The Potential Global Distribution of the White Peach Scale Pseudaulacaspis pentagona (Targioni Tozzetti) under Climate Change
Abstract
1. Introduction
2. Materials and Methods
2.1. Species Occurrence Data
2.2. Environmental Variables
2.2.1. Current Environmental Variables
2.2.2. Future Environmental Variables
2.3. Environmental Niche Models
2.4. Model Evaluation
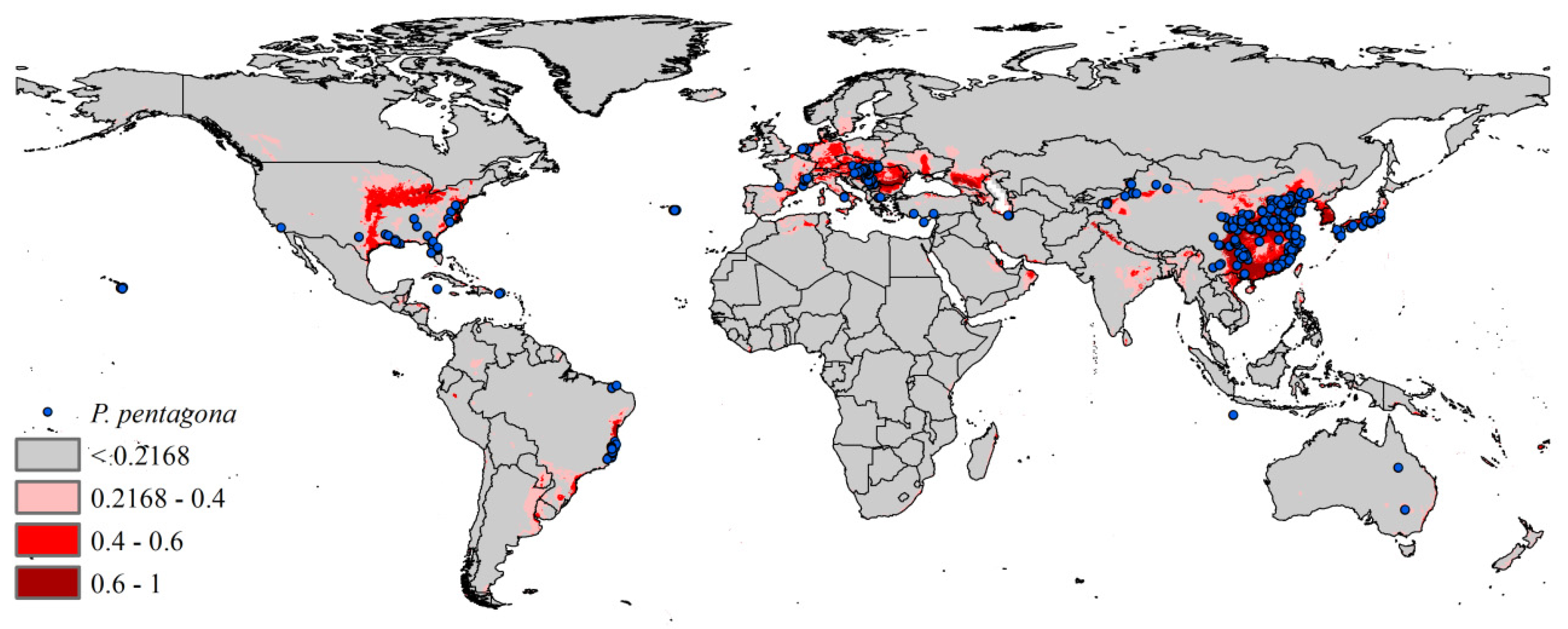
3. Results
3.1. Statistical Model Performance
3.2. Current Potential Distribution
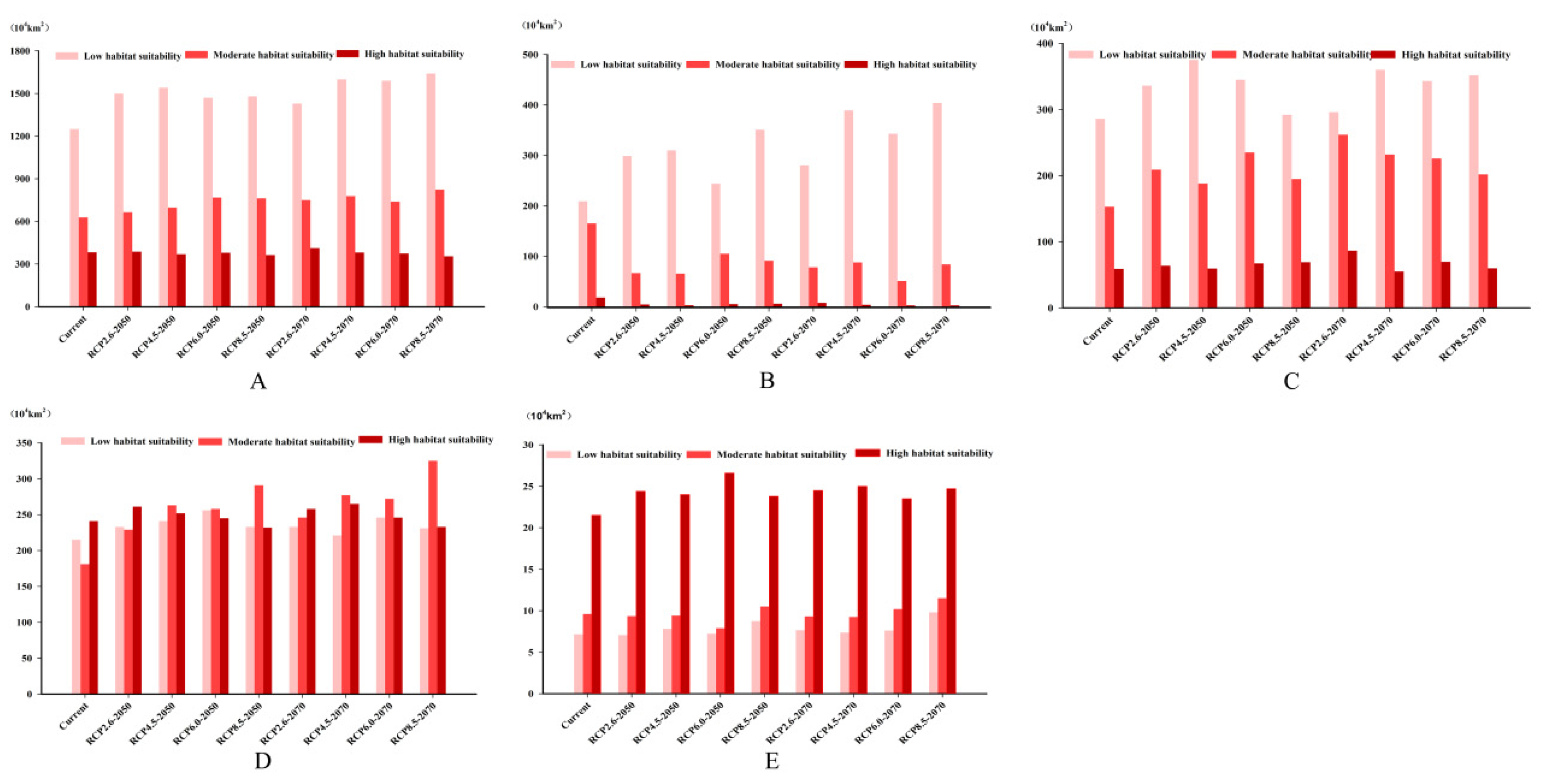
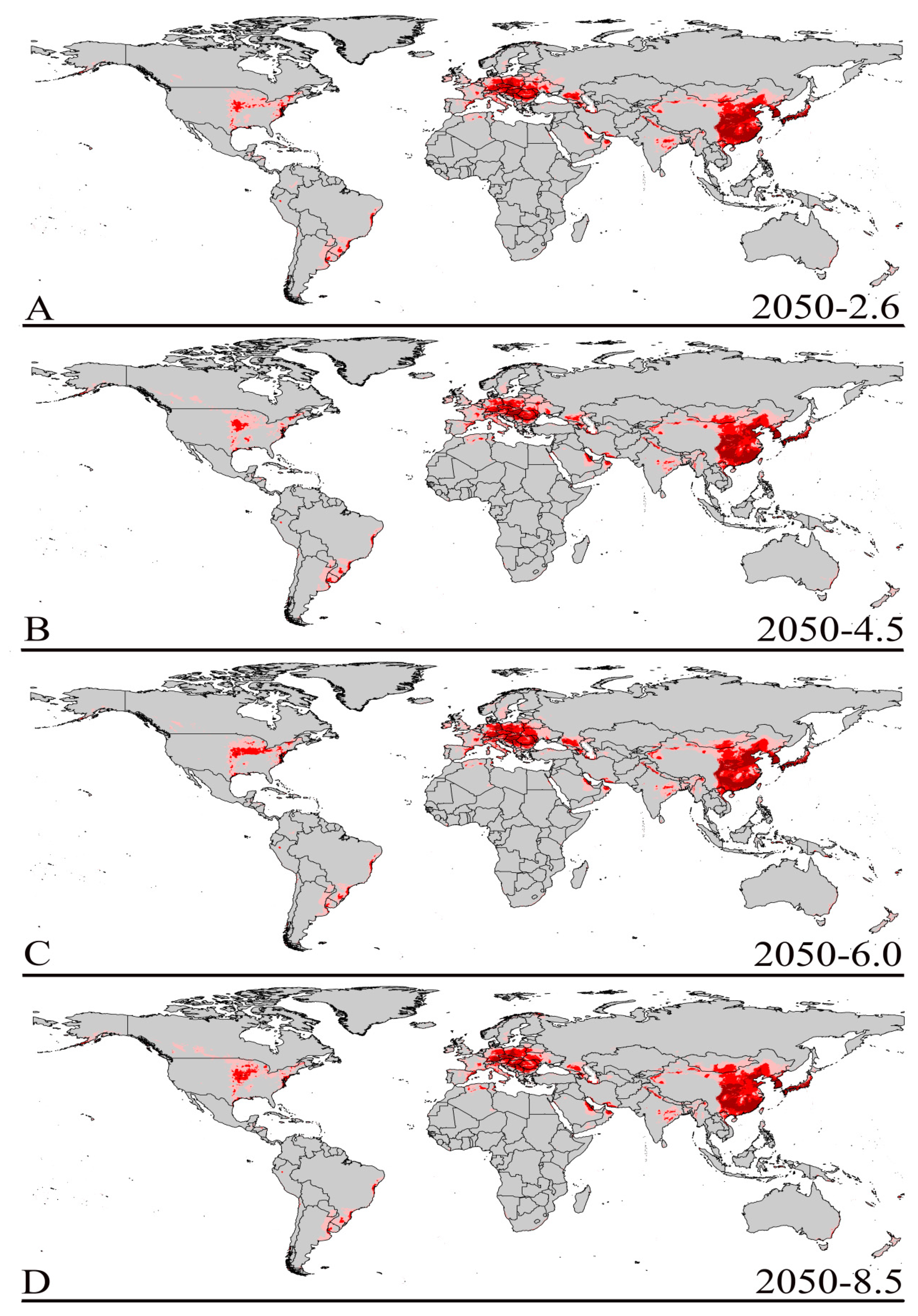
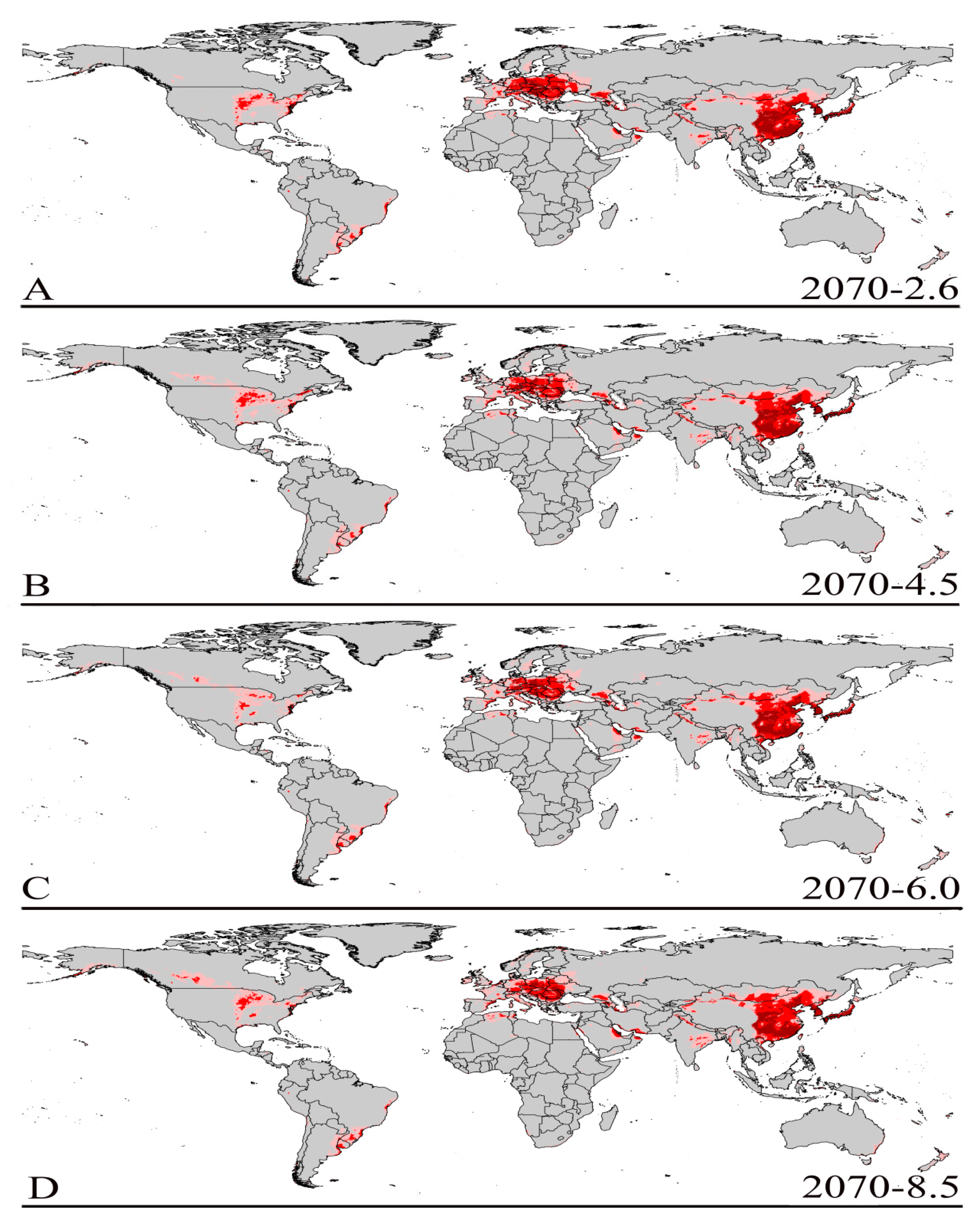
3.3. Future Climate Predictions
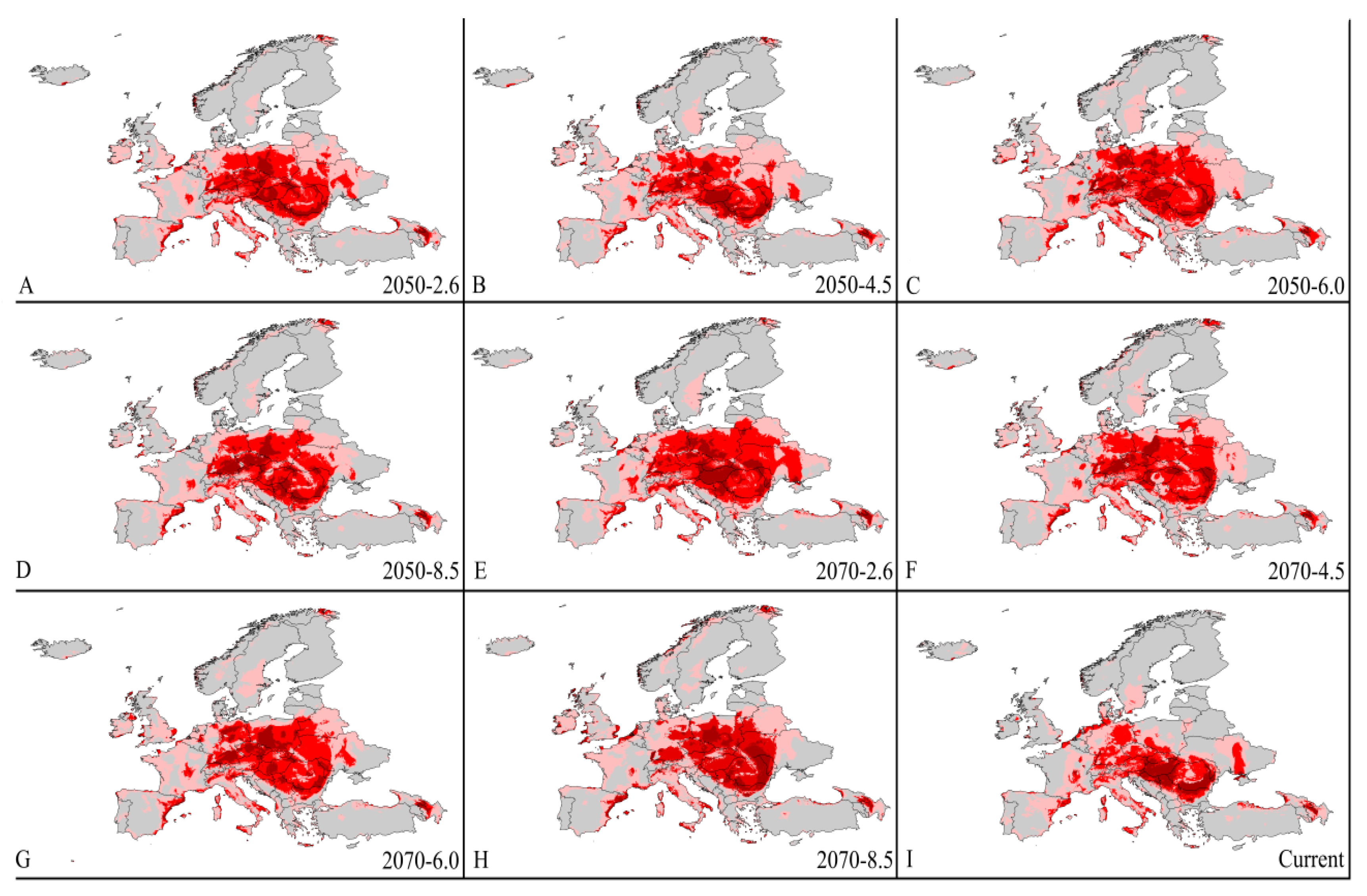
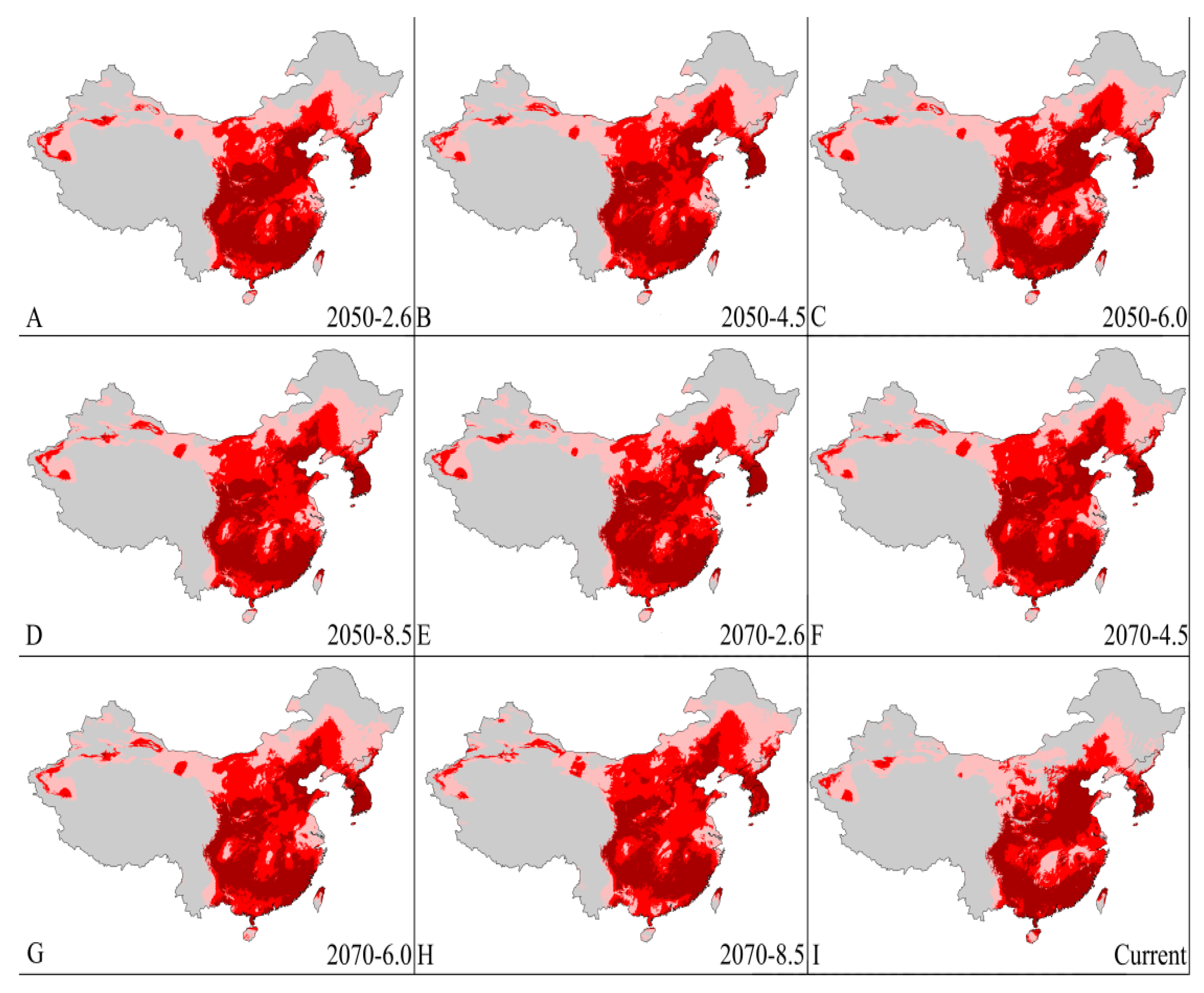
3.4. Four Regions Future Climate Predictions
3.4.1. North America
3.4.2. Europe
3.4.3. China and Korean Peninsula
3.4.4. Japan
3.5. Performance for the Predictive Model and Key Factors for Creating a Suitable Place
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adriaens, T.; Sutton-Croft, M.; Owen, K.; Brosens, D.; Valkenburg, J.V.; Kilbey, D.; Groom, Q.; Ehmig, C.; Thürkow, F.; Hende, P.D.; et al. Trying to engage the crowd in recording invasive alien species in Europe: Experiences from two smartphone applications in northwest Europe. Manag. Biol. Invasions 2015, 6, 215–225. [Google Scholar] [CrossRef]
- Kiritani, K. Invasive Insect Pests and Plant Quarantine in Japan; Food & Fertilizer Technology Center: Taipei, Taiwan, 2001. [Google Scholar]
- Schei, P.J. Conclusions and recommendations from the UN/Norway conference on alien species. Sci. Int. 1996, 63, 32–36. [Google Scholar]
- Liebhold, A.M.; Berec, L.; Brockerhoff, E.G.; Epanchin-Niell, R.S.; Hastings, A.; Herms, D.A.; Kean, J.M.; Mccullough, D.D.; Suckling, D.M.; Tobin, P.C.; et al. Eradication of invading insect populations: From concepts to applications. Annu. Rev. Entomol. 2016, 61, 335–352. [Google Scholar] [CrossRef] [PubMed]
