The Effects of Copper and Silver Nanoparticles on Container-Grown Scots Pine (Pinus sylvestris L.) and Pedunculate Oak (Quercus robur L.) Seedlings
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Nanoparticles and Treatments
2.3. Chlorophyll a Fluorescence Measurements
2.4. Samplings and Biometric Parameters
2.5. Microscopic Investigations
2.6. Data Analysis
3. Results
3.1. Biometric Parameters of Seedlings
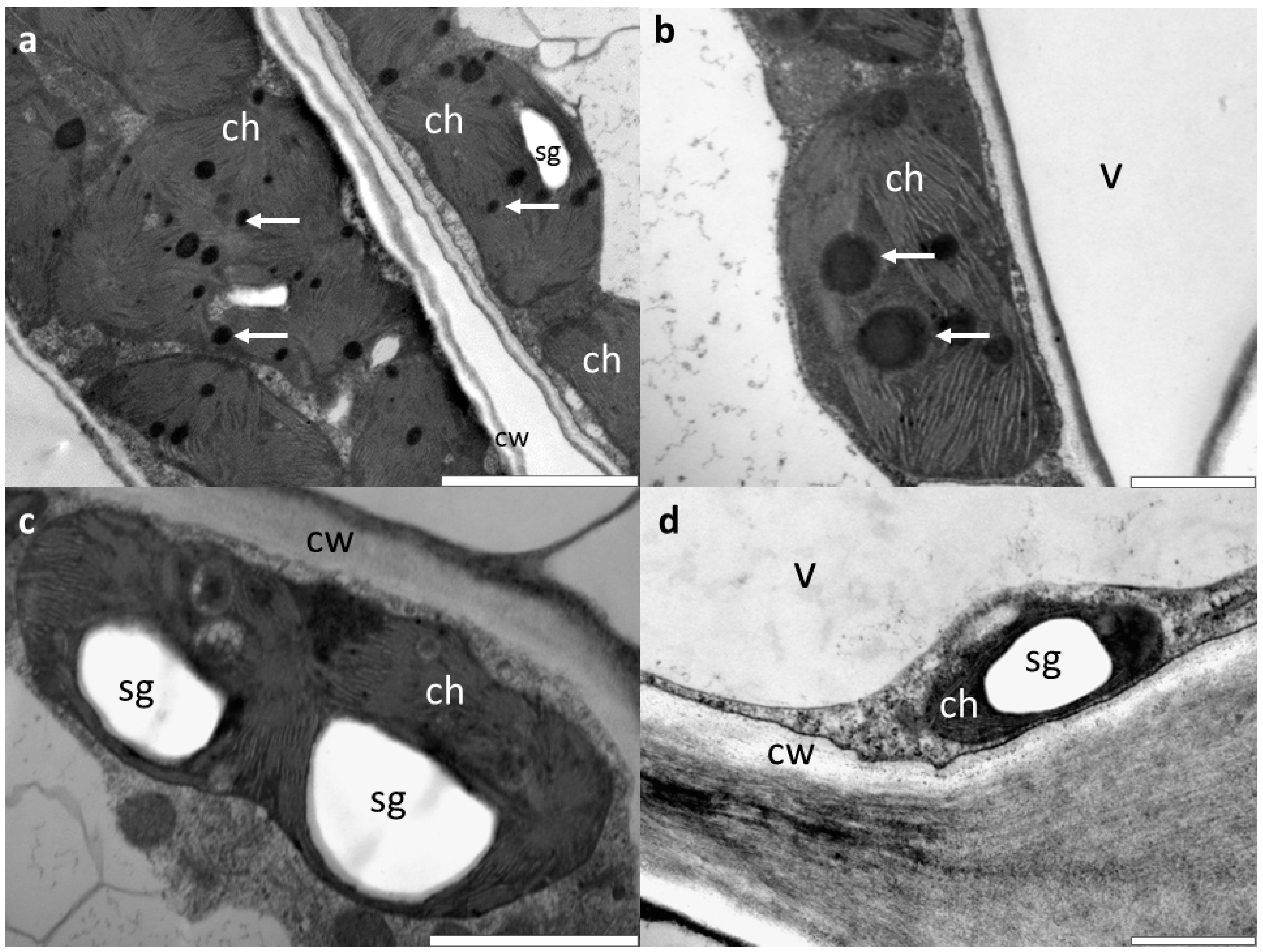
3.2. Microscopic Investigations
3.3. Chlorophyll a Fluorescence
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bernhardt, E.S.; Colman, B.P.; Hochella, M.F.; Cardinale, B.J.; Nisbet, M.; Richardson, C.J.; Yin, L. An ecological perspective on nanomaterial impacts in the environment. J. Environ. Qual. 2010, 39, 1–12. [Google Scholar] [CrossRef]
- Ruffini Castiglione, M.; Cremonini, R. Nanoparticles and higher plants. Caryologia 2009, 62, 161–165. [Google Scholar]
- Klaine, S.; Alvarez, P.J.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. 2008, 27, 1825–1851. [Google Scholar] [CrossRef]
- Masarovičá, E.; Kráľová, K. Metal nanoparticles and plants. Ecol. Chem. Eng. S 2013, 20, 9–22. [Google Scholar] [CrossRef]
- Duhan, J.S.; Kumar, R.; Kumar, N.; Nehra, K.; Duhan, S. Nanotechnology: The new perspective in precision agriculture. Biotechnol. Rep. 2017, 15, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Lombi, E.; Zhao, F.-J.; Kopittke, P.M. Nanotechnology: A new opportunity in plant sciences. Trends Plant Sci. 2016, 21, 699–712. [Google Scholar] [CrossRef]
- Peharec Štefanić, P.; Cvjetko, P.; Biba, R.; Domijan, A.-M.; Letofsky-Papst, I.; Tkalec, M.; Šikić, S.; Cindrić, M.; Balen, B. Physiological, ultrastructural and proteomic responses of tobacco seedlings exposed to silver nanoparticles and silver nitrate. Chemosphere 2018, 209, 640–655. [Google Scholar] [CrossRef]
- Ruttkay-Nedecky, B.; Krystofova, O.; Nejdl, L.; Adam, V. Nanoparticles based on essential metals and their phytotoxicity. J. Nanobiotechnol. 2017, 15, 33. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J.; Herth, S. Plant nanotoxicology. Trends Plant Sci. 2011, 16, 582–589. [Google Scholar] [CrossRef]
- Schwab, F.; Zhai, G.; Kern, M.; Turner, A.; Schnoor, J.L.; Wiesner, M.R. Barriers, pathways and processes for uptake, translocation and accumulation of nanomaterials in plants—Critical review. Nanotoxicology 2016, 10, 257–278. [Google Scholar] [CrossRef]
