Priorities for Conservation and Sustainable Use of Forest Genetic Resources in Four Mexican Pines
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources
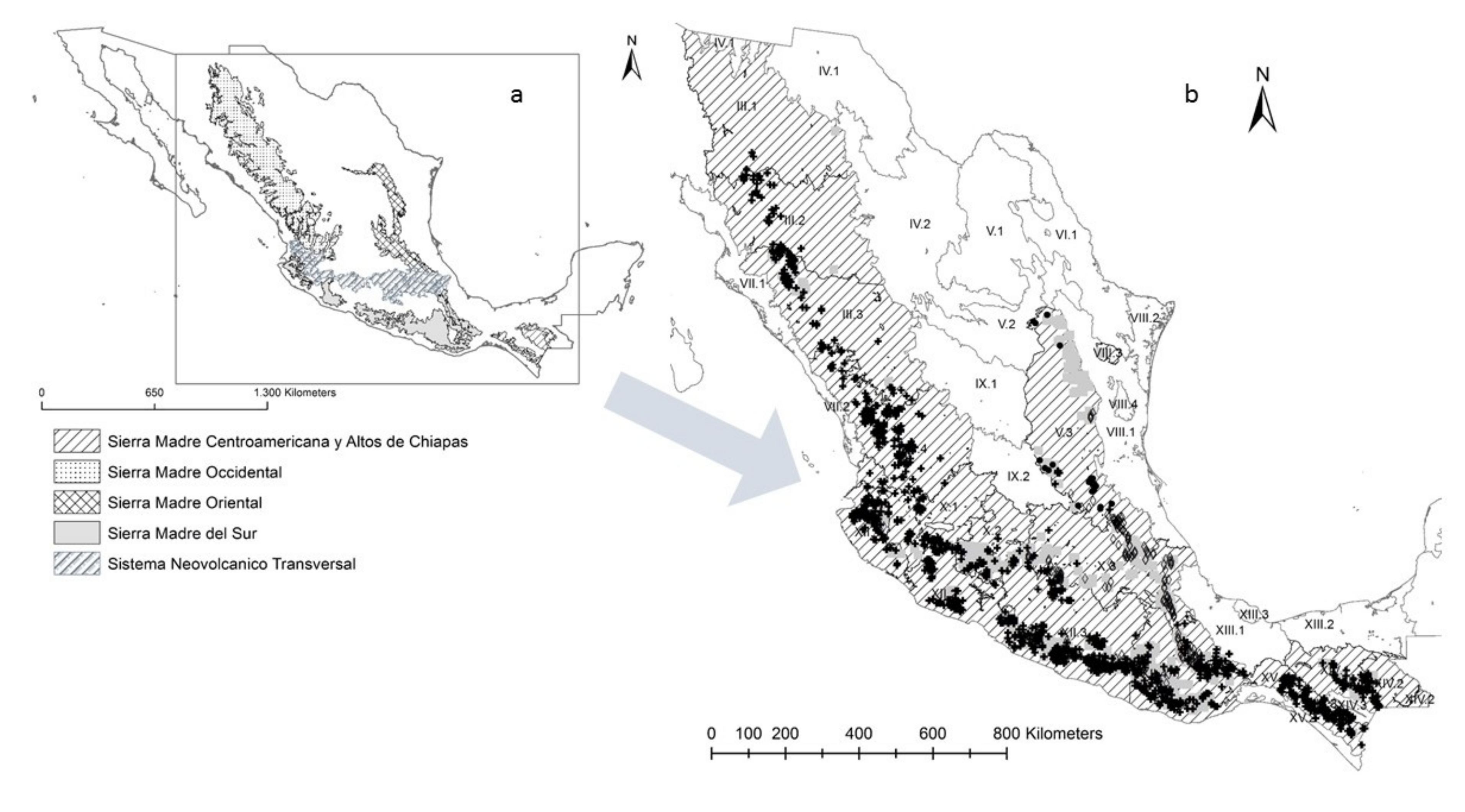
2.2. Genetic Zones
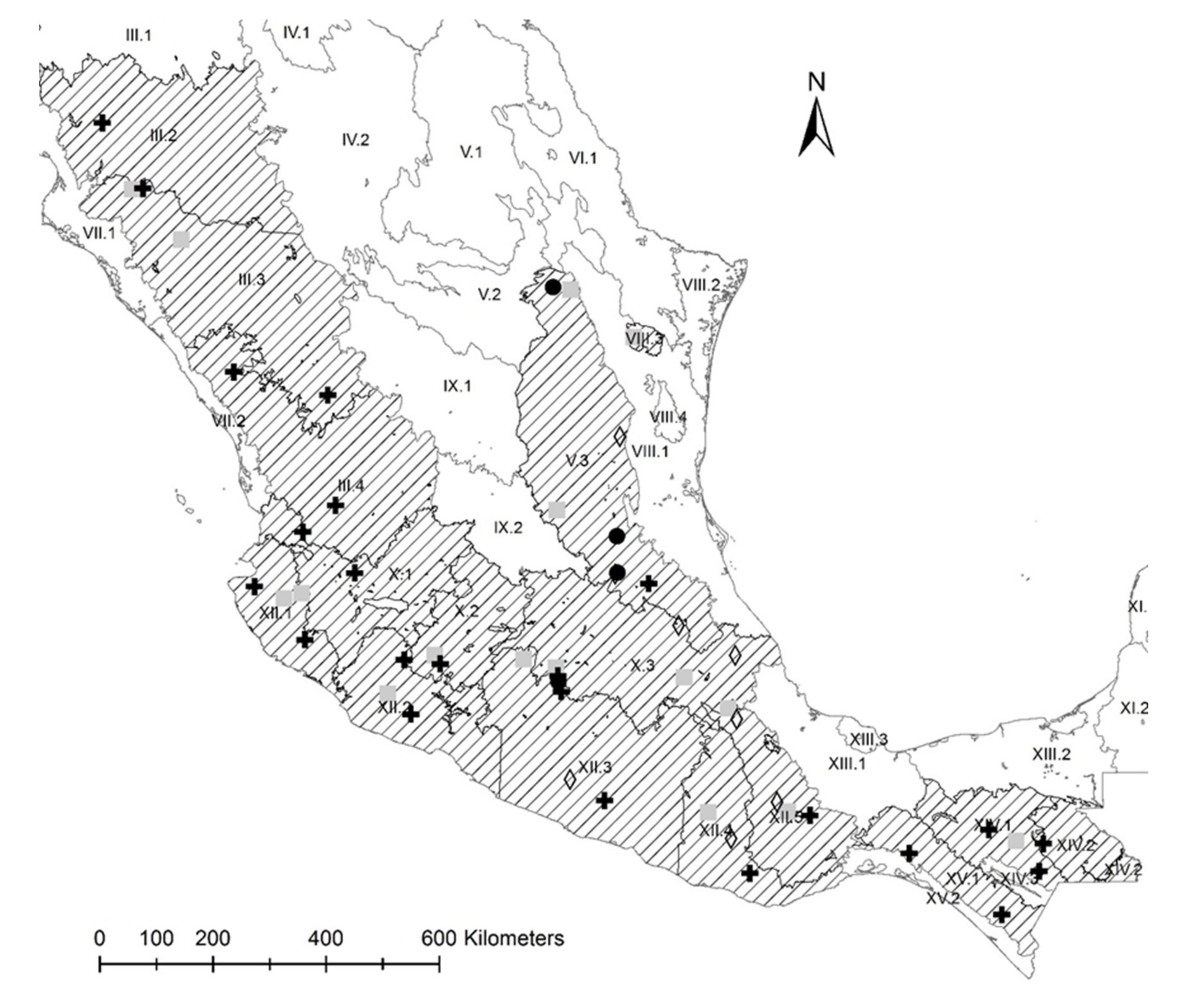
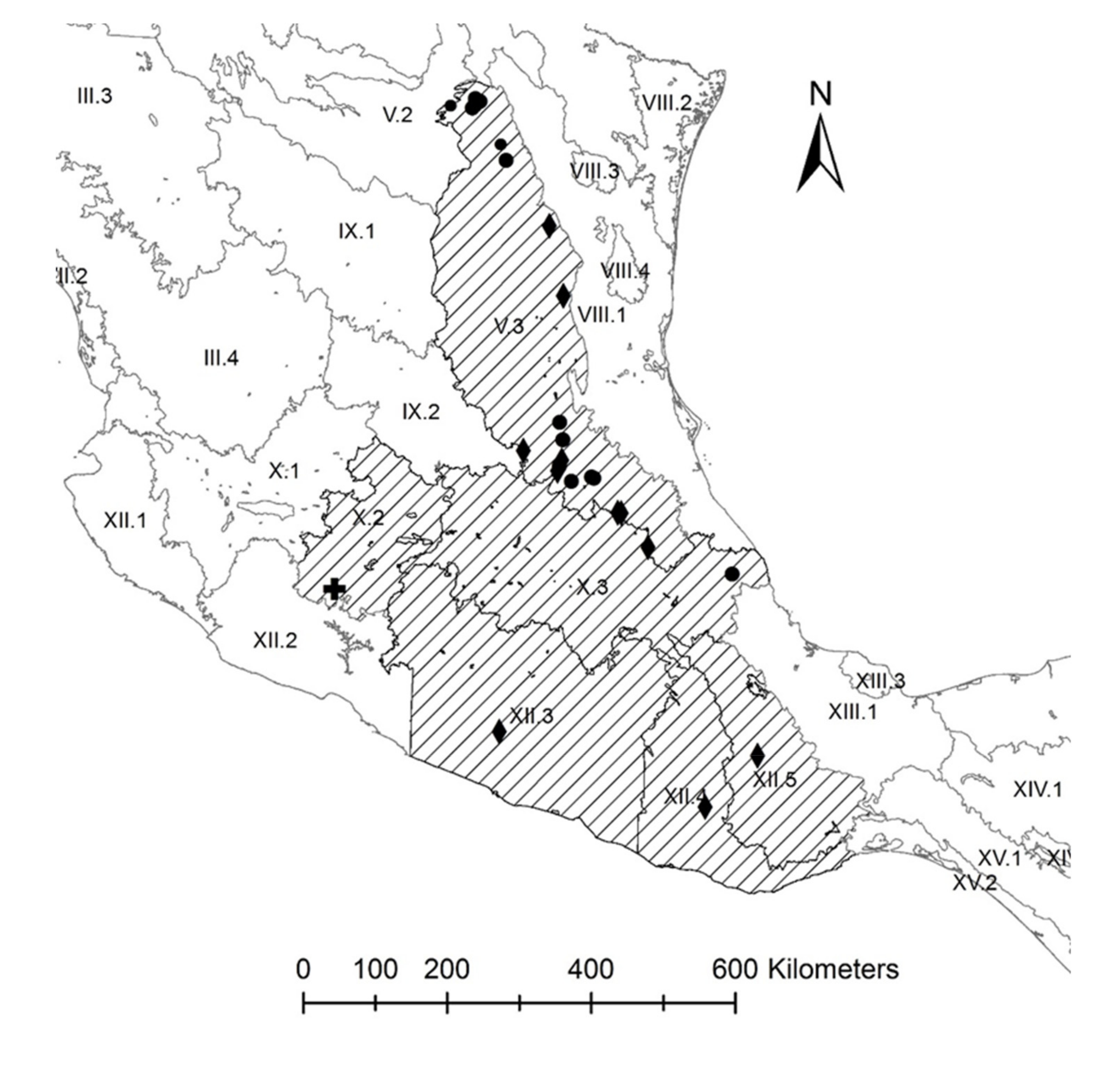
2.3. Conservation Units
2.4. Use of Genetics Resources
2.5. Importance of Genetic Zones for the Conservation and Sustainable Use of Forest Genetic Resources
3. Results
3.1. Genetic Zones
3.2. Conservation Units
3.3. Use of Genetic Resources
3.4. Efforts for Conservation and Use of Genetic Resources
3.5. Prioritization of Regions for Conservation and Use of Genetic Resources in Mexico
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. The State of the World’s Forest Genetics Resources; FAO: Rome, Italy, 2014; ISBN 978-92-5-108403-8. [Google Scholar]
- Savolainen, O.; Pyhäjärvi, T.; Knürr, T. Gene flow and local adaptation in trees. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 595–619. [Google Scholar] [CrossRef]
- Konnert, M.; Fady, B.; An Gömöry, D.; A’hara, S.; Wolter, F.; Ducci, F.; Koskela, J.; Bozzano, M.; Maaten, T.; Kowalczyk, J. Use and Transfer of Forest Reproductive Material in Europe in the Context of Climate Change; European Forest Genetic Resources Programme (EUFORGEN), Bioversity International: Rome, Italy, 2015; ISBN 9789292550318. [Google Scholar]
- De Vries, S.M.G.; Alan, M.; Bozzano, M.; Burianek, V.; Collin, E.; Cottrell, J.; Ivankovic, M.; Kelleher, C.T.; Koskela, J.; Rotach, P.; et al. Pan-European Strategy for Genetic Conservation of Forest Trees and Establishment of a Core Network of Dynamic Conservation Units; European Forest Genetic Resources Programme (EUFORGEN), Bioversity International: Rome, Italy, 2015; ISBN 9789292550295. [Google Scholar]
