Habitat Selection by the European Nightjar Caprimulgus europaeus in North-Eastern Poland: Implications for Forest Management
Abstract
1. Introduction
2. Materials and Methods
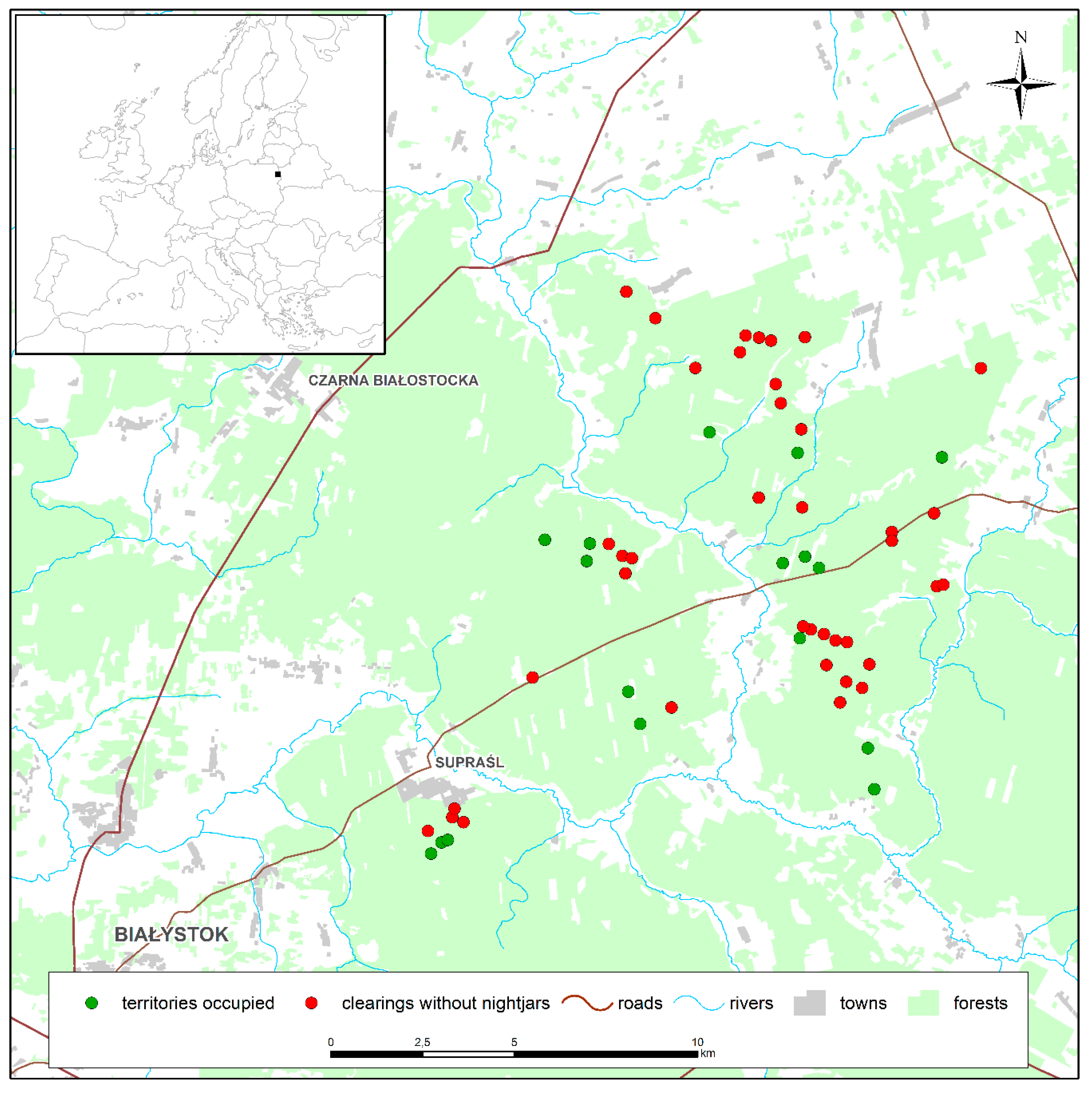
2.1. Study Area
2.2. Methods
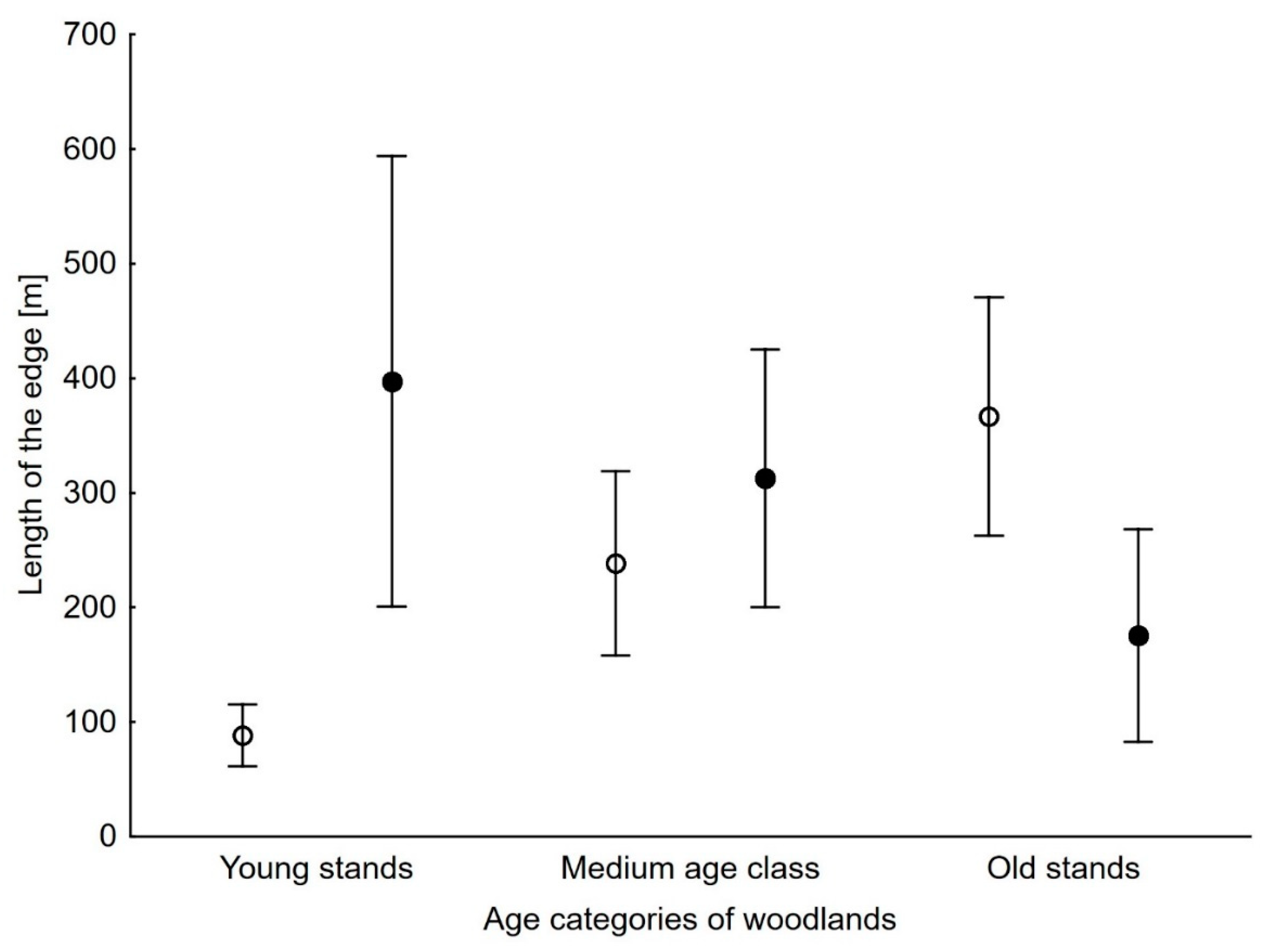
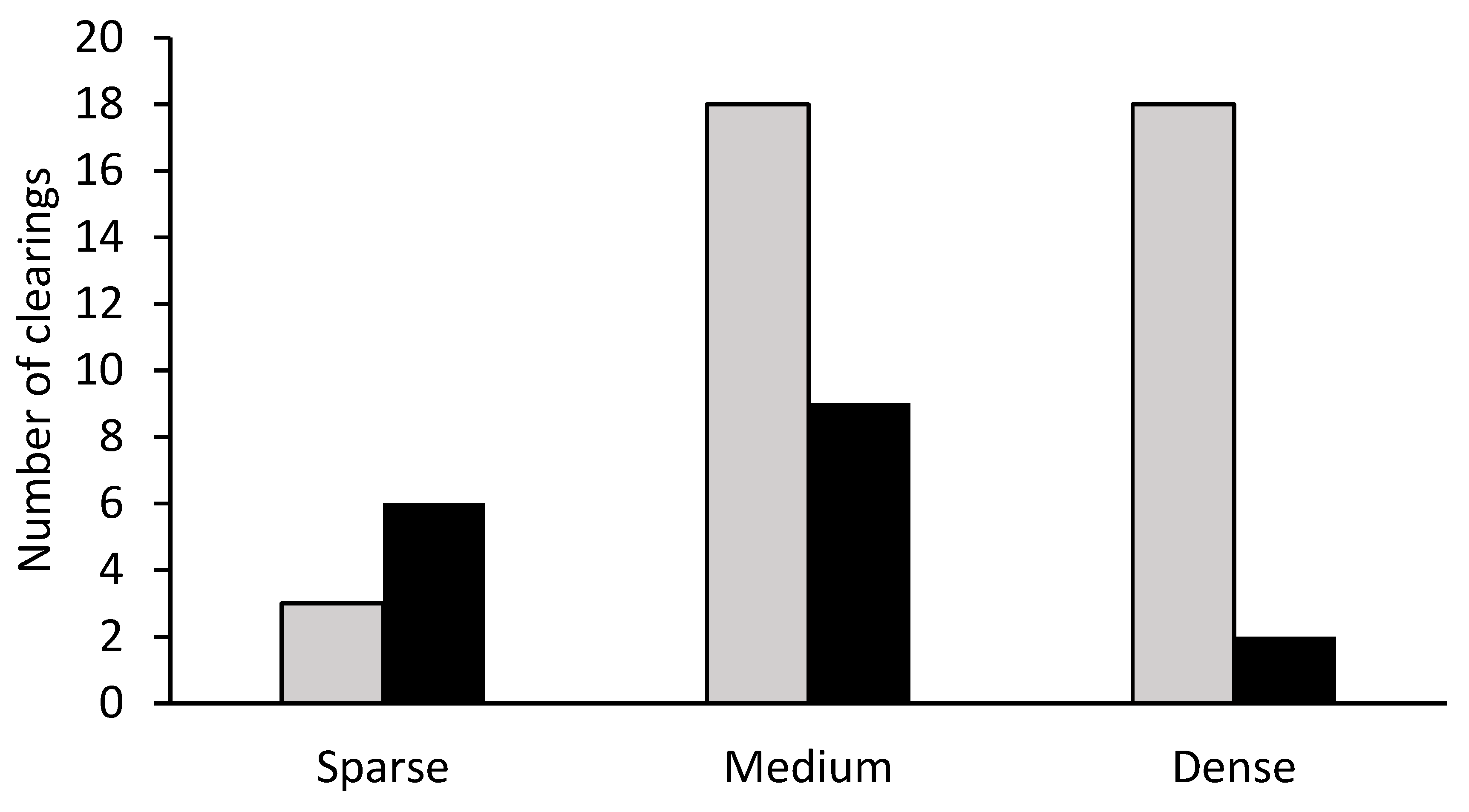
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- von Gadow, K.; Pukkala, T.; Tomé, M. Managing Forest Ecosystem. In Sustainable Forest Management; Springer: Dordrecht, The Netherlands, 2000; p. 355. [Google Scholar]
- Seymour, R.S.; White, A.S.; DeMaynadier, P.G. Natural disturbance regimes in northeastern North America—Evaluating silvicultural systems using natural scales and frequencies. For. Ecol. Manag. 2002, 155, 357–367. [Google Scholar] [CrossRef]
- Sing, L.; Metzger, M.J.; Paterson, J.S.; Ray, D. A review of the effects of forest management intensity on ecosystem services for northern European temperate forests with a focus on the UK. Forestry 2017, 91, 151–164. [Google Scholar] [CrossRef]
- Bartniczak, B.; Raszkowski, A. Sustainable forest management in Poland. Manag. Environ. Qual. 2018, 29, 666–677. [Google Scholar] [CrossRef]
