Linkages between Phosphorus and Plant Diversity in Central European Forest Ecosystems—Complementarity or Competition?
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Data Sources
2.2. Organic Layer, Mineral Soil, and Foliar Sampling
2.3. Alpha-Diversity Indices
- Shannon diversity index: the degree of diversity in a finite forest stand
- Number of different species
- Proportional cover (%) of the ith species
- Cover (%) of individuals in the ith species
- Total cover (%) of individuals
- Complement Simpson diversity index, which captures the variance of the species cover distribution
- Number of different species
- Proportional cover (%) of the ith species
- Cover (%) of individuals in the ith species
- Total cover (%) of individuals
- Degree of evenness in species cover
- Euler’s number: approximately equal to 2.71828
- Shannon diversity index: the degree of diversity in a finite forest stand
- The number of different species
2.4. Phosphorus Use Efficiency Indices
- Phosphorus content (g P/kg) of European beech leaves and Norway spruce needles, respectively
- Constant dry mass (105 °C) of 100 European beech leaves or 1000 Norway spruce needles
2.5. Statistical Analysis
3. Results
3.1. P-Related Parameters in European Beech and Norway Spruce Forests
3.2. Plant Diversity of Beech and Spruce Forests
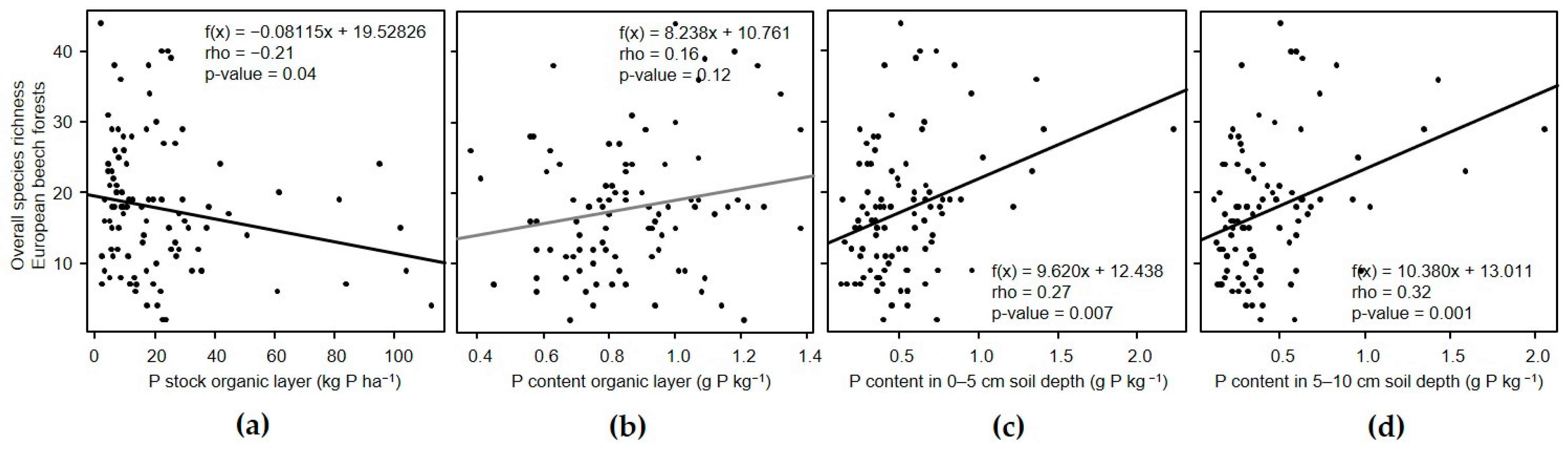
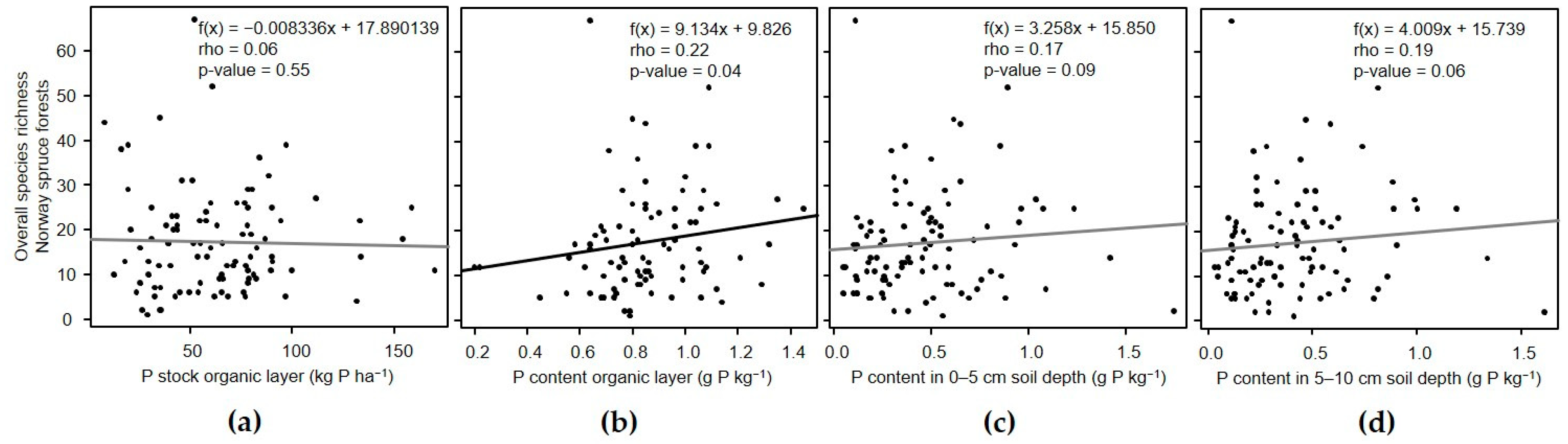
3.3. Relation of α-Diversity with Phosphorus in the Organic Layer and Soil
3.3.1. European Beech Forests
3.3.2. Norway Spruce Forests
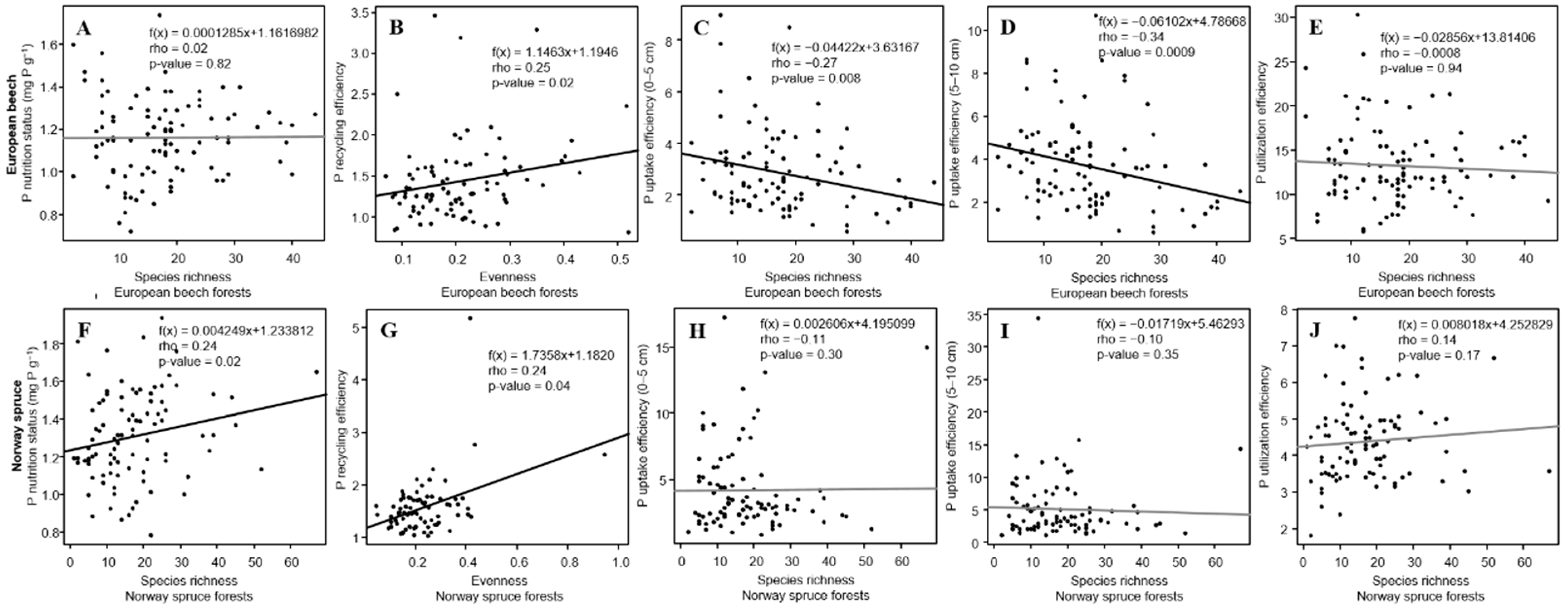
3.4. P Nutrition Status and PUE in Relation to α-Diversity
4. Discussion
4.1. Phosphorus in Foliage, Organic Layer, and Soil
4.2. Plant Diversity by Vegetation Layer
4.3. Linkages between Organic Layer and Soil P, Biodiversity, and PUE
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus uptake by plants: From soil to cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, F.; Blume, H.-P.; Thiele, S. Lehrbuch der Bodenkunde, 16th ed.; Spektrum Akad. Verl: Heidelberg, Germany, 2010; ISBN 978-3-8274-1444-1. [Google Scholar]
- Lambers, H. Root structure and functioning for efficient acquisition of phosphorus: Matching morphological and physiological traits. Ann. Bot. 2006, 98, 693–713. [Google Scholar] [CrossRef] [PubMed]
- Feller, I.C.; McKee, K.L.; Whigham, D.F.; O’Neill, J.P. Nitrogen vs. phosphorus limitation across an ecotonal gradient in a mangrove forest. Biogeochemistry 2003, 62, 145–175. [Google Scholar] [CrossRef]
- Cross, A.F.; Schlesinger, W.H. A literature review and evaluation of the Hedley fractionation: Applications to the biogeochemical cycle of soil phosphorus in natural ecosystems. Geoderma 1995, 64, 197–214. [Google Scholar] [CrossRef]
- Gleason, S.M.; Read, J.; Ares, A.; Metcalfe, D.J. Phosphorus economics of tropical rainforest species and stands across soil contrasts in Queensland, Australia: Understanding the effects of soil specialization and trait plasticity. Funct. Ecol. 2009, 23, 1157–1166. [Google Scholar] [CrossRef]
- Herbert, D.A.; Fownes, J.H. Phosphorus limitation of forest leaf area and net primary production on a highly weathered soil. Biogeochemistry 1995, 29, 223–235. [Google Scholar] [CrossRef]