- Jiménez, P.; Diaz-Fernandez, P.M.; Iglesias, S.; Prada, M.A.; Garcia del Barrio, J.M.; Alba, N.; Alía, R.; Díaz-Fernández, P.M.; Iglesias, S.; Prada, A.; et al. Strategy for the Conservation and Sustainable Use of Spanish Forest Genetic Resources. For. Syst. 2009, 18, 13–19. [Google Scholar] [CrossRef]
- Rajora, O.P.; Mosseler, A. Challenges and opportunities for conservation of forest genetic resources. Euphytica 2001, 118, 197–212. [Google Scholar] [CrossRef]
- Alberto, F.J.; Aitken, S.N.; Alía, R.; González-Martínez, S.C.; Hänninen, H.; Kremer, A.; Lefèvre, F.; Lenormand, T.; Yeaman, S.; Whetten, R.; et al. Potential for evolutionary responses to climate change—Evidence from tree populations. Glob. Chang. Biol. 2013, 19, 1645–1661. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, G.; Namkoong, G.; Roberds, J.H. Dynamic gene conservation for uncertain futures. For. Ecol. Manag. 1993, 62, 15–37. [Google Scholar] [CrossRef]
- Van Buijtenen, J.P. Fundamental genetic principles. In Handbook of Quantitative Forest Genetics; Fins, L., Friedman, S., Brotschol, J., Eds.; Springer: Dordrecht, The Netherlands, 1992; pp. 29–68. ISBN 978-90-481-4112-8. [Google Scholar]
- Kanowski, P. In situ forest conservation: A broader vision for the 21 st century. In In Situ and EX Situ Conservation of Commercial Tropical Trees; Gadjah Mada University and ITTO: Yogyakarta, Indonesia, 2001; pp. 11–36. ISBN 9799665205. [Google Scholar]
- Koskela, J.; Lefèvre, F.; Schueler, S.; Kraigher, H.; Olrik, D.C.; Hubert, J.; Longauer, R.; Bozzano, M.; Yrjänä, L.; Alizoti, P.; et al. Translating conservation genetics into management: Pan-European minimum requirements for dynamic conservation units of forest tree genetic diversity. Biol. Conserv. 2013, 157, 39–49. [Google Scholar] [CrossRef]
