Effects on Greenhouse Gas (CH4, CO2, N2O) Emissions of Conversion from Over-Mature Forest to Secondary Forest and Korean Pine Plantation in Northeast China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Soil Gas Flux Measurements
2.3. Determination of Environmental Variables
2.4. Global Warming Potential
2.5. GHG Flux Directions
2.6. Data Analysis
3. Results
3.1. Soil Properties
3.2. Seasonal Variation of GHG Fluxes
3.2.1. CH4 Fluxes
3.2.2. CO2 Fluxes
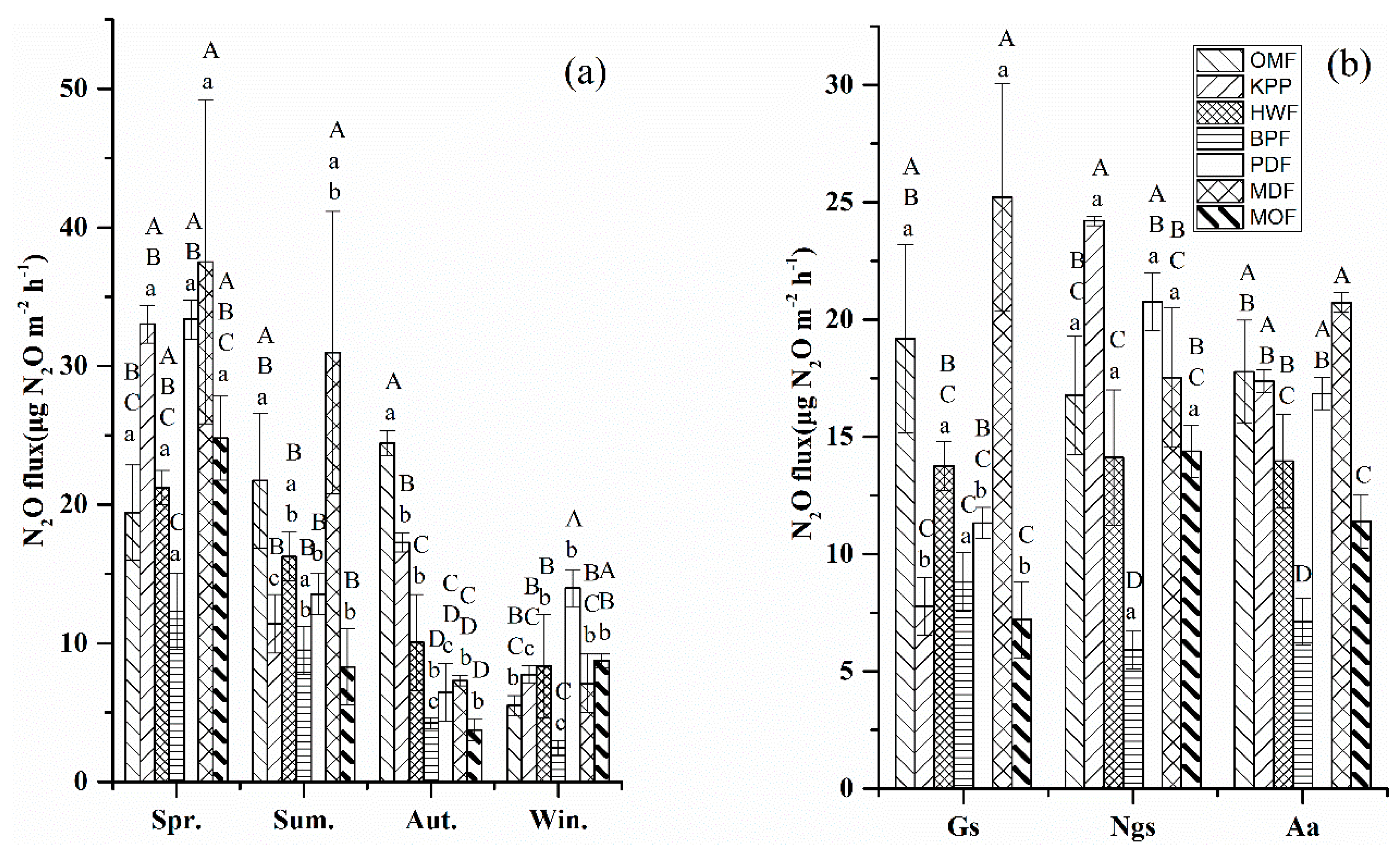
3.2.3. N2O Fluxes
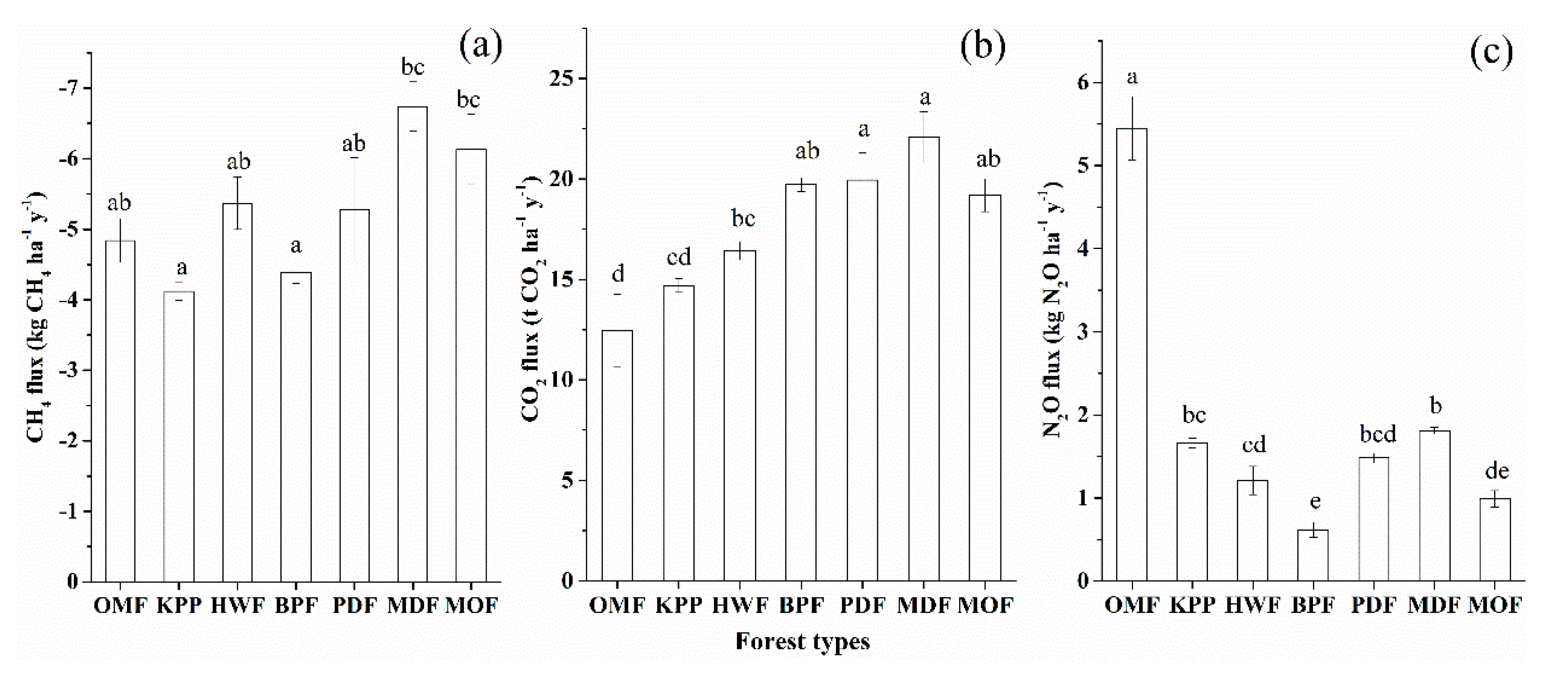
3.3. Estimation of GHG Emissions and GWPs
4. Discussion
4.1. Factors Controlling GHG Fluxes
4.1.1. Methane
4.1.2. Carbon Dioxide
4.1.3. Nitrous Oxide
4.2. Effects of Different Forest Types on GHG Fluxes
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lavoie, M.; Kellman, L.; Risk, D. The effects of clear-cutting on soil CO₂, CH₄, and N₂O flux, storage and concentration in two Atlantic temperate forests in Nova Scotia, Canada. For. Ecol. Manag. 2013, 304, 355–369. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group1 to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Farías, L.; Florez-Leiva, L.; Besoain, V.; Sarthou, G.; Fernández, C. Dissolved greenhouse gases (nitrous oxide and methane) associated with the naturally iron-fertilized Kerguelen region (KEOPS 2 cruise) in the Southern Ocean. Biogeosci. Discuss. 2015, 11, 12531–12569. [Google Scholar] [CrossRef]
- Rowlings, D.W.; Grace, P.R.; Kiese, R.; Weier, K.L. Environmental factors controlling temporal and spatial variability in the soil-atmosphere exchange of CO2, CH4 and N2O from an Australian subtropical rainforest. Glob. Chang. Biol. 2012, 18, 726–738. [Google Scholar] [CrossRef]
- Li, Y.; Dong, S.; Liu, S.; Zhou, H.; Gao, Q.; Cao, G.; Wang, X.; Su, X.; Zhang, Y.; Tang, L.; et al. Seasonal changes of CO2, CH4 and N2O fluxes in different types of alpine grassland in the Qinghai-Tibetan Plateau of China. Soil Biol. Biochem. 2015, 80, 306–314. [Google Scholar] [CrossRef]
- IPCC. Climate Change 1994: Radiative Forcing of Climate Change and an Evaluation of the IPCC IS92 Emissions Scenarios; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1995. [Google Scholar]
- Ehhalt, D.; Prather, M. Atmospheric chemistry and greenhouse gases. In Climate Change 2001: The Scientific Basis; Cambridge University Press: London, UK, 2001; pp. 228–248. [Google Scholar]
- Maljanen, M.; Hytönen, J.; Martikainen, P.J. Fluxes of N2O, CH4 and CO2 on afforested boreal agricultural soils. Plant Soil 2001, 231, 113–121. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Impacts, Adaptation, and Vulnerability Working Group II Contribution to the Fifth Assessment Report; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Mills, R.T.E.; Dewhirst, N.; Sowerby, A.; Emmett, B.A.; Jones, D.L. Interactive effects of depth and temperature on CH4 and N2O flux in a shallow podzol. Soil Biol. Biochem. 2013, 62, 1–4. [Google Scholar] [CrossRef]
