Nutrient Status of Tree Seedlings in a Site Recovering from a Landslide
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Soil Sampling
2.2. Laboratory Analysis
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Binkley, D.; Vitousek, P. Soil nutrient availability. In Plant Physiological Ecology; Pearcy, R.W., Ehleringer, J.R., Mooney, H.A., Rundel, P.W., Eds.; Springer: Dordrecht, The Netherlands, 1989; pp. 75–96. [Google Scholar]
- Marschner, P.; Rengel, Z. Nutrient Availability in Soil. In Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Academic Press: London, UK; Waltham, MA, USA, 2012; pp. 315–330. [Google Scholar]
- Shiels, A.B.; Walker, L.R.; Thompson, D.B. Organic matter inputs create variable resource patches on Puerto Rican landslides. Plant Ecol. 2006, 184, 223–236. [Google Scholar] [CrossRef]
- Walker, L.R.; Neris, L. Posthurricane seed rain dynamics in Puerto Rico. Biotropica 1993, 25, 408–418. [Google Scholar] [CrossRef]
- Adams, P.W.; Sidle, R.C. Soil conditions in three recent landslide in Southeast Alaska. For. Ecol. Mang. 1987, 18, 93–102. [Google Scholar] [CrossRef]
- Walker, L.R.; Shiels, A.B. Physical Causes and Consequences for Landslide Ecology. In Landslide Ecology; Walker, L.R., Shiels, A.B., Eds.; Cambridge University Press: Cambridge, UK, 2013; p. 1640. [Google Scholar]
- Cheng, C.; Hsiao, S.; Huang, Y.; Hung, C.; Pai, C.; Chen, C.; Menyailo, O.V. Landslide-induces chan ges of soil physicochemical properties in Xitou, Central Taiwan. Geoderma 2016, 265, 187–195. [Google Scholar] [CrossRef]
- Błońska, E.; Lasota, J.; Piaszczyk, W.; Wiecheć, M.; Klamerus-Iwan, A. The effect of landslide on soil organic carbon Stock and biochemical properties of soil. J. Soils Sediments 2018, 18, 2727–2737. [Google Scholar] [CrossRef]
- Walker, L.R.; Zarin, D.J.; Fetcher, N.; Myster, R.W.; Johnson, A.H. Ecosystem Development and Plant Succession on Landslides in the Caribbean. Biotropica 1996, 28, 566–576. [Google Scholar] [CrossRef]
- Walker, L.R.; Velázquez, E.; Shiels, A.B. Applying lessons from ecological succession to the restoration of landslides. Plant Soil 2009, 324, 157–168. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, C.; Lin, S. Mechanisms of Forest Restoration in Landslide Treatment Areas. Sustainability 2014, 6, 6766–6780. [Google Scholar] [CrossRef]
