Norway Spruce Survival Rate in Two Forested Landscapes, 1975–2016
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Data
2.3. Data Analysis
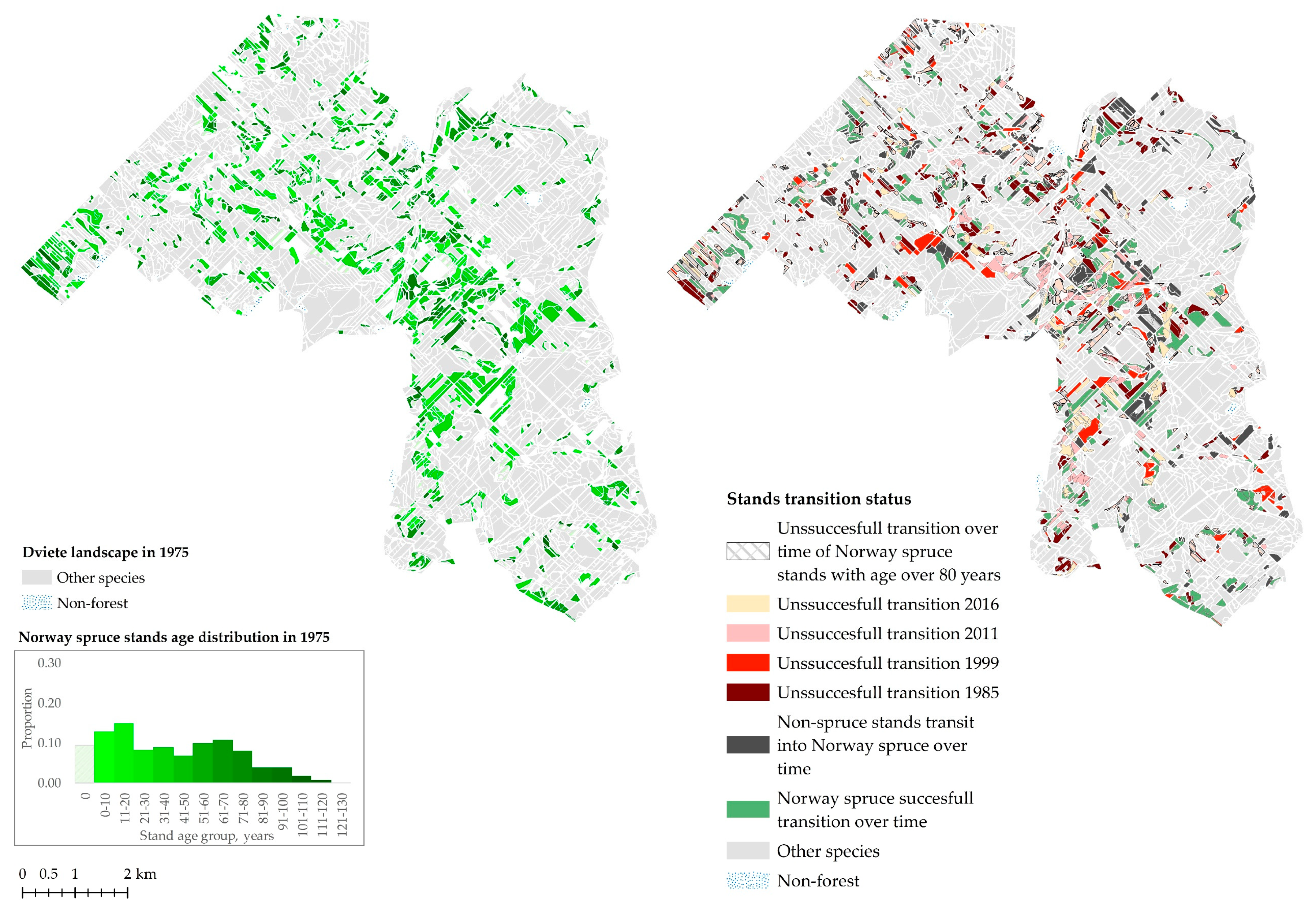
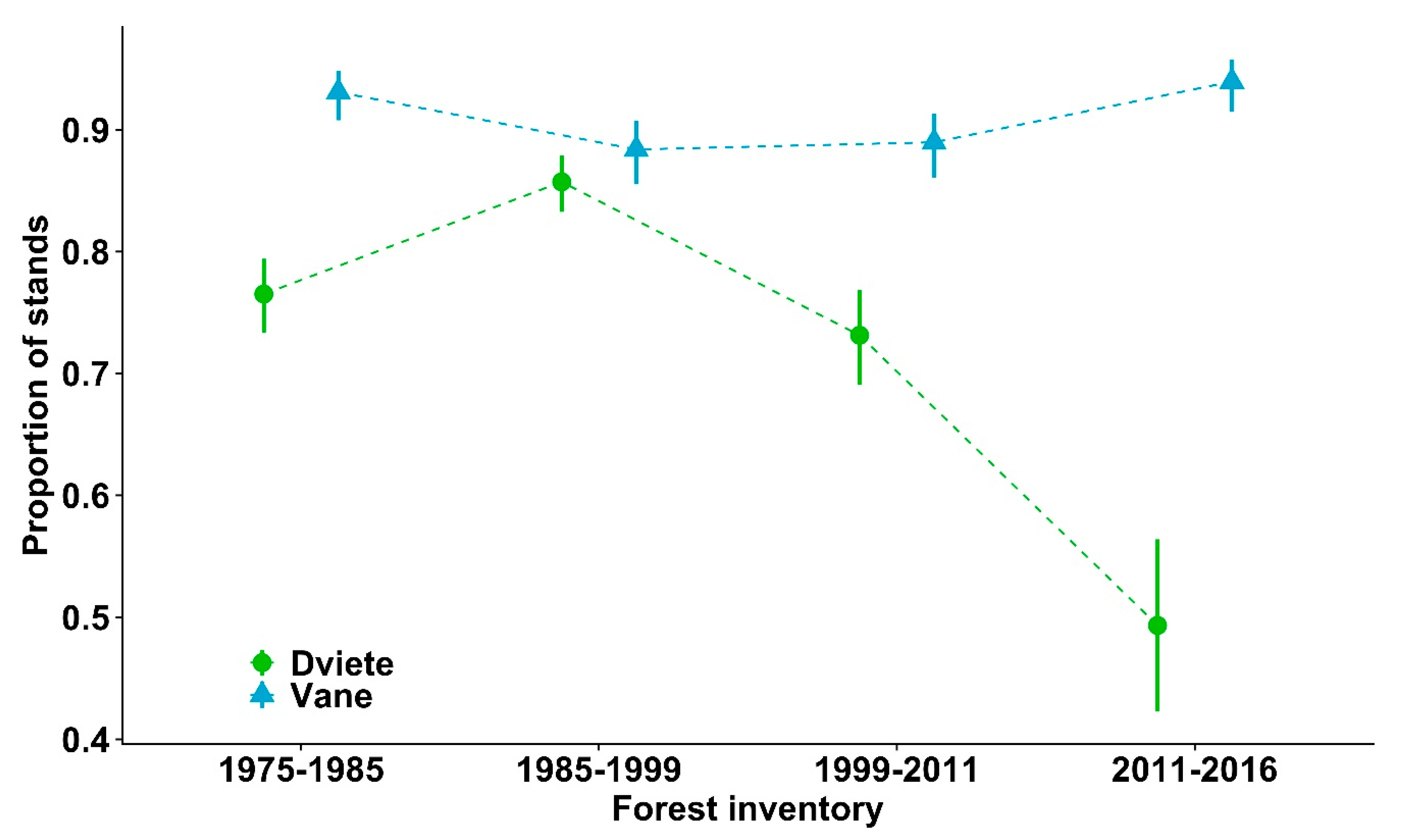
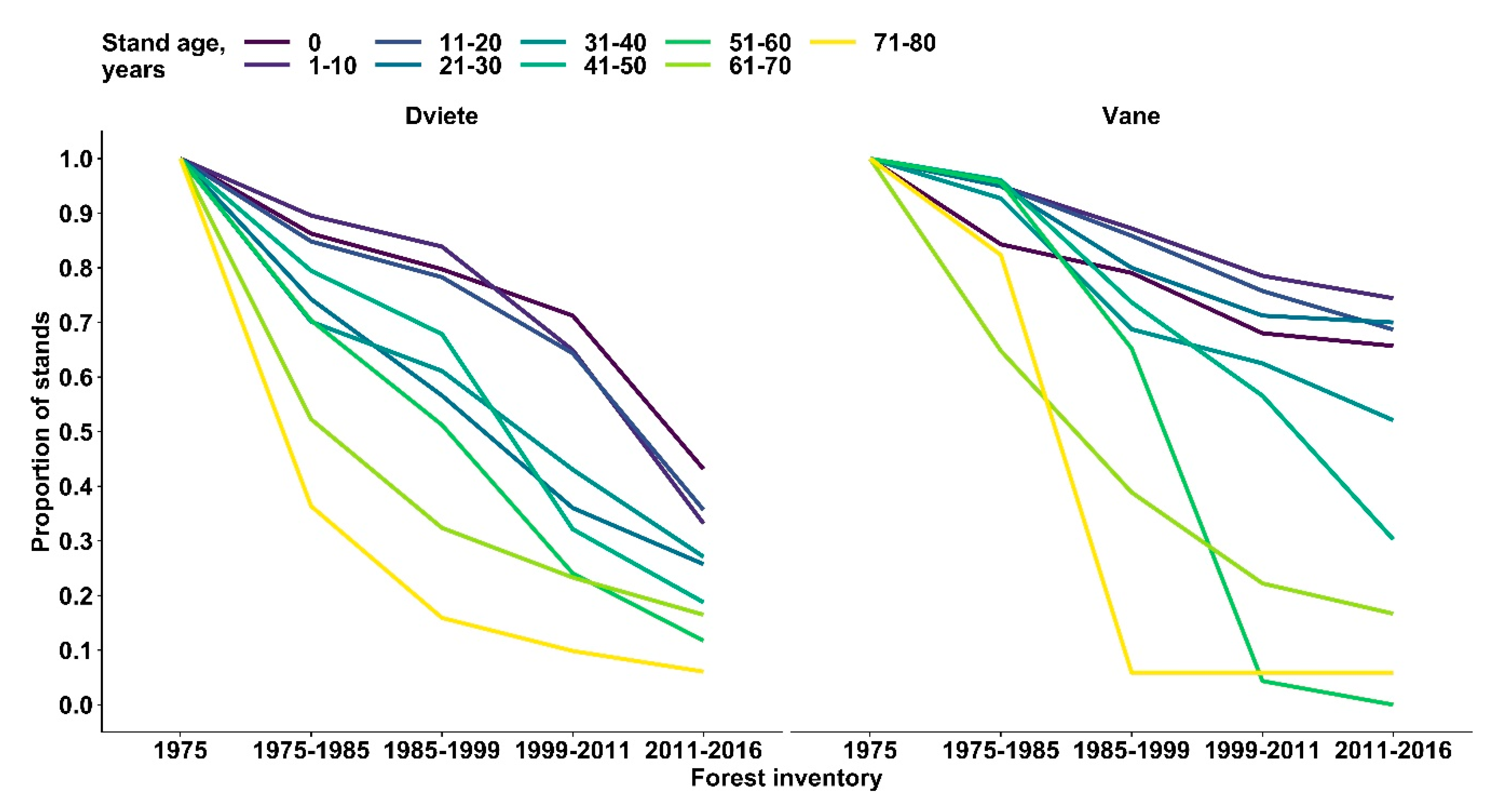
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crookston, N.L.; Rehfeldt, G.E.; Dixon, G.E.; Weiskittel, A.R. Addressing climate change in the forest vegetation simulator to assess impacts on landscape forest dynamics. For. Ecol. Manag. 2010, 260, 1198–1211. [Google Scholar] [CrossRef]
- Bugmann, H. Forests in a greenhouse atmosphere Predicting the unpredictable? In Forests and Global Change; Coomes, D.A., Burslem, D.F.R.P., Simonson, W.D., Eds.; Cambridge University Press: Cambridge, UK, 2014; pp. 359–380. [Google Scholar] [CrossRef]
- Pabst, R.J.; Goslin, M.N.; Garman, S.L.; Spies, T.A. Calibrating and testing a gap model for simulating forest management in the Oregon Coast Range. For. Ecol. Manag. 2008, 256, 958–972. [Google Scholar] [CrossRef]
- Fontes, L.; Bontemps, J.; Bugmann, H.; van Oijen, M.; Gracia, C.A.; Kramer, K.; Lindner, M.; Rotzer, T.; Skovsgaard, J.P. Models for supporting forest management in a changing environment. For. Syst. 2011, 3, 8. [Google Scholar] [CrossRef]