- Paini, D.R.; Sheppard, A.W.; Cook, D.C.; De Barro, P.J.; Worner, S.P.; Thomas, M.B. Global threat to agriculture from invasive species. Proc. Natl. Acad. Sci. USA 2016, 113, 7575–7579. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.R.; Miller, G.L.; Hodges, G.S.; Davidson, J.A. Introduced scale insects (Hemiptera: Coccoidea) of the United States and their impact on US agriculture. Proc. Entomol. Soc. Wash. 2005, 107, 123–158. [Google Scholar]
- Morales, M.G.; Denno, B.D.; Miller, D.R.; Miller, G.L.; Ben-Dov, Y.; Hardy, N.B. ScaleNet: A literature-based model of scale insect biology and systematics. Database 2016, 2016, article ID bav118. [Google Scholar] [CrossRef]
- Toorani, A.H.; Abbasipour, H.; Dehghan-Dehnavi, L. Biodiversity and population fluctuations of parasitoids of the white peach scale, Pseudaulacaspis pentagona (Targioni-Tozzetti) (Hemiptera: Diaspididae), in kiwifruit orchards in Northern Iran. Hell. Plant Prot. J. 2019, 12, 12–21. [Google Scholar] [CrossRef]
- Howard, L.O. The activity of Prospaltella berlesei Howard against Diaspis pentagona Targ. J. Econ. Entomol. 1912, 5, 325–328. [Google Scholar]
- Mohammed, E.M.A.M.; Ülgentürk, S.; Uygun, N.; Garonna, A.P.; Szentkirályi, F.; Fent, M.; Hayat, M. The distribution, host plants and natural enemies of white peach scale, Pseudaulacaspis pentagona (Targioni-Tozzetti) (Hemiptera: Diaspididae) in Ankara province. Munis Entomol. Zool. 2016, 11, 650–656. [Google Scholar]
- Hank, L.M.; Denno, R.F. The white peach scale, Pseudaulacaspis pentagona (Targioni-Tozzetti) (Homoptera: Diaspididae): Life history in Maryland, host plants, and natural enemies. Proc. Entomol. Soc. Wash. 1993, 95, 79–98. [Google Scholar]
