Drivers of the Distribution of Ecological Species Groups in Temperate Deciduous Managed Forests in the Western Carpathian Mountains
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Study Plots
2.3. Vegetation Sampling
2.4. Soil Samplings
2.5. Topographical Variables
2.6. Statistical Analysis
3. Results
3.1. Ecological Species Group
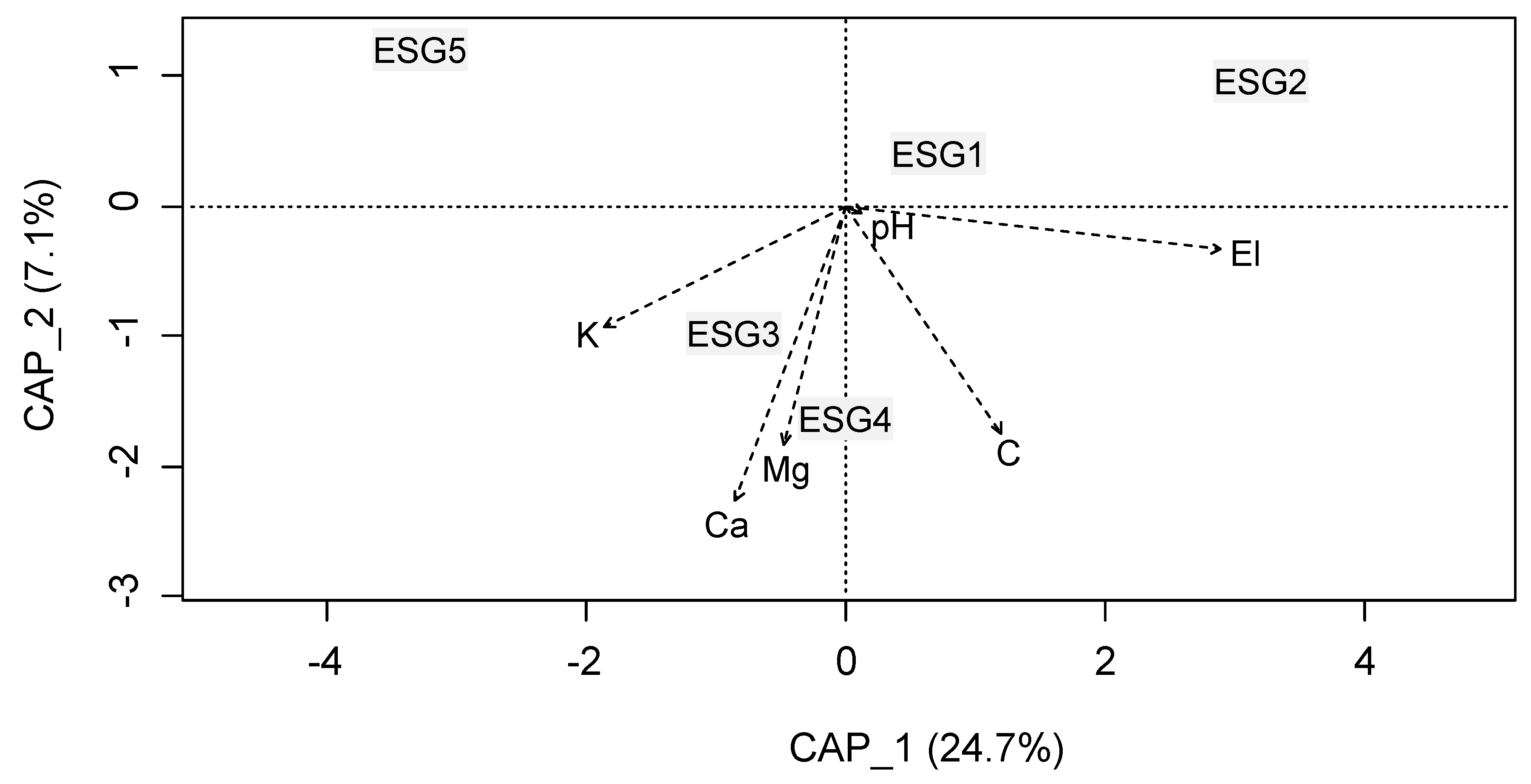
3.2. Ecological Species Group-Environment Relation
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Zn | Fe | Ca | K | Al | Si | S | Mg | C | N | pH | El | Sl | PrC | PlC | TC | TWI | TPI | East | South | SH | SR | MSP | VDChN | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pb | 0.7275 | 0.6911 | −0.0358 | −0.2264 | −0.0807 | −0.3482 | −0.1684 | 0.0367 | 0.6665 | 0.1041 | −0.4277 | 0.7831 | −0.0227 | 0.0269 | −0.0189 | −0.1042 | −0.0848 | 0.1013 | 0.0900 | −0.0253 | 0.1418 | 0.1055 | −0.0297 | 0.2254 |
| Zn | 1.0000 | 0.8751 | 0.2093 | −0.3631 | 0.0169 | −0.1826 | −0.2981 | 0.1204 | 0.4926 | 0.0108 | −0.1334 | 0.5964 | 0.0491 | −0.1022 | −0.0600 | 0.0026 | 0.0503 | 0.0384 | −0.0387 | −0.0700 | 0.2565 | 0.0082 | −0.0377 | 0.1391 |
| Fe | 1.0000 | 0.1418 | −0.2970 | 0.0808 | −0.2647 | −0.2509 | 0.1389 | 0.5087 | −0.0400 | −0.1554 | 0.6055 | 0.0551 | −0.0553 | −0.0232 | 0.0033 | 0.0737 | −0.0382 | −0.0625 | −0.1080 | 0.1648 | 0.0514 | −0.0043 | 0.1182 | |
| Ca | 1.0000 | 0.3450 | 0.4706 | −0.4892 | 0.4025 | 0.3078 | 0.2603 | −0.0904 | 0.4103 | −0.3189 | 0.1113 | −0.0383 | 0.0444 | 0.2571 | −0.0239 | 0.1373 | −0.1349 | −0.1941 | 0.0479 | −0.1371 | −0.0149 | 0.1075 | ||
| K | 1.0000 | 0.6300 | −0.4624 | 0.9661 | 0.4440 | 0.0347 | 0.0128 | 0.1833 | −0.3549 | −0.1377 | 0.0409 | 0.0859 | 0.1680 | −0.0252 | 0.1179 | 0.1195 | −0.0447 | −0.2772 | −0.0310 | 0.0449 | 0.0460 | |||
| Al | 1.0000 | −0.1477 | 0.7134 | 0.7081 | 0.1094 | −0.2373 | 0.1513 | −0.3384 | 0.0908 | −0.0057 | 0.0651 | 0.1669 | 0.0231 | 0.1243 | 0.0407 | −0.0359 | 0.0212 | 0.0025 | −0.0717 | 0.0643 | ||||
| Si | 1.0000 | −0.4523 | −0.0188 | −0.5817 | −0.1908 | 0.0467 | −0.1812 | 0.2763 | 0.0371 | −0.1072 | −0.1150 | 0.0753 | −0.1649 | −0.0224 | 0.1390 | 0.2080 | 0.0704 | −0.0917 | −0.1608 | |||||
| S | 1.0000 | 0.5011 | 0.0943 | −0.0219 | 0.1656 | −0.3532 | −0.1160 | 0.0467 | 0.1119 | 0.1927 | −0.0394 | 0.1602 | 0.0995 | −0.0341 | −0.2221 | −0.0373 | 0.0203 | 0.0690 | ||||||
| Mg | 1.0000 | 0.0468 | −0.0937 | 0.1158 | −0.1841 | 0.1099 | −0.0973 | 0.0012 | 0.1108 | 0.0163 | −0.0139 | −0.0140 | −0.0092 | 0.0207 | 0.0521 | −0.0562 | 0.0413 | |||||||
| C | 1.0000 | 0.1012 | −0.4576 | 0.4951 | −0.0988 | 0.0682 | 0.1234 | 0.0560 | −0.0290 | 0.1776 | 0.0651 | −0.0535 | 0.0501 | 0.0525 | −0.0167 | 0.2388 | ||||||||
| N | 1.0000 | −0.1498 | 0.1807 | −0.3186 | −0.1921 | −0.0626 | −0.1096 | −0.1757 | −0.0802 | 0.0473 | −0.0439 | −0.2707 | −0.1202 | 0.2557 | 0.1083 | |||||||||
| pH | 1.0000 | −0.5175 | 0.1485 | −0.0507 | −0.0694 | 0.2624 | −0.0768 | −0.0129 | −0.1274 | −0.1848 | 0.0072 | −0.2584 | 0.0814 | −0.0787 | ||||||||||
| El | 1.0000 | −0.1397 | 0.0272 | −0.0778 | −0.2232 | 0.0562 | 0.0579 | 0.0825 | 0.0557 | 0.1585 | 0.2818 | −0.0150 | 0.1520 | |||||||||||
| Sl | 1.0000 | −0.0079 | −0.1276 | 0.3536 | −0.1355 | −0.2181 | −0.1003 | −0.1127 | 0.1903 | −0.1513 | −0.1281 | −0.0240 | ||||||||||||
| PrC | 1.0000 | 0.2376 | −0.0903 | −0.0057 | 0.2901 | 0.0811 | 0.0318 | 0.1891 | 0.0734 | 0.0301 | 0.1084 | |||||||||||||