- King, D.I.; De Graaf, R.M. Bird species diversity and nestingsuccess in mature, clearcut and shelterwood forest in northern NewHampshire, USA. For. Ecol. Manag. 2000, 129, 227–235. [Google Scholar] [CrossRef]
- Vanderwel, M.C.; Malcolm, J.R.; Mills, S.C. Ameta-analysis of bird responses to uniform partial harvestingacross North America. Conserv. Biol. 2007, 21, 1230–1240. [Google Scholar] [CrossRef] [PubMed]
- Stachura-Skierczyńska, K.; Tumiel, T.; Skierczyński, M. Habitat prediction model for three-toed woodpecker and its implications for the conservation of biologically valuable forests. For. Ecol. Manag. 2009, 258, 697–703. [Google Scholar] [CrossRef]
- Carrillo-Rubio, E.; Kery, M.; Morreale, S.J.; Sullivan, P.J.; Gardner, B.; Cooch, E.G.; Lassoie, J.P. Use of multispecies occupancy models to evaluate the response of bird communities to forest degradation associated with logging. Conserv. Biol. 2014, 28, 1034–1044. [Google Scholar] [CrossRef]
- Czeszczewik, D.; Zub, K.; Stanski, T.; Sahel, M.; Kapusta, A.; Walankiewicz, W. Effect of forest management on bird assemblages in the Białowieża Forest, Poland. iForest 2014, 8, 377–385. [Google Scholar] [CrossRef]
- Camprodon, J.; Brotons, L. Effects of undergrowth clearing on the bird communities of the Northwestern Mediterranean Coppice Holm oak forests. For. Ecol. Manag. 2006, 221, 72–82. [Google Scholar] [CrossRef]
