Root-Plate Characteristics of Common Aspen in Hemiboreal Forests of Latvia: A Case Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
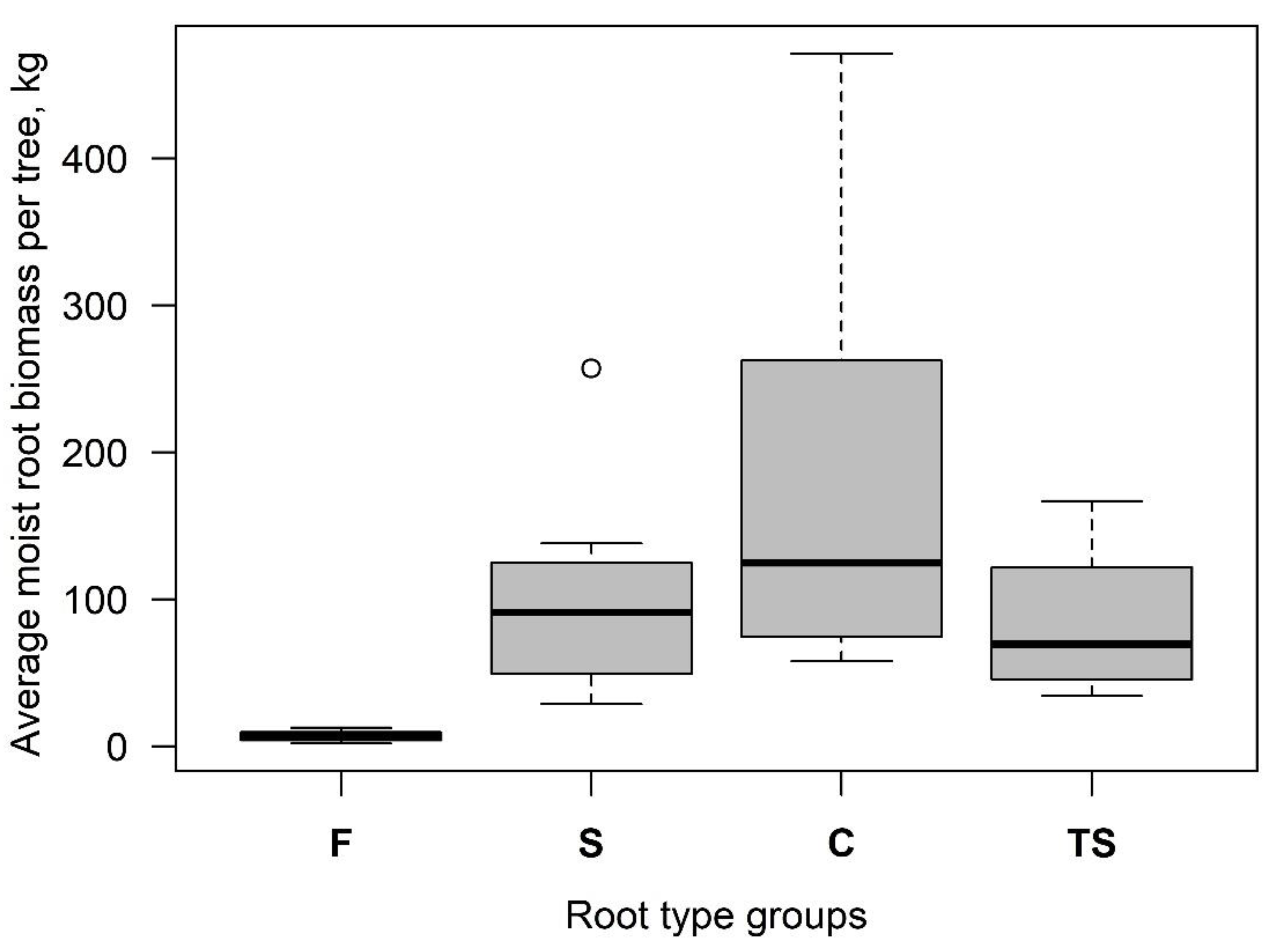
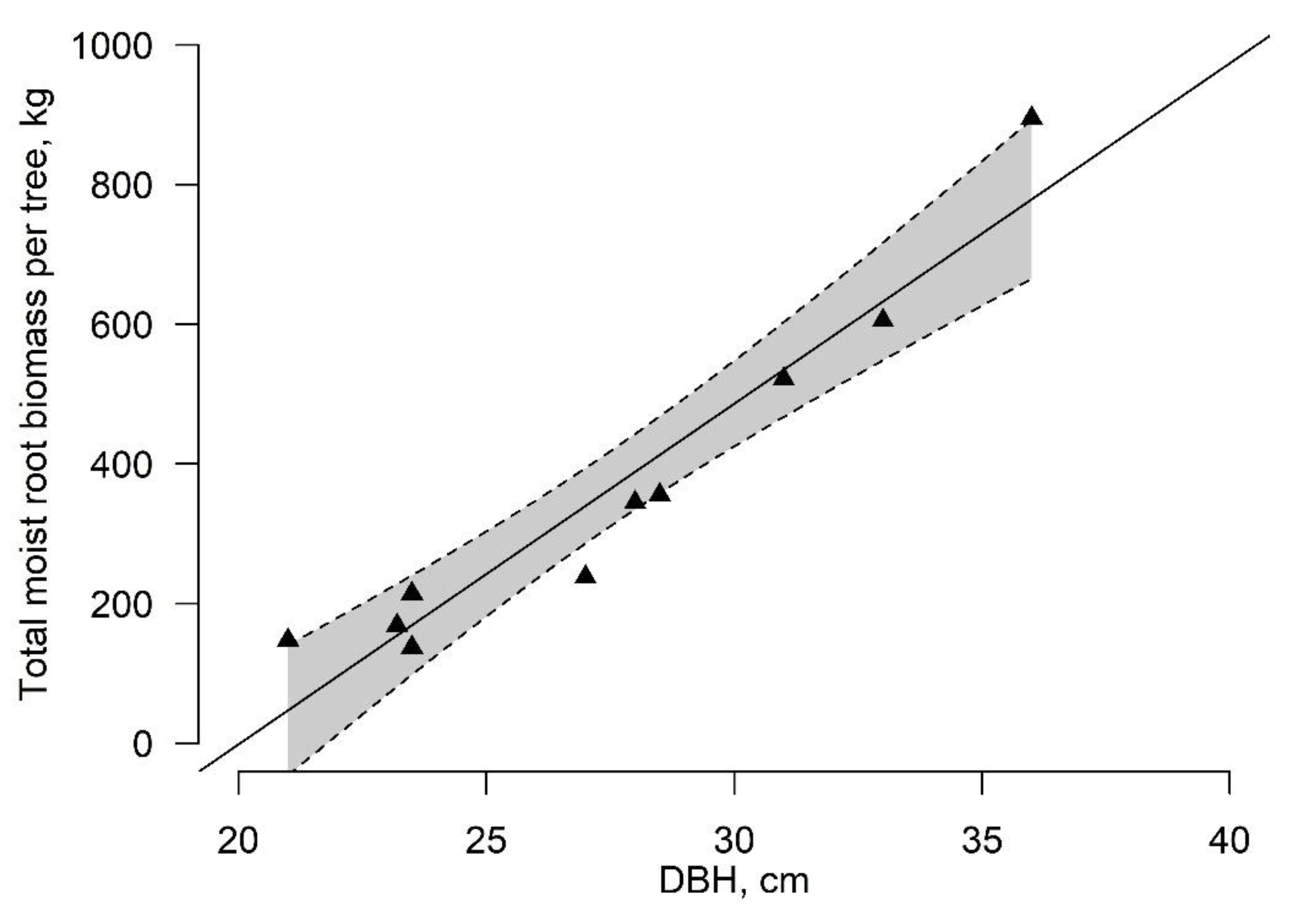
3.1. Root-Plate Biomass
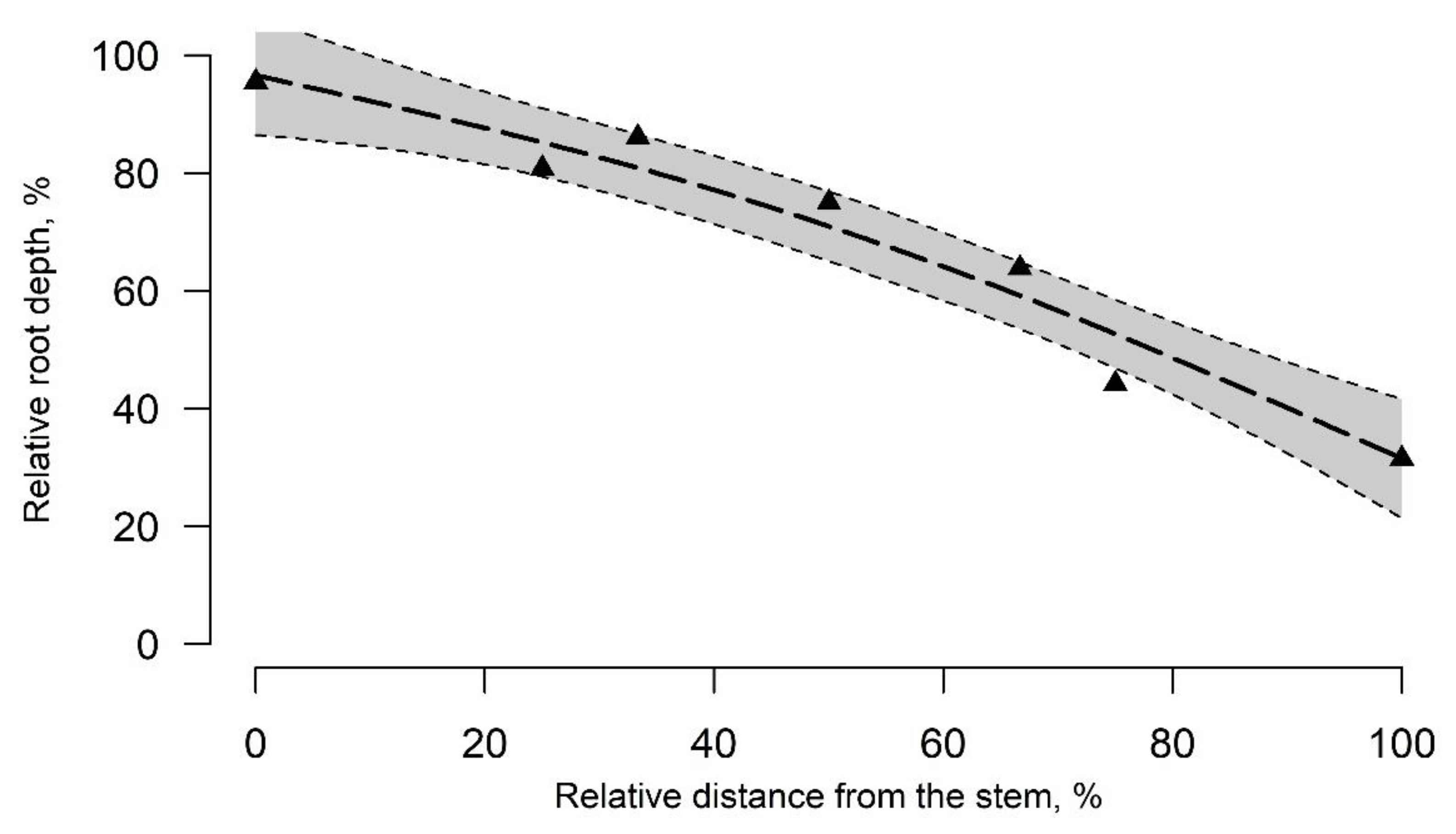
3.2. Structural Root-Plate Depth Distribution
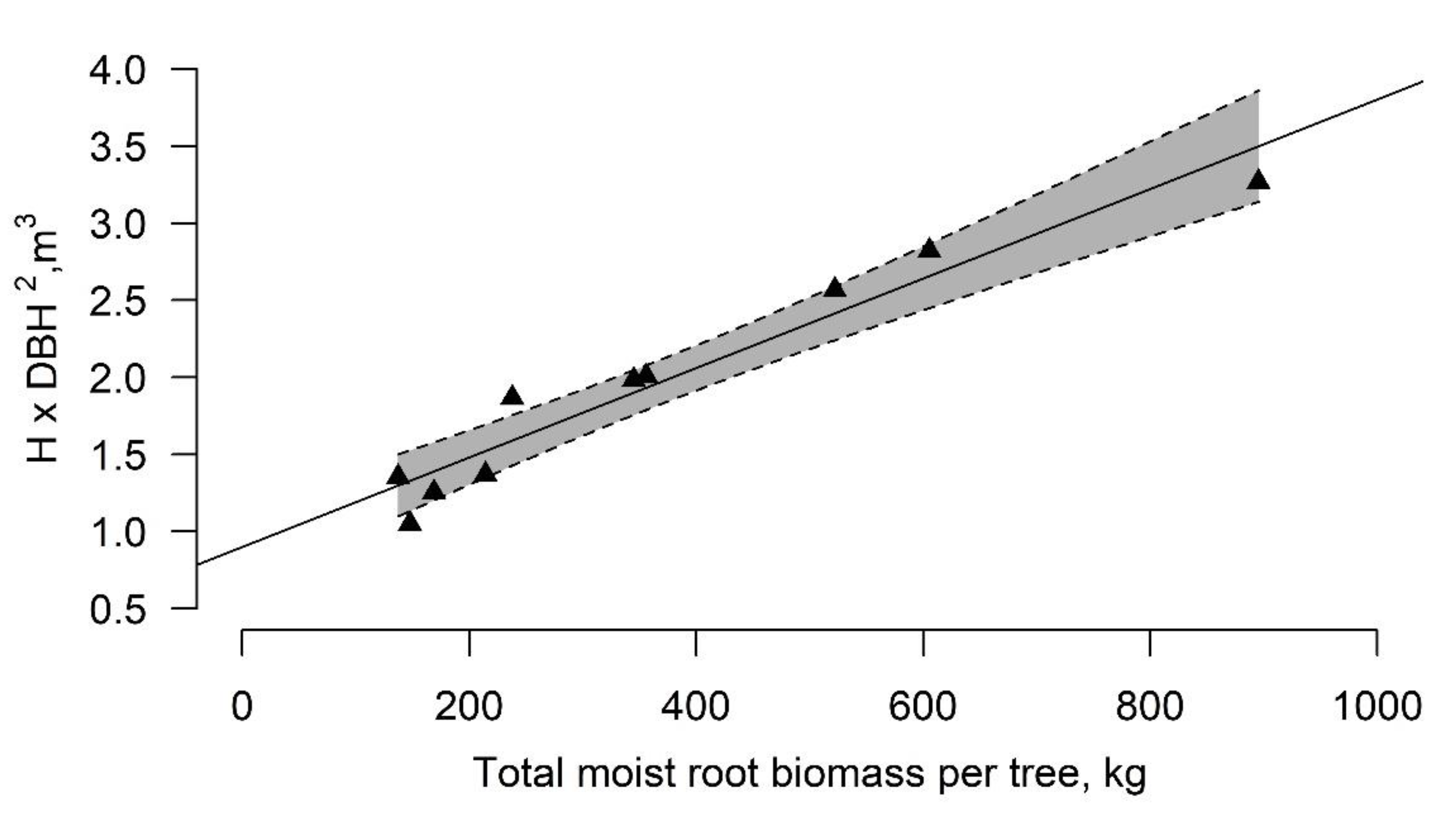
3.3. Tree Wind Resistance Indicator
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. 2019: Summary for Policymakers. In Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; Shukla, P.R., Skea, J., Buendia, E.C., Masson-Delmotte, V., Pörtner, H.-O., Roberts, D.C., Zhai, P., Slade, R., Connors, S., van Diemen, R., et al., Eds.; IPCC: Geneva, Switzerland, 2019; in press. [Google Scholar]
