Plant Diversity Along the Urban–Rural Gradient and Its Relationship with Urbanization Degree in Shanghai, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling Design and Plant Field Survey
2.3. Plant Diversity Index Calculation
2.4. Urbanization Degree Calculation
2.5. Data Analyses
3. Results
3.1. General Plant Species Composition
3.2. The Distribution Pattern of Plant Diversity along the Urban to Rural Gradient
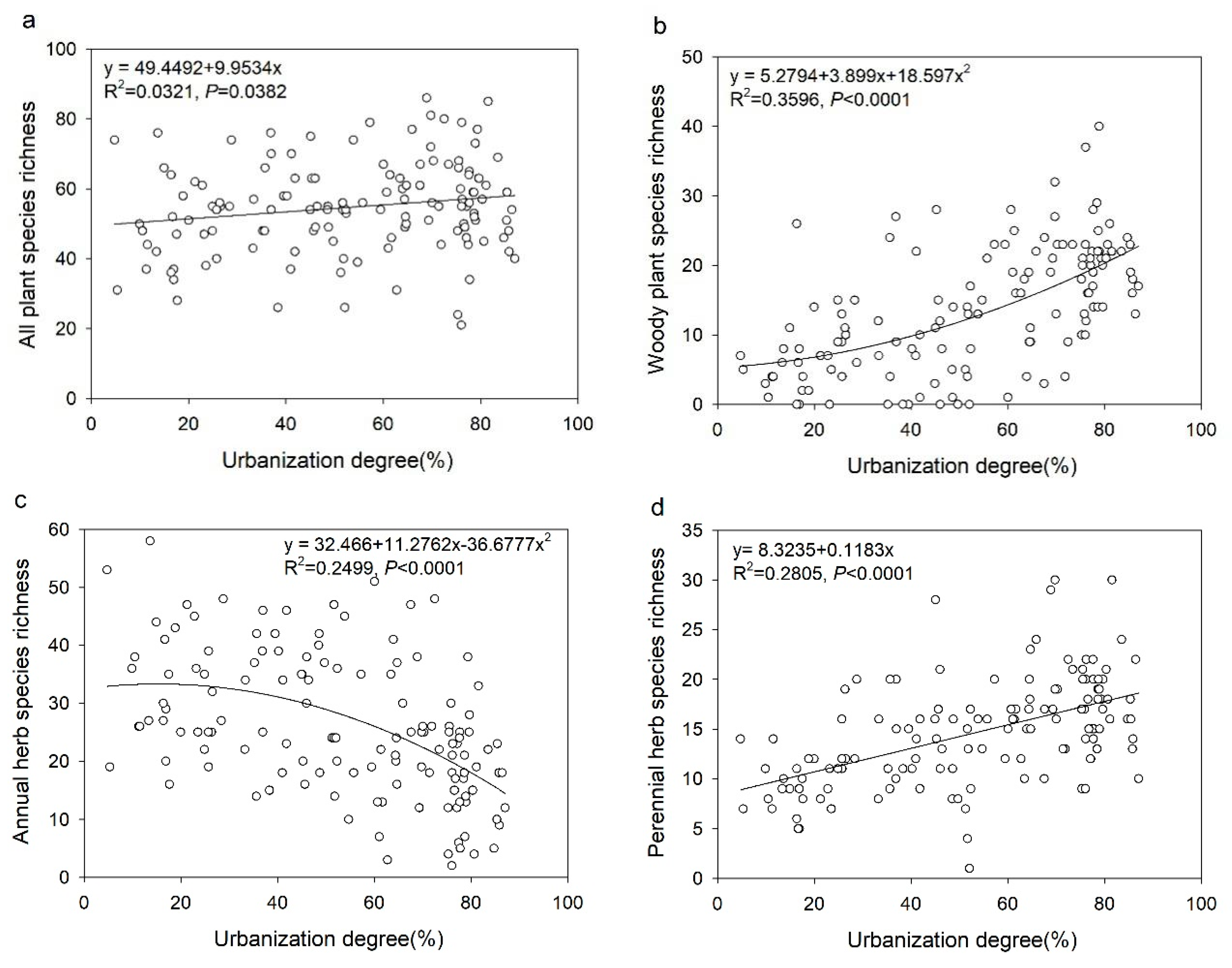
3.3. The Relationship between Plant Diversity and Urbanization Degree
4. Discussion
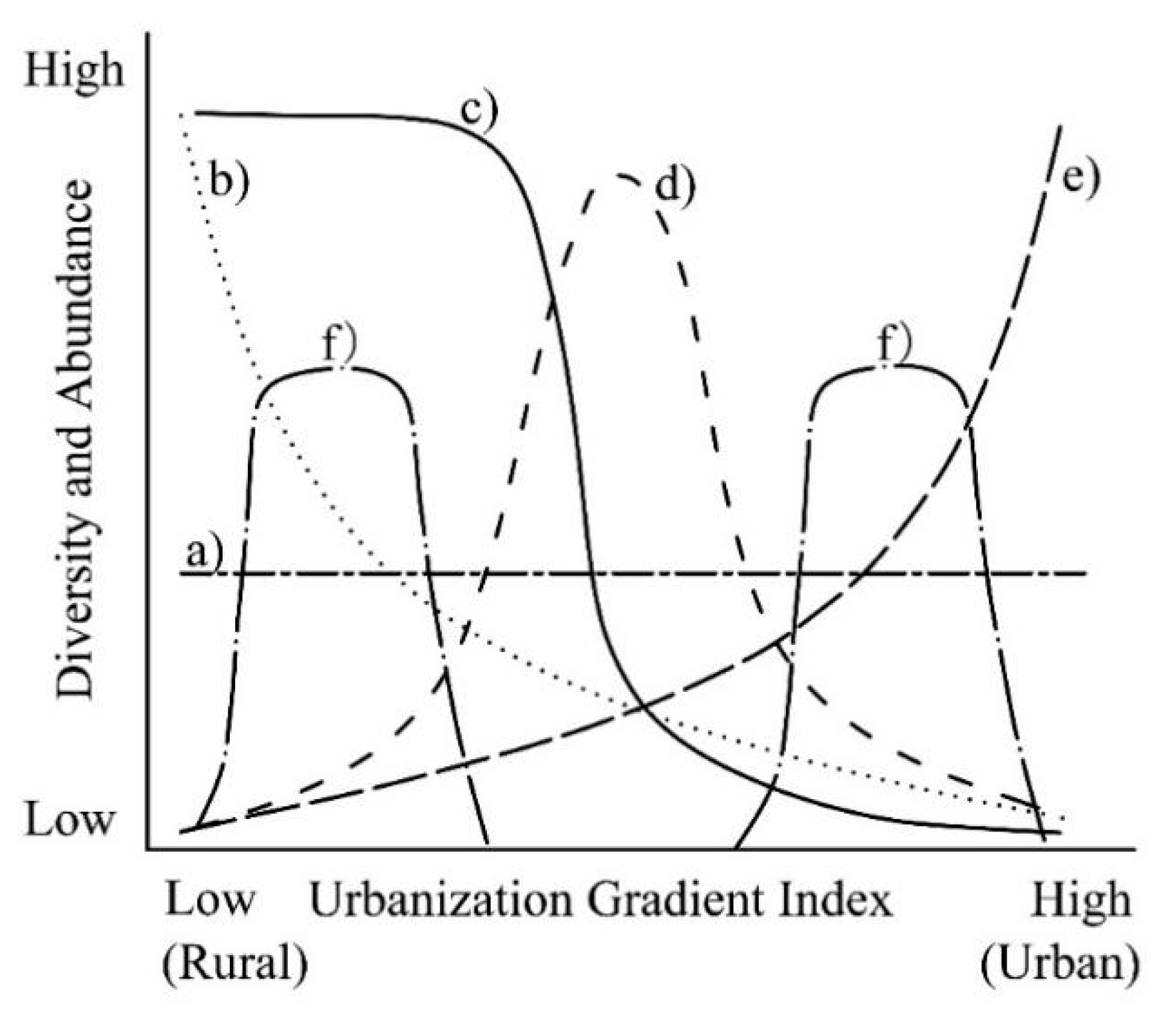
4.1. Multiple Patterns of Plant Species Diversity along the Urban-rural Gradient
4.2. The Impacts of Urbanization on Plant Diversity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- United Nations, Department of Economic and Social Affairs, Population Division. World Population Prospects: The 2017 Revision, Key Findings and Advance Tables; No. ESA/P/WP/248; Working Paper; United Nations, Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2017. [Google Scholar]
- Knapp, S.; Kcohn, I.; Bakker, J.P.; Kleyer, M.; Klotz, S.; Ozinga, W.A.; Poschlod, P.; Thompson, K.; Thuiller, W.; Rcemermann, C. How species traits and affinity to urban land use control large-scale species frequency. Divers. Distrib. 2009, 15, 533–546. [Google Scholar] [CrossRef]
- Ives, C.D.; Lentini, P.E.; Threlfall, C.G.; Ikin, K.; Shanahan, D.F.; Garrard, G.E.; Bekessy, S.A.; Fuller, R.A.; Mumaw, L.; Rayner, L.; et al. Cities are hotspots for threatened species. Glob. Ecol. Biogeogr. 2016, 25, 117–126. [Google Scholar] [CrossRef]
- McKinney, M.L. Effects of urbanization on species richness: A review of plants and animals. Urban Ecosyst. 2008, 11, 161–176. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, H.; Singh, J. Plant diversity in cities: Call for assessment and conservation. Curr. Sci. 2018, 115, 428. [Google Scholar] [CrossRef]
- Soanes, K.; Lentini, P.E. When cities are the last chance for saving species. Front. Ecol. Environ. 2019, 17, 225–231. [Google Scholar] [CrossRef]
- Piana, M.R.; Aronson, M.F.; Pickett, S.T.; Handel, S.N. Plants in the city: Understanding recruitment dynamics in urban landscapes. Front. Ecol. Environ. 2019, 17, 455–463. [Google Scholar] [CrossRef]
- Filazzola, A.; Shrestha, N.; MacIvor, J.S. The contribution of constructed green infrastructure to urban biodiversity: A synthesis and meta-analysis. J. Appl. Ecol. 2019, 56, 2131–2143. [Google Scholar] [CrossRef]
