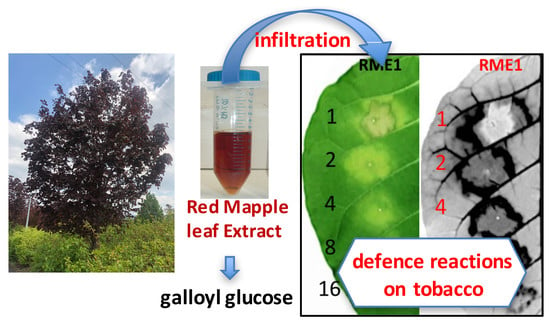
Inducing Plant Defense Reactions in Tobacco Plants with Phenolic-Rich Extracts from Red Maple Leaves: A Characterization of Main Active Ingredients
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Total Polyphenols Extraction and Quantification
2.3. Tobacco Treatments
2.4. Chemicals
2.5. Scopoletin Assay
2.6. Semi-Quantitative Real-Time RT-PCR
2.7. HPLC-UV and UPLC-HRMS Analyses
2.8. Alkaline Hydrolysis of RME1 and RME2
2.9. Glucose Quantification
2.10. Statistical Analysis
3. Results and Discussion
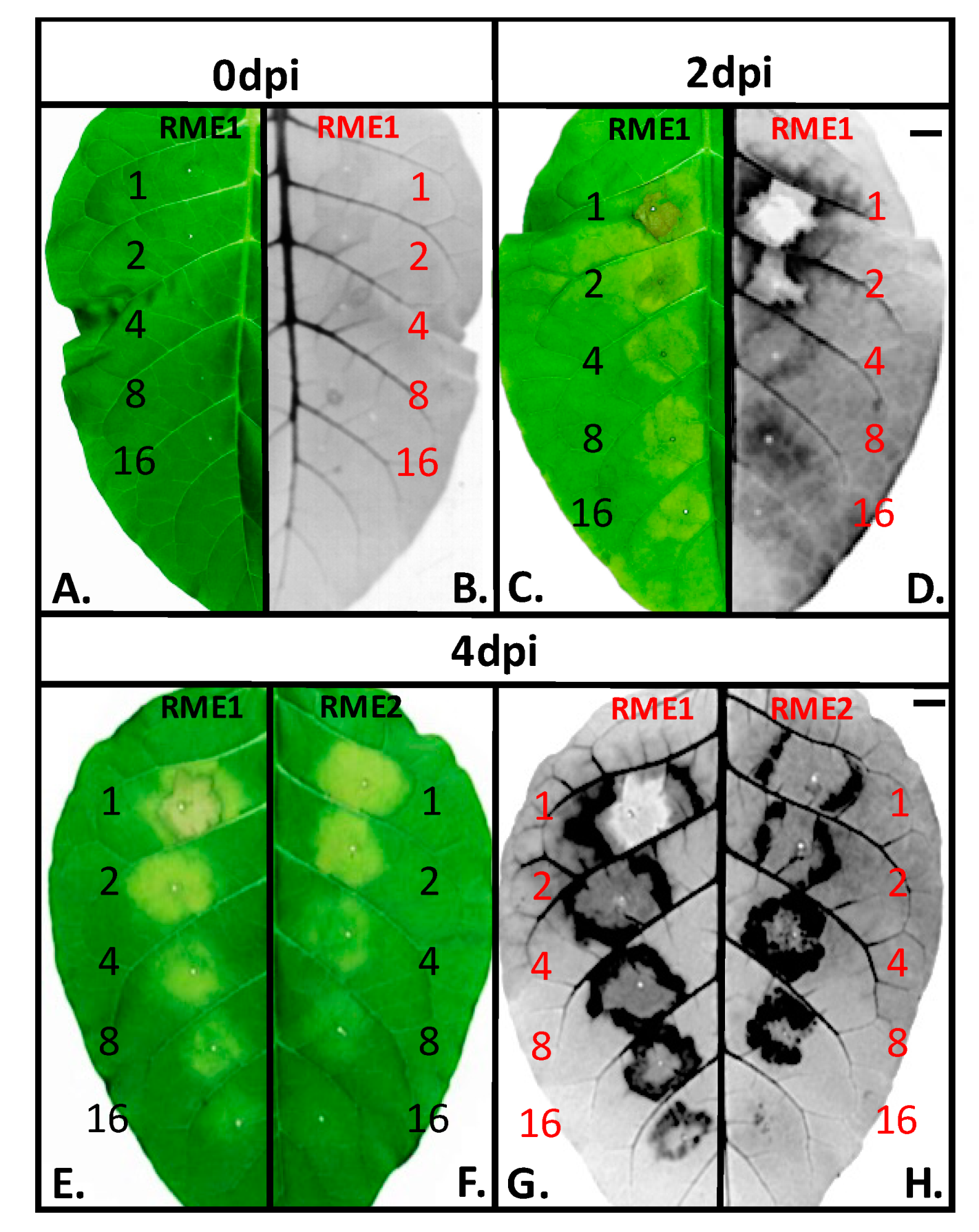
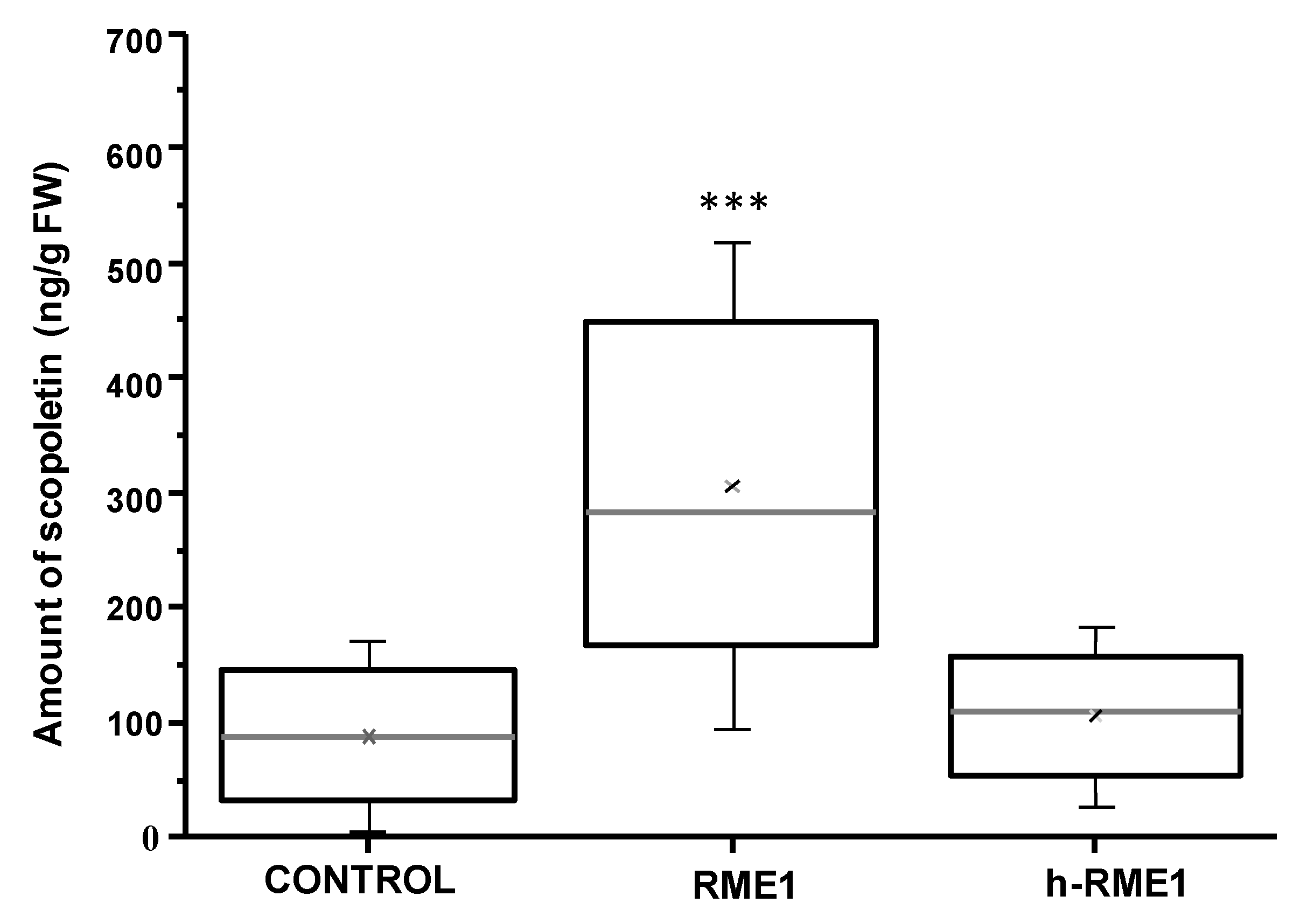
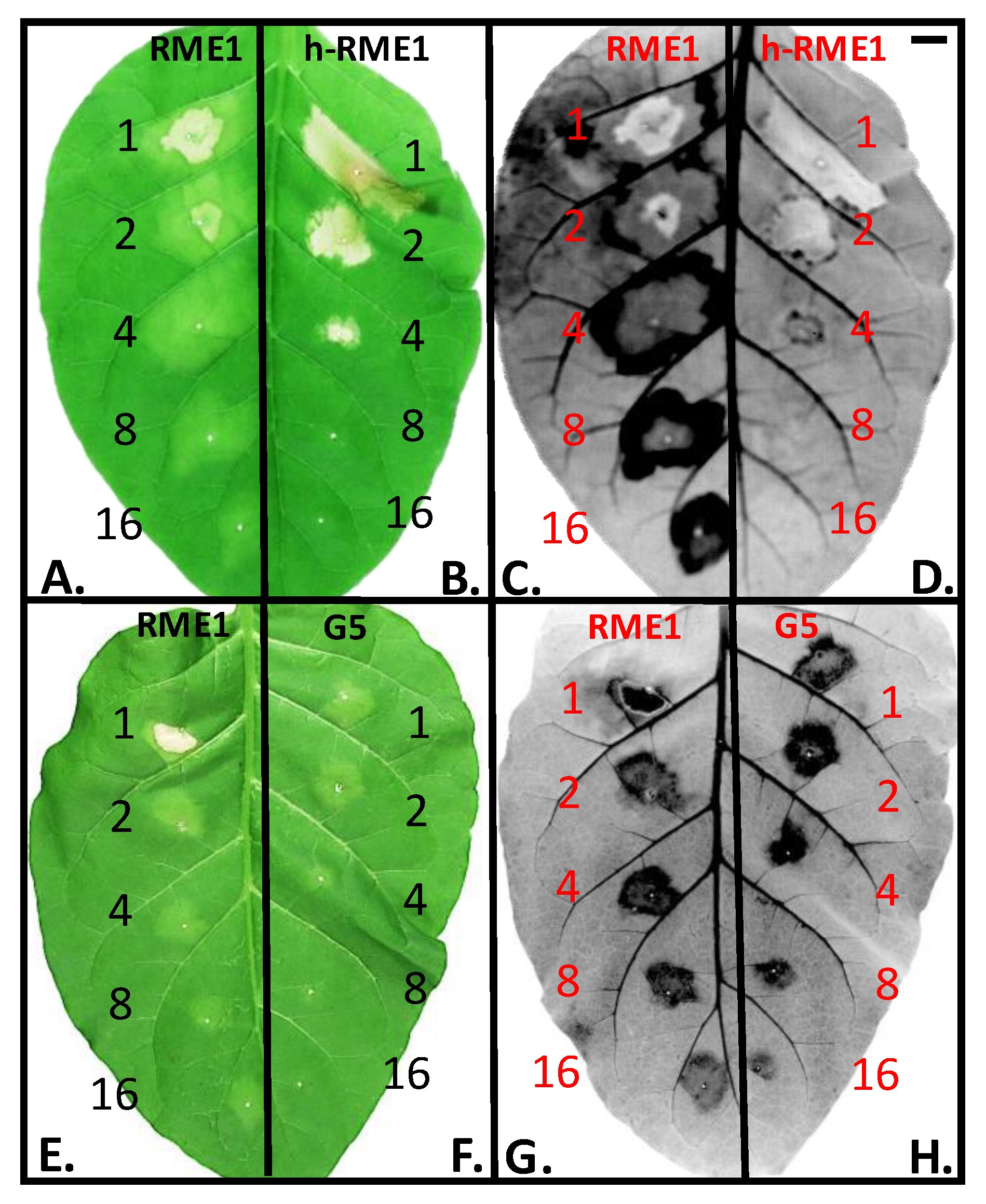
3.1. Plant Defense Inducer (PDI) Activity of Enriched-Polyphenol Red Maple Extracts (RME)
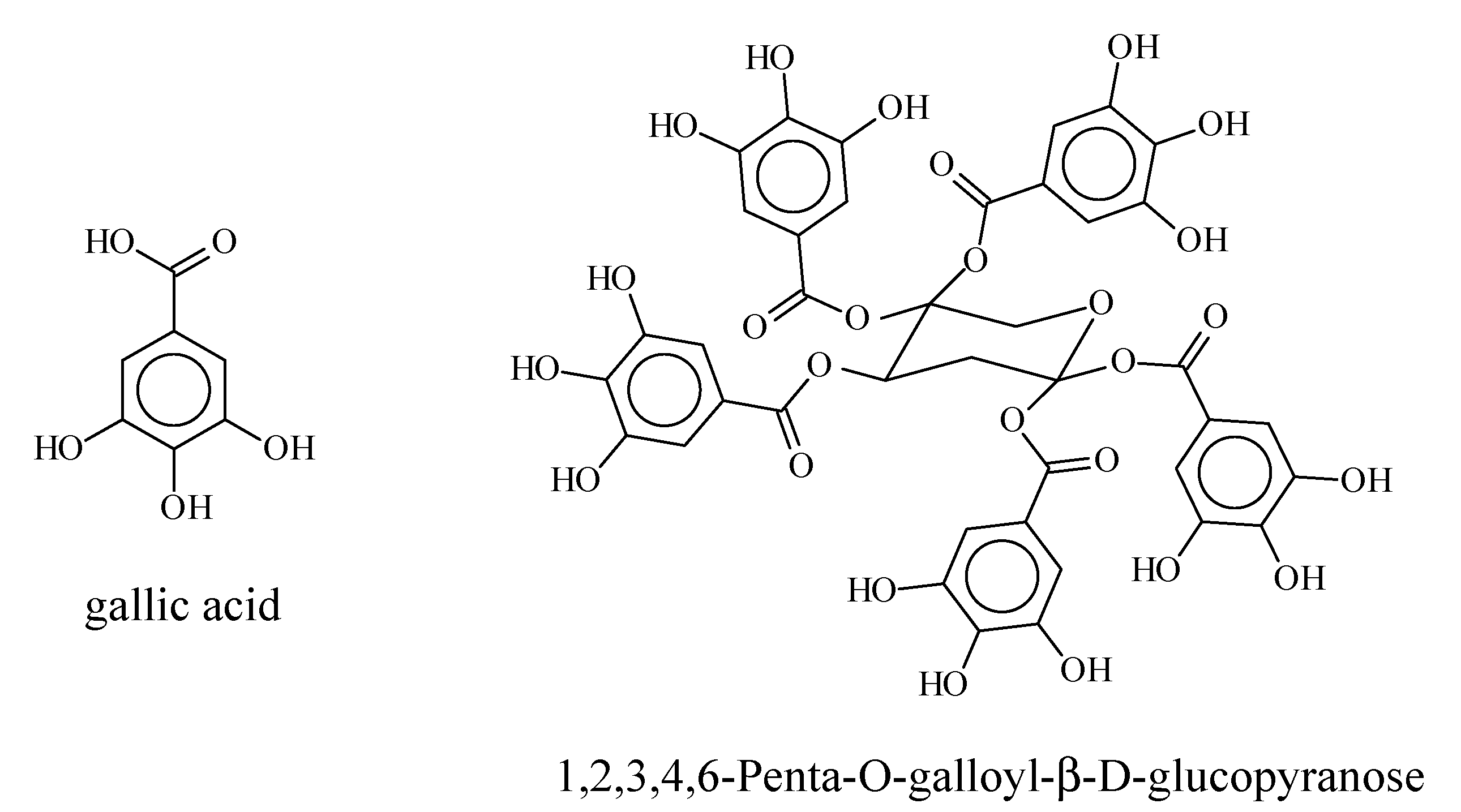
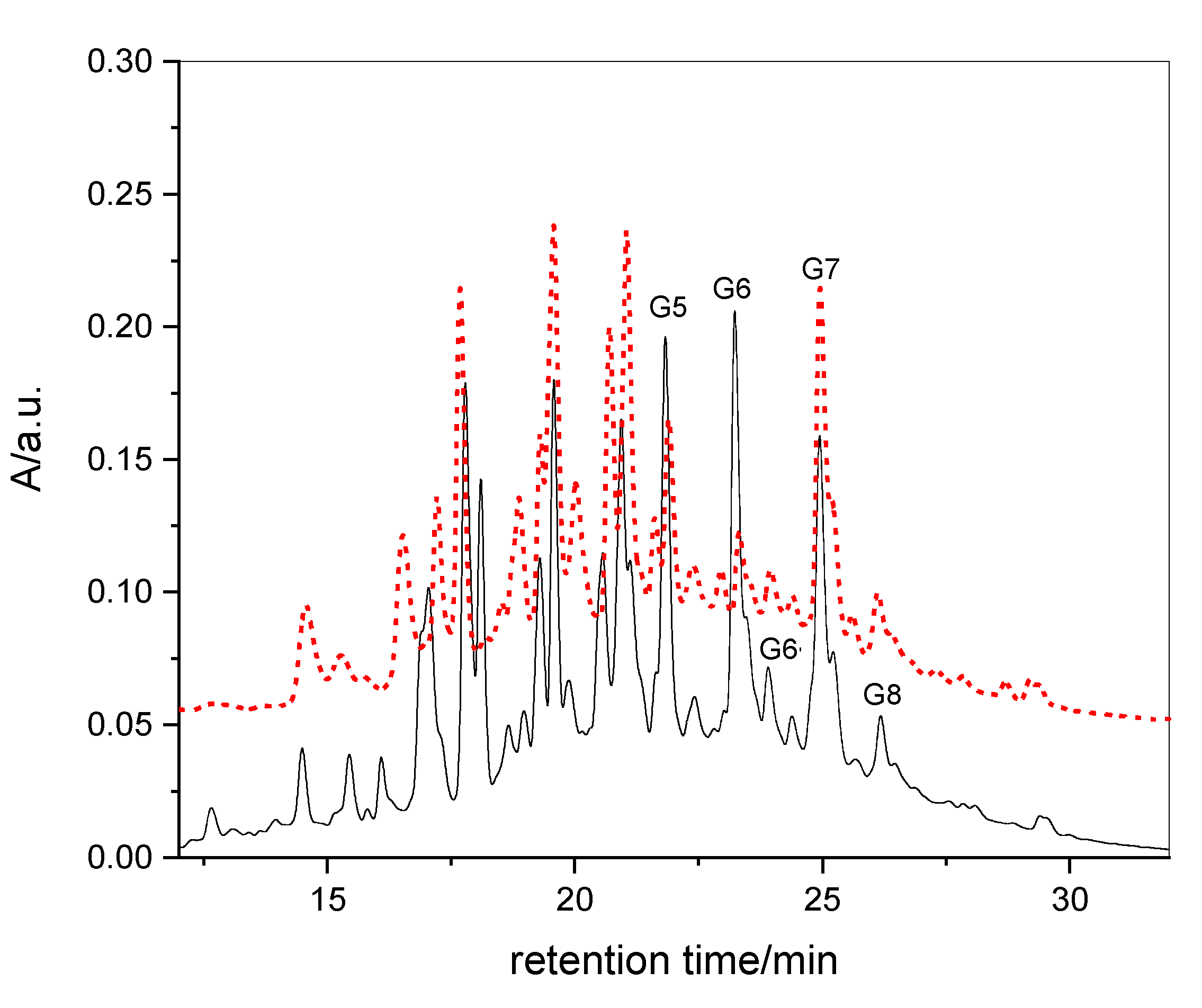
3.2. HPLC-UV Fingerprints and UPLC-HR-MS Analysis of RME1 and RME2
3.3. Quantification of Gallate Functions by Alkaline Hydrolysis
3.4. Quantification of Gallotannins in RME1
3.5. Suppression of Topical Symptoms Induced by Alkaline Hydrolysed RME1
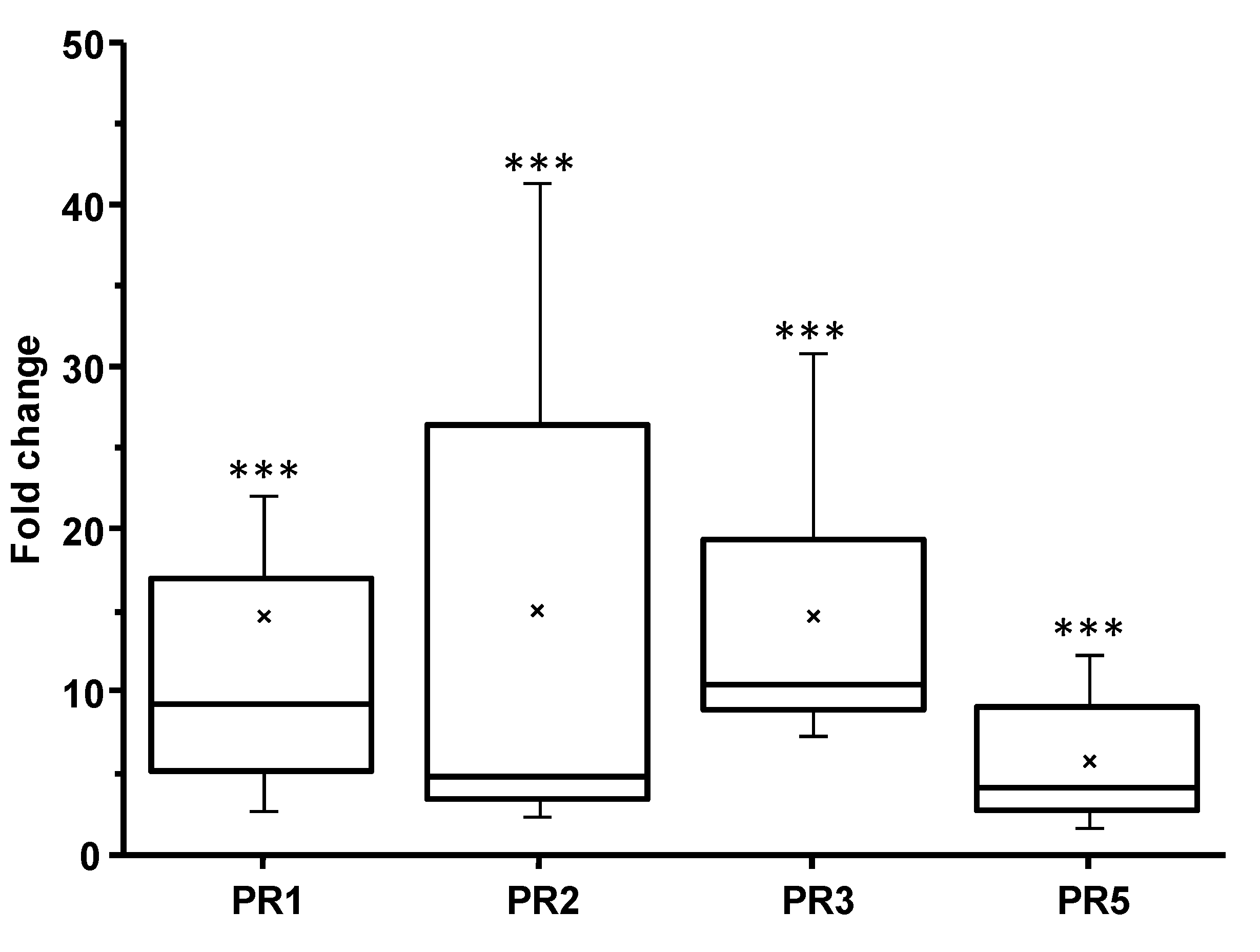
3.6. PDI Activity of Pentagalloyl Glucose
3.7. Acer Leaf Extracts and Gallotannins as PDI
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ishihara, A.; Ando, K.; Yoshioka, A.; Murata, K.; Kokubo, Y.; Morimoto, N.; Ube, N.; Yabuta, Y.; Ueno, M.; Tebayashi, S. Induction of defense responses by extracts of