- Morris, D.L.; Porneluzi, P.A.; Haslerig, J.; Clawson, R.L.; Faaborg, J. Results of 20 years of experimental forest management on breeding birds in Ozark forests of Missouri, USA. For. Ecol. Manag. 2013, 310, 747–760. [Google Scholar] [CrossRef]
- Nyland, R.D. Silviculture Concepts and Applications; McGraw-Hill Co. Inc.: New York, NY, USA, 1996; p. 633. [Google Scholar]
- Spiecker, H. Silvicultural management in maintaining biodiversity and resistance of forests in Europe-temperate zone. J. Environ. Manag. 2003, 67, 55–65. [Google Scholar] [CrossRef]
- Zimmerling, J.R.; Francis, C.M.; Roy, C.; Calvert, A.M. How well does forestry in Ontario’s boreal forest emulate natural disturbances from the perspective of birds? Avian Conserv. Ecol. 2017, 12, 10. [Google Scholar] [CrossRef]
- Ameztegui, A.; Gil-Tena, A.; Faus, J.; Piqué, M.; Brotons, L.; Camprodon, J. Bird community response in mountain pine forests of the Pyrenees managed under a shelterwood system. For. Ecol. Manag. 2018, 407, 95–105. [Google Scholar] [CrossRef]
- Cramp, S.; Simmons, K.E.L. The Birds of the Western Palearctic; Oxford University Press: Oxford, UK, 1983; Volume 3, p. 913. [Google Scholar]