| PlC | 1.0000 | −0.1194 | −0.1191 | 0.1554 | −0.0844 | 0.0488 | 0.0900 | 0.0174 | −0.0200 | 0.0223 | ||||||||||||||
| TC | 1.0000 | −0.1813 | −0.1406 | −0.0162 | −0.0140 | −0.1918 | −0.2956 | 0.0095 | 0.0159 | |||||||||||||||
| TWI | 1.0000 | −0.0938 | 0.0166 | −0.1005 | 0.0266 | 0.3862 | −0.1172 | −0.1858 | ||||||||||||||||
| TPI | 1.0000 | −0.1133 | 0.2313 | 0.5728 | 0.0802 | 0.0763 | 0.6302 | |||||||||||||||||
| East | 1.0000 | 0.0709 | −0.1787 | 0.2024 | −0.1752 | −0.0976 | ||||||||||||||||||
| Sout | 1.0000 | 0.0727 | 0.0785 | 0.0268 | −0.0058 | |||||||||||||||||||
| SH | 1.0000 | 0.1088 | −0.0846 | 0.4594 | ||||||||||||||||||||
| SR | 1.0000 | −0.2085 | −0.0650 | |||||||||||||||||||||
| MSP | 1.0000 | 0.1340 |
References
- Solon, J.; Degórski, M.; Roo-Zielińska, E. Vegetation response to a topographical-soil gradient. Catena 2007, 71, 309–320. [Google Scholar] [CrossRef]
- Oprsal, Z.; Harmacek, J.; Pavlik, P.; Machar, I. What Factors can Influence the Expansion of Protected Areas around the World in the Context of International Environmental and Development Goals? Probl. Ekorozw. 2018, 13, 145–157. [Google Scholar]
- Pechanec, V.; Machar, I.; Pohanka, T.; Opršal, Z.; Petrovič, F.; Švajda, J.; Šálek, L.; Chobot, K.; Filippovova, J.; Cudlín, P.; et al. Effectiveness of Natura 2000 system for habitat types protection: A case study from the Czech Republic. Nat. Conserv. 2018, 24, 21–41. [Google Scholar] [CrossRef]
- Machar, I. Conservation and Management of Floodplain Forests in the Protected Landscape Area Litovelske Pomoravi (Czech Republic) Introduction. In Conservation and Management of Floodplain Forests in the Protected Landscape Area Litovelske Pomoravi (Czech Republic); Machar, I., Ed.; Palacky University: Olomouc, Czech Republic, 2009; pp. 7–108, Accession Number: WOS: 000331015800001; ISBN 978-80-244-2355-5. [Google Scholar]
- Leuschner, C.; Meier, I.C.; Hertel, D. On the niche breadth ofFagus sylvatica: Soil nutrient status in 50 Central European beech stands on a broad range of bedrock types. Ann. For. Sci. 2006, 63, 355–368. [Google Scholar] [CrossRef]
- Kilianová, H.; Pechanec, V.; Brus, J.; Kirchner, K.; Machar, I. Analysis of the development of land use in the Morava River floodplain, with special emphasis on the landscape matrix. Morav. Geogr. Rep. 2017, 25, 46–59. [Google Scholar]
- Chmura, D. The slope aspect affects the heterogenity and growth of ground vegetation in deciduous forest. Pol. J. Ecol. 2008, 56, 463–470. [Google Scholar]
- Langenbruch, C.; Helfrich, M.; Flessa, H. Effects of beech (Fagus sylvatica), ash (Fraxinus excelsior) and lime (Tilia spec.) on soil chemical properties in a mixed deciduous forest. Plant Soil 2012, 352, 389–403. [Google Scholar] [CrossRef]
- Slezak, M.; Hrivnak, R.; Machava, J. Environmental controls of plant species richness and species composition in black alder floodplain forests of central Slovakia. Tuexenia 2017, 37, 79–94. [Google Scholar] [CrossRef]
- Simon, J.; Machar, I.; Brus, J.; Pechanec, V. Combining a growth-simulation model with acoustic-wood tomography as a decision-support tool for adaptive management and conservation of forest ecosystems. Ecol. Inform. 2015, 30, 309–312. [Google Scholar] [CrossRef]
