; Herrera, R. Soil moisture increment as a controlling variable of the “Birch effect”. Interactions with the pre-wetting soil moisture and litter addition. Plant Soil 2014, 379, 21–34. [Google Scholar] [CrossRef]
- Almagro, M.; Martínez-Mena, M. Exploring short-term leaf-litter decomposition dynamics in a Mediterranean ecosystem: Dependence on litter type and site conditions. Plant Soil 2012, 358, 323–335. [Google Scholar] [CrossRef]
- Ludovici, K.H.; Zarnoch, S.J.; Richter, D.D. Modeling in-situ pine root decomposition using data from a 60-year chronosequence. Can. J. For. Res. 2002, 32, 1675–1684. [Google Scholar] [CrossRef][Green Version]
- Scott, N.A.; Binkley, D. Foliage litter quality and annual net N mineralization: Comparison across North American forest sites. Oecologia 1997, 111, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, J.B. Fine-scale patterns of leaf litterfall and nitrogen cycling in an old-growth forest. Can. J. For. Res. 1999, 29, 291–302. [Google Scholar] [CrossRef]
- Killingbeck, K.T.; Whitford, W.G. High foliar nitrogen in desert shrubs: An important ecosystem trait or defective desert doctrine? Ecology 1996, 77, 1728–1737. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J.; Rodà, F. Changes in nutrient use efficiency, status and retranslocation in young post-fire regeneration Pinus halepensis in response to sudden N and P input, irrigation and removal of competing vegetation. Trees 2005, 19, 233–250. [Google Scholar] [CrossRef]



| Block | Treatment | Corg (%) | N (%) | C/N (g g−1) | Average Content N-NH4+ (mg kg−1) | Average Content N-NO3- (mg kg−1) | Annual Mineralization (mg N kg−1) | Annual Nitrification (mg N kg−1) |
|---|---|---|---|---|---|---|---|---|
| TU | T0 | 5.4 ± 0.5 ab | 0.26 ± 0.05 b | 20.8 ± 2.2 | 1.48 ± 1.06 | 0.43 ± 0.47 | 22.8 ± 12.2 b | 14.2 ± 6.5 b |
| T60 | 5.5 ± 1.4 ab | 0.30 ± 0.07 b | 18.4 ± 3.4 | 1.60 ± 1.70 | 0.32 ± 1.00 | 44.4 ± 21.8 ab | 22.7 ± 11.8 ab | |
| T75 | 3.8 ± 0.7 b | 0.22 ± 0.03 b | 17.2 ± 3.7 | 1.38 ± 1.38 | 0.32 ± 0.51 | 50.8 ± 23.9 a | 33.3 ± 18.2 a | |
| T100 | 7.2 ± 1.4 a | 0.43 ± 0.11 a | 17.2 ± 3.3 | 1.90 ± 2.05 | 0.43 ± 0.66 | 44.1 ± 15.8 ab | 33.1 ± 17.0 a | |
| p-value | 0.001 | 0.002 | 0.268 | 0.432 | 0.840 | 0.021 | 0.000 | |
| CH | T0 | 1.1 ± 0.7 b | 0.03 ± 0.02 b | 69.3 ± 62.3 | 1.45 ± 0.91 b | 0.38 ± 0.42 | 17.9 ± 12.4 b | 15.0 ± 11.6 b |
| T60 | 3.3 ± 1.0 a | 0.15 ± 0.03 a | 20.4 ± 3.2 | 2.46 ± 2.08 a | 0.67 ± 1.06 | 50.3 ± 15.7 a | 36.8 ± 15.5 a | |
| T75 | 1.7 ± 0.8 b | 0.07 ± 0.05 b | 28.9 ± 9.9 | 2.43 ± 1.29 a | 0.33 ± 0.37 | 44.0 ± 21.9 a | 33.2 ± 18.4 a | |
| T100 | 1.5 ± 0.3 b | 0.07 ± 0.01 b | 20.8 ± 2.1 | 1.91 ± 1.44 ab | 0.62 ± 0.70 | 48.7 ± 26.0 a | 35.5 ± 22.4 a | |
| p-value | 0.002 | 0.000 | 0.079 | 0.014 | 0.313 | 0.000 | 0.000 |
| Block | Treatment | Organic Layer Depth (mm) |
|---|---|---|
| TU | T0 | 6.9 ± 3.5 b |
| T60 | 4.0 ± 2.9 ab | |
| T75 | 2.5 ± 3.2 a | |
| T100 | 1.8 ± 2.7 a | |
| p-value | 0.007 | |
| CH | T0 | 3.1 ± 1.9 |
| T60 | 2.9 ± 2.7 | |
| T75 | 1.8 ± 1.5 | |
| T100 | 1.4 ± 1.2 | |
| p-value | NS |
| Plant Cover | NH4+ (kg N ha−1 y−1) | NO3− (kg N ha−1 y−1) | Mineral N (kg N ha−1 y−1) |
|---|---|---|---|
| B | 1.89 ± 1.37 | 0.82 ± 0.19 | 2.71 ± 1.53 |
| S | 1.12 ± 0.12 | 0.70 ± 0.24 | 1.82 ± 0.84 |
| T | 3.98 ± 3.32 | 3.20 ± 2.07 | 7.18 ± 5.39 |
| TS | 3.48 ± 1.33 | 3.82 ± 1.86 | 7.30 ± 2.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bautista, I.; Lado-Monserrat, L.; Lull, C.; Lidón, A. Mid-Term Effects of Forest Thinning on N Mineralization in a Semi-Arid Aleppo Pine Forest. Forests 2021, 12, 1470. https://doi.org/10.3390/f12111470
Bautista I, Lado-Monserrat L, Lull C, Lidón A. Mid-Term Effects of Forest Thinning on N Mineralization in a Semi-Arid Aleppo Pine Forest. Forests. 2021; 12(11):1470. https://doi.org/10.3390/f12111470
Chicago/Turabian StyleBautista, Inmaculada, Luis Lado-Monserrat, Cristina Lull, and Antonio Lidón. 2021. "Mid-Term Effects of Forest Thinning on N Mineralization in a Semi-Arid Aleppo Pine Forest" Forests 12, no. 11: 1470. https://doi.org/10.3390/f12111470
APA StyleBautista, I., Lado-Monserrat, L., Lull, C., & Lidón, A. (2021). Mid-Term Effects of Forest Thinning on N Mineralization in a Semi-Arid Aleppo Pine Forest. Forests, 12(11), 1470. https://doi.org/10.3390/f12111470