- Benanti, G.; Saunders, M.; Tobin, B.; Osborne, B. Contrasting impacts of afforestation on nitrous oxide and methane emissions. Agric. For. Meteorol. 2014, 198–199, 82–93. [Google Scholar] [CrossRef]
- Mishurov, M.; Kiely, G. Nitrous oxide flux dynamics of grassland undergoing afforestation. Agric. Ecosyst. Environ. 2010, 139, 59–65. [Google Scholar] [CrossRef]
- Carmo, J.B.D.; Neto, E.R.D.S.; Duarte-Neto, P.J.; Ometto, J.P.H.B.; Martinelli, L.A. Conversion of the coastal Atlantic forest to pasture: Consequences for the nitrogen cycle and soil greenhouse gas emissions. Agric. Ecosyst. Environ. 2012, 148, 37–43. [Google Scholar] [CrossRef]
- Maljanen, M.; Shurpali, N.; Hytönen, J.; Mäkiranta, P.; Aro, L.; Potila, H.; Laine, J.; Li, C.; Martikainen, P.J. Afforestation does not necessarily reduce nitrous oxide emissions from managed boreal peat soils. Biogeochemistry 2012, 108, 199–218. [Google Scholar] [CrossRef]
- Lagomarsino, A.; Agnelli, A.E.; Pastorelli, R.; Pallara, G.; Rasse, D.P.; Silvennoinen, H. Past water management affected GHG production and microbial community pattern in Italian rice paddy soils. Soil Biol. Biochem. 2015. [Google Scholar] [CrossRef]
- Dou, X.; Zhou, W.; Zhang, Q.; Cheng, X. Greenhouse gas (CO2, CH4, N2O) emissions from soils following afforestation in central China. Atmos. Environ. 2015, 126, 98–106. [Google Scholar] [CrossRef]
- Ding, W.; Cai, Z.; Tsuruta, H. Methane concentration and emission as affected by methane transport capacity of plants in freshwater marsh. Water Air Soil Pollut. 2004, 158, 99–111. [Google Scholar] [CrossRef]
- Sun, X.; Mu, C.; Song, C. Seasonal and spatial variations of methane emissions from montane wetlands in Northeast China. Atmos. Environ. 2011, 45, 1809–1816. [Google Scholar] [CrossRef]
- Tate, K.R. Soil methane oxidation and land-use change–from process to mitigation. Soil Biol. Biochem. 2015, 80, 260–272. [Google Scholar] [CrossRef]
- Konda, R.; Ohta, S.; Ishizuka, S.; Heriyanto, J.; Wicaksono, A. Seasonal changes in the spatial structures of N2O, CO2, and CH4 fluxes from Acacia mangium plantation soils in Indonesia. Soil Biol. Biochem. 2010, 42, 1512–1522. [Google Scholar] [CrossRef]
- Priemé, A.; Christensen, S. Natural perturbations, drying–wetting and freezing–thawing cycles, and the emission of nitrous oxide, carbon dioxide and methane from farmed organic soils. Soil Biol. Biochem. 2001, 33, 2083–2091. [Google Scholar] [CrossRef]
- Wang, C.K.; Yang, J.Y.; Zhang, Q.Z. Soil respiration in six temperate forests in China. Glob. Chang. Biol. 2006, 12, 2103–2114. [Google Scholar] [CrossRef]
