1. Introduction
The profitability of hybrid poplar biomass production and its biochemical conversion to transportation fuels is dependent on genetically improved interspecific varieties. Both farm and refinery economics are influenced by genetic gains in biomass yield [
1,
2] and biomass quality, the latter manifested in the ease of cell wall deconstruction and sugar release during pretreatment and hydrolysis [
3,
4,
5]. The improvement cycle is not trivial, encompassing up to seven years to complete the process of hybridization, clonal propagation, and yield testing through the first coppice cycle and laboratory analyses of biomass composition and conversion efficiency. Additionally, once superior varieties have been identified, it remains for them to be multiplied to levels for widespread initial varietal deployment. Considering the density of bioenergy plantations approximates 3500–4500 stems per hectare (ha) and the sizable acreage that needs to be cultivated to produce the tonnage required by refinery operations, the time to fully expand the supply of planting stock of newly-selected varieties impedes the expeditious delivery of gains in yield and conversion efficiency. While traditional nursery propagation may take seven years and 150 ha to introduce a new varietal into a moderately sized plantation (i.e., 10,000 ha), a combination of laboratory and greenhouse propagation has demonstrated a condensed delivery timeline [
6]. An argument for such rapid multiplication propagation techniques as indispensable to the initial propagation of new hybrid varieties for bioenergy farms has been made since the beginnings of clonal forestry [
7].
Commercial biomass farms employing interspecific poplar hybrids between sections Aigeiros and Tacamahaca are normally planted as clonal stands using inexpensive one-year-old hardwood cuttings that establish in the field by formation of adventitious roots that elongate from initials formed on nursery stock the previous growing season during shoot development. Although measurable genetic variance in vegetative propagation has been reported, field rooting of Aigeiros × Tacamahaca hybrid taxa using hardwood cuttings is a generally reliable propagation method [
8]. However, for the initial scaling of clonal selections, varieties are rapidly multiplied in greenhouses using succulent cuttings rooted under mist propagation in soil or hydroponically to produce containerized planting stock [
9,
