Abstract
Despite the widespread use and strong promotion of the sustainable forest management approach, there are still uncertainties about the actual contribution of current forest management practices to sustainability. We studied the problem of sustainable timber production in four tropical countries (Belize, Guyana, Suriname, and Trinidad and Tobago). Data assessed on experimental plots covering 10 km2 were used to compare management practices of four forest tenure types that commonly exist in the study countries: large scale concessions (LSC), private forests (PR), periodic block system forests (PBS), and community managed forests (CM). As an indicator of sustainable timber production, we calculated the recovery times expected under the initial condition of the stands and compared them with currently practiced cutting cycles. Three growth scenarios were simulated using diameter growth rates (1.6/2.7/4.5 mm year−1) from empirical data from studies in the region. Initial volumes were determined for all commercial trees as well as for commercial trees with a DBH-threshold ≥45 cm. Highest initial volumes were found in LSC and PBS managed forests. Lowest volumes were found in CM and PR forests. Assuming the lowest growth rate for all commercial trees, none of the stands studied reached the initial pre-harvest volumes within the currently practiced cutting cycles. Assuming the highest growth rate for all trees, LSC, PBS, and PR forests reach the initial pre-harvest volume. Looking at the subset of commercial trees with a DBH ≥45 cm, all stands will reach the initial volume within 30 years only if the highest growth rate is assumed. We show that general harvest codes do not guarantee sustainable forest management in the tropics. Local stand conditions must always be one of the guiding principles of sustainable timber utilization. Applying the rigid rules, which do not take into account the current conditions of the stands, entails long-term risk of forest degradation.
1. Introduction
The holistic approach of sustainable forest management (SFM) has become an integral part of modern tropical forest management and addresses the multiple ecological, economic, and social functions of forests [1,2,3]. Although there is no generally accepted definition of SFM, the concept has been strongly promoted by the international community and is an important foundation of the United Nations Convention on Biological Diversity (CBD), the United Nations Convention to Combat Desertification (CCD) and the United Nations Framework Convention on Climate Change (UNFCCC). The ITTO and FAO have launched criteria and indicators as well as guidelines for strengthening SFM of tropical forests [1,4,5].
Despite the widespread use and strong promotion of SFM, there are uncertainties as to whether current tropical forest management is sustainable [6]. Forest degradation by timber harvesting and wood fuel extraction is considered to have a significant impact on tropical forest ecosystems, though varying from region to region [7,8,9]. According to Blaser and ITTO [2], around 403 million hectares (ha) of tropical forests were managed under selective logging, and around 183 million ha were managed with a management plan until 2010. Several studies showed an increase in forest growth in logged compared to non-logged forests [10,11,12,13,14,15,16], which is caused by varying reasons. The level of disturbance caused by the intensity of trees removed from the current growing stock has a marked influence on the rate of recovery of the remaining stand; increasing intensity of disturbance generally reduced the growth of the remaining stand [10,17,18,19,20,21]. It is widely accepted that the implementation of reduce impact logging (RIL) and post-harvesting silvicultural treatments shows a positive impact on growing stock recovery [16,21,22,23,24]. Various studies examined and questioned cutting cycles and harvest intensities in tropical South America (e.g., Piponiot et al. [25], Piponiot et al. [26], Macpherson et al. [27], ter Steege et al. [28]), with the joint consensus questioning the sustainability of current management. Avila et al. [29] studied the effect of logging intensities on tree species composition on 41 permanent sample plots in the Brazilian Amazon over a period of 30 years and showed that high logging intensities with a basal area reduction of > 6.6 m2 ha−1 had a substantial influence on tree species abundance with no signs of return to the pre-logging species composition. Schwartz et al. [30] compared areas where reduced impact logging (RIL) was applied with unlogged areas in the Tapajós National Forest, Eastern Amazon—Brazil and suggest additional silvicultural techniques such as liberation of future crop trees for maintaining the ecological outcome of RIL on the long run. Shima et al. [31] studied the diversity of unlogged and selectively logged Malaysian forests and found that a period of 40 years is not sufficient for selectively logged forests to regain their diversity. In the Guiana Shield, the sustainability of common forest exploitation was studied by Yguel et al. [32]. They studied 12 plots of 6.25 ha each in French Guiana, consisting of control plots, plots with selective logging, with selective logging and thinning, and with selective logging, thinning, and fuelwood harvesting. Harvesting has not affected species richness, but even the slightest form of disturbance has resulted in a decrease in total and commercial biomass. Based on these results, they conclude that the rotation periods commonly used in tropical forests are not sufficient for recovery. Lévesque et al. [33] studied the recovery rate and stem turnover in a primary tropical dry forest in Jamaica on a total sample area of 0.27 ha. Tree height, basal area, and tree diversity in partially cut plots had recovered by more than 80% 10 years after experimental cutting. Size classes with DBH ≥ 14 cm had fewer individuals compared with the pre-disturbance size-class distribution and the biomass lost by cutting could not be recovered.
The present paper utilizes data from study sites covering a total area of 10 km² and are located in four Caribbean countries to examine the sustainability of timber production by comparing four common forest tenure types. The production and regenerative capacity of forest stands is used as an indicator of sustainable timber production.
2. Materials and Methods
2.1. Countries, Study Sites, and Forest Tenure Types
The present study was carried out in four Caribbean countries: Belize, Guyana, Suriname, and Trinidad and Tobago. The climate in the selected countries is tropical with dry and rainy seasons. Suriname and Guyana show two rainy and two dry seasons, with the dry seasons extending from February to April and from August to November. Belize and Trinidad and Tobago exhibit one dry season from January to May. In Trinidad and Tobago and Belize, forests are threatened by hurricanes. All four countries have experience in forest use and management dating back to the beginning of the 20th century [34,35,36].
For each country, at least two research sites were selected. Main decision criteria for the selection of sites were:
- logging was practiced at least once within the past 30 years;
- logging activities were carried out within the project period;
- the implemented forest management system was representative for the Caribbean;
- minimum size of 100 ha;
- participation of granted concessionaires, forest owners, or communities was secured
Four forest tenure types were covered by the site selection (Table 1): (1) large scale concession managed forest (LSC), (2) periodic block system (PBS), (3) private owned forest (PR), and (4) community managed forest (CM). The analyses shown refer to the respective tenure types and not to the countries involved. As the data and results are representative of the tenure types, they cannot be used to assess and compare individual countries.

Table 1.
Sites and tenure types
Large scale concession (LSC): Large scale concession managed in this case means a semi-/controlled management, which includes measurements e.g., establishment of annual cutting areas of 100 ha, pre-harvest inventory of harvestable species, planned skidding, directional felling, tree selection, and marking. The concessionaire has to prepare a management plan which has to be approved by the national forest authority prior to harvesting. The harvest has to follow guidelines published by the forest authority which include the maximum allowable cut per hectare (normally between 20 and 25 m³ ha−1) within 30 years; minimum distance of 10 m between harvest trees; protection of soil, water, and conservation values; block alignment; and the maximum area of roads to be constructed in a felling compartment [2,37].
Periodic Block System (PBS): The periodic block system is a polycyclic selective timber harvesting system. At least one block per year is opened and the trees within the open block are to be sold over a two-year period. After two years the block is closed and allowed to regenerate for a period of 30 years. The trees for sale are selected and marked by forest officers following guidelines for tree selection. So-called ‘replacement trees’ are required for each tree selected for harvest. These trees were to be of the same species and will form the residual stand after the logging. There is no pre-harvest inventory and skid trails are not pre-planned; they are created by loggers in an unplanned manner. Although a limited amount of timber is supposed to be removed from each block on a 30-year cycle, there are blocks that have been clearly over-harvested and trees that are not supposed to be taken are felled and sold to the loggers [38].
Private owned forest (PR): The private owned forest areas for this study are located within the Rio Bravo Conservation and Management Area (RBCMA) in north-western Belize. The RBCMA was part of a logging concession from the mid-19th century until 1982 [39]. Logging was done mainly for mahogany without any management prescriptions until a minimum girth limit was introduced in 1922. The first inventory was conducted in 1975 resulting in a commercial species volume of only 36 m³ ha−1 [40]. The area is managed by the non-governmental organisation Programme for Belize (PFB) since 1988. PFB uses a yield model for the selection of the harvest stand which was developed based on the data of the national permanent sample plot network [41]. Before logging the owner has to apply for a cutting permission by presenting an annual plan of operations to the national forest authorities. A pre-harvest inventory has to be done, skid trails are pre-planned, and a post-harvest inventory has to be executed after logging. The cutting cycle is 40 years [40].
Community managed forest (CM): The communities participating in this study log their forest on an annual basis. The forest is state owned but managed by a community with conventional logging. Cutting permits, so called state forest permits (SFP), are granted on an annual basis. The SFP holder is not committed to present a management plan or to do pre-harvest activities like pre-harvest inventory or skid trail planning [2]. Measures of sustainable forest management (SFM) are written in a code of practice [42] which was adopted by the forests act of Guyana in 2009 [43].
2.2. Block Layout
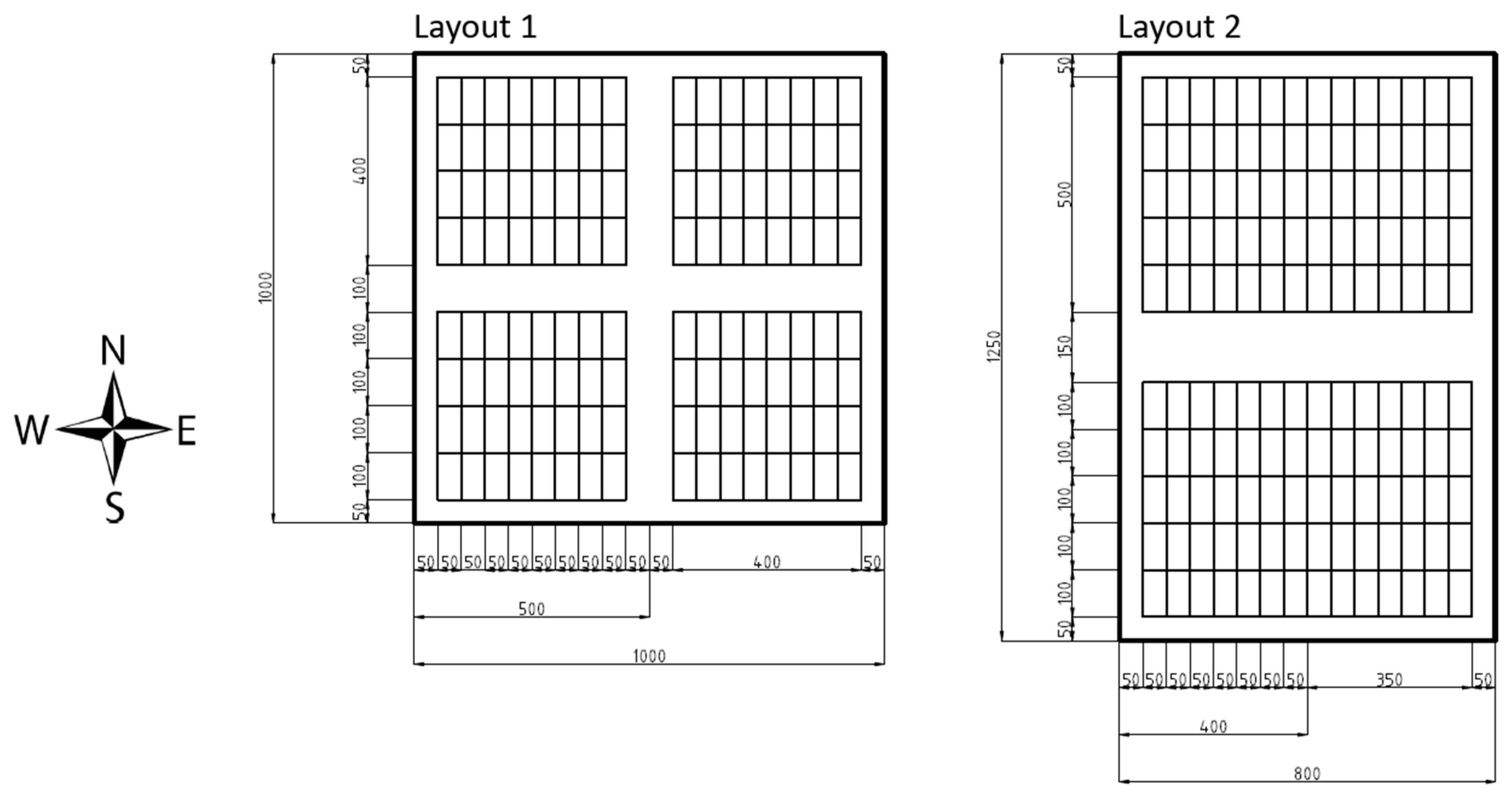
In order to investigate the effect of silvicultural management systems in an objective manner, a randomized block design was chosen for the study. With exception of one site in Suriname, all sites had an area of 1 × 1 km. In each 1 × 1 km site, four blocks containing 32 plots of 50 × 100 m were installed (Figure 1). The individual blocks and the entire 1 × 1 km site were surrounded by a buffer-zone to avoid influences from neighboring stocks. The 32 plots were 0.5 ha in size. Due to the concessionaire’s pre-set logging area alignment for one site in Suriname a modified block design had to be used: The site size was set to 0.8 × 1.25 km with two blocks inside. Within the two blocks, 140 sample plots with a size of 0.5 ha were installed. Both blocks were surrounded by buffer zones.