- Lang, F.; Bauhus, J.; Frossard, E.; George, E.; Kaiser, K.; Kaupenjohann, M.; Krüger, J.; Matzner, E.; Polle, A.; Prietzel, J.; et al. Phosphorus in forest ecosystems: New insights from an ecosystem nutrition perspective. Z. Pflanzenernähr. Bodenk. 2016, 179, 129–135. [Google Scholar] [CrossRef]
- Gartlan, J.S.; Newbery, D.M.; Thomas, D.W.; Waterman, P.G. The influence of topography and soil phosphorus on the vegetation of Korup Forest Reserve, Cameroun. Vegetatio 1986, 65, 131–148. [Google Scholar] [CrossRef]
- Malizia, A.; Grau, H.R.; Lichstein, J.W. Soil phosphorus and disturbance influence liana communities in a subtropical montane forest. J. Veg. Sci. 2010, 21, 551–560. [Google Scholar] [CrossRef]
- Tilman, D. Plant Strategies and the Dynamics and Structure of Plant Communities; Princeton University Press: Princeton, NJ, USA, 1988; ISBN 9780691084893. [Google Scholar]
- Hooper, D.U. The role of complementarity and competition in ecosystem responses to variation in plant diversity. Ecology 1998, 79, 704–719. [Google Scholar] [CrossRef]
- Fridley, J.D. The influence of species diversity on ecosystem productivity: How, where, and why? Oikos 2001, 93, 514–526. [Google Scholar] [CrossRef]
- Tilman, D. The influence of functional diversity and composition on ecosystem processes. Science 1997, 277, 1300–1302. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S.; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Grime, J.P. Competitive exclusion in herbaceous vegetation. Nature 1973, 242, 344–347. [Google Scholar] [CrossRef]
- Newman, E.I. Competition and diversity in herbaceous vegetation. Nature 1973, 244, 310. [Google Scholar] [CrossRef]
- Oksanen, J. Is the humped relationship between species richness and biomass an artefact due to plot size? J. Ecol. 1996, 84, 293–295. [Google Scholar] [CrossRef]
- Rajaniemi, T.K. Why does fertilization reduce plant species diversity? Testing three competition-based hypotheses. J. Ecol. 2002, 90, 316–324. [Google Scholar] [CrossRef]
- Craine, J.M.; Dybzinski, R.; Robinson, D. Mechanisms of plant competition for nutrients, water and light. Funct. Ecol. 2013, 27, 833–840. [Google Scholar] [CrossRef]
- Nakamura, N. Species richness and aggregation effects on the productivity of ruderal plant communities under drought perturbation. Biosci. Horiz. 2008, 1, 128–135. [Google Scholar] [CrossRef]
- Smith, R.G.; Mortensen, D.A.; Ryan, M.R. A new hypothesis for the functional role of diversity in mediating resource pools and weed−crop competition in agroecosystems. Weed Res. 2010, 50, 37–48. [Google Scholar] [CrossRef]
- Davidson, E.A.; Reis de Carvalho, C.J.; Vieira, I.C.G.; Figueiredo, R.d.O.; Moutinho, P.; Yoko Ishida, F.; Primo dos Santos, M.T.; Benito Guerrero, J.; Kalif, K.; Tuma Sabá, R. Nitrogen and phosphorus limitation of biomass growth in a tropical secondary forest. Ecol. Appl. 2004, 14, 150–163. [Google Scholar] [CrossRef]
- Homeier, J.; Breckle, S.-W.; Günter, S.; Rollenbeck, R.T.; Leuschner, C. Tree diversity, forest structure and productivity along altitudinal and topographical gradients in a species-rich Ecuadorian montane rain forest. Biotropica 2010, 42, 140–148. [Google Scholar] [CrossRef]
- Campo, J.; Maass, M.; Jaramillo, V.J.; Martínez-Yrízar, A.; Sarukhán, J. Phosphorus cycling in a Mexican tropical dry forest ecosystem. Biogeochemistry 2001, 53, 161–179. [Google Scholar] [CrossRef]