- Ülgentürk, S.; Çanakçioğlu, H. Scale insect pests on ornamental plants in urban habitats in Turkey. J. Pest. Sci. 2004, 77, 79–84. [Google Scholar]
- Erkiliç, L.B.; Uygun, N. Development time and fecundity of the white peach scale, Pseudaulacaspis pentagona, in Turkey. Phytoparasitica 1997, 25, 9–16. [Google Scholar] [CrossRef]
- Takeda, M. Effects of temperature on oviposition in overwintering females and hatch in first-generation larvae of Pseudaulacaspis pentagona (Hemiptera: Diaspididae). Appl. Entomol. Zool. 2004, 39, 15–26. [Google Scholar] [CrossRef]
- Kosztarab, M. Armored scale insects: Their biology, natural enemies and control. Econ. Importance 1990, 4, 307–311. [Google Scholar]
- Neumann, G.; Follett, P.A.; Hollingsworth, R.G.; de León, J.H. High host specificity in Encarsia diaspidicola (Hymenoptera: Aphelinidae), a biological control candidate against the white peach scale in Hawaii. Biol. Control. 2010, 54, 107–113. [Google Scholar] [CrossRef]
- Liebregts, W.M.; Sands, D.A.; Bourne, A.S. Population studies and biological control of Pseudaulacaspis pentagona (Targioni-Tozzetti) (Hemiptera: Diaspididae) on passion fruit in Western Samoa. Bull. Entomol. Res. 1989, 79, 163–171. [Google Scholar] [CrossRef]
- Penchevа, A.; Yovkova, M. New data on alien insect pests of ornamental plants in Bulgaria. For. Ideas 2016, 22, 17–33. [Google Scholar]
- Hodgson, C.J.; Lagowska, B. New scale insect (Hemiptera: Sternorrhyncha: Coccoidea) records from Fiji: Three new species, records of several new invasive species and an updated checklist of Coccoidea. Zootaxa 2011, 2766, 1–29. [Google Scholar] [CrossRef]
- Murakami, Y. Review of biology and ecology of Diaspine scales in Japan (Homoptera, Coccoidea). Mushi 1970, 43, 65–114. [Google Scholar]
- Gossard, H.A. Two Peach Scales; Florida Agricultural Experiment Station: Gainesville, FL, USA, 1902. [Google Scholar]