- Zhai, G.; Walters, K.S.; Peate, D.W.; Alvarez, P.J.J.; Schnoor, J.L. Transport of gold nanoparticles through plasmodesmata and precipitation of gold ions on woody poplar. Environ. Sci. Technol. Lett. 2014, 1, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Song, Y.; Wu, K.; Tao, Q.; Liang, Y.; Li, T. Effects of Cr2O3 nanoparticles on the chlorophyll fluorescence and chloroplast ultrastructure of soybean (Glycine max). Environ. Sci. Pollut. Res. 2018, 25, 19446–19457. [Google Scholar] [CrossRef] [PubMed]
- Servin, A.; Elmer, W.; Mukherjee, A.; De la Torre-Roche, R.; Hamidi, H.; White, J.C.; Bindraban, P.; Dimkpa, C. A review of the use of engineered nanomaterials to suppress plant disease and enhance crop yield. J. Nanopart. Res. 2015, 17, 92. [Google Scholar] [CrossRef]
- Rizwan, M.; Ali, S.; Qayyum, M.F.; Ok, Y.S.; Adrees, M.; Ibrahim, M.; Zia-ur-Rehman, M.; Farid, M.; Abbas, F. Effect of metal and metal oxide nanoparticles on growth and physiology of globally important food crops: A critical review. J. Hazard. Mat. 2017, 322, 2–16. [Google Scholar] [CrossRef]
- Linglan, M.; Chao, L.; Chunxiang, Q.; Sitao, Y.; Jie, L.; Fengqing, G.; Fashui, H. Rubisco activase mRNA expression in spinach: Modulation by nanoanatase treatment. Biol. Trace Elem. Res. 2008, 122, 168–178. [Google Scholar] [CrossRef]
- Prasad, T.N.V.K.V.; Sudhakar, P.; Sreenivasulu, Y.; Latha, P.; Munaswamy, V.; Raja Reddy, K.; Sreeprasad, T.S.; Sajanlal, P.R.; Pradeep, T. Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J. Plant Nutr. 2012, 35, 905–927. [Google Scholar] [CrossRef]
- Jaberzadeh, A.; Moaveni, P.; Tohidi Moghadam, H.R.; Zahedi, H. Influence of bulk and nanoparticles titanium foliar application on some agronomic traits, seed gluten and starch contents of wheat subjected to water deficit stress. Not. Bot. Horti Agrobo. 2013, 41, 201–207. [Google Scholar] [CrossRef]
- Khodakovskaya, M.V.; Kim, B.S.; Kim, J.N.; Alimohammadi, M.; Dervishi, E.; Mustafa, T.; Cernigla, C.E. Carbon nanotubes as plant growth regulators: Effects on tomato growth, reproductive system, and soil microbial community. Small 2013, 9, 115–123. [Google Scholar] [CrossRef]
- Shaw, A.K.; Hossain, Z. Impact of nano-CuO stress on rice (Oryza sativa L.) seedlings. Chemosphere 2013, 93, 906–915. [Google Scholar] [CrossRef]
- Dimkpa, C.O.; McLean, J.E.; Latta, D.E.; Manangón, E.; Britt, W.D.; Johnson, W.P.; Boyanov, M.I.; Anderson, A.L. CuO and ZnO nanoparticles: Phytotoxicity, metal speciation, and induction of oxidative stress in sand-grown wheat. J. Nanopart. Res. 2012, 14, 1125. [Google Scholar] [CrossRef]
