Localized and Moderate Phosphorus Application Improves Plant Growth and Phosphorus Accumulation in Rosa multiflora Thunb. ex Murr. via Efficient Root System Development
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Design
2.2. Sample Work
2.3. Statistical Analysis
3. Results
3.1. Interactive Effects of Treatment Days, P Fertilizer Supply Methods and Levels
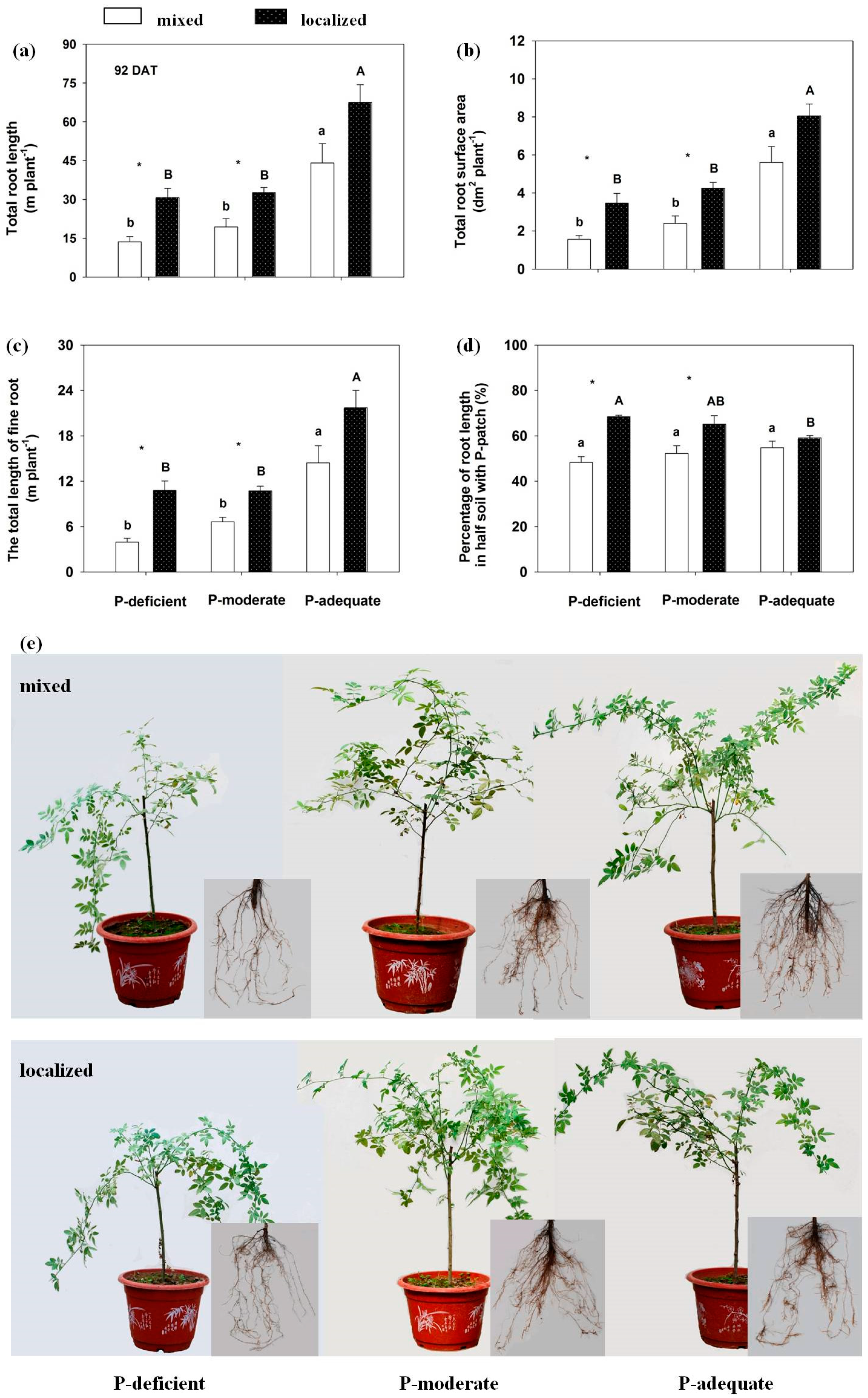
3.2. Plant Growth
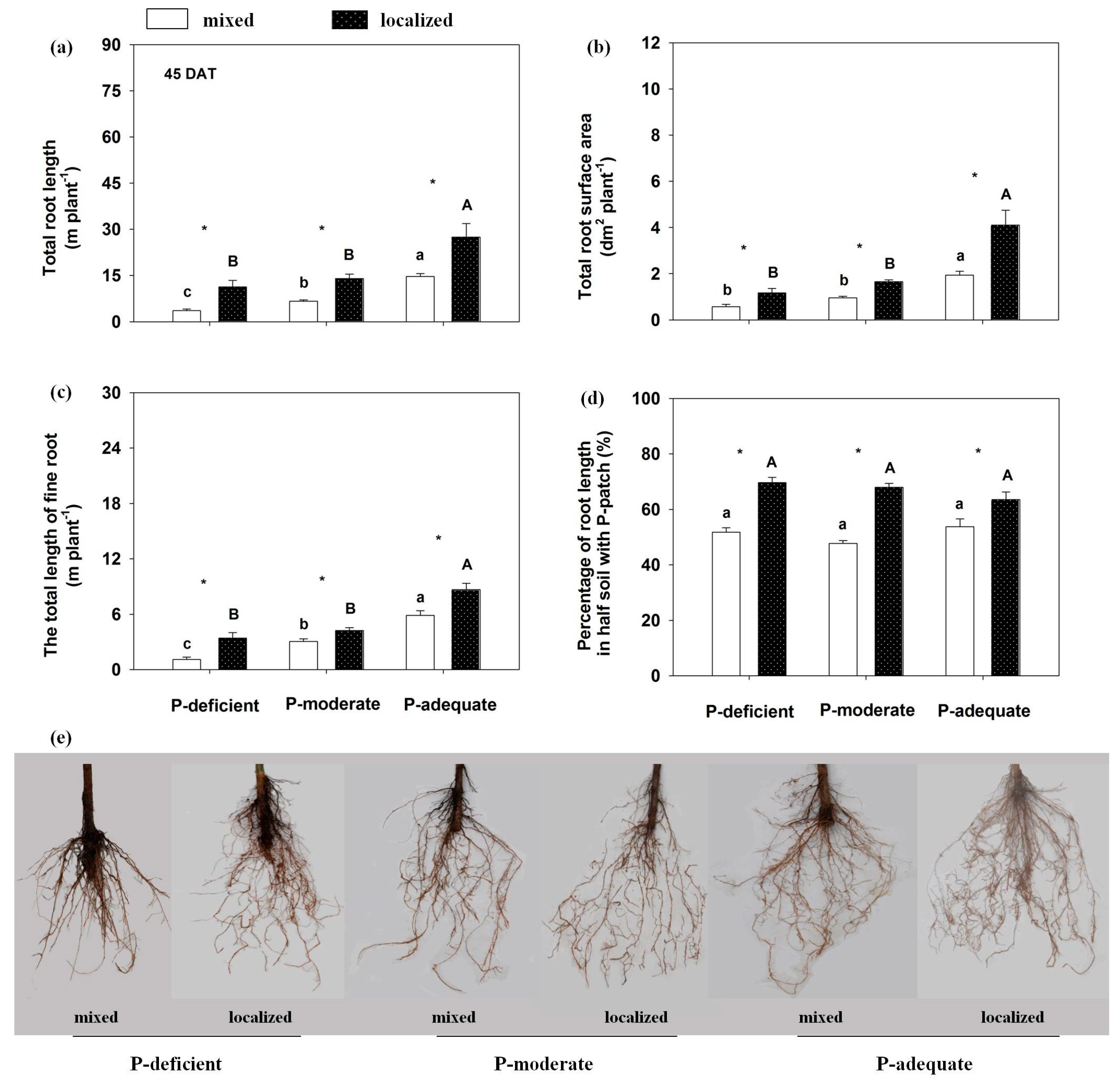
3.3. Effects on Root Length, Root Surface Area, Fine Root and Root Distribution
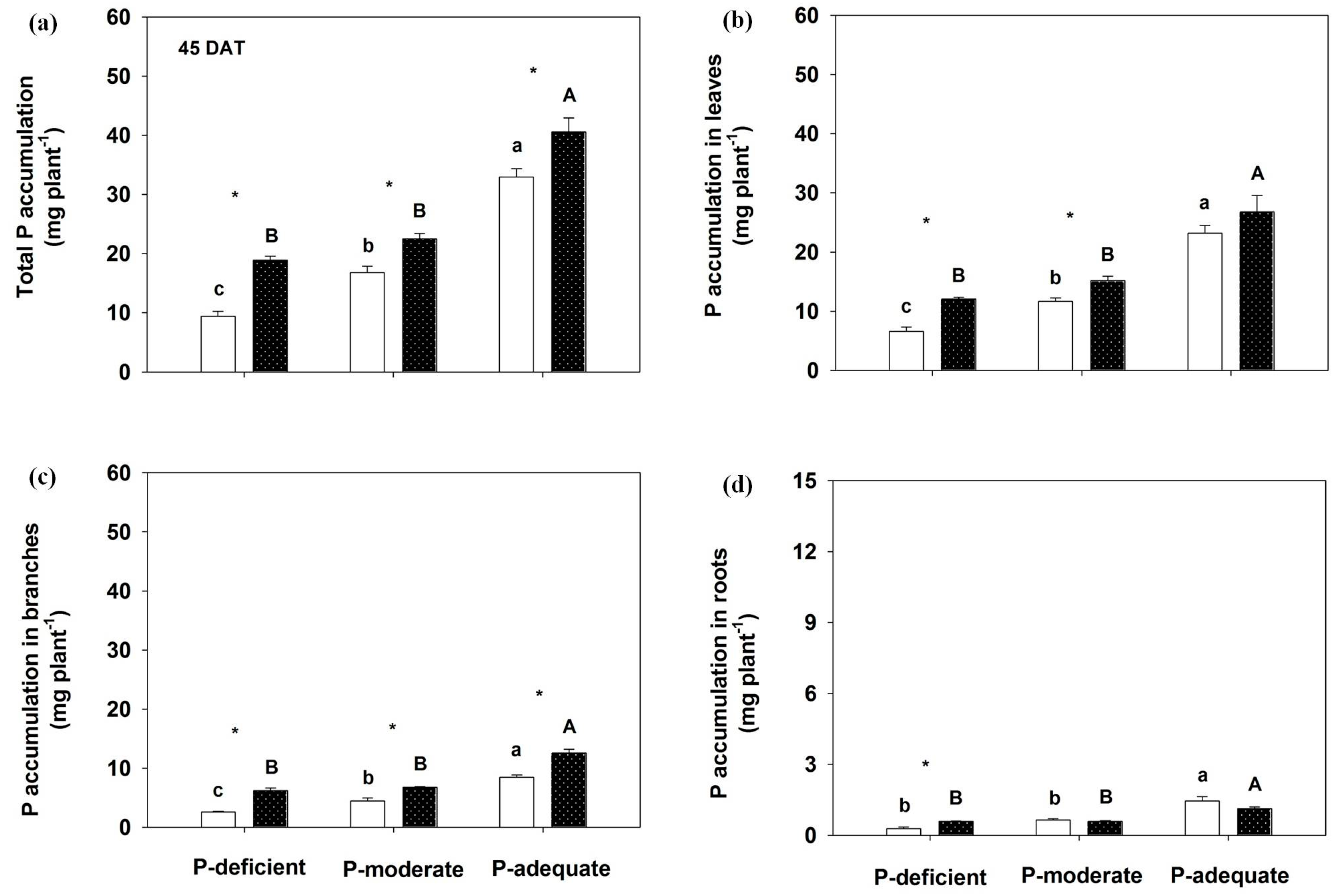
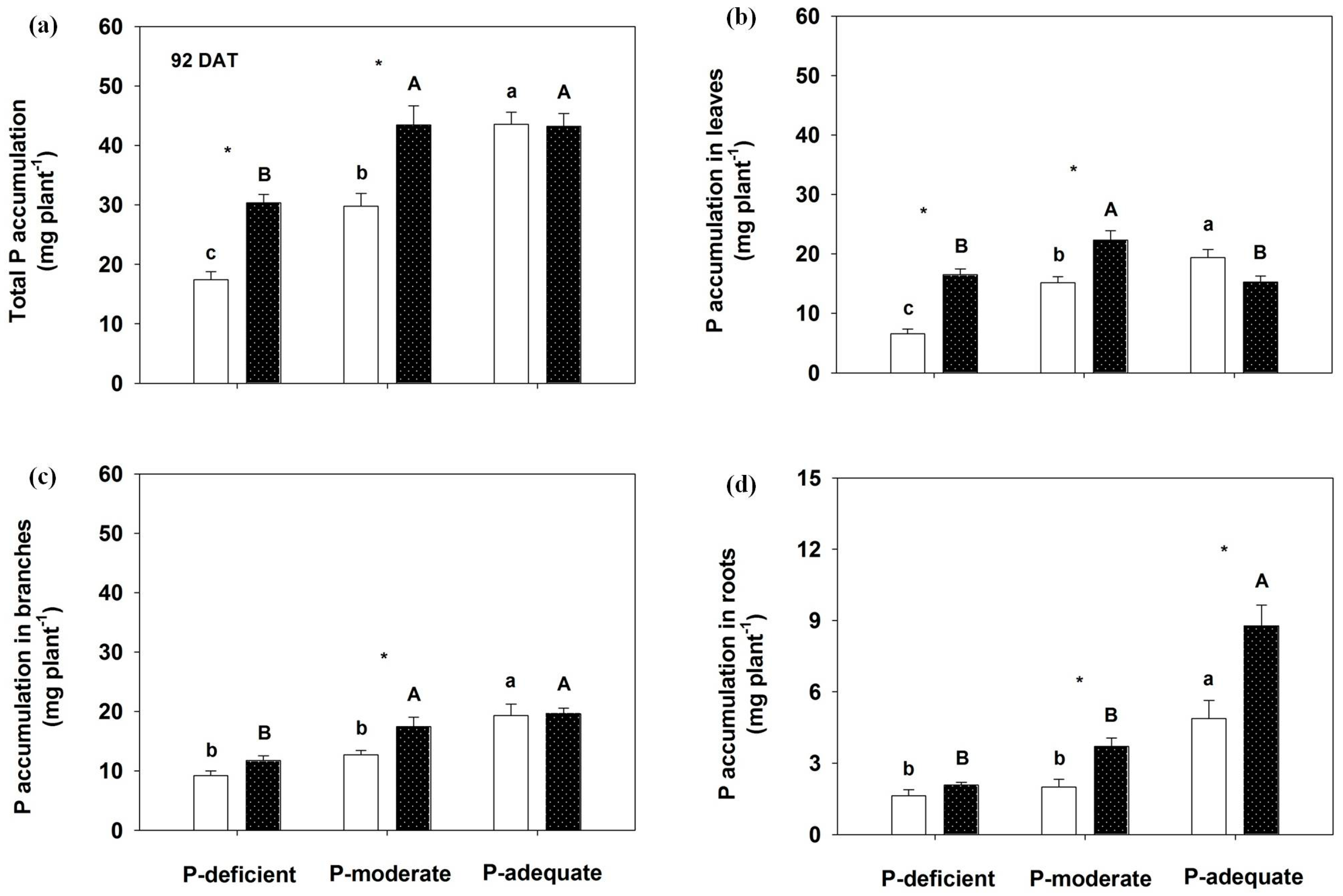
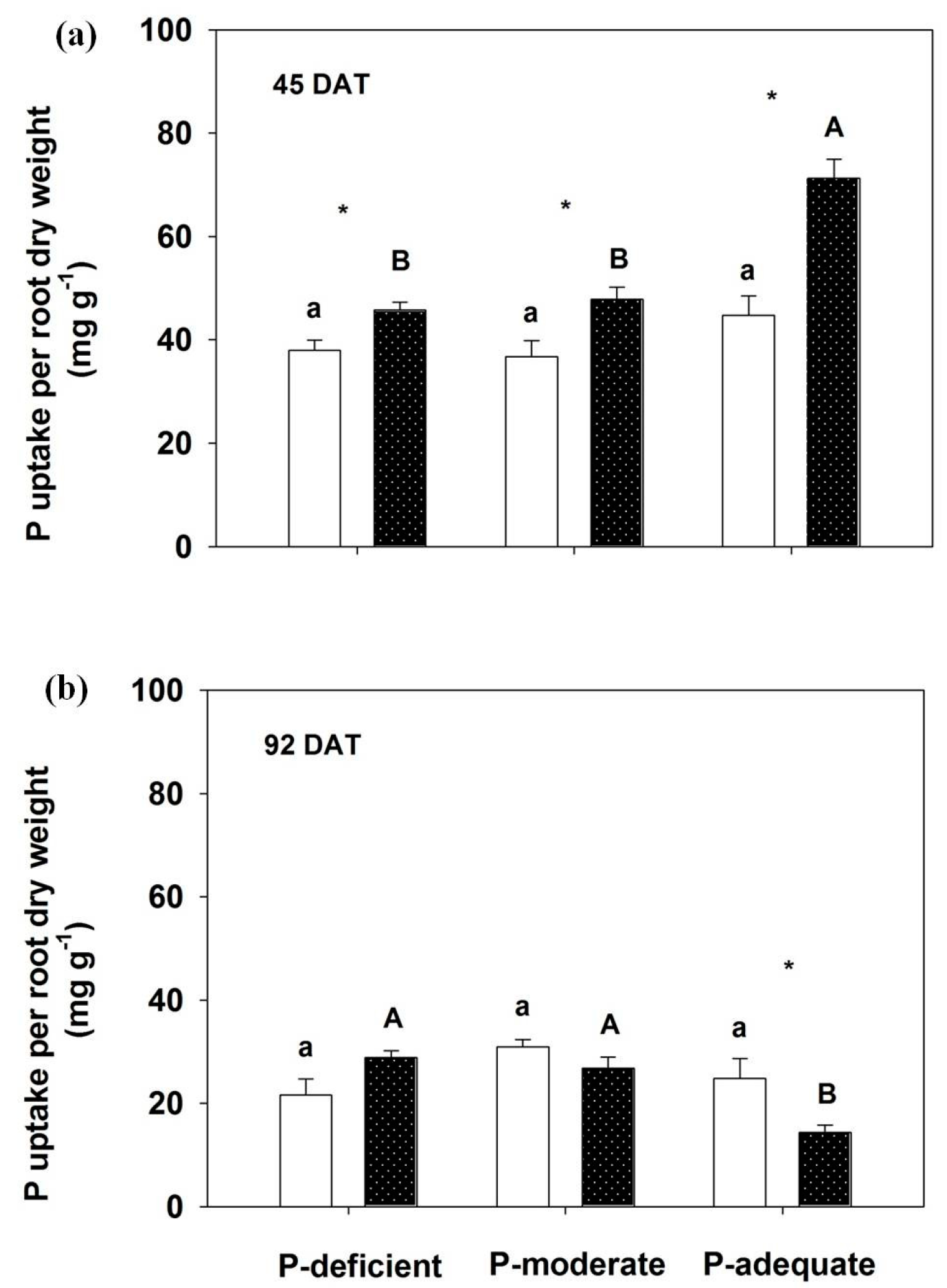
3.4. Effects on P Accumulation in Annual Leaves, Branches, Roots and the Whole Plant and P Uptake Rate
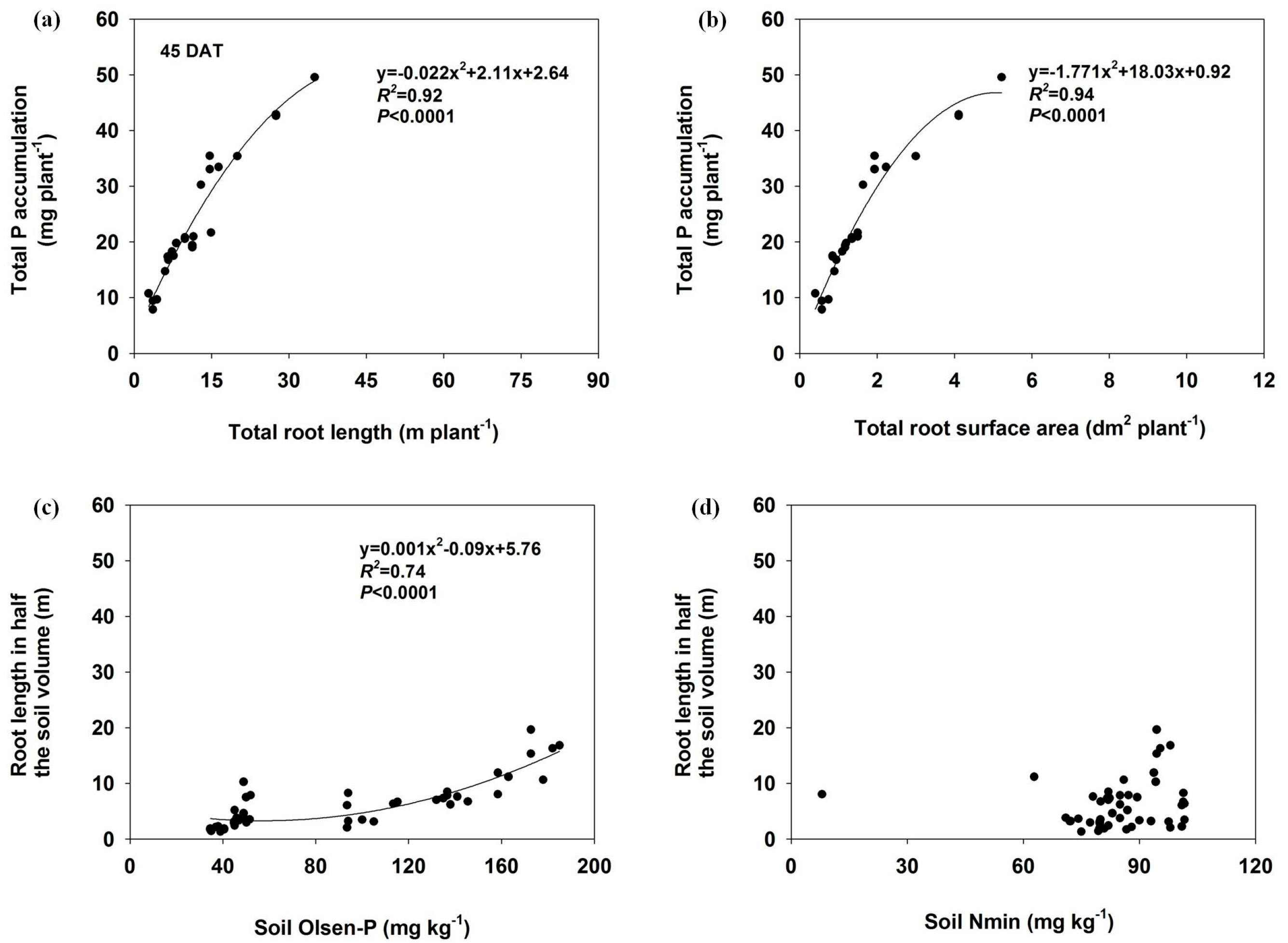
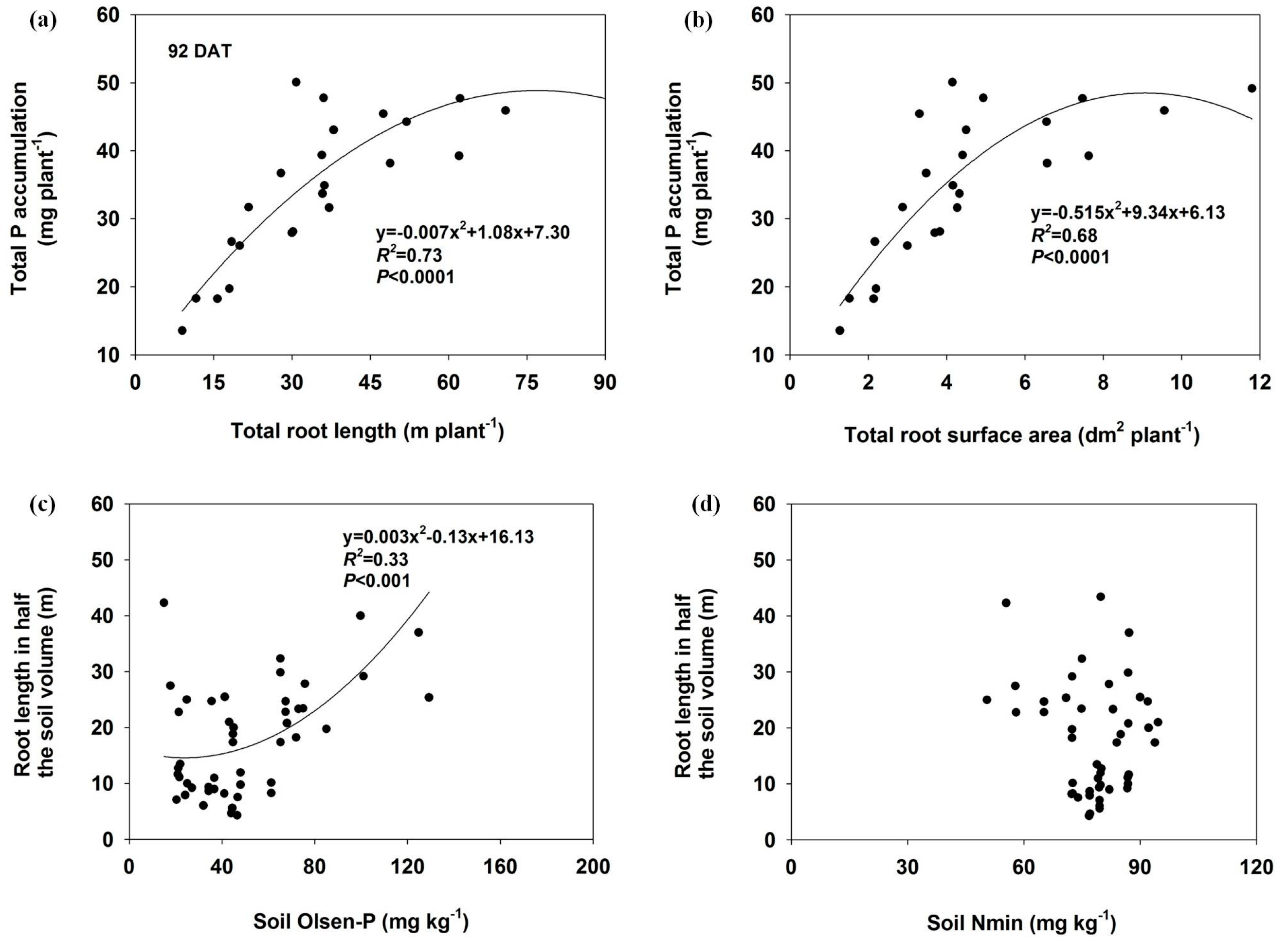
3.5. Relationship Between Soil Nutrients (Nmin and Olsen-P), Root Length and P Accumulation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Lambers, H.; Plaxton, W.C. Phosphorus: Back to the roots. Annu. Plant Rev. Online 2018, 48, 3–22. [Google Scholar]
- Michigami, T.; Kawai, M.; Yamazaki, M.; Ozono, K. Phosphate as a signaling molecule and its sensing mechanism. Physiol. Rev. 2018, 98, 2317–2348. [Google Scholar] [CrossRef] [PubMed]
- Johnston, A.E.; Poulton, P.R.; Fixen, P.E.; Curtin, D. Phosphorus: Its efficient use in agriculture. Adv. Agron. 2014, 123, 177–228. [Google Scholar]
- Herrera-Estrella, L.; López-Arredondo, D. Phosphorus: The underrated element for feeding the world. Trends Plant Sci. 2016, 21, 461–463. