Twenty-Five Years of Aboveground Biomass and Carbon Accumulation Following Extreme Wind Damage in an Old-Growth Forest
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Methods
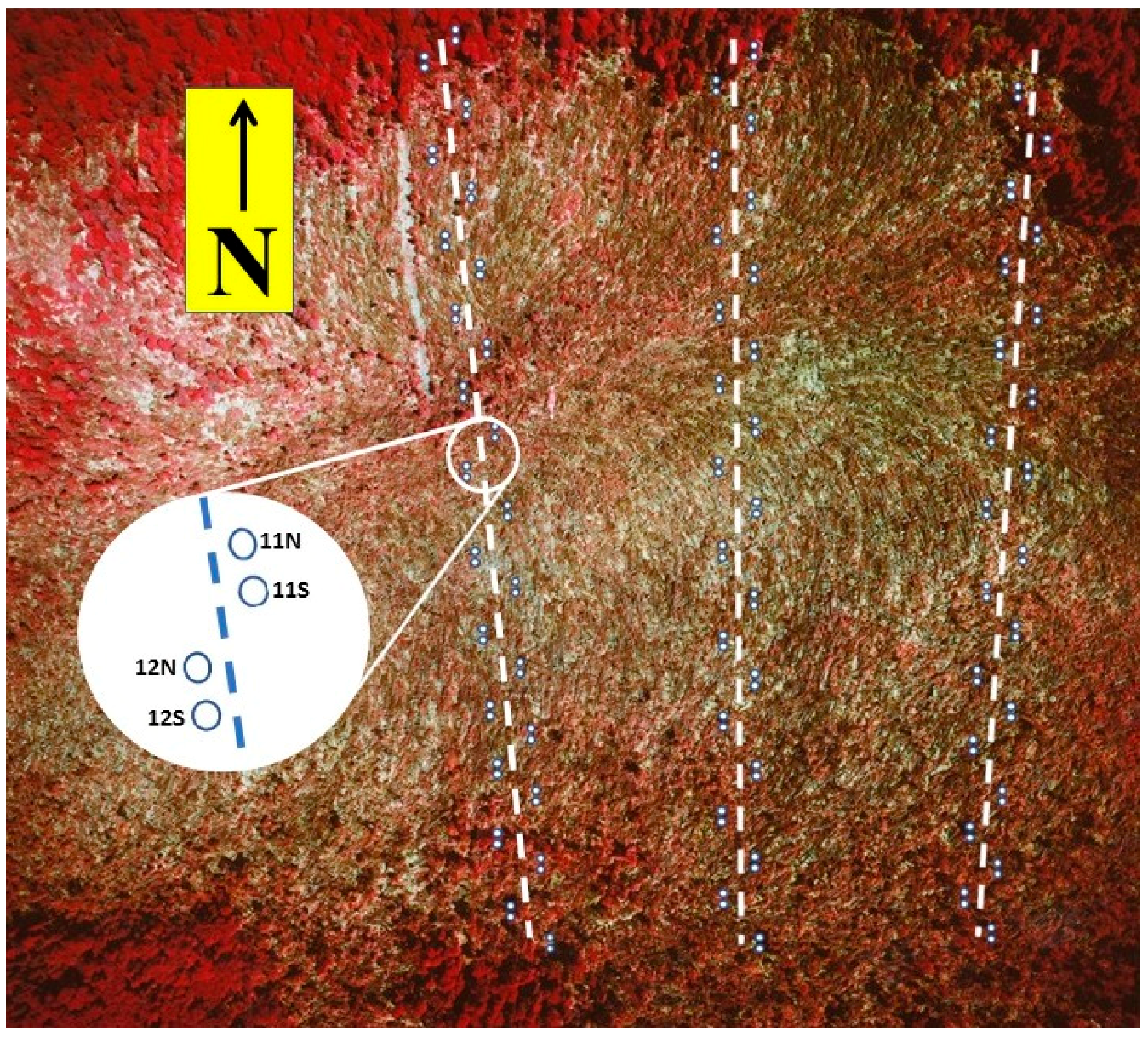
2.2.1. Field Sampling
2.2.2. Estimation of Intact Forest Aboveground Biomass
2.2.3. Estimation of Survivor Tree Biomass
2.2.4. Estimation of Regeneration Biomass
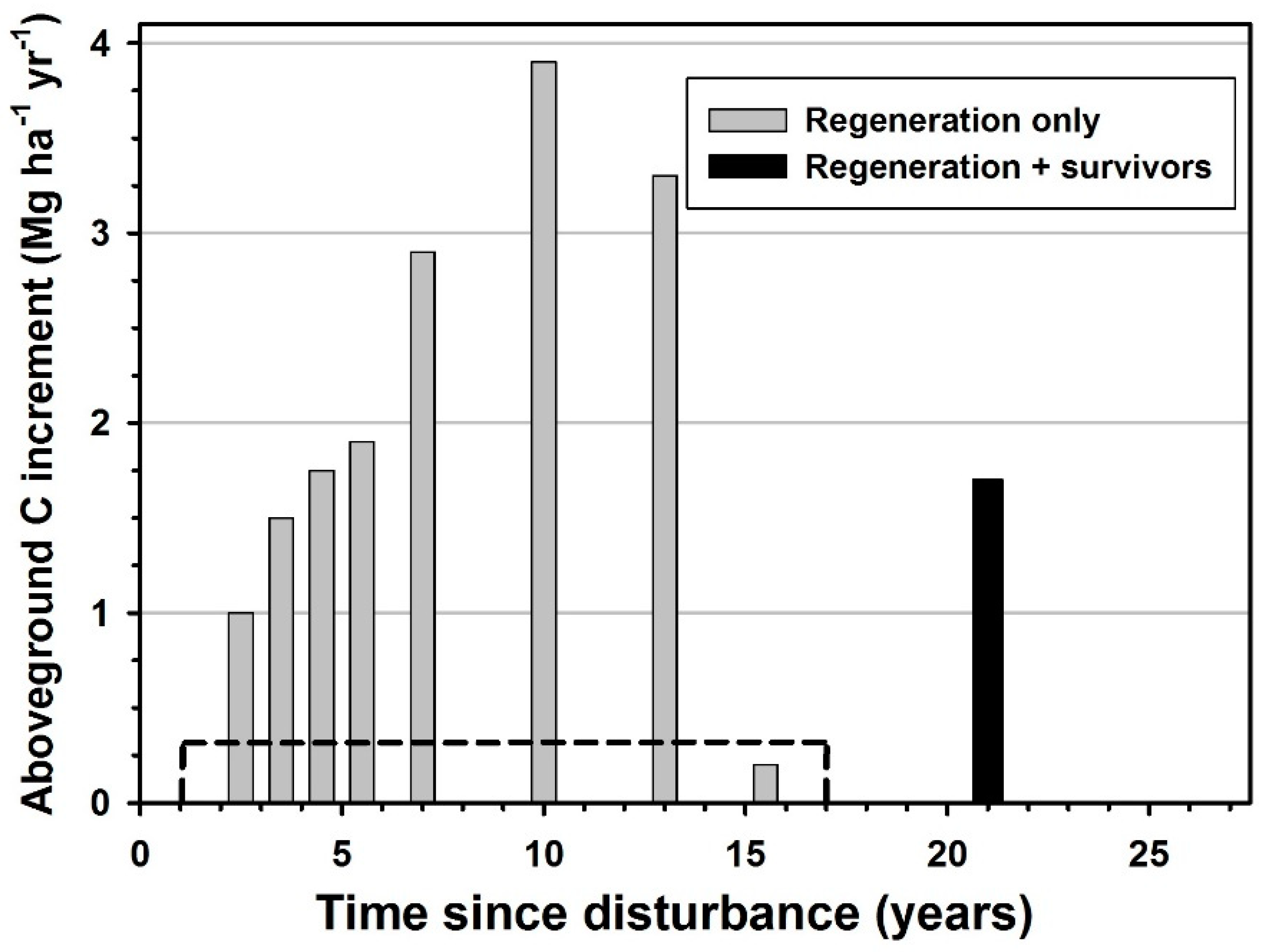
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Domke, G.; Williams, C.A.; Birdsey, R.; Coulston, J.; Finzi, A.; Gough, C.; Haight, B.; Hicke, J.; Janowiak, M.; de Jon, B.; et al. Chapter 9: Forests. In Second State of the Carbon Cycle Report (SOCCR2): A Sustained Assessment Report; U.S. Global Change Research Program: Washington, DC, USA, 2018; pp. 365–398. [Google Scholar]
- Curtis, P.S.; Gough, C.M. Forest age, disturbance and the carbon cycle. New Phytol. 2018, 218, 1–6. [Google Scholar] [CrossRef]
- Kashian, D.M.; Romme, W.H.; Tinker, D.B.; Turner, M.G.; Ryan, M.G. Carbon storage on landscapes with stand-replacing fires. BioScience 2008, 56, 598–606. [Google Scholar] [CrossRef]
- Williams, C.A.; Gu, H.; MacLean, R.; Masek, J.G.; Collatz, G.J. Disturbance and the carbon balance of US forests: A quantitative review of impacts from harvests, fires, insects, and droughts. Glob. Planet. Chang. 2016, 143, 66–80. [Google Scholar] [CrossRef]
- Amiro, B.D.; Barr, A.G.; Barr, J.G.; Black, T.A.; Bracho, R.; Brown, M.; Chen, J.; Clark, K.L.; Davis, K.J.; Desai, A.R.; et al. Ecosystem carbon dioxide fluxes after disturbance in forests of North America. J. Geophys. Res. 2010, 115, G00K02. [Google Scholar] [CrossRef]
- Ghimire, B.; Williams, C.A.; Collatz, G.J.; Vanderhoof, M. Fire-induced carbon emissions and regrowth uptake in western U.S. forests: Documenting variation across forest types, fire severity, and climate regions. J. Geophys. Res. 2012, 117, G03036. [Google Scholar] [CrossRef]
- Kasischke, E.S.; Amiro, B.D.; Barger, N.N.; French, N.H.F.; Goetz, S.J.; Grosse, G.; Harmon, M.E.; Hicke, J.A.; Liu, S.; Masek, J.G. Impacts of disturbance on the terrestrial carbon budget of North America. J. Geophys. Res. Biogeosci. 2013, 118, 303–316. [Google Scholar] [CrossRef]
- Everham, E.E.; Brokaw, N.V.L. Forest damage and recovery from catastrophic wind. Bot. Rev. 1996, 62, 113–185. [Google Scholar] [CrossRef]
- Mitchell, S.J. Wind as a natural disturbace agent in forests—A synthesis. Forestry 2013, 86, 147–157. [Google Scholar] [CrossRef]