- Keith, H.; Raison, R.J.; Jacobson, K.L. Allocation of carbon in a mature eucalypt forest and some effects of soil phosphorus availability. Plant Soil 1997, 196, 81–99. [Google Scholar] [CrossRef]
- Jäger, H.; Alencastro, M.J.; Kaupenjohann, M.; Kowarik, I. Ecosystem changes in Galápagos highlands by the invasive tree Cinchona pubescens. Plant Soil 2013, 371, 629–640. [Google Scholar] [CrossRef]
- Romanyà i Socoró, J. Phosphorus Cycling in Fast Growing Forest Plantations: Availability, Plant Uptake and the Role of Forest Floor. Ph.D. Thesis, Universitat de Barcelona, Barcelona, Spain, 1993. [Google Scholar]
- Sardans, J.; Peñuelas, J. Increasing drought decreases phosphorus availability in an evergreen Mediterranean forest. Plant Soil 2004, 267, 367–377. [Google Scholar] [CrossRef]
- Oelmann, Y.; Richter, A.K.; Roscher, C.; Rosenkranz, S.; Temperton, V.M.; Weisser, W.W.; Wilcke, W. Does plant diversity influence phosphorus cycling in experimental grasslands? Geoderma 2011, 167–168, 178–187. [Google Scholar] [CrossRef]
- Ceulemans, T.; Stevens, C.J.; Duchateau, L.; Jacquemyn, H.; Gowing, D.J.G.; Merckx, R.; Wallace, H.; van Rooijen, N.; Goethem, T.; Bobbink, R.; et al. Soil phosphorus constrains biodiversity across European grasslands. Glob. Chang. Biol. 2014, 20, 3814–3822. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Ball, M.C.; Choat, B.; Engelbrecht, B.M.; Holbrook, N.M.; Feller, I.C. Linking physiological processes with mangrove forest structure: Phosphorus deficiency limits canopy development, hydraulic conductivity and photosynthetic carbon gain in dwarf Rhizophora mangle. Plant Cell Environ. 2006, 29, 793–802. [Google Scholar] [CrossRef]
- Bustamante, M.M.C.; de Brito, D.Q.; Kozovits, A.R.; Luedemann, G.; de Mello, T.R.B.; de Siqueira Pinto, A.; Munhoz, C.B.R.; Takahashi, F.S.C. Effects of nutrient additions on plant biomass and diversity of the herbaceous-subshrub layer of a Brazilian savanna (Cerrado). Plant Ecol. 2012, 213, 795–808. [Google Scholar] [CrossRef]
- Lannes, L.S.; Bustamante, M.M.C.; Edwards, P.J.; Venterink, H.O. Alien and endangered plants in the Brazilian Cerrado exhibit contrasting relationships with vegetation biomass and N:P stoichiometry. New Phytol. 2012, 196, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Prober, S.M.; Wiehl, G. Relationships among soil fertility, native plant diversity and exotic plant abundance inform restoration of forb-rich eucalypt woodlands. Divers. Distrib. 2012, 18, 795–807. [Google Scholar] [CrossRef]
- Sander, J.; Wardell-Johnson, G. Impacts of soil fertility on species and phylogenetic turnover in the high-rainfall zone of the Southwest Australian global biodiversity hotspot. Plant Soil 2011, 345, 103–124. [Google Scholar] [CrossRef]
- Tyler, G. Soil chemical limitations to growth and development of Veronica officinalis L. and Carex pilulifera L. Plant Soil 1996, 184, 281–289. [Google Scholar] [CrossRef]
- Wellbrock, N.; Eickenscheidt, N.; Grüneberg, E.; Bögelein, R. Environmental settings and their changes in the last decades. In Status and Dynamics of Forests in Germany: Results of the National Forest Monitoring Ecological Studies (Analysis and Synthesis), 1st ed.; Wellbrock, N., Bolte, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; ISBN 978-3-030-15734-0. [Google Scholar]