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus limitation: Mechanisms, implications, and nitrogen–phosphorus interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, F. Phosphorus dynamics: From soil to plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Yuan, J.; Zhang, T.; Hao, F.; Jose, S.; Zhang, S. Soil degradation and the decline of available nitrogen and phosphorus in soils of the main forest types in the Qinling Mountains of China. Forests 2017, 8, 460. [Google Scholar] [CrossRef] [Green Version]
- Heinonen, T.; Pukkala, T.; Asikainen, A.; Peltola, H. Scenario analyses on the effects of fertilization, improved regeneration material, and ditch network maintenance on timber production of Finnish forests. Eur. J. Forest Res. 2018, 137, 93–107. [Google Scholar] [CrossRef] [Green Version]
- de Melo, E.A.S.C.; de Gonçalves, J.L.M.; Rocha, J.H.T.; Hakamada, R.E.; Bazani, J.H.; Wenzel, A.V.A.; Arthur, J.C., Jr.; Borges, J.S.; Malheiros, R.; de Lemos, C.C.Z.; et al. Responses of clonal eucalypt plantations to N, P and K fertilizer application in different edaphoclimatic conditions. Forests 2015, 7, 2. [Google Scholar] [CrossRef] [Green Version]
- Wu, P.F.; Ma, X.Q.; Tigabu, M.; Huang, Y.; Zhou, L.L.; Cai, L.; Oden, P.C. Comparative growth, dry matter accumulation and photosynthetic rate of seven species of Eucalypt in response to phosphorus supply. J. Forestry Res. 2014, 25, 377–383. [Google Scholar] [CrossRef]
- Grant, C.A.; Flaten, D.N.; Tomasiewicz, D.J.; Sheppard, S.C. The importance of early season phosphorus nutrition. Can. J. Plant Sci. 2001, 81, 211–224. [Google Scholar] [CrossRef]
- Plassard, C.; Dell, B. Phosphorus nutrition of mycorrhizal trees. Tree Physiol. 2010, 30, 1129–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, M.G.; Gama-Rodrigues, A.C.; Gonçalves, J.L.D.M.; Gama-Rodrigues, E.F.; Sales, M.V.D.S.; Aleixo, S. Labile and non-labile fractions of phosphorus and its transformations in soil under eucalyptus plantations, Brazil. Forests 2016, 7, 15. [Google Scholar] [CrossRef] [Green Version]