- Merino, A.; Real, C.; Álvarez-González, J.G.; Rodríguez-Guitián, M.A. Forest structure and C stocks in natural Fagus sylvatica forest in southern Europe: The effects of past management. For. Ecol. Manag. 2007, 250, 206–214. [Google Scholar] [CrossRef]
- Shen, Z.H.; Fang, J.Y.; Chiu, C.A.; Chen, T.Y. The geographical distribution and differentiation of Chinese beech forests and the association with Quercus. Appl. Veg. Sci. 2015, 18, 23–33. [Google Scholar] [CrossRef]
- Cerny, T.; Kopecky, M.; Petrik, P.; Song, J.; Srutek, M.; Valachovic, M.; Altman, J.; Dolezal, J. Classification of Korean forests: Patterns along geographic and environmental gradients. Appl. Veg. Sci. 2015, 18, 5–22. [Google Scholar] [CrossRef]
- Rad, J.E.; Shafiei, A.B. The distribution of ecological species groups in Fagetum communities of Caspian forests: Determination of effective environmental factors. Flora Morphol. Distrib. Funct. Ecol. Plants 2010, 205, 721–727. [Google Scholar] [CrossRef]
- Boyle, M.F.; Abella, S.R.; Shelburne, V.B. An ecosystem classification approach to assessing forest change in the southern Appalachian Mountains. For. Ecol. Manag. 2014, 323, 85–97. [Google Scholar] [CrossRef]
- Hoersch, B.; Braun, G.; Schmidt, U. Relation between landform and vegetation in alpine regions of Wallis, Switzerland. A multiscale remote sensing and GIS approach. Comput. Environ. Urban Syst. 2002, 26, 113–139. [Google Scholar] [CrossRef]
- Machar, I.; Simon, J.; Rejsek, K.; Pechanec, V.; Brus, J.; Kilianova, H. Assessment of Forest Management in Protected Areas Based on Multidisciplinary Research. Forests 2016, 7, 285. [Google Scholar] [CrossRef]
- Brosofske, K.; Chen, J.; Crow, T. Understory vegetation and site factors: Implications for a managed Wisconsin landscape. For. Ecol. Manag. 2001, 146, 75–87. [Google Scholar] [CrossRef]
- Willner, W.; Di Pietro, R.; Bergmeier, E. Phytogeographical evidence for post-glacial dispersal limitation of European beech forest species. Ecography 2009, 32, 1011–1018. [Google Scholar] [CrossRef]
- Machar, I. Attempt to summarize the problems: Is a sustainable management of floodplain forest geobiocenoses possible. In Biodiversity and Target Management of Floodplain Forests in the Morava River Basin (Czech Republic); Machar, I., Ed.; Palacky University: Olomouc, Czech Republic, 2010; pp. 189–226. ISBN 978-80-244-2530-6. [Google Scholar]
- Adel, M.N.; Pourbabaei, H.; Dey, D.C. Ecological species group—Environmental factors relationships in unharvested beech forests in the north of Iran. Ecol. Eng. 2014, 69, 1–7. [Google Scholar] [CrossRef]
- Host, G.E.; Pregitzer, K.S. Ecological species groups for upland forest ecosystems of northwestern Lower Michigan. For. Ecol. Manag. 1991, 43, 87–102. [Google Scholar] [CrossRef]
- Decocq, G.; Aubert, M.; Dupont, F.; Bardat, J.; Wattez-Franger, A.; Saguez, R.; De Foucault, B.; Alard, D.; Delelis-Dusollier, A. Silviculture-driven vegetation change in a European temperate deciduous forest. Ann. For. Sci. 2005, 62, 313–323. [Google Scholar] [CrossRef]
- Hansen, A.; Rotella, J. Abiotic factors. In Maintaining Biodiversity in Forest Ecosystems; Malcolm, L., Hunter, J.R., Eds.; Cambridge University Press: Cambridge, UK, 1999; pp. 161–209. ISBN 0-521-63104-1. [Google Scholar]