- Kimmins, J.P.; Welham, C.; Seely, B.; Meitner, M.; Rempel, R.; Sullivan, T. Science in forestry: Why does it sometimes disappoint or even fail us? For. Chron. 2005, 81, 723–734. [Google Scholar] [CrossRef][Green Version]
- Rennolls, K.; Tome, M.; Mcroberts, R.E.; Vanclay, J.K.; Lemay, V.; Guan, B.T.; Gertner, G.Z. Potential Contributions of Statistics and Modelling to Sustainable Forest Management: Review and Synthesis. In Sustainable Forestry: From Monitoring and Modelling to Knowledge Management & Policy Science; Reynolds, K.M., Thomson, A.J., Kohl, M., Shannon, M.A., Ray, D., Rennolls, K., Eds.; CABI Publishing: Boston, MA, USA, 2007; pp. 314–341. [Google Scholar] [CrossRef]
- Peng, C. Growth and yield models for uneven-aged stands: Past, present and future. For. Ecol. Manag. 2000, 132, 259–279. [Google Scholar] [CrossRef]
- Radeloff, V.C.; Mladenoff, D.J.; Boyce, M.S. The changing relation of landscape patterns and jack pine budworm populations during an outbreak. Oikos 2000, 90, 417–430. [Google Scholar] [CrossRef]
- Seidl, R.; Fernandes, P.M.; Fonseca, T.; Gillet, F.; Jonsson, A.M.; Merganicova, K.; Netherer, S.; Arpaci, A.; Bontemps, J.; Bugmann, H.; et al. Modelling natural disturbances in forest ecosystems: A review. Ecol. Model. 2011, 222, 903–924. [Google Scholar] [CrossRef]