- Jansons, Ā. Forest management in the context of climate change. In Meža Apsaimniekošana Klimata Izmaiņu Kontekstā; LVMI Silava: Salaspils, Latvia, 2010; p. 31. (In Latvian) [Google Scholar]
- Donis, J. Evaluation of extreme wind speed impact on steadiness of forest stand, elaboration of the support system of decision making. In Ekstrēmu Vēju Ātrumu Ietekmes uz Kokaudzes Noturību Novērtējums, Lēmuma Pieņemšanas Atbalsta Sistēmas Izstrāde; LVMI Silava: Salaspils, Latvia, 2006; p. 64. (In Latvian) [Google Scholar]
- Yang, M.; Défossez, P.; Danjon, F.; Fourcaud, T. Tree stability under wind: Simulating uprooting with root breakage using a finite element method. Ann. Bot. 2014, 114, 695–709. [Google Scholar] [CrossRef] [PubMed]
- Šņepsts, G.; Donis, J.; Strēlnieks, K.; Krišāns, O.; Desaine, I.; Adamovičs, A. Post-storm regeneration of Norway spruce. In Proceedings of the 25th Annual International Scientific Conference “Research for Rural Development 2019”, Jelgava, Latvia, 17–19 May 2019; Volume 1, pp. 71–75. [Google Scholar] [CrossRef]
