Temporal Progress of Candeia Rust Caused by Puccinia velata in Clonal Candeia Plantation (Eremanthus erythropappus (DC.) McLeisch)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site Description
2.2. Disease Assessment and Area under the Disease Progress Curve (AUDPC)
- AUDPC = area under the disease progress curve
- yi = proportion of disease at the ith observation
- ti = time in days at the ith observation
- n = total number of observations
2.3. Disease Progress Curve and Its Relationship with Climatic Variables
2.4. Fitting of Empirical Models to Disease Progression
2.5. Statistical Analysis
3. Results
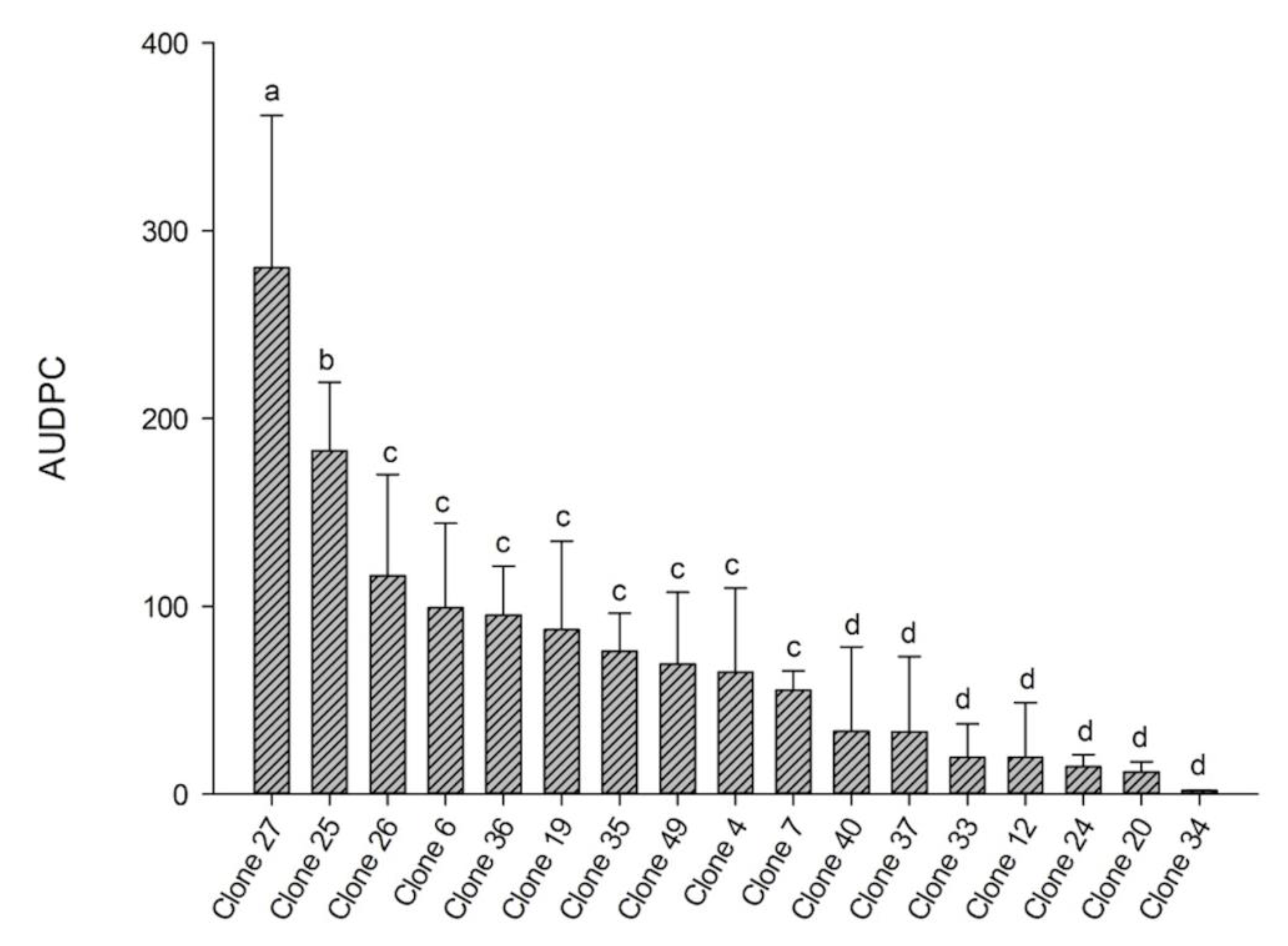
3.1. Area under the Disease Progress Curve
3.2. Disease Progress Curve and Its Relationship with Climatic Variables
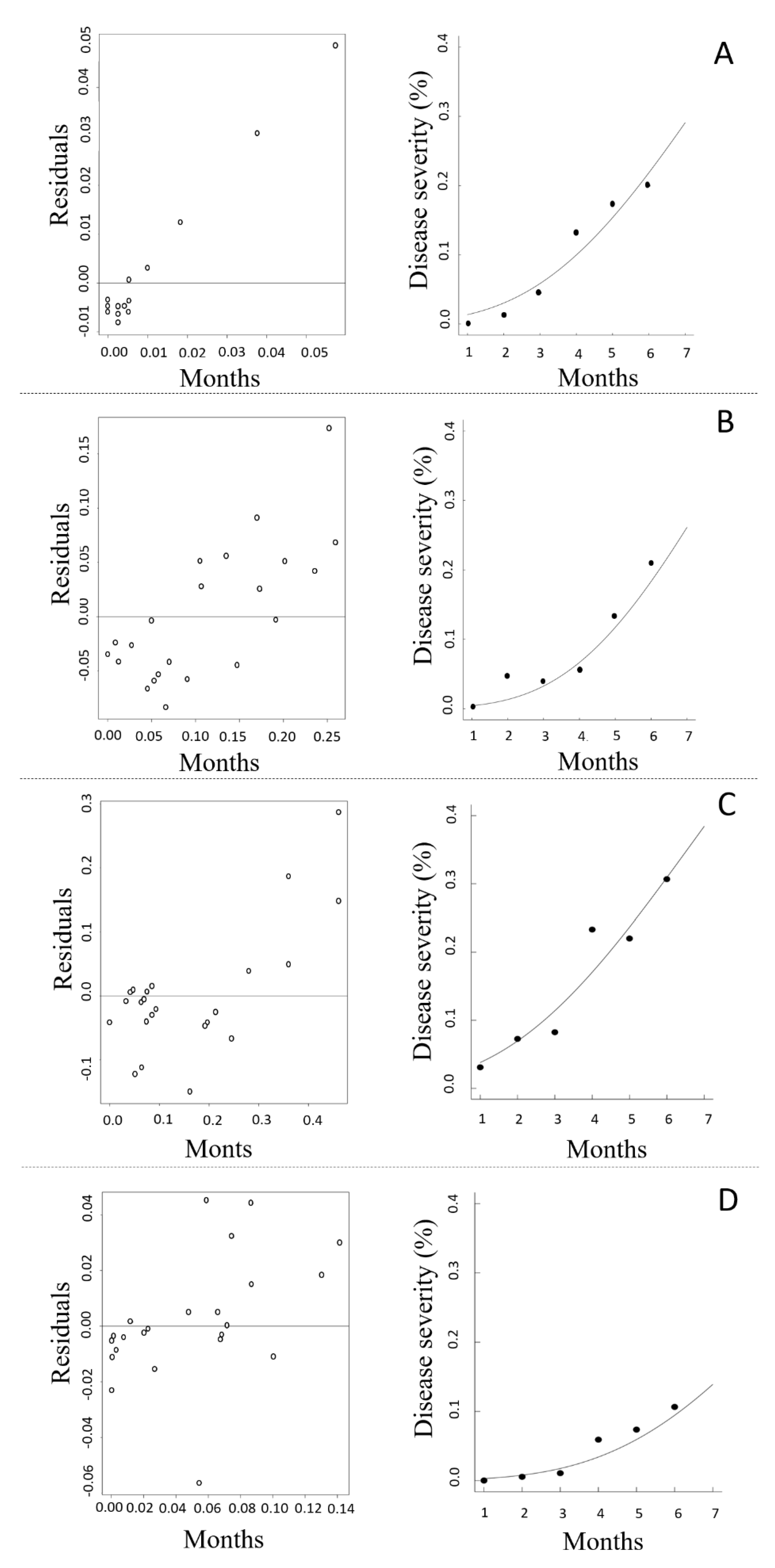
3.3. Fitting of Empirical Models to Candeia Rust Progress
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- de Araújo, E.J.G.; Pelissari, A.L.; David, H.C.; Scolforo, J.R.S.; Netto, S.P.; Morais, V.A. Relação hipsométrica para candeia (Eremanthus erythropappus) com diferentes espaçamentos de plantio em Minas Gerais, Brasil. Pesqui. Florest. Bras. 2012, 32, 257–268. [Google Scholar] [CrossRef]
- Carvalho, P.E.R. Espécies Florestais Brasileiras: Recomendações Silviculturais, Potencialidade e Uso Da Madeira; EMBRAPA-CNPF: Brasília, Brazil, 1994; p. 640. [Google Scholar]
- Teixeira, M.C.B.; Nunes, Y.R.F.; Maia, K.M.P.; Ribeiro, R.N. Influência da Luz na Germinação de Sementes de Candeia (Vanillosmopsis erythropappa Shuh. Bip.); Encontro regional de botânica, 28., 1996, Belo Horizonte; Anais. Belo Horizonte: SBB; Pontifícia Universidade Católica de Minas Gerais: Belo horizonte, Brazil, 1996; pp. 35–41. [Google Scholar]
- Scolforo, J.R.S.; Oliveira, A.D.; Davide, A.C. Manejo Sustentável da Candeia Eremanthus erythropappus e Eremanthus incanus; Relatório técnico científico; UFLA-FAEPE: Lavras, Brazil, 2002; p. 350. [Google Scholar]