10]. Alternatively, in vitro micropropagation systems of exceedingly greater capacity are available, albeit costly [
11,
12]. Recent micropropagation research—producing plantlets in liquid-phase bioreactors for ex vitro greenhouse rooting—may ultimately prove cost-effective for industrial clonal propagation [
13], but as currently practiced, micropropagation is prohibitively expensive for commercial planting stock quantities [
14]. Until the affordability of in vitro systems is proven, the rapid multiplication technique as developed in Finland for hybrid aspen (
Populus ×
wettsteinii Hämet-Ahti) may be the most cost-effective avenue for scaling new selections [
15]. This approach utilizes in vitro micro-cuttings that are produced in laboratories, rooted in greenhouses, and subsequently expanded by hedge propagation of macro-cuttings to generate multiple serial propagation tracks. In vitro propagation takes place in a laboratory using culture media and glass vessels [
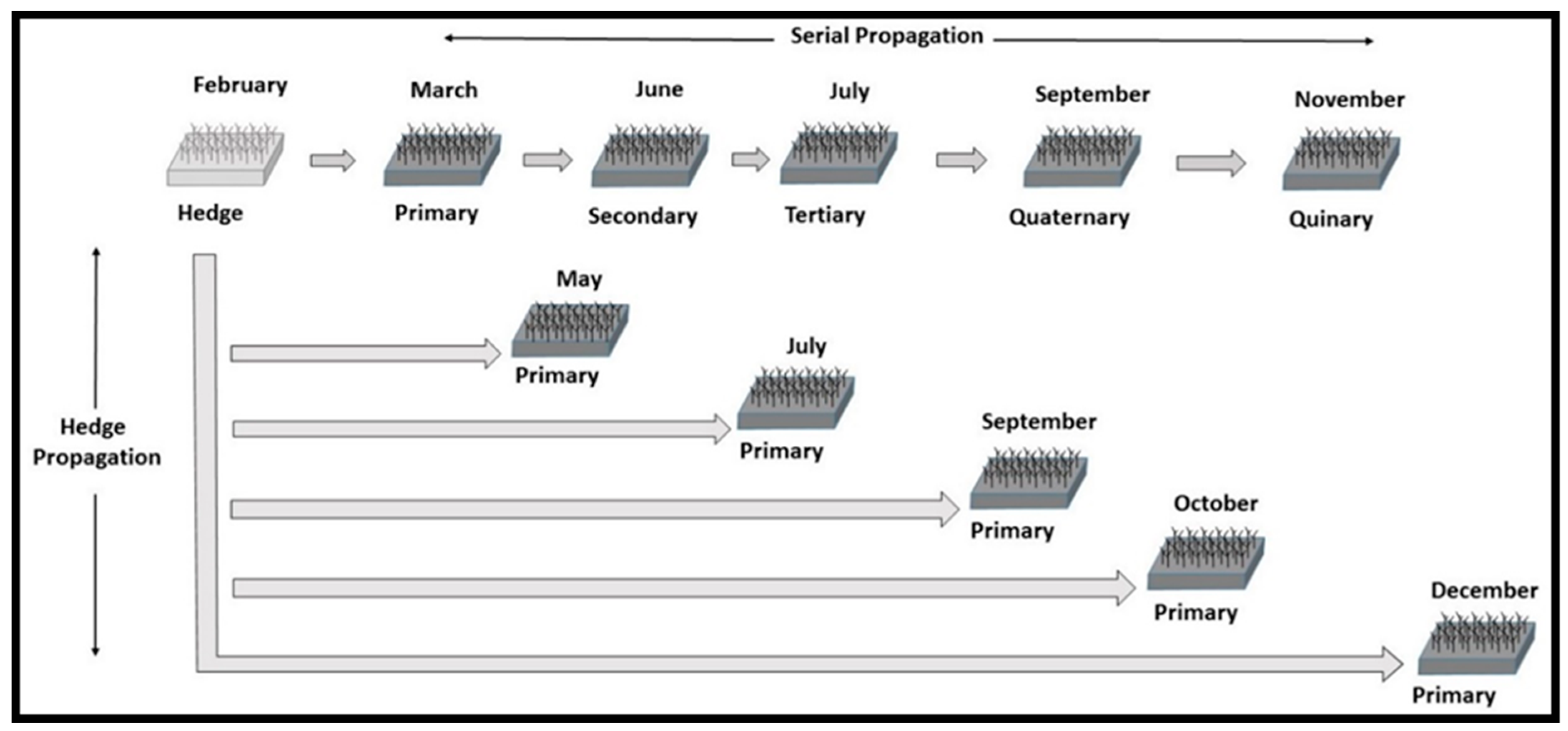
16]. Hedge propagation is the continuous harvesting of re-sprouting plants for re-planting under greenhouse conditions. Serial propagation begins with greenhouse propagation of cuttings that become primary ramets, from which cuttings are collected that become secondary ramets that lead to tertiary ramets and so on.
The economics of this combined approach has not been studied for the Aigeiros × Tacamahaca taxa, although generally recognized to be more expensive than conventional nursery propagation. A factorial study was therefore conducted to assess the economics of a rapid multiplication method based on the propagation of micro- and macro-cuttings for Aigeiros × Tacamahaca hybrid varieties patterned on the Finnish
P. ×
wettsteinii model. For this paper, micro-cuttings are shoots produced through multiple rounds of in vitro propagation from explants containing a shoot-tip or nodal meristem. Conversely, shoots produced using ex vitro greenhouse rooting techniques for containerized planting stock are referred to as macro-cuttings in this study. The intent of the investigation was to: (1) Quantify the efficiency of greenhouse hedge and serial macro-cutting multiplication initiated with in vitro micro-cuttings for several varieties of diverse taxa and (2) determine the profitability of greenhouse rapid multiplication as a function of genetic gains in biomass yield and biomass quality using the internal rate of return as a standard metric of economic performance [
17].