- Meehl, G.; Stoker, T.; Collins, W.; Friedlingstein, P.; Gaye, A.; Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M. Climate Change, 2007: The Physical Science Basis; Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; The Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2007. [Google Scholar]
- Brignall, A.P.; Hossell, J.E.; Rounsevell, M.D.A. Climate change and crop potential in England and Wales. J. R. Agric. Soc. Engl. 1994, 155, 140–161. [Google Scholar]
- Bale, J.S.; Masters, G.J.; Hodkinson, L.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfied, J.; Buse, A.; Conlson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Battisti, A.; Stastny, M.; Netherer, S.; Robinet, C.; Schopf, A.; Roques, A.; Larsson, S. Expansion of geographic range in the pine processionary moth caused by increased winter temperatures. Ecol. Appl. 2005, 15, 2084–2096. [Google Scholar] [CrossRef]
- Williams, D.W.; Liebhold, A.M. Climate change and the outbreak ranges of two North American bark beetles. Agric. For. Entomol. 2002, 4, 87–99. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Guisan, A.; Lehmann, A.; Ferrier, S.; Austin, M.; Overton, J.M.C.; Aspinall, R.; Hastie, T. Making better biogeographical predictions of species’ distributions. J. Appl. Ecol. 2006, 43, 386–392. [Google Scholar] [CrossRef]
- Stockwell, D. The GARP modelling system: Problems and solutions to automated spatial prediction. Int. J. Geogr. Inf. Sci. 1999, 13, 143–158. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M. Modeling of species distributions with Maxent: New extensions and a comprehensive evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Jiménez-Valverde, A.; Peterson, A.T.; Soberón, J.; Overton, J.M.; Aragón, P.; Lobo, J.M. Use of niche models in invasive species risk assessment. Biol. Invasions 2011, 13, 2785–2797. [Google Scholar] [CrossRef]