- Barr, J.G.; Engle, V.; Fuentes, J.D.; Zieman, J.C.; O’Halloran, T.L.; Smith, T.J.; Anderson, G.H. Controls on mangrove forest-atmosphere carbon dioxide exchanges in western Everglades National Park. J. Geophys. Res. 2010, 115, G02020. [Google Scholar] [CrossRef]
- Barr, J.G.; Engel, V.; Smith, T.J.; Fuentes, J.D. Hurricane disturbance and recovery of energy balance, CO2 fluxes and canopy structure in a mangrove forest of the Florida Everglades. Agric. For. Meteorol. 2012, 153, 54–66. [Google Scholar] [CrossRef]
- Li, J.; Powell, T.L.; Seiler, T.J.; Johnson, D.P.; Anderson, H.P.; Bracho, R.; Hungate, B.A.; Hinkle, C.R.; Drake, B.G. Impacts of Hurricane Frances on Florida scrub-oak ecosystem processes: Defoliation, net CO2 exchange and interactions with elevated CO2. Glob. Chang. Biol. 2007, 13, 1101–1113. [Google Scholar] [CrossRef]
- Chambers, J.Q.; Fisher, J.I.; Zeng, H.C.; Chapman, E.L.; Baker, D.B.; Hurtt, G.C. Hurricane Katrina’s carbon footprint on U.S. Gulf Coast forests. Science 2007, 318, 1107. [Google Scholar] [CrossRef]
- Zeng, H.C.; Chambers, J.Q.; Negrón-Juárez, R.I.; Hurtt, G.C.; Baker, D.B.; Powell, M.D. Impacts of tropical cyclones on U.S. forest tree mortality and carbon flux from 1851 to 2000. Proc. Natl. Acad. Sci. USA 2009, 106, 7888–7892. [Google Scholar] [CrossRef] [PubMed]
- Fisk, J.P. The impacts of tropical cyclones on the net carbon balance of eastern US forests (1851–2000). Environ. Res. Lett. 2013, 8, 045017. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Orwig, D.A.; Foster, D.R.; Plotkin, A.B.; Schoonmaker, P.K.; Wagner, M.R. Long-term structural and biomass dynamics of virgin Tsuga canadensis-Pinus strobus forests after hurricane disturbance. Ecology 2017, 98, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Meigs, G.W.; Keeton, W.S. Intermediate-severity wind disturbance in mature temperate forests: Legacy structure, carbon storage, and stand dynamics. Ecol. Appl. 2018, 28, 798–815. [Google Scholar] [CrossRef] [PubMed]
- Magnabosco Marra, D.; Trumbore, S.E.; Higuchi, N.; Ribeiro, G.H.P.M.; Negrón-Juárez, R.I.; Holzwarth, F.; Rifai, S.W.; dos Santos, J.; Lima, A.J.N.; Kinupp, V.F.; et al. Windthrows control biomass patterns and functional composition of Amazon forests. Glob. Chang. Biol. 2018, 2018, 1–15. [Google Scholar] [CrossRef]
- Parker, G.; Martinez-Yrizar, A.; Alvarez-Yepiz, J.C.; Maass, M.; Araiza, S. Effects of hurricane disturbance on a tropical dry forest canopy in western Mexico. For. Ecol. Manag. 2018, 426, 39–52. [Google Scholar] [CrossRef]