- Pang, C.; Ma, X.K.; Hung, T.T.; Hau, B.C. Early ecological succession on landslide trails, Hong Kong, China. Ėcoscience 2018, 25, 153–161. [Google Scholar] [CrossRef]
- Schomakers, J.; Jien, S.; Lee, T.; Huang, J.; Hseu, Z.; Lin, Z.; Lee, L.; Hein, T.; Mantler, A.; Zehetner, F. Soil and biomass carbon re-accumulation after landslide disturbances. Geomorphology 2019, 288, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Błońska, E.; Lasota, J.; Zwydak, M.; Klamerus-Iwan, A.; Gołąb, J. Restoration of forest soil and vegetation 15 years after landslides in a lower zone of mountains in temperate climates. Ecol. Eng. 2016, 97, 503–515. [Google Scholar] [CrossRef]
- Restrepo, C.; Walker, L.R.; Shiels, A.B.; Bussmann, R.; Claessens, L.; Fisch, S.; Lozano, P.; Negi, G.; Paolini, L.; Poveda, G.; et al. Landsliding and Its Multiscale Influence on Mountainscapes. BioScience 2009, 59, 685–698. [Google Scholar] [CrossRef]
- Błońska, E.; Lasota, J.; Gruba, P. Enzymatic activity and stabilization of organic matter in soil with different detritus inputs. J. Soil Sci. Plant Nutr. 2017, 63, 242–247. [Google Scholar]
- Lasota, J.; Błońska, E. Polycyclic aromatic hydrocarbons content in contaminated forest soils with different humus type. Water Air Soil Pollut. 2018, 229, 204. [Google Scholar] [CrossRef] [PubMed]
- Lasota, J.; Błońska, E.; Łyszczarz, S.; Tibbett, M. Forest humus type governs heavy metal accumulation in specific organic matter fractions. Water Air Soil Pollut. 2020, 231, 80. [Google Scholar] [CrossRef]
- Kandeler, E.; Palli, S.; Stemmer, M.; Gerzabek, M.H. Tillage changes microbial biomass and enzymeactivities in particle size fractions. Soil Biol. Biochem. 1999, 31, 1253–1264. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Dick, W.A. Enzymes in soil. In Enzymes in the Environment: Activity, Ecology and Applications; Burns, R.G., Dick, R.P., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 567–596. [Google Scholar]
- Adetunji, A.T.; Lewu, F.B.; Mulidzi, R.; Ncube, B. The biological activities of β-glucosidase, phosphatase and urease as soil quality indicators: A review. J. Soil Sci. Plant Nutr. 2017, 17, 794–807. [Google Scholar] [CrossRef]