- Zielonka, T. When does dead wood turn into a substrate for spruce replacement. J. Veg. Sci. 2006, 17, 739–746. [Google Scholar] [CrossRef]
- Gardiner, B.; Schuck, A.R.T.; Schelhaas, M.J.; Orazio, C.; Blennow, K.; Nicoll, B. Living with Storm Damage to Forests: What Science Can Tell Us, 3rd ed.; European Forest Institute: Joensuu, Finland, 2013; p. 133. [Google Scholar]
- Peltola, H.; Ikonen, V.P.; Gregow, H.; Strandman, H.; Kilpeläinen, A.; Venäläinen, A.; Kellomäki, S. Impacts of climate change on timber production and regional risks of wind-induced damage to forests in Finland. For. Ecol. Manag. 2020, 260, 833–845. [Google Scholar] [CrossRef]
- Bussotti, F.; Pollastrini, M. Traditional and novel indicators of climate change impacts on European forest trees. Forests 2017, 8, 137. [Google Scholar] [CrossRef]
- Grime, J.P. Plant Strategies, Vegetation Processes and Ecosystem Properties; Wiley: Chichester, UK, 2001; p. 456. [Google Scholar]
- Stokes, A.; Nicoll, B.C.; Coutts, M.P.; Fitter, A.H. Responses of young Sitka spruce clones to mechanical perturbation and nutrition: Effects on biomass allocation, root development, and resistance to bending. Can. J. For. Res. 1997, 27, 1049–1057. [Google Scholar] [CrossRef]
- Nicoll, B.C.; Ray, D. Adaptive growth of tree root systems in response to wind action and site conditions. Tree Physiol. 1996, 16, 891–898. [Google Scholar] [CrossRef]
- Liepiņš, J. Methodology Development for Forest Stand Biomass and Carbon Stock Estimates in Latvia. Ph.D. Thesis, Latvia University of Life Sciences and Technologies, Jelgava, Latvia, 2019. [Google Scholar]
- Caudullo, G.; de Rigo, D. Populus tremula in Europe: Distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; San-Miguel-Ayanz, J., de Rigo, D., Caudullo, G., Houston Durrant, T., Mauri, A., Eds.; Publication Office of the European Union Luxembourg: Luxembourg, 2016; pp. 138–139. [Google Scholar]
- MacKenzie, N.A. Ecology, Conservation and Management of Aspen. A Literature Review; Scottish Native Woods: Aberfeldy, UK, 2010; p. 40. [Google Scholar]
- Worrell, R. European aspen (Populus tremula L.)—A review with particular reference to Scotland. Distribution, ecology and genetic variation. Forestry 1995, 68, 93–105. [Google Scholar] [CrossRef]
- State Forest Service. Annual Report 2019. Available online: https://www.zm.gov.lv/public/files/CMS_Static_Page_Doc/00/00/01/80/15/Publiskais_parskats_2019.pdf (accessed on 4 November 2020).