- Mckinney, M.L. Urbanization, Biodiversity, and Conservation. Bioscience 2002, 52, 883–890. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Pickett, S.T.A.; Groffman, P.; Bohlen, P.; Pouyat, R.V.; Zipperer, W.C.; Parmelee, R.W.; Carreiro, M.M.; Medley, K. Ecosystem processes along an urban-to-rural gradient. Urban Ecosyst. 1997, 1, 21–36. [Google Scholar] [CrossRef]
- Vakhlamova, T.; Rusterholz, H.-P.; Kanibolotskaya, Y.; Baur, B. Changes in plant diversity along an urban–rural gradient in an expanding city in Kazakhstan, Western Siberia. Landsc. Urban Plan. 2014, 132, 111–120. [Google Scholar] [CrossRef]
- Ranta, P.; Viljanen, V. Vascular plants along an urban-rural gradient in the city of Tampere, Finland. Urban Ecosyst. 2011, 14, 361–376. [Google Scholar] [CrossRef]
- Lippe, M.V.D.; Kowarik, I. Do cities export biodiversity? Traffic as dispersal vector across urban-rural gradients. Divers. Distrib. 2008, 14, 18–25. [Google Scholar] [CrossRef]
- Tian, Z.; Song, K.; Da, L. Distribution patterns and traits of weed communities along an urban–rural gradient under rapid urbanization in Shanghai, China. Weed Biol. Manag. 2015, 15, 27–41. [Google Scholar] [CrossRef]
- Stenchly, K.; Lippmann, S.; Waongo, A.; Nyarko, G.; Buerkert, A. Weed species structural and functional composition of okra fields and field periphery under different management intensities along the rural-urban gradient of two West African cities. Agric. Ecosyst. Environ. 2017, 237, 213–223. [Google Scholar] [CrossRef]
- Shea, K.; Roxburgh, S.H.; Rauschert, E.S.J. Moving from pattern to process: Coexistence mechanisms under intermediate disturbance regimes. Ecol. Lett. 2004, 7, 491–508. [Google Scholar] [CrossRef]
- Connell, J.H. Intermediate-disturbance hypothesis. Science 1979, 204, 1345. [Google Scholar] [CrossRef]
- Aronson, M.F.; Handel, S.N.; La Puma, I.P.; Clemants, S.E. Urbanization promotes non-native woody species and diverse plant assemblages in the New York metropolitan region. Urban Ecosyst. 2015, 18, 31–45. [Google Scholar] [CrossRef]
- Wang, Y.; Meng, D.; Zhu, Y.; Zhang, F. Impacts of regional urbanization development on plant diversity within boundary of built-up areas of different settlement categories in Jinzhong Basin, China. Landsc. Urban Plan. 2009, 91, 212–218. [Google Scholar] [CrossRef]