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus uptake by plants: From soil to cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, J.P. Root phenes for enhanced soil exploration and phosphorus acquisition: Tools for future crops. Plant Physiol. 2011, 156, 1041–1049. [Google Scholar] [CrossRef] [Green Version]
- Ma, Q.; Zhang, F.; Rengel, Z.; Shen, J. Localized application of NH4+-N plus P at the seedling and later growth stages enhances nutrient uptake and maize yield by inducing lateral root proliferation. Plant Soil 2013, 372, 65–80. [Google Scholar] [CrossRef]
- Kaiser, D.; Mallarino, A.P.; Bermudez, M. Corn grain yield, early growth, and early nutrient uptake as affected by broadcast and in-furrow starter fertilization. Agron. J. 2005, 97, 620–626. [Google Scholar] [CrossRef]
- Roth, G.W.; Beegle, D.B.; Heinbaugh, S.M.; Antle, M.E. Starter fertilizers for corn on soils testing high in phosphorus in the northeastern USA. Agron. J. 2006, 98, 1121–1127. [Google Scholar] [CrossRef]
- Hodge, A. The plastic plant: Root responses to heterogeneous supplies of nutrients. New Phytol. 2004, 162, 9–24. [Google Scholar] [CrossRef]
- Zhou, C.; Jiang, W.; Li, Y.; Hou, X.; Liu, A.; Cai, L. Morphological plasticity and phosphorus uptake mechanisms of hybrid Eucalyptus roots under spatially heterogeneous phosphorus stress. J. Forestry Res. 2017, 28, 713–724. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, C.; Tang, X.; Li, H.; Zhang, F.; Rengel, Z.; Shen, J. Increased soil phosphorus availability induced by faba bean root exudation stimulates root growth and phosphorus uptake in neighbouring maize. New Phytol. 