Figure 1.
Block layout.
2.3. Forest Stock Assessment
A forest stock assessment (pre-harvest inventory) was implemented to obtain information about forest stand attributes: (1) diameter at 1.30 m height (DBH), (2) spatial distribution of trees, (3) log grade (LG), (4) species composition, (5) standing volume, and (6) harvestable timber volume. Log grade was determined using the categories presented in Table 2. Tree species was further categorized in commercial and non-commercial species. The classification was done using local species classification lists provided by the national forest authorities or forest management units. Within the sample plots, every tree with DBH ≥ 25 cm was recorded and mapped.

Table 2.
Log grade categories
2.4. Commercial Species Classes
Only a proportion of the total volume is made of so called commercial species which have a merchantable value on the timber market. All commercial tree species were assigned to four classes (CSC): (1) class A includes species with the highest market value and demand on the timber market; (2) class B species are less valuable species, but with a high acceptance on the timber market; (3) class C species are marketable, but with low demand on the timber market; and (4) class D species are commercial species but with a weaker marketability. The species were categorised based on national species classifications which can be found in Alder [44], GFC [45], Ramnarine et al. [38], and SBB [46,47].
2.5. Tree Selection and Stand Terminology
The selection of trees to be harvested within the forest management units (FMU) is limited by several factors, e.g., the actual timber market demand, or the harvesting capacity of the FMU. In order to be able to compare the intensity of logging in a standardized way, two approaches for selecting harvestable trees were applied: (1) the FMU applied their local tree selection practices and selected the trees to be harvested on the basis of national criteria, and (2) the minimum harvesting diameter (MHD) and log quality were used as uniform, systematic criteria to determine trees to be harvested. Actual logging activities were performed based on the selection decisions of the FMUs.
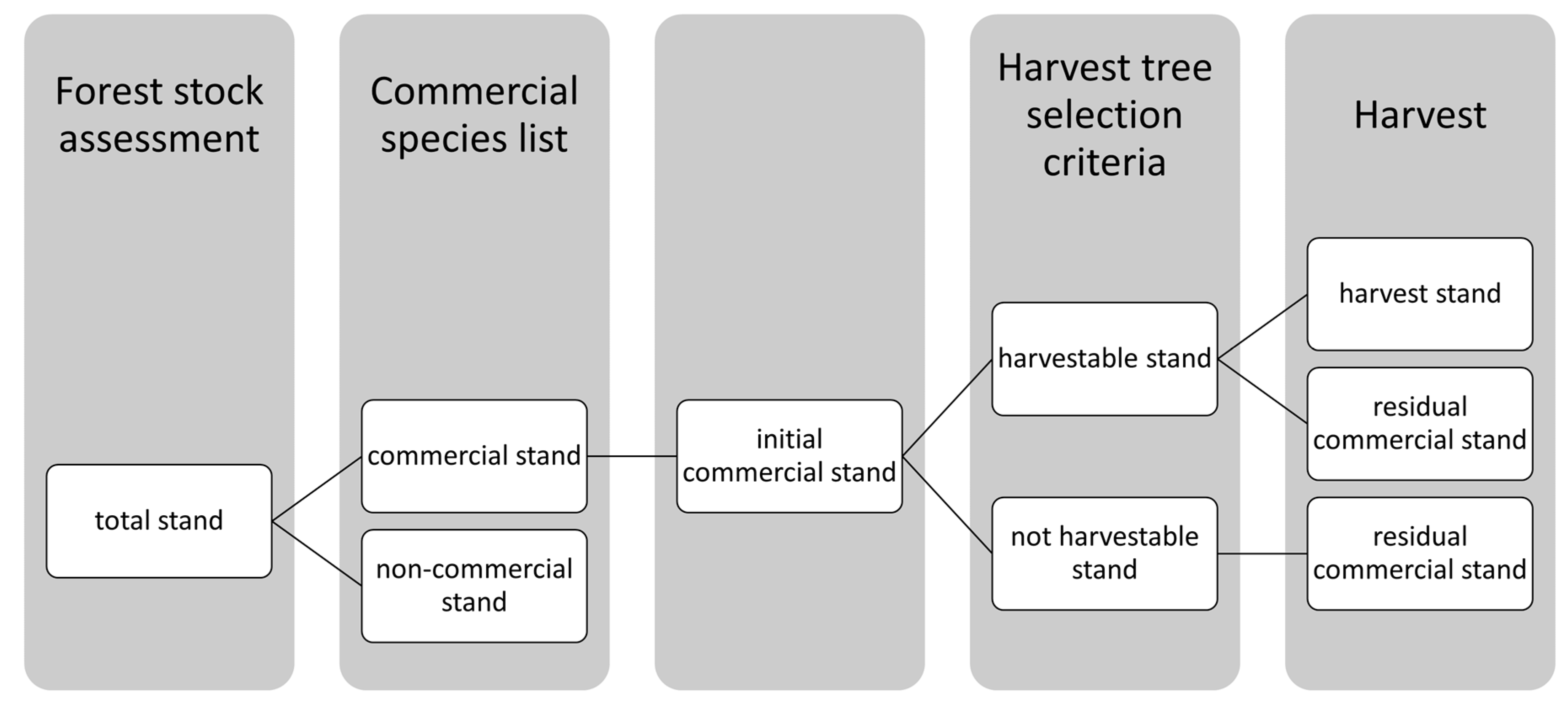
Depending on the selection, the trees were divided into groups: (1) harvestable trees = trees that met the criteria for harvest (see Table 3) and that were potential harvest trees, (2) harvest trees = trees of the harvestable stand which were selected to be harvested during the actual or upcoming harvest, and (3) residual trees = trees of commercial species that formed the remaining commercial stand after logging. The harvested and the residual commercial stand together formed the initial commercial stand (Figure 2).

Table 3.
Harvest tree selection criteria
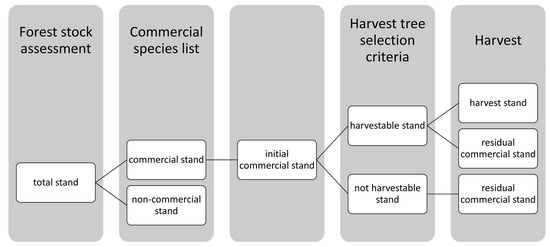
Figure 2.
Systematics of stand terminology.
2.6. Volume Equation
The tree volume was estimated using a DBH-based allometric equation (Equation (1)) according to Alder and van Kuijk [48]. The equation was derived from 1849 felled sample trees covering 137 species.
where
V = volume in m3
DBH = diameter at breast height in cm
2.7. Scenario Analysis
No post-harvest assessments were carried out after previous harvest operations, which did not allow for an empirical estimation of post-intervention tree growth. Therefore, growth rates were taken from former studies conducted in the region and implemented in a scenario analysis. The scenario analysis approach was chosen to include a wide range of potential site specific growth rates. We used a straightforward diameter-increment-approach to simulate the growth of the individual trees. The scenario analysis served to evaluate the quantity of harvestable volume, the resulting harvest intensity, and the tenure type specific cutting cycles (see Section 2.1). For the scenario analysis we used the selection of the harvest stand made by the forest management units.
2.7.1. Simulation Parameters
The parameters mean growth rate and mean annual tree mortality rate were varied for the simulation study. Mean growth rate here is expressed in annual diameter growth. In logged tropical forests diameter growth between 2.3 mm year−1 and 4.6 mm year−1 (Table 4) were reported by Werger [36] and Jonkers [49]. Smaller growth rates were usually observed in undisturbed forests whereas high growth rates result from silvicultural interventions or high intensity logging [36,50]. Vieira et al. [51] found growth rates from 1.7 mm year−1 (mean of minimum growth rates), 3.1 mm year−1 (mean of medium growth rates), to 3.9 mm year−1 (mean of maximum growth rates) for trees of DBH ≥ 10 cm. Lieberman et al. [52] reported growth rates from tropical wet forests in Costa Rica from 0.8 mm year−1 (mean of minimum growth rates), 2.7 mm year−1 (mean of medium growth rates), to 5.1 mm year−1 (mean of maximum growth rates) for trees DBH ≥ 10 cm. Mean growth rates of 2.3 mm year−1 for trees from logged tropical forests of French Guiana were published by Herault et al. [53]. For this study, we used fixed mean growth rates of 1.6 mm year−1, 2.7 mm year−1, and 4.5 mm year−1.

Table 4.
Mean growth rate references
A mean annual mortality rate of 2.6% year−1 in logged forests was observed by Sist and Nguyen-Thé [54]. Vidal et al. [20] published mortality rates of 1.4% year−1 for RIL, 1.7% year−1 for CL within 15 years after logging and 5.9% year−1 for RIL and 6.3% year−1 for CL within 20 years after logging. Johnson et al. [55] observed mortality rates from the Guyana shield of 1.66% year−1. In order to compensate for the recruitment of young trees, which was not included in our calculations, we applied a mean annual mortality rate of 1% year−1 for the overall volume of the stand over the entire simulation period [50,54,56].