- Shi, B.; Gao, W.; Jin, G. Effects on rhizospheric and heterotrophic respiration of conversion from primary forest to secondary forest and plantations in northeast China. Eur. J. Soil Biol. 2015, 66, 11–18. [Google Scholar] [CrossRef]
- Chen, D.K.; Zhou, X.F.; Zhao, H.X.; Wang, Y.H.; Jing, Y.Y. Study on the Structure, Function and Succession of the Four Types in Natural Secondary Forest; Northeast Forestry University Press: Harbin, China, 1982. [Google Scholar]
- Smith, K.A.; Dobbie, K.E.; Ball, B.C.; Bakken, L.R.; Sitaula, B.K.; Hansen, S.; Brumme, R.; Borken, W.; Christensen, S.; Priemé, A.; et al. Oxidation of atmospheric methane in Northern European soils, comparison with other ecosystems, and uncertainties in the global terrestrial sink. Glob. Chang. Biol. 2010, 6, 791–803. [Google Scholar] [CrossRef]
- Wang, C.K. Biomass allometric equations for 10 co-occurring tree species in Chinese temperate forests. For. Ecol. Manag. 2006, 222, 9–16. [Google Scholar] [CrossRef]
- Wang, X.; Wang, C.; Guo, Q.; Wang, J. Improving the CO2 storage measurements with a single profile system in a tall-dense-canopy temperate forest. Agric. For. Meteorol. 2016, 228–229, 327–338. [Google Scholar] [CrossRef]
- Tang, X.; Liu, S.; Zhou, G.; Zhang, D.; Zhou, C. Soil-atmospheric exchange of CO₂, CH₄, and N₂O in three subtropical forest ecosystems in southern China. Glob. Chang. Biol. 2006, 12, 546–560. [Google Scholar] [CrossRef]
- Li, M.; Zhou, X.; Zhang, Q.; Cheng, X. Consequences of afforestation for soil nitrogen dynamics in central China. Agric. Ecosyst. Environ. 2014, 183, 40–46. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2007: Synthesis Report; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Shi, B.; Jin, G. Variability of soil respiration at different spatial scales in temperate forests. Biol. Fertil. Soils 2016, 52, 561–571. [Google Scholar] [CrossRef]
- Liu, S. Effluxes of Soil Carbon Dioxide, Methane and Nitrous Oxide in Four Chinese Temperate Forests. Master’s Thesis, Northeast Forestry University, Harbin, China, 2010. (In Chinese). [Google Scholar]
- Yang, G.; Chen, H.; Wu, N.; Tian, J.; Peng, C.; Zhu, Q.; Zhu, D.; He, Y.; Zheng, Q.; Zhang, C. Effects of soil warming, rainfall reduction and water table level on CH4 emissions from the Zoige peatland in China. Soil Biol. Biochem. 2014, 78, 83–89. [Google Scholar] [CrossRef]
- Nesbit, S.P.; Breitenbeck, G.A. A laboratory study of factors influencing methane uptake by soils. Agric. Ecosyst. Environ. 1992, 41, 39–54. [Google Scholar] [CrossRef]
- Castro, M.S.; Steudler, P.A.; Melillo, J.M.; Aber, J.D.; Bowden, R.D. Factors controlling atmospheric methane consumption by temperate forest soils. Glob. Biogeochem. Cycles 1995, 9, 1–10. [Google Scholar] [CrossRef]
- Geng, S.C.; Chen, Z.J.; Zhang, J.H.; Lou, X.; Wang, X.X.; Dai, G.H.; Han, S.J.; Yu, D.D. Soil methane fluxes of three forest types in Changbai Mountain of Northeast China. Chin. J. Ecol. 2013, 32, 1091–1096. [Google Scholar]