2016, 209, 823–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, R.D.; Dunsford, K.; McLaughlin, M.J.; McBeath, T.; Mason, S.; Dunbabin, V.M. Phosphorus and nitrogen fertiliser use efficiency of wheat seedlings grown in soils from contrasting tillage systems. Plant Soil 2015, 396, 297–309. [Google Scholar] [CrossRef]
- Li, H.; Mollier, A.; Ziadi, N.; Shi, Y.; Parent, L.; Morel, C. The long-term effects of tillage practice and phosphorus fertilization on the distribution and morphology of corn root. Plant Soil 2017, 412, 97–114. [Google Scholar] [CrossRef]
- Weligama, C.; Tang, C.; Sale, P.W.G.; Conyers, M.; Liu, D. Localised nitrate and phosphate application enhances root proliferation by wheat and maximises rhizosphere alkalisation in acid subsoil. Plant Soil 2008, 312, 101–115. [Google Scholar] [CrossRef]
- Nkebiwe, P.M.; Weinmann, M.; Bar-Tal, A.; Müller, T. Fertilizer placement to improve crop nutrient acquisition and yield: A review and meta-analysis. Field Crop. Res. 2016, 196, 389–401. [Google Scholar] [CrossRef]
- Gao, W.; Blaser, S.R.; Schlüter, S.; Shen, J.; Vetterlein, D. Effect of localised phosphorus application on root growth and soil nutrient dynamics in situ–comparison of maize (Zea mays) and faba bean (Vicia faba) at the seedling stage. Plant Soil 2019, 441, 469–483. [Google Scholar] [CrossRef]
- Mallarino, A.P.; Bergmann, N.; Kaiser, D.E. Corn responses to in-furrow phosphorus and potassium starter fertilizer applications. Agron. J. 2011, 103, 685–694. [Google Scholar] [CrossRef]
- Jing, J.; Rui, Y.; Zhang, F.; Rengel, Z.; Shen, J. Localized application of phosphorus and ammonium improves growth of maize seedlings by stimulating root proliferation and rhizosphere acidification. Field Crop. Res. 2010, 119, 355–364. [Google Scholar] [CrossRef]