- Scatena, F.N.; Moya, S.; Estrada, C.; Chinea, J.D. The first five years in the reorganization of aboveground biomass and nutrient use following Hurricane Hugo in the Bisley Experimental Watersheds, Luquillo Experimental Forest, Puerto Rico. Biotropica 1996, 28, 424–440. [Google Scholar] [CrossRef]
- Mascaro, J.; Perfecto, I.; Barros, O.; Boucher, D.H.; Granzow de la Cerda, I.; Ruiz, J.; Vandermeer, J. Aboveground biomass accumulation in a tropical wet forest in Nicaragua following a catastrophic hurricane disturbance. Biotropica 2005, 37, 600–608. [Google Scholar] [CrossRef]
- Scalley, T.H.; Scatena, F.N.; Lugo, A.E.; Moya, S.; Estrada Ruiz, C.R. Changes in structure, composition, and nutrients during 15 years of hurricane-induced succession in a subtropical wet forest in Puerto Rico. Biotropica 2010, 42, 455–463. [Google Scholar] [CrossRef]
- Sano, T.; Hirano, T.; Liang, N.; Hirata, R.; Fujinuma, Y. Carbon dioxide exchange of a larch forest after a typhoon disturbance. For. Ecol. Manag. 2010, 260, 2214–2223. [Google Scholar] [CrossRef]
- Yamanoi, K.; Mizoguchi, Y.; Utsugi, H. Effects of a windthrow disturbance on the carbon balance of a broadleaf deciduous forest in Hokkaido, Japan. Biogeosciences 2015, 12, 6837–6851. [Google Scholar] [CrossRef]
- Yazaki, T.; Hirano, T.; Sano, T. Biomass accumulation and net primary production during the early stage of secondary succession after a severe forest disturbance in northern Japan. Forests 2016, 7, 287. [Google Scholar] [CrossRef]
- Kobler, J.; Jandl, R.; Dirnbock, T.; Mirtl, M.; Schindlbacher, A. Effects of stand patchiness due to windthrow and bark beetle abatement measures on soil CO2 efflux and net ecosystem productivity of a managed temperate mountain forest. Eur. J. For. Res. 2015, 134, 683–692. [Google Scholar] [CrossRef]
- Knohl, A.; Kolle, O.; Minayeva, T.Y.; Milyukova, I.J.; Vygodskaya, N.N.; Foken, T.; Schulze, E.D. Carbon dioxide exchange of a Russian boreal forest after disturbance by wind throw. Glob. Chang. Biol. 2002, 8, 231–246. [Google Scholar] [CrossRef]
- Lindroth, A.; Lagergren, F.; Grelle, A.; Klemedtsson, L.; Langvall, O.; Weslien, P.; Tuulik, J. Storms can cause Europe-wide reduction in forest carbon sink. Glob. Chang. Biol. 2009, 15, 346–355. [Google Scholar] [CrossRef]
- Fortin, M.; Albrecht, A.; Kohnle, U.; Ningre, F. The impact of windstorm damage in the assessment of the carbon balance in even-aged Fagus sylvatica L. stands. Forests 2014, 5, 784–801. [Google Scholar] [CrossRef]