- Ivanova, M.; Michaeli, E.; Boltiziar, M.; Fazekasova, D. The analysis of changes ecological stability of landscape in the contrasting region of the mountain range and a lowland. In Proceedings of the International Multidisciplinary Scientific GeoConference: SGEM: Surveying Geology & Mining Ecology Management, Albena, Bulgaria,, 16–22 June 2013. [Google Scholar]
- Belčáková, I. The impact of floodplain vegetation on the erosion-sedimentation processess in fluvisols during flood events. Appl. Ecol. Environ. Res. 2019, 17, 3. [Google Scholar] [CrossRef]
- Thomas, P.A.; Packham, J.R. Nutrients dynamics in different forests. In Ecology of Woodland and Forests; Thomas, P.A., Packham, J.R., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 341–345. ISBN 978-0-521-83452-0. [Google Scholar]
- De Nicola, C.; Fanelli, G.; Testi, A.; Costa, C.; D’Angeli, D.; Pignatti, S. Recovering ability of deciduous Oak Forests after different stages of tree cutting in Central Italy. Rend. Lincei 2017, 28, 53–64. [Google Scholar] [CrossRef]
- Roo-Zelinska, E. Ecological groups of vascular plant species in the herb layer of the pine forests of Northern and Central Europe. Pol. J. Ecol. 2003, 51, 493–506. [Google Scholar]
- Ujhazyova, M.; Ujhazy, K.; Chytry, M.; Williner, W.; Ciliak, M.; Malis, F.; Slezak, M. Diversity of beech forests vegetation it the Eastern Alps, Bohemian Massif and the Western Carpathians. Preslia 2016, 88, 435–457. [Google Scholar]
- Zastocki, D.; Lachowicz, H.; Sadowski, J.; Moskalik, T. Changes in the Assortment and Species Structure of Timber Harvested from the Polish Managed Part of Białowieża Forest. Sustainability 2018, 10, 3279. [Google Scholar] [CrossRef]
- Szymura, T.H.; Murak, S.; Szymura, M.; Raduła, M.W. Changes in forest cover in Sudety Mountains during the last 250 years: Patterns, drivers, and landscape-scale implications for nature conservation. Acta Soc. Bot. Pol. 2018, 87, 3576. [Google Scholar] [CrossRef]
- Machar, I.; Schlossarek, M.; Pechanec, V.; Uradnicek, L.; Praus, L.; Sıvacıoğlu, A. Retention Forestry Supports Bird Diversity in Managed, Temperate Hardwood Floodplain Forests. Forests 2019, 10, 300. [Google Scholar] [CrossRef]
- Schulze, E.D.; Bouriaud, O.; Weber, U.; Roscher, C.; Hessenmoeller, D.; Kroiher, F.; Schall, P. Management breaks the natural productivity-biodiversity relationship in forests and grassland: An opinion. For. Ecosyst. 2018, 5, 3. [Google Scholar] [CrossRef]
- Řepka, R.; Šebesta, J.; Maděra, P.; Vahalík, P. Comparison of the floodplain forest floristic composition of two riparian corridors: Species richness, alien species and the effect of water regime changes. Biologia 2015, 70, 208–217. [Google Scholar] [CrossRef]
- Madera, P.; Machala, M.; Slach, T.; Friedl, M.; Cernušáková, L.; Volarík, D.; Buček, A. Predicted occurrence of ancient coppice woodlands in the Czech Republic. iForest-Biogeosci. For. 2017, 10, 788–795. [Google Scholar] [CrossRef]
- Hresko, J.; Petrovič, F.; Misovicova, R. Mountain landscape archetypes of the Western Carpathians (Slovakia). Biodivers. Conserv. 2015, 24, 3269–3283. [Google Scholar] [CrossRef]
- Mikulova, K.; Stastny, P.; Bochnicek, O.; Borsanyi, P.; Cepcekova, E.; Ondruska, P. Klimatologické Normály Teploty Vzduchu na Slovensku [Climate in Slovakia]; SHMU: Bratislava, Slovensko, 2015; p. 135. [Google Scholar]
- Kovařík, P.; Kutal, M.; Machar, I. Sheep and wolves: Is the occurrence of large predators a limiting factor for sheep grazing in the Czech Carpathians? J. Nat. Conserv. 2014, 22, 479–486. [Google Scholar] [CrossRef]