2.7.2. Harvesting Percent and Recovery Time
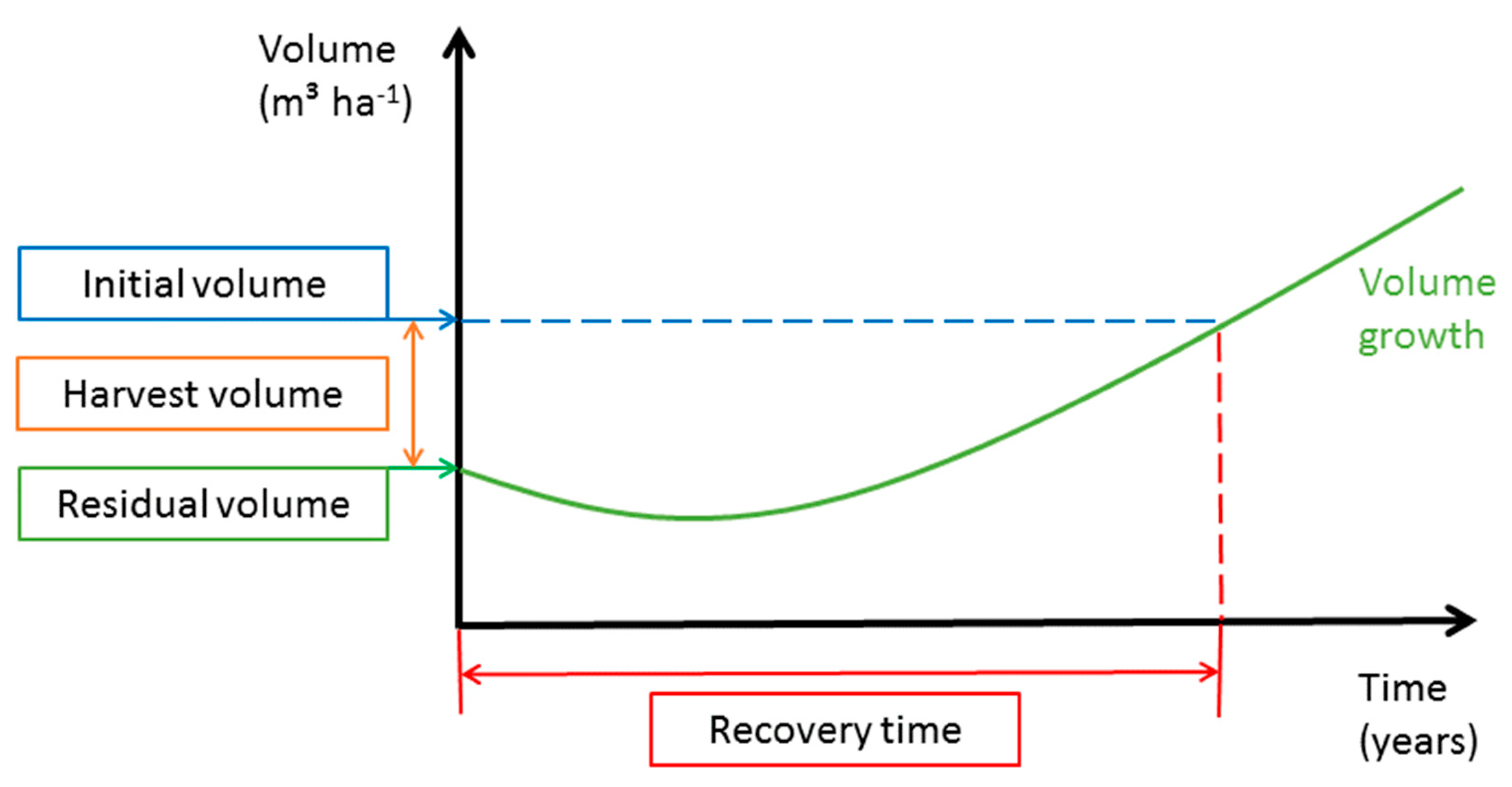
We calculated the harvesting percent (Equation (2)) based on the ratio of initial commercial volume and harvested volume. Recovery time (Equation( 30) here means (1) the time the forest needs to recover its initial commercial volume ignoring the commercial DBH classification and (2) the time the forest needs to recover its initial commercial volume of trees with DBH ≥ MHD. The recovery time was calculated using the annual volume increment and the volume of the initial commercial stand. The results of the growth simulation are presented in graphical form showing the residual volume in relation to initial volume and harvest volume and the time needed for recovery (see Figure 3 as an example).
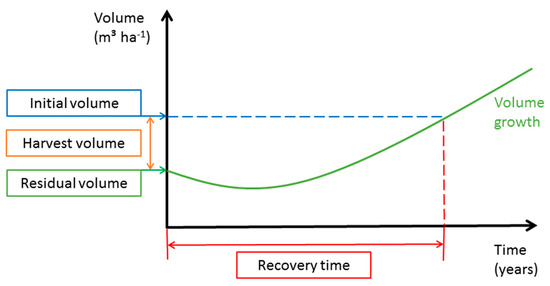
Figure 3.
Growth simulation.
2.7.3. Simulation Scenarios
We calculated the time needed for the stand to reach its initial volume after harvesting (recovery time, see Section 2.7.2). On the one hand, a diameter-independent initial volume, which ignores the commercial DBH classification, was taken as a basis and on the other hand, an initial volume which only takes trees with a DBH ≥ 45 cm (MHD) into account (Table 5). To calculate the recovery time, we assumed three diameter growth levels (see Section 2.7.1): 1.6 mm year−1 (G1.6), 2.7 mm year−1 (G2.7) and 4.5 mm year−1 (G4.5). A mean mortality rate (MR) of 1% year−1 was applied.

Table 5.
Simulation subjects
2.8. Descriptive Statistics
We used the statistical computing environment R in version 3.6.0 [57] and the R-package lme4 [58] to perform a linear mixed effects analysis of the effect of the present forest tenure type on the harvested and commercial residual volume. With a mixed effects model we were able to incorporate both fixed- and random-effects terms in a linear predictor expression. As fixed effects, we entered tenure type into the model. As random effects, we added the study sites.
We used the R-package emmeans [59] to calculate the 95%-confidence intervals of the least-squares means from the fitted linear mixed effects model. To evaluate the differences in volume between the forest tenure types, we used pairwise comparisons of the least-square means and calculated the 95%-confidence intervals of the differences.
3. Results
3.1. Forest Stock Assessment
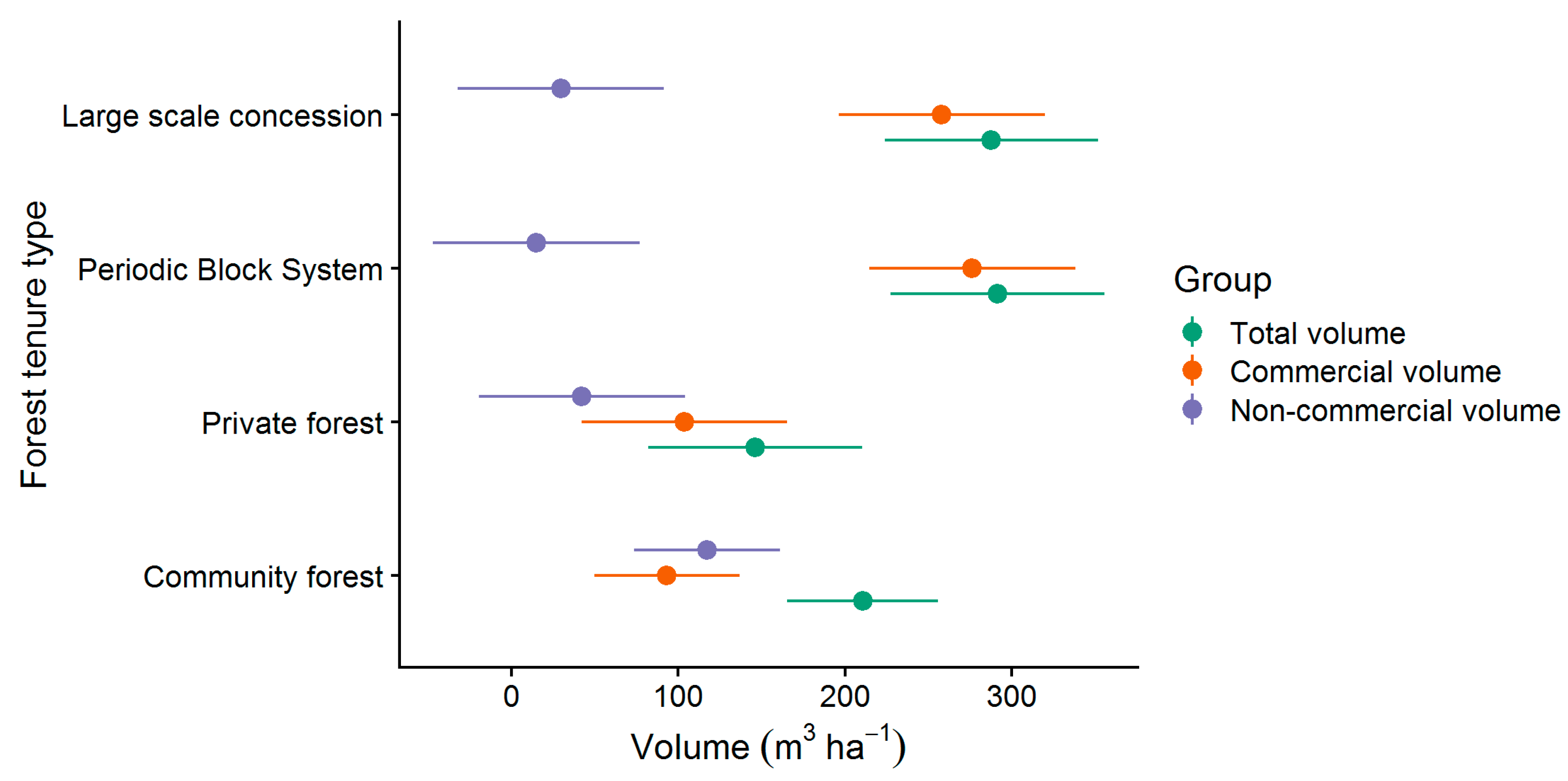
As described in Section 2.3, all trees with DBH ≥ 25 cm were recorded at the forest stock assessment. Total volumes (Figure 4) of 288 m³ ha−1 (224–352 m³ ha−1) and 291 m³ ha−1 (227–355 m³ ha−1) were found at large scale concession (LSC) and periodic block system (PBS), respectively. As indicated by the confidence intervals, significantly lower total volume was found at private forest (PR) with 146 m³ ha−1 (82–210 m³ ha−1). As explained in Section 2.4, only a proportion of the total volume is made up of commercial species. Commercial volumes of LSC (258 ± 62 m³ ha−1) and PBS (276 ± 62 m³ ha−1) were significant higher than commercial volumes of CM (93 ± 62 m³ ha−1) and PR (104 ± 62 m³ ha−1).
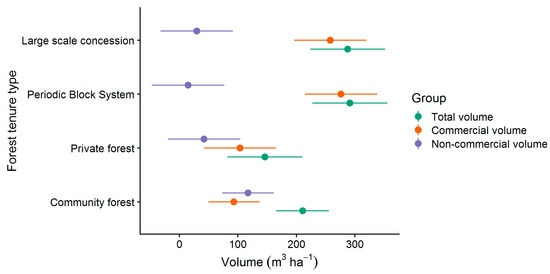
Figure 4.
Commercial and non-commercial volumes and 95%-confidence intervals of the present forest tenure types.