- Wu, X.; Yao, Z.; Brüggemann, N.; Shen, Z.Y.; Wolf, B.; Dannenmann, M.; Zheng, X.; Butterbach-Bahl, K. Effects of soil moisture and temperature on CO2 and CH4 soil–atmosphere exchange of various land use/cover types in a semi-arid grassland in Inner Mongolia, China. Soil Biol. Biochem. 2010, 42, 773–787. [Google Scholar] [CrossRef]
- Aronson, E.L.; Helliker, B.R. Methane flux in non-wetland soils in response to nitrogen addition: A meta-analysis. Ecology 2010, 91, 3242–3251. [Google Scholar] [CrossRef] [PubMed]
- Yonemura, S.; Nouchi, I.; Nishimura, S.; Sakurai, G.; Togami, K.; Yagi, K. Soil respiration, N2O, and CH4 emissions from an Andisol under conventional-tillage and no-tillage cultivation for 4 years. Biol. Fertil. Soils 2014, 50, 63–74. [Google Scholar] [CrossRef]
- Bodelier, P.L. Interactions between nitrogenous fertilizers and methane cycling in wetland and upland soils. Curr. Opin. Environ. Sustain. 2011, 3, 379–388. [Google Scholar] [CrossRef]
- Xu, M.; Qi, Y. Spatial and Seasonal Variations of Q10 Determined by Soil Respiration Measurements at a Sierra Nevadan Forest. Glob. Biogeochem. Cycles 2001, 15, 687–696. [Google Scholar] [CrossRef]
- Almaraz, J.J.; Zhou, X.; Mabood, F.; Madramootoo, C.; Rochette, P.; Ma, B.L.; Smith, D.L. Greenhouse gas fluxes associated with soybean production under two tillage systems in southwestern Quebec. Soil Tillage Res. 2009, 104, 134–139. [Google Scholar] [CrossRef]
- Jain, N.; Arora, P.; Tomer, R.; Mishra, S.V.; Bhatia, A.; Pathak, H.; Chakraborty, D.; Kumar, V.; Dubey, D.S.; Harit, R.C.; et al. Greenhouse gases emission from soils under major crops in Northwest India. Sci. Total Environ. 2016, 542 Pt A, 551. [Google Scholar] [CrossRef]
- Weintraub, M.N.; Schimel, J.P. Interactions between carbon and nitrogen mineralization and soil organic matter chemistry in Arctic tundra soils. Ecosystems 2003, 6, 129–143. [Google Scholar] [CrossRef]
- Wang, Q.K.; Wang, S.L.; He, T.X.; Liu, L.; Wu, J.B. Response of organic carbon mineralization and microbial community to leaf litter and nutrient additions in subtropical forest soils. Soil Biol. Biochem. 2014, 71, 13–20. [Google Scholar] [CrossRef]
- Bae, K.; Lee, D.K.; Fahey, T.J.; Woo, S.Y.; Quaye, A.K.; Lee, Y.K. Seasonal variation of soil respiration rates in a secondary forest and agroforestry systems. Agrofor. Syst. 2013, 87, 131–139. [Google Scholar] [CrossRef]
- Gomes, J.; Bayer, C.; Costa, F.D.S.; Piccolo, M.D.C.; Zanatta, J.A.; Vieira, F.C.B.; Six, J. Soil nitrous oxide emissions in long-term cover crops-based rotations under subtropical climate. Soil Tillage Res. 2009, 106, 36–44. [Google Scholar] [CrossRef]
- Goodroad, L.L.; Keeney, D.R. Nitrous oxide production in aerobic soils under varying pH, temperature and water content. Soil Biol. Biochem. 1984, 16, 39–43. [Google Scholar] [CrossRef]