- Machar, I.; Vozenilek, V.; Simon, J.; Pechanec, V.; Brus, J.; Fulnecek, P.; Vitek, T. Joining of the historical research and future prediction as a support tool for the assessment of management strategy for European beech-dominated forests in protected areas. Nat. Conserv. 2017, 22, 51–78. [Google Scholar] [CrossRef]
- Kimmins, J.P.; Keenan, R.J. The ecological effects of clear-cutting. Environ. Rev. 1993, 1, 121–144. [Google Scholar]
- Braun-Blanquet, J.; Fuller, G.D.; Conard, H.S. Plant Sociology, the Study of Plant Communities, 1st ed.; McGraw-Hill Book Company: New York, NY, USA; London, UK, 1932; p. 476. [Google Scholar]
- Hennkens, S.M.; Schaminée, J.H.J. Turboveg, a comprehensive database management system for vegetation data. J. Veg. Sci. 2001, 12, 589–591. [Google Scholar] [CrossRef]
- Tichý, L. JUICE, software for vegetation classification. J. Veg. Sci. 2002, 13, 451–453. [Google Scholar] [CrossRef]
- Weigel, R.; Gilles, J.; Klisz, M.; Manthey, M.; Kreyling, J. Forest understory vegetation is more related to soil than to climate towards the cold distribution margin of European beech. J. Veg. Sci. 2019, 30, 746–755. [Google Scholar] [CrossRef]
- Kubala-Kukus, A.; Banas, D.; Braziewicz, J.; Dziadowicz, M.; Kopec, E.; Majewska, U.; Mazurek, M.; Pajek, M.; Sobisz, M.; Stabrawa, I.; et al. X-ray spectrometry and X-ray microtomography techniques for soil and geological samples analysis. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 2015, 364, 85–92. [Google Scholar] [CrossRef]
- Parsons, C.; Grabulosa, E.M.; Pili, E.; Floor, G.H.; Román-Ross, G.; Charlet, L. Quantification of trace arsenic in soils by field-portable X-ray fluorescence spectrometry: Considerations for sample preparation and measurement conditions. J. Hazard. Mater. 2013, 262, 1213–1222. [Google Scholar] [CrossRef]
- Svobodova, J.; Vozenilek, V. Relief for models of natural phenomena. In Landscape Modelling: Geographical Space, Transformation and Future Scenarios (Urban and Landscape Perspectives); Anděl, J., Bičík, I., Dostál, P., Shasneshin, S., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 183–196. [Google Scholar]
- Deng, Y.; Chen, X.; Chuvieco, E.; Warner, T.; Wilson, J.P. Multi-scale linkages between topographic attributes and vegetation indices in a mountainous landscape. Remote Sens. Environ. 2007, 111, 122–134. [Google Scholar] [CrossRef]
- Roleček, J.; Tichý, L.; Zelený, D.; Chytrý, M. Modified TWINSPAN classification in which the hierarchy respects cluster heterogeneity. J. Veg. Sci. 2009, 20, 596–602. [Google Scholar] [CrossRef]
- Tichý, L.; Chytrý, M. Statistical determination of diagnostic species for site groups of unequal size. J. Veg. Sci. 2006, 17, 809–818. [Google Scholar] [CrossRef]
- Anderson, M.J.; Willis, T.J. Canonical analysis of principal coordinates: A useful method of contrained ordination for ecology. Ecology 2003, 84, 511–525. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environmenta for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Zlatník, A. Přehled skupin typů geobiocénů původně lesních a křovinných v ČSSR. Zpr. Geogr. Úst. ČSAV 1976, 13, 55–64. [Google Scholar]
- Comission of European Communities. Corine biotopes manual. In Habitats of the European Community; Office for Publications of the EC: Luxembourg, 1991; p. 122. [Google Scholar]
