Black Alder (Alnus glutinosa (L.) Gaertn.) on Compacted Skid Trails: A Trade-off between Greenhouse Gas Fluxes and Soil Structure Recovery?
Abstract
1. Introduction
2. Materials and Methods
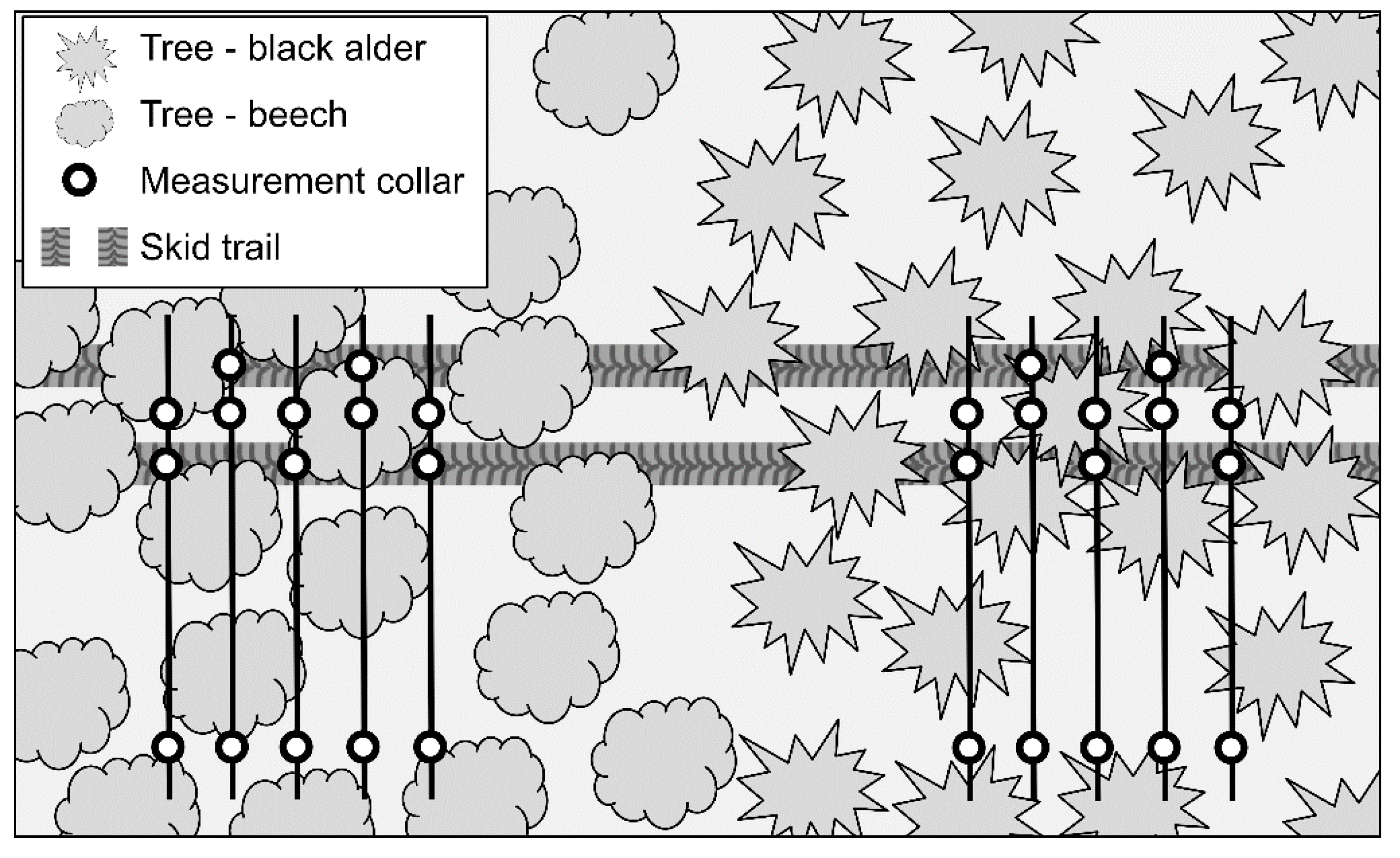
2.1. Study Site and Experimental Design
2.2. Field Measurements
2.3. Soil Sampling and Laboratory Analyses
2.4. Statistical Analyses
3. Results
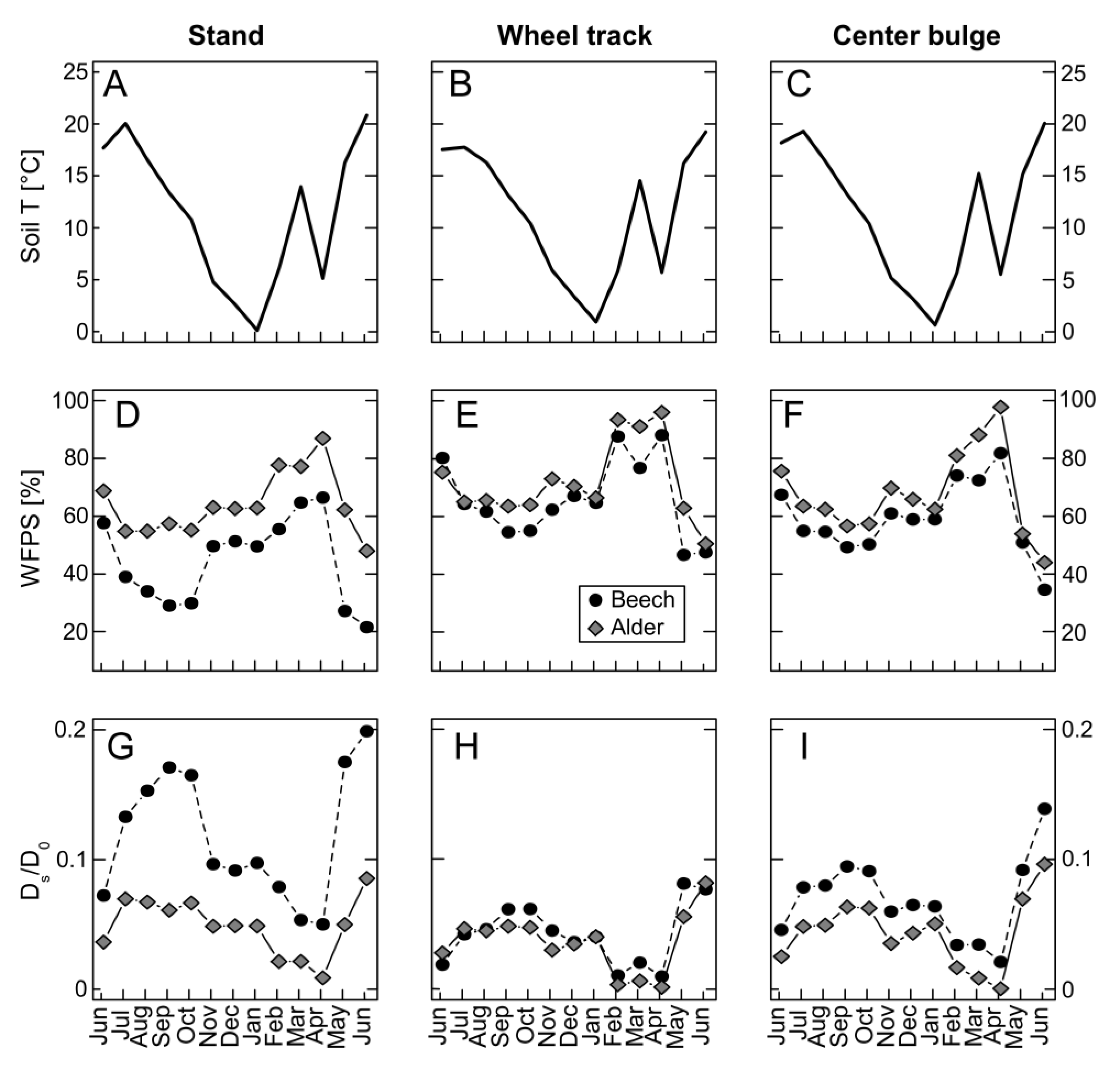
3.1. Environmental Conditions during the Measurement Period
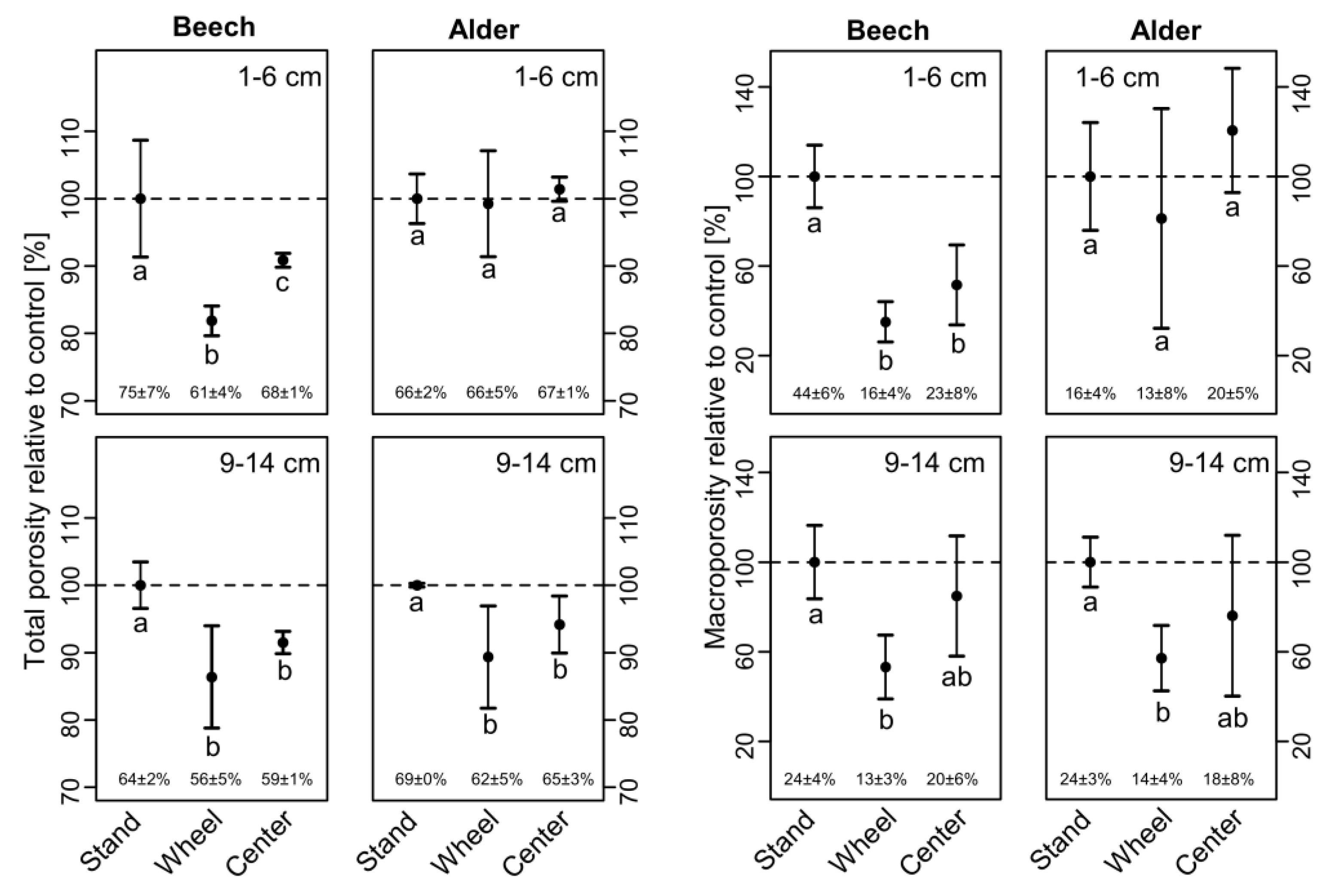
3.2. Soil Physical Properties
3.3. Soil Chemical Properties
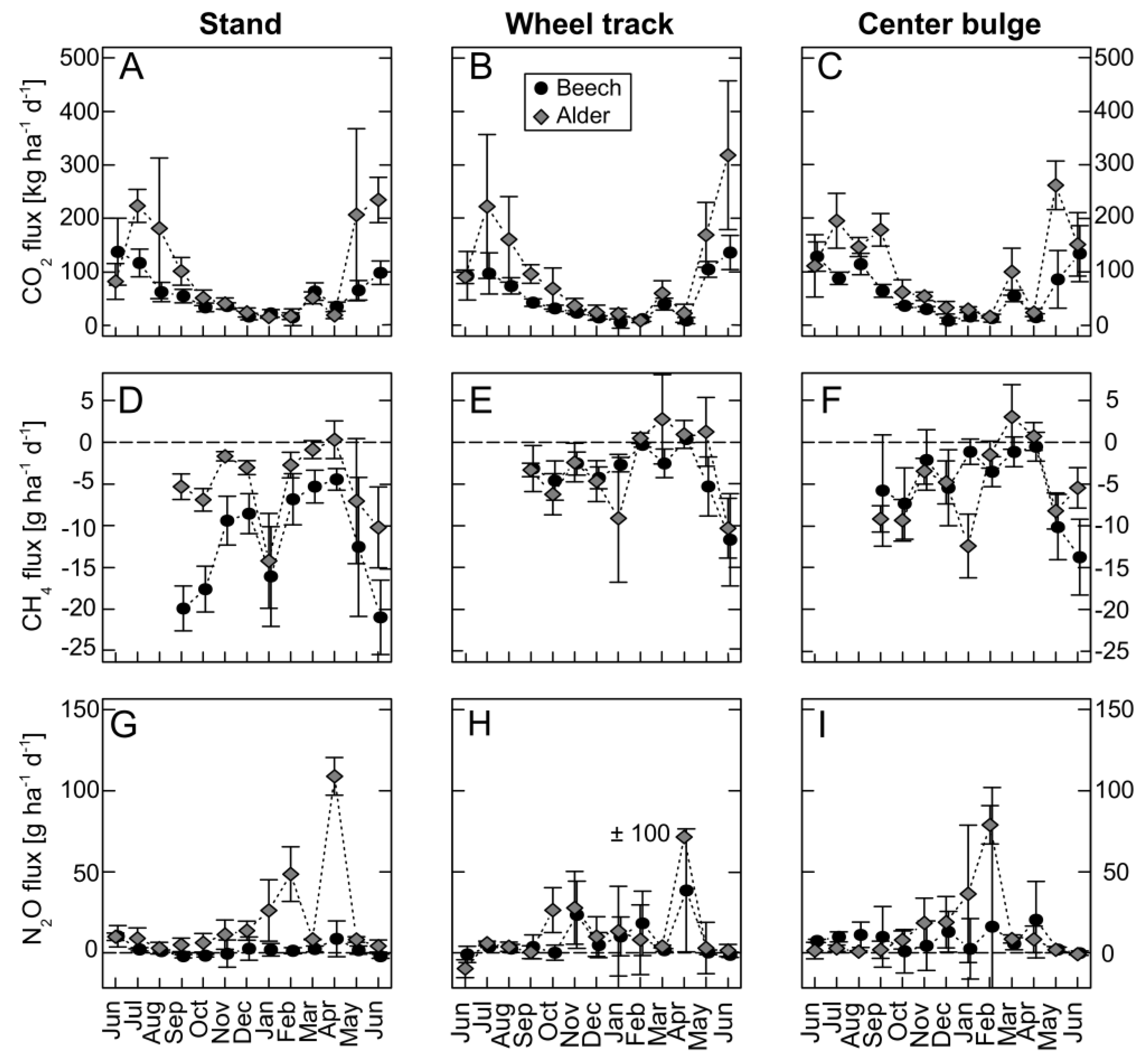
3.4. Greenhouse Gas Fluxes
3.4.1. Carbon Dioxide
3.4.2. Methane
3.4.3. Nitrous Oxide
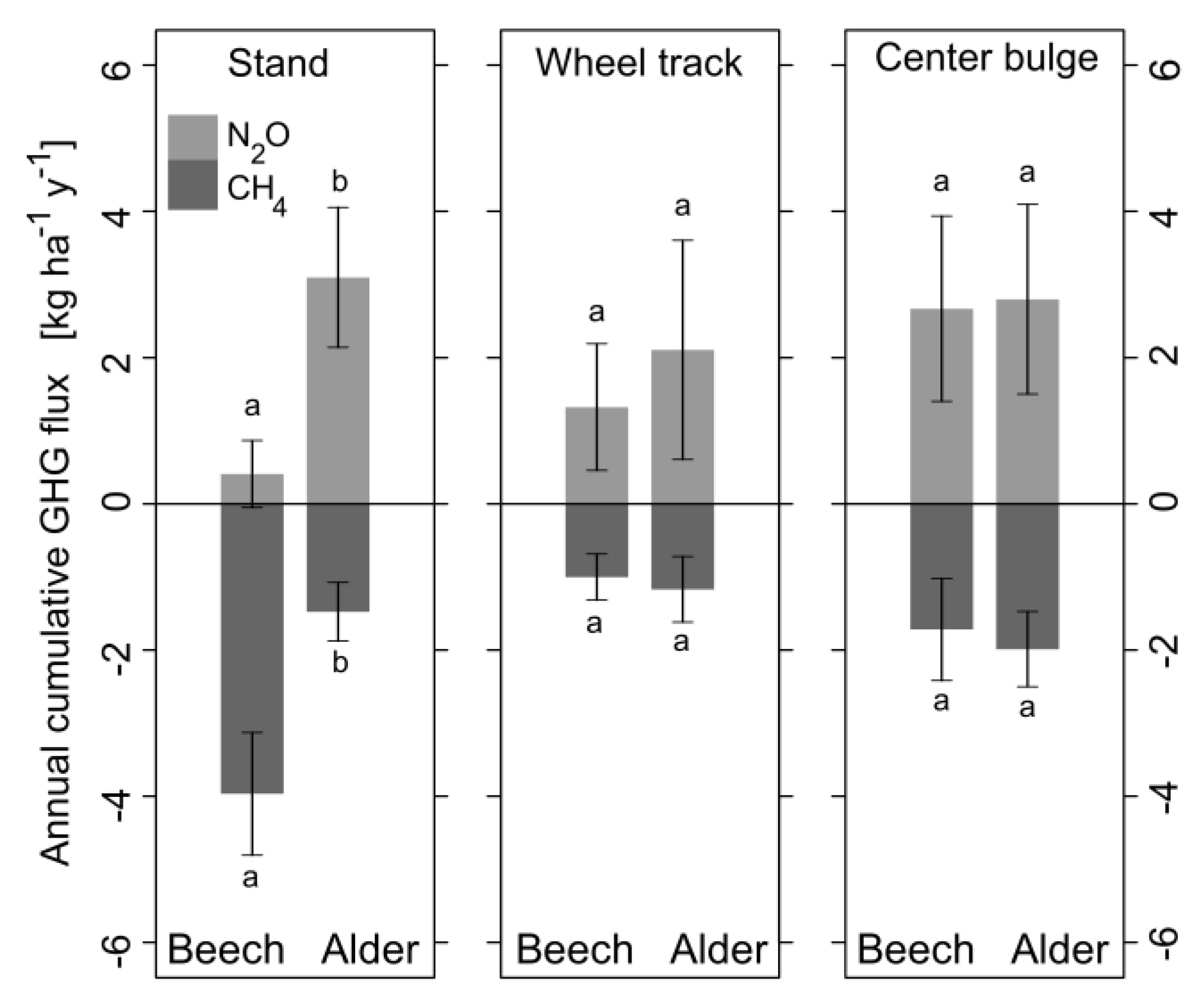
3.4.4. Greenhouse Gas Fluxes on Stand-Level
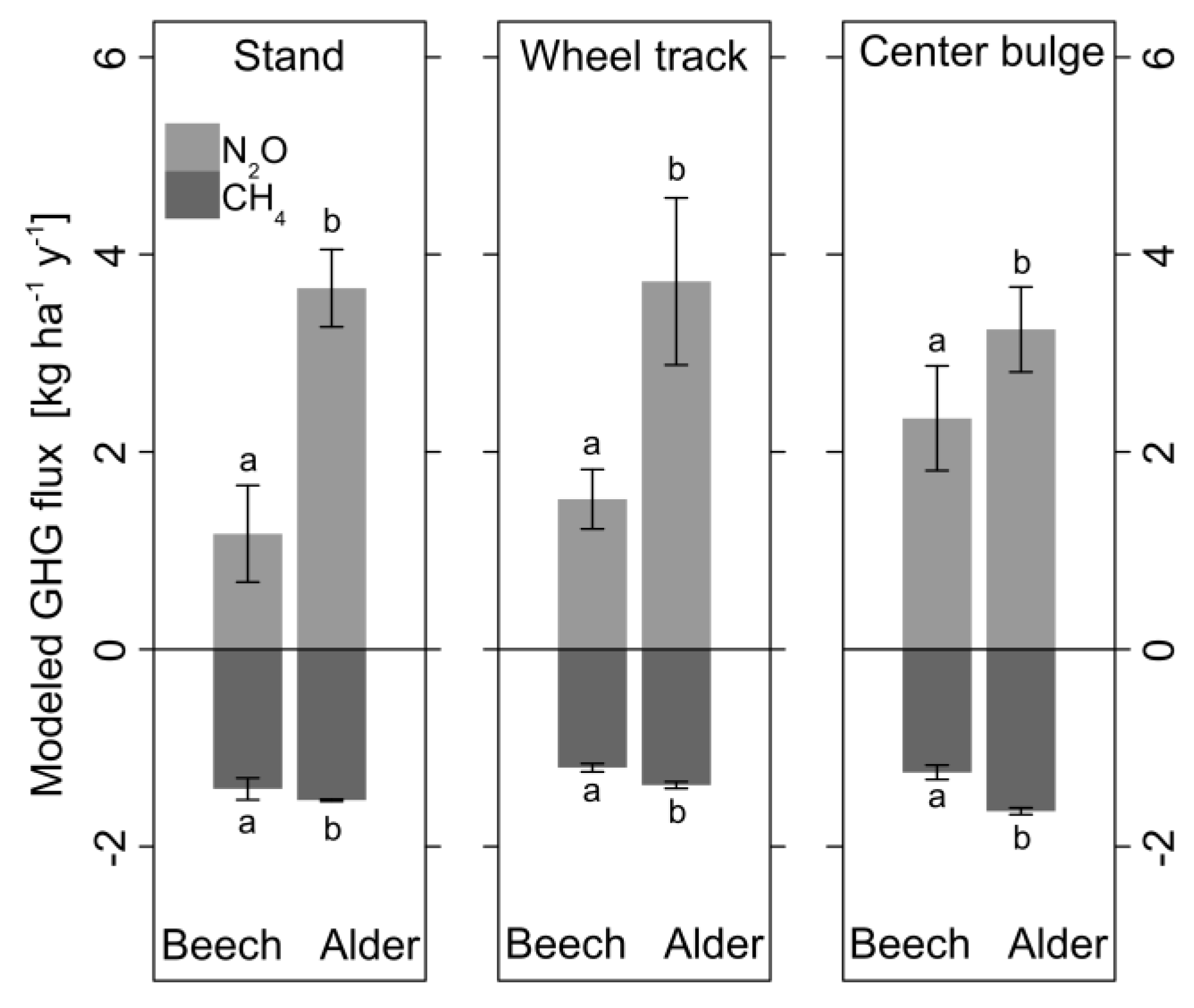
3.5. Mixed Effects Models for CH4 and N2O Fluxes
4. Discussion
4.1. Soil Chemical Parameters
4.2. Soil Physical Parameters
4.3. Greenhouse Gas Fluxes
4.3.1. Carbon Dioxide
4.3.2. Methane
4.3.3. Nitrous Oxide
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References and Notes
- Garland, J.J. Designated Skid Trails Minimize Soil Compaction; Oregon State University, Extension Service: Corvallis, OR, USA, 1983. [Google Scholar]
- Teepe, R.; Brumme, R.; Beese, F.; Ludwig, B. Nitrous Oxide Emission and Methane Consumption Following Compaction of Forest Soils. Soil Sci. Soc. Am. J. 2004, 68, 605. [Google Scholar] [CrossRef]
- Ampoorter, E.; De Schrijver, A.; De Frenne, P.; Hermy, M.; Verheyen, K. Experimental Assessment of Ecological Restoration Options for Compacted Forest Soils. Ecol. Eng. 2011, 37, 1734–1746. [Google Scholar] [CrossRef]
- Horn, R.; Vossbrink, J.; Peth, S.; Becker, S. Impact of Modern Forest Vehicles on Soil Physical Properties. For. Ecol. Manag. 2007, 248, 56–63. [Google Scholar] [CrossRef]
