Excess Nitrogen in Temperate Forest Ecosystems Decreases Herbaceous Layer Diversity and Shifts Control from Soil to Canopy Structure
Abstract
1. Introduction
2. Methods
2.1. Study Site
2.2. Field Methods
2.3. Data Analysis
3. Results and Discussion
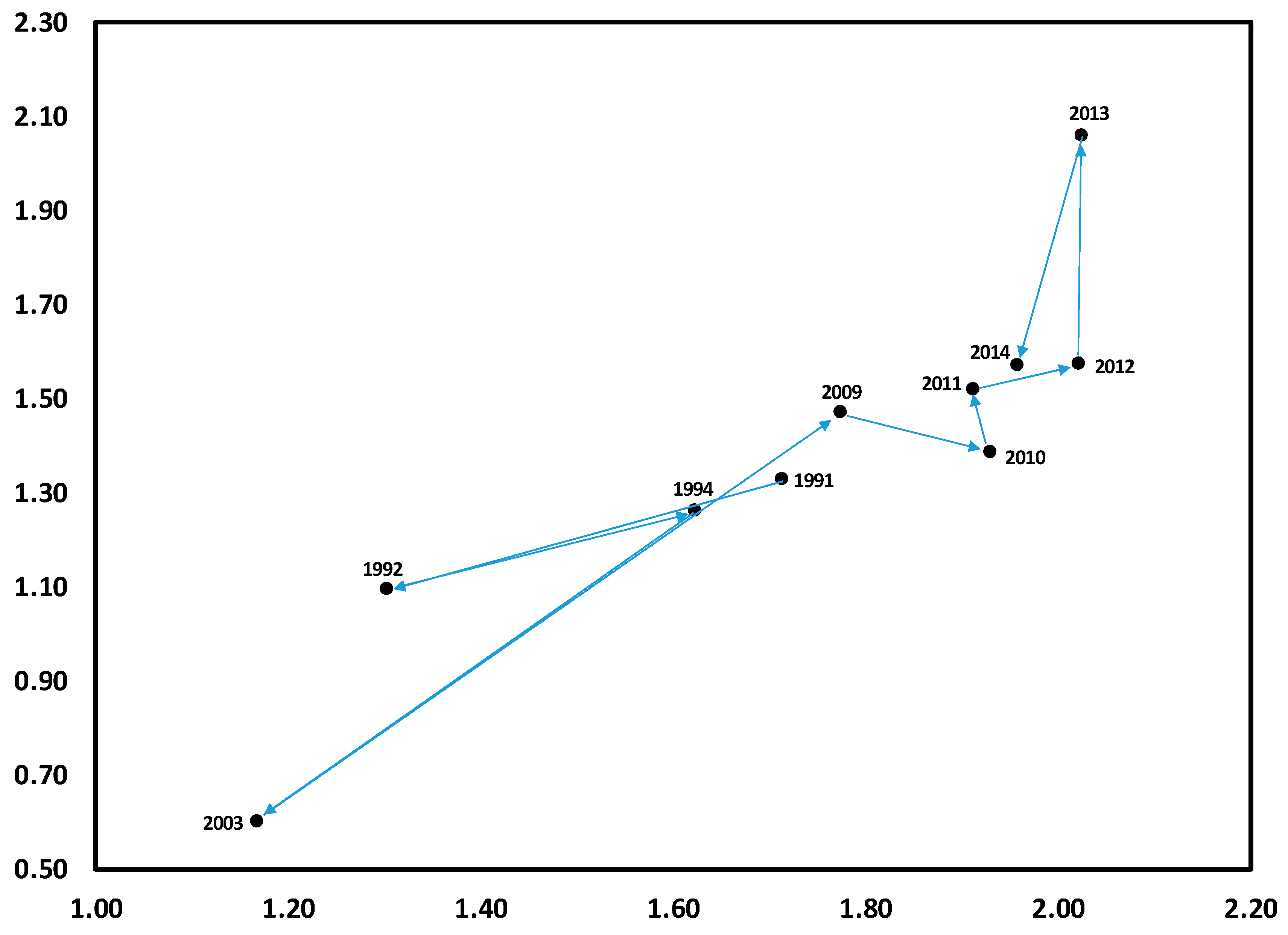
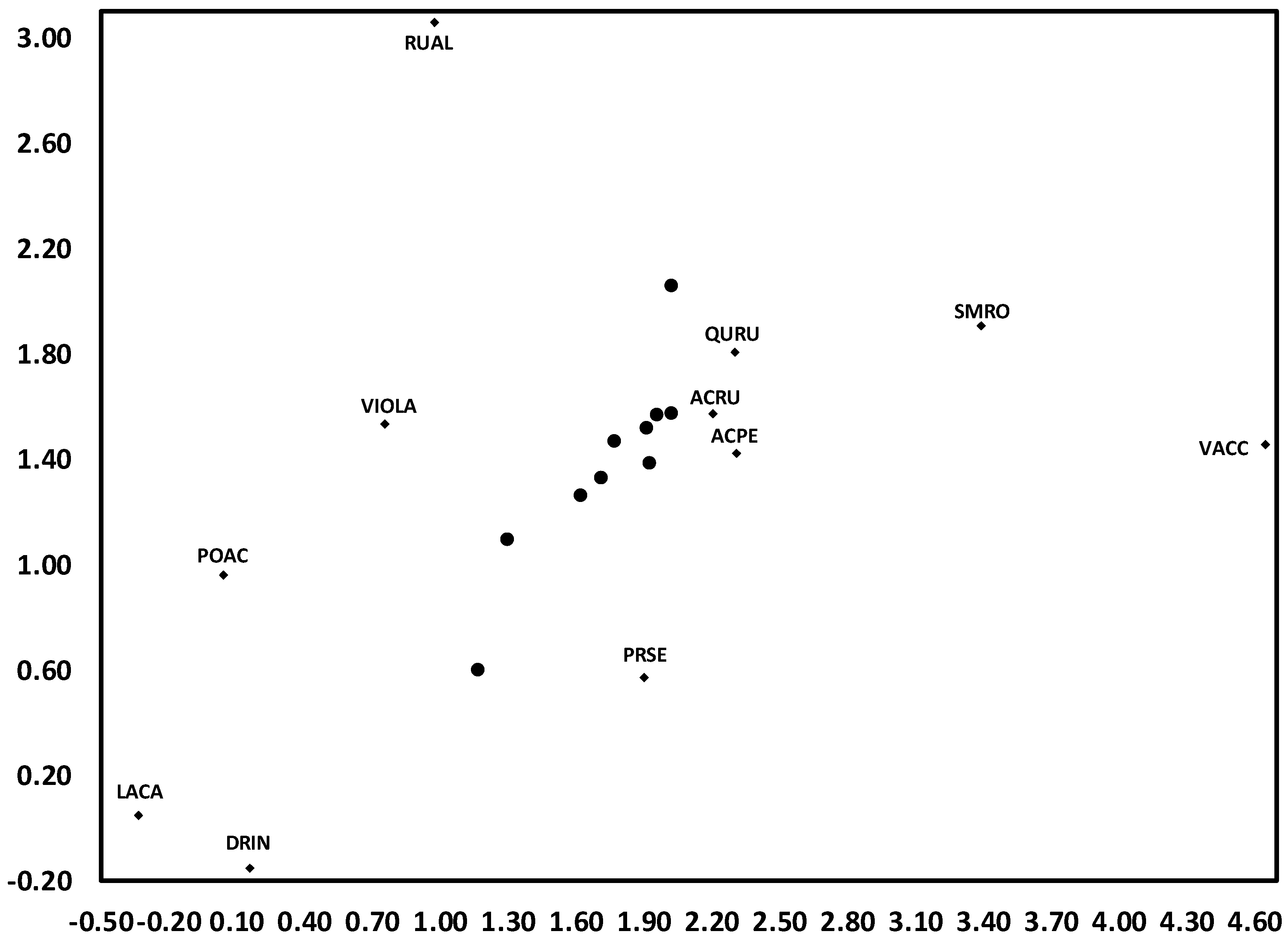
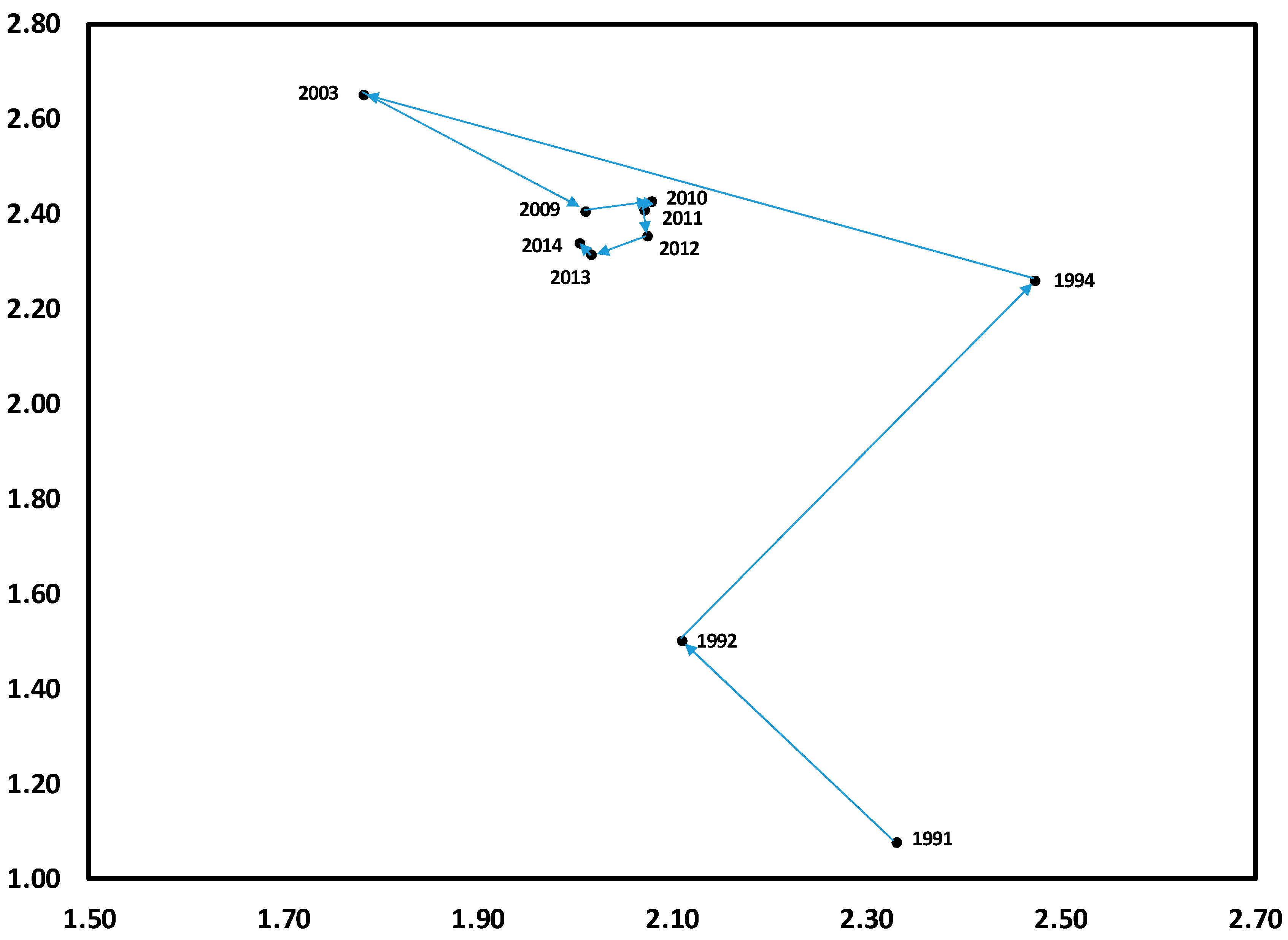
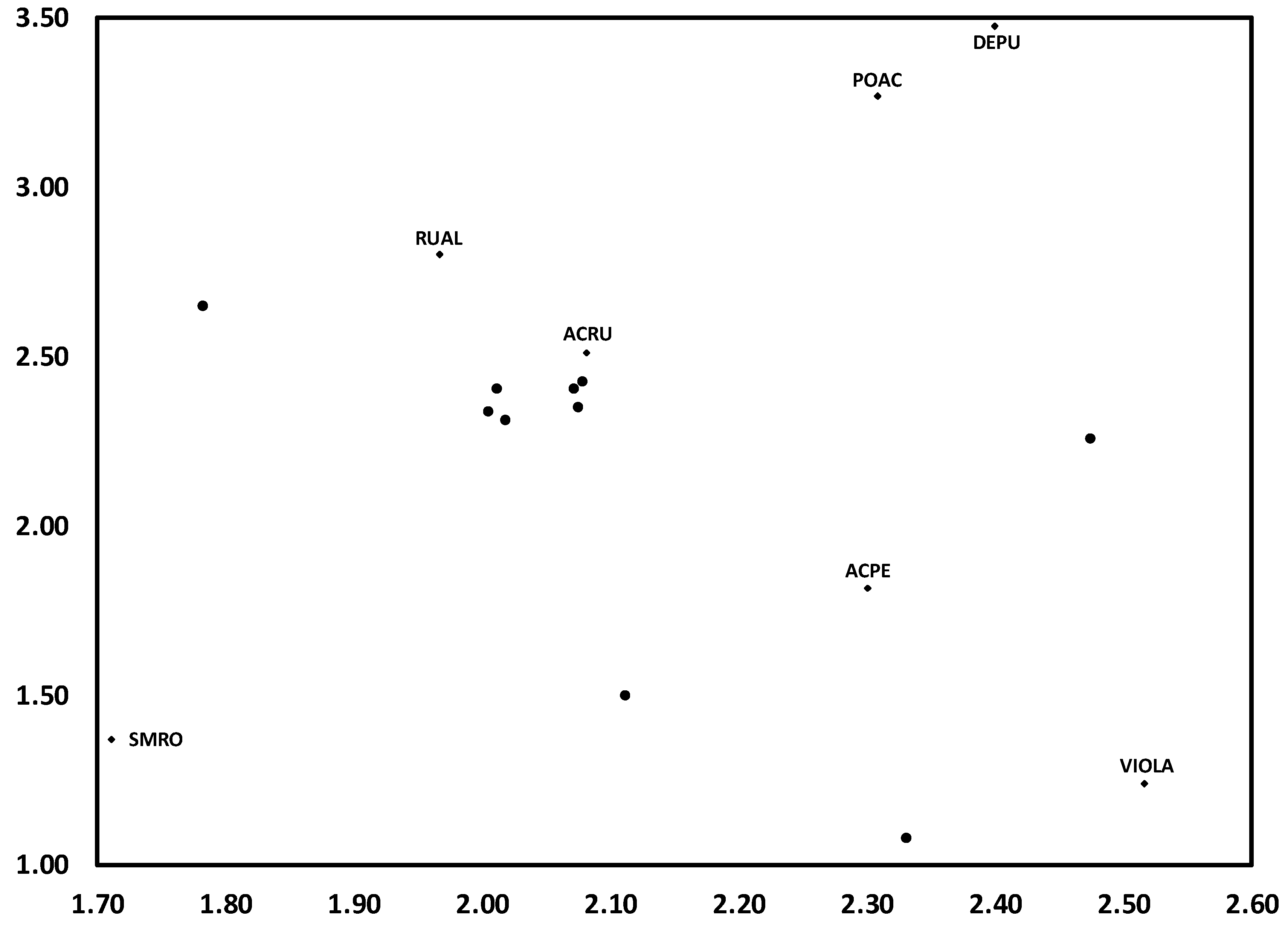
3.1. Temporal Variation in Composition of the Herbaceous Layer
3.2. Change in Herb Diversity Metrics over Time
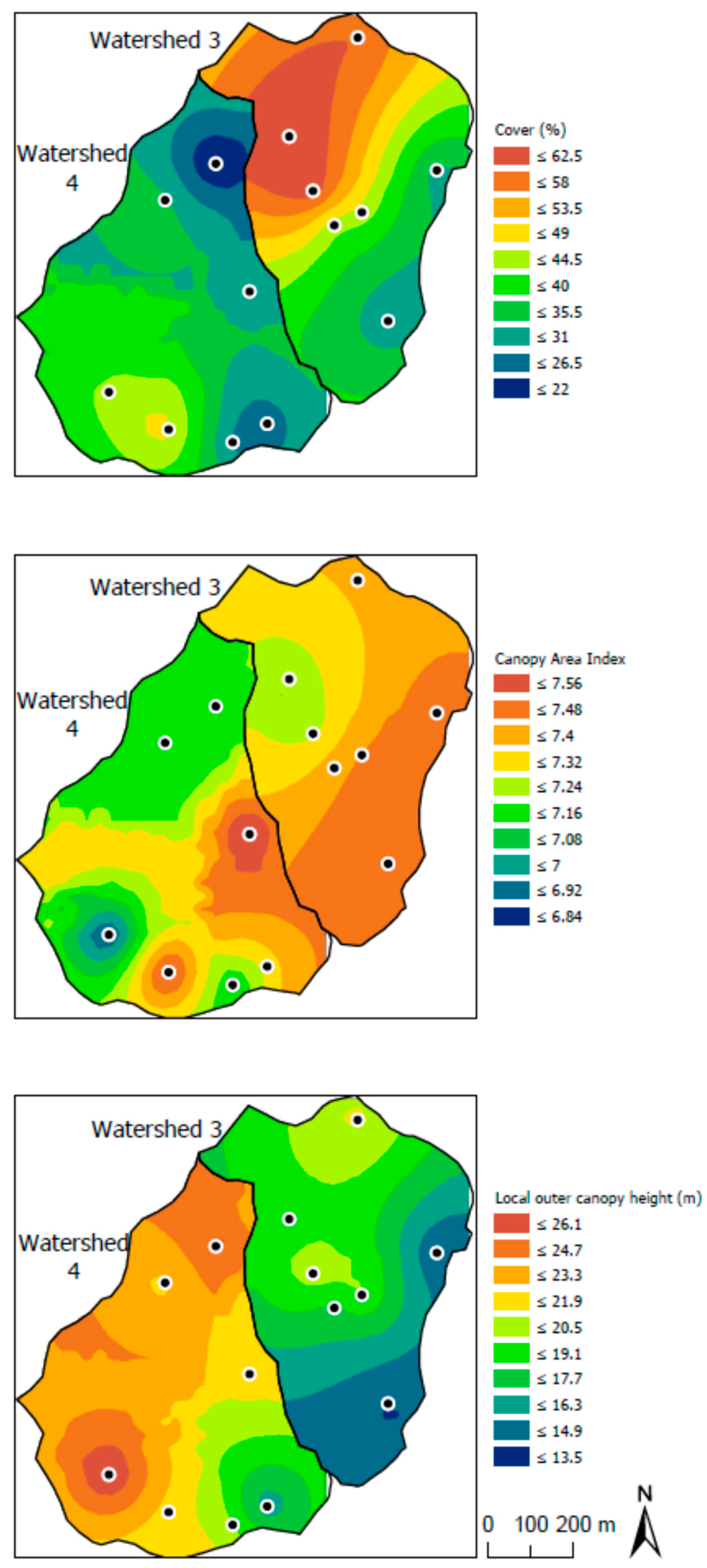
3.3. Influences of Canopy and Soil Variables on Herbaceous Biodiversity and Cover
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Gilliam, F.S. The ecological significance of the herbaceous layer in forest ecosystems. BioScience 2007, 57, 845–858. [Google Scholar] [CrossRef]