- Hamann, A.; Smets, P.; Yanchuk, A.D.; Aitken, S.N. An ecogeographic framework for in situ conservation of forest trees in British Columbia. Can. J. For. Res. 2005, 35, 2553–2561. [Google Scholar] [CrossRef]
- Yanchuk, A.D. Techniques in forest tree breeding. In Forests and Forest Plants; Owens, J.N., Lund, H.G., Eds.; EOLSS Publications: Oxford, UK, 2009; Volume 3, pp. 40–47. ISBN 9781905839407. [Google Scholar]
- Prober, S.M.; Byrne, M.; Mclean, E.H.; Steane, D.A.; Potts, B.M.; Vaillancourt, R.E. Climate-adjusted provenancing: A strategy for climate-resilient ecological restoration. Front. Ecol. Evol. 2015, 3, 65. [Google Scholar] [CrossRef]
- Koskela, J.; Vinceti, B.; Dvorak, W.; Bush, D.; Dawson, I.K.; Loo, J.; Kjaer, E.D.; Navarro, C.; Padolina, C.; Bordács, S.; et al. Utilization and transfer of forest genetic resources: A global review. For. Ecol. Manag. 2014, 333, 22–34. [Google Scholar] [CrossRef]
- Rymer, L. Pine plantations in australia as a habitat for native animals. Environ. Conserv. 1981, 8, 95–96. [Google Scholar] [CrossRef]
- Ehrlich, P.R.; Mooney, H.A. Extinction, substitution, and ecosystem services. Bioscience 1983, 33, 248–254. [Google Scholar] [CrossRef]
- Gernandt, D.S.; Pérez-de la Rosa, J.A. Biodiversidad de Pinophyta (coníferas) en México. Rev. Mex. Biodivers. 2014, 85, 126–133. [Google Scholar] [CrossRef]
- Sánchez-González, A. Una visión actual de la diversidad y distribución de los pinos de México. Madera Bosques 2008, 14, 107–120. [Google Scholar] [CrossRef]