- Wei, J.F.; Zhang, H.F.; Zhao, W.Q.; Zhao, Q. Niche shifts and the potential distribution of Phenacoccus solenopsis (Hemiptera: Pseudococcidae) under climate change. PLoS ONE 2017, 12, e0180913. [Google Scholar] [CrossRef] [PubMed]
- Hanks, L.M.; Denno, R.F. Local adaptation in the armored scale insect Pseudaulacaspis pentagona (Homoptera: Diaspididae). Ecology 1994, 75, 2301–2310. [Google Scholar] [CrossRef]
- Kramer-Schadt, S.; Niedballa, J.; Pilgrim, J.D.; Schröder, B.; Lindenborn, J.; Reinfelder, V.; Stillfried, M.; Heckmann, I.; Scharf, A.K.; Augeri, D.M.; et al. The importance of correcting for sampling bias in MaxEnt species distribution models. Divers. Distrib. 2013, 19, 1366–1379. [Google Scholar] [CrossRef]
- Abolmaali, S.M.R.; Tarkesh, M.; Bashari, H. MaxEnt modeling for predicting suitable habitats and identifying the effects of climate change on a threatened species, Daphne mucronata, in central Iran. Ecol. Inf. 2018, 43, 116–123. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- Ning, S.Y.; Wei, J.F.; Feng, J.N. Predicting the current potential and future world wide distribution of the onion maggot, Delia antiqua using maximum entropy ecological niche modeling. PLoS ONE 2017, 12, e0171190. [Google Scholar] [CrossRef]
- Woodward, F.I.; Williams, B.G. Climate and plant distribution at global and local scales. Vegetatio 1987, 69, 189–197. [Google Scholar] [CrossRef]
- Nomura, M.; Ishikawa, Y. Biphasic effect of low temperature on completion of winter diapause in the onion maggot, Delia Antiq. J. Insect Physiol. 2000, 46, 373–377. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Yamashita, T.; Nomura, M. Characteristics of summer diapause in the onion maggot, Delia antiqua (Diptera: Anthomyiidae). J. Insect Physiol. 2000, 46, 161–167. [Google Scholar] [CrossRef]