- Ilg, K.; Wellbrock, N.; Lux, W. Phosphorus supply and cycling at long-term forest monitoring sites in Germany. Eur. J. For. Res. 2009, 128, 483–492. [Google Scholar] [CrossRef]
- Talkner, U.; Meiwes, K.J.; Potočić, N.; Seletković, I.; Cools, N.; de Vos, B.; Rautio, P. Phosphorus nutrition of beech (Fagus sylvatica L.) is decreasing in Europe. Ann. For. Sci. 2015, 72, 919–928. [Google Scholar] [CrossRef]
- Wellbrock, N.; Bolte, A.; Flessa, H. (Eds.) Dynamik und räumliche Muster Forstlicher Standorte in Deutschland. Ergebnisse der Bodenzustandserhebung im Wald 2006 bis 2008; Johann Heinrich von Thünen-Institut: Braunschweig, Germany, 2016; ISBN 978-3-86576-161-3. [Google Scholar]
- Ma, M. Species richness vs evenness: Independent relationship and different responses to edaphic factors. Oikos 2005, 111, 192–198. [Google Scholar] [CrossRef]
- Marini, L.; Scotton, M.; Klimek, S.; Isselstein, J.; Pecile, A. Effects of local factors on plant species richness and composition of Alpine meadows. Agric. Ecosyst. Environ. 2007, 119, 281–288. [Google Scholar] [CrossRef]
- Merunková, K.; Chytrý, M. Environmental control of species richness and composition in upland grasslands of the southern Czech Republic. Plant Ecol. 2012, 213, 591–602. [Google Scholar] [CrossRef]
- Wassen, M.J.; Venterink, H.O.; Lapshina, E.D.; Tanneberger, F. Endangered plants persist under phosphorus limitation. Nature 2005, 437, 547–550. [Google Scholar] [CrossRef]
- Karanika, E.D.; Alifragis, D.A.; Mamolos, A.P.; Veresoglou, D.S. Differentiation between responses of primary productivity and phosphorus exploitation to species richness. Plant Soil 2007, 297, 69–81. [Google Scholar] [CrossRef]
- Ball, B.A.; Bradford, M.A.; Hunter, M.D. Nitrogen and phosphorus release from mixed litter layers is lower than predicted from single species decay. Ecosystems 2009, 12, 87–100. [Google Scholar] [CrossRef]
- Talkner, U.; Jansen, M.; Beese, F.O. Soil phosphorus status and turnover in central-European beech forest ecosystems with differing tree species diversity. Eur. J. Soil Sci. 2009, 60, 338–346. [Google Scholar] [CrossRef]
- Jacob, M.; Viedenz, K.; Polle, A.; Thomas, F.M. Leaf litter decomposition in temperate deciduous forest stands with a decreasing fraction of beech (Fagus sylvatica). Oecologia 2010, 164, 1083–1094. [Google Scholar] [CrossRef] [PubMed]
- Helgason, T.; Merryweather, J.W.; Denison, J.; Wilson, P.; Young, J.P.W.; Fitter, A.H. Selectivity and functional diversity in arbuscular mycorrhizas of co-occurring fungi and plants from a temperate deciduous woodland. J. Ecol. 2002, 90, 371–384. [Google Scholar] [CrossRef]
- Baxter, J.W.; Dighton, J. Phosphorus source alters host plant response to ectomycorrhizal diversity. Mycorrhiza 2005, 15, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Ewald, J.; Ziche, D. Giving meaning to Ellenberg nutrient values: National Forest Soil Inventory yields frequency-based scaling. Appl. Veg. Sci. 2017, 20, 115–123. [Google Scholar] [CrossRef]
- Fu, B.J.; Liu, S.L.; Ma, K.M.; Zhu, Y.G. Relationships between soil characteristics, topography and plant diversity in a heterogeneous deciduous broad-leaved forest near Beijing, China. Plant Soil 2004, 261, 47–54. [Google Scholar] [CrossRef]
- Ziche, D.; Seidling, W. Homogenisation of climate time series from ICP Forests Level II monitoring sites in Germany based on interpolated climate data. Ann. For. Sci. 2010, 67, 804. [Google Scholar] [CrossRef]