spent mushroom substrates in rice. J. Pestic. Sci. 2019, 44, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Dewen, Q.; Yijie, D.; Yi, Z.; Shupeng, L.; Fachao, S. Plant immunity inducer development and application. Mol. Plant-Microbe Interact. 2017, 30, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Brugger, A.; Lamotte, O.; Vandelle, E.; Bourque, S.; Lecourieux, D.; Poinssot, B.; Wendehenne, D.; Pugin, A. Early signaling events induced by elicitors of plant defenses. Mol. Plant-Microbe Interact. 2006, 19, 711–724. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Henry, G.; Thonart, P.; Ongena, M. PAMPs, MAMPs, DAMPs and others: An update on the diversity of plant immunity elicitors. Biotechnol. Agron. Soc. Environ. 2012, 16, 257–268. [Google Scholar]
- Zhang, W.; Zhao, F.; Jiang, L.; Chen, C.; Wu, L.; Liu, Z. Different pathogen strategies in Arabidopsis: More than pathogen recognition. Cells 2018, 7, 252. [Google Scholar] [CrossRef]
- Torres, M.A.; Jones, J.D.G.; Dangl, J.L. Reactive oxygen species signaling in response to pathogens. Plant Physiol. 2006, 141, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Heller, J.; Tudzynski, P. Reactive oxygen species in phytopathogenic fungi: Signaling. Annu. Rev. Phytopathol. 2011, 49, 369–390. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef] [PubMed]
- Loebenstein, G. Local lesions and induced resistance. Adv. Virus Res. 2009, 75, 73–117. [Google Scholar]