- Hagemeijer, W.J.; Blair, M.J. The EBCC Atlas of European Breeding Birds: Their Distribution and Abundance; Poyser: London, UK, 1997; pp. 1–903. [Google Scholar]
- Berry, R. Nightjar habitats and breeding in East Anglia. Br. Birds 1979, 72, 207–218. [Google Scholar]
- Cadbury, C.J. Nightjar census methods. Bird Study 1981, 28, 1–4. [Google Scholar] [CrossRef]
- Ravenscroft, N.O.M. The status and habitat of the Nightjar Caprimulgus europaeus in coastal Suffolk. Bird Study 1989, 36, 161–169. [Google Scholar] [CrossRef]
- Scott, G.W.; Jardine, D.C.; Hills, G.; Sweeney, B. Changes in Nightjar Caprimulgus europaeus populations in upland forests in Yorkshire. Bird Study 1998, 45, 219–225. [Google Scholar] [CrossRef][Green Version]
- Verstraeten, G.; Baeten, L.; Verheyen, K. Habitat preferences of European Nightjars Caprimulgus europaeus in forests on sandy soils. Bird Study 2011, 58, 120–129. [Google Scholar] [CrossRef]
- Langston, R.H.W.; Wotton, S.R.; Conway, G.J.; Wright, L.J.; Mallord, J.W.; Currie, F.A.; Drewitt, A.L.; Grice, P.V.; Hoccom, D.G.; Symes, N. Nightjar Caprimulgus europaeus and Woodlark Lullula arborea–recovering species in Britain? Ibis 2007, 149, 250–260. [Google Scholar] [CrossRef]
- Birdlife International. Species Factsheet: Caprimulgus Europaeus. Available online: http://www.birdlife.org (accessed on 15 December 2019).