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, A.; Chhabra, R.; DeFries, R.; Galloway, J.; Heimann, M.; et al. Carbon and Other Biogeochemical Cycles. In Climate Change 2013: The Physical Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; pp. 465–570. [Google Scholar]
- Le Mer, J.; Roger, P. Production, Oxidation, Emission and Consumption of Methane by Soils: A Review. Eur. J. Soil Biol. 2001, 37, 25–50. [Google Scholar] [CrossRef]
- Dobbie, K.E.; Smith, K.A. Nitrous Oxide Emission Factors for Agricultural Soils in Great Britain: The Impact of Soil Water-filled Pore Space and Other Controlling Variables. Glob. Chang. Biol. 2003, 9, 204–218. [Google Scholar] [CrossRef]
- Bateman, E.J.; Baggs, E.M. Contributions of Nitrification and Denitrification to N2O Emissions from Soils at Different Water-Filled Pore Space. Biol. Fertil. Soils 2005, 41, 379–388. [Google Scholar] [CrossRef]
- Butterbach-Bahl, K.; Baggs, E.M.; Dannenmann, M.; Kiese, R.; Zechmeister-Boltenstern, S. Nitrous Oxide Emissions from Soils: How Well Do We Understand the Processes and Their Controls? Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130122. [Google Scholar] [CrossRef] [PubMed]
- Weier, K.L.; Doran, J.W.; Power, J.F.; Walters, D.T. Denitrification and the Dinitrogen/Nitrous Oxide Ratio as Affected by Soil Water, Available Carbon, and Nitrate. Soil Sci. Soc. Am. J. 1993, 57, 66. [Google Scholar] [CrossRef]
- Linn, D.M.; Doran, J.W. Effect of Water-Filled Pore Space on Carbon Dioxide and Nitrous Oxide Production in Tilled and Nontilled Soils1. Soil Sci. Soc. Am. J. 1984, 48, 1267. [Google Scholar] [CrossRef]
- Chapuis-Lardy, L.; Wrage, N.; Metay, A.; Chotte, J.-L.; Bernoux, M. Soils, a Sink for N2O? A Review. Glob. Chang. Biol. 2007, 13, 1–17. [Google Scholar] [CrossRef]
- Frey, B.; Niklaus, P.A.; Kremer, J.; Lüscher, P.; Zimmermann, S. Heavy-Machinery Traffic Impacts Methane Emissions as Well as Methanogen Abundance and Community Structure in Oxic Forest Soils. Appl. Environ. Microbiol. 2011, 77, 6060–6068. [Google Scholar] [CrossRef] [PubMed]