- Kachroo, A.; Vincelli, P.; Kachroo, P. Signaling mechanisms underlying resistance responses: What have we learned, and how is it being applied? Phytopathology 2017, 107, 1452–1461. [Google Scholar] [CrossRef] [PubMed]
- Klessig, D.F.; Choi, H.W.; Dempsey, D.A. Systemic acquired resistance and salicylic acid: Past, present, and future. Mol. Plant-Microbe Interact. 2018, 31, 871–888. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Hébrard, C.; Deville, M.-A.; Cordelier, S.; Dorey, S.; Aziz, A.; Crouzet, J. Deciphering the role of phytoalexins in plant-microorganism interactions and human health. Molecules 2014, 19, 18033–18056. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P. Phytoalexins: Current progress and future prospects. Molecules 2015, 20, 2770–2774. [Google Scholar] [CrossRef]
- Pusztahelyi, T.; Holb, I.J.; Pócsi, I. Secondary metabolites in fungus-plant interactions. Front. Plant Sci. 2015, 6, 573. [Google Scholar] [CrossRef] [PubMed]
- Stringlis, I.A.; de Jonge, R.; Pieterse, C.M.J. The age of coumarins in plant–microbe interactions. Plant Cell Physiol. 2019, 60, 1405–1419. [Google Scholar] [CrossRef]
- Goupil, P.; Benouaret, R.; Charrier, O.; Ter Halle, A.; Richard, C.; Eyheraguibel, B.; Thiery, D.; Ledoigt, G. Grape marc extract acts as elicitor of plant defence responses. Ecotoxicology 2012, 21, 1541–1549. [Google Scholar] [CrossRef]