- White, R.J.; Carreiro, M.M.; Zipperer, W.C. Woody plant communities along urban, suburban, and rural streams in Louisville, Kentucky, USA. Urban Ecosyst. 2014, 17, 1061–1094. [Google Scholar] [CrossRef]
- Moffatt, S.F.; McLachlan, S.M.; Kenkel, N.C. Impacts of land use on riparian forest along an urban-rural gradient in southern Manitoba. Plant Ecol. 2004, 174, 119–135. [Google Scholar] [CrossRef]
- Zhang, D.; Zheng, H.; He, X.; Ren, Z.; Zhai, C.; Yu, X.; Mao, Z.; Wang, P. Effects of forest type and urbanization on species composition and diversity of urban forest in Changchun, Northeast China. Urban Ecosyst. 2016, 19, 455–473. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, G.; Zhou, Y.; Liu, Q.; Ji, Y.; Wang, S.; Chen, S.; Hui, L. Biodiversity conservation in a fast-growing metropolitan area in China: A case study of plant diversity in Beijing. Biodivers. Conserv. 2007, 16, 4025–4038. [Google Scholar] [CrossRef]
- Schwoertzig, E.; Poulin, N.; Hardion, L.; Trémolières, M. Plant ecological traits highlight the effects of landscape on riparian plant communities along an urban–rural gradient. Ecol. Indic. 2016, 61, 568–576. [Google Scholar] [CrossRef]
- Kühn, I.; Klotz, S. Urbanization and homogenization—comparing the floras of urban and rural areas in Germany. Biol. Conserv. 2006, 127, 292–300. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Hahs, A.K. The use of gradient analysis studies in advancing our understanding of the ecology of urbanizing landscapes: Current status and future directions. Landsc. Ecol. 2008, 23, 1143–1155. [Google Scholar] [CrossRef]
- Ricketts, T.; Imhoff, M. Biodiversity, Urban Areas, and Agriculture: Locating Priority Ecoregions for Conservation. Conserv. Ecol. 2003, 8, 1. [Google Scholar] [CrossRef]
- Heckmann, K.E.; Manley, P.N.; Schlesinger, M.D. Ecological integrity of remnant montane forests along an urban gradient in the Sierra Nevada. For. Ecol. Manag. 2008, 255, 2453–2466. [Google Scholar] [CrossRef]
- Burton, M.L.; Samuelson, L.J. Influence of urbanization on riparian forest diversity and structure in the Georgia Piedmont, US. Plant Ecol. 2008, 195, 99–115. [Google Scholar] [CrossRef]
- Li, J.; Li, C.; Zhu, F.; Song, C.; Wu, J. Spatiotemporal pattern of urbanization in Shanghai, China between 1989 and 2005. Landsc. Ecol. 2013, 28, 1545–1565. [Google Scholar] [CrossRef]
- Shanghai Municipal Statistics Bureau. ShangHai Statistical Yearbook 2017; China Statistics Press: Beijing, China, 2017.