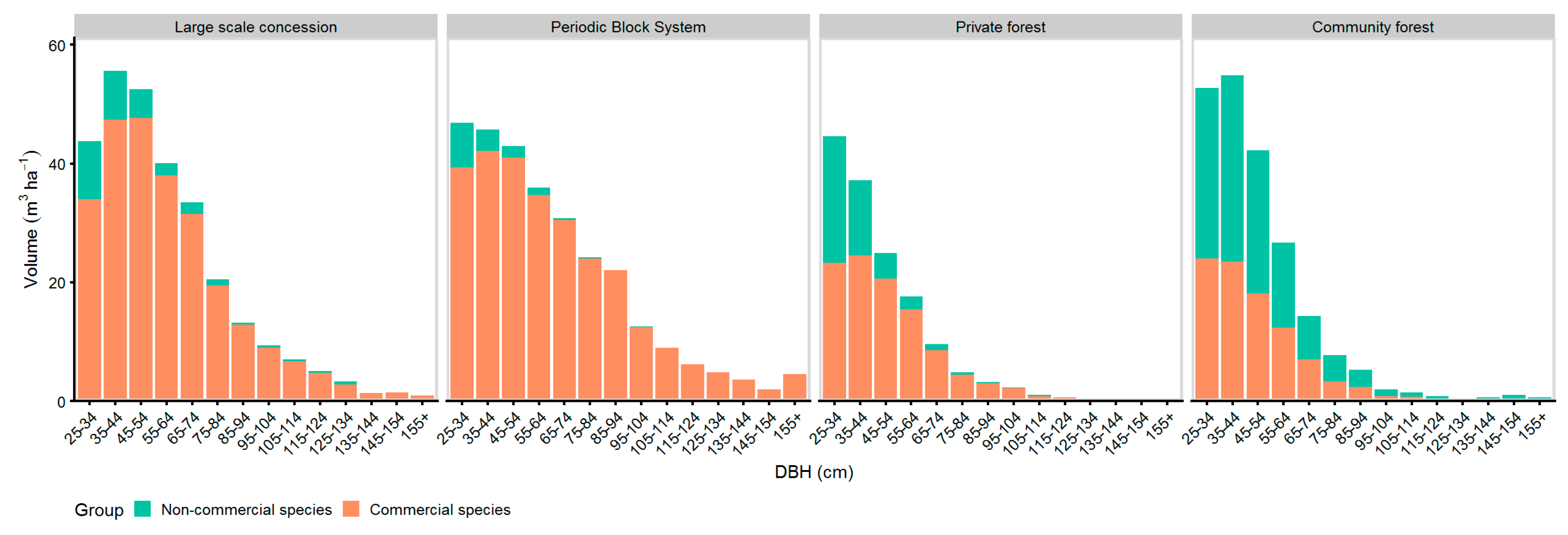
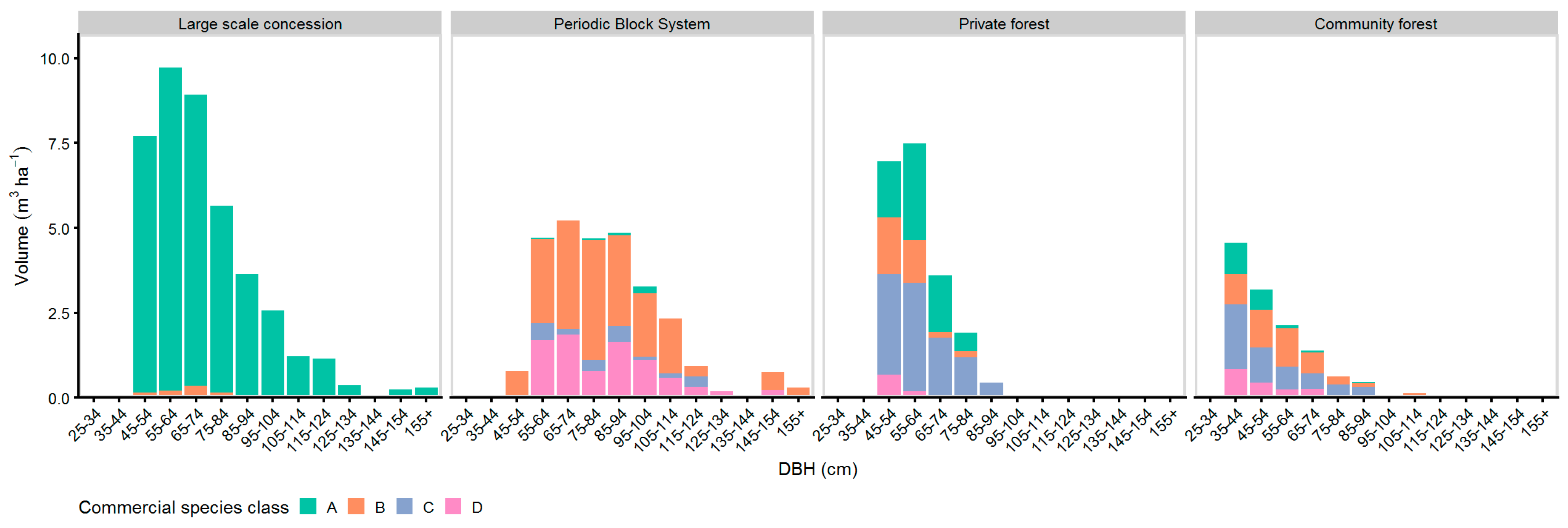
Figure 5 shows the volume diameter distribution of commercial and non-commercial species. The percentage of commercial volume was 90% in LSC and 95% in PBS, while the proportion of commercial volume was 44% in CM. Commercial volume was present at all diameter classes in LSC as well as in PBS, and only in diameter classes up to 105 cm in CM.
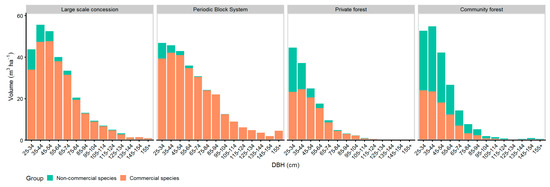
Figure 5.
Volume-DBH distribution of commercial and non-commercial species.
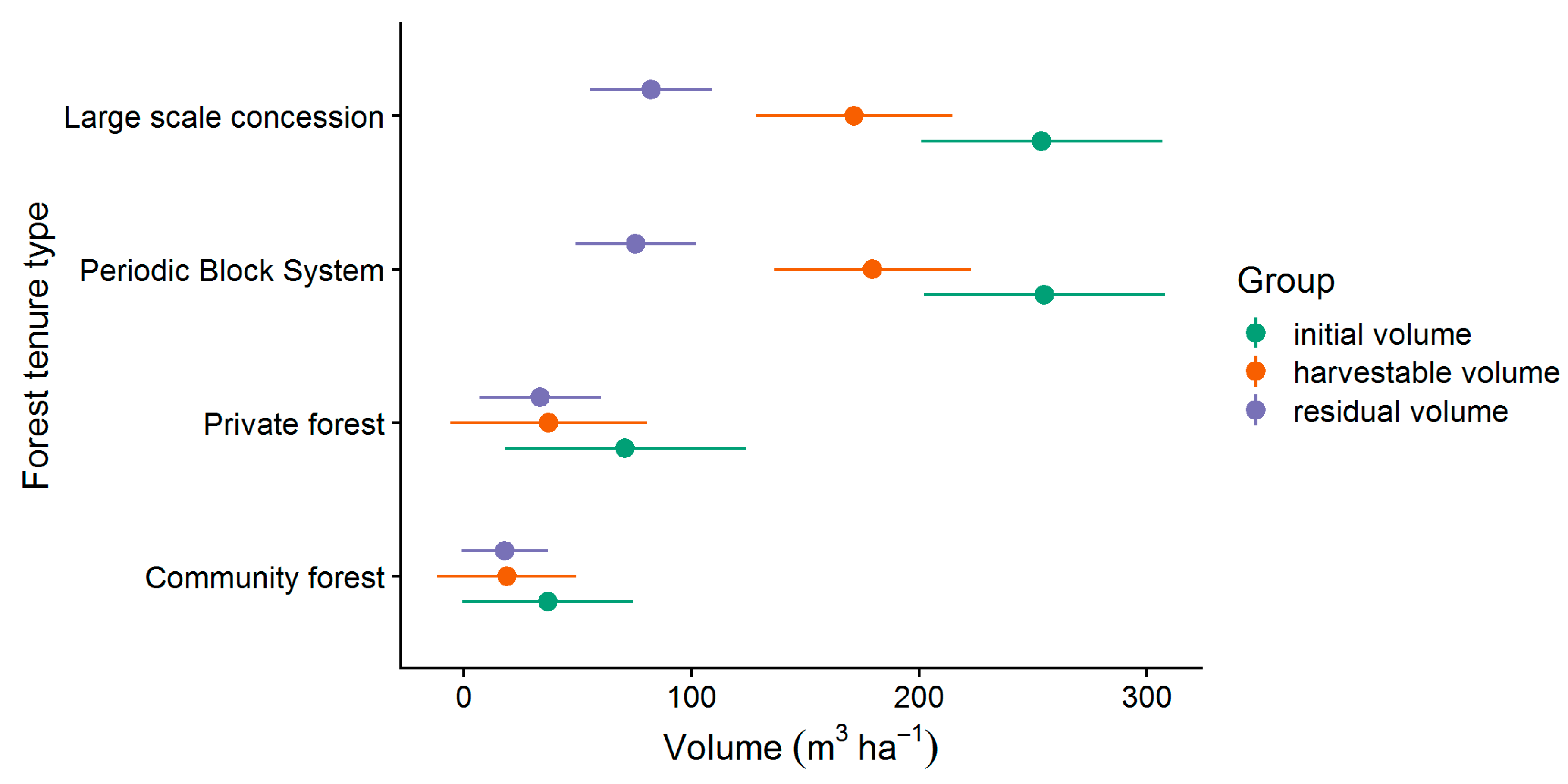
However, only the commercial stand of marketable or potentially marketable trees (all CSC, LG 1 & 2) is relevant for the following considerations, which is why the non-commercial stand is not discussed further. The results for the mean volumes and the corresponding 95%-confidence intervals of the commercial stand (all CSC, LG 1 & 2) are presented in Figure 6. The two tenure types large scale concession (LSC) and periodic block system (PBS) had the highest initial volumes of 254 m³ ha−1 (201–307 m³ ha−1) and 255 m³ ha−1 (202–308 m³ ha−1) respectively. The lowest initial volumes were found in private forest (PR) with 71 m³ ha−1 (18–124 m³ ha−1) and community forest (CM) with 37 m³ ha−1 (−0.5–74 m³ ha−1). While the volumes of LSC and PBS as well as CM and PR did not differ significantly from each other, we could find strong differences between the volumes of the two tenure types LSC and PBS compared to CM and PR. The tenure types also differed in the distribution of the harvestable and residual volume. While the harvestable volume of 172 m³ ha−1 (128–215 m³ ha−1) for LSC and 179 m³ ha−1 (136–223 m³ ha−1) for PBS was significantly higher than the residual volume of 82 m³ ha−1 (56–109 m³ ha−1) and 76 m³ ha−1 (49–102 m³ ha−1), respectively, the harvestable volume at CM with 19 m³ ha−1 (−12–50 m³ ha−1) and PR with 37 m³ ha−1 (−6–81 m³ ha−1) did not differ significantly from the residual volume of 18 m³ ha−1 (−0.8–37 m³ ha−1) for CM and 34 m³ ha−1 (7–60 m³ ha−1) for PR.
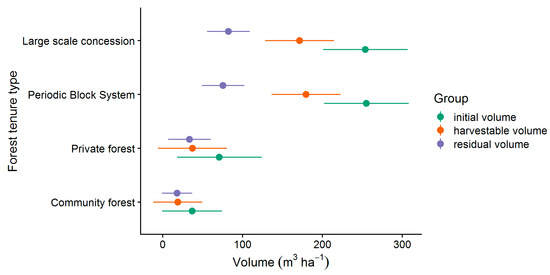
Figure 6.
Mean commercial volumes (all CSC, LG 1 & 2) and 95%-confidence intervals.
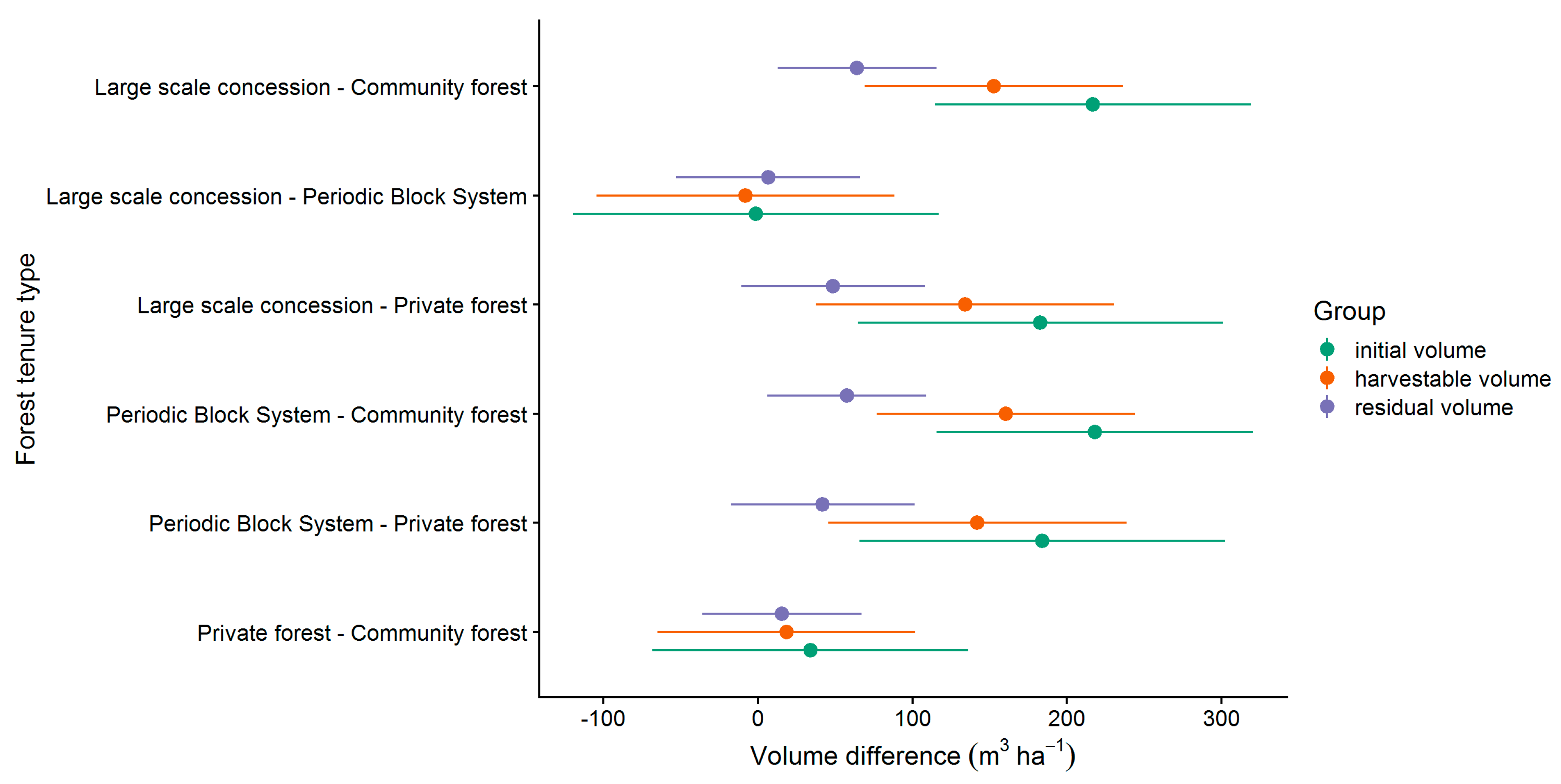
In order to analyze the effect of the tenure types on the volume in more detail, a pairwise comparison was applied (see Section 2.8). The results of the pairwise comparison is presented in Figure 7. The effect of the tenure type differed significantly from zero except in the comparisons LSC with PBS and PR with CM. In all other comparisons, the tenure types LSC and PBS had a positive effect on volume compared to PR and CM. The largest differences were found in the comparisons between LSC or PBS and CM.