- Su, W.; Liu, B.; Liu, X.; Li, X.; Ren, T.; Cong, R.; Lu, J. Effect of depth of fertilizer banded-placement on growth, nutrient uptake and yield of oilseed rape (Brassica napus L.). Eur. J. Agron. 2015, 62, 38–45. [Google Scholar] [CrossRef]
- Hansel, F.; Diaz, D.; Rosa, A.; Moorberg, C. Phosphorus fertilizer placement and rate affect soybean root growth and nutrient uptake in soil with high fertility. Agron. Sci. Biotechnol. 2019, 5, 62–73. [Google Scholar] [CrossRef] [Green Version]
- Feng, H.; Wang, M.L.; Cong, R.C.; Dai, S.L. Colchicine-and trifluralin-mediated polyploidization of Rosa multiflora Thunb. var. inermis and Rosa roxburghii f. normalis. J. Hortic. Sci. Biotech. 2017, 92, 279–287. [Google Scholar] [CrossRef]
- Otiende, M.A.; Nyabundi, J.O.; Ngamau, K.; Opala, P. Effects of cutting position of rose rootstock cultivars on rooting and its relationship with mineral nutrient content and endogenous carbohydlevels. Sci. Hortic. 2017, 225, 204–212. [Google Scholar] [CrossRef]
- Westerman, R.L. Soil Testing and Plant Analysis, 3rd ed.; American Society of Agronomy and Soil Science of America: Madison, WI, USA, 1991. [Google Scholar]
- Johnson, C.M.; Ulrich, A. Analytical Methods for Use in Plant Analysis. Bull. Calif. Agric. Exp. Stn. 1959, 766, 78. [Google Scholar]
- Sattari, S.Z.; Bouwman, A.F.; Rodriguez, R.M.; Beusen, A.H.W.; Van Ittersum, M.K. Negative global phosphorus budgets challenge sustainable intensification of grasslands. Nat. Commun. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Meister, R.; Rajani, M.S.; Ruzicka, D.; Schachtman, D.P. Challenges of modifying root traits in crops for agriculture. Trends Plant Sci. 2014, 19, 779–788. [Google Scholar] [CrossRef]