- Pająk, M.; Błońska, E.; Szostak, M.; Gąsiorek, M.; Pietrzykowski, M.; Urban, O.; Derbis, P. Restoration of Vegetation in Relation to Soil Properties of Spoil Heap Heavily Contaminated with Heavy Metals. Water Air Soil Pollut. 2018, 229, 392. [Google Scholar] [CrossRef]
- Kacprzyk, M.; Błońska, E.; Lasota, J. Effect of spot burning of logging residues on the properties of mountain forest soils and the occurrence of ground beetles (Coleoptera, Carabidae). J. Mt. Sci. 2019, 17, 31–41. [Google Scholar] [CrossRef]
- Gawęda, T.; Błońska, E.; Małek, S. Soil organic carbon accumulation in post-agricultural soils under the influence birch stands. Sustainability 2019, 11, 4300. [Google Scholar] [CrossRef]
- IUSS Working Group WRB, World Reference Base for Soil Resources 2014. International Soil Classification System for Naming Soil and Creating Legends for Soil Maps; Update 2015, World Soil Resources Reports No. 106; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2015; p. 190. [Google Scholar]
- Pritsch, K.; Raidl, S.; Marksteiner, E.; Blaschke, H.; Agerer, R.; Schloter, M.; Hartmann, A. A rapid and highly sensitive method for measuring enzyme activities in single mycorrhizal tips using 4-methylumbelliferone-labelled fluorogenic substrates in a microplate system. J. Microbiol. Methods 2004, 58, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Sanaullah, M.; Razavi, B.S.; Blagodatskaya, E.; Kuzyakov, Y. Spatial distribution and catalytic mechanisms of β-glucosidase activity at the root-soil interface. Biol. Fertil. Soils 2016, 52, 505–514. [Google Scholar] [CrossRef]
- Turner, B.L. Variation in pH Optima of Hydrolytic Enzyme Activities in Tropical Rain Forest Soils. Appl. Environ. Microbiol. 2010, 76, 6485–6493. [Google Scholar] [CrossRef] [PubMed]
- Małek, S.; Januszek, K.; Keeton, W.; Barszcz, J.; Kroczek, M.; Błońska, E.; Wanic, T. Preliminary effects of fertilization on ecochemical soil condition in mature spruce stands experiencing dieback in the Beskid Śląski and Żywiecki Mts., Poland. Water Air Soil Pollut. 2014, 225, 1971. [Google Scholar] [CrossRef] [PubMed]