- Waring, R.H.; Running, S.W. Forest EcosystemsAnalysis at Multiple Scales; Elsevier Academic Press: San Diego, CA, USA, 2007; Volume 63. [Google Scholar] [CrossRef]
- Reyer, C.P.O.; Bathgate, S.; Blennow, K.; Borges, J.G.; Bugmann, H.; Delzon, S.; Faias, S.P.; Garciagonzalo, J.; Gardiner, B.; Gonzalezolabarria, J.R.; et al. Are forest disturbances amplifying or canceling out climate change-induced productivity changes in European forests? Environ. Res. Lett. 2017, 12. [Google Scholar] [CrossRef]
- Keane, R.E.; Austin, M.P.; Field, C.B.; Huth, A.; Lexer, M.J.; Peters, D.P.C.; Solomon, A.M.; Wyckoff, P.H. Tree mortality in gap models: Application to climate change. Clim. Chang. 2001, 51, 509–540. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Im, S.T.; Dvinskaya, M.L.; Golukov, A.S.; Ranson, K.J. Climate-induced mortality of spruce stands in Belarus. Environ. Res. Lett. 2015, 10. [Google Scholar] [CrossRef]
- Lischke, H.; Zimmermann, N.E.; Bolliger, J.; Rickebusch, S.; Löffler, T.J. TreeMig: A forest-landscape model for simulating spatio-temporal patterns from stand to landscape scale. Ecol. Model. 2006, 199, 409–420. [Google Scholar] [CrossRef]
- Rasche, L.; Fahse, L.; Bugmann, H. Key factors affecting the future provision of tree-based forest ecosystem goods and services. Clim. Chang. 2013, 118, 579–593. [Google Scholar] [CrossRef]