- Ennos, A. The mechanics of root anchorage. Adv. Bot. Res. 2000, 33, 133–157. [Google Scholar] [CrossRef]
- Stubbs, C.J.; Cook, D.D.; Niklas, K.J. A general review of the biomechanics of root anchorage. J. Exp. Bot. 2019, 70, 3439–3451. [Google Scholar] [CrossRef]
- Coutts, M.P.; Nielsen, C.C.N.; Nicoll, B.C. The development of symmetry, rigidity and anchorage in the structural root system of conifers. Plant Soil. 1999, 217, 1–15. [Google Scholar] [CrossRef]
- Nadelhoffer, K.J.; Raich, J.W. Fine root production estimates and belowground carbon allocation in forest ecosystems. Ecology 1992, 73, 1139–1147. [Google Scholar] [CrossRef]
- Coutts, M.P. Developmental process in tree root systems. Can. J. For. Res. 1987, 17, 761–767. [Google Scholar] [CrossRef]
- Coutts, M.P. Root architecture and tree stability. In Tree Root Systems and Their Mycorrhizas; Atkinson, D., Bhat, K.K.S., Coutts, M.P., Mason, P.A., Read, D.J., Eds.; Developments in Plant and Soil Sciences; Springer: Dordrecht, The Netherlands, 1983; Volume 71, pp. 171–188. [Google Scholar] [CrossRef]
- Coutts, M.P. Components of tree stability in Sitka spruce on peaty gley soils. Forestry 1986, 59, 173–197. [Google Scholar] [CrossRef]
- James, K.R.; Haritos, N.; Ades, P.K. Mechanical stability of trees under dynamic loads. Am. J. Bot. 2006, 93, 1522–1530. [Google Scholar] [CrossRef]
- Miezite, O.; Ruba, J. Sanitary state of naturally and artificially regenerated Picea abies (L.) H. Karst young stands in Oxalidosa. In Proceedings of the 24th Annual International Scientific Conference “Research for Rural Development 2018”, Jelgava, Latvia, 16–18 May 2018; Volume 1, pp. 72–77. [Google Scholar] [CrossRef]
- Ruba, J.; Miezite, O.; Baltmanis, R.; Luguza, S. Abiotic risks of managing young forest stands of Norway spruce (Picea abies (L.) karst.). In Proceedings of the 19th Annual International Scientific Conference “Research for Rural Development 2013”, Jelgava, Latvia, 15–17 May 2013; Volume 2, pp. 27–33. [Google Scholar]
- Gardiner, B.; Berry, P.; Moulia, B. Review: Wind impacts on plant growth, mechanics and damage. Plant Sci. 2016, 245, 94–118. [Google Scholar] [CrossRef]
- Rahardjo, H.; Harnas, F.R.; Leong, E.C.; Tan, P.Y.; Fong, Y.K.; Sim, E.K. Tree stability in an improved soil to withstand wind loading. Urban For. Urban Green. 2009, 8, 237–247. [Google Scholar] [CrossRef]
- Peltola, H.; Kellomäki, S.; Hassinen, A.; Granander, M. Mechanical stability of Scots pine, Norway spruce and birch: An analysis of tree-pulling experiments in Finland. For. Ecol. Manag. 2000, 135, 143–153. [Google Scholar] [CrossRef]
- Bolte, A.; Tahmann, T.; Kuhr, M.; Pogoda, P.; Murach, D.; Gadow, K.V. Relationship between tree dimension and coarse root biomass in mixed stands of European beech (Fagus sylvatica L.) and Norway spruce (Picea abies [L.] Karst.). Plant Soil. 2004, 264, 1–11. [Google Scholar] [CrossRef]
- Liepiņš, J.; Lazdiņš, A.; Liepiņš, K. Equations for estimating above- and belowground biomass of Norway spruce, Scots pine, birch spp. and European aspen in Latvia. Scand. J. For. Res. 2017, 33, 58–70. [Google Scholar] [CrossRef]
- Kennedy, F. The Identification of Soils for Forest Management; Forestry Commission Field Guide; Forestry Commission: Edinburgh, UK, 2002; p. 56. [Google Scholar]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015. [Google Scholar]
- Bušs, K. Forest ecosystem classification in Latvia. In Proceedings of the Latvian Academy of Sciences B, Rīga, Latvia, 15 January 1997; Volume 51, pp. 204–218. [Google Scholar]
- Krisans, O.; Matisons, R.; Rust, S.; Burnevica, N.; Bruna, L.; Elferts, D.; Kalvane, L.; Jansons, A. Presence of Root Rot Reduces Stability of Norway Spruce (Picea abies): Results of Static Pulling Tests in Latvia. Forests 2020, 11, 416. [Google Scholar] [CrossRef]
- Cools, N.; De Vos, B. 2010: Sampling and Analysis of Soil. Manual Part X, 208 pp. In Manual on Methods and Criteria for Harmonized Sampling, Assessment, Monitoring and Analysis of the Effects of Air Pollution on Forests; UNECE, ICP Forests: Hamburg, Germany, 2010; ISBN 978-3-926301-03-1. Available online: http://www.icp-forests.org/Manual.htm (accessed on 2 November 2020).
- ISO 11465: 1993/ACl:1994 Soil Quality. Determination of Dry Matter and Water Content on a Mass Basis. Gravimetric Method. Available online: https://www.iso.org/obp/ui/#iso:std:iso:11465:en (accessed on 15 December 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 22 April 2020).