- Błońska, E.; Małek, S.; Januszek, K.; Barszcz, J.; Wanic, T. Changes in forest soil properties and in spruce stands after dolomite, magnesite and serpentinite fertilization. Eur. J Forest Res. 2015, 134, 981–990. [Google Scholar] [CrossRef][Green Version]
- Kreutzer, K. Effects of forest liming on soil processes. Plant Soil 1995, 168, 447–470. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology; Sinauer Associates, Inc.: Sunderland, MA, USA, 2002; p. 690. [Google Scholar]
- Rennenberg, H.; Dannenmann, M. Nitrogen Nutrition of Trees in Temperate Forests—The Significance of Nitrogen Availability in the Pedosphere and Atmosphere. Forests 2015, 6, 2820–2835. [Google Scholar] [CrossRef]
- Robertson, G.P.; Groffman, P.M. Nitrogen transformations. In Soil Microbiology, Ecology and Biochemistry, 4th ed.; Paul, E.A., Ed.; Academic Press: Burlington, MA, USA, 2015; pp. 421–446. [Google Scholar]
- Lemanowicz, J. Dynamics of phosphorus content and the activity of phosphatase in forest soil in the sustained nitrogen compounds emissions zone. Environ. Sci. Pollut. Res. 2018, 25, 33773–33782. [Google Scholar] [CrossRef]
- Goldberg, S.; Sposito, G. On the mechanizm of specific phosphate adsorption by hydroxylated mineral surfaces: A review. Commun. Soil Sci. Plant 1985, 16, 801–821. [Google Scholar] [CrossRef]
- Giesler, R.; Petersson, T.; Högberg, P. Phosphorus Limitation in Boreal Forests: Effects of Aluminum and Iron Accumulation in the Humus Layer. Ecosystems 2002, 5, 300–314. [Google Scholar] [CrossRef]
- Kunito, T.; Hiruta, N.; Miyagishi, Y.; Sumi, H.; Moro, H. Changes in phosphorus fractions caused by increased microbial activity in forest soil in a short-term incubation study. Chem. Spec. Bioavailab. 2018, 30, 9–13. [Google Scholar] [CrossRef]
- Perala, D.; Alm, A.A. Reproductive Ecology of Birch: A Review. For. Ecol. Manag. 1990, 32, 1–38. [Google Scholar] [CrossRef]
- Fiedler, H.J.; Nebe, W.; Hoffmann, F. Forstliche Pflanzenernährung und Düngung; VEB Verlag Gustav Fischer: Jena, Germany, 1973; p. 481. [Google Scholar]
- Sobczak, R. Forest Nursery Production; Świat Publishing House: Warsaw, Poland, 1992; p. 192. (In Polish) [Google Scholar]
- Hagen-Thorn, A.; Varnagiryte, I.; Nihlgård, B.; Armolaitis, K. Autumn nutrient resorption and losses in four deciduous forest tree species. For. Ecol. Manag. 2006, 228, 33–39. [Google Scholar] [CrossRef]