- Perera, A.H.; Sturtevant, B.R.; Buse, L.J. Simulation Modeling of Forest Landscape Disturbances; Springer International Publishing: Geneva, Switzerland, 2015; pp. 1–321. [Google Scholar] [CrossRef]
- Hülsmann, L.; Bugmann, H.; Brang, P. How to predict tree death from inventory data lessons from a systematic assessment of European tree mortality models. Can. J. For. Res. 2017, 47, 890–900. [Google Scholar] [CrossRef]
- Spiecker, H. Silvicultural management in maintaining biodiversity and resistance of forests in Europe—Temperate zone. J. Environ. Manag. 2003, 67, 55–65. [Google Scholar] [CrossRef]
- Schlyter, P.; Stjernquist, I.; Bärring, L.; Jönsson, A.M.; Nilsson, C. Assessment of the impacts of climate change and weather extremes on boreal forests in northern Europe, focusing on Norway spruce. Clim. Res. 2006, 31, 75–84. [Google Scholar] [CrossRef]
- Schelhaas, M.J.; Nabuurs, G.J.; Schuck, A. Natural disturbances in the European forests in the 19th and 20th centuries. Glob. Chang. Biol. 2003, 9, 1620–1633. [Google Scholar] [CrossRef]
- Gardiner, B.; Blennow, K. Destructive Storms in European Forests: Past and Forthcoming Impacts. 2010. Available online: http://www.cabdirect.org/abstracts/20113168903.html (accessed on 24 February 2015).