- Gilliam, F.S. Introduction: The herbaceous layer—The forest between the trees. In The Herbaceous Layer in Forests of Eastern North America, 2nd ed.; Gilliam, F.S., Ed.; Oxford University Press: New York, NY, USA, 2014; pp. 1–9. ISBN 978-0-19-983765-6. [Google Scholar]
- Thrippleton, T.; Bugmann, H.; Kramer-Priewasser, K.; Snell, R.S. Herbaceous understorey: An overlooked player in forest landscape dynamics? Ecosystems 2016, 19, 1240–1254. [Google Scholar] [CrossRef]
- Neufeld, H.S.; Young, D.R. Ecophysiology of the herbaceous layer in temperate deciduous forests. In The Herbaceous Layer in Forests of Eastern North America, 2nd ed.; Gilliam, F.S., Ed.; Oxford University Press: New York, NY, USA, 2014; pp. 35–95. ISBN 978-0-19-983765-6. [Google Scholar]
- De Frenne, P.; Rodríguez-Sánchez, F.; Coomes, D.A.; Baeten, L.; Verstraeten, G.; Vellend, M.; Bernhardt-Römermann, M.; Brown, C.D.; Brunet, J.; Cornelis, J.; et al. Microclimate moderates plant responses to macroclimate warming. Proc. Natl. Acad. Sci. USA 2013, 110, 18561–18565. [Google Scholar] [CrossRef] [PubMed]