- Epron, D.; Plain, C.; Ndiaye, F.K.; Bonnaud, P.; Pasquier, C.; Ranger, J. Effects of Compaction by Heavy Machine Traffic on Soil Fluxes of Methane and Carbon Dioxide in a Temperate Broadleaved Forest. For. Ecol. Manage. 2016, 382, 1–9. [Google Scholar] [CrossRef]
- Ebeling, C.; Lang, F.; Gaertig, T. Structural Recovery in Three Selected Forest Soils after Compaction by Forest Machines in Lower Saxony, Germany. For. Ecol. Manag. 2016, 359, 74–82. [Google Scholar] [CrossRef]
- Von Wilpert, K.; Schäffer, J. Ecological Effects of Soil Compaction and Initial Recovery Dynamics: A Preliminary Study. Eur. J. For. Res. 2006, 125, 129–138. [Google Scholar] [CrossRef]
- Meyer, C.; Lüscher, P.; Schulin, R. Recovery of Forest Soil from Compaction in Skid Tracks Planted with Black Alder (Alnus glutinosa (L.) Gaertn.). Soil Tillage Res. 2014, 143, 7–16. [Google Scholar] [CrossRef]
- Flores Fernández, J.L.; Hartmann, P.; Schäffer, J.; Puhlmann, H.; von Wilpert, K. Initial Recovery of Compacted Soil—Planting and Technical Treatments Decrease CO2 concentrations in Soil and Promote Root Growth. Ann. For. Sci. 2017, 74. [Google Scholar] [CrossRef]
- Haas, J.; Kühne, A.; Schack-Kirchner, H.; Lang, F. Does Juncus Effusus L. And Carex Brizoides L. Help to Regenerate a Sufficient Aeration in Compacted Forest Soils? Allg. For. Jagdzeitung 2017, 188, 85–94. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; Word Soil Ressource Reports; FAO: Rome, Italy, 2015. [Google Scholar]
- Angers, D.A.; Caron, J. Plant-Induced Changes in Soil Structure: Processes and Feedbacks. Biogeochemistry 1998, 42, 55–72. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Mommer, L.; De Vries, F.T. Going Underground: Root Traits as Drivers of Ecosystem Processes. Trends Ecol. Evol. 2014, 29, 692–699. [Google Scholar] [CrossRef]
- Gill, C.J. The Ecological Significance of Adventitious Rooting as a Response to Flooding in Woody Species, with Special Reference to Alnus glutinosa (L.) Gaertn. Flora 1975, 164, 85–97. [Google Scholar] [CrossRef]