- Rigg, G.B.; Harrar, E.S. The root systems of trees growing in sphagnum. Am. J. Bot. 1931, 18, 391–397. [Google Scholar] [CrossRef]
- Gardiner, B.; Byrne, K.; Hale, S.; Kamimura, K.; Mitchell, S.J.; Peltola, H.; Ruel, J.C. A review of mechanistic modelling of wind damage risk to forests. Forestry 2008, 81, 447–463. [Google Scholar] [CrossRef]
- Coder, K.D. Tree Anchorage & Root Strength. University of Georgia Warnell School of Forestry & Natural Resources Monograph Publication. WSFNR14-9. 2014. Available online: https://www.warnell.uga.edu/outreach/publications/individual/tree-anchorage-root-strength-manual (accessed on 4 November 2020).
- Krišāns, O.; Samariks, V.; Donis, J.; Jansons, A. Structural Root-plate characteristics of wind-thrown Norway spruce in hemiboreal forests of Latvia. Forests 2020, 11, 1143. [Google Scholar] [CrossRef]
- Henwood, K. A structural model of forces in buttressed tropical rain forest trees. Biotropica 1973, 5, 83–93. [Google Scholar] [CrossRef]
- Fayle, D.C.F. Radial growth in tree roots. In Technical Report No. 9; University of Toronto, Faculty of Forestry: Toronto, ON, Canada, 1968; p. 183. [Google Scholar]
- Schmid, I.; Kazda, M. Root distribution of Norway spruce in monospecific and mixed stands on different soils. For. Ecol. Manag. 2002, 159, 37–47. [Google Scholar] [CrossRef]
- Persson, H.Å.; Stadenberg, I. Fine root dynamics in a Norway spruce forest (Picea abies (L.) Karst) in eastern Sweden. Plant Soil. 2010, 330, 329–344. [Google Scholar] [CrossRef]
- Samariks, V.; Istenais, N.; Seipulis, A.; Miezite, O.; Krisans, O.; Jansons, A. Root-soil plate characteristics of silver birch on wet and dry mineral soils in Latvia. Forests 2020. under review. [Google Scholar] [CrossRef]
- Crotteau, J.S.; Ritchie, M.W. Long-term stand growth of interior ponderosa pine stands in response to structural modifications and burning treatments in northeastern California. J. For. 2014, 112, 412–423. [Google Scholar] [CrossRef]
- Puhe, J. Growth and development of the root system of Norway spruce (Picea abies) in forest stands—A review. For. Ecol. Manag. 2003, 175, 253–273. [Google Scholar] [CrossRef]
| Variable | Tree Age (Years) | H (m) | DBH (cm) | Root-Plate Width (m) | Root-Plate Depth (m) |
|---|---|---|---|---|---|
| Min | 41–60 | 23.3 | 21.0 | 1.5 | 0.2 |
| Max | 26.7 | 36.0 | 2.5 | 1.2 | |
| Average (±CI *) | 25.0 ± 0.7 | 27.5 ± 3.3 | 1.2 ± 0.2 | 0.8 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samariks, V.; Brizga, D.; Rūba, J.; Seipulis, A.; Jansons, Ā. Root-Plate Characteristics of Common Aspen in Hemiboreal Forests of Latvia: A Case Study. Forests 2021, 12, 32. https://doi.org/10.3390/f12010032
Samariks V, Brizga D, Rūba J, Seipulis A, Jansons Ā. Root-Plate Characteristics of Common Aspen in Hemiboreal Forests of Latvia: A Case Study. Forests. 2021; 12(1):32. https://doi.org/10.3390/f12010032
Chicago/Turabian StyleSamariks, Valters, Dace Brizga, Jeļena Rūba, Andris Seipulis, and Āris Jansons. 2021. "Root-Plate Characteristics of Common Aspen in Hemiboreal Forests of Latvia: A Case Study" Forests 12, no. 1: 32. https://doi.org/10.3390/f12010032
APA StyleSamariks, V., Brizga, D., Rūba, J., Seipulis, A., & Jansons, Ā. (2021). Root-Plate Characteristics of Common Aspen in Hemiboreal Forests of Latvia: A Case Study. Forests, 12(1), 32. https://doi.org/10.3390/f12010032