- Zhou, X. The main natural vegetation types of Shanghai and their distribution. Acta Phytoecol. Et Geobot. Sin. 1984, 8, 189–198. [Google Scholar]
- Gao, J. Study on the basic characteristics of natural vegetation, vegetation regionalization and protection of Shanghai. Geogr. Res. 1997, 61, 891–895. [Google Scholar]
- Science and Technology Academy of Shanghai. The Plants of Shanghai; Shanghai Scientific and Technological Literature Publishing House: Shanghai, China, 1999. [Google Scholar]
- Zhao, S.; Da, L.; Tang, Z.; Fang, H.; Song, K.; Fang, J. Ecological consequences of rapid urban expansion: Shanghai, China. Front. Ecol. Environ. 2006, 4, 341–346. [Google Scholar] [CrossRef]
- Hope, D.; Gries, C.; Zhu, W.; Fagan, W.F.; Redman, C.L.; Grimm, N.B.; Nelson, A.L.; Martin, C.; Kinzig, A. Socioeconomics drive urban plant diversity. Proc. Natl. Acad. Sci. USA 2003, 100, 8788–8792. [Google Scholar] [CrossRef]
- Stohlgren, T.J.; Falkner, M.B.; Schell, L.D. A Modified-Whittaker nested vegetation sampling method. Plant Ecol. 1995, 117, 113–121. [Google Scholar] [CrossRef]
- He, R.; Yang, J.; Song, X. Quantifying the Impact of Different Ways to Delimit Study Areas on the Assessment of Species Diversity of an Urban Forest. Forests 2016, 7, 42. [Google Scholar] [CrossRef]
- Stohlgren, T.J.; Coughenour, M.B.; Chong, G.W.; Binkley, D.; Kalkhan, M.A.; Schell, L.D.; Buckley, D.J.; Berry, J.K. Landscape analysis of plant diversity. Landsc. Ecol. 1997, 12, 155–170. [Google Scholar] [CrossRef]
- Rüter, S.; Matthies, S.A.; Zoch, L. Applicability of Modified Whittaker plots for habitat assessment in urban forests: Examples from Hannover, Germany. Urban For. Urban Green. 2017, 21, 116–128. [Google Scholar] [CrossRef]
- Ma, J. The Checklist of Shanghai Vascular Plants; Higher Education Press: Beijing, China, 2013. [Google Scholar]
- Flora of China. Available online: http://www.iplant.cn/foc/ (accessed on 4 February 2020).
- Magurran, A.E. Ecological Diversity and Its Measurement; Princeton University Press: Princeton, NJ, USA, 1988; pp. 81–99. [Google Scholar]
- Wilsey, B.; Stirling, G. Species richness and evenness respond in a different manner to propagule density in developing prairie microcosm communities. Plant Ecol. 2007, 190, 259–273. [Google Scholar] [CrossRef]
- Wilsey, B.J.; Potvin, C. Biodiversity and Ecosystem Functioning: Importance of Species Evenness in an Old Field. Ecology 2000, 81, 887–892. [Google Scholar] [CrossRef]
- Smith, B.; Wilson, J.B. A Consumer’s Guide to Evenness Indices. Oikos 1996, 76, 70–82. [Google Scholar] [CrossRef]
- Jost, L. Entropy and diversity. Oikos 2006, 113, 363–375. [Google Scholar] [CrossRef]
- Macarthur, R.H. Patterns of species diversity. Biol. Rev. 1965, 40, 510–533. [Google Scholar] [CrossRef]
- Whittaker, R.H.; Niering, W.A. Vegetation of the Santa Catalina Mountains, Arizona: A Gradient Analysis of the South Slope. Ecology 1965, 46, 429–452. [Google Scholar] [CrossRef]
- Kowarik, I. Some responses of flora and vegetation to urbanization in central Europe. In Urban Ecology: Plants and Plant Communities in Urban Environments; Sukopp, H., Hejny, S., Kowarik, I., Eds.; SPB Academic Publishing bv: The Hague, The Netherlands, 1990; pp. 45–74. [Google Scholar]
- Mcdonnell, M.J.; Pickett, S.T.A. Ecosystem Structure and Function along Urban-Rural Gradients: An Unexploited Opportunity for Ecology. Ecology 1990, 71, 1232–1237. [Google Scholar] [CrossRef]
- Luck, M.; Wu, J. A gradient analysis of urban landscape pattern: A case study from the Phoenix metropolitan region, Arizona, USA. Landsc. Ecol. 2002, 17, 327–339. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language And Environment for Statistical Computing, Reference Index version 2.2.1. In R Foundation for Statistical Computing; R Development Core Team: Vienna, Austria, 2005. [Google Scholar]