- Castaldi, S. Responses of nitrous oxide, dinitrogen and carbon dioxide production and oxygen consumption to temperature in forest and agricultural light-textured soils determined by model experiment. Biol. Fertil. Soils 2000, 32, 67–72. [Google Scholar] [CrossRef]
- Levine, J.S.; Cofer, W.R.; Sebacher, D.I.; Winstead, E.L.; Sebacher, S.; Boston, P.J. The effects of fire on biogenic soil emissions of nitric oxide and nitrous oxide. Glob. Biogeochem. Cycles 1988, 2, 445–449. [Google Scholar] [CrossRef]
- Teepe, R.; Brumme, R.; Beese, F.; Ludwig, B. Nitrous oxide emission and methane consumption following compaction of forest soils. Soil Sci. Soc. Am. J. 2004, 68, 605–611. [Google Scholar] [CrossRef]
- Dalal, R.C.; Wang, W.; Robertson, G.P.; Parton, W.J. Nitrous oxide emission from Australian agricultural lands and mitigation options: A review. Aust. J. Soil Res. 2003, 41, 165–195. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Matson, P.A.; Cleve, K.V. Nitrogen availability and nitrification during succession: Primary, secondary and old-field seres. Plant Soil 1989, 115, 229–239. [Google Scholar] [CrossRef]
- Gao, W.; Kou, L.; Zhang, J.; Müller, C.; Yang, H.; Li, S. Ammonium fertilization causes a decoupling of ammonium cycling in a boreal forest. Soil Biol. Biochem. 2016, 101, 114–123. [Google Scholar] [CrossRef]
- Tate, K.R.; Ross, D.J.; Saggar, S.; Hedley, C.B.; Dando, J.; Singh, B.K.; Lambie, S.M. Methane uptake in soils from Pinus radiata plantations, a reverting shrubland and adjacent pastures: Effects of land-use change, and soil texture, water and mineral nitrogen. Soil Biol. Biochem. 2007, 39, 1437–1449. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, X.; Ren, X.; Zhu, B. Afforestation and deforestation enhanced soil CH4 uptake in a subtropical agricultural landscape: Evidence from multi-year and multi-site field experiments. Sci. Total Environ. 2019, 662, 313–323. [Google Scholar] [CrossRef]
- Kolb, S. The quest for atmospheric methane oxidizers in forest soils. Environ. Microbiol. Rep. 2009, 1, 336–346. [Google Scholar] [CrossRef]
- Maurer, D.; Kolb, S.; Haumaier, L.; Borken, W. Inhibition of atmospheric methane oxidation by monoterpenes in Norway spruce and European beech soils. Soil Biol. Biochem. 2008, 40, 3014–3020. [Google Scholar] [CrossRef]
- Sheng, H.; Yang, Y.; Yang, Z.; Chen, G.; Xie, J.; Guo, J.; Zou, S. The dynamic response of soil respiration to land-use changes in subtropical China. Glob. Chang. Biol. 2010, 16, 1107–1121. [Google Scholar] [CrossRef]
- Xu, X.; Zou, X.; Cao, L.; Zhamangulova, N.; Zhao, Y.; Tang, D.; Liu, D. Seasonal and spatial dynamics of greenhouse gas emissions under various vegetation covers in a coastal saline wetland in southeast China. Ecol. Eng. 2014, 73, 469–477. [Google Scholar] [CrossRef]