- Lindauer, M.; Schmid, H.P.; Grote, R.; Mauder, M.; Steinbrecher, R.; Wolpert, B. Net ecosystem exchange over a non-cleared wind-throw-disturbed upland spruce forest—Measurements and simulations. Agric. For. Meteorol. 2014, 197, 219–234. [Google Scholar] [CrossRef]
- Matthews, B.; Mayer, M.; Katzensteiner, K.; Godbold, D.L.; Schume, H. Turbulent energy and carbon dioxide exchange along an early-successional windthrow chronosequence in the European Alps. Agric. For. Meteorol. 2017, 232, 576–594. [Google Scholar] [CrossRef]
- Liu, S.; Bond-Lamberty, B.; Hicke, J.A.; Vargas, R.; Zhao, S.; Chen, J.; Edburg, S.L.; Hu, Y.; Liu, J.; McGuire, A.D.; et al. Simulating the impacts of disturbances on forest carbon cycling in North America: Processes, data, models, and challenges. J. Geophys. Res. 2011, 116, G00K08. [Google Scholar] [CrossRef]
- Bjorkbom, J.C.; Larson, R.G. The Tionesta Scenic and Research Natural Areas. In USDA Forest Service General Technical Report; NE-31; U.S. Department of Agriculture, Forest Service, Northeastern Forest Experimental Station: Upper Darby, PA, USA, 1977. [Google Scholar]
- Peterson, C.J.; Pickett, S.T.A. Forest reorganization: A case study in an old-growth forest catastrophic blowdown. Ecology 1995, 76, 763–774. [Google Scholar] [CrossRef]
- Peterson, C.J.; Carson, W.P. Generalizing forest regeneration models: The dependence of propagule availability on disturbance history and stand size. Can. J. For. Res. 1996, 26, 45–52. [Google Scholar] [CrossRef]
- Peterson, C.J.; Carson, W.P.; McCarthy, B.C.; Pickett, S.T.A. Microsite variation and soil dynamics within newly created treefall pits and mounds. Oikos 1990, 58, 39–46. [Google Scholar] [CrossRef]
- Peterson, C.J.; Pickett, S.T.A. Microsite and elevational influences on forest regeneration three years after catastrophic windthrow. J. Veg. Sci. 1990, 1, 657–662. [Google Scholar] [CrossRef]
- Long, Z.T.; Carson, W.P.; Peterson, C.J. Can disturbance create refugia from herbivores: An example with hemlock regeneration on treefall mounds. J. Torrey Bot. Soc. 1998, 125, 165–168. [Google Scholar] [CrossRef]
- Peterson, C.J. Damage and recovery of tree species after two different tornadoes in the same old growth forest: A comparison of infrequent wind disturbances. For. Ecol. Manag. 2000, 135, 237–252. [Google Scholar] [CrossRef]
- Krueger, L.M.; Peterson, C.J. Effects of white-tailed deer on Tsuga canadensis regeneration: Evidence of microsites as refugia from browsing. Am. Midl. Nat. 2006, 156, 353–362. [Google Scholar] [CrossRef]