- Hammill, E.; Hawkins, C.P.; Greig, H.S.; Kratina, P.; Shurin, J.B.; Atwood, T.B. Landscape heterogeneity strengthens the relationship between β-diversity and ecosystem function. Ecology 2018, 99, 2467–2475. [Google Scholar] [CrossRef]
- Redon, M.; Bergès, L.; Cordonnier, T.; Luque, S. Effects of increasing landscape heterogeneity on local plant species richness: How much is enough? Landsc. Ecol. 2014, 29, 773–787. [Google Scholar] [CrossRef]
- Fahrig, L.; Baudry, J.; Brotons, L.; Burel, F.G.; Crist, T.O.; Fuller, R.J.; Sirami, C.; Siriwardena, G.M.; Martin, J.-L. Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol. Lett. 2011, 14, 101–112. [Google Scholar] [CrossRef]
- Haines-Young, R. Land use and biodiversity relationships. Land Use Policy 2009, 26, S178–S186. [Google Scholar] [CrossRef]
- Velázquez, A.; Durán, E.; Ramírez, I.; Mas, J.-F.; Bocco, G.; Ramírez, G.; Palacio, J.-L. Land use-cover change processes in highly biodiverse areas: The case of Oaxaca, Mexico. Glob. Environ. Chang. 2003, 13, 175–184. [Google Scholar] [CrossRef]
- Gerstner, K.; Dormann, C.F.; Stein, A.; Manceur, A.M.; Seppelt, R. Effects of land use on plant diversity – A global meta-analysis. J. Appl. Ecol. 2014, 51, 1690–1700. [Google Scholar] [CrossRef]
- Jentsch, A.; Steinbauer, M.J.; Alt, M.; Retzer, V.; Buhk, C.; Beierkuhnlein, C. A systematic approach to relate plant-species diversity to land use diversity across landscapes. Landsc. Urban Plan. 2012, 107, 236–244. [Google Scholar] [CrossRef]
- Luck, G.; Smallbone, L.; O’Brien, R. Socio-Economics and Vegetation Change in Urban Ecosystems: Patterns in Space and Time. Ecosystems 2009, 12, 604–620. [Google Scholar] [CrossRef]
- Martin, C.A.; Warren, P.S.; Kinzig, A.P. Neighborhood socioeconomic status is a useful predictor of perennial landscape vegetation in residential neighborhoods and embedded small parks of Phoenix, AZ. Landsc. Urban Plan. 2004, 69, 355–368. [Google Scholar] [CrossRef]
- Hanspach, J.; Loos, J.; Dorresteijn, I.; Abson, D.J.; Fischer, J. Characterizing social–ecological units to inform biodiversity conservation in cultural landscapes. Divers. Distrib. 2016, 22, 853–864. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, Y.; He, Y.; Wang, M.; Kuang, S.; Chen, G.; Li, J. Effects of socioeconomic factors on urban plant diversity of residential areas in Shanghai, China. J. Appl. Ecol. 2019, 30, 3403–3410. [Google Scholar]
- Kinzig, A.P.; Warren, P.; Martin, C.; Hope, D.; Katti, M. The effects of human socioeconomic status and cultural characteristics on urban patterns of biodiversity. Ecol. Soc. 2005, 10, 23. [Google Scholar] [CrossRef]
- Uchiyama, Y.; Kohsaka, R. Spatio-temporal Analysis of Biodiversity, Land-use Mix and Human Population in a Socio-ecological Production Landscape: A Case Study in the Hokuriku Region, Japan. Procedia Eng. 2017, 198, 219–226. [Google Scholar] [CrossRef]
- Aronson, M.F.J.; Nilon, C.H.; Lepczyk, C.A.; Parker, T.S.; Warren, P.S.; Cilliers, S.S.; Goddard, M.A.; Hahs, A.K.; Herzog, C.; Katti, M.; et al. Hierarchical filters determine community assembly of urban species pools. Ecology 2016, 97, 2952–2963. [Google Scholar] [CrossRef]
- Lopez, B.E.; Urban, D.; White, P.S. Testing the effects of four urbanization filters on forest plant taxonomic, functional, and phylogenetic diversity. Ecol. Appl. 2018, 28, 2197–2205. [Google Scholar] [CrossRef]
- Carvajal-Endara, S.; Hendry, A.P.; Emery, N.C.; Davies, T.J. Habitat filtering not dispersal limitation shapes oceanic island floras: Species assembly of the Galápagos archipelago. Ecol. Lett. 2017, 20, 495–504. [Google Scholar] [CrossRef]
- Jakovac, C.C.; Bongers, F.; Kuyper, T.W.; Mesquita, R.C.G.; Peña-Claros, M. Land use as a filter for species composition in Amazonian secondary forests. J. Veg. Sci. 2016, 27, 1104–1116. [Google Scholar] [CrossRef]