- Yamulki, S.; Anderson, R.; Peace, A.; Morison, J.I.L. Soil CO2 CH4 and N2O fluxes from an afforested lowland raised peatbog in Scotland: Implications for drainage and restoration. Biogeosciences 2013, 10, 7623–7630. [Google Scholar] [CrossRef]
- Dusenbury, M.P.; Engel, R.E.; Miller, P.R.; Lemke, R.L.; Wallander, R. Nitrous oxide emissions from a Northern Great Plains soil as influenced by nitrogen management and cropping systems. J. Environ. Qual. 2008, 37, 542. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Majumdar, D.; Jain, M.C. Nitrous oxide emissions from kharif and rabi legumes grown on an alluvial soil. Biol. Fertil. Soils 2002, 35, 473–478. [Google Scholar] [CrossRef]
- Mu, Z.; Kimura, S.D.; Hatano, R. Estimation of global warming potential from upland cropping systems in central Hokkaido, Japan. Soil Sci. Plant Nutr. 2010, 52, 371–377. [Google Scholar] [CrossRef]
- Shrestha, R.K.; Lal, R.; Penrose, C. Greenhouse Gas Emissions and Global Warming Potential of Reclaimed Forest and Grassland Soils. J. Environ. Qual. 2009, 38, 426–436. [Google Scholar] [CrossRef]
- Jauhiainen, J.; Silvennoinen, H.; Hämäläinen, R.; Kusin, K.; Limin, S.; Raison, R.J.; Vasander, H. Nitrous oxide fluxes from tropical peat with different disturbance history and management. Biogeosciences 2012, 9, 1337–1350. [Google Scholar] [CrossRef]
- Melling, L.; Hatano, R.; Goh, K. Global warming potential from soils in tropical peatland of Sarawak, Malaysia. Phyton 2005, 45, 275–284. [Google Scholar]
- Ishikura, K.; Darung, U.; Inoue, T.; Hatano, R. Variation in Soil Properties Regulate Greenhouse Gas Fluxes and Global Warming Potential in Three Land Use Types on Tropical Peat. Atmosphere 2018, 9, 465. [Google Scholar] [CrossRef]
- Ruan, L. Greenhouse Gases Emissions from Different Land Use Types and Their Global Warming Potential at Xianning, Hubei, China. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2007. (In Chinese). [Google Scholar]
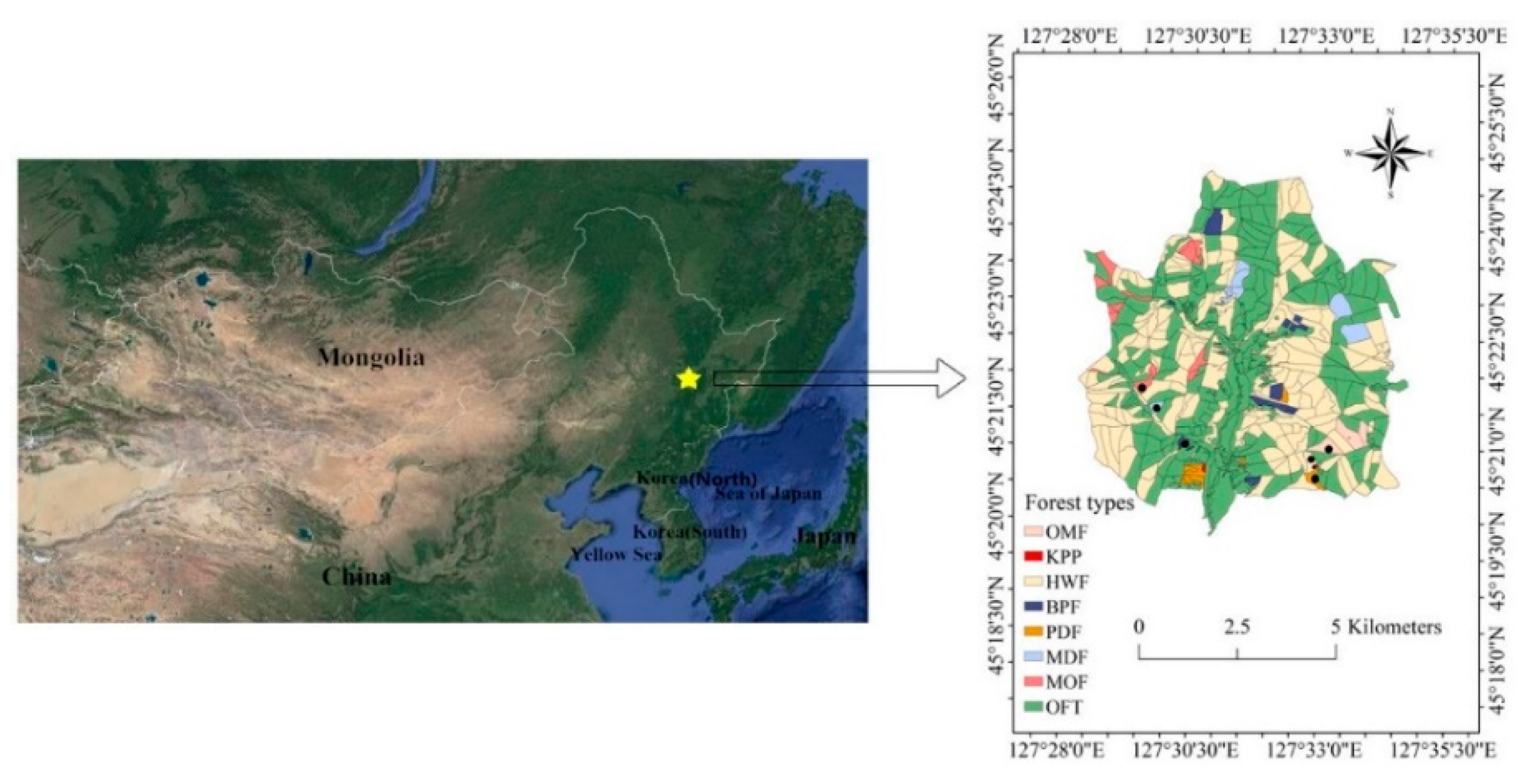
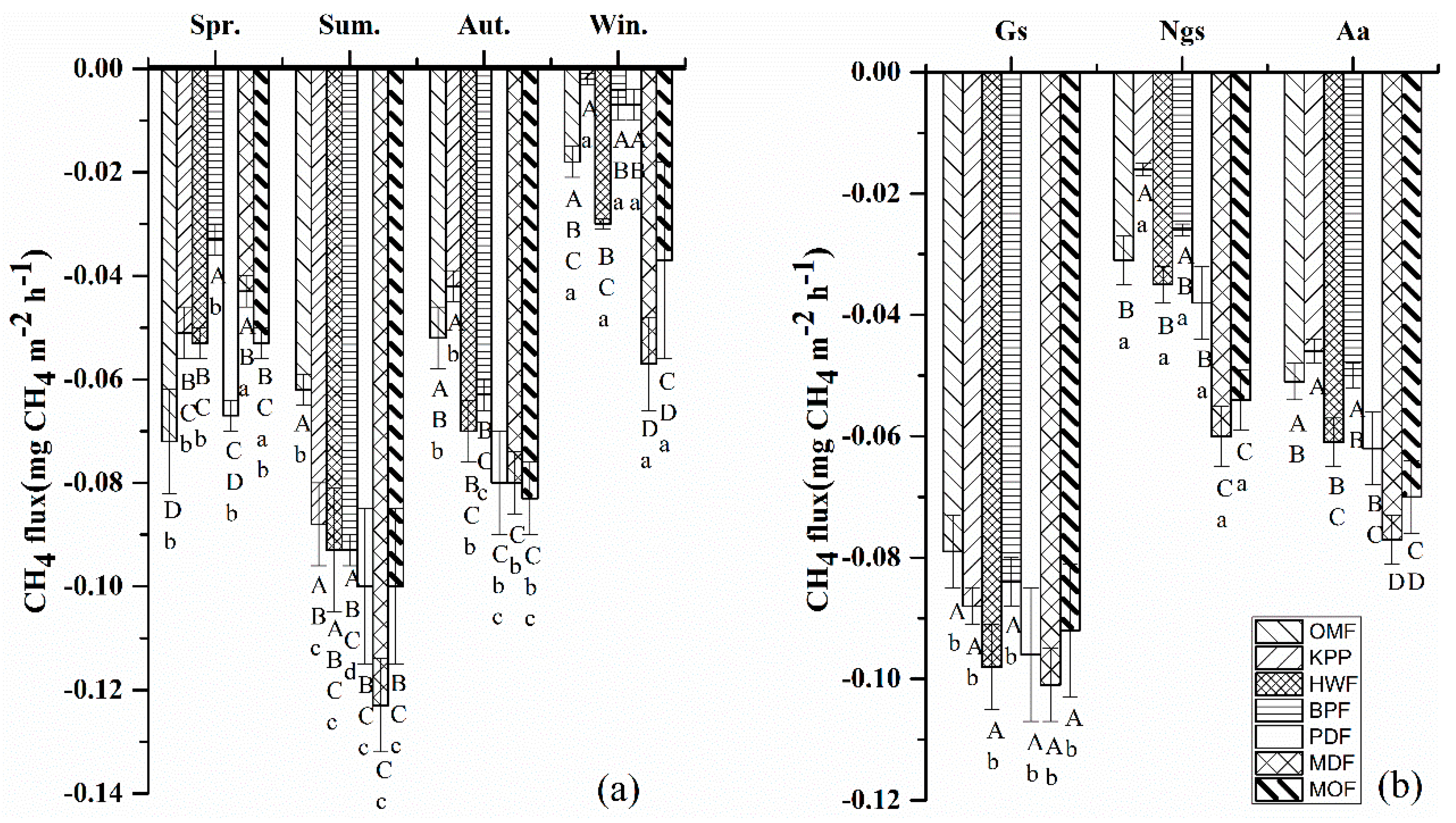
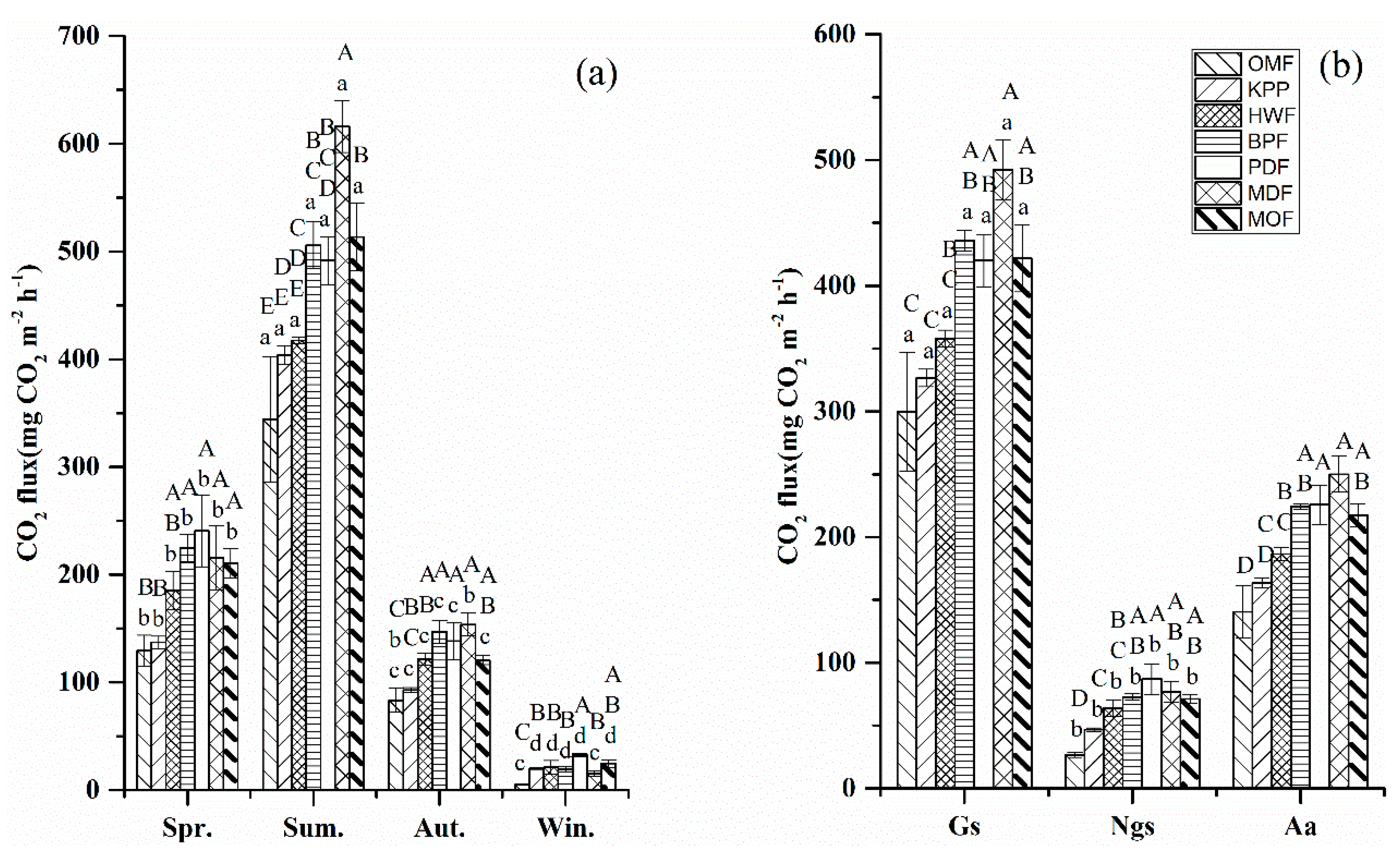
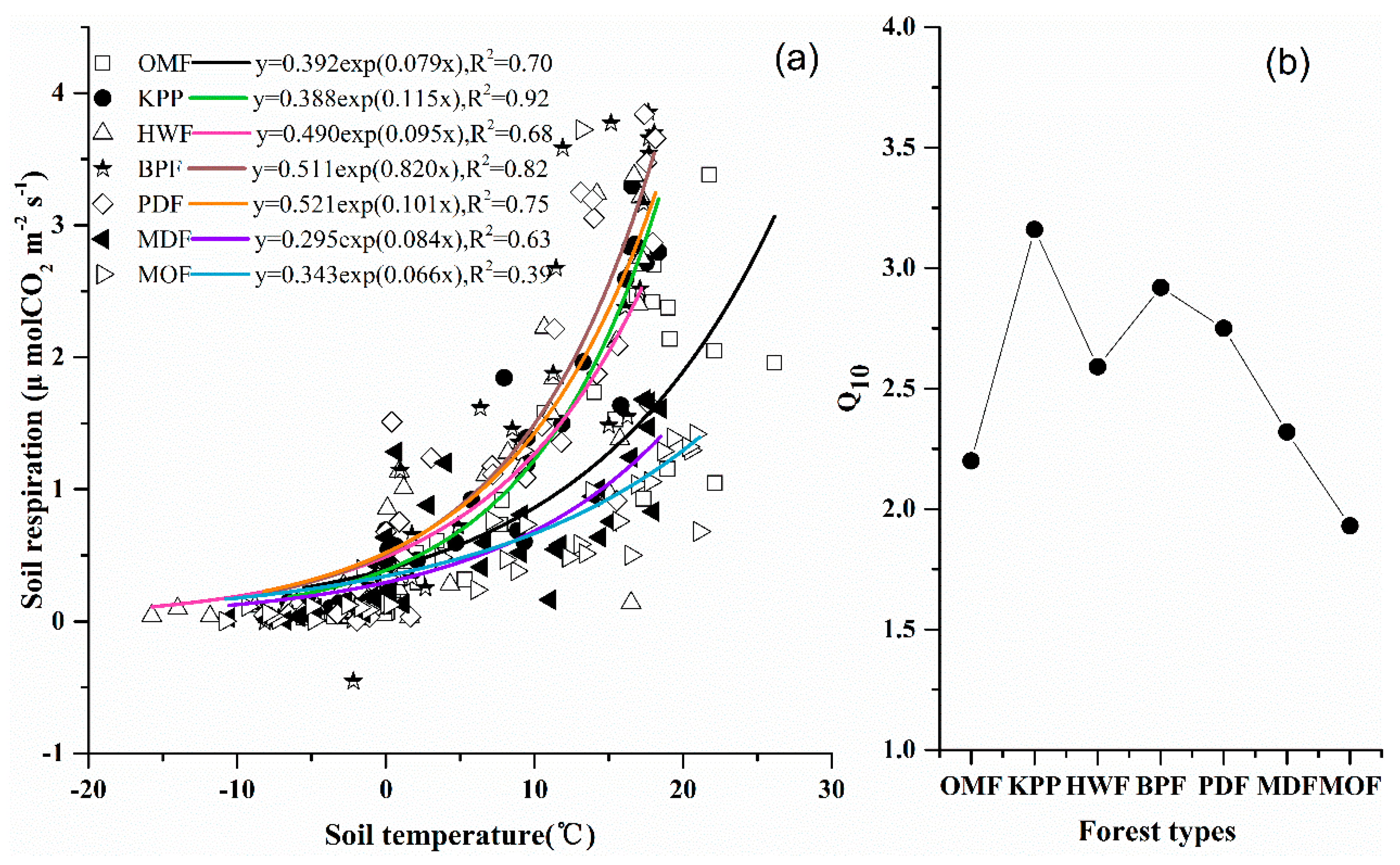
| Forest Type | Age (Year) | Main Species Composition b (Dominant Species) | Land-Use History | Areal Extent (ha) | Density (Trees ha–1) | BA (m2 ha–1) | DBH (cm) | |
|---|---|---|---|---|---|---|---|---|
| Mean | Range | |||||||
| OMF | >160 | (8), 9, 2, 3, 10, 11, 12, 13, 14 | Natural forest | 58.87 | 789 | 54.6 | 25.3 | 4.0–69.1 |
| KPP | 51 | (8), 6, 3, 9, 5, 7, 6 | Reforestation in 1965 | 2.3 | 2111 | 42.1 | 15.0 | 3.9–34.4 |
| HWF | 60 | (4), (3), 2, (7) | Natural regeneration after clearcutting in 1955 | 2083.41 | 1578 | 26.2 | 10.1 | 1.6–50.3 |
| BPF | 65 | (6), 15, 12, 17, 18 | Natural regeneration after clearcutting in 1950 | 80.7 | 3022 | 25.4 | 7.4 | 1.4–34.8 |