- Benouaret, R.; Goujon, E.; Goupil, P. Grape marc extract causes early perception events, defence reactions and hypersensitive response in cultured tobacco cells. Plant Physiol. Biochem. 2014, 77, 84–89. [Google Scholar] [CrossRef]
- Benouaret, R.; Goujon, E.; Trivella, A.; Richard, C.; Ledoigt, G.; Joubert, J.-M.; Mery-Bernardon, A.; Goupil, P. Water extracts from winery by-products as tobacco defense inducers. Ecotoxicology 2014, 23, 1574–1581. [Google Scholar] [CrossRef]
- Benouaret, R.; Goupil, P. Grape marc extract-induced defense Reactions and Protection against Phytophthora Parasitica Are Impaired in NahG Tobacco Plants. J. Agric. Food Chem. 2015, 63, 6653–6659. [Google Scholar] [CrossRef]
- Goupil, P.; Benouaret, R.; Richard, C. Ethyl gallate displays elicitor activities in tobacco plants. J. Agric. Food Chem. 2017, 65, 9006–9012. [Google Scholar] [CrossRef] [PubMed]
- Hillis, W.E.; Inoue, T. The formation of polyphenols in Trees-IV. The polyphenols formed in Pinus Radiata after sirex attack. Phytochemistry 1968, 7, 13–22. [Google Scholar] [CrossRef]
- Haddock, E.A.; Gupta, R.K.; Al-Shafi, S.M.K.; Haslam, E.; Magnolato, D. The metabolism of gallic acid and hexahydroxydiphenic acid in plants. Part 1. introduction. Naturally occurring galloyl esters. J. Chem. Soc. Perkin Trans. 1982, 1, 2515–2524. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, H.; Yuan, T.; Seeram, N.P. Red maple (Acer rubrum) aerial parts as a source of bioactive phenolics. Nat. Prod. Commun. 2015, 10, 1409–1412. [Google Scholar] [CrossRef]