- Poff, N.L. Landscape filters and species traits: Towards mechanistic understanding and prediction in stream ecology. J. North Am. Benthol. Soc. 1997, 16, 391–409. [Google Scholar] [CrossRef]
- Evans, B.S.; Reitsma, R.; Hurlbert, A.H.; Marra, P.P. Environmental filtering of avian communities along a rural-to-urban gradient in Greater Washington, D.C., USA. Ecosphere 2018, 9, e02402. [Google Scholar] [CrossRef]
- Hough-Snee, N.; Laub, B.G.; Merritt, D.M.; Long, A.L.; Nackley, L.L.; Roper, B.B.; Wheaton, J.M. Multi-scale environmental filters and niche partitioning govern the distributions of riparian vegetation guilds. Ecosphere 2015, 6, 1–22. [Google Scholar] [CrossRef]
- Batriu, E.; Ninot, J.M.; Pino, J. Filtering of plant functional traits is determined by environmental gradients and by past land use in a Mediterranean coastal marsh. J. Veg. Sci. 2015, 26, 492–500. [Google Scholar] [CrossRef]
- Kühn, I.; Brandl, R.; Klotz, S. The flora of German cities is naturally species rich. Evol. Ecol. Res. 2004, 6, 749–764. [Google Scholar]
- Mack, R.N.; Lonsdale, W.M. Humans as Global Plant Dispersers: Getting More Than We Bargained For. Bioscience 2001, 51, 95–102. [Google Scholar] [CrossRef]
- Kowarik, I. On the Role of Alien Species in Urban Flora and Vegetation. In Urban Ecology: An International Perspective on the Interaction Between Humans and Nature; Marzluff, J.M., Shulenberger, E., Endlicher, W., Alberti, M., Bradley, G., Ryan, C., Simon, U., ZumBrunnen, C., Eds.; Springer: Boston, MA, USA, 2008; pp. 321–338. [Google Scholar] [CrossRef]
- Wang, H.-F.; MacGregor-Fors, I.; López-Pujol, J. Warm-temperate, immense, and sprawling: Plant diversity drivers in urban Beijing, China. Plant Ecol. 2012, 213, 967–992. [Google Scholar] [CrossRef]
- Knapp, S.; Kühn, I.; Stolle, J.; Klotz, S. Changes in the functional composition of a Central European urban flora over three centuries. Perspect. Plant Ecol. Evol. Syst. 2010, 12, 235–244. [Google Scholar] [CrossRef]
- Clarke, L.W.; Jenerette, G.D.; Davila, A. The luxury of vegetation and the legacy of tree biodiversity in Los Angeles, CA. Landsc. Urban Plan. 2013, 116, 48–59. [Google Scholar] [CrossRef]
- Sukopp, H. Human-caused impact on preserved vegetation. Landsc. Urban Plan. 2004, 68, 347–355. [Google Scholar] [CrossRef]
- Wang, H.; Qin, J.; Zhao, B.; Chen, J.; Dong, L.; Hu, Y. Spatiotemporal dynamics of plant diversity in response to farmers’ evolved settlements in Shanghai. Urban For. Urban Green. 2017, 22, 64–73. [Google Scholar] [CrossRef]
- Knapp, S.; Dinsmore, L.; Fissore, C.; Hobbie, S.E.; Jakobsdottir, I.; Kattge, J.; King, J.Y.; Klotz, S.; McFadden, J.P.; Cavender-Bares, J. Phylogenetic and functional characteristics of household yard floras and their changes along an urbanization gradient. Ecology 2012, 93, S83–S98. [Google Scholar] [CrossRef]
- Walker, J.S.; Grimm, N.B.; Briggs, J.M.; Gries, C.; Dugan, L. Effects of urbanization on plant species diversity in central Arizona. Front. Ecol. Environ. 2009, 7, 465–470. [Google Scholar] [CrossRef]
- Yang, Y.; Fujihara, M.; Li, B.; Yuan, X.; Hara, K.; Da, L.; Tomita, M.; Zhao, Y. Structure and diversity of remnant natural evergreen broad-leaved forests at three sites affected by urbanization in Chongqing metropolis, Southwest China. Landsc. Ecol. Eng. 2014, 10, 137–149. [Google Scholar] [CrossRef]
- Palma, E.; Catford, J.A.; Corlett, R.T.; Duncan, R.P.; Hahs, A.K.; McCarthy, M.A.; McDonnell, M.J.; Thompson, K.; Williams, N.S.G.; Vesk, P.A. Functional trait changes in the floras of 11 cities across the globe in response to urbanization. Ecography 2016, 40, 875–886. [Google Scholar] [CrossRef]
- Seto, K.C.; Güneralp, B.; Hutyra, L.R. Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc. Natl. Acad. Sci. USA 2012, 109, 16083–16088. [Google Scholar] [CrossRef]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.; Bai, X.; Briggs, J.M. Global Change and the Ecology of Cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef]