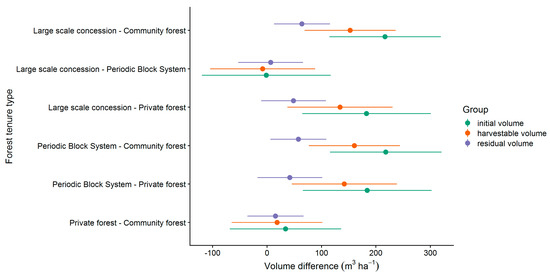
Figure 7.
Pairwise comparison of the effect of the forest tenure types on the commercial volume (all CSC, LG 1 & 2).
3.2. Tree Selection and Species Class Composition
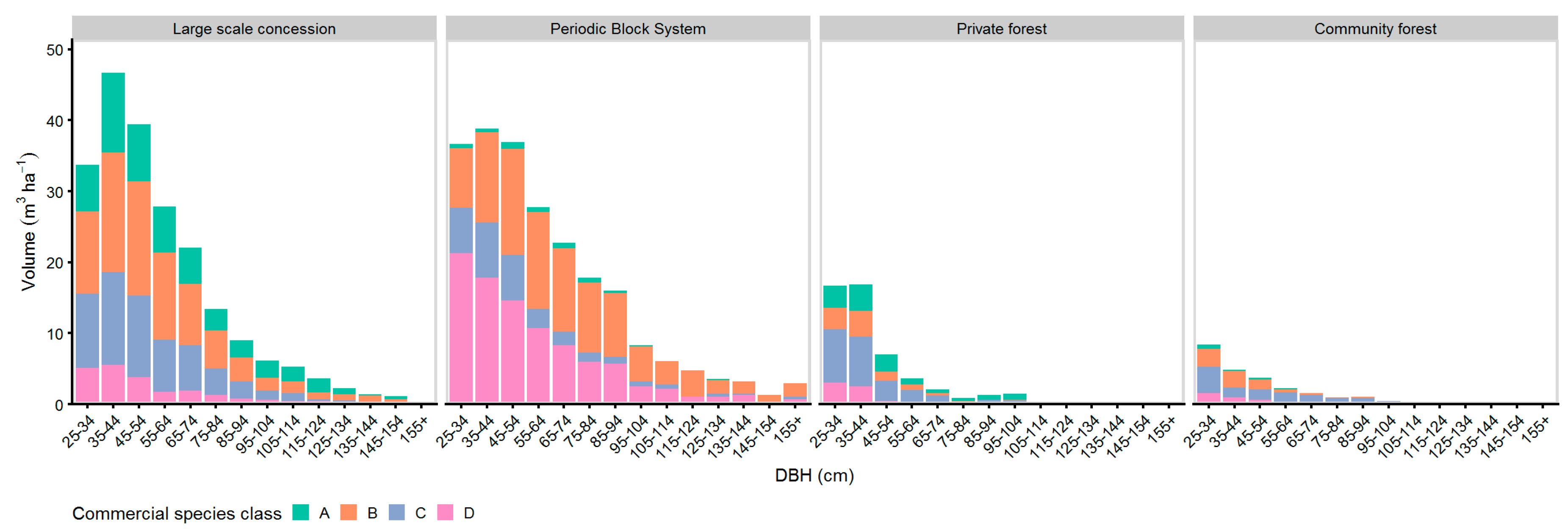
Highest harvest intensities in terms of removals from the growing stock could be found at CM forests showing a harvesting percent of 36%. Despite the high harvesting percent, the harvest volume was the lowest (13 m³ ha−1) of all tenure types. The low harvest volume with the simultaneously high harvesting percent resulted from the low initial commercial volume (37 m³ ha−1) of CM. In contrast, the relatively high initial commercial volume (254 m³ ha−1) at the LSC resulted in the highest harvest volumes (42 m³ ha−1) of all tenure types, while the harvesting percent was low (17%) compared to the CM and PR stands. The comparison of the national harvest tree selection with the standardized harvest tree selection (see Section 2.5) showed that CM used almost the entire potential of the harvestable volume when selecting the harvest stand. All three other tenure types used only part of the potentially harvestable stand.
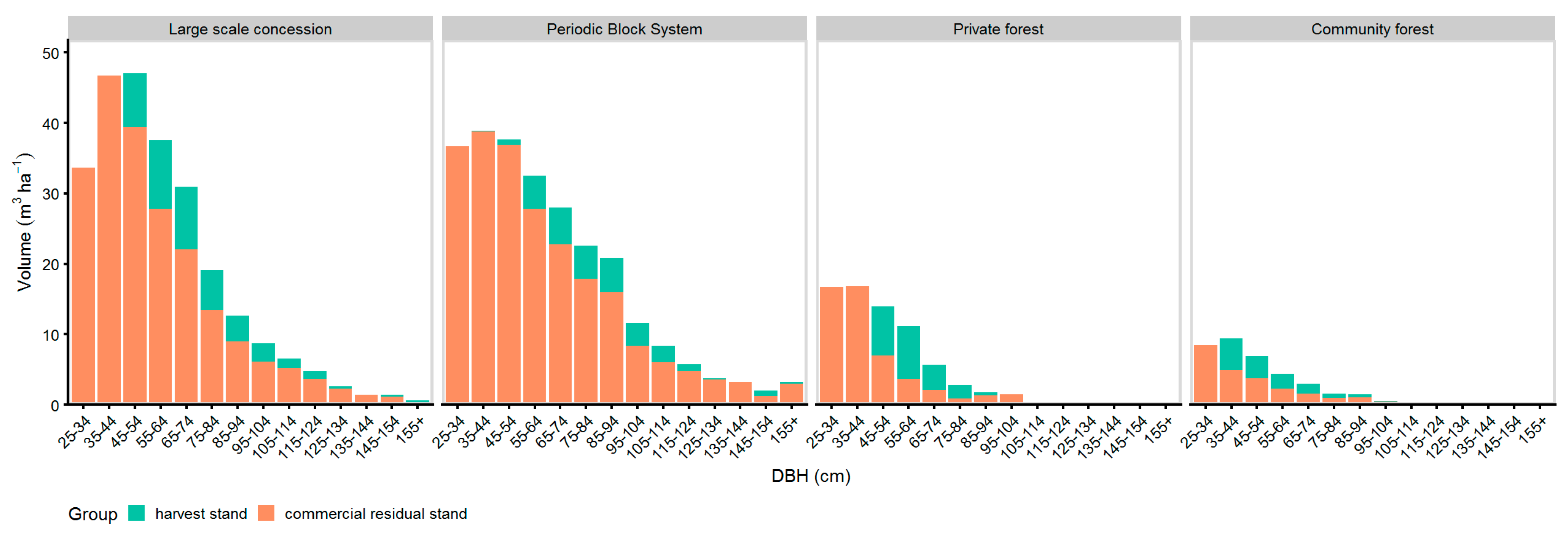
Figure 8 shows the distribution of the harvest intensity (see Table 6) over the diameter classes. In LSC, PBS, and PR managed forests, the harvest trees were mostly selected from DBH ≥ 45 cm. Trees with DBH < 45 cm were selected for harvest only at the community managed forests and a small amount at the PBS managed forest. The highest commercial residual stand remained at the LSC and PBS sites, whereas the lowest commercial residual stand was found at the community forests. The highest distribution of the commercial residual stand over the DBH classes was found at the LSC and PBS managed sites.
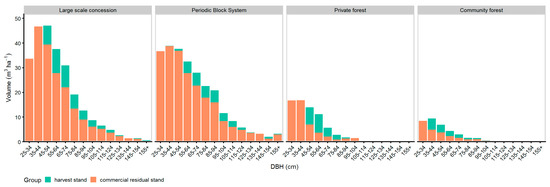
Figure 8.
DBH-histogram of national harvest tree selection and commercial residual stand of trees with LG 1 & 2.

Table 6.
Mean harvest intensities per forest tenure type
Figure 9 and Figure 10 show the tree species class distribution of the harvest stand and the residual stand across the diameter classes. The amount of marketable timber species of class ‘A’ harvest trees was highest at the LSC managed sites and lowest at the PBS. At the commercial residual stand, the highest amount of class ‘A’ species was found again at the LSC managed sites, but lowest amounts were found at the CM sites.
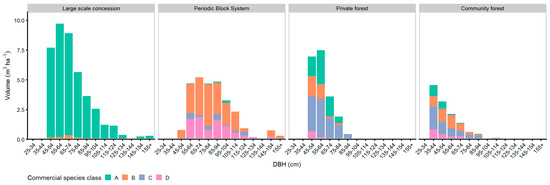
Figure 9.
DBH-histogram of commercial species classes of trees with LG 1 & 2 selected for harvest.
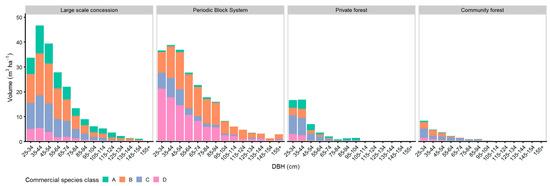
Figure 10.
DBH-histogram of commercial species classes of commercial residual trees with LG 1 & 2.