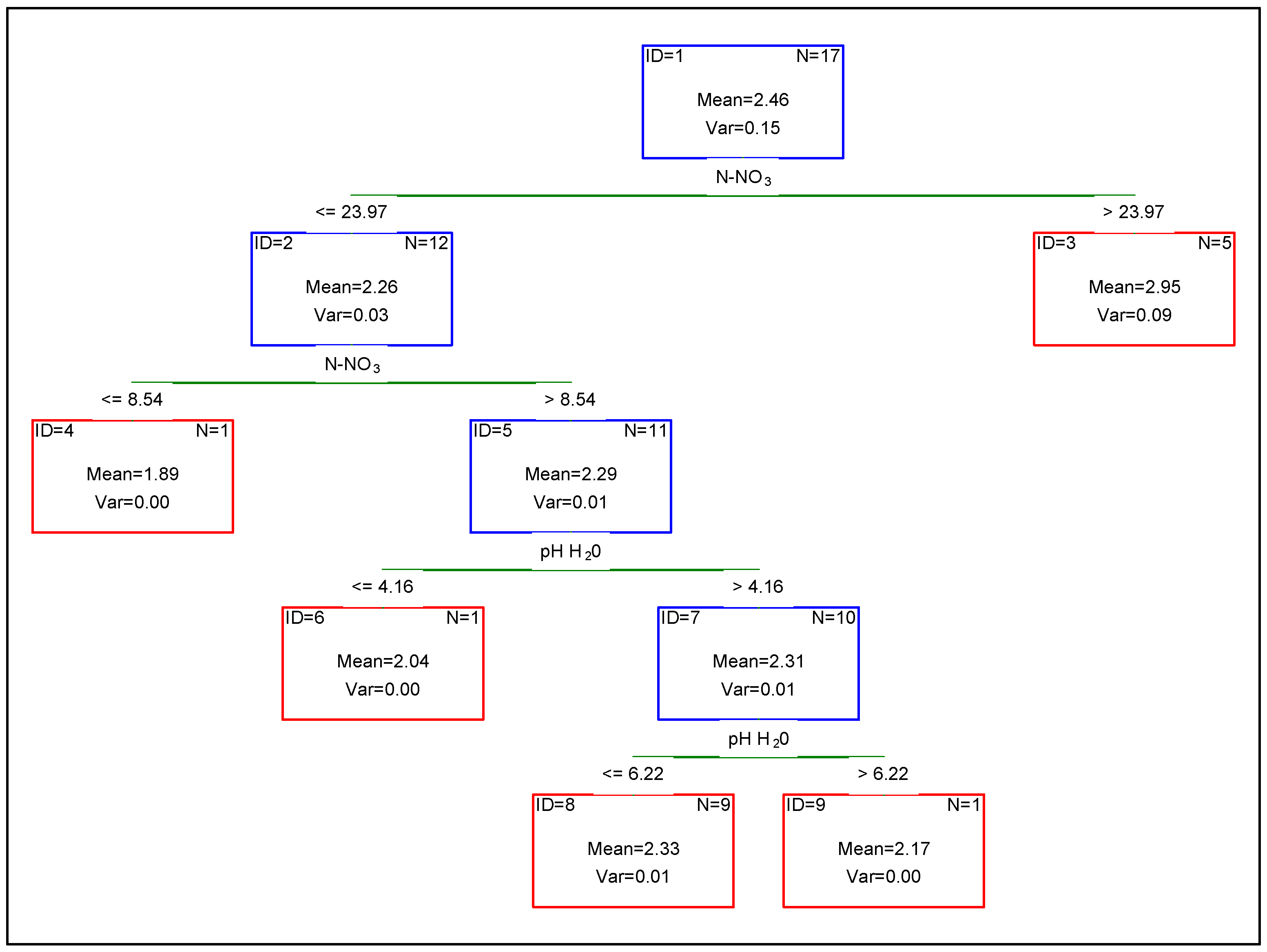
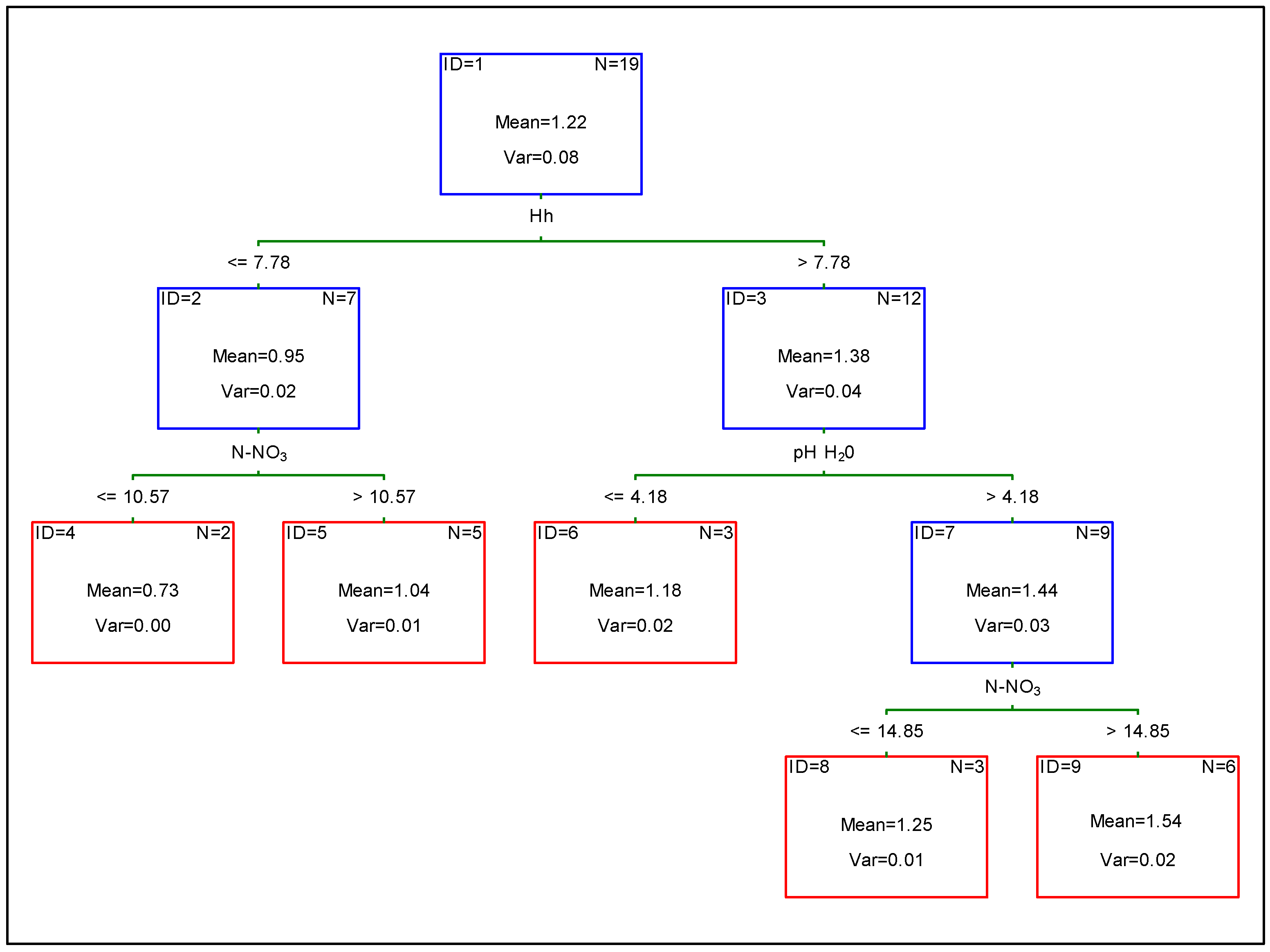
| pH H2O | pH KCl | Hh | He | C | N | C/N | Sand | Silt | Clay | |
|---|---|---|---|---|---|---|---|---|---|---|
| cmol (+)·kg−1 | % | % | ||||||||
| Zone of depletion | 5.12 ±0.74 a | 3.87 ±0.40 a | 7.26 ±3.62 a | 6.86 ±4.60 a | 0.92 ±0.20 b | 0.07 ±0.01 b | 14.20 ±2.34 a | 16.1 ±6.2 a | 69.4 ±3.7 a | 14.5 ±3.8 a |
| Zone of accumulation | 4.72 ±0.88 a | 3.83 ±0.82 a | 10.30 ±3.92 a | 8.79 ±3.81 a | 1.91 ±0.49 a | 0.14 ±0.03 a | 14.08 ±1.22 a | 20.5 ±11.1 a | 67.3 ±6.8 a | 12.1 ±4.8 a |
| Al | Ca | Mg | K | Na | P | Cu | Mn | Ni | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|
| mg·kg−1 | ||||||||||
| Zone of depletion | 24886.5 ±5340.5 a | 1377.3 ±899.2 a | 3706.9 ±1110.8 a | 8033.6 ±2893.6 a | 310.2 ±46.6 a | 182.4 ±65.4 b | 12.5 ±6.2 a | 568.8 ±233.1 a | 24.9 ±12.5 a | 43.0 ±11.5 a |
| Zone of accumulation | 24170.9 ±7445.8 a | 1151.3 ±1195.5 a | 3414.9 ±1743.4 a | 7593.8 ±4547.4 a | 312.7 ±95.2 a | 242.5 ±40.4 a | 11.5 ±8.17 a | 705.7 ±503.5 a | 25.1 ±24.3 a | 43.4 ±14.9 a |
| CB | BG | XYL | NAG | SP | PH | N-NH4 | N-NO3 | |
|---|---|---|---|---|---|---|---|---|
| nmol MUB·g−1d.s.·h−1 | mg·kg−1 | |||||||
| Zone of depletion | 21.62 ±9.95 b | 83.11 ±51.54 b | 24.28 ±19.23 a | 56.11 ±31.20 b | 4.51 ±3.84 a | 310.76 ±141.47 b | 0.94 ±0.13 b | 12.48 ±4.19 b |
| Zone of accumulation | 45.85 ±10.74 a | 169.62 ±48.65 a | 33.34 ±30.04 a | 253.98 ±72.45 a | 7.66 ±7.77 a | 686.35 ±294.69 a | 1.51 ±0.32 a | 24.33 ±3.84 a |
| Zone | Species | C | N | C/N | Ca | Mg | K | Na | Al |
|---|---|---|---|---|---|---|---|---|---|
| % | mg·kg−1 d.m. | ||||||||
| Zone of depletion | Birch | 44.43 ±4.54 a | 2.27 ±0.17 b | 19.60 ±1.53 a | 3546.2 ±718.8 a | 2159.2 ±606.4 a | 7494.1 ±968.5 a | 117.2 ±18.8 a | 1993.9 ±1703.4 a |
| Fir | 46.43 ±1.45 a | 1.10 ±0.26 b | 44.58 ±11.53 a | 5992.9 ±968.8 a | 791.6 ±382.2 a | 6266.6 ±1141.2 a | 88.2 ±25.2 a | 1453.9 ±1620.0 a | |
| Zone of accumulation | Birch | 43.86 ±3.88 a | 2.74 ±0.47 a | 16.25 ±1.78 b | 3171.1 ±555.5 a | 1918.3 ±358.0 a | 7755.3 ±1005.3 a | 132.1 ±32.8 a | 2122.3 ±1627.0 a |