- Wellbrock, N.; Ahrends, B.; Bögelein, R.; Bolte, A.; Eickenscheidt, N.; Grüneberg, E.; König, N.; Schmitz, A.; Fleck, S.; Ziche, D. Concept and Methodology of the National Forest Soil Inventory. In Status and Dynamics of Forests in Germany: Results of the National Forest Monitoring Ecological Studies (Analysis and Synthesis), 1st ed.; Wellbrock, N., Bolte, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; ISBN 978-3-030-15734-0. [Google Scholar]
- Ad-hoc-AG Boden. Bodenkundliche Kartieranleitung. 5.; Verbesserte und erweiterte Auflage: Hannover, Germany, 2005. [Google Scholar]
- Grüneberg, E.; Ziche, D.; Wellbrock, N. Organic carbon stocks and sequestration rates of forest soils in Germany. Glob. Chang. Biol. 2014, 20, 2644–2662. [Google Scholar] [CrossRef]
- Handbuch Forstliche Analytik. Eine Loseblatt-Sammlung der Analysemethoden im Forstbereich, Gutachterausschuss Forstliche Analytik; Bundesministerium für Ernährung und Landwirtschaft, Ed.; HFA: Berlin, Germany, 2006. [Google Scholar]
- Wellbrock, N.G.; Aydin, C.-T.; Block, J.; Bussian, B.; Deckert, M.; Diekmann, O.; Evers, J.G.; Fetzer, K.D.; Gauer, J.; Gehrmann, J.; et al. Bodenzustandserhebung im Wald (BZE II). Arbeitsanleitung für die Außenaufnahmen; BEML: Bonn, Germany, 2006. [Google Scholar]
- Rautio, P.; Fürst, A.; Stefan, K.; Raitio, H.; Bartels, U. Part XII: Sampling and analysis of needles and leaves. In Manual on Methods and Criteria for Harmonized Sampling, Assessment, Monitoring and Analysis of the Effects of Air Pollution on Forests; UNECE ICP Forests Programme Co-ordinating Centre, Ed.; Thünen Institute: Eberswalde, Germany, 2016; p. 19. [Google Scholar]
- Wellbrock, N.; Bolte, A. Status and Dynamics of Forests in Germany. Available online: https://link.springer.com/book/10.1007/978-3-030-15734-0 (accessed on 18 November 2019).
- Magurran, A.E. Measuring Biological Diversity; Blackwell Pub: Middlesex, MA, USA, 2004; ISBN 978-0-632-05633-0. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Grundon, N.J. Mineral nutrition of some Queensland heath plants. J. Ecol. 1972, 60, 171. [Google Scholar] [CrossRef]
- Shaver, G.R.; Melillo, J.M. Nutrient budgets of marsh plants: Efficiency concepts and relation to availability. Ecology 1984, 65, 1491–1510. [Google Scholar] [CrossRef]
- Vitousek, P. Nutrient cycling and nutrient use efficiency. Am. Nat. 1982, 119, 553–572. [Google Scholar] [CrossRef]
- Jiang, D.; Geng, Q.; Li, Q.; Luo, Y.; Vogel, J.; Shi, Z.; Ruan, H.; Xu, X. Nitrogen and phosphorus resorption in planted forests worldwide. Forests 2019, 10, 201. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A protocol for data exploration to avoid common statistical problems. Method. Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- Schmidt, M.; Veldkamp, E.; Corre, M.D. Tree species diversity effects on productivity, soil nutrient availability and nutrient response efficiency in a temperate deciduous forest. For. Ecol. Manag. 2015, 338, 114–123. [Google Scholar] [CrossRef]
- Wolff, B.; Riek, W. Deutscher Waldbodenbericht 1996. Ergebnisse der Bundesweiten Bodenzustandserhebung im Wald von 1987–1993 (BZE). Band 1, Bonn. 1997. Available online: http://bfh-web.fh-eberswalde.de/bze/upload/BZE_I_Bericht/band1.pdf (accessed on 24 November 2015).