- García-Villalba, R.; Espín, J.C.; Tomás-Barberán, F.A.; Rocha-Guzmán, N.E. Comprehensive characterization by LC-DAD-MS/MS of the phenolic composition of seven Quercus leaf teas. J. Food Compos. Anal. 2017, 63, 38–46. [Google Scholar] [CrossRef]
- Armitage, R.; Bayliss, G.S.; Gramshaw, J.W.; Haslam, E.; Haworth, R.D.; Jones, K.; Rogers, H.J.; Searle, T. 360. Gallotannins. Part III. The constitution of Chinese, Turkish, sumach, and tara tannins. J. Chem. Soc. 1961, 1842–1853. [Google Scholar] [CrossRef]
- Britton, G.; Haslam, E. Gallotannins. Part XII. Phenolic constituents of Arctostaphylos Uva-Ursi L. Spreng. J. Chem. Soc. 1965, 1342, 7312–7319. [Google Scholar] [CrossRef]
- Abou-Zaida, M.M.; Nozzolillo, C. 1-O-galloyl-a-L-rhamnose from Acer rubrum. Phytochemistry 1999, 52, 1629–1631. [Google Scholar] [CrossRef]
- Yuan, T.; Wan, C.; Liu, K.; Seeram, N.P. New maplexins F-I and phenolic glycosides from red maple (Acer rubrum) bark. Tetrahedron 2012, 68, 959–964. [Google Scholar] [CrossRef]
- Zhang, L.; Tu, Z.-C.; Xie, X.; Lu, Y.; Wang, Z.-X.; Wang, H.; Sha, X.-M. Antihyperglycemic, antioxidant activities of two Acer palmatum cultivars, and identification of phenolics profile byUPLC-QTOF-MS/MS: New natural sources of functional constituents. Ind. Crops Prod. 2016, 89, 522–532. [Google Scholar] [CrossRef]