- Armstrong, W.; Armstrong, J. Stem Photosynthesis Not Pressurized Ventilation Is Responsible for Light-Enhanced Oxygen Supply to Submerged Roots of Alder (Alnus glutinosa). Ann. Bot. 2005, 96, 591–612. [Google Scholar] [CrossRef][Green Version]
- Kätzel, R. Zum Physiologischen Anpassungspotenzial Der Schwarz-Erle (Alnus glutinosa [L.] GAERTN.). Eberswalder Forstl. Schriftenr. 2003, 17, 39–46. [Google Scholar]
- Bühlmann, T.; Körner, C.; Hiltbrunner, E. Shrub Expansion of Alnus Viridis Drives Former Montane Grassland into Nitrogen Saturation. Ecosystems 2016, 19, 968–985. [Google Scholar] [CrossRef]
- Mogge, B.; Kaiser, E.A.; Munch, J.C. Nitrous Oxide Emissions and Denitrification N-Losses from Forest Soils in the Bornhoved Lake Region (Northern Germany). Soil Biol. Biochem. 1998, 30, 703–710. [Google Scholar] [CrossRef]
- Bühlmann, T.; Caprez, R.; Hiltbrunner, E.; Körner, C.; Niklaus, P.A. Nitrogen Fixation by Alnus Species Boosts Soil Nitrous Oxide Emissions. Eur. J. Soil Sci. 2017, 68, 740–748. [Google Scholar] [CrossRef]
- Reay, D.S.; Nedwell, D.B.; McNamara, N.; Ineson, P. Effect of Tree Species on Methane and Ammonium Oxidation Capacity in Forest Soils. Soil Biol. Biochem. 2005, 37, 719–730. [Google Scholar] [CrossRef]
- MacDonald, J.A.; Skiba, U.; Sheppard, L.J.; Ball, B.; Roberts, J.D.; Smith, K.A.; Fowler, D. The Effect of Nitrogen Deposition and Seasonal Variability on Methane Oxidation and Nitrous Oxide Emission Rates in an Upland Spruce Plantation and Moorland. Atmos. Environ. 1997, 31, 3693–3706. [Google Scholar] [CrossRef]
- Gulledge, J.; Doyle, A.P.; Schimel, J.P. Different NH4+-Inhibition Patterns of Soil CH4 Consumption: A Result of Distinct CH4-Oxidizer Populations Across Sites? Soil Biol. Biochem. 1997, 29, 13–21. [Google Scholar] [CrossRef]