- Dvorak, W.S. CAMCORE: Industry and governments’ efforts to conserve threatened forest species in Guatemala, Honduras and Mexico. For. Ecol. Manag. 1990, 35, 151–157. [Google Scholar] [CrossRef]
- Wehenkel, C.; Del Rocío Mariscal-Lucero, S.; Jaramillo-Correa, J.P.; López-Sánchez, C.A.; Vargas-Hernández, J.J.; Sáenz-Romero, C. Genetic diversity and conservation of Mexican forest trees. In Biodiversity and Conservation of Woody Plants; Ahuja, M., Jain, S., Eds.; Sustainable Development and Biodiversity; Springer International Publishing: Cham, Switzerland, 2017; Volume 17, pp. 37–67. ISBN 9783642209246. [Google Scholar]
- Perry, J.P. The Pines of Mexico and Central America; Timber Press, Inc.: Portland, OR, USA, 1991; ISBN 0881921742. [Google Scholar]
- Fuentes López, M.E.; García Salazar, J.A.; Hernández Martínez, J. Factores que afectan el mercado de madera aserrada de pino en México. Madera Bosques 2006, 12, 17–28. [Google Scholar] [CrossRef]
- López-Upton, J.; Donahue, J.K.; Plascencia-Escalante, F.O.; Ramírez-Herrera, C. Provenance variation in growth characters of four subtropical pine species planted in Mexico. New For. 2005, 29, 1–13. [Google Scholar] [CrossRef]
- Dvorak, W. The strategic importance of applied tree conservation programs to the forest industry in South Africa. South. For. J. For. Sci. 2012, 74, 1–6. [Google Scholar] [CrossRef]
- Gwaze, D.P.; Dungey, H.S.; Dieters, M.J.; Toon, P.G.; Nikles, D.G. Interspecific pine hybrids. I. Genetic parameter estimates in Australia. For. Genet. 2000, 7, 11–20. [Google Scholar]
- CONAFOR Inventario Nacional Forestal y de Suelo (INFyS) 2013–2014, 32 volume collection. Available online: http://transparencia01.cnf.gob.mx/OpenData/Inventario// (accessed on 2 January 2018).