- Li, C.; Seeram, N.P. Ultra-fast liquid chromatography coupled with electrospray ionization time-of-flight mass spectrometry for the rapid phenolic profiling of red Maple (Acer rubrum) leaves. J. Sep. Sci. 2018, 41, 2331. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, L.; Ye, Y.-H.; Zhu, M.-F.; Li, J.; Tu, Z.-C.; Yang, S.-H.; Liao, H. Phytochemical profiles and screening of α-glucosidase inhibitors of four Acer species leaves with ultra-filtration combined with UPLC-QTOF-MS/MS. Ind. Crops Prod. 2019, 129, 156–168. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Mamaní, A.; Filippone, M.P.; Grellet, C.; Welin, B.; Castagnaro, A.P.; Ricci, J.C.D. Pathogen-induced accumulation of an ellagitannin elicits plant defense response. Mol. Plant-Microbe Interact. 2012, 25, 1430–1439. [Google Scholar] [CrossRef] [PubMed]
- Emmons, C.L.; Peterson, D.M. Antioxidant activity and phenolic content of oat as affected by cultivar and location. Crop Sci. 2001, 41, 1676–1681. [Google Scholar] [CrossRef]
- Chen, Z.; Huang, S.; Su, Q.; Zhu, X. Pressurized liquid extraction and HPLC analysis for determination of polyphenols in tobacco. Asian J. Chem. 2013, 25, 3889–3892. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing Real-Time PCR Data by the Comparative C(T) Method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Cordelier, S.; De Ruffray, P.; Fritig, B.; Kauffmann, S. Biological and molecular comparison between localized and systemic acquired resistance induced in tobacco by a Phytophthora Megasperma glycoprotein elicitin. Plant Mol. Biol. 2003, 51, 109–118. [Google Scholar] [CrossRef]