- Deutscher Wetterdienst, DWD. Monthly Mean of Air Temperature, 1 km Grid for Germany. Available online: http://werdis.dwd.de/werdis/retrieve_data.do?pidpat=de.dwd.nkdz-grid.TAMM17.30.00&toplevel=false (accessed on 15 February 2019).
- Loftfield, N.; Flessa, H.; Augustin, J.; Beese, F. Automated Gas Chromatographic System for Rapid Analysis of the Atmospheric Trace Gases Methane, Carbon Dioxide, and Nitrous Oxide. J. Environ. Qual. 1997, 26, 560–564. [Google Scholar] [CrossRef]
- Hutchinson, G.L.; Livingston, G.P. Soil-Atmosphere Gas Exchange. In Methods of Soil Analysis: Part 4 Physical Methods; Soil Science Society of America: Madison, WI, USA, 2002; pp. 1159–1182. [Google Scholar]
- Maechler, M.; Rousseeuw, P.; Croux, C.; Todorov, V.; Ruckstuhl, A.; Salibian-Barrera, M.; Verbeke, T.; Koller, M. Robustbase: Basic Robust Statistics, 2016.
- Myhre, G.; Shindell, D.; Bréon, F.-M.; Collins, W.; Fuglestvedt, J.; Huang, J.; Koch, D.; Lamarque, J.-F.; Lee, D.; Mendoza, B.; et al. Anthropogenic and Natural Radiative Forcing. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; pp. 659–740. [Google Scholar]
- Hartge, K.H.; Horn, R. Die Physikalische Untersuchung von Böden, 3rd ed.; Ferdinand Enke Verlag: Stuttgart, Germany, 2011. [Google Scholar]
- Kühne, A.; Schack-Kirchner, H.; Hildebrand, E.E. Gas Diffusivity in Soils Compared to Ideal Isotropic Porous Media. J. Plant Nutr. Soil Sci. 2012, 175, 34–45. [Google Scholar] [CrossRef]
- Schack-Kirchner, H.; Gaertig, T.; Wilpert, K.V.; Hildebrand, E.E. A Modified Mcintyre and Phillip Approach to Measure Top-Soil Gas Diffusivity in-Situ. J. Plant Nutr. Soil Sci. 2001, 164, 253–258. [Google Scholar] [CrossRef]
- Bassler, R.; Hoffmann, G. Bestimmung von Mineralischem, (Nitrat-) Stickstoff in Bodenprofilen, Nmin-Labormethode. In VDLUFA Methodenbuch Band I; Deller, B., Ed.; VDLUFA-Verlag: Darmstadt, Germany, 1997. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Dinno, A. Dunn.Test: Dunn’s Test of Multiple Comparisons Using Rank Sums, 2016.