- CONAFOR Inventario Nacional Forestal y de Suelos. Informe 2004–2009. Available online: https://www.snieg.mx/DocumentacionPortal/iin/acuerdo_3_X/Manual_y_Procedimientos_para_el_Muestreo_de_Campo_INFyS_2004-2009.pdf (accessed on 2 January 2018).
- CONAFOR. Manual Para el Establecimiento de Unidades Productoras de Germoplasma Forestal; CONAFOR: Zapopan, Mexico, 2016. [Google Scholar]
- Flores, A. Phenotypic Variation among Natural Populations of Pines: Implications for the Management and Conservation of Genetic Resources. Ph.D. Thesis, Universidad de Valladolid, Valladolid, Spain, 2018. [Google Scholar]
- Secretaria de Economía Declaratoria de vigencia de la Norma Mexicana NMX-AA-169-SCFI-2016. Available online: http://dof.gob.mx/nota_detalle.php?codigo=5455455&fecha=03/10/2016 (accessed on 3 November 2017).
- Flores Flores, C.; López-Upton, J.; Valencia Manzo, S. Manual Técnico Para el Establecimiento de Ensayos de Procedencias y Progenies; CONAFOR: Zapopan, Mexico, 2014. [Google Scholar]
- Ledig, F.T. The Conservation of Diversity in Forest Trees. Bioscience 1988, 38, 471–479. [Google Scholar] [CrossRef]
- ESRI ArcGIS Desktop: Release 10. 2011. Available online: http://desktop.arcgis.com/es/arcmap/ (accessed on 10 June 2019).
- INEGI Mexico National Institute of Statistics, Geography and Informatics. Available online: https://www.inegi.org.mx/ (accessed on 11 January 2018).
- SEMARNAT Plantation area. Available online: https://datos.gob.mx/busca/dataset/indicadores-basicos-del-desempeno-ambiental--recursos-forestales (accessed on 2 January 2018).
- CONAFOR Zonificación forestal. Available online: https://snigf.cnf.gob.mx/zonificacion/ (accessed on 1 January 2018).
- EUFORGEN. Revised indicator on genetic resources (4.6) of the pan-European criteria and indicators for sustainable forest management. REPORT of EUFORGEN working group. Unpublished work. 2019. [Google Scholar]
- Villers-Ruiz, L.; Trejo-Vázquez, I. Impacto del cambio climático en los bosques y áreas naturales protegidas de México. Interciencia 1998, 23, 10–19. [Google Scholar]
- Rojas-Soto, O.R.; Sosa, V.; Ornelas, J.F. Forecasting cloud forest in eastern and southern Mexico: Conservation insights under future climate change scenarios. Biodivers. Conserv. 2012, 21, 2671–2690. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Snively, A.E.; Lindig-Cisneros, R. Conservation and restoration of pine forest genetic resources in México. Silvae Genet. 2003, 52, 233–237. [Google Scholar]
- Merino-Perez, L. Community action for conservation: Mexican experiences. In Community Action for Conservation: Mexican Experiences; Porter-Bolland, L., Ruiz-Mallén, I., Camacho-Benavides, C., McCandless, S.R., Eds.; Springer Science & Business Media: New York, NY, USA, 2013; pp. 25–44. ISBN 9781461479567. [Google Scholar]
- Parraguirre, L.C.; Vargas, H.J.J.; Ramírez, V.P.; Azpíroz, R.H.S.; Jasso, M.J. Estructura de la diversidad Genética en poblaciones naturales de Pinus greggü Engelm. Rev. Fitotec. Mex. 2002, 25, 279–287. [Google Scholar]
- Hernández-Velasco, J.; Hernández-Díaz, J.C.; Fladung, M.; Cañadas-López, Á.; Prieto-Ruíz, J.Á.; Wehenkel, C. Spatial genetic structure in four Pinus species in the Sierra. Can. J. For. Res. 2017, 47, 73–80. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Guzmán-Reyna, R.R.; Rehfeldt, G.E. Altitudinal genetic variation among Pinus oocarpa populations in Michoacán, Mexico. For. Ecol. Manag. 2006, 229, 340–350. [Google Scholar] [CrossRef]