- Zeeman, B.J.; McDonnell, M.J.; Kendal, D.; Morgan, J.W. Biotic homogenization in an increasingly urbanized temperate grassland ecosystem. J. Veg. Sci. 2017, 28, 550–561. [Google Scholar] [CrossRef]
- McKinney, M.L. Urbanization as a major cause of biotic homogenization. Biol. Conserv. 2006, 127, 247–260. [Google Scholar] [CrossRef]
- Qian, S.; Qi, M.; Huang, L.; Zhao, L.; Lin, D.; Yang, Y. Biotic homogenization of China’s urban greening: A meta-analysis on woody species. Urban For. Urban Green. 2016, 18, 25–33. [Google Scholar] [CrossRef]
- Peng, Y.; Mi, K.; Wang, H.; Liu, Z.; Lin, Y.; Sang, W.; Cui, Q. Most suitable landscape patterns to preserve indigenous plant diversity affected by increasing urbanization: A case study of Shunyi District of Beijing, China. Urban For. Urban Green. 2019, 38, 33–41. [Google Scholar] [CrossRef]
- Cui, Y.C.; Song, K.; Guo, X.Y.; van Bodegom, P.M.; Pan, Y.J.; Tian, Z.H.; Chen, X.S.; Wang, J.; Da, L.J. Phylogenetic and functional structures of plant communities along a spatiotemporal urbanization gradient: Effects of colonization and extinction. J. Veg. Sci. 2019, 30, 341–351. [Google Scholar] [CrossRef]
- Johnson, A.L.; Tauzer, E.C.; Swan, C.M. Human legacies differentially organize functional and phylogenetic diversity of urban herbaceous plant communities at multiple spatial scales. Appl. Veg. Sci. 2015, 18, 513–527. [Google Scholar] [CrossRef]
- Youngsteadt, E.; Dale, A.G.; Terando, A.J.; Dunn, R.R.; Frank, S.D. Do cities simulate climate change? A comparison of herbivore response to urban and global warming. Glob. Chang. Biol. 2015, 21, 97–105. [Google Scholar] [CrossRef]
- Jiang, Z.; Ma, K.; Liu, H.; Tang, Z. A trait-based approach reveals the importance of biotic filter for elevational herb richness pattern. J. Biogeogr. 2018, 45, 2288–2298. [Google Scholar] [CrossRef]
- Wania, A.; Kuhn, I.; Klotz, S. Plant richness patterns in agricultural and urban landscapes in Central Germany—spatial gradients of species richness. Landsc. Urban Plan. 2006, 75, 97–110. [Google Scholar] [CrossRef]
- Hahs, A.K.; Mcdonnell, M.J. Selecting independent measures to quantify Melbourne’s urban–rural gradient. Landsc. Urban Plan. 2006, 78, 435–448. [Google Scholar] [CrossRef]
- Dallimer, M.; Rouquette, J.R.; Skinner, A.M.J.; Armsworth, P.R.; Maltby, L.M.; Warren, P.H.; Gaston, K.J. Contrasting patterns in species richness of birds, butterflies and plants along riparian corridors in an urban landscape. Divers. Distrib. 2012, 18, 742–753. [Google Scholar] [CrossRef]
- Pautasso, M. Scale dependence of the correlation between human population presence and vertebrate and plant species richness. Ecol. Lett. 2007, 10, 16–24. [Google Scholar] [CrossRef]
- Luck, G.W. A review of the relationships between human population density and biodiversity. Biol. Rev. 2007, 82, 607–645. [Google Scholar] [CrossRef]
- Just, M.G.; Dale, A.G.; Long, L.C.; Frank, S.D. Urbanization drives unique latitudinal patterns of insect herbivory and tree condition. Oikos 2019, 128, 984–993. [Google Scholar] [CrossRef]
- Concepción, E.D.; Obrist, M.K.; Moretti, M.; Altermatt, F.; Baur, B.; Nobis, M.P. Impacts of urban sprawl on species richness of plants, butterflies, gastropods and birds: Not only built-up area matters. Urban Ecosyst. 2016, 19, 225–242. [Google Scholar] [CrossRef]
- Nilon, C.H.; Aronson, M.F.J.; Cilliers, S.S.; Dobbs, C.; Frazee, L.J.; Goddard, M.A.; O’Neill, K.M.; Roberts, D.; Stander, E.K.; Werner, P.; et al. Planning for the Future of Urban Biodiversity: A Global Review of City-Scale Initiatives. BioScience 2017, 67, 332–342. [Google Scholar] [CrossRef]