- Sultana, S.; Baumgartner, J.B.; Dominiak, B.C.; Royer, J.E.; Beaumont, L.J. Potential impacts of climate change on habitat suitability for the Queensland fruit fly. Sci. Rep. 2017, 7, 13025. [Google Scholar] [CrossRef]
- Moss, R.H.; Edmonds, J.A.; Hibbard, K.A.; Manning, M.R.; Rose, S.K.; Vuuren, D.P.V.; Carter, T.R.; Emori, S.; Kainuma, M.; Kram, T.; et al. The next generation of scenarios for climate change research and assessment. Nature 2010, 463, 747. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.F.; Li, X.Z.; Lu, Y.Y.; Zhao, L.; Zhang, H.F.; Zhao, Q. Modeling the potential global distribution of Phenacoccus madeirensis green under various climate change scenarios. Forests 2019, 10, 773. [Google Scholar] [CrossRef]
- Graham, C.H.; Hijmans, R.J. A comparison of methods for mapping species ranges and species richness. Glob. Ecol. Biogeogr. 2006, 15, 578–587. [Google Scholar] [CrossRef]
- Wang, R.L.; Li, Q.; He, S.S.; Liu, Y.; Wang, M.T.; Jiang, G. Modeling and mapping the current and future distribution of Pseudomonas syringae pv. actinidiae under climate change in China. PLoS ONE 2018, 13, e0192153. [Google Scholar] [CrossRef] [PubMed]
- Booth, T.H.; Nix, H.A.; Busby, J.R.; Hutchinson, M.F. BIOCLIM: The first species distribution modelling package, its early applications and relevance to most current MAXENT studies. Divers. Distrib. 2013, 20, 1–9. [Google Scholar] [CrossRef]
- Qin, Z.; Zhang, J.E.; DiTommaso, A.; Wang, R.L.; Wu, R.S. Predicting invasions of Wedelia trilobata (L.) Hitchc. with Maxent and GARP models. J. Plant Res. 2015, 128, 763–775. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Dudík, M.; Schapire, R.E.; Blair, M.E. Opening the black box: An open-source release of Maxent. Ecography 2017, 40, 887–893. [Google Scholar] [CrossRef]