- Bellemare, J.; Moeller, D.A. Climate change and forest herbs of temperate deciduous forests. In The Herbaceous Layer in Forests of Eastern North America, 2nd ed.; Gilliam, F.S., Ed.; Oxford University Press: New York, NY, USA, 2014; pp. 460–493. ISBN 978-0-19-983765-6. [Google Scholar]
- Parker, G.G.; Brown, M.J. Forest canopy stratification—Is it useful? Am. Nat. 2000, 473–484. [Google Scholar]
- Gilliam, F.S. The dynamic nature of the herbaceous layer: A brief synthesis. In The Herbaceous Layer in Forests of Eastern North America, 2nd ed.; Gilliam, F.S., Ed.; Oxford University Press: New York, NY, USA, 2014; pp. 497–508. ISBN 978-0-19-983765-6. [Google Scholar]
- Hutchings, M.J.; John, E.A.; Wijesinghe, D.K. Toward understanding the consequences of soil heterogeneity for plant populations and communities. Ecology 2003, 84, 2322–2334. [Google Scholar] [CrossRef]
- Bartels, S.F.; Chen, H.Y.N. Is understory plant species diversity driven by resource quantity or resource heterogeneity? Ecology 2010, 91, 1931–1938. [Google Scholar] [CrossRef] [PubMed]