3.3. Scenario Analysis
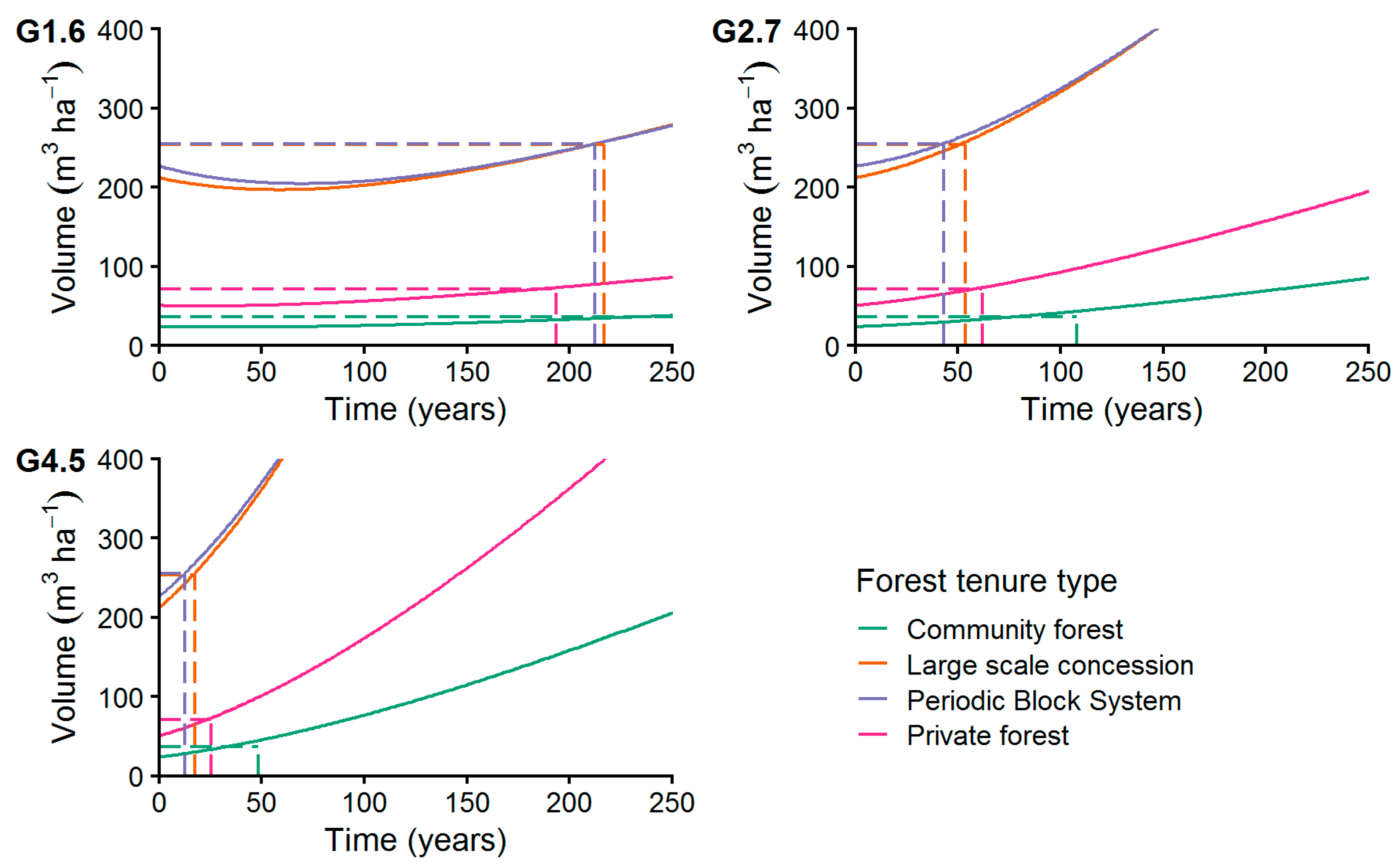
The analysis of the growth scenarios (Figure 11) resulted in a recovery time from 13 to 292 years to recover from harvest back to the initial volume, ignoring the commercial DBH classification of the trees (see Section 2.7.3). We found shortest recovery times at the PBS (13 years) and the LSC (18 years) managed stands at the highest diameter growth rate of 4.5 mm year−1. Applying the highest growth rate at CM and PR managed stands resulted in a recovery time of 48 years (CM) and 26 years (PR). Applying a medium growth rate of 2.7 mm year−1 resulted in recovery times from 43 years in PBS managed forests to 108 years in CM forest. At the lowest growth rate of 1.6 mm year−1, recovery times from 194 years in PR forests to 292 years in CM forests were calculated.
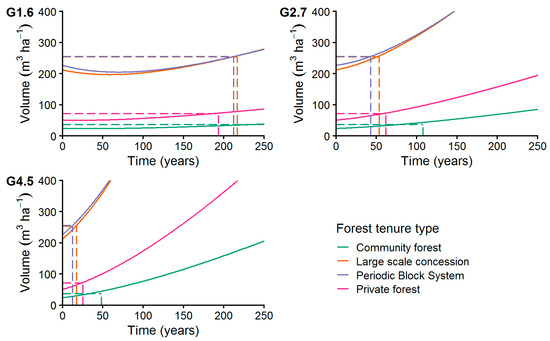
Figure 11.
Increment of commercial species volume applying growth levels G1.6, G2.7, and G4.5 with MR = 1% p.a. The dashed lines connect the initial volume before harvesting with the years needed to achieve it after harvesting (recovery time, see Section 2.7.2).
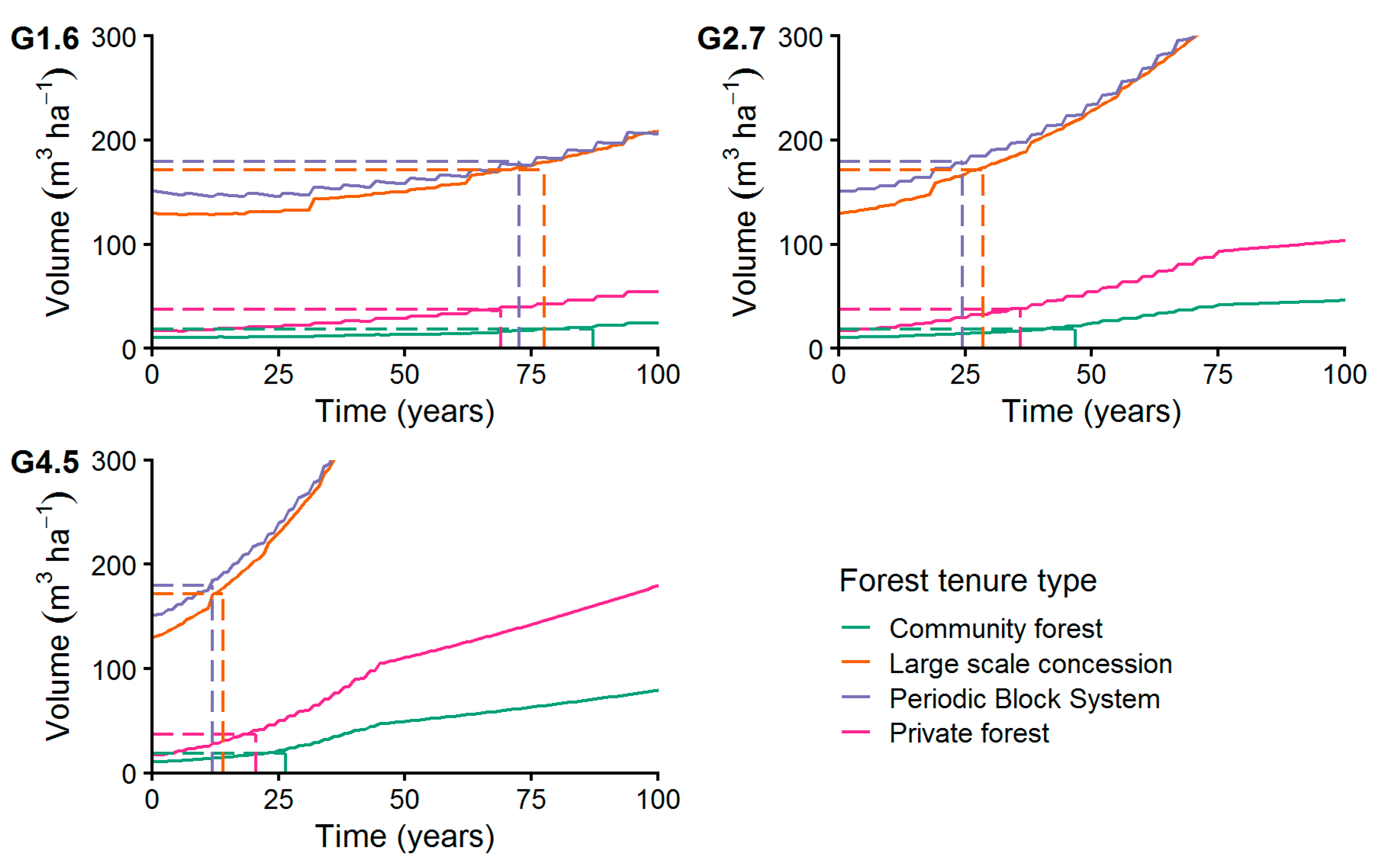
Considering only trees with DBH ≥ MHD for the growth simulation (Figure 12) resulted in recovery times from 12 years (PBS) to 87 years (CM). Applying the highest growth rate (G4.5) of 4.5 mm year−1 we found shortest recovery times at the PBS stands (12 years) and LSC managed stands (14 years). Longest recovery times at the highest growth rate were calculated for PR forests (21 years) and CM forests (27 years). At a low growth rate of 1.6 mm year−1 (G1.6) shortest recovery times were reached by PR stands (69 years) and the PBS managed sites (73 years). The CM stand recovered after 87 years, applying a low growth rate (G1.6).
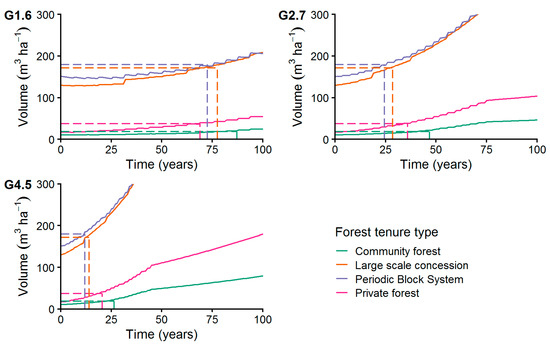
Figure 12.
Increment of commercial species volume of trees with DBH ≥ 45 cm applying growth levels G1.6, G2.7, and G4.5 with MR = 1% p.a. The dashed lines connect the initial volume before harvesting with the years needed to achieve it after harvesting (recovery time, see Section 2.7.2).
4. Discussion
In defining sustainable forest management, economic, ecological, and social aspects can be combined and emphasized differently. Accordingly, the chosen definition plays a decisive role in the assessment of sustainability. For monocyclic management systems in which harvesting operations are carried out at long time intervals without interim maintenance operations, we define sustainability as the time a stand takes to recover the volume used. The recovery time is used to determine the sustainable cutting cycles. From this it can be deduced whether the cutting cycles of 30 to 40 years currently practised in the region under study are in fact sustainable. The initial volumes were determined for all commercial trees (total volume) as well as for commercial trees with a DBH over 45 cm.
Assuming an average annual growth rate of 1.6 mm for each tree, none of the stands studied reach the initial total volume within the currently practiced cutting cycles of 30 to 40 years. Assuming the highest annual growth rate of 4.5 mm for all trees, LSC, PBS, and PR reach the initial total volume. If the sub-set of trees with a diameter over 45 cm is considered, all stands reach the initial volume within 30 years only, if the highest annual growth rate (4.5 mm) is assumed. The time needed by a stand to recover the initial stand volume before harvesting serves as an indicator for sustainability. This renders the assessment of the initial stand before any logging operations necessary, because otherwise a stand volume that has already been degraded is used as a comparative measure. This does not permit an assessment of sustainability, as—on the contrary—this would in retrospect legitimize stand degradation.
The stands studied show clear differences in initial volumes and diameter distribution of commercial species classes. The initial volumes of commercial tree species of the systems PBS and LSC exceed the initial volumes of CM and PR by 200 m³ ha−1. No significant differences could be found between the initial volumes of PBS and LSC and between CM and PR. For forests of the Guyana Shield studies of Jonkers [60] as well as Alder and van Kuijk [48] indicate commercial volumes of 172 to 243 m³ ha−1 and 195 to 237 m³ ha−1, respectively. Alder and van Kuijk [48] consider these volumes as relatively low for tropical forests. The volumes of CM and PR are well below the volumes reported by Alder and van Kuijk [48] and Jonkers [60] while the volumes of PBS and LSC are higher.
The reasons for the observed volumes are not only due to the different management activities, but also to differences in site conditions and geographical location. Three of the four CM stands, as well as the LSC stands, are located in regions with poor soils [61,62]. The two PR stands have been hit several times by hurricanes in the past [40]. LSC and CM are both in similar geographical locations and yet show large volume differences, so that the differences in stand volumes are likely caused by previous management practices. Compared to LSC, stands managed under CM are subject to a less restrictive set of rules and fewer controls. The connection between looser rules and controls for their compliance and the current CM stand structures is obvious.
Beside the market driven, selective extraction of timber species, the management of the LSC stands studied is subject to a code of practice and government controls [2,37], which define parameters such as maximum logging intensities, cutting cycles, or distance rules. The largely regulated use has led to stands with the highest proportion of merchantable volume within the stands studied. To ensure sustainability, it is not so much the permitted harvesting volumes as the cutting cycles that are decisive. This applies as well to individual timber species, which can be harvested in two or more successive cycles. Nevertheless, the Code of Practice [37] alone does not guarantee sustainable use, unless the currently defined cutting cycles of 30 years in LSC allow forests to regenerate. The currently practiced cutting cycles of 30 years have to be corrected in order to safeguard sustainable use. Currently practiced cutting cycles were also criticized by Piponiot et al. [25], Piponiot et al. [26], and Sist and Ferreira [50] who studied forest stands in Amazonia.