- Nair, P.M.G.; Chung, I.M. A mechanistic study on the toxic effect of copper oxide nanoparticles in soybean (Glycine max L.) root development and lignification of root cells. Biol. Trace Elem. Res. 2014, 162, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Vannini, C.; Domingo, G.; Onelli, E.; De Mattia, F.; Bruni, I.; Marsoni, M.; Bracale, M. Phytotoxic and genotoxic effects of silver nanoparticles exposure on germinating wheat seedlings. J. Plant Physiol. 2014, 171, 1142–1148. [Google Scholar] [CrossRef] [PubMed]
- Raliya, R.; Tarafdar, J.C. ZnO nanoparticle biosynthesis and its effect on phosphorous-mobilizing enzyme secretion and gum contents in clusterbean (Cyamopsis tetragonoloba L.). Agric. Res. 2013, 2, 48–57. [Google Scholar] [CrossRef]
- Tarafdar, J.C.; Raliya, R.; Mahawar, H.; Rathore, I. Development of zinc nanofertilizer to enhance crop production in pearl millet (Pennisetum americanum). Agric. Res. 2014, 3, 257–262. [Google Scholar] [CrossRef]
- Qian, H.; Peng, X.; Han, X.; Ren, J.; Sun, L.; Fu, Z. Comparison of the toxicity of silver nanoparticles and silver ions on the growth of terrestrial plant model Arabidopsis thaliana. J. Environ. Sci. 2013, 25, 1947–1955. [Google Scholar] [CrossRef]
- Rastogi, A.; Zivcak, M.; Sytar, O.; Kalaji, H.M.; He, X.; Mbarki, S.; Brestic, M. Impact of metal and metal oxide nanoparticles on plant: A critical Review. Front. Chem. 2017, 5, 78. [Google Scholar] [CrossRef]
- Yuan, J.; Chen, Y.; Li, H.; Lu, J.; Zhao, H.; Liu, M.; Nechitaylo, G.S.; Glushchenko, N.N. New insights in to the cellular responses to iron nanoparticles in Capsicum annuum. Sci. Rep. 2018, 8, 3228. [Google Scholar] [CrossRef]
- Nhan, L.V.; Ma, C.; Rui, Y.; Liu, S.; Li, X.; Xing, B.; Liu, L. Phytotoxic mechanism of nanoparticles: Destruction of chloroplast and vascular bundles and alteration of nutrient absorption. Sci. Rep. 2015, 5, 11618. [Google Scholar] [CrossRef] [PubMed]
- Cvjetko, P.; Zovko, M.; Peharec Štefanić, P.; Biba, R.; Tkalec, M.; Domijan, A.-M.; Vinković Vrček, I.; Letofsky-Papst, I.; Šikić, S.; Balen, B. Phytotoxic effects of silver nanoparticles in tobacco plants. Environ. Sci. Pollut. Res. 2018, 25, 5590–5602. [Google Scholar] [CrossRef] [PubMed]
- Aleksandrowicz-Trzcińska, M.; Szaniawski, A.; Studnicki, M.; Bederska-Błaszczyk, M.; Urban, A. The effect of silver and copper nanoparticles on the growth and mycorrhizal colonisation of Scots pine (Pinus sylvestris L.) in a container nursery experiment. iForest 2018, 11, 690–697. [Google Scholar] [CrossRef]
- Yang, J.; Cao, W.; Rui, Y. Interactions between nanoparticles and plants: Phytotoxicity and defense mechanisms. J. Plant Interact. 2017, 12, 158–169. [Google Scholar] [CrossRef]