- Laliberté, E.; Grace, J.B.; Huston, M.A.; Lambers, H.; Teste, F.P.; Turner, B.L.; Wardle, D.A. How does pedogenesis drive plant diversity? Trends Ecol. Evol. 2013, 28, 331–340. [Google Scholar] [CrossRef]
- Rogers, R.S. Hemlock stands from Wisconsin to Nova Scotia: Transitions in understory composition along a floristic gradient. Ecology 1980, 61, 178–193. [Google Scholar] [CrossRef]
- Rogers, R.S. Mature mesophytic hardwood forest: Community transitions, by layer, from east-central Minnesota to southeastern Michigan. Ecology 1981, 62, 1634–1647. [Google Scholar] [CrossRef]
- Rogers, R.S. Early spring herb communities in mesophytic forests of the Great Lakes Region. Ecology 1982, 63, 1050–1063. [Google Scholar] [CrossRef]
- Rogers, R.S. Annual variability in community organization of forest herbs: Effect of an extremely warm and dry early spring. Ecology 1983, 64, 1086–1091. [Google Scholar] [CrossRef]
- Rogers, R.S. Local coexistence of deciduous-forest groundlayer species growing in different seasons. Ecology 1985, 66, 701–707. [Google Scholar] [CrossRef]
- Reiners, W.A. Twenty years of ecosystem reorganization following experimental deforestation and regrowth suppression. Ecol. Monogr. 1992, 62, 503–523. [Google Scholar] [CrossRef]
- Reich, P.B.; Frelich, L.E.; Voldseth, R.A.; Bakken, P.; Adair, E.C. Understorey diversity in southern boreal forests is regulated by productivity and its indirect impacts on resource availability and heterogeneity. J. Ecol. 2012, 100, 539–545. [Google Scholar] [CrossRef]
- Kumar, P.; Chen, H.Y.N.; Thomas, S.C.; Shahi, C. Linking resource availability and heterogeneity to understorey species diversity through succession in boreal forest of Canada. J. Ecol. 2018, 106, 1266–1276. [Google Scholar] [CrossRef]