- Yan, X.L.; Wang, C.; Ma, X.; Wu, P. Root morphology and seedling growth of three tree species in southern China in response to homogeneous and heterogeneous phosphorus supplies. Trees 2019, 33, 1283–1297. [Google Scholar] [CrossRef]
- Pan, F.; Liang, Y.; Wang, K.; Zhang, W. Responses of fine root functional traits to soil nutrient limitations in a karst ecosystem of Southwest China. Forests 2018, 9, 743. [Google Scholar] [CrossRef] [Green Version]
- Yaseen, M.; Ahmad, T.; Sablok, G.; Standardi, A.; Hafiz, I.A. Role of carbon sources for in vitro plant growth and development. Mol. Biol. Rep. 2013, 40, 2837–2849. [Google Scholar] [CrossRef]
- Hermans, C.; Hammond, J.P.; White, P.J.; Verbruggen, N. How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci. 2006, 11, 610–617. [Google Scholar] [CrossRef]
- Rose, T.J.; Rengel, Z.; Ma, Q.; Bowden, J.W. Phosphorus accumulation by field-grown canola crops and the potential for deep phosphorus placement in a Mediterranean-type climate. Crop. Pasture Sci. 2009, 60, 987–994. [Google Scholar] [CrossRef]
- Ågren, G.I.; Wetterstedt, J.M.; Billberger, M.F. Nutrient limitation on terrestrial plant growth–modeling the interaction between nitrogen and phosphorus. New Phytol. 2012, 194, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Li, H.; Shen, J.; Rengel, Z. Maize responds to low shoot P concentration by altering root morphology rather than increasing root exudation. Plant Soil 2017, 416, 377–389. [Google Scholar] [CrossRef]
- Scott, D.A.; Bliss, C.M. Phosphorus fertilizer rate, soil P availability, and long-term growth response in a loblolly pine plantation on a weathered ultisol. Forests 2012, 3, 1071–1085. [Google Scholar] [CrossRef]