- Anderson, R.P. Real vs. artefactual absences in species distributions: Tests for Oryzomys albigularis (Rodentia: Muridae) in Venezuela. J. Biogeogr. 2003, 30, 591–605. [Google Scholar] [CrossRef]
- Muscarella, R.; Galante, P.J.; Soley-Guardia, M.; Boria, R.A.; Kass, J.M.; Uriarte, M.; Anderson, R.P. ENM eval: An R package for conducting spatially independent evaluations and estimating optimal model complexity for Maxent ecological niche models. Methods Ecol. Evol. 2014, 5, 1198–1205. [Google Scholar] [CrossRef]
- Wang, F.; Wang, D.; Guo, G.; Hu, Y.H.; Wei, J.F.; Liu, J.Z. Species delimitation of the Dermacentor ticks based on phylogenetic clustering and niche modeling. PeerJ 2019, 7, e6911. [Google Scholar] [CrossRef]
- Huang, J.; Ling, C.X. Using AUC and accuracy in evaluating learning algorithms. IEEE Trans. Knowl. Data Eng. 2005, 17, 299–310. [Google Scholar] [CrossRef]
- Lobo, J.M.; Jiménez-Valverde, A.; Real, R. AUC: A misleading measure of the performance of predictive distribution models. Glob. Ecol. Biogeogr. 2008, 17, 145–151. [Google Scholar] [CrossRef]
- Miller, D.R.; Davidson, J.A. Armored Scale Insect Pests of Trees and Shrubs (Hemiptera: Diaspididae); Cornell University Press: Ithaca, NY, USA, 2005; pp. 360–361. [Google Scholar]
- Richardson, D.M.; Iponga, D.M.; Roura-Pascual, N.; Krug, R.M.; Milton, S.J.; Hughes, G.O.; Thuiller, W. Accommodating scenarios of climate change and management in modelling the distribution of the invasive tree Schinus molle in South Africa. Ecography 2010, 33, 1049–1061. [Google Scholar] [CrossRef]
- Potgieter, L.J.; Richardson, D.M.; Wilson, J.R.U. Casuarina cunninghamiana in the Western Cape, South Africa: Determinants of naturalisation and invasion, and options for management. S. Afr. J. Bot. 2014, 92, 134–146. [Google Scholar] [CrossRef]