- Nel, A.; Xia, T.; Mädler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, K.; Johnson, N.G. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Bhatt, D.; Zaidi, M.G.; Saradhi, P.P.; Khanna, P.K.; Arora, S. Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica juncea. Appl. Biochem. Biotechnol. 2012, 167, 2225–2233. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Singh, S.; Singh, S.; Srivastava, P.K.; Singh, V.P.; Singh, S.; Prasad, S.M.; Singh, P.K.; Dubey, N.K.; Pandey, A.C.; et al. Nitric oxide alleviates silver nanoparticles (AgNps)-induced phytotoxicity in Pisum sativum seedlings. Plant Physiol. Biochem. 2017, 110, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Vishwakarma, K.; Shweta, U.N.; Singh, J.; Liu, S.; Singh, V.P.; Prasad, S.M.; Chauhan, D.K.; Tripathi, D.K.; Sharma, S. Differential phytotoxic impact of plant mediated silver nanoparticles (AgNPs) and silver nitrate (AgNO3) on Brassica sp. Front. Plant Sci. 2017, 8, 1501. [Google Scholar] [CrossRef]
- Karnovsky, M.J. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol. 1965, 27, 137. [Google Scholar]
- R Core Team. R. A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2012. Available online: http://www.R-project.org/ (accessed on 7 February 2019).
- Seeger, E.M.; Baun, A.; Kästner, M.; Trapp, S. Insignificant acute toxicity of TiO2 nanoparticles to willow trees. J. Soils Sediments 2009, 9, 46–53. [Google Scholar] [CrossRef]
- Zarafshar, M.; Akbarinia, M.; Askari, H.; Hossein, S.M.; Rahaie, M.; Struve, D. Toxicity Assessment of SiO2 nanoparticles to pear seedlings. Int. J. Nanosci. Nanotechnol. 2015, 11, 13–22. [Google Scholar]
- Olchowik, J.; Bzdyk, R.; Studnicki, M.; Bednarska-Błaszczyk, M.; Urban, A.; Aleksandrowicz-Trzcińska, M. The effects of silver and copper nanoparticles on the condition of English oak (Quercus robur L.) seedlings in a container nursery experiment. Forests 2017, 8, 310. [Google Scholar] [CrossRef]
- Sweet, M.J.; Singleton, I. Soil contamination with silver nanoparticles reduces Bishop pine growth and ectomycorrhizal diversity on pine roots. J. Nanopart. Res. 2015, 17, 448. [Google Scholar] [CrossRef] [PubMed]
- Adani, F.; Papa, G.; Schievano, A.; Cardinale, G.; D’Imporzano, G.; Tambone, F. Nanoscale structure of the cell wall protecting cellulose from enzyme attack. Environ. Sci. Technol. 2010, 45, 1107–1113. [Google Scholar] [CrossRef]
- Rajput, V.D.; Minkina, T.; Sushkova, S.; Tsitsishvili, V.; Mandzhieva, S.; Gorovtsov, A.; Nevidomskyaya, D.; Gromakova, N. Effect of nanoparticles on crops and soil microbial communities. J. Soils Sediments 2018, 18, 2179–2187. [Google Scholar] [CrossRef]