| 65 | (6), (5), 7, 4, 3, 2 | Natural regeneration after clearcutting in 1950 | 44.4 | 1833 | 34.1 | 12.1 | 1.6–49.4 | |
| MDF | 65 | 2, 7, 4, 3, 5, 6 | Natural regeneration after clearcutting in 1950 | 99.47 | 2022 | 34.8 | 11.3 | 1.9–49.6 |
| MOF | 66 | (1), 2, 3, 4, 5, 6 | Natural regeneration after clearcutting in 1949 | 101.06 | 1511 | 36.6 | 13.7 | 2.0–55.4 |
| Properties | OMF | KPP | HWF | BPF | MDF | MOF | |
|---|---|---|---|---|---|---|---|
| Air T (°C) | 5.92 ± 0.01B | 5.63 ± 0.01B | 3.44 ± 0.16E | 4.79 ± 0.02C | 4.05 ± 0.21D | 4.93 ± 0.05C | 6.93 ± 0.05A |
| Soil T (°C) | 7.20 ± 0.01A | 4.42 ± 0.01E | 4.17 ± 0.06F | 5.24 ± 0.07D | 5.63 ± 0.04C | 5.17 ± 0.15D | 6.90 ± 0.11B |
| Soil moisture (%) | 35.45 ± 0.49A | 27.90 ± 0.12C | 34.42 ± 0.33A | 26.58 ± 0.32D | 30.84 ± 0.41B | 27.35 ± 0.56D | 23.96 ± 0.47E |
| Soil pH | 5.51 ± 0.04C | 6.02 ± 0.03A | 5.66 ± 0.16B | 5.43 ± 0.05BC | 5.47 ± 0.24BC | 5.53 ± 0.06B | 5.72 ± 0.05B |
| NO3−-N (mg kg−1) | 5.42 ± 0.35B | 9.76 ± 0.19A | 2.58 ± 0.26C | 1.09 ± 0.13D | 2.22 ± 0.42C | 2.18 ± 0.35C | 1.98 ± 0.20C |
| NH4+-N (mg kg−1) | 6.46 ± 0.48B | 24.12 ± 2.32A | 4.97 ± 0.07B | 4.95 ± 0.66B | 4.60 ± 1.69B | 4.38 ± 0.75B | 4.32 ± 0.19B |
| Soil organic C (g kg−1) | 72.02 ± 7.25B | 55.26 ± 1.08CD | 96.37 ± 8.15A | 51.96 ± 3.32D | 61.77 ± 2.07BCD | 69.92 ± 4.52BC | 48.42 ± 2.37D |
| Plot | AT | ST | SVWC | pH | NO3−-N | NH4+-N | SOC | Intercept | R2 | p |
|---|---|---|---|---|---|---|---|---|---|---|
| Models for soil CH4 | ||||||||||
| OMF | −0.008 * | −0.005 ** | 0.587 * | 0.663 | <0.05 | |||||
| KPP | −0.002 ** | 0.004 * | −0.129 * | 0.842 | <0.0071 | |||||
| HWF | 0.003 * | −0.007 ** | −0.045 ** | 0.612 | <0.05 | |||||
| BPF | −0.004 ** | 0.002 * | 0.198 ** | −0.117 * | 0.927 | <0.0071 | ||||
| −0.003 ** | −0.047 ** | 0.626 | <0.0071 | |||||||
| MDF | 0.004 * | −0.007 * | −0.057 ** | 0.441 | <0.1 | |||||
| MOF | 0.836 | 0.330 | <0.1 | |||||||
| Models for soil CO2 | ||||||||||
| OMF | 12.920 ** | 15.382 * | −512.687 * | 0.891 | <0.0071 | |||||
| KPP | 21.477 ** | −6.017 ** | −476.729 | 0.973 | <0.0071 | |||||
| HWF | 16.692 ** | 114.102 ** | 0.833 | <0.0071 | ||||||
| BPF | 23.067 ** | 57.177 * | 0.895 | <0.0071 | ||||||
| 25.094 ** | 2928.966 * | 0.944 | <0.0071 | |||||||
| MDF | 33.999 ** | −10.888 * | 454.856 | 0.936 | <0.0071 | |||||
| MOF | −19.018 * | 49.746 ** | −112.542 * | 388.194 ** | 0.943 | <0.0071 | ||||
| Models for soil N2O | ||||||||||
| OMF | 0.018 ** | −0.068 * | 0.851 | <0.0071 | ||||||
| KPP | 0.016 * | 0.018 | nsc | |||||||
| HWF | 0.01 * | 0.009 * | −0.040 | 0.537 | <0.05 | |||||
| BPF | 0.005 | 0.284 | <0.1 | |||||||
| −0.121 * | −0.058 * | 0.891 * | 0.640 | <0.05 | ||||||
| MDF | 0.001 ** | −0.002 * | −0.155 ** | −0.018 * | 0.994 ** | 0.797 | <0.05 | |||