- Prance, L. Cosmetics Design. Available online: https://www.cosmeticsdesign.com/Article/2007/06/07/Worldwide-manufacturers-harness-powers-of-bisabolol (accessed on 16 March 2020).
- Santos, R.C.; Mendes, L.M.; Mori, F.A.; Mendes, R.F. Aproveitamento de resíduos da madeira de candeia (Eremanthus erythropappus) para produção de painéis cimentomadeira. Cerne 2008, 14, 241–250. [Google Scholar]
- Silva, C.S.J.E.; de Oliveira, A.D.; Junior, L.M.C.; Scolforo, J.R.S.; de Souza, Á.N. Viabilidade econômica e rotação florestal de plantios de candeia (Eremanthus erythropappus), em condições de risco. Cerene 2014, 20, 113–122. [Google Scholar] [CrossRef]
- Dietel, P. Uredineae brasilienses a cl. E. Ulelectae. Hedwigia 1897, 26–37. [Google Scholar]
- Sales, N.I.S.; Correia, L.C.M.D.A.; Siqueira, C.D.A.; dos Santos, G.R.; Leão, E.U. Temporal progress of teak rust in a tropical area of Tocantins State, Brazil. Acta Amaz. 2017, 47, 277–280. [Google Scholar] [CrossRef]
- Galdino, A.P.P.; Brito, J.O.; Garcia, R.F.; Scolforo, J.R. Estudo sobre o rendimento e qualidade do óleo de candeia (Eremanthuss sp.) e an influência das diferentes origens comerciais da sua madeira. Rev. Brasil. Plantas Med. 2006, 8, 44–46. [Google Scholar]
- Bergamin, A.F.; Amorim, L.; Hau, B. Análise temporal e espacial de epidemias. In O essencial da fitopatologia: Epidemiologia de doenças de Planta; UFV: Viçosa, Brazil, 2014; pp. 101–165. [Google Scholar]
- Figueiredo, M.B.; Passador, M.M. Morfologia, Funções dos Soros e Variações dos Ciclos Vitais das Ferrugens; Arquivos do Instituto Biológico: São Paulo, Brazil, 2008; pp. 117–134. [Google Scholar]
- Carvalho, A.; Hennen, J.F. The species of Puccinia on Piptocarpha and Vanillosmopsis in the Neotropics. Mycologia 2012, 104, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Madden, L.V. Introduction to Plant Disease Epidemiology; John Wiley: New, York, NY, USA, 1990; p. 560. [Google Scholar]