- Hough, A.F.; Forbes, R.D. The ecology and silvics of forests in the high plateaus of Pennsylvania. Ecol. Monogr. 1943, 13, 299–320. [Google Scholar] [CrossRef]
- Hough, A.F. A climax forest community on East Tionesta Creek in northwestern Pennsylvania. Ecology 1936, 17, 9–28. [Google Scholar] [CrossRef]
- Kain, M.; Battaglia, L.; Royo, A.; Carson, W.P. Over-browsing in Pennsylvania creates a depauperate forest dominated by an understory tree: Results from a 60-year-old deer exclosure. J. Torrey Bot. Soc. 2011, 138, 322–326. [Google Scholar] [CrossRef]
- Jenkins, J.C.; Chojnacky, D.C.; Heath, L.S.; Birdsey, R.A. National-scale biomass estimators for United States tree species. For. Sci. 2003, 49, 12–35. [Google Scholar]
- Warde, W.; Petranka, J.W. A correction factor table for missing point-center quarter data. Ecology 1981, 62, 491–494. [Google Scholar] [CrossRef]
- Fatemi, F.R.; Yanai, R.D.; Hamburg, S.P.; Vadeboncoeur, M.A.; Arthur, M.A.; Briggs, R.D.; Levine, C.R. Allometric equations for young northern hardwoods: The importance of age-specific equations for estimating aboveground biomass. Can. J. For. Res. 2011, 41, 881–891. [Google Scholar] [CrossRef]
- Woods, K.D. Multi-decade biomass dynamics in an old-growth hemlock-northern hardwood forest, Michigan, USA. PeerJ 2014, e598. [Google Scholar] [CrossRef]
- Keeton, W.S.; Whitman, A.A.; McGee, G.C.; Goodale, C.L. Late-successional biomass development in northern hardwood-conifer forests of the northeastern United States. For. Sci. 2011, 57, 489–505. [Google Scholar]
- Canham, C.D.; Loucks, O.L. Catastrophic windthrow in the presettlement forests of Wisconsin. Ecology 1984, 65, 803–809. [Google Scholar] [CrossRef]
- Canham, C.D.; Marks, P.L. The response of woody plants to disturbance: Patterns of establishment and growth. In The Ecology of Natural Disturbance and Patch Dynamics; Pickett, S.T.A., White, P.S., Eds.; Academic Press: New York, NY, USA, 1985; pp. 197–216. [Google Scholar]
- Seischab, F.K.; Orwig, D. Catastrophic disturbances in the presettlement forests of western New York. Bull. Torrey Bot. Club 1991, 118, 117–122. [Google Scholar] [CrossRef]
- Frelich, L.E.; Lorimer, C.G. Natural disturbance regimes in hemlock-hardwood forests of the upper Great Lakes region. Ecol. Monogr. 1991, 61, 145–164. [Google Scholar] [CrossRef]
- Dunn, C.P.; Guntenspergen, G.R.; Dorney, J.R. Catastrophic wind disturbance in an old-growth hemlock-hardwood forest, Wisconsin. Can. J. Bot. 1983, 61, 211–217. [Google Scholar] [CrossRef]