- Möykkynen, T.; Miina, J. Optimizing the management of a butt-rotted Picea abies stand infected by Heterobasidion annosum from the previous rotation. Scand. J. For. Res. 2002, 17, 47–52. [Google Scholar] [CrossRef]
- Nikolov, C.; Konôpka, B.; Kajba, M.; Galko, J.; Kunca, A.; Janský, L. Post-disaster Forest Management and Bark Beetle Outbreak in Tatra National Park, Slovakia. Mt. Res. Dev. 2014, 34, 326–335. [Google Scholar] [CrossRef]
- Seidl, R.; Rammer, W. Climate change amplifies the interactions between wind and bark beetle disturbances in forest landscapes. Landsc. Ecol. 2017, 32, 1485–1498. [Google Scholar] [CrossRef]
- Krisans, O.; Saleniece, R.; Rust, S.; Elferts, D.; Kapostins, R.; Jansons, A.; Matisons, R. Effect of bark-stripping on mechanical stability of Norway Spruce. Forests 2020, 11, 357. [Google Scholar] [CrossRef]
- Mäkinen, H.; Nöjd, P.; Mielikäinen, K. Climatic signal in annual growth variation in damaged and healthy stands of Norway spruce [Picea abies (L.) Karst.] in southern Finland. Trees Struct. Funct. 2001, 15, 177–185. [Google Scholar] [CrossRef]
- Solberg, S. Summer drought: A driver for crown condition and mortality of Norway spruce in Norway. For. Pathol. 2004, 34, 93–104. [Google Scholar] [CrossRef]
- Seidl, R.; Schelhaas, M.; Rammer, W.; Verkerk, P.J. Increasing forest disturbances in Europe and their impact on carbon storage. Nat. Clim. Chang. 2014, 4, 806–810. [Google Scholar] [CrossRef]
- Libiete, Z.; Zalitis, P. Determining the growth potential for even-aged stands of Norway spruce (Picea abies (L.) karst.). Balt. For. 2007, 13, 2–9. [Google Scholar]
- ESRI. ArcGIS Desktop: Release 10; Environmental Systems Research Institute: Redlands, CA, USA, 2014. [Google Scholar]
- Bergstedt, J.; Milberg, P. The impact of logging intensity on field-layer vegetation in swedish boreal forests. For. Ecol. Manag. 2001, 154, 105–115. [Google Scholar] [CrossRef]
- Fridman, J.; Ståhl, G. A three-step approach for modelling tree mortality in Swedish forests. Scand. J. For. Res. 2001, 16, 455–466. [Google Scholar] [CrossRef]
- Hefti, R.; Schmid-Haas, P.; Buhler, U. Zustand Und Gefahrdung Der Davoser Waldungen; MAB-Schlussberichte, 23; Bundesamt fur Umweltschutz: Bern, Switzerland, 1986. [Google Scholar]