- Vale, F.X.R.; Fernandes Filho, E.I.; Liberato, J.R. QUANT. A software plant disease severity assessment. In Proceedings of the 8th International Congress of Plant Pathology, Christchurch, New Zealand, 2–7 February 2003; p. 105. [Google Scholar]
- Colhoun, J. Effects of Environmental Factors on Plant Disease. Annu. Rev. Phytopathol. 1973, 11, 343–364. [Google Scholar] [CrossRef]
- Junior, W.C.D.J.; Bassanezi, R.B. Análise da dinâmica e estrutura de focos da morte súbita dos citros. Fitopatol. Bras. 2004, 29, 399–405. [Google Scholar] [CrossRef][Green Version]
- Vanderplank, J.E. Plant Diseases: Epidemics and Control; Academic Press: New York, NY, USA, 1963. [Google Scholar]
- Dantas, A.A.A.; de Carvalho, L.G.; Ferreira, E. Classificação e tendências climáticas em Lavras, MG. Ciência Agrotecnol. 2007, 31, 1862–1866. [Google Scholar] [CrossRef]
- Pereira, R.C.M.; Filho, C.A.L.; Melo, L.A.; Alozen, P.C.; Mafia, R.G.; Barros, A.F.; Ferreira, M.A. Diagrammatic scale for severity evaluation of the Eremanthus erythropappus—Puccinia velata pathosystem. J. Phtyopathol. 2019, 168, 135–143. [Google Scholar] [CrossRef]
- Anonymous. The R Project for Statistical Computing. Available online: http://www.r-project.org/ (accessed on 13 February 2012).
- Ruiz, R.A.R.; Alfenas, A.C.; Maffia, L.A.; Barbosa, M.D.M. Progresso da ferrugem do eucalipto, causada por Puccinia psidii, em condições de campo. Fitopatol. Brasil. 1989, 14, 73–81. [Google Scholar]
- Zauza, E.A.V.; Lana, V.M.; Maffia, L.A.; Araujo, M.M.F.C.; Alfenas, R.; Silva, F.F.; Alfenas, A.C. Wind dispersal of Puccinia psidii urediniospores and progress of eucalypt rust. For. Pathol. 2014, 45, 102–110. [Google Scholar] [CrossRef]
- Ruiz, R.A.R.; Alfenas, A.C.; Ferreira, F.A.; Vale, F.X.R. Influência da temperatura, do tempo de molhamento foliar, fotoperíodo e da intensidade de luz sobre an infeccão de Puccinia psidii em eucalipto. Fitopatol. Brasil. 1989, 14, 55–61. [Google Scholar]
- Fulton, J.D. Microorganisms of the upper atmosphere. V. Relationship between frontal activity and the micropopulation at altitude. J. Appl. Microbiol. 1966, 14, 245–250. [Google Scholar] [CrossRef]