The upper limits set for harvest volumes refer only to the extracted stem volume. However, during harvesting operations, additional stand volume are caused by logging losses, e.g., due to skidding trails, or felling gaps [63,64]. Without taking these harvesting losses into account, the anticipated growth assumptions would require a cutting cycle for LSC to be extended to at least 40 years if average growth is assumed, so that the extracted volume can be compensated by growth. If logging losses are taken into account, the recovery periods are extended accordingly.
While distinct rules for the utilization of concessions are enforced, the use of CM is usually uncontrolled. This led to strong harvesting interventions in the past, which benefited from the exploitation of the access infrastructure created by large-scale concessions. As a result, CMs today contain significantly fewer numbers and volumes of commercial trees than LSCs and PBSs. Likewise, the volume of trees with larger diameters is smaller than in LSC and PBS, so that only small harvest volumes are available for future utilization. The uncontrolled and still on-going harvesting activities have put the CM stocks in a state from which they can no longer recover within the periods considered. The available data do not allow an indication of the period over which harvesting measures would need to be suspended to ensure recovery of stocks. However, it is evident that the current management of these stands is not sustainable. Our results therefore show, that community based forest management does not necessarily safeguard sustainability and therefore contradicts studies that see community forestry as a guarantee of the sustainability of forests [65,66,67].
The PR stands were subject to uncontrolled exploitation until 1988, which resulted in a low commercial volume of 36 m³ ha−1 [40]. Despite the uncontrolled previous use and regular past disruptions by hurricanes, the volume under current management doubled during the last 30 years. This was made possible by management adapted to the individual stands. Before each intervention, a pre-harvest inventory is conducted and forms the basis for adjusting the harvesting levels to the respective stand conditions. The PR managed stands are thus an example for the improvement of stand conditions by introducing control measures and silvicultural measures adapted to the current status of stands. However, it remains to be proven through permanent observations that such an approach ensures sustainability in the long term.
Our work is based on case studies, which limits its validity. Specific management alternatives were examined for the countries considered. Therefore, no conclusions for the overall sustainability of forest management in the individual countries are possible. The simulations carried out have some limitations. Constant growth values were used, which do not take into account diameter distributions, neighborhood relations, or the social position of the individual trees. Recruitment and mortality were also not taken into account. Thus, the differences in individual tree growth, which show a high variability, especially in tropical natural forests, cannot be mapped [68,69]. Since there are no long-term observations in the investigated forest populations that allow a differentiated consideration of individual tree growth, mortality and recruitment, a simplifying approach based on stand volume and unified growth functions had to be chosen.
Despite the case study approach, our study allows some general statements. Our results clearly show that the sustainable use of tropical natural forests is not guaranteed by applying general harvesting rules. Rather, the current conditions of the stands must be assessed by pre-harvest inventories and taken into account when planning harvest interventions. Harvesting operations must be based on the current volume of commercial trees, their number, and diameter distribution. If these are not sufficient, harvesting operations must be postponed to a later point in time. This allows stand growth and recovery time to be used as benchmarks for determining harvesting intensities and harvesting cycles. The resulting flexible consideration of the specific conditions of any stand is a prerequisite for sustainable forest management.
5. Conclusions
Forest management through selective logging in the tropics aims at the production of valuable timber over several cutting cycles without fostering measures to stabilize or increase the population size of valuable timber species. When a species disappears, the next valuable species is used instead. We show that the investigated forest tenure types lead to different stand structures after earlier harvest and recovery cycles, and cause tensions between degradation and sustainable harvest volume. The simulated growth scenarios clearly show, that not under all local conditions will past harvest regimes enable sustainable forest management in the future. This also applies when past harvesting operations have been in accordance with existing harvest codes. The application of universal harvesting codes for the use of tropical forests is therefore no guarantee of sustainable forest management. Past experience of close-to-nature sustainable forest management practiced in other regions of the world (e.g., Western Europe, North America, and New Zealand) indicated that the law of locality must be taken into account when assessing sustainability; this is no exception for tropical forests if their sustainable forest management is desired.
Our study shows that the sustainability of forest utilization according to rigid rules must be critically evaluated. The application of rigid rules, which do not take into account the current conditions of the stands, entails the long-term risk of forest degradation. The harvesting quantities and harvesting cycles often defined in harvesting codes must therefore be reconsidered. Rather, local stand conditions must always be the guiding principle of sustainable use. Two demands for the sustainable management of tropical forests can be derived from this. On the one hand, pre-harvest inventories must be carried out in order to provide an objective information basis for the planning of harvesting interventions. Minimum standards, such as standing volume or balanced DBH-distribution, should also be defined for the stand conditions that must be met in order to justify any harvesting intervention. On the other hand, for already logged-over stands, repeated inventories must be carried out to determine growth patterns, natural mortality, and recruitment under local conditions.
Further research on the regeneration ecology and growth dynamics of heterogeneous tropical forests is urgently needed to safeguard the sustainable utilization of timber. Studies on silvicultural treatments are also indispensable to improve diameter growth and to reduce the recovery time until the next intervention. Silvicultural treatments are mainly aiming at (value) growth and tree species distribution, while costs and profitability are the guiding criteria for management measures. Therefore, studies on the economic aspects of silvicultural treatments are necessary. In order to expand the range of marketable species and to reduce the pressure on the relatively few merchantable timber species, studies on the market potential of lesser-known species need to be urgently addressed.
Author Contributions
Conceptualization, M.K. and C.-M.E.; Methodology, S.G. and M.K.; Data curation, S.G.; Validation, S.G. and M.K.; Formal analysis, S.G.; Investigation, S.G., M.P., M.P.O., R.P., and Q.B.; Writing—original draft preparation, S.G. and M.K.; Writing—review and editing, M.P., M.P.O., R.P., and Q.B.; Visualization, S.G.; Supervision, M.K.; Project administration, C.-M.E. and M.K.; Funding acquisition, M.K. and C.-M.E. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the German Ministry for Food and Agriculture (BMEL) through the project “Ensuring long-term productivity of lowland tropical forests in the Caribbean” (GCP/SLC/205/GER). The main objective of this research project is to develop guidelines for the management of logged-over natural forests in the Caribbean. The project is being implemented as a regional government trust fund project conducted in partnership with the University of Hamburg and partner organizations in the participating countries.
Acknowledgments
We would like to express our gratitude to the Belize Forest Department, Belize, the Guyana Forestry Commission, Guyana, the Foundation for Forest Management and Production Control, Suriname, and the Forestry Division of Trinidad and Tobago, for contributing their valuable time and sharing their knowledge and insights to the realization of this research. We are thankful to Thomas Hausmann and Matthias Schwörer, BMEL, for their support and rewarding discussions. Philip Mundhenk deserves thanks for his patient assistance in the application of R. Two unknown reviewers carefully read the first draft of the manuscript and gave valuable hints for revision. We are deeply indebted to them for their contribution to improve the quality of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- International Tropical Timber Organization (ITTO). Voluntary Guidelines for the Sustainable Management of Natural Tropical Forests; International Tropical Timber Organization (ITTO): Yokohama, Japan, 2015; ISBN 978-4-86507-015-6. [Google Scholar]
- Blaser, J.; International Tropical Timber Organization (ITTO). Status of Tropical Forest Management 2011; International Tropical Timber Organization (ITTO): Yokohama, Japan, 2011; ISBN 978-4-902045-78-9. [Google Scholar]
- Pancel, L.; Köhl, M. (Eds.) Tropical Forestry Handbook, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 9783642546020. [Google Scholar]
- International Tropical Timber Organization (ITTO). Criteria and Indicators for the Sustainable Management of Tropical Forests; International Tropical Timber Organization (ITTO): Yokohama, Japan, 2016; ISBN 978-4-86507-028-6. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Voluntary Guidelines on National Forest Monitoring; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2017; ISBN 978-92-5-109619-2. [Google Scholar]
- Nasi, R.; Frost, P. Sustainable forest management in the tropics: Is everything in order but the patient still dying? Ecol. Soc. 2009, 14, 40. [Google Scholar] [CrossRef]
- Achard, F.; Stibig, H.-J.; Eva, H.D.; Lindquist, E.J.; Bouvet, A.; Arino, O.; Mayaux, P. Estimating tropical deforestation from Earth observation data. Carbon Manag. 2010, 1, 271–287. [Google Scholar] [CrossRef]
- Köhl, M.; Lasco, R.; Cifuentes, M.; Jonsson, Ö.; Korhonen, K.T.; Mundhenk, P.; de Jesus Navar, J.; Stinson, G. Changes in forest production, biomass and carbon: Results from the 2015 UN FAO Global Forest Resource Assessment. For. Ecol. Manag. 2015, 352, 21–34. [Google Scholar] [CrossRef]
- Pearson, T.R.H.; Brown, S.; Murray, L.; Sidman, G. Greenhouse gas emissions from tropical forest degradation: An underestimated source. Carbon Balance Manag. 2017, 12, 3. [Google Scholar] [CrossRef]
- Chapman, C.A.; Chapman, L.J. Forest Regeneration in Logged and Unlogged Forests of Kibale National Park, Uganda. Biotropica 1997, 29, 396–412. [Google Scholar] [CrossRef]
- Pélissier, R.; Pascal, J.-P.; Houllier, F.; Laborde, H. Impact of selective logging on the dynamics of a low elevation dense moist evergreen forest in the Western Ghats (South India). For. Ecol. Manag. 1998, 105, 107–119. [Google Scholar] [CrossRef]
- Bischoff, W.; Newbery, D.M.; Lingenfelder, M.; Schnaeckel, R.; Petol, G.H.; Madani, L.; Ridsdale, C.E. Secondary succession and dipterocarp recruitment in Bornean rain forest after logging. For. Ecol. Manag. 2005, 218, 174–192. [Google Scholar] [CrossRef]
- Berry, N.J.; Phillips, O.L.; Lewis, S.L.; Hill, J.K.; Edwards, D.P.; Tawatao, N.B.; Ahmad, N.; Magintan, D.; Khen, C.V.; Maryati, M.; et al. The high value of logged tropical forests: Lessons from northern Borneo. Biodivers. Conserv. 2010, 19, 985–997. [Google Scholar] [CrossRef]
- Hawthorne, W.D.; Sheil, D.; Agyeman, V.K.; Abu Juam, M.; Marshall, C.A.M. Logging scars in Ghanaian high forest: Towards improved models for sustainable production. For. Ecol. Manag. 2012, 271, 27–36. [Google Scholar] [CrossRef]