- Song, X.; Wan, F.; Chang, X.; Zhang, J.; Sun, M.; Liu, Y. Effects of nutrient deficiency on root morphology and nutrient allocation in Pistacia chinensis bunge Seedlings. Forests 2019, 10, 1035. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.; Chu, C. Nitrogen–phosphorus interplay: Old story with molecular tale. New Phytol. 2020, 225, 1455–1460. [Google Scholar] [CrossRef] [Green Version]
- Robinson, D. The responses of plants to non-uniform supplies of nutrients. New Phytol. 1994, 127, 635–674. [Google Scholar] [CrossRef]
- Graciano, C.; Tambussi, E.A.; Castán, E.; Guiamet, J.J. Dry mass partitioning and nitrogen uptake by Eucalyptus grandis plants in response to localized or mixed application of phosphorus. Plant Soil 2009, 319, 175–184. [Google Scholar] [CrossRef]
- Mazlouzi, M.E.; Morel, C.; Robert, T.; Yan, B.; Mollier, A. Phosphorus uptake and partitioning in two durum wheat cultivars with contrasting biomass allocation as affected by different P supply during grain filling. Plant Soil 2020, 449, 179–192. [Google Scholar] [CrossRef]
| Source of Variation | Shoot DW | Root DW | Root/Shoot Ratio | Total Root Length | Total Root Surface Area | Total P Accumulation |
|---|---|---|---|---|---|---|
| Days | 6.03 * | 133.163 | 106.97 | 118.68 | 91.98 | 129.52 |
| Methods | 51.13 | 17.09 *** | 1.75 | 29.16 | 31.37 | 70.56 |
| Levels | 80.09 | 37.37 | 10.45 *** | 48.23 | 44.02 | 159.94 |
| Days × Methods | 0.48 | 15.79 *** | 6.75 * | 3.95 | 4.60 * | 0.69 |
| Days × Levels | 19.59 | 15.34 *** | 13.79 *** | 9.86 *** | 4.99 * | 11.77 *** |
| Methods × Levels | 0.56 | 1.61 | 2.27 | 2.33 | 4.26 * | 3.51 * |
| Days × Methods × Levels | 6.74 ** | 5.46 ** | 5.61 ** | 0.19 | 0.19 | 6.72 ** |
| Sampling Time | Parameters | Mixed Application | Localized Application | ||||||
|---|---|---|---|---|---|---|---|---|---|
| P-Deficient | P-Moderate | P-Adequate | Mean | P-Deficient | P-Moderate | P-Adequate | Mean | ||