- Ewald, J. Ist Phosphormangel für die geringe Vitalität von Buchen (Fagus sylvatica L.) in den Bayerischen Alpen verantwortlich? Forstw. Cbl. 2000, 119, 276–296. [Google Scholar] [CrossRef]
- Gallardo, A.; Covelo, F. Spatial pattern and scale of leaf N and P concentration in a Quercus robur population. Plant Soil 2005, 273, 269–277. [Google Scholar] [CrossRef]
- Mölder, A.; Bernhardt-Römermann, M.; Schmidt, W. Herb-layer diversity in deciduous forests: Raised by tree richness or beaten by beech? For. Ecol. Manag. 2008, 256, 272–281. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, S.; Ma, K.; Fu, B.; Anand, M. Woody species diversity in forest plantations in a mountainous region of Beijing, China: Effects of sampling scale and species selection. PLoS ONE 2014, 9, e115038. [Google Scholar] [CrossRef]
- Harpole, W.S.; Sullivan, L.L.; Lind, E.M.; Firn, J.; Adler, P.B.; Borer, E.T.; Chase, J.; Fay, P.A.; Hautier, Y.; Hillebrand, H.; et al. Addition of multiple limiting resources reduces grassland diversity. Nature 2016, 537, 93–96. [Google Scholar] [CrossRef]
- Canadell, J.; Jackson, R.B.; Ehleringer, J.R.; Mooney, H.A.; Sala, O.E.; Schulze, E.-D. Maximum rooting depth of vegetation types at the global scale. Oecologia 1996, 108, 583–595. [Google Scholar] [CrossRef] [PubMed]
- Jansa, J.; Finlay, R.; Wallander, H.; Smith, F.A.; Smith, S.E. Role of mycorrhizal symbiosis in phosphorus cycling. In Phosphorus in Action; Varma, A., Bünemann, E., Oberson, A., Frossard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 137–168. ISBN 978-3-642-15270-2. [Google Scholar]
- Akhmetzhanova, A.A.; Soudzilovskaia, N.A.; Onipchenko, V.G.; Cornwell, W.K.; Agafonov, V.A.; Selivanov, I.A.; Cornelissen, J.H.C. A rediscovered treasure: Mycorrhizal intensity database for 3000 vascular plant species across the former Soviet Union. Ecology 2012, 93, 689–690. [Google Scholar] [CrossRef]
- Harley, J.L.; Smith, S.E. Mycorrhizal Symbiosis; Academic Press: London, UK, 1983. [Google Scholar]
- Bodeker, I.T.M.; Clemmensen, K.E.; Boer, W.d.; Martin, F.; Olson, A.; Lindahl, B.D. Ectomycorrhizal Cortinarius species participate in enzymatic oxidation of humus in northern forest ecosystems. New Phytol. 2014, 203, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Soudzilovskaia, N.A.; van der Heijden, M.G.A.; Cornelissen, J.H.C.; Makarov, M.I.; Onipchenko, V.G.; Maslov, M.N.; Akhmetzhanova, A.A.; van Bodegom, P.M. Quantitative assessment of the differential impacts of arbuscular and ectomycorrhiza on soil carbon cycling. New Phytol. 2015, 208, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Rosling, A.; Midgley, M.G.; Cheeke, T.; Urbina, H.; Fransson, P.; Phillips, R.P. Phosphorus cycling in deciduous forest soil differs between stands dominated by ecto- and arbuscular mycorrhizal trees. New Phytol. 2016, 209, 1184–1195. [Google Scholar] [CrossRef] [PubMed]
- Herben, T. Bryophytes in grassland vegetation sample plots: What is their correlation with vascular plants? Folia Geobot. Phytotax. 1987, 22, 35–41. [Google Scholar] [CrossRef]
- Vellak, K.; Paal, J.; Liira, J. Diversity and distribution pattern of bryophytes and vascular plants in a boreal spruce forest. Silva Fenn. 2003, 37. [Google Scholar] [CrossRef]
- Albers, D. Decomposition of beech leaves (Fagus sylvatica) and spruce needles (Picea abies) in pure and mixed stands of beech and spruce. Soil. Biol. Biochem. 2004, 36, 155–164. [Google Scholar] [CrossRef]
- Berger, T.W.; Berger, P. Greater accumulation of litter in spruce (Picea abies) compared to beech (Fagus sylvatica) stands is not a consequence of the inherent recalcitrance of needles. Plant Soil 2012, 358, 349–369. [Google Scholar] [CrossRef]