- Davies, C.E.; Moss, D. EUNIS Habitat Classification. In European Topic Centre on Nature Conservation; European Environment Agency: Paris, France, 1997; p. 98. [Google Scholar]
- Miko, L. Nature and landscape protection in the European context. In Ochrana Přírody a Krajiny v České Republice, 1st ed.; Machar, I., Drobilová, L., Eds.; Palacky University Olomouc: Olomouc, Czech Republic, 2012; pp. 43–49. ISBN 978-80-244-3041-6. [Google Scholar]
- Jarolímek, I.; Šibík, J.; Hegedüšová, K.; Janišová, M.; Kliment, J.; Kučera, P.; Májeková, J.; Michálková, D.; Sadloňová, J.; Šibíková, I.; et al. A list of vegetation units of Slovakia. In Diagnostic, Constant and Dominant Species of the Higher Vegetation Units of Slovakia, 1st ed; Jarolimek, I., Sibik, J., Eds.; Veda: Bratislava, Slovakia, 2008; pp. 295–329. [Google Scholar]
- Petrovič, F. Landscape-ecological optimization of hydric potential in foothills region with dispersed settlements—A case study of nová bošáca, slovakia. Appl. Ecol. Environ. Res. 2017, 15, 379–400. [Google Scholar] [CrossRef]
- Súľovský, M.; Falťan, V.; Skokanová, H.; Havlíček, M.; Petrovič, F. Spatial analysis of long-term land-use development in regard to physiotopes: Case studies from the Carpathians. Phys. Geogr. 2017, 25, 1–19. [Google Scholar] [CrossRef]
- Skokanova, H.; Havlicek, M.; Klusacek, P.; Martinat, S. Five military training areas—Five different trajectories of land cover development? Case studies from the Czech Republic. Geogr. Cassoviensis 2017, 11, 201–213. [Google Scholar]
- Salekl, L.; Sivacioglu, A.; Topacoglu, O.; Zahradnile, D.; Jerabkoval, L.; Machar, I. Crowns of Old Remnant Oak Standards. Fresenius Environmental Bulletin 2017, 26, 4023–4032. [Google Scholar]
- Miklós, L.; Izakovičová, Z.; Offertálerová, M.; Miklósová, V. The Institutional Tools of Integrated Landscape Management in Slovakia for Mitigation of Climate Change and Other Natural Disasters. Eur. Countrys. 2017, 9, 647–657. [Google Scholar] [CrossRef]
- Lieskovský, J.; Kaim, D.; Balázs, P.; Boltižiar, M.; Chmiel, M.; Grabska, E.; Király, G.; Konkoly-Gyuró, E.; Kozak, J.; Antalová, K.; et al. Historical land use dataset of the Carpathian region (1819–1980). J. Maps 2018, 14, 644–651. [Google Scholar] [CrossRef]
- Boltiziar, M.; Olah, B.; Gallay, I.; Gallayova, Z. Transformation of the Slovak cultural landscape and its recent trends. In Proceedings of the 17th International Symposium on Landscape Ecology - Landscape and Landscape Ecology, Nitra, Slovakia, 27–29 May 2015. [Google Scholar]
- Munteanu, C.; Kuemmerle, T.; Boltiziar, M.; Lieskovský, J.; Mojses, M.; Kaim, D.; Konkoly-Gyuró, É.; Mackovčin, P.; Műller, D.; Ostapowicz, K.; et al. Nineteenth-century land-use legacies affect contemporary land abandonment in the Carpathians. Reg. Environ. Chang. 2017, 17, 2209–2222. [Google Scholar] [CrossRef]
- Machar, I.; Vozenilek, V.; Kirchner, K.; Vlckova, V.; Bucek, A. Biogeographic model of climate conditions for vegetation zones in Czechia. Geografie 2017, 122, 64–82. [Google Scholar]
- Stanova, V.; Valachovic, M. Katalog Biotopov Slovenska [Catalogue of Habitats in Slovakia]; Daphne: Bratislava, Slovakia, 2002; ISBN 80-89133-00-2. [Google Scholar]
- Slezák, M. Environmental conditions in relation to variability of deciduous forest vegetation — A study from volcanic mountain in central Slovakia. Biologia 2012, 67, 310–322. [Google Scholar]