| 45 DAT | Shoot DW (g plant−1) | 4.35c | 7.14b | 12.67a | 8.05B | 7.99b | 9.38b | 17.18a | 11.51A |
| Root DW (g plant−1) | 0.20c | 0.47b | 0.75a | 0.47A | 0.42b | 0.43b | 0.61a | 0.49A | |
| Root/shoot ratio | 0.05a | 0.06a | 0.06a | 0.06A | 0.05a | 0.05a | 0.04a | 0.05A | |
| 92 DAT | Shoot DW (g plant−1) | 6.69b | 9.17b | 12.24a | 9.37B | 9.77b | 13.86a | 12.55ab | 12.06A |
| Root DW (g plant−1) | 0.85b | 0.92b | 1.83a | 1.20B | 1.06b | 1.65ab | 3.23a | 1.98A | |
| Root/shoot ratio | 0.13a | 0.10a | 0.16a | 0.12B | 0.11b | 0.12b | 0.26a | 0.15A | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Q.; Chen, L.; Du, M.; Zhang, Y.; Zhang, Y. Localized and Moderate Phosphorus Application Improves Plant Growth and Phosphorus Accumulation in Rosa multiflora Thunb. ex Murr. via Efficient Root System Development. Forests 2020, 11, 570. https://doi.org/10.3390/f11050570
Ma Q, Chen L, Du M, Zhang Y, Zhang Y. Localized and Moderate Phosphorus Application Improves Plant Growth and Phosphorus Accumulation in Rosa multiflora Thunb. ex Murr. via Efficient Root System Development. Forests. 2020; 11(5):570. https://doi.org/10.3390/f11050570
Chicago/Turabian StyleMa, Qinghua, Lin Chen, Manyi Du, Yongan Zhang, and Yaoxiang Zhang. 2020. "Localized and Moderate Phosphorus Application Improves Plant Growth and Phosphorus Accumulation in Rosa multiflora Thunb. ex Murr. via Efficient Root System Development" Forests 11, no. 5: 570. https://doi.org/10.3390/f11050570
APA StyleMa, Q., Chen, L., Du, M., Zhang, Y., & Zhang, Y. (2020). Localized and Moderate Phosphorus Application Improves Plant Growth and Phosphorus Accumulation in Rosa multiflora Thunb. ex Murr. via Efficient Root System Development. Forests, 11(5), 570. https://doi.org/10.3390/f11050570