| MOF | 0.001 * | −0.005 | 0.494 | <0.05 | ||||||
| Sites | Total CH4 Flux (kg CH4 year−1) | Total CO2 Flux (t CO2 year−1) | Total N2O Flux (kg N2O year−1) |
|---|---|---|---|
| OMF | −284.84 ± 18.32a (2.13%) | 733.22 ± 105.79de (1.76%) | 321.01 ± 22.60b (9.90%) |
| KPP | −9.48 ± 0.30a (0.07%) | 33.80 ± 0.76e (0.08%) | 3.82 ± 0.13b (0.12%) |
| HWF | −11,201.8 ± 774.17b (83.76%) | 34,230.43 ± 915.36a (82.27%) | 2520.93 ± 358.65a (77.75%) |
| BPF | −354.00 ± 13.36a (2.65%) | 1591.67 ± 15.97bcd (3.83%) | 50.57 ± 7.13b (1.56%) |
| −234.28 ± 32.66a (1.75%) | 885.19 ± 60.78cde (2.13%) | 65.86 ± 2.63b (2.03%) | |
| MDF | −670.43 ± 34.61a (5.01%) | 2194.97 ± 126.88b (5.28%) | 180.37 ± 3.27b (5.56%) |
| MOF | −619.50 ± 50.34a (4.63%) | 1939.68 ± 83.08bc (4.66%) | 99.71 ± 10.04b (3.08%) |
| Total flux | −13,374.33 ± 841.90 | 41,608.96 ± 814.40 | 3242.27 ± 336.40 |
| Sites | GWPCH4 (t CO2 eq. ha−1 year−1) | GWPCO2 (t CO2 eq. ha−1 year−1) | GWPN2O (t CO2 eq. ha−1 year−1) | Total GWP (t CO2 eq. ha−1 year−1) | Cumulative GWP (t CO2 eq year−1) |
|---|---|---|---|---|---|
| OMF | −0.12 ± 0.01ab | 12.46 ± 1.80d | 1.63 ± 0.11a | 12.46 ± 1.80d | 733.26 ± 105.80cd (1.74%) |
| KPP | −0.10 ± 0.01a | 14.70 ± 0.33cd | 0.49 ± 0.02bc | 14.69 ± 0.33cd | 33.79 ± 0.76d (0.08%) |
| HWF | −0.14 ± 0.01abc | 16.43 ± 0.44bc | 0.36 ± 0.05cd | 16.66 ± 0.38bc | 34,702.67 ± 794.28a (82.33%) |
| BPF | −0.11 ± 0.01a | 19.72 ± 0.35ab | 0.19 ± 0.03e | 19.80 ± 0.22ab | 1597.59 ± 17.90bc (3.79%) |
| −0.13 ± 0.02abc | 19.94 ± 1.37a | 0.44 ± 0.02bcd | 20.25 ± 1.40a | 898.95 ± 61.95cd (2.13%) | |
| MDF | −0.17 ± 0.01c | 22.07 ± 1.28a | 0.54 ± 0.01b | 22.44 ± 1.28a | 2231.78 ± 127.62b (5.29%) |
| MOF | −0.15 ± 0.01bc | 19.19 ± 0.82ab | 0.29 ± 0.03de | 19.33 ± 0.80ab | 1953.83 ± 79.86b (4.64%) |
| Total | 42,151.87 ± 698.53 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, B.; Mu, C. Effects on Greenhouse Gas (CH4, CO2, N2O) Emissions of Conversion from Over-Mature Forest to Secondary Forest and Korean Pine Plantation in Northeast China. Forests 2019, 10, 788. https://doi.org/10.3390/f10090788
Wu B, Mu C. Effects on Greenhouse Gas (CH4, CO2, N2O) Emissions of Conversion from Over-Mature Forest to Secondary Forest and Korean Pine Plantation in Northeast China. Forests. 2019; 10(9):788. https://doi.org/10.3390/f10090788
Chicago/Turabian StyleWu, Bin, and Changcheng Mu. 2019. "Effects on Greenhouse Gas (CH4, CO2, N2O) Emissions of Conversion from Over-Mature Forest to Secondary Forest and Korean Pine Plantation in Northeast China" Forests 10, no. 9: 788. https://doi.org/10.3390/f10090788
APA StyleWu, B., & Mu, C. (2019). Effects on Greenhouse Gas (CH4, CO2, N2O) Emissions of Conversion from Over-Mature Forest to Secondary Forest and Korean Pine Plantation in Northeast China. Forests, 10(9), 788. https://doi.org/10.3390/f10090788