| Fir | 45.76 ±1.28 a | 1.40 ±0.24 a | 33.66 ±6.33 b | 5895.6 ±1355.0 a | 659.2 ±251.7 a | 5466.0 ±921.3 a | 70.4 ±18.3 a | 1248.2 ±777.9 a | |
| Zone | Species | Ca/Al | Mg/Al | Ca/Mg | P | Cu | Co | Mn | Ni | Zn |
|---|---|---|---|---|---|---|---|---|---|---|
| mg·kg−1 | ||||||||||
| Zone of depletion | Birch | 3.56 ± 4.14 a | 2.13 ± 2.65 a | 1.72 ± 0.44 a | 2051.6 ± 515.2 a | 16.60 ± 1.53 b | 2.10 ± 1.53 a | 1668.4 ± 411.8 a | 11.26 ± 4.22 a | 120.13 ± 22.73 a |
| Fir | 6.01 ± 2.26 a | 0.69 ± 0.22 a | 8.45 ± 2.29 a | 1150.0 ± 474.7 a | 16.39 ± 5.94 a | 0.83 ± 0.74 a | 1302.5 ± 519.6 a | 12.81 ± 4.93 a | 29.39 ± 8.06 a | |
| Zone of accumulation | Birch | 2.73 ± 2.09 a | 1.56 ± 1.20 a | 1.68 ± 0.34 a | 1879.3 ± 288.7 a | 19.21 ± 2.90 a | 1.89 ± 1.19 a | 1786.9 ± 132.1 a | 11.64 ± 6.65 a | 137.71 ± 35.08 a |
| Fir | 6.25 ± 3.31 a | 0.66 ± 0.34 a | 9.57 ± 2.79 a | 1120.8 ± 387.0 a | 12.11 ± 9.39 a | 0.60 ± 0.28 a | 1439.4 ± 650.0 a | 12.12 ± 8.10 a | 34.78 ± 6.98 a | |
| C | N | C/N | Ca | Mg | K | Na | Al | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | p | F | p | F | p | F | p | F | p | F | p | F | p | F | p | |
| Zone of landslide | 0.33 | 0.568 | 15.19 | 0.000 | 8.89 | 0.006 | 0.53 | 0.471 | 1.58 | 0.218 | 0.60 | 0.445 | 0.03 | 0.855 | 0.01 | 0.941 |
| Tree species | 3.29 | 0.079 | 160.64 | 0.000 | 78.40 | 0.000 | 63.74 | 0.000 | 78.32 | 0.000 | 25.48 | 0.000 | 30.81 | 0.000 | 1.90 | 0.178 |
| Zone of landslide × Tree species | 0.00 | 0.964 | 0.76 | 0.389 | 2.50 | 0.124 | 0.18 | 0.671 | 0.13 | 0.717 | 2.32 | 0.138 | 3.99 | 0.054 | 0.11 | 0.747 |
| Ca/Al | Mg/Al | Ca/Mg | P | Cu | Co | Mn | Ni | Zn | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | p | F | p | F | p | F | p | F | p | F | p | F | p | F | p | F | p | |
| Zone of landslide | 0.08 | 0.784 | 0.34 | 0.566 | 0.75 | 0.394 | 0.45 | 0.507 | 0.19 | 0.670 | 0.37 | 0.548 | 0.45 | 0.508 | 0.01 | 0.940 | 2.70 | 0.111 |
| Tree species | 7.81 | 0.009 | 4.96 | 0.033 | 136.39 | 0.000 | 30.61 | 0.000 | 3.52 | 0.070 | 12.36 | 0.001 | 3.51 | 0.071 | 0.25 | 0.623 | 191.76 | 0.000 |
| Zone of landslide × Tree species | 0.25 | 0.620 | 0.27 | 0.608 | 0.85 | 0.363 | 0.23 | 0.637 | 3.13 | 0.087 | 0.00 | 0.987 | 0.00 | 0.962 | 0.07 | 0.794 | 0.76 | 0.390 |