- de Oliveira, M.E.S.; Fernandes, F.S.; Junior, M.A.G.; de Oliveira, A.S.; Mafia, R.G.; Ferreira, M.A. Temporal Analysis of Bacterial Leaf Blight in Clonal Eucalyptus Plantations in Brazil. Forests 2019, 10, 839. [Google Scholar] [CrossRef]
- Berger, R.D. Comparison of the Gompertz and Logistic Equations to Describe Plant Disease Progress. Phytopathology 1981, 71, 716. [Google Scholar] [CrossRef]
- de Mio, L.L.M.; Amorim, L.; Moreira, L.M. Progresso de epidemias e avaliação de danos da ferrugem em clones de álamo. Fitopatol. Bras. 2006, 31, 133–139. [Google Scholar] [CrossRef][Green Version]
- Masson, V.M.; Ohto, C.T.; Furtado, E.L.; Silva, A.S. Zoneamento climático do eucalipto no estado de São Paulo visando o controle da ferrugem. Summa Phytopathol. 2007, 33, 67. [Google Scholar]


| Climatic Variables | Clone | |||
|---|---|---|---|---|
| C25 | C27 | C24 | C35 | |
| Insolation | 0.205 | −0.03 | −0.117 | −0.069 |
| Cumulative rainfall | −0.698 * | −0.637 * | −0.287 | −0.601 * |
| Maximum temperature | −0.584 * | −0.816 * | −0.598 * | −0.799 * |
| Minimum temperature | −0.729 * | −0.833 * | −0.466 * | −0.845 * |
| Mean temperature | −0.703 * | −0.884 * | −0.602 * | −0.869 * |
| Relative humidity | 0.133 | 0.009 | 0.251 | 0.090 |
| Actual evapotranspiration | −0.772 * | −0.799 * | −0.359 | −0.818 * |
| Wind speed | −0.464 | −0.334 | 0.057 | −0.288 |
| Clone | Models | Rate (r) | y0 | R2 | MSR |
|---|---|---|---|---|---|
| Clone 24 | Linear | 0.044 *** | −0.063 * | 0.95 | 0.00140 |
| Monomolecular | 0.049 *** | −0.072 * | 0.94 | 0.00055 | |
| Exponential | 0.425 * | 0.017 | 0.88 | 0.00108 | |
| Logistic | 0.493 * | 0.014 | 0.89 | 0.00094 | |
| Gompertz | 0.208 ** | 0.005 | 0.93 | 0.00059 | |
| Clone 25 | Linear | 0.037 ** | −0.048 | 0.84 | 0.00108 |
| Monomolecular | 0.039 * | −0.052 | 0.82 | 0.00121 | |
| Exponential | 0.532 ** | 0.008 | 0.96 | 0.00026 | |