- Niemetz, R.; Gross, G.G. Enzymology of gallotannin and ellagitannin biosynthesis. Phytochemistry 2005, 66, 2001–2011. [Google Scholar] [CrossRef]
- Burketova, L.; Trda, L.; Ott, P.G.; Valentova, O. Bio-based resistance inducers for sustainable plant protection against pathogens. Biotechnol. Adv. 2015, 33, 994–1004. [Google Scholar] [CrossRef]
- Sieniawska, E.; Baj, T. Tannins. In Pharmacognosy. Fundamentals, Applications and Strategies; Badal, S., Delgoda, R., Eds.; Academic Press: London, UK, 2017; pp. 199–227. [Google Scholar]
- Goupil, P.; Richard, C.; Ter Halle, A. Available online: https://patents.google.com/WO2015136195A1/en (accessed on 17 September 2015).
| Name | m/z in ES− | Fragments in ES− | Δppm | Formula of Neutral Molecules |
|---|---|---|---|---|
| G5 | 469.0531 (z = 2) | 393.0486 317.0416 241.0360 169.0139 125.0238 | 5.1 (z = 2) | C41H32O26 |
| 939.1143 (z = 1) | 4.7 (z = 1) | |||
| 1,2,3,4,6-penta-O-galloyl-β-D-glucopyranose | 469.0509 (z = 2) | 0.40 (z = 2) | C41H32O26 | |
| 939.1105 (z = 1) | 0.70 (z = 1) | |||
| G6/G6′ | 545.0593 (z = 2) | Those of G5 + 469.0534 | 5.8 (z = 2) | C48H36O30 |
| 1091.1252 (z = 1) | 4.1 (z = 1) | |||
| G7 | 621.0652 (z = 2) | Those of G6 + 545.0604 | 5.8 (z = 2) | C55H40O34 |
| 1243.1362 (z = 1) | 3.8 (z = 1) | |||
| G8 | 697.0717 (z = 2) | Those of G6 | 6.6 (z = 2) | C62H44O38 |