- Barve, N.; Barve, Y.; Jiménez-Valverde, A.; Lira-Noriega, A.; Maher, S.P.; Peterson, A.T.; Soberón, J.; Villalobos, F. The crucial role of the accessible area in ecological niche modeling and species distribution modeling. Ecol. Model. 2011, 222, 1810–1819. [Google Scholar] [CrossRef]
- Chapman, D.; Pescott, O.L.; Roy, H.E.; Tanner, R. Improving species distribution models for invasive non-native species with biologically informed pseudo-absence selection. J. Biogeogr. 2019, 46, 1029–1040. [Google Scholar] [CrossRef]
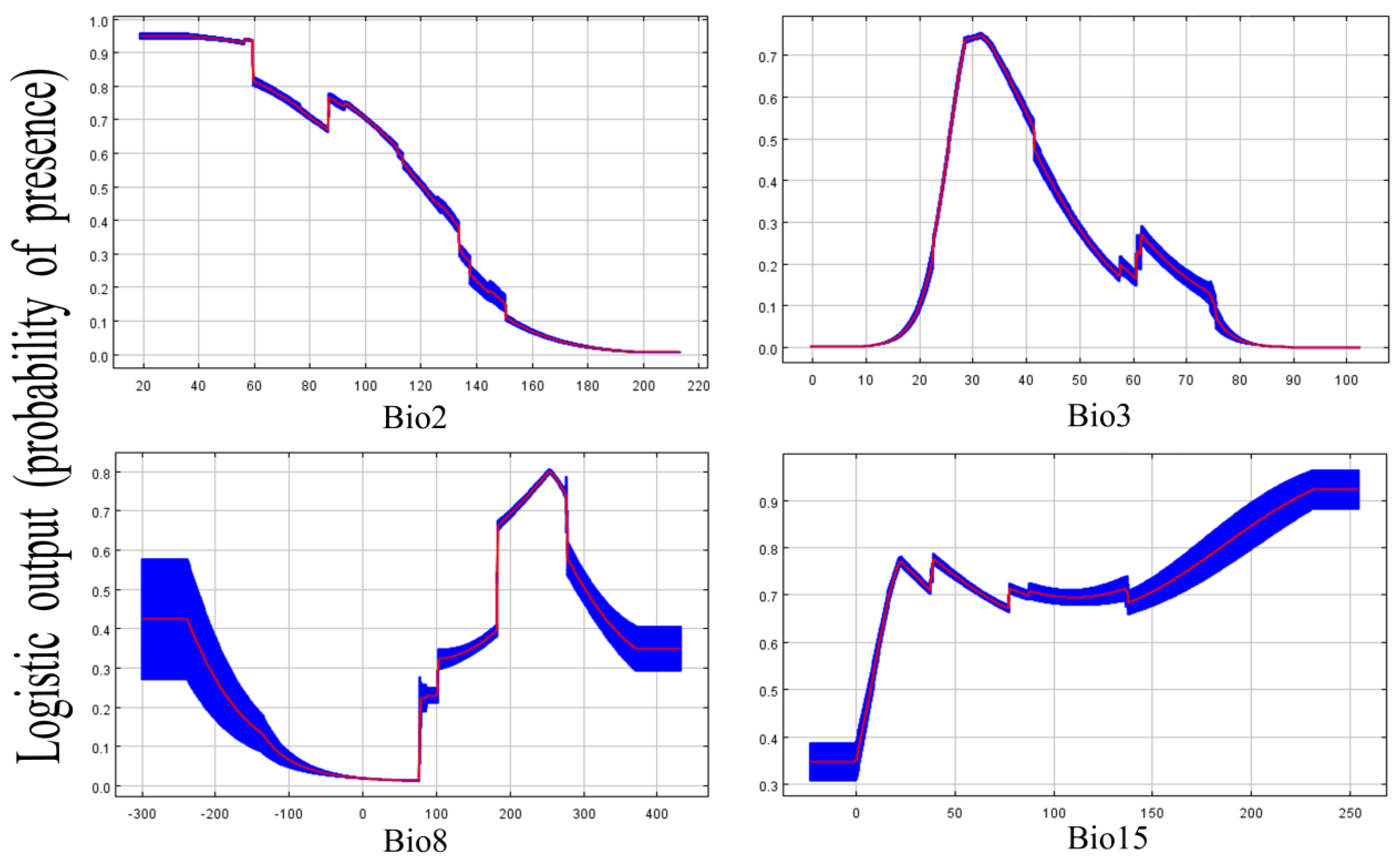


| Environment Variables | Relative Contribution |
|---|---|
| BIO3 | 48.6% |
| BIO8 | 28.8% |
| BIO2 | 20.1% |
| BIO15 | 2.5% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Y.; Zhao, Q.; Cheng, L.; Zhao, L.; Zhang, H.; Wei, J. The Potential Global Distribution of the White Peach Scale Pseudaulacaspis pentagona (Targioni Tozzetti) under Climate Change. Forests 2020, 11, 192. https://doi.org/10.3390/f11020192
Lu Y, Zhao Q, Cheng L, Zhao L, Zhang H, Wei J. The Potential Global Distribution of the White Peach Scale Pseudaulacaspis pentagona (Targioni Tozzetti) under Climate Change. Forests. 2020; 11(2):192. https://doi.org/10.3390/f11020192
Chicago/Turabian StyleLu, Yunyun, Qing Zhao, Lifang Cheng, Ling Zhao, Hufang Zhang, and Jiufeng Wei. 2020. "The Potential Global Distribution of the White Peach Scale Pseudaulacaspis pentagona (Targioni Tozzetti) under Climate Change" Forests 11, no. 2: 192. https://doi.org/10.3390/f11020192
APA StyleLu, Y., Zhao, Q., Cheng, L., Zhao, L., Zhang, H., & Wei, J. (2020). The Potential Global Distribution of the White Peach Scale Pseudaulacaspis pentagona (Targioni Tozzetti) under Climate Change. Forests, 11(2), 192. https://doi.org/10.3390/f11020192