- Lang, K.D.; Schulte, L.A.; Guntenspergen, G.R. Windthrow and salvage logging in an old-growth hemlock-northern hardwoods forest. For. Ecol. Manag. 2009, 259, 56–64. [Google Scholar] [CrossRef]
- Bradford, J.B.; Fraver, S.; Milo, A.M.; D’Amato, A.W.; Palik, B.; Shinneman, D.J. Effects of multiple interacting disturbances and salvage logging on forest carbon stocks. For. Ecol. Manag. 2012, 267, 209–214. [Google Scholar] [CrossRef]
- Lewis, R.J.; Banner-Martin, K.H. The impact of Cyclone Fanele on a tropical dry forest in Madagascar. Biotropica 2012, 44, 135–140. [Google Scholar] [CrossRef]
- Lugo, A.E. Visible and invisible effects of hurricanes on forest ecosystems: An international review. Austral Ecol. 2008, 33, 368–398. [Google Scholar] [CrossRef]
- Elliott, K.J.; Boring, L.R.; Swank, W.T. Aboveground biomass and nutrient accumulation 20 years after clear-cutting a southern Appalachian watershed. Can. J. For. Res. 2002, 32, 667–683. [Google Scholar] [CrossRef]
- Barker-Plotkin, A.; Foster, D.; Carlson, J.; Magill, A. Survivors, not invaders, control forest development following simulated hurricane. Ecology 2013, 94, 414–423. [Google Scholar] [CrossRef]
- Shure, D.J.; Phillips, D.L.; Bostick, P.E. Gap size and succession in cutover southern Appalachian forests: An 18 year study of vegetation dynamics. Plant Ecol. 2006, 185, 299–318. [Google Scholar] [CrossRef]
- Reiners, W.A. Twenty years of ecosystem reorganization following experimental deforestation and regrowth suppression. Ecol. Monogr. 1992, 62, 503–523. [Google Scholar] [CrossRef]
- Fu, Z.; Li, D.; Hararuk, O.; Schwalm, C.; Luo, Y.; Yan, L.; Niu, S. Recovery time and state change of terrestrial carbon cycle after disturbance. Environ. Res. Lett. 2017, 12, 104004. [Google Scholar] [CrossRef]

| Species | Form of Equation | a | b | R2 |
|---|---|---|---|---|
| A. Stems 25–300 cm tall | lnAGB = a + (b × ln(Height)) | |||
| Acer pensylvanicum | −7.702 | 2.569 | n.a. | |
| Acer rubrum. | −6.509 | 2.135 | 0.768 | |
| Betula spp. | −9.105 | 2.646 | 0.821 | |
| Fagus grandifolia | −7.702 | 2.569 | 0.768 | |
| Prunus spp. | −8.569 | 2.498 | 0.839 | |
| Tsuga canadensis | −6.509 | 2.135 | n.a. | |
| B. Stems > 300 cm tall and <10 cm dbh | log10AGB = a + (b × log10(dbh) | |||
| Acer pensylvanicum | 2.342 | 2.155 | n.a. | |
| Acer rubrum | 2.130 | 2.237 | 0.988 | |
| Betula spp. | 2.260 | 2.513 | 0.972 | |
| Fagus grandifolia | 2.342 | 2.155 | 0.959 | |