- Orazio, C.; Montoya, R.C.; Régolini, M.; Borges, J.G.; Garcia-Gonzalo, J.; Barreiro, S.; Botequim, B.; Marques, S.; Sedmák, R.; Smrecek, R.; et al. Decision support tools and strategies to simulate forest landscape evolutions integrating forest owner behaviour: A review from the case studies of the European project, INTEGRAL. Sustainability 2017, 9, 599. [Google Scholar] [CrossRef]
- Pukkala, T.; Laiho, O.; Lähde, E. Continuous cover management reduces wind damage. For. Ecol. Manag. 2016, 372, 120–127. [Google Scholar] [CrossRef]
- Zeng, H.; Peltola, H.; Talkkari, A.; Strandman, H.; Venalainen, A.; Wang, K.; Kellomaki, S. Simulations of the influence of clear-cutting on the risk of wind damage on a regional scale over a 20-year period. Can. J. For. Res. 2006, 36, 2247–2258. [Google Scholar] [CrossRef]
- Blennow, K.; Sallnäs, O. WINDA—A system of models for assessing the probability of wind damage to forest stands within a landscape. Ecol. Model. 2004, 175, 87–99. [Google Scholar] [CrossRef]
- Baders, E.; Jansons, A.; Matisons, R.; Elferts, D.; Desaine, I. Landscape diversity for reduced risk of insect damage: A case study of spruce bud scale in Latvia. Forests 2018, 9, 545. [Google Scholar] [CrossRef]
- Hanewinkel, M.; Breidenbach, J.; Neeff, T.; Hanewinkel, E.K.M. Seventy-seven years of natural disturbances in a mountain forest area—The influence of storm, snow, and insect damage analysed with a long-term time series. Can. J. For. Res. 2008, 38, 2249–2261. [Google Scholar] [CrossRef]
- Miezite, O.; Okmanis, M.; Indriksons, A. Assessment of sanitary conditions in stands of Norway spruce (Picea abies Karst.) damaged by spruce bud scale (Physokermes piceae Schrnk.). iForest 2013, 6, 73–78. [Google Scholar] [CrossRef]
- Bengtsson, A.; Nilsson, C. Extreme value modelling of storm damage in Swedish forests. Nat. Hazards Earth Syst. Sci. 2007, 7, 515–521. [Google Scholar] [CrossRef]
- Hanewinkel, M.; Hummel, S.; Albrecht, A. Assessing natural hazards in forestry for risk management: A review. Eur. J. For. Res. 2011, 130, 329–351. [Google Scholar] [CrossRef]
- Vitali, V.; Büntgen, U.; Bauhus, J. Silver Fir and Douglas Fir Are More Tolerant to Extreme Droughts than Norway Spruce in South-Western Germany; The Physical Science Basis. Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar] [CrossRef]