- McGill, R.; Tukey, J.W.; Larsen, W.A. Variation of Box Plots. Am. Stat. 1978, 32, 12–16. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models, 2016.
- John, J.A.; Draper, N.R. An Alternative Family of Transformations. J. R. Stat. Soc. 1980, 29, 190–197. [Google Scholar] [CrossRef]
- Lefcheck, J.S. PiecewiseSEM: Piecewise Structural Equation Modelling in R for Ecology, Evolution, and Systematics. Methods Ecol. Evol. 2016, 7, 573–579. [Google Scholar] [CrossRef]
- Wang, F.; Li, Z.; Xia, H.; Zou, B.; Li, N.; LIu, J.; Zhu, W. Effects of Nitrogen-Fixing and Non-Nitrogen-Fixing Tree Species on Soil Properties and Nitrogen Transformation during Forest Restoration in Southern China. Soil Sci. Plant Nutr. 2010, 56, 297–306. [Google Scholar] [CrossRef]
- Uri, V.; Aosaar, J.; Varik, M.; Becker, H.; Ligi, K.; Padari, A.; Kanal, A.; Lõhmus, K. The Dynamics of Biomass Production, Carbon and Nitrogen Accumulation in Grey Alder (Alnus incana (L.) Moench) Chronosequence Stands in Estonia. For. Ecol. Manag. 2014, 327, 106–117. [Google Scholar] [CrossRef]
- Rothe, A.; Kermit Cromack, J.; Resh, S.C.; Makineci, E.; Son, Y. Soil Carbon and Nitrogen Changes Under Douglas-Fir With and Without Red Alder. Soil Sci. Soc. Am. J. 2002, 66, 1988–1995. [Google Scholar] [CrossRef]
- Binkley, D. How Nitrogen-Fixing Trees Change Soil Carbon. In Tree Species Effects on Soils: Implications for Global Change; Binkley, D., Menyailo, O., Eds.; Implications for Global Change, NATO Sciences Series; Kluwer Academic Publishers: Dordrecht, The Netherland, 2005; pp. 155–164. [Google Scholar]
- Resh, S.C.; Binkley, D.; Parrotta, J.A. Greater Soil Carbon Sequestration under Nitrogen-Fixing Trees Compared with Eucalyptus Species. Ecosystems 2002, 5, 217–231. [Google Scholar] [CrossRef]
- Rytter, L.; Rytter, R.M. Growth and Carbon Capture of Grey Alder (Alnus incana (L.) Moench.) under North European Conditions—Estimates Based on Reported Research. For. Ecol. Manag. 2016, 373, 56–65. [Google Scholar] [CrossRef]
- Rytter, R.M. The Effect of Limited Availability of N or Water on C Allocation to Fine Roots and Annual Fine Root Turnover in Alnus incana and Salix Viminalis. Tree Physiol. 2013, 33, 924–939. [Google Scholar] [CrossRef]
- Kalyn, A.L.; Van Rees, K.C.J. Contribution of Fine Roots to Ecosystem Biomass and Net Primary Production in Black Spruce, Aspen, and Jack Pine Forests in Saskatchewan. Agric. For. Meteorol. 2006, 140, 236–243. [Google Scholar] [CrossRef]
- Hertel, D.; Strecker, T.; Müller-Haubold, H.; Leuschner, C. Fine Root Biomass and Dynamics in Beech Forests across a Precipitation Gradient—Is Optimal Resource Partitioning Theory Applicable to Water-Limited Mature Trees? J. Ecol. 2013, 101, 1183–1200. [Google Scholar] [CrossRef]
- Burke, M.K.; Raynal, J. Fine Root Growth Phenology, Production, and Turnover in a Northern Hardwood Forest Ecosystem. Plant Soil 1994, 162, 135–146. [Google Scholar] [CrossRef]
- Uri, V.; Lõhmus, K.; Mander, Ü.; Ostonen, I.; Aosaar, J.; Maddison, M.; Helmisaari, H.S.; Augustin, J. Long-Term Effects on the Nitrogen Budget of a Short-Rotation Grey Alder (Alnus incana (L.) Moench) Forest on Abandoned Agricultural Land. Ecol. Eng. 2011, 37, 920–930. [Google Scholar] [CrossRef]
- Van Miegroet, H.; Cole, D.W. Acidification Sources in Red Alder and Douglas Fir Soils-Importance of Nitrification. Soil Sci. Soc. Am. J. 1985, 49, 1274–1279. [Google Scholar] [CrossRef]
- Dilly, O.; Munch, J.C. Microbial Biomass Content, Basal Respiration and Enzyme Activities during the Course of Decomposition of Leaf Litter in a Black Alder (Alnus glutinosa (L.) Gaertn.) Forest. Soil Biol. Biochem. 1996, 28, 1073–1081. [Google Scholar] [CrossRef]
- Wedderburn, M.E.; Carter, J. Litter Decomposition by Four Functional Tree Types for Use in Silvopastoral Systems. Soil Biol. Biochem. 1999, 31, 455–461. [Google Scholar] [CrossRef]
- De Vries, W.; Breeuwsma, A. Relative Importance of Natural and Anthropogenic Proton Sources in Soils in The Netherlands. Water. Air. Soil Pollut. 1986, 28, 173–184. [Google Scholar] [CrossRef]
- Neirynck, J.; Mirtcheva, S.; Sioen, G.; Lust, N. Impact of Tilia platyphyllos Scop., Fraxinus Excelsior L., Acer pseudoplatanus L., Quercus robur L. and Fagus sylvatica L. on Earthworm Biomass and Physico-Chemical Properties of a Loamy Topsoil. For. Ecol. Manag. 2000, 133, 275–286. [Google Scholar] [CrossRef]
- Haynes, R.J.; Naidu, R. Influence of Lime, Fertilizer and Manure Applications on Soil Organic Matter Content and Soil Physical Conditions: A Review. Nutr. Cycl. Agroecosyst. 1998, 51, 123–137. [Google Scholar] [CrossRef]
- Rawls, W.J.; Pachepsky, Y.A.; Ritchie, J.C.; Sobecki, T.M.; Bloodworth, H. Effect of Soil Organic Carbon on Soil Water Retention. Geoderma 2003, 116, 61–76. [Google Scholar] [CrossRef]