- Viveros-Viveros, H.; Sáenz-Romero, C.; Vargas-Hernández, J.J.; López-Upton, J.; Ramírez-Valverde, G.; Santacruz-Varela, A. Altitudinal genetic variation in Pinus hartwegii Lindl I: Height growth, shoot phenology, and frost damage in seedlings. For. Ecol. Manag. 2009, 257, 836–842. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Rehfeldt, G.E.; Soto-Correa, J.C.; Aguilar-Aguilar, S.; Zamarripa-Morales, V.; López-Upton, J. Altitudinal genetic variation among Pinus pseudostrobus populations from Michoacán, México. Two location shadehouse test results. Rev. Fitotec. Mex. 2012, 35, 111–120. [Google Scholar]
- Loya-Rebollar, E.; Sáenz-Romero, C.; Lindig-Cisneros, R.A.; Lobit, P.; Villegas-Moreno, J.A.; Sánchez-Vargas, N.M. Clinal variation in Pinus hartwegii populations and its application for adaptation to climate change. Silvae Genet. 2013, 62, 86–95. [Google Scholar] [CrossRef]
- Ruiz-Talonia, L.F.; Sánchez-Vargas, N.M.; Bayuelo-Jiménez, J.S.; Lara-Cabrera, S.I.; Sáenz-Romero, C. Altitudinal genetic variation among native Pinus patula provenances: Performance in two locations, seed zone delineation and adaptation to climate change. Silvae Genet. 2014, 63, 139–149. [Google Scholar] [CrossRef]
- Ortiz-Bibian, M.A.; Blanco-García, A.; Lindig-Cisneros, R.A.; Gómez-Romero, M.; Castellanos-Acuña, D.; Herrerías-Diego, Y.; Sánchez-Vargas, N.M.; Sáenz-Romero, C. Genetic variation in Abies religiosa for quantitative traits and delineation of elevational and climatic zoning for maintaining monarch butterfly overwintering sites in Mexico, considering climatic change. Silvae Genet. 2017, 66, 14–23. [Google Scholar] [CrossRef]
- Ramírez-Herrera, C.; Vargas-Hernández, J.J.; López-Upton, J. Distribución y conservación de las poblaciones naturales de Pinus greggii. Acta Botánica Mex. 2005, 72, 1–16. [Google Scholar] [CrossRef]
- Flores, A.; Climent, J.; Pando, V.; López-Upton, J.; Alía, R. Intraspecific variation in pines from the Trans-Mexican Volcanic Belt grown under two watering regimes: Implications for management of genetic resources. Forests 2018, 9, 71. [Google Scholar] [CrossRef]
- Koski, V.; Skrøppa, T.; Paule, L.; Wolf, H.; Turok, J. Technical Guidelines for Genetic Conservation of Norway Spruce (Picea Abies (L.) Karst.); International Plant Genetic Resources Institute: Rome, Italy, 1997; ISBN 92-9043-331-X. [Google Scholar]
- Wallace, J.; Aquilué, N.; Archambault, C.; Carpentier, S.; Francoeur, X.; Greffard, M.H.; Laforest, I.; Galicia, L.; Messier, C. Present forest management structures and policies in temperate forests of Mexico: Challenges and prospects for unique tree species assemblages. For. Chron. 2015, 91, 306–317. [Google Scholar] [CrossRef][Green Version]
- National Research Council In situ Conservation of Genetic Resources. Managing Global Genetic Resources: Agricultural Crop Issues and Policies; The National Academies Press: Washington, DC, USA, 1993; p. 451. ISBN 978-0-309-04430-1. [Google Scholar]
- Aravanopoulos, F.A. Conservation and monitoring of tree genetic resources in temperate forests. Curr. For. Rep. 2016, 2, 119–129. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Rehfeldt, G.E.; Crookston, N.L.; Duval, P.; St-Amant, R.; Beaulieu, J.; Richardson, B.A. Spline models of contemporary, 2030, 2060 and 2090 climates for Mexico and their use in understanding climate-change impacts on the vegetation. Clim. Chang. 2010, 102, 595–623. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Lindig-Cisneros, R.A.; Joyce, D.G.; Beaulieu, J.; Clair, J.B.S.; Jaquish, B.C. Assisted migration of forest populations for adapting trees to climate change. Rev. Chapingo Ser. Cienc. For. Ambiente 2016, 22, 303–323. [Google Scholar]