- Lazdina, D.T.; Stals, S.C. Experimental forest regeneration after selected initially highly productive, but later withered spruce stand on peat land (Oxalidosa turf. mel.). In Forest and Earth Entrails Resources: Research and Sustainable Utilization—New Products and Technologies (ResProd); National Research Programme, 2014–2018, Proceedings; Latvian State Institute of Wood Chemistry: Rīga, Latvia, 2018; pp. 37–43. [Google Scholar]
- Zeng, H.; Pukkala, T.; Peltola, H. The use of heuristic optimization in risk management of wind damage in forest planning. For. Ecol. Manag. 2007, 241, 189–199. [Google Scholar] [CrossRef]
- Peltola, H.; Kellomäki, S.; Väisänen, H.; Ikonen, V.P. A mechanistic model for assessing the risk of wind and snow damage to single trees and stands of Scots pine, Norway spruce, and birch. Can. J. For. Res. 1999, 29, 647–661. [Google Scholar] [CrossRef]
- Terauds, A.; Brumelis, G.; Nikodemus, O. Seventy-year changes in tree species composition and tree ages in state-owned forests in Latvia. Scand. J. For. Res. 2011, 26, 446–456. [Google Scholar] [CrossRef]
- Piri, T. The spreading of the S type of Heterobasidion annosum from Norway spruce stumps to the subsequent tree stand. Eur. J. For. Pathol. 1996, 26, 193–204. [Google Scholar] [CrossRef]
- Arhipova, N.; Gaitnieks, T.; Donis, J.; Stenlid, J.; Vasaitis, R. Butt rot incidence, causal fungi, and related yield loss in Picea abies stands in Latvia. Can. J. For. Res. 2011, 41, 2337–2345. [Google Scholar] [CrossRef]
- Bušs, K. Mežu Ekosistēmu Daudzveidība Un Stabilitāte [Forest Ecosystem Diversity and Stability]; Mežsaimniecība un mežrūpniecība: Rīga, Latvija, 1984. [Google Scholar]
- Zālītis, P. Mežkopības Priekšnosacījumi [Prerequisites Offorest Silviculture]; SIA et Cetera: Rīga, Latvija, 2006. [Google Scholar]
- Thorsen, B.J.; Helles, F. Optimal stand management with endogenous risk of sudden destruction. For. Ecol. Manag. 1998, 108, 287–299. [Google Scholar] [CrossRef]
- Katrevičs, J.; Džeriņa, B.; Neimane, U.; Desaine, I.; Bigača, Z.J.Ā. Production and profitability of low density Norway spruce (Picea abies (L.) Karst.) plantation at 50 years of age: Case study from eastern Latvia. Agron. Res. 2018, 16. [Google Scholar] [CrossRef]
- Donis, J.; Saleniece, R.; Krisans, O.; Dubrovskis, E.; Kitenberga, M.; Jansons, A. A Financial Assessment of Windstorm Risks for Scots Pine Stands in Hemiboreal Forests. Forests 2020, 11, 566. [Google Scholar] [CrossRef]
- Samariks, V.; Krisans, O.; Donis, J.; Silamikele, I.; Katrevics, J. Cost-Benefit Analysis of Measures to Reduce Windstorm Impact in Pure Norway Spruce (Picea abies L. Karst.) Stands in Latvia. Forests 2020, 11, 576. [Google Scholar] [CrossRef]