- Minasny, B.; McBratney, A.B. Limited Effect of Organic Matter on Soil Available Water Capacity. Eur. J. Soil Sci. 2018, 69, 39–47. [Google Scholar] [CrossRef]
- Kutsch, W.L.; Staack, A.; Wötzel, J.; Middelhoff, U.; Kappen, L. Field Measurements of Root Respiration and Total Soil Respiration in an Alder Forest. New Phytol. 2001, 150, 157–168. [Google Scholar] [CrossRef]
- Butnor, J.R.; Johnsen, K.H.; Oren, R.; Katul, G.G. Reduction of Forest Floor Respiration by Fertilization on Both Carbon Dioxide-Enriched and Reference 17-Year-Old Loblolly Pine Stands. Glob. Chang. Biol. 2003, 9, 849–861. [Google Scholar] [CrossRef]
- Lloyd, J.; Taylor, J.A. On the Temperature Dependence of Soil Respiration. Funct. Ecol. 1994, 8, 315–323. [Google Scholar] [CrossRef]
- Goutal, N.; Parent, F.; Bonnaud, P.; Demaison, J.; Nourrisson, G.; Epron, D.; Ranger, J. Soil CO2 concentration and Efflux as Affected by Heavy Traffic in Forest in Northeast France. Eur. J. Soil Sci. 2012, 63, 261–271. [Google Scholar] [CrossRef]
- Conlin, T.S.S.; Driessche, R. Response of Soil CO2 and O2 Concentrations to Forest Soil Compaction at the Long-Term Soil Productivity Sites in Central British Columbia. Can. J. Soil Sci. 2000, 80, 625–632. [Google Scholar] [CrossRef]
- Smith, K.A.; Dobbie, K.E.; Ball, B.C.; Bakken, L.R.; Sitaula, B.K.; Hansen, S.; Brumme, R.; Borken, W.; Christensen, S.; Priemé, A.; et al. Oxidation of Atmospheric Methane in Northern European Soils, Comparison with Other Ecosystems, and Uncertainties in the Global Terrestrial Sink. Glob. Chang. Biol. 2000, 6, 791–803. [Google Scholar] [CrossRef]
- Reay, D.S.; Radajewski, S.; Murrell, J.C.; McNamara, N.; Nedwell, D.B. Effects of Land-Use on the Activity and Diversity of Methane Oxidizing Bacteria in Forest Soils. Soil Biol. Biochem. 2001, 33, 1613–1623. [Google Scholar] [CrossRef]
- Gallardo, A.; Rodr, J.J.; Covelo, F.; Fern, R.; Rodriguez-Saucedo, J.J.; Fernandez-Ales, R. Soil Nitrogen Heterogeneity in a Dehesa Ecosystem. Plant Soil 2000, 222, 71–82. [Google Scholar] [CrossRef]
- Rusch, H.; Rennenberg, H. Black Alder (Alnus glutinosa (L.) Gaertn.) Trees Mediate Methane and Nitrous Oxide Emission from the Soil to the Atmosphere. Plant Soil 1998, 201, 1–7. [Google Scholar] [CrossRef]
- Maier, M.; Machacova, K.; Lang, F.; Svobodova, K.; Urban, O. Combining Soil and Tree-Stem Flux Measurements and Soil Gas Profiles to Understand CH4 Pathways in Fagus Sylvatica Forests. J. Plant Nutr. Soil Sci. 2017, 1–5. [Google Scholar] [CrossRef]
- Eickenscheidt, T.; Heinichen, J.; Augustin, J.; Freibauer, A.; Drösler, M. Nitrogen Mineralization and Gaseous Nitrogen Losses from Waterlogged and Drained Organic Soils in a Black Alder (Alnus glutinosa (L.) Gaertn.) Forest. Biogeosciences 2014, 11, 2961–2976. [Google Scholar] [CrossRef]
- Butterbach-Bahl, K.; Willibald, G.; Papen, H.; Gasche, R. Exchange of N-Gases at the Spruce and Beech Sites at the Höglwald Forest—A Summary. Plant Soil 2002, 240, 117–123. [Google Scholar] [CrossRef]
- Borken, W.; Beese, F. Methane and Nitrous Oxide Fluxes of Soils in Pure and Mixed Stands of European Beech and Norway Spruce. Eur. J. Soil Sci. 2006, 57, 617–625. [Google Scholar] [CrossRef]
- Zechmeister-Boltenstern, S.; Hahn, M.; Meger, S.; Jandl, R. Nitrous Oxide Emissions and Nitrate Leaching in Relation to Microbial Biomass Dynamics in a Beech Forest Soil. Soil Biol. Biochem. 2002, 34, 823–832. [Google Scholar] [CrossRef]
- Teepe, R.; Brumme, R.; Beese, F. Nitrous Oxide Emissions from Soil during Freezing and Thawing Periods. Soil Biol. Biochem. 2001, 33, 1269–1275. [Google Scholar] [CrossRef]
- Bierderbeck, V.O.; Campbell, C.A. Influence of Simulated Fall and Spring Conditions on Soil System. I. Effect on Soil Microflora. Soil Sci. Soc. Am. J. 1971, 35, 474–479. [Google Scholar] [CrossRef]
- Müller, C.; Martin, M.; Stevens, R.J.; Laughlin, R.J.; Kammann, C.; Ottow, J.C.G.; Jäger, H.J. Processes Leading to N2O Emissions in Grassland Soil during Freezing and Thawing. Soil Biol. Biochem. 2002, 34, 1325–1331. [Google Scholar] [CrossRef]
- Risk, N.; Snider, D.; Wagner-Riddle, C. Mechanisms Leading to Enhanced Soil Nitrous Oxide Fluxes Induced by Freeze–Thaw Cycles. Can. J. Soil Sci. 2013, 93, 401–414. [Google Scholar] [CrossRef]
- Papen, H.; Butterbach-Bahl, K. A 3-Year Continuous Record of Nitrogen Trace Gas Fluxes from Untreated and Limed Soil of a N-Saturated Spruce and Beech Forest Ecosystem in Germany 1. N2O Emissions. J. Geophys. Res. 1999, 104, 18487–18503. [Google Scholar] [CrossRef]
- Luo, G.J.; Brüggemann, N.; Wolf, B.; Gasche, R.; Grote, R.; Butterbach-Bahl, K. Decadal Variability of Soil CO2, NO, N2O, and CH4 fluxes at the Höglwald Forest, Germany. Biogeosciences 2012, 9, 1741–1763. [Google Scholar] [CrossRef]