| Logistic | 0.603 ** | 0.006 | 0.96 | 0.00027 | |
| Gompertz | 0.234 ** | 0.001 | 0.94 | 0.00028 | |
| Clone 27 | Linear | 0.056 ** | −0.039 | 0.91 | 0.00130 |
| Monomolecular | 0.066 ** | −0.055 | 0.90 | 0.01180 | |
| Exponential | 0.532 ** | 0.008 | 0.88 | 0.01216 | |
| Logistic | 0.427 ** | 0.034 | 0.89 | 0.01214 | |
| Gompertz | 0.205 ** | 0.018 | 0.91 | 0.00130 | |
| Clone 35 | Linear | 0.022 ** | −0.035 * | 0.92 | 0.00017 |
| Monomolecular | 0.023 ** | −0.037 | 0.91 | 0.00019 | |
| Exponential | 0.506 ** | 0.005 | 0.92 | 0.00018 | |
| Logistic | 0.541 ** | 0.004 | 0.92 | 0.00017 | |
| Gompertz | 0.179 ** | 0.001 | 0.94 | 0.00012 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, R.C.M.; Ferreira, M.A.; Soares, T.P.F.; Andrade, M.F.C.M.; Filho, C.A.L.; Pozza, E.A.; Avelar, M.L.M.; Melo, L.A. Temporal Progress of Candeia Rust Caused by Puccinia velata in Clonal Candeia Plantation (Eremanthus erythropappus (DC.) McLeisch). Forests 2020, 11, 720. https://doi.org/10.3390/f11070720
Pereira RCM, Ferreira MA, Soares TPF, Andrade MFCM, Filho CAL, Pozza EA, Avelar MLM, Melo LA. Temporal Progress of Candeia Rust Caused by Puccinia velata in Clonal Candeia Plantation (Eremanthus erythropappus (DC.) McLeisch). Forests. 2020; 11(7):720. https://doi.org/10.3390/f11070720
Chicago/Turabian StylePereira, Renata C. M., Maria A. Ferreira, Thaissa P. F. Soares, Mario F. C. M. Andrade, Cézar A. L. Filho, Edson A. Pozza, Maria L. M. Avelar, and Lucas A. Melo. 2020. "Temporal Progress of Candeia Rust Caused by Puccinia velata in Clonal Candeia Plantation (Eremanthus erythropappus (DC.) McLeisch)" Forests 11, no. 7: 720. https://doi.org/10.3390/f11070720
APA StylePereira, R. C. M., Ferreira, M. A., Soares, T. P. F., Andrade, M. F. C. M., Filho, C. A. L., Pozza, E. A., Avelar, M. L. M., & Melo, L. A. (2020). Temporal Progress of Candeia Rust Caused by Puccinia velata in Clonal Candeia Plantation (Eremanthus erythropappus (DC.) McLeisch). Forests, 11(7), 720. https://doi.org/10.3390/f11070720