- Subramanian, N.; Bergh, J.; Johansson, U.; Nilsson, U.; Sallnäs, O. Adaptation of forest management regimes in southern Sweden to increased risks associated with climate change. Forests 2016, 7, 8. [Google Scholar] [CrossRef]
- Eerikäinen, K.; Valkonen, S.; Saksa, T. Ingrowth, survival and height growth of small trees in uneven-aged picea abies stands in southern Finland. For. Ecosyst. 2014, 1, 5. [Google Scholar] [CrossRef]
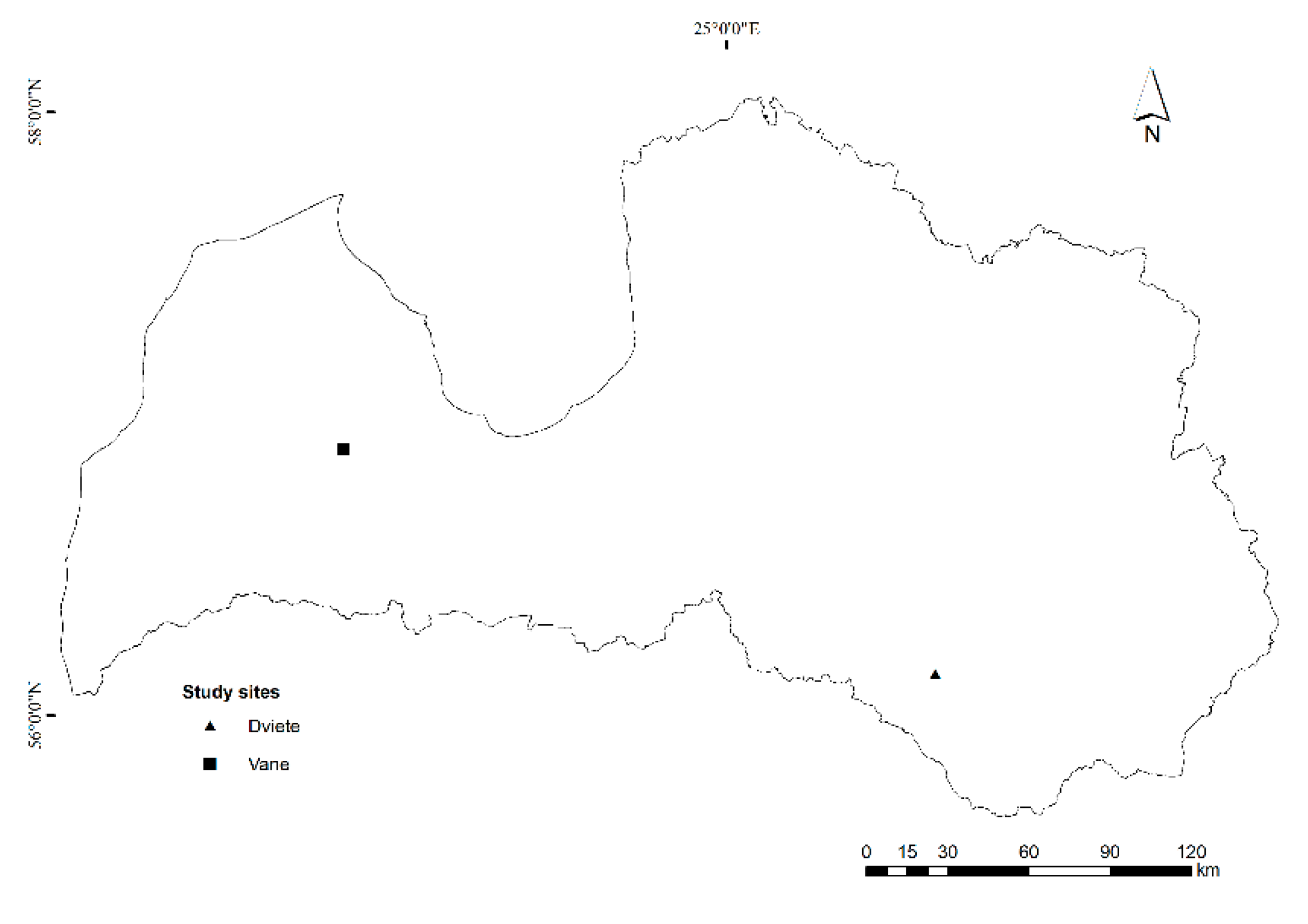



| Landscape | Latitude | Longitude | Species | Area, ha | Area, % | Mean Annual Precipitation, mm | Mean Annual Temperature, °C |
|---|---|---|---|---|---|---|---|
| Vane | 56°53′ N | 22°38′ E | Picea abies ((L.) Karst.) | 1307.1 | 37% | 650 | 6.2 |
| Pinus sylvestris L. | 784.2 | 22% | |||||
| Non-forest | 764.8 | 22% | |||||
| Betula pendula Roth | 513.7 | 15% | |||||
| Populus tremula L. | 101.0 | 3% | |||||
| Alnus glutinosa (L.) Gaertn. | 9.9 | 0% | |||||
| Alnus incana (L.) | 9.9 | 0% | |||||
| Total area | 3490.5 | ||||||
| Dviete | 56°08′ N | 26°15′ E | Pinus sylvestris L. | 3086.3 | 43% | 713 | 5.9 |
| Betula pendula Roth | 1534.7 | 21% | |||||
| Picea abies (L.) Karst.) | 1404.0 | 20% | |||||
| Non-forest | 709.4 | 10% | |||||
| Alnus glutinosa (L.) Gaertn. | 420.9 | 6% | |||||
| Populus tremula L. | 26.1 | 0% | |||||
| Alnus incana (L.) | 8.0 | 0% | |||||
| Total area | 7189.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bāders, E.; Krišāns, O.; Donis, J.; Elferts, D.; Jaunslaviete, I.; Jansons, Ā. Norway Spruce Survival Rate in Two Forested Landscapes, 1975–2016. Forests 2020, 11, 745. https://doi.org/10.3390/f11070745
Bāders E, Krišāns O, Donis J, Elferts D, Jaunslaviete I, Jansons Ā. Norway Spruce Survival Rate in Two Forested Landscapes, 1975–2016. Forests. 2020; 11(7):745. https://doi.org/10.3390/f11070745
Chicago/Turabian StyleBāders, Endijs, Oskars Krišāns, Jānis Donis, Didzis Elferts, Ieva Jaunslaviete, and Āris Jansons. 2020. "Norway Spruce Survival Rate in Two Forested Landscapes, 1975–2016" Forests 11, no. 7: 745. https://doi.org/10.3390/f11070745
APA StyleBāders, E., Krišāns, O., Donis, J., Elferts, D., Jaunslaviete, I., & Jansons, Ā. (2020). Norway Spruce Survival Rate in Two Forested Landscapes, 1975–2016. Forests, 11(7), 745. https://doi.org/10.3390/f11070745