| 1395.1481 (z = 1) | 4.6 (z = 1) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peghaire, E.; Hamdache, S.; Galien, A.; Sleiman, M.; ter Halle, A.; El Alaoui, H.; Kocer, A.; Richard, C.; Goupil, P. Inducing Plant Defense Reactions in Tobacco Plants with Phenolic-Rich Extracts from Red Maple Leaves: A Characterization of Main Active Ingredients. Forests 2020, 11, 705. https://doi.org/10.3390/f11060705
Peghaire E, Hamdache S, Galien A, Sleiman M, ter Halle A, El Alaoui H, Kocer A, Richard C, Goupil P. Inducing Plant Defense Reactions in Tobacco Plants with Phenolic-Rich Extracts from Red Maple Leaves: A Characterization of Main Active Ingredients. Forests. 2020; 11(6):705. https://doi.org/10.3390/f11060705
Chicago/Turabian StylePeghaire, Elodie, Samar Hamdache, Antonin Galien, Mohamad Sleiman, Alexandra ter Halle, Hicham El Alaoui, Ayhan Kocer, Claire Richard, and Pascale Goupil. 2020. "Inducing Plant Defense Reactions in Tobacco Plants with Phenolic-Rich Extracts from Red Maple Leaves: A Characterization of Main Active Ingredients" Forests 11, no. 6: 705. https://doi.org/10.3390/f11060705
APA StylePeghaire, E., Hamdache, S., Galien, A., Sleiman, M., ter Halle, A., El Alaoui, H., Kocer, A., Richard, C., & Goupil, P. (2020). Inducing Plant Defense Reactions in Tobacco Plants with Phenolic-Rich Extracts from Red Maple Leaves: A Characterization of Main Active Ingredients. Forests, 11(6), 705. https://doi.org/10.3390/f11060705