- Berger, T.W.; Berger, P. Does mixing of beech (Fagus sylvatica) and spruce (Picea abies) litter hasten decomposition? Plant Soil 2014, 377, 217–234. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.H. Mineral nutrition. In Bryophyte Ecology; Smith, A.J.E., Ed.; Chapman & Hall: London, UK, 1982; pp. 383–444. [Google Scholar]
- Frahm, J.-P. Moose als Bioindikatoren; Quelle und Meyer: Wiesbaden, Germany, 1998; ISBN 3-494-01271-7. [Google Scholar]
- Beike, A.K.; Decker, E.L.; Frank, W.; Lang, D.; Vervliet-Scheebaum, M.; Zimmer, A.D.; Reski, R. Applied Bryology—Bryotechnology. Trop. Bryol. 2010, 31, 22–32. [Google Scholar]
- Chapin, F.S.; Oechel, W.C.; van Cleve, K.; Lawrence, W. The role of mosses in the phosphorus cycling of an Alaskan black spruce forest. Oecologia 1987, 74, 310–315. [Google Scholar] [CrossRef] [PubMed]
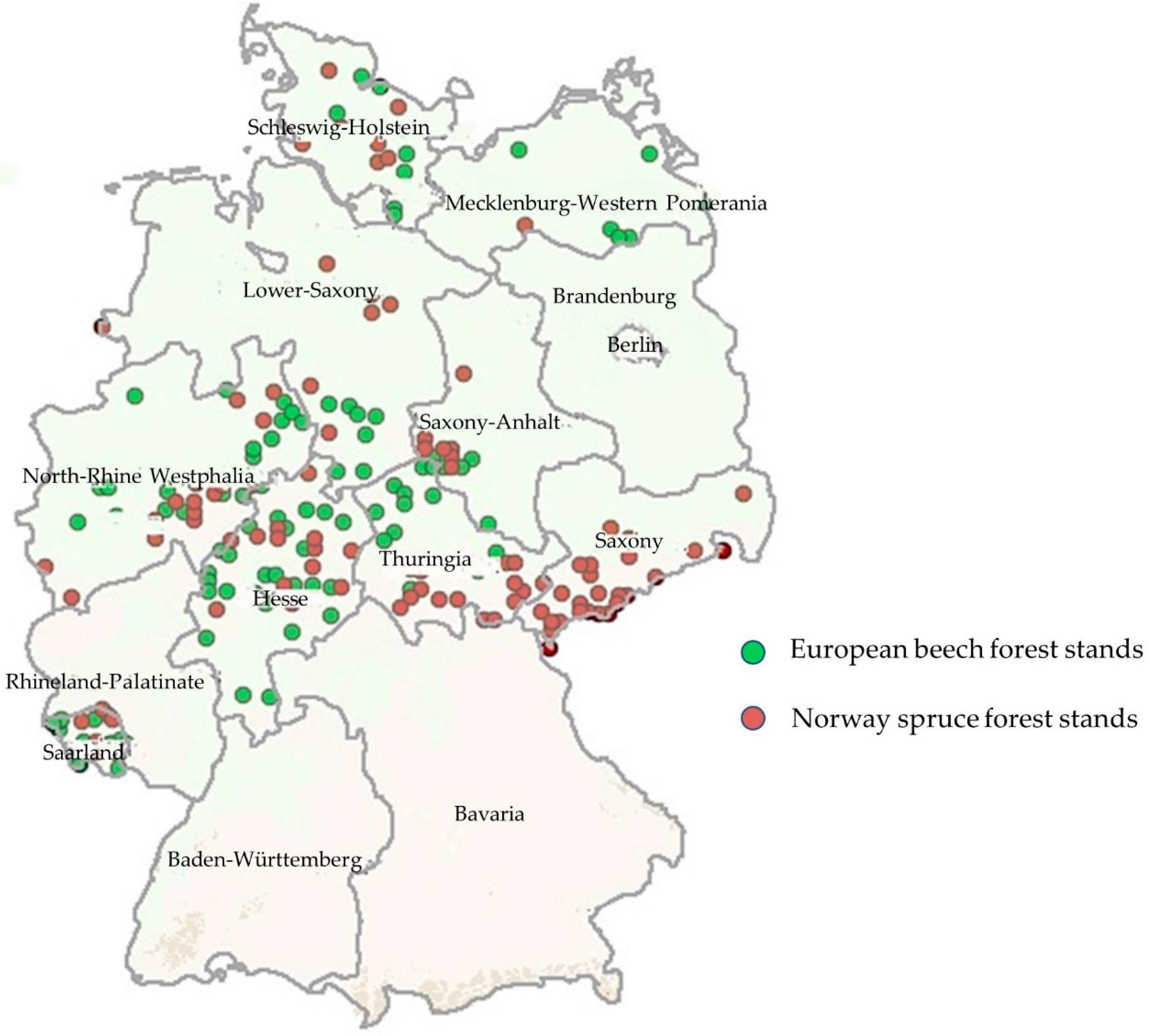
| Parameter | European Beech (n = 101) | Norway Spruce (n = 99) |
|---|---|---|
| Tree age (years) | 87 (14–200) | 61 (21–129) |
| Altitude (m above sea level) | 300 (9–660) | 390 (18–935) |
| Temperature (°C) | 8.3 (6.3–10.5) | 7.7 (4.8–9.7) |
| Precipitation (mm) | 824 (337–1783) | 727 (359–1782) |
| pH (CaCl2, mineral soil 0–90 cm) | 4.5 (3.5–7.6) | 3.9 (3.2–7.4) |
| Soil depth (cm) | 61 (0–230) | 97 (0–210) |
| Soil types | ||
| Cambisol | 56 | 61 |
| Gleysol | 1 | 2 |
| Leptosol | 7 | 0 |
| Luvisol | 21 | 8 |
| Podzol | 0 | 11 |
| Stagnosol | 16 | 16 |
| Parameters | European Beech | Norway Spruce | ||||
|---|---|---|---|---|---|---|
| Mean (SE) | Range | NAs | Mean (SE) | Range | NAs | |
| P nutrition status (g P kg−1) | 1.16 (0.02) | 0.72–1.74 | 3 | 1.31 (0.02) | 0.78–1.94 | 7 |
| P recycling efficiency (g P kg−1) | 1.4 (0.05) | 0.8–3.5 | 12 | 1.6 (0.07) | 0.0–5.2 | 21 |
| P uptake efficiency 0–5 cm (g P kg−1) | 2.9 (0.16) | (0.6–9.0) | 5 | 4.2 (0.33) | (0.0–17.3) | 9 |
| P uptake efficiency 5−10 cm (g P kg−1) | 3.7 (0.21) | 0.6–10.7 | 5 | 5.1 (0.46) | 0.0–34.5 | 9 |
| P utilization efficiency | 13.3 (0.44) | 5.8–30.3 | 3 | 4.3 (0.11) | 1.8–7.8 | 7 |
| P stock organic layer (kg P ha−1) | 22.0 (2.2) | 2.0–27.0 | 0 | 71.9 (9.7) | 7.8–969.0 | 0 |
| P content organic layer (g P kg−1) | 0.86 (0.02) | 0.38–1.38 | 9 | 0.86 (0.02) | 0.20–1.45 | 13 |
| P stock mineral soil 0–5 cm (kg P ha−1) | 194.0 (12.3) | 29.7–919.5 | 2 | 159.2 (11.1) | 10.9–762.4 | 2 |
| P stock mineral soil 5–10 cm (kg P ha−1) | 195.4 (12.3) | 65.3–964.0 | 2 | 160.5 (12.3) | 6.34–905.1 | 2 |
| P content mineral soil 0–5 cm (g P kg−1) | 0.53 (0.03) | 0.14–2.23 | 2 | 0.46 (0.03) | 0.05–1.75 | 0 |
| P content mineral soil 5–10 cm (g P kg−1) | 0.44 (0.03) | 0.12–2.1 | 2 | 0.41 (0.03) | 0.03–1.61 | 0 |
| C/P soil stock ratio 0–10 cm (kg P ha−1) | 113.7 (5.1) | 18.5–327.2 | 2 | 166.7 (12.4) | 19.1–602.6 | 0 |
| Vegetation Layer | Forest Type | Diversity Indices | ||||
|---|---|---|---|---|---|---|
| Total | European beech | 101 | 18 (2–44) | 1.05 (0–2.5) | 0.44 (0–0.9) | 0.21 (0–0.5) |
| Norway spruce | 99 | 17 (1–67) | 1.07 (0–4.1) | 0.44 (0–1.0) | 0.24 (0–1) | |
| Tree | European beech | 101 | 2 (1–8) | 0.31 (0‒1.5) | 0.18 (0‒0.7) | 0.75 (0.1‒1) |
| Norway spruce | 99 | 2 (1‒5) | 0.22 (0‒1.0) | 0.12 (0‒0.6) | 0.78 (0.1‒1) | |
| Shrub | European beech | 79 | 2 (1–11) | 0.28 (0–1.6) | 0.15 (0–0.8) | 0.85 (0.3–1) |
| Norway spruce | 62 | 3 (1–9) | 0.56 (0–1.8) | 0.30 (0–0.8) | 0.69 (0.1–1) | |
| Herb | European beech | 101 | 16 (1–43) | 1.83 (0–3.4) | 0.76 (0–1.0) | 0.57 (0.1–1) |
| Norway spruce | 97 | 17 (1–66) | 1.70 (0–4.1) | 0.67 (0–1.0) | 0.53 (0.1–1) | |
| Moss | European beech | 33 | 3 (1–5) | 0.77 (0–1.6) | 0.45 (0–0.8) | 0.99 (0.8–1) |
| Norway spruce | 66 | 5 (1–12) | 0.98 (0–2.0) | 0.50 (0–0.9) | 0.70 (0.2–1) | |
| P Stock Organic Layer | P Content Org. Layer | P Content in Soil (0–5 cm) | P Content in Soil (5–10 cm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VL | SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E |