- Manhães, A.P.; Mazzochini, G.G.; Oliveira-Filho, A.T.; Ganade, G.; Carvalho, A.R. Spatial associations of ecosystem services and biodiversity as a baseline for systematic conservation planning. Divers. Distrib. 2016, 22, 932–943. [Google Scholar] [CrossRef]
- Botzat, A.; Fischer, L.K.; Kowarik, I. Unexploited opportunities in understanding liveable and biodiverse cities. A review on urban biodiversity perception and valuation. Glob. Environ. Chang. 2016, 39, 220–233. [Google Scholar] [CrossRef]
- Bourne, K.S.; Conway, T.M. The influence of land use type and municipal context on urban tree species diversity. Urban Ecosyst. 2014, 17, 329–348. [Google Scholar] [CrossRef]
- Martin, Y.; Dyck, H.V.; Dendoncker, N.; Titeux, N.J.G.E. Testing instead of assuming the importance of land use change scenarios to model species distributions under climate change. Glob. Ecol. Biogeogr. 2013, 22, 1204–1216. [Google Scholar] [CrossRef]
- Jankovska, I.; Brūmelis, G.; Nikodemus, O.; Kasparinskis, R.; Amatniece, V.; Straupmanis, G. Tree Species Establishment in Urban Forest in Relation to Vegetation Composition, Tree Canopy Gap Area and Soil Factors. Forests 2015, 6, 4451–4461. [Google Scholar] [CrossRef]





| LULC Type | Subtype of LULC |
|---|---|
| Urban land | Industry land, traffic land, public facility land, residential land, in construction land, etc. |
| Green land | Park, botanic garden, zoo garden, forest |
| Agriculture land | Farmland, orchard, nursery |
| Water | River, lake, pond and wetland |
| Other land | Unused land and Wasted land |
| Variables | Equation | Parameters |
|---|---|---|
| Urbanization (W - E) | y = 73.81 − 3.71x − 0.81x2 + 0.07x3 + 0.006x4 − 2.35e−4x5 − 1.54e−5x6 | R2 = 0.89, p= 2.08e−07 |
| Urbanization (S - N) | y = 74.47 + 0.68x − 0.59x2 − 0.002x3 + 0.002x4 | R2 = 0.86, p = 3.80e−08 |
| All plant - E | y = 54.85 + 11.64x − 4.95x2 + 0.68x3 − 0.03x4 | R2 = 0.79, p = 0.032 |
| Woody plant - W | y = 23.07 + 3.87x + 0.37x2 + 0.01x3 | R2 = 0.74, p = 0.005 |
| Woody plant - E | y = 22.15 − 2.41x + 0.13x2 | R2 = 0.50, p = 0.015 |
| Woody plant - S | y = 20.50 + 1.11x | R2 = 0.48, p = 0.002 |
| Annual herb - W | y = 13.54 − 3.55x − 0.2x2 | R2 = 0.54, p = 0.020 |
| Annual herb - S | y = 14.60 − 4.11x − 0.20x2 | R2 = 0.56, p = 0.003 |
| Annual herb - N | y = 12.82 + 2.54x | R2 = 0.71, p = 0.004 |
| Perennial herb - W | y = 15.31 + 0.53x | R2 = 0.67, p = 0.0006 |
| Perennial herb - E | y = 14.52 + 6.60x − 2.77x2 + 0.35x3 − 0.01x4 | R2 = 0.79, p = 0.032 |
| Perennial herb - S | y = 17.37 + 0.84x + 0.04x2 | R2 = 0.36, p = 0.046 |
| Variables | Equation | Parameters |
|---|---|---|
| Exotic plant - W | y = 25.93 + 2.29x + 0.4x2 + 0.02x3 | R2 = 0.67, p = 0.016 |
| Exotic plant - E | y = 25.73 + 6.95x − 3.14x2 + 0.43x3 − 0.02x4 | R2 = 0.75, p = 0.047 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Li, J.; Kuang, S.; He, Y.; Chen, G.; Huang, Y.; Song, C.; Anderson, P.; Łowicki, D. Plant Diversity Along the Urban–Rural Gradient and Its Relationship with Urbanization Degree in Shanghai, China. Forests 2020, 11, 171. https://doi.org/10.3390/f11020171
Wang M, Li J, Kuang S, He Y, Chen G, Huang Y, Song C, Anderson P, Łowicki D. Plant Diversity Along the Urban–Rural Gradient and Its Relationship with Urbanization Degree in Shanghai, China. Forests. 2020; 11(2):171. https://doi.org/10.3390/f11020171
Chicago/Turabian StyleWang, Meng, Junxiang Li, Shengjian Kuang, Yujuan He, Guojian Chen, Yue Huang, Conghe Song, Pippin Anderson, and Damian Łowicki. 2020. "Plant Diversity Along the Urban–Rural Gradient and Its Relationship with Urbanization Degree in Shanghai, China" Forests 11, no. 2: 171. https://doi.org/10.3390/f11020171
APA StyleWang, M., Li, J., Kuang, S., He, Y., Chen, G., Huang, Y., Song, C., Anderson, P., & Łowicki, D. (2020). Plant Diversity Along the Urban–Rural Gradient and Its Relationship with Urbanization Degree in Shanghai, China. Forests, 11(2), 171. https://doi.org/10.3390/f11020171