- Wu, X.; Bruggemann, N.; Gasche, R.; Shen, Z.; Wolf, B.; Butterbach-Bahl, K. Environmental Controls over Soil-Atmosphere Exchange of N2O, NO, and CO2 in a Temperate Norway Spruce Forest. Global Biogeochem. Cycles 2010, 24, 1–16. [Google Scholar] [CrossRef]
- Jungkunst, H.F.; Freibauer, A.; Neufeldt, H.; Bareth, G. Nitrous Oxide Emissions from Agricultural Land Use in Germany—A Synthesis of Available Annual Field Data. J. Plant Nutr. Soil Sci. 2006, 169, 341–351. [Google Scholar] [CrossRef]
| Depth [cm] | Stand | Wheel Track | Center Bulge | ||||
|---|---|---|---|---|---|---|---|
| Beech | Alder | Beech | Alder | Beech | Alder | ||
| DB | 1–6 | 0.76 ± 0.04 a | 0.92 ± 0.02 b | 1.05 ± 0.00 a | 0.94 ± 0.03 b | 0.95 ± 0.02 a | 0.93 ± 0.00 a |
| 9–14 | 1.06 ± 0.02 a | 0.87 ± 0.00 b | 1.20 ± 0.02 a | 1.07 ± 0.04 b | 1.20 ± 0.01 a | 0.96 ± 0.02 b | |
| RMD | 1–6 | 0.88 ± 0.38 a | 0.89 ± 0.30 a | 1.34 ± 0.45 a | 0.63 ± 0.26 a | 1.93 ± 2.31 a | 0.93 ± 0.43 a |
| 9–14 | 1.00 ± 0.65 a | 0.66 ± 0.14 a | 0.13 ± 0.27 a | 0.57 ± 0.23 a | 0.47 ± 0.87 a | 0.30 ± 0.34 a | |
| Depth [cm] | Stand | Wheel Track | Center Bulge | ||||
|---|---|---|---|---|---|---|---|
| Beech | Alder | Beech | Alder | Beech | Alder | ||
| pH | 1–6 | 5.0 ± 0.02 a | 5.7 ± 0.03 b | 5.6 ± 0.04 a | 5.9 ± 0.04 b | 5.2 ± 0.08 a | 5.6 ± 0.01 b |
| 9–14 | 5.0 ± 0.09 a | 5.9 ± 0.02 b | 5.7 ± 0.02 a | 6.1 ± 0.02 b | 5.6 ± 0.02 a | 6.0 ± 0.03 b | |
| C [mg g−1] | 1–6 | 41.2 ± 0.3 a | 57.7 ± 0.4 b | 38.4 ± 0.3 a | 58.2 ± 0.5 b | 41.0 ± 0.4 a | 54.5 ± 0.3 b |
| 9–14 | 36.8 ± 0.4 a | 47.9 ± 0.6 b | 22.6 ± 0.3 a | 47.4 ± 0.6 b | 25.7 ± 0.3 a | 41.6 ± 0.2 b | |
| N [mg g−1] | 1–6 | 3.2 ± 0.2 a | 4.4 ± 0.1 b | 3.0 ± 0.2 a | 4.4 ± 0.4 b | 3.2 ± 0.20 a | 4.3 ± 0.1 b |
| 9–14 | 2.8 ± 0.2 a | 3.5 ± 0.3 b | 1.9 ± 0.2 a | 3.4 ± 0.4 b | 2.2 ± 0.2 a | 3.1 ± 0.2 b | |
| C/N | 1–6 | 13.1 ± 0.5 a | 12.8 ± 1.0 a | 12.6 ± 0.63 a | 13.0 ± 0.2 a | 12.5 ± 0.64 a | 12.4 ± 0.5 a |
| 9–14 | 12.2 ± 1.4 a | 13.8 ± 0.5 a | 11.7 ± 0.4 a | 14.0 ± 0.7 b | 11.7 ± 0.2 a | 13.0 ± 0.2 b | |
| Stand (86%) | Wheel Track (7%) | Center Bulge (7%) | ||||
|---|---|---|---|---|---|---|
| Beech | Alder | Beech | Alder | Beech | Alder | |
| CO2 [kg ha−1 y−1] | 14,359 ± 2093 a | 17,862 ± 5700 b | 820 ± 267 a | 1697 ± 430 b | 988 ± 238 a | 1933 ± 572 b |
| N2O in CO2 eq [kg ha−1 y−1] | 93.0 ± 104.4 a | 706.0 ± 218.4 b | 24.6 ± 16.1 a | 39.1 ± 27.8 a | 49.5 ± 23.5 a | 51.9 ± 24.1 a |
| CH4 in CO2 eq [kg ha−1 y−1] | −95.5 ± 20.0 a | −35.5 ± 9.6 b | −2.0 ± 0.6 a | −2.3 ± 0.9 a | −3.4 ± 1.4 a | −3.9 ± 1.0 a |
| ∑ CO2 eq [kg ha−1 y−1] | −2.5 | 670.5 | 22.6 | 36.8 | 46.1 | 48 |
| CH4 Flux Beech | Estimate | Standard Error | Effect [%] | p-Value | Conditional R2 | RMSE |
| Intercept | 0.56 | 0.68 | - | n.s. | 0.64 | 4.13 |
| Soil T [°C] | −0.04 | 0.06 | −7.1 per 1 °C | n.s. | ||
| Ds/D0 [−] | −93.6 | 6.57 | −167.1 per 0.01 | *** | ||
| CH4 Flux Alder | Estimate | Standard Error | Effect [%] | p-Value | Conditional R2 | |
| Intercept | 0.71 | 0.81 | - | n.s. | 0.26 | 4.99 |
| Soil T [°C] | 0.13 | 0.07 | 18.3 per 1 °C | * | ||
| Ds/D0 [−] | −115.08 | 14.78 | −162.1 per 0.01 | *** | ||
| N2O Flux Beech | Estimate | Standard Error | Effect [%] | p-Value | Conditional R2 | |
| Intercept | 2.73 | 0.31 | - | *** | 0.48 | 1.26 |
| Soil T [°C] | −0.04 | 0.02 | −1.5 per 1 °C | * | ||
| Ds/D0 [−] | −13.36 | 2.50 | −4.9 per 0.01 | *** | ||
| N2O Flux Alder | Estimate | Standard Error | Effect [%] | p-Value | Conditional R2 | |
| Intercept | 4.21 | 0.27 | - | *** | 0.50 | 1.25 |
| Soil T [°C] | −0.12 | 0.02 | −2.9 per 1 °C | *** | ||
| Ds/D0 [−] | −19.80 | 3.93 | −4.7 per 0.01 | *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warlo, H.; von Wilpert, K.; Lang, F.; Schack-Kirchner, H. Black Alder (Alnus glutinosa (L.) Gaertn.) on Compacted Skid Trails: A Trade-off between Greenhouse Gas Fluxes and Soil Structure Recovery? Forests 2019, 10, 726. https://doi.org/10.3390/f10090726
Warlo H, von Wilpert K, Lang F, Schack-Kirchner H. Black Alder (Alnus glutinosa (L.) Gaertn.) on Compacted Skid Trails: A Trade-off between Greenhouse Gas Fluxes and Soil Structure Recovery? Forests. 2019; 10(9):726. https://doi.org/10.3390/f10090726
Chicago/Turabian StyleWarlo, Hannes, Klaus von Wilpert, Friederike Lang, and Helmer Schack-Kirchner. 2019. "Black Alder (Alnus glutinosa (L.) Gaertn.) on Compacted Skid Trails: A Trade-off between Greenhouse Gas Fluxes and Soil Structure Recovery?" Forests 10, no. 9: 726. https://doi.org/10.3390/f10090726
APA StyleWarlo, H., von Wilpert, K., Lang, F., & Schack-Kirchner, H. (2019). Black Alder (Alnus glutinosa (L.) Gaertn.) on Compacted Skid Trails: A Trade-off between Greenhouse Gas Fluxes and Soil Structure Recovery? Forests, 10(9), 726. https://doi.org/10.3390/f10090726