| OV | −0.21 * | ns | ns | ns | ns | ns | ns | ns | 0.27 ** | 0.21 * | ns | ns | 0.32 *** | 0.27 ** | 0.22 * | ns |
| TL | ns | −0.23 * | −0.23 * | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| SL | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| HL | −0.23 * | ns | ns | ns | ns | ns | ns | ns | 0.25 * | ns | ns | −0.24 * | 0.31 ** | ns | ns | −0.35 *** |
| ML | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| P Stock Organic Layer | P Content Organic Layer | P Content in Soil (0–5 cm) | P Content in Soil (5–10 cm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VL | SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E |
| OV | ns | ns | ns | ns | 0.22 * | ns | ns | −0.35 ** | ns | ns | ns | ns | ns | ns | ns | ns |
| TL | ns | ns | ns | ns | ns | ns | −0.21 * | ns | −0.28 ** | −0.31 ** | −0.31 ** | −0.21 * | −0.31 ** | −0.36 *** | −0.36 *** | 0.23 * |
| SL | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| HL | ns | −0.33 *** | −0.38 *** | −0.39 *** | 0.23 * | ns | ns | ns | 0.23 * | 0.23 * | 0.21 * | ns | 0.23 * | 0.2 * | ns | ns |
| ML | 0.39 ** | 0.25 * | ns | −0.31 * | 0.38 ** | 0.34 ** | 0.34 ** | ns | ns | ns | ns | 0.35 ** | ns | ns | ns | ns |
| Phosphorus Use Efficiencies | Forest Ecosystem | Tree Layer | Shrub Layer | Herb Layer | Moss Layer | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E | SR | SH | SI | E | ||
| P nutrition status | European beech | ns | ns | ns | ns | ns | 0.21 | 0.22 | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Norway spruce | −0.18 | −0.21 * | −0.21 * | ns | ns | ns | ns | ns | 0.24 * | ns | ns | ns | ns | ns | ns | ns | |
| P recycling efficiency | European beech | ns | 0.18 | 0.17 | ns | ns | ns | ns | ns | ns | −0.25 * | −0.23 * | ns | ns | ns | ns | ns |
| Norway spruce | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | −0.29 * | ns | ns | ns | |
| P uptake eff. (0–5 cm) | European beech | ns | ns | ns | ns | ns | ns | ns | ns | −0.23 * | ns | ns | 0.20 * | ns | ns | ns | ns |
| Norway spruce | 0.22 * | 0.28 ** | 0.29 ** | ns | ns | ns | ns | ns | ns | −0.18 | −0.18 | ns | ns | ns | ns | −0.37 ** | |
| P uptake eff. (5–10 cm) | European beech | ns | ns | ns | ns | ns | ns | ns | ns | −0.31 ** | ns | ns | 0.32 ** | ns | −0.32 | −0.31 | ns |
| Norway spruce | 0.25 * | 0.32 ** | 0.32 ** | −0.17 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | −0.29 * | |
| P utilization efficiency | European beech | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Norway spruce | ns | ns | ns | ns | ns | ns | ns | −0.36 ** | ns | ns | ns | −0.21 * | 0.32 * | ns | 0.26 * | −0.24 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rieger, I.; Kowarik, I.; Ziche, D.; Wellbrock, N.; Cierjacks, A. Linkages between Phosphorus and Plant Diversity in Central European Forest Ecosystems—Complementarity or Competition? Forests 2019, 10, 1156. https://doi.org/10.3390/f10121156
Rieger I, Kowarik I, Ziche D, Wellbrock N, Cierjacks A. Linkages between Phosphorus and Plant Diversity in Central European Forest Ecosystems—Complementarity or Competition? Forests. 2019; 10(12):1156. https://doi.org/10.3390/f10121156
Chicago/Turabian StyleRieger, Isaak, Ingo Kowarik, Daniel Ziche, Nicole Wellbrock, and Arne Cierjacks. 2019. "Linkages between Phosphorus and Plant Diversity in Central European Forest Ecosystems—Complementarity or Competition?" Forests 10, no. 12: 1156. https://doi.org/10.3390/f10121156
APA StyleRieger, I., Kowarik, I., Ziche, D., Wellbrock, N., & Cierjacks, A. (2019). Linkages between Phosphorus and Plant Diversity in Central European Forest Ecosystems—Complementarity or Competition? Forests, 10(12), 1156. https://doi.org/10.3390/f10121156