| Nbirch | Nfir | Pbirch | Pfir | Kbirch | Kfir | Mgbirch | Mgfir | Cabirch | Cafir | Ca/Albirch | Ca/Alfir | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH H2O | 0.151 | −0.504 * | 0.512 * | 0.622 * | 0.408 | 0.400 | 0.702 * | 0.637 * | 0.661 * | 0.598 * | 0.567 * | −0.118 |
| pH KCl | 0.362 | −0.315 | 0.439 | 0.646 * | 0.279 | 0.327 | 0.504 * | 0.463 | 0.428 | 0.640 * | 0.430 | 0.070 |
| Hh | −0.000 | 0.584 * | −0.483 * | −0.550 * | −0.336 | −0.398 | −0.766 * | −0.653 * | −0.678 * | −0.556 * | −0.476 | 0.265 |
| He | −0.053 | 0.523 * | −0.462 | −0.566 * | −0.394 | −0.345 | −0.775 * | −0.659 * | −0.682 * | −0.544 * | −0.502 * | 0.240 |
| C | 0.417 | 0.498 * | −0.358 | −0.301 | −0.050 | −0.458 | −0.457 | −0.341 | −0.401 | −0.287 | −0.249 | 0.200 |
| N | 0.445 | 0.571 * | −0.313 | −0.323 | −0.046 | −0.457 | −0.494 * | −0.377 | −0.443 | −0.307 | −0.215 | 0.245 |
| N-NH4 | 0.338 | 0.521 * | −0.375 | −0.320 | −0.141 | −0.400 | −0.505 * | −0.320 | −0.482 | −0.330 | −0.249 | 0.272 |
| N-NO3 | 0.700 * | 0.574 * | −0.054 | −0.182 | 0.200 | −0.417 | −0.450 | −0.354 | −0.192 | −0.189 | 0.053 | 0.159 |
| silt | −0.010 | −0.465 | 0.392 | 0.238 | 0.510* | 0.367 | 0.494* | 0.343 | 0.486 * | 0.192 | 0.421 | −0.147 |
| clay | −0.066 | −0.127 | 0.334 | 0.421 | 0.152 | 0.433 | 0.271 | 0.469 | 0.352 | 0.365 | 0.140 | −0.359 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lasota, J.; Kraj, W.; Honkowicz, B.; Staszel, K.; Błońska, E. Nutrient Status of Tree Seedlings in a Site Recovering from a Landslide. Forests 2020, 11, 709. https://doi.org/10.3390/f11060709
Lasota J, Kraj W, Honkowicz B, Staszel K, Błońska E. Nutrient Status of Tree Seedlings in a Site Recovering from a Landslide. Forests. 2020; 11(6):709. https://doi.org/10.3390/f11060709
Chicago/Turabian StyleLasota, Jarosław, Wojciech Kraj, Bożena Honkowicz, Karolina Staszel, and Ewa Błońska. 2020. "Nutrient Status of Tree Seedlings in a Site Recovering from a Landslide" Forests 11, no. 6: 709. https://doi.org/10.3390/f11060709
APA StyleLasota, J., Kraj, W., Honkowicz, B., Staszel, K., & Błońska, E. (2020). Nutrient Status of Tree Seedlings in a Site Recovering from a Landslide. Forests, 11(6), 709. https://doi.org/10.3390/f11060709