| Prunus spp. | 1.833 | 2.597 | 0.872 | |
| Tsuga canadensis | 2.130 | 2.237 | n.a. | |
| C. Stems ≥ 10 cm dbh | AGB = exp(a + b × ln(dbh)) | |||
| Acer rubrum | −1.912 | 2.365 | 0.958 | |
| Acer saccharum | −2.013 | 2.434 | 0.988 | |
| Betula spp. | −1.912 | 2.365 | 0.958 | |
| Fagus grandifolia | −2.013 | 2.434 | 0.988 | |
| Prunus pensylvanicum | −2.480 | 2.483 | 0.980 | |
| Prunus serotina | −2.480 | 2.483 | 0.980 | |
| Tsuga canadensis | 2.538 | 2.481 | 0.992 | |
| Location | Forest Type | Disturbance Type | Years Post-Disturbance | AGBI * | Citation |
|---|---|---|---|---|---|
| Hokkaido, Japan | Temperate broadleaf | Windthrow | 2 | 1.84 | 1 |
| 8 | 3.96 | ||||
| Hokkaido, Japan | Temperate coniferous | Windthrow | 2–5 | 0.43 | 2 |
| 5–7 | 0.98 | ||||
| 7–10 | −0.02 | ||||
| Austria | Temperate coniferous | Windthrow + bark beetles | 1–4 | 4.62 | 3 |
| Massachusetts, USA | Temperate broadleaf | Simulated windthrow | 4–24 | 4.7 | 4 |
| New Hampshire, USA | Temperate mixed conifer-broadleaf | Windthrow | 51–71 | 1.75 ** | 5 |
| Pennsylvania, USA | Temperate broadleaf | Windthrow | 2–3 | 2 | This study |
| 3–4 | 3 | ||||
| 4–5 | 3.5 | ||||
| 5–6 | 3.8 | ||||
| 6–8 | 5.8 | ||||
| 8–12 | 7.8 | ||||
| 12–14 | 6.6 | ||||
| 14–17 | 0.4 | ||||
| 17–25 | 3.4 | ||||
| Puerto Rico, USA | Subtropical broadleaf | Windthrow | 0–5 | 16.2 | 6 |
| 5–10 | 6.6 | ||||
| 10–15 | 1.2 | ||||
| Central Amazon, Brazil | Tropical broadleaf | Windthrow | 4–8 | 2.1–7.6 | 7 |
| Nicaragua | Tropical broadleaf | Windthrow | 1–10 | 5.67 | 8 |
| 10–12 | 4.6 | ||||
| Mexico | Tropical dry | Windthrow | 0–2 | 1.83 | 9 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peterson, C.J. Twenty-Five Years of Aboveground Biomass and Carbon Accumulation Following Extreme Wind Damage in an Old-Growth Forest. Forests 2019, 10, 289. https://doi.org/10.3390/f10030289
Peterson CJ. Twenty-Five Years of Aboveground Biomass and Carbon Accumulation Following Extreme Wind Damage in an Old-Growth Forest. Forests. 2019; 10(3):289. https://doi.org/10.3390/f10030289
Chicago/Turabian StylePeterson, Chris J. 2019. "Twenty-Five Years of Aboveground Biomass and Carbon Accumulation Following Extreme Wind Damage in an Old-Growth Forest" Forests 10, no. 3: 289. https://doi.org/10.3390/f10030289
APA StylePeterson, C. J. (2019). Twenty-Five Years of Aboveground Biomass and Carbon Accumulation Following Extreme Wind Damage in an Old-Growth Forest. Forests, 10(3), 289. https://doi.org/10.3390/f10030289