4. Discussion
The present study looked at returns that accrue over a 20-year rotation when rapid multiplication is used to produce planting stock of new varieties directly for operations, compared with the coincident use of less expensive hardwood cuttings to initiate planting with standard varieties that are available in commercial quantities. Thus, this comparison focused on the last component of the entire tree improvement cycle when newly-released varieties are propagated en masse as containerized rooted cuttings to originate a bioenergy planting that later transitions to serial coppice regeneration. An implicit assumption is that if the route of rapid propagation of new varieties is chosen, that process takes place at the same time that land is acquired and prepared, meaning that the new varieties are available at the corresponding time that standard varieties are purchased from a commercial nursery. Returns were estimated under both options over the course of a 20-year rotation. Research costs in developing new hybrids were not factored into the analysis, reasoning that a timber investment management organization produces proprietary varieties as leverage in raising capital into its bioenergy investment funds.
Investments into hybrid poplar bioenergy operations at all four AHB regions with current commercial varieties were previously shown to generate insufficient returns to attract private-sector capital and indicated the need for marked increases in biomass pricing and yields [
19]. Other AHB analyses have suggested that the adoption of poplar feedstock operations is also impacted by the opportunity cost and the demand for competing crop commodities [
26]. Despite the influence of biomass yields, selling prices and alternative crop options, dedicated energy crops are nonetheless expected to provide a significant component of biorefinery feedstock supply in the AHB region at a benchmark biofuel price of
$19.6 GJ
−1 [
27].
Rapid multiplication was clearly superior to the base case at the two highest rates of yield irrespective of market pricing and region. For investments into rapid multiplication to make sense at the Washington, Oregon, and Idaho locations within the BTS range, yields of 22.5 Mg ha−1 year−1 and above would be needed for the most part. Conversely, a decision to forego investment into rapid multiplication in favor of the base case analysis of current commercial varieties would be generally expected at yields of 18–22.5 Mg−1 year−1 at all but the California location; this is true especially at market prices above the BTS range. However, it is doubtful whether current commercial hybrid poplar varieties that have been bred and selected for markets other than bioenergy would have the preferred biomass chemistry that would incentivize the market to offer a premium represented by prices above the BTS range. The regional influence of the study was noteworthy: The most favorable investment analysis, in terms of the overall mean and number of positive IRRs, was noted for the two non-irrigated farms, Washington and Oregon; of the two, the former is pasture land with the lowest lease rate in the study, while the Oregon farm was situated on cropland that commanded a higher lease rate. The two dryland farms requiring irrigation-Idaho and California-had lower overall IRRs and counts of positive returns. The California farm with the highest lease rate in the study exhibited the least favorable profile of returns under both the rapid multiplication and the base case. Returns at California under the base case were uniformly negative, and appreciable returns for the most part under the rapid multiplication case were not seen until yields of 27 and 31.5 Mg−1 year−1 at market prices above the BTS range.
The argument for rapid multiplication is that the exploitation of gains in biomass yield and chemistry requires a swift and seamless introduction of those gains into operations that regenerate by frequent coppicing to justify the expense of propagation [
7]. However, are the genetic gains in yield and feedstock quality presented in this study realistic as requisites to poplar bioenergy farms investment? They may be. Poplar growth and yield simulations support the yields modeled in this study up to 22.5 to 27.0 Mg ha
−1 year
−1 [
25,
28,
29], and preliminary estimates from bioenergy plantation density trials of 67 hybrid poplar varieties support theoretical yields of 35 Mg ha
−1 year
−1 for the most productive varieties [
30]. Achieving yields above this level through ongoing hybridization is conceivable based on projections of 90% biomass gains for hybrid poplar when using reciprocal, intraspecific recurrent breeding in advance of first generation interspecific hybridization and within-family clonal selection [
31]. Similarly, to accomplish improvements in biomass quality, gains in cell wall deconstruction, sugar release and biomass hydrolyzability may be anticipated using genomic selection of naturally-occurring mutant alleles in the lignin biosynthetic pathway [
32,
33,
34], as well as lignin transformation [
35,
36], with the expectation that low-lignin biomass may significantly reduce the operating cost of mechanical and chemical pre-treatment. To illustrate, research at Oak Ridge National Laboratory’s Bioenergy Research Center demonstrated a 15% increase in sugar yield associated with the biochemical conversion of low-lignin poplar varieties [
37]. AHB research confirms this with reports of increases of 19% in sugar yield leading to 10% increases in biorefinery revenue that accompany individual hybrid selections [
38]. However, it should be recognized that achieving these gains in large-scale plantations where soil quality and water supply vary at the landscape scale will be challenging.