- Bult, H. Nightjars Caprimulgus europaeus in the SW Netherlands. Limosa 2002, 75, 91–102, (In Dutch, with English Summary). [Google Scholar]
- Conway, G.; Wotton, S.; Henderson, I.; Langston, R.; Drewitt, A.; Currie, F. Status and distribution of European Nightjars Caprimulgus europaeus in the UK in 2004. Bird Study 2007, 54, 98–111. [Google Scholar] [CrossRef]
- Wichmann, G. Habitat use of nightjar (Caprimulgus europaeus) in an Austrian pine forest. J. Ornithol. 2004, 145, 69–73. [Google Scholar] [CrossRef]
- Tumiel, T.; Białomyzy, P.; Grygoruk, G.; Korniluk, M.; Świętochowski, P.; Wereszczuk, M.; Skierczyński, M. Valuable and scarce breeding birds of the Special Protection Area Knyszyn Forest. Ornis Polonica 2013, 54, 170–186, (In Polish, with English Abstract). [Google Scholar]
- Winiger, N.; Korner, P.; Arlettaz, R.; Jacot, A. Vegetation structure and decreased moth abundance limit the recolonisation of restored habitat by the European Nightjar. Rethink. Ecol. 2018, 3, 19–33. [Google Scholar] [CrossRef][Green Version]
- Langston, R.H.W.; Liley, D.; Murison, G.; Woddfield, E.; Clarke, R.T. What effects do walkers and dogs have on the distribution and productivity of breeding European Nightjar Caprimulgus europaeus? Ibis 2007, 149, 27–36. [Google Scholar] [CrossRef]
- Sokołowski, A. Lasy Północno—Wschodniej Polski; Centrum Informacyjne Lasów Państwowych: Warszawa, Poland, 2006; pp. 1–359. (In Polish)
- Wilk, T.; Jujka, M.; Krogulec, J.; Chylarecki, P. Important Bird Areas of International Importance in Poland; OTOP: Marki, Poland, 2010; pp. 1–595, (In Polish, with English Summary). [Google Scholar]
- Tumiel, T. Knyszyn Forest. In Important Bird Areas of International Importance in Poland, 1st ed.; Wilk, T., Jujka, M., Krogulec, J., Chylarecki, P., Eds.; OTOP: Marki, Poland, 2010; pp. 199–201, (In Polish, with English Summary). [Google Scholar]
- Google Earth. Available online: https://www.google.pl/intl/pl/earth/ (accessed on 29 July 2014).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach; Springer: New York, NY, USA, 2002; pp. 1–454. [Google Scholar]
- Bartoń, K. MuMln: Multi-Model Inference. R Package Version 1.15.6. 2016. Available online: https://cran.r-project.org/web/packages/MuMIn (accessed on 15 December 2019).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Alexander, I.; Cresswell, B. Foraging by Nightjars Caprimulgus europaeus away from their nesting areas. Ibis 1990, 132, 568–574. [Google Scholar] [CrossRef]
- Sierro, A.; Arlettaz, R.; Naef-Daenzer, B.; Strebel, S.; Zbinden, N. Habitat use and foraging ecology of the nightjar Caprimulgus europaeus in the Swiss Alps: Towards a conservation scheme. Biol. Conserv. 2001, 98, 325–331. [Google Scholar] [CrossRef]
- Sharps, K. The Conservation Ecology of the European Nightjar (Caprimulgus europaeus) in a Complex Heathland-Plantation Landscape. Ph.D. Thesis, University of East Anglia, Norwich, UK, 2013. [Google Scholar]
- Cresswell, B. Nightjars—Some aspects of their behaviour and conservation. Br. Wildl. 1996, 7, 297–304. [Google Scholar]