- Knollova, I.; Chytry, M. Oak-hornbeam forests of the Czech Republic: Geographical and ecological approaches to vegetation classification. Preslia 2004, 76, 291–311. [Google Scholar]
- Fontaine, M.; Aerts, R.; Özkan, K.; Mert, A.; Gülsoy, S.; Süel, H.; Waelkens, M.; Muys, B. Elevation and exposition rather than soil types determine communities and site suitability in Mediterranean mountain forests of southern Anatolia, Turkey. For. Ecol. Manag. 2007, 247, 18–25. [Google Scholar] [CrossRef]
- Wang, J.-J.; Pisani, O.; Lin, L.H.; Lun, O.O.; Bowden, R.D.; Lajtha, K.; Simpson, A.J.; Simpson, M.J. Long-term litter manipulation alters soil organic matter turnover in a temperate deciduous forest. Sci. Total. Environ. 2017, 607, 865–875. [Google Scholar] [CrossRef]
- Havlicek, M.; Skokanova, H.; Sarapatka, B.; Netopil, P.; Pavelkova, R. Development of land use of the kyjovska pahorkatina hilly land. Geogr. Inf. 2018, 22, 103–117. [Google Scholar] [CrossRef]
- Kozová, M.; Dobšinská, Z.; Pauditšová, E.; Tomčíková, I.; Rakytová, I. Network and participatory governance in urban forestry: An assessment of examples from selected Slovakian cities. For. Policy Econ. 2018, 89, 31–41. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R. Species Distribution Models: Ecological Explanation and Prediction Across Space and Time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
- Spulerova, J. The influence of exposition on the succession in a sedge-beech forest. Ekologia 2000, 19, 181–186. [Google Scholar]
- Garamvoelgyi, A.; Hufnagel, L. Impacts f climate change on vegetation distribution no.1. Climate change induced vegetation shifts in the Palearctic region. Appl. Ecol. Environ. Res. 2013, 11, 79–122. [Google Scholar] [CrossRef]
- Naqinezhad, A.; Hamzeh’ee, B.; Attar, F. Vegetation–environment relationships in the alderwood communities of Caspian lowlands, N. Iran (toward an ecological classification). Flora-Morphol. Distrib. Funct. Ecol. Plants 2008, 203, 567–577. [Google Scholar] [CrossRef]
- Moradi, H.; Naqinezhad, A.; Siadati, S.; Yousefi, Y.; Attar, F.; Etemad, V.; Reif, A. Elevational gradient and vegetation-environmental relationships in the central Hyrcanian forests of northern Iran. Nord. J. Bot. 2016, 34, 1–14. [Google Scholar] [CrossRef]
- Fu, B.; Liu, S.; Ma, K.; Zhu, Y.-G. Relationships between soil characteristics, topography and plant diversity in a heterogeneous deciduous broad-leaved forest near Beijing, China. Plant. Soil 2004, 261, 47–54. [Google Scholar] [CrossRef]
- Jafari, M.; Chahouki, M.; Tavili, A.; Azarnivand, H.; Amiri, G. Effective environmental factors in the distribution of vegetation types in Poshtkouh rangelands of Yazd Province (Iran). J. Arid. Environ. 2004, 56, 627–641. [Google Scholar] [CrossRef]
- He, M.; Zheng, J.; Li, X.; Qian, Y. Environmental factors affecting vegetation composition in the Alxa Plateau, China. J. Arid. Environ. 2007, 69, 473–489. [Google Scholar] [CrossRef]

| No. of ESG | I | II | III | IV | V |
|---|---|---|---|---|---|
| No. of relevés | 29 | 48 | 10 | 19 | 44 |
| Fagus sylvatica | 10036,6 | 9834,5 | 10 | 79 | 39 |
| Luzula luzuloides | 3452,0 | − | − | − | 2 |
| Moehringia trinervia | 6638,7 | 2 | − | 5 | 7751,5 |
| Tithymalus amyglaoides | 3127,8 | − | 10 | 11 | 11 |
| Dentaria bulbifera | 5521,0 | 6329,4 | 10 | 47 | − |