Although gains in both biomass quantity and quality are likely, genetic improvement in the former is preferred from the perspective of an investor into biomass farms: Farm management is far better positioned to exploit increases in yield rather than bargaining the price at which the biomass is sold in to the energy markets. Increased yield directly provides incremental revenue, while an increase in price could be difficult to negotiate to the investor’s favor, recognizing that biorefineries may not be able to adjust their processes to specific biomass characteristic-the superiority of cell wall chemistry notwithstanding-when managing a diversity of cellulosic feedstock sources.
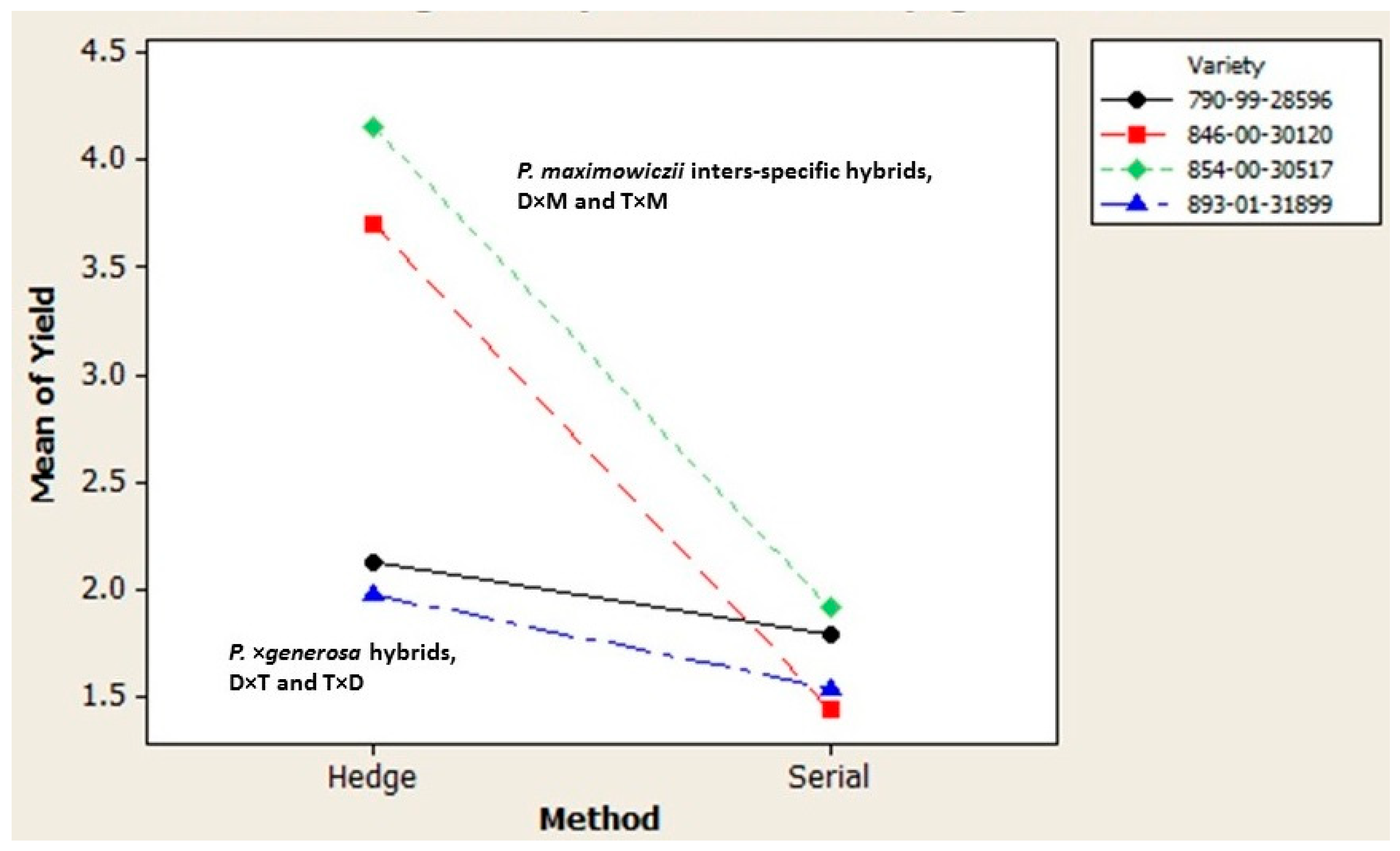
Beyond the genetic gains that strengthen the attractiveness of rapid multiplication, its appeal may be improved per se by exploiting differences in the varietal performance during both the in vitro and greenhouse propagation phases [
39,
40,
41]. For instance, the statistically-significant yield differences in macro-cutting propagation noted can be exploited to optimize the system, as recognized elsewhere [
42]. This is especially true of the
P. maximowiczii interspecific varieties and their strong response to hedge propagation that increased cutting yield by approximately twofold. The superior greenhouse performance of the
P. maximowiczii hybrids mirrored their exceptional height growth and extremely low rates of sylleptic branching that maximizes nursery production of hardwood cuttings. Beyond these manipulations, the process can be shortened considerably using test trees from final stage yield trials to collect succulent macro-cuttings during spring shoot extension which are then used to initiate greenhouse propagation circumventing the in vitro production of micro-cuttings with cost and time savings.