- Castellanos-Acuña, D.; Vance-Borland, K.W.; Clair, J.B.S.; Hamann, A.; López-Upton, J.; Gómez-Pineda, E.; Ortega-Rodríguez, J.M.; Sáenz-Romero, C. Climate-based seed zones for Mexico: Guiding reforestation under observed and projected climate change. New For. 2017, 49, 297–309. [Google Scholar] [CrossRef]
- El-Kassaby, Y.A.; Lstiburek, M. Breeding without Breeding. Genet. Res. 2009, 91, 111–120. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Tapia-Olivares, B.L. Genetic variation in frost damage and seed zone delineation within an altitudinal transect of Pinus devoniana (P. michoacana) in Mexico. Silvae Genet. 2008, 57, 165–170. [Google Scholar] [CrossRef]
- Burney, O.; Aldrete, A.; Alvarez Reyes, R.; Prieto Ruíz, J.A.; Sánchez Velazquez, J.R.; Mexal, J.G. Mexico-addressing challenges to reforestation. J. For. 2015, 113, 404–413. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Lindig-Cisneros, R. Evaluación y propuestas para el programa de reforestación en Michoacán. Cienc. Nicolaıta 2004, 37, 107–122. [Google Scholar]
- Stead, J.W.; Styles, B.T. Studies of Central American pines: A revision of the ‘pseudostrobus’ group (Pinaceae). Bot. J. Linn. Soc. 1984, 89, 249–275. [Google Scholar] [CrossRef]
| Indicator | Description | |
|---|---|---|
| I1 | Number of genetic zones | Number of genetic zones with presence of the species |
| I2 | Molecular characterization effort | % of genetic zones with at least 1 sample in molecular studies |
| I3 | Provenance characterization effort | % of genetic zones with at least 1 population in provenance test |
| I4 | Progeny characterization effort | % of genetic zones with plus trees in progeny tests |
| I5 | Seed stands index | % of genetic zones with at least 1 forest seed production units |
| I6 | Seed orchard index | % of genetic zones with at least 1 seed orchard |
| I7 | Genetic conservation index | % of genetic zones with at least 1 conservation units identified |
| Genetic Zone | Trees National Forest Inventory (ni) | Molecular Characterization (nmk) | Seed Stands (nst) | Progeny Testing (nis) | Provenance Testing (npt) | Seed Orchards (nso) | Gene Conservation Units (ng) | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PS | OC | PA | GR | PS | OC | PA | GR | PS | OC | PA | GR | PS | OC | PA | GR | PS | OC | PA | GR | PS | OC | PA | GR | PS | OC | PA | GR | |
| III.2 | 55 | 587 | - | - | 0 | 1 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 1 | - | - |
| III.3 | 199 | 593 | - | - | 0 | 1 | - | - | 0 | 1 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 2 | - | - |
| III.4 | - | 1201 | - | - | - | 3 | - | - | - | 1 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 2 | - | - |
| V.3 | 1136 | 56 | 538 | 308 | 0 | 2 | 3 | 19 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 | 22 | 0 | 0 | 0 | 0 | 2 | 1 | 2 | 3 |
| VIII.3 | 27 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 1 | - | - | - |
| X.1 | 61 | 699 | - | - | 0 | 1 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 2 | - | - |
| X.2 | 777 | 581 | 126 | - | 2 | 8 | 0 | - | 2 | 1 | 0 | - | 13 | 0 | 0 | - | 0 | 5 | 0 | - | 0 | 5 | 0 | - | 1 | 1 | 0 | - |
| X.3 | 1262 | 177 | 1028 | 23 | 4 | 2 | 1 | 0 | 8 | 0 | 5 | 0 | 0 | 0 | 36 | 120 | 0 | 0 | 4 | 2 | 0 | 0 | 1 | 0 | 2 | 1 | 2 | 1 |
| XII.1 | 240 | 1222 | - | - | 0 | 1 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 2 | - | - |
| XII.2 | 268 | 1503 | - | - | 1 | 1 | - | - | 1 | 2 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 2 | - | - |