- Izumi, H.; Anderson, I.C.; Killham, K. Diversity of predominant endophytic bacteria in European deciduous and coniferous trees. Can. J. Microbiol. 2008, 54, 173–179. [Google Scholar] [CrossRef]
- Tashi-Oshnoei, F.; Harighi, B.; Abdollahzadeh, J. Isolation and identification of endophytic bacteria with plant growth promoting and biocontrol potential from oak trees. For. Pathol. 2017, 47, e12360. [Google Scholar] [CrossRef]
- Ryan, R.P.; Germaine, K.; Franks, A.; Ryan, D.J.; Dowling, D.N. Bacterial endophytes: Recent developments and applications. FEMS Microbiol. Lett. 2008, 278, 1–9. [Google Scholar] [CrossRef]
- Birbaum, K.; Brogioli, R.; Schellenberg, M.; Martinoia, E.; Stark, W.J.; Gunter, D.; Limbach, L.K. No evidence for cerium dioxide NP translocation in maize plants. Environ. Sci. Technol. 2010, 44, 8718–8723. [Google Scholar] [CrossRef] [PubMed]
- Selga, T.; Selga, M. Response of Pinus silvestris L. needles to electromagnetic fields. Cytological and ultrastructural aspects. Sci. Total Environ. 1996, 180, 65–73. [Google Scholar] [CrossRef]
- Piller, L.E.; Abraham, M.; Dormann, P.; Kessler, F.; Besagni, C. Plastid lipid droplets at the crossroads of prenylquinone metabolism. J. Exp. Bot. 2012, 63, 1609–1618. [Google Scholar] [CrossRef]
- Kuc, T.; Aleksandrowicz-Trzcińska, M. Sterowana mikoryzacja i doglebowa aplikacja fungicydów w hodowli dębu szypułkowego. I. Kolonizacja mikoryzowa i wzrost sadzonek z zakrytym systemem korzeniowym w szkółce. Sylwan 2012, 156, 765–775. [Google Scholar] [CrossRef]
- Aleksandrowicz-Trzcińska, M.; Hamera-Dzierżanowska, A.; Żybura, H.; Drozdowski, S. Wpływ mikoryzacji i chitozanu na wzrost sosny zwyczajnej (Pinus sylvestris L.) w szkółce i na uprawie. Sylwan 2013, 157, 899–908. [Google Scholar] [CrossRef]

| Treatment (n = 40) | Length of Shoot [cm] | Root Collar Diameter [mm] | Total Root Length [cm] | Dry Mass of Aboveground part [g] | Dry Mass of Root [g] |
|---|---|---|---|---|---|
| Scots pine | |||||
| Ag0 | 9.4 ± 2.70 b | 2.43 ± 0.451 a | 283.4 ± 71.3 a | 0.788 ± 0.369 a | 0.421 ± 0.101 b |
| Ag5 | 7.8 ± 1.75 a | 2.29 ± 0.363 a | 293.4 ± 76.8 a | 0.719 ± 0.332 a | 0.348 ± 0.165 ab |
| Ag25 | 9.7 ± 2.41 b | 2.27 ± 0.463 a | 295.0 ± 80.5 a | 0.883 ± 0.355 a | 0.338 ± 0.147 ab |
| Ag50 | 7.6 ± 2.57 a | 2.36 ± 0.542 a | 287.5 ± 74.4 a | 0.876 ± 0.326 a | 0.325 ± 0.116 a |
| Cu0 | 9.5 ± 2.17 bc | 2.63 ± 0.375 b | 269.6 ± 80.9 a | 0.986 ± 0.371 ab | 0.431 ± 0.159 b |
| Cu5 | 8.7 ± 1.86 ab | 2.25 ± 0.469 a | 290.4 ± 87.9 a | 1.314 ± 0.443 c | 0.294 ± 0.113 a |
| Cu25 | 10.7 ± 2.14 c | 2.22 ± 0.547 a | 269.3 ± 89.3 a | 0.944 ± 0.381 b | 0.332 ± 0.162 ab |
| Cu50 | 8.0 ± 1.59 a | 2.03 ± 0.453 a | 296.0 ± 84.9 a | 0.730 ± 0.323 a | 0.404 ± 0.125 b |