| Acer pseudoplatanus | − | 2214,6 | 20 | 21 | − |
| Acer campestre | 3 | 2 | 3021,2 | 3730,8 | 2 |
| Carpinus betulus | − | − | 9054,8 | 32 | 6427,5 |
| Quercus petraea agg. | 3 | 4 | 8048,8 | 32 | 5017,1 |
| Tilia Cordata | − | − | 6035,3 | 7956,3 | 2 |
| Galium sylvaticum agg. | 3 | − | 5063,6 | − | − |
| Hedera helix | − | − | 3019,2 | 4743,0 | 2 |
| Impatiens parviflora | 3 | 6 | 8033,5 | 8437,7 | 5912,5 |
| Melica uniflora | − | − | 10066,5 | 7439,1 | 7 |
| Galium aparine agg. | − | − | 4023,4 | 5845,4 | 7 |
| Fraxinus excelsior | − | 2 | − | 4256,5 | 2 |
| Corydalis cava | − | − | 10 | 6368,7 | − |
| Galium odoratum | 10 | − | 10 | 4730,3 | 4325,3 |
| Geranium robertianum agg. | 7 | − | 50 | 5322,2 | 5019,4 |
| Ficaria bulbifera | 10 | − | − | 4731,4 | 5034,6 |
| Alliaria petiolata | − | − | 50 | 5322,3 | 5726,7 |
| Polygonatum multiflorum | − | − | 10 | 4251,9 | − |
| Anemone ranunculoides | − | 8 | − | 1625,8 | − |
| Calystegia sepium agg. | 7 | 2 | 30 | 32 | 6139,7 |
| Viola reichenbachiana | 10 | − | − | 11 | 1819,3 |
| Mycelis muralis | 10 | − | − | 26 | 4539,2 |
| Cerasus avium | 7 | − | − | 5 | 1622,4 |
| Carex sylvatica | − | 4 | 10 | − | 1114,3 |
| Urtica dioica | 41 | 2 | 20 | 11 | 7549,4 |
| Rubus fruticosus agg. | 17 | 14 | − | 11 | 5040,8 |
| Poa nemoralis agg. | 3 | − | − | 5 | 1625,4 |
| ESG 1 | ESG 2 | ESG 3 | ESG 4 | ESG 5 | F-Ratio | p-Value | |
|---|---|---|---|---|---|---|---|
| Ca (%) | 0.412 ± 0.069 | 0.374 ± 0.053 | 0.653 ± 0.117 | 0.572 ± 0.085 | 0.358 ± 0.059 | 2.273 | 0.064 |
| K (%) | 1.494 ± 0.051 | 1.377 ± 0.039 | 1.814 ± 0.085 | 1.692 ± 0.062 | 1.716 ± 0.043 | 12.712 | <0.05 |
| Mg (%) | 0.595 ± 0.031 | 0.577 ± 0.023 | 0.826 ± 0.052 | 0.723 ± 0.038 | 0.615 ± 0.026 | 6.799 | <0.05 |
| C (%) | 1.868 ± 0.199 | 2.435 ± 0.151 | 1.924 ± 0.334 | 2.851 ± 0.242 | 1.509 ± 0.167 | 7.165 | <0.05 |
| pH | 3.723 ± 0.116 | 3.604 ± 0.088 | 3.661 ± 0.195 | 3.498 ± 0.142 | 3.558 ± 0.098 | 0.480 | 0.75 |
| El (m) | 425.752 ± 12.644 | 470.023 ± 9.559 | 287.258 ± 21.159 | 398.017 ± 15.350 | 279.992 ± 10.579 | 52.468 | <0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabor, M.; Beracko, P.; Faltan, V.; Matecny, I.; Karlik, L.; Petrovič, F.; Vallo, D.; Machar, I. Drivers of the Distribution of Ecological Species Groups in Temperate Deciduous Managed Forests in the Western Carpathian Mountains. Forests 2019, 10, 798. https://doi.org/10.3390/f10090798
Gabor M, Beracko P, Faltan V, Matecny I, Karlik L, Petrovič F, Vallo D, Machar I. Drivers of the Distribution of Ecological Species Groups in Temperate Deciduous Managed Forests in the Western Carpathian Mountains. Forests. 2019; 10(9):798. https://doi.org/10.3390/f10090798
Chicago/Turabian StyleGabor, Marian, Pavel Beracko, Vladimir Faltan, Igor Matecny, Lukas Karlik, František Petrovič, Dusan Vallo, and Ivo Machar. 2019. "Drivers of the Distribution of Ecological Species Groups in Temperate Deciduous Managed Forests in the Western Carpathian Mountains" Forests 10, no. 9: 798. https://doi.org/10.3390/f10090798
APA StyleGabor, M., Beracko, P., Faltan, V., Matecny, I., Karlik, L., Petrovič, F., Vallo, D., & Machar, I. (2019). Drivers of the Distribution of Ecological Species Groups in Temperate Deciduous Managed Forests in the Western Carpathian Mountains. Forests, 10(9), 798. https://doi.org/10.3390/f10090798