| XII.3 | 608 | 2650 | 81 | - | 1 | 2 | 1 | - | 0 | 2 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 1 | 2 | 1 | - |
| XII.4 | 1944 | 1534 | 66 | - | 0 | 1 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 1 | 1 | 1 | - |
| XII.5 | 1122 | 924 | 820 | - | 0 | 0 | 7 | - | 1 | 0 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 0 | 0 | 0 | - | 2 | 1 | 2 | - |
| XIV.1 | 88 | 404 | - | - | 1 | 4 | - | - | 1 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 0 | 0 | - | - | 1 | 2 | - | - |
| XIV.2 | - | 312 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 1 | - | - |
| XV.1 | - | 1123 | - | - | - | 3 | - | - | - | 1 | - | - | - | 0 | - | - | - | 0 | - | - | - | 0 | - | - | - | 2 | - | - |
| Total | 7787 | 13566 | 2659 | 331 | 9 | 30 | 12 | 19 | 14 | 8 | 5 | 0 | 13 | 0 | 36 | 120 | 0 | 5 | 9 | 24 | 0 | 0 | 1 | 0 | 16 | 23 | 8 | 4 |
| Indicator | P. pseudostrobus | P. oocarpa | P. patula | P. greggii | |
|---|---|---|---|---|---|
| I1 | Number genetic zones | 13 | 15 | 6 | 2 |
| I2 | Molecular characterization effort | 38.46 | 86.66 | 66.66 | 50.00 |
| I3 | Provenance characterization effort | 0.00 | 6.66 | 33.33 | 100.00 |
| I4 | Progeny characterization effort | 7.69 | 0.00 | 16.66 | 50.00 |
| I3 | Seed stands index | 46.15 | 40.00 | 16.66 | 0.00 |
| I6 | Seed orchard index | 0.00 | 0.00 | 16.66 | 0.00 |
| I7 | Genetic conservation index * | 100.00 | 100.00 | 83.33 | 100.00 |
| Genetic Zone | Presence of the Pine Species | Value for Conservation (1 to 4) | Value for Breeding (1 to 4) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| PS | OC | PA | GR | PS | OC | PA | GR | ||
| III.2 | 50 | 4 | 2 | - | - | 1 | 2 | - | - |
| III.3 | 88 | 3 | 2 | - | - | 2 | 2 | - | - |
| III.4 | 137 | - | 2 | - | - | - | 3 | - | - |
| V.3 | 143 | 1 | 4 | 4 | 4 | 3 | 1 | 4 | 4 |
| VIII.3 | 2 | 4 | - | - | - | 1 | - | - | - |
| X.1 | 66 | 3 | 2 | - | - | 1 | 2 | - | - |
| X.2 | 66 | 2 | 4 | 2 | - | 3 | 4 | 2 | - |
| X.3 | 144 | 1 | 4 | 4 | 4 | 3 | 1 | 4 | 4 |
| XII.1 | 93 | 3 | 2 | - | - | 2 | 3 | - | - |
| XII.2 | 114 | 3 | 2 | - | - | 2 | 3 | - | - |
| XII.3 | 300 | 2 | 2 | 4 | - | 2 | 3 | 4 | - |
| XII.4 | 213 | 1 | 2 | 4 | - | 3 | 3 | 4 | - |
| XII.5 | 188 | 1 | 2 | 4 | - | 3 | 3 | 4 | - |
| XIV.1 | 60 | 4 | 3 | - | - | 1 | 2 | - | - |
| XIV.2 | 18 | - | 3 | - | - | - | 2 | - | - |
| XV.1 | 109 | - | 2 | - | - | - | 3 | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores, A.; López-Upton, J.; Rullán-Silva, C.D.; Olthoff, A.E.; Alía, R.; Sáenz-Romero, C.; Garcia del Barrio, J.M. Priorities for Conservation and Sustainable Use of Forest Genetic Resources in Four Mexican Pines. Forests 2019, 10, 675. https://doi.org/10.3390/f10080675
Flores A, López-Upton J, Rullán-Silva CD, Olthoff AE, Alía R, Sáenz-Romero C, Garcia del Barrio JM. Priorities for Conservation and Sustainable Use of Forest Genetic Resources in Four Mexican Pines. Forests. 2019; 10(8):675. https://doi.org/10.3390/f10080675
Chicago/Turabian StyleFlores, Andrés, Javier López-Upton, Cristobal D. Rullán-Silva, Adriana E. Olthoff, Ricardo Alía, Cuauhtémoc Sáenz-Romero, and José M. Garcia del Barrio. 2019. "Priorities for Conservation and Sustainable Use of Forest Genetic Resources in Four Mexican Pines" Forests 10, no. 8: 675. https://doi.org/10.3390/f10080675
APA StyleFlores, A., López-Upton, J., Rullán-Silva, C. D., Olthoff, A. E., Alía, R., Sáenz-Romero, C., & Garcia del Barrio, J. M. (2019). Priorities for Conservation and Sustainable Use of Forest Genetic Resources in Four Mexican Pines. Forests, 10(8), 675. https://doi.org/10.3390/f10080675