Finally, there are other valuable features of the rapid multiplication system associated with the use of containerized rooted planting stock that reduce the risk of plantation failures. First, vagaries of the weather-frost injury and drought-are much more easily mitigated when planting stock is grown in controlled greenhouse environments in comparison to field nurseries. Second, greenhouse production of newly-deployed varieties allows for far greater flexibility than field nurseries when turning over and initiating new varieties in to scale-up operations when, for example, such varieties must be removed from production due to a loss of pest resistance or a change in market preference. Third, rooted cuttings greatly reduce the risk of planting failures compared to the risk incurred using unrooted hardwood cuttings when an unfavorable environment-protracted dry or cold periods-is encountered during the time of establishment.
5. Conclusions
1. No variety failed to respond to in vitro micro-cutting propagation or greenhouse macro-cutting propagation.
2. The Populus deltoides × P. maximowiczii and the Populus trichocarpa × P. maximowiczii varieties greatly exceeded the multiplication rate of the P. × generosa varieties under greenhouse propagation. This was largely due to their exceptional response to repeated hedging required to initiate multiple tracks of serial propagation. It mirrored the stronger performance of the P. maximowiczii taxon under traditional stoolbed culture.
3. Rapid multiplication was clearly superior to the base case at the two highest rates of yield irrespective of market pricing and region. For investments into rapid multiplication to make sense at the Washington, Oregon, and Idaho locations within the BTS range, yields of 22.5 Mg ha−1 year−1 and above would be needed for the most part.
4. Conversely, a decision to forego investment into rapid multiplication in favor of the base case analysis of current commercial varieties would be generally expected at yields of 18.0–22.5 Mg−1 year−1 at all but the California location; this is true especially at market prices above the BTS range. However, it is doubtful whether current commercial hybrid poplar varieties for which preferred biomass chemistry has not been an improvement emphasis would justify market prices above the BTS range.