| Pedunculate oak | |||||
| Ag0 | 28.7 ± 4.33 a | 5.28 ± 0.622 a | 408.8 ± 122.3 b | 3.145 ± 1.082 a | 1.633 ± 1.194 a |
| Ag5 | 27.1 ± 5.64 a | 4.62 ± 0.989 a | 400.1 ± 115.2 b | 3.010 ± 1.444 a | 2.415 ± 1.249 a |
| Ag25 | 27.4 ± 6.42 a | 4.98 ± 0.906 a | 366.2 ± 127.6 a | 2.761 ± 1.133 a | 2.365 ± 1.199 a |
| Ag50 | 25.9 ± 7.92 a | 5.06 ± 1.012 a | 410.9 ± 102.8 b | 2.988 ± 1.530 a | 1.753 ± 1.210 a |
| Cu0 | 28.0 ± 4.68 ab | 4.77 ± 0.786 a | 401.3 ± 122.5 ab | 2.690 ± 0.968 a | 1.607 ± 1.133 a |
| Cu5 | 25.3 ± 7.37 ab | 5.06 ± 0.962 a | 371.0 ± 113.5 a | 2.660 ± 1.245 a | 1.763 ± 1.272 ab |
| Cu25 | 28.5 ± 5.61 b | 5.11 ± 1.110 a | 379.2 ± 123.6 a | 3.063 ± 1.481 a | 2.543 ± 1.155 b |
| Cu50 | 25.0 ± 6.02 a | 5.05 ± 0.911 a | 432.8 ± 124.0 b | 2.543 ± 1.079 a | 2.072 ± 1.167 b |
| Treatment | Scots Pine | Pedunculate Oak |
|---|---|---|
| Ag0 | 0.8026 ± 0.0276 a | 0.7905 ± 0.0467 b |
| Ag5 | 0.8086 ± 0.0339 a | 0.7803 ± 0.0533 ab |
| Ag25 | 0.8049 ± 0.0360 a | 0.7798 ± 0.0630 ab |
| Ag50 | 0.8074 ± 0.0282 a | 0.7629 ± 0.0901 a |
| Cu0 | 0.8011 ± 0.0391 a | 0.7905 ± 0.0425 b |
| Cu5 | 0.8119 ± 0.0263 b | 0.7780 ± 0.0679 ab |
| Cu25 | 0.8013 ± 0.0352 a | 0.7628 ± 0.0634 a |
| Cu50 | 0.8075 ± 0.0355 ab | 0.7651 ± 0.0611 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aleksandrowicz-Trzcińska, M.; Bederska-Błaszczyk, M.; Szaniawski, A.; Olchowik, J.; Studnicki, M. The Effects of Copper and Silver Nanoparticles on Container-Grown Scots Pine (Pinus sylvestris L.) and Pedunculate Oak (Quercus robur L.) Seedlings. Forests 2019, 10, 269. https://doi.org/10.3390/f10030269
Aleksandrowicz-Trzcińska M, Bederska-Błaszczyk M, Szaniawski A, Olchowik J, Studnicki M. The Effects of Copper and Silver Nanoparticles on Container-Grown Scots Pine (Pinus sylvestris L.) and Pedunculate Oak (Quercus robur L.) Seedlings. Forests. 2019; 10(3):269. https://doi.org/10.3390/f10030269
Chicago/Turabian StyleAleksandrowicz-Trzcińska, Marta, Magdalena Bederska-Błaszczyk, Adam Szaniawski, Jacek Olchowik, and Marcin Studnicki. 2019. "The Effects of Copper and Silver Nanoparticles on Container-Grown Scots Pine (Pinus sylvestris L.) and Pedunculate Oak (Quercus robur L.) Seedlings" Forests 10, no. 3: 269. https://doi.org/10.3390/f10030269
APA StyleAleksandrowicz-Trzcińska, M., Bederska-Błaszczyk, M., Szaniawski, A., Olchowik, J., & Studnicki, M. (2019). The Effects of Copper and Silver Nanoparticles on Container-Grown Scots Pine (Pinus sylvestris L.) and Pedunculate Oak (Quercus robur L.) Seedlings. Forests, 10(3), 269. https://doi.org/10.3390/f10030269