- Mazzei, L.; Sist, P.; Ruschel, A.; Putz, F.E.; Marco, P.; Pena, W.; Ferreira, J.E.R. Above-ground biomass dynamics after reduced-impact logging in the Eastern Amazon. For. Ecol. Manag. 2010, 259, 367–373. [Google Scholar] [CrossRef]
- Gourlet-Fleury, S.; Mortier, F.; Fayolle, A.; Baya, F.; Ouedraogo, D.; Benedet, F.; Picard, N. Tropical forest recovery from logging: A 24 year silvicultural experiment from Central Africa. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120302. [Google Scholar] [CrossRef]
- Bonnell, T.R.; Reyna-Hurtado, R.; Chapman, C.A. Post-logging recovery time is longer than expected in an East African tropical forest. For. Ecol. Manag. 2011, 261, 855–864. [Google Scholar] [CrossRef]
- Peña-Claros, M.; Peters, E.M.; Justiniano, M.J.; Bongers, F.; Blate, G.M.; Fredericksen, T.S.; Putz, F.E. Regeneration of commercial tree species following silvicultural treatments in a moist tropical forest. For. Ecol. Manag. 2008, 255, 1283–1293. [Google Scholar] [CrossRef]
- Sist, P.; Mazzei, L.; Blanc, L.; Rutishauser, E. Large trees as key elements of carbon storage and dynamics after selective logging in the Eastern Amazon. For. Ecol. Manag. 2014, 318, 103–109. [Google Scholar] [CrossRef]
- Vidal, E.; West, T.A.P.; Putz, F.E. Recovery of biomass and merchantable timber volumes twenty years after conventional and reduced-impact logging in Amazonian Brazil. For. Ecol. Manag. 2016, 376, 1–8. [Google Scholar] [CrossRef]
- Villegas, Z.; Peña-Claros, M.; Mostacedo, B.; Alarcón, A.; Licona, J.C.; Leaño, C.; Pariona, W.; Choque, U. Silvicultural treatments enhance growth rates of future crop trees in a tropical dry forest. For. Ecol. Manag. 2009, 258, 971–977. [Google Scholar] [CrossRef]
- Graaf, N.R.; de Poels, R.L.H.; Van Rompaey, R.S.A.R. Effect of silvicultural treatment on growth and mortality of rainforest in Surinam over long periods. For. Ecol. Manag. 1999, 124, 123–135. [Google Scholar] [CrossRef]
- Priyadi, H.; Gunarso, P.; Kanninen, M. (Eds.) Permanent Sample Plots: More than Just Forest Data; Center for International Forestry Research (CIFOR): Bogor, Indonesia, 2006; ISBN 979244632X. [Google Scholar]
- Peña-Claros, M.; Fredericksen, T.S.; Alarcón, A.; Blate, G.M.; Choque, U.; Leaño, C.; Licona, J.C.; Mostacedo, B.; Pariona, W.; Villegas, Z.; et al. Beyond reduced-impact logging: Silvicultural treatments to increase growth rates of tropical trees. For. Ecol. Manag. 2008, 256, 1458–1467. [Google Scholar] [CrossRef]
- Piponiot, C.; Derroire, G.; Descroix, L.; Mazzei, L.; Rutishauser, E.; Sist, P.; Hérault, B. Assessing timber volume recovery after disturbance in tropical forests—A new modelling framework. Ecol. Model. 2018, 384, 353–369. [Google Scholar] [CrossRef]
- Piponiot, C.; Rödig, E.; Putz, F.E.; Rutishauser, E.; Sist, P.; Ascarrunz, N.; Blanc, L.; Derroire, G.; Descroix, L.; Guedes, M.C.; et al. Can timber provision from Amazonian production forests be sustainable? Environ. Res. Lett. 2019, 14, 64014. [Google Scholar] [CrossRef]
- Macpherson, A.J.; Carter, D.R.; Schulze, M.D.; Vidal, E.; Lentini, M.W. The sustainability of timber production from Eastern Amazonian forests. Land Use Policy 2012, 29, 339–350. [Google Scholar] [CrossRef]
- Ter Steege, H.; Welch, I.; Zagt, R. Long-term effect of timber harvesting in the Bartica Triangle, Central Guyana. For. Ecol. Manag. 2002, 170, 127–144. [Google Scholar] [CrossRef]
- De Avila, A.L.; Ruschel, A.R.; de Carvalho, J.O.P.; Mazzei, L.; Silva, J.N.M.; do Carmo Lopes, J.; Bauhus, J. Medium-term dynamics of tree species composition in response to silvicultural intervention intensities in a tropical rain forest. Biol. Conserv. 2015, 191, 577–586. [Google Scholar] [CrossRef]
- Schwartz, G.; Peña-Claros, M.; Lopes, J.C.A.; Mohren, G.M.J.; Kanashiro, M. Mid-term effects of reduced-impact logging on the regeneration of seven tree commercial species in the Eastern Amazon. For. Ecol. Manag. 2012, 274, 116–125. [Google Scholar] [CrossRef]
- Shima, K.; Yamada, T.; Okuda, T.; Fletcher, C.; Kassim, A.R. Dynamics of Tree Species Diversity in Unlogged and Selectively Logged Malaysian Forests. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Yguel, B.; Piponiot, C.; Mirabel, A.; Dourdain, A.; Hérault, B.; Gourlet-Fleury, S.; Forget, P.-M.; Fontaine, C. Beyond species richness and biomass: Impact of selective logging and silvicultural treatments on the functional composition of a neotropical forest. For. Ecol. Manag. 2019, 433, 528–534. [Google Scholar] [CrossRef]
- Lévesque, M.; McLaren, K.P.; McDonald, M.A. Recovery and dynamics of a primary tropical dry forest in Jamaica, 10 years after human disturbance. For. Ecol. Manag. 2011, 262, 817–826. [Google Scholar] [CrossRef]
- Record, S.J. Forestry in British Honduras. J. For. 1926, 24, 562–568. [Google Scholar] [CrossRef]
- Wadsworth, F.H. Forest Production for Tropical America; USDA Forest Service: Washington, DC, USA, 1997.
- Werger, M.J.A. Sustainable management of tropical rainforests. In The CELOS Management System; Tropenbos International: Wageningen, The Netherlands, 2011. [Google Scholar]
- Van der Hout, P. Praktijkrichtlijnen voor duurzame houtgoogst in Suriname. In Code of Practice; Foundation for Forest Management and Production Control (SBB): Paramaribo, Suriname, 2011. [Google Scholar]
- Ramnarine, S.; Jhilmit, S.; John, K. Manual for the Establishment and Management of Periodic Blocks in Trinidad; Ministry of Public Utilities and the Environment: Port of Spain, Trinidad and Tobago, 2002.
- Shono, K.; Snook, L.K. Growth of big-leaf mahogany (Swietenia macrophylla King) in natural forests in Belize. J. Trop. For. Sci. 2006, 18, 66–73. [Google Scholar]
- Programme for Belize (PFB). Sustainable Forest Management Plan 2017 to 2021 for the Rio Bravo Conservation and Management Area; Programme for Belize (PFB): Belize City, Belize, 2016. [Google Scholar]
- Cho, P.; Blackburn, G.A.; Bird, N.M.; Brewer, S.W.; Barlow, J. The FORMNET-B database: Monitoring the biomass and dynamics of disturbed and degraded tropical forests. J. Veg. Sci. 2013, 24, 1204–1207. [Google Scholar] [CrossRef]
- Guyana Forestry Commission (GFC). Code of Practice for Forest Operations. In State Forest Permission Holders (Small-Scale Forest Operators), 3rd ed.; Guyana Forestry Commission (GFC): Georgetown, Guyana, 2014. [Google Scholar]
- Parliament of Guyana. Forests Act 2009. 2009. Available online: https://www.forestry.gov.gy/wp-content/uploads/2016/07/Forests-Act-2009.pdf (accessed on 18 September 2019).
- Alder, D. An Assessment of Broadleaf Forest Resources and Sustainable Yield in Belize; Belize Forest Planning and Management Project Consultancy Report No. 11. 1993. Available online: https://mayaforestbelize.files.wordpress.com/2016/11/alder-forest-resources-1993.pdf (accessed on 9 August 2019).
- Guyana Forestry Commission (GFC). Forest Sector Information Report—Annual Review 2016; Guyana Forestry Commission (GFC): Georgetown, Guyana, 2016.
- Foundation for Forest Management and Production Control (SBB). Suriname Forestry Sector 2013; Foundation for Forest Management and Production Control (SBB): Paramaribo, Suriname, 2014. [Google Scholar]
- Foundation for Forest Management and Production Control (SBB). Production, Export and Import of Timber and Timber Products in 2012; Foundation for Forest Management and Production Control (SBB): Paramaribo, Suriname, 2013. [Google Scholar]
- Alder, D.; van Kuijk, M. A Baseline Assessment of Forest Carbon in Guyana. 2009. Available online: http://www.denisalder.net/pdf/gybasass09.pdf (accessed on 31 October 2018).
- Jonkers, W.B.J.; Tjon, K.; Wirjosentono, J. Long-term effects of logging in a Neotropical rain forest in Suriname. In Proceedings of the International Expert Meeting on the Development and Implementation of National Codes of Practice for Forest Harvesting: Issues and Options, Chiba, Japan, 17–20 November 2003; Japan International Forestry Cooperation Office: Tokyo, Japan, 2003; pp. 157–166. [Google Scholar]
- Sist, P.; Ferreira, F.N. Sustainability of reduced-impact logging in the Eastern Amazon. For. Ecol. Manag. 2007, 243, 199–209. [Google Scholar] [CrossRef]
- Vieira, S.; de Camargo, P.B.; Selhorst, D.; da Silva, R.; Hutyra, L.; Chambers, J.Q.; Brown, I.F.; Higuchi, N.; dos Santos, J.; Wofsy, S.C.; et al. Forest structure and carbon dynamics in Amazonian tropical rain forests. Oecologia 2004, 140, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, D.; Lieberman, M.; Hartshorn, G.; Peralta, R. Growth rates and age-size relationships of tropical wet forest trees in Costa Rica. J. Trop. Ecol. 1985, 1, 97–109. [Google Scholar] [CrossRef]
- Herault, B.; Ouallet, J.; Blanc, L.; Wagner, F.; Baraloto, C. Growth responses of neotropical trees to logging gaps. J. Appl. Ecol. 2010, 47, 821–831. [Google Scholar] [CrossRef]
- Sist, P.; Nguyen-Thé, N. Logging damage and the subsequent dynamics of a dipterocarp forest in East Kalimantan (1990–1996). For. Ecol. Manag. 2002, 165, 85–103. [Google Scholar] [CrossRef]
- Johnson, M.O.; Galbraith, D.; Gloor, M.; de Deurwaerder, H.; Guimberteau, M.; Rammig, A.; Thonicke, K.; Verbeeck, H.; von Randow, C.; Monteagudo, A.; et al. Variation in stem mortality rates determines patterns of above-ground biomass in Amazonian forests: Implications for dynamic global vegetation models. Glob. Chang. Biol. 2016, 22, 3996–4013. [Google Scholar] [CrossRef]
- Sheil, D.; May, R.M. Mortality and Recruitment Rate Evaluations in Heterogeneous Tropical Forests. J. Ecol. 1996, 84, 91. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org (accessed on 1 July 2019).
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67. [Google Scholar] [CrossRef]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2018. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 29 June 2018).
- Jonkers, W.B.J. Vegetation Structure, Logging Damage and Silviculture in a Tropical Rain Forest in Suriname. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 1987. [Google Scholar]
- Ter Steege, H.; Boot, R.G.A.; Brouwer, L.C. Ecology and Logging in a Tropical Rain Forest in Guyana: With Recommendations for Forest Management; Backhuys Publ: Leiden, The Netherlands, 1996; ISBN 9051130260. [Google Scholar]
- Clarke, G. Law Compliance and Prevention and Control of Illegal Activities in the Forest Sector in Guyana: Preliminary Report Prepared for the World Bank; The World Bank No. 64607. 2006. Available online: http://www-wds.worldbank.org/external/default/WDSContentServer/WDSP// (accessed on 13 August 2019).
- Enters, T. Trash or treasure? Logging and Mill Residues in Asia and the Pacific; Food and Agricultural Organization of the United Nations Regional Office for Asia and the Pacific: Bangkok, Thailand, 2001; ISBN 974-7946-12-2. [Google Scholar]
- Butarbutar, T.; Köhl, M.; Neupane, P.R. Harvested wood products and REDD+: Looking beyond the forest border. Carbon Balance Manag. 2016, 11, 4. [Google Scholar] [CrossRef][Green Version]
- Jafari, A.; Sadeghi Kaji, H.; Azadi, H.; Gebrehiwot, K.; Aghamir, F.; van Passel, S. Assessing the sustainability of community forest management: A case study from Iran. For. Policy Econ. 2018, 96, 1–8. [Google Scholar] [CrossRef]
- Baral, S.; Gautam, A.P.; Vacik, H. Ecological and economical sustainability assessment of community forest management in Nepal: A reality check. J. Sustain. For. 2018, 37, 820–841. [Google Scholar] [CrossRef]
- Ojha, H.; Timinsa, N.P.; Kumar, C.; Belcher, B.; Banjade, M.R.; Chhetri, R.; Nightingale, A. Community-based Forest Management Programmes in Nepal: An Overview of Contexts, Policies, Practices and Issues. In Communities, Forests, and Governance: Policy and Institutional Innovations from Nepal; Ojha, H.R., Ed.; Adroit: New Delhi, India, 2008; pp. 25–54. ISBN 9788187392842. [Google Scholar]
- Newbery, D.M.; Ridsdale, C.E. Neighbourhood abundance and small-tree survival in a lowland Bornean rainforest. Ecol. Res. 2016, 31, 353–366. [Google Scholar] [CrossRef]
- Köhl, M.; Neupane, P.R.; Lotfiomran, N.; Delzon, S. The impact of tree age on biomass growth and carbon accumulation capacity: A retrospective analysis using tree ring data of three tropical tree species grown in natural forests of Suriname. PLoS ONE 2017, 12, e0181187. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).