- Jackson, H.D. A field survey to investigate why nightjars frequent roads at night. Ostrich J. Afr. Ornithol. 2003, 74, 97–101. [Google Scholar] [CrossRef]
| Code | Description |
|---|---|
| AREA | Area of monitored clearings (ha) |
| AGE | Age of monitored clearings (year) |
| TYPE | Type of clearings: clear-felling and patch-felling |
| VEGETAT | Categories of undergrowth vegetation: sparse, medium and dense |
| SEEDTREE | Occurrence of the seed tree: present and absent |
| CLEARNUMB | Number of clearings in radius of 0.5 km |
| OLD | Edge lengths of old forest (m) |
| MEDIUM | Edge lengths of medium age trees (m) |
| YOUNG | Edge lengths of the young stands of the trees (m) |
| TREEAGE | Average age of surrounding trees (year) |
| DISCLEAR | Distance to nearest clearing (km) |
| DISROAD | Distance to nearest roads (km) |
| DISSETTL | Distance to nearest settlements (km) |
| DISEDGE | Distance to forest edge (km) |
| Model (Fixed Effects) | df | LL | AICc | Δ AICc | AICcwt |
|---|---|---|---|---|---|
| CLEARNUMB + VEGETAT + YOUNG + OLD | 6 | −14.245 | 42.2 | 0.00 | 0.019 |
| CLEARNUMB + VEGETAT + YOUNG + MEDIUM | 6 | −14.521 | 42.8 | 0.55 | 0.014 |
| CLEARNUMB + VEGETAT + YOUNG + MEDIUM + OLD | 7 | −13.348 | 43.0 | 0.83 | 0.012 |
| CLEARNUMB + VEGETAT + YOUNG + OLD + TREEAGE | 7 | −13.376 | 43.1 | 0.88 | 0.012 |
| CLEARNUMB + VEGETAT + YOUNG + TREEAGE | 6 | −14.726 | 43.2 | 0.96 | 0.011 |
| CLEARNUMB + VEGETAT + YOUNG | 5 | −16.281 | 43.8 | 1.56 | 0.008 |
| CLEARNUMB + VEGETAT + YOUNG + OLD + DISEDGE | 7 | −13.824 | 44.0 | 1.78 | 0.008 |
| Parameters | Estimate | Standard Error | Z Value | p Value |
|---|---|---|---|---|
| Intercept | 1.194 | 2.061 | 0.579 | 0.562 |
| OLD | −0.004 | 0.003 | −1.589 | 0.112 |
| YOUNG | 0.007 | 0.003 | 2.285 | 0.023 |
| CLEARNUMB | 1.237 | 0.494 | 2.505 | 0.012 |
| VEGETAT: Medium | −4.127 | 2.085 | −1.979 | 0.048 |
| VEGETAT: Dense | −6.170 | 2.388 | −2.583 | 0.010 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polakowski, M.; Broniszewska, M.; Kirczuk, L.; Kasprzykowski, Z. Habitat Selection by the European Nightjar Caprimulgus europaeus in North-Eastern Poland: Implications for Forest Management. Forests 2020, 11, 291. https://doi.org/10.3390/f11030291
Polakowski M, Broniszewska M, Kirczuk L, Kasprzykowski Z. Habitat Selection by the European Nightjar Caprimulgus europaeus in North-Eastern Poland: Implications for Forest Management. Forests. 2020; 11(3):291. https://doi.org/10.3390/f11030291
Chicago/Turabian StylePolakowski, Michał, Monika Broniszewska, Lucyna Kirczuk, and Zbigniew Kasprzykowski. 2020. "Habitat Selection by the European Nightjar Caprimulgus europaeus in North-Eastern Poland: Implications for Forest Management" Forests 11, no. 3: 291. https://doi.org/10.3390/f11030291
APA StylePolakowski, M., Broniszewska, M., Kirczuk, L., & Kasprzykowski, Z. (2020). Habitat Selection by the European Nightjar Caprimulgus europaeus in North-Eastern Poland: Implications for Forest Management. Forests, 11(3), 291. https://doi.org/10.3390/f11030291