- Kumar, P.; Chen, H.Y.N.; Searle, E.B.; Shahi, C. Dynamics of understorey biomass, production and turnover associated with long-term overstorey succession in boreal forest of Canada. For. Ecol. Manag. 2018, 427, 152–161. [Google Scholar] [CrossRef]
- Clark, C.M.; Morefield, P.; Gilliam, F.S.; Pardo, L.H. Estimated losses of plant biodiversity across the U.S. from historical N deposition from 1985–2010. Ecology 2013, 94, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Sutton, M.A.; Mason, K.E.; Sheppard, L.J.; Sverdrup, H.; Haeuber, R.; Hicks, W.K. Nitrogen Deposition, Critical Loads and Biodiversity: Proceedings of the International Nitrogen Initiatives Workshop, Linking Experts of the Convention on Long-Range Transboundary Air Pollution and the Convention on Biological Diversity; Springer: New York, NY, USA, 2014; ISBN 978-94-007-7938-9. [Google Scholar]
- Simkin, S.M.; Allen, E.B.; Bowman, W.D.; Clark, C.M.; Belnap, J.; Brooks, M.L.; Cade, B.S.; Collins, S.L.; Geiser, L.H.; Gilliam, F.S.; et al. A continental analysis of ecosystem vulnerability to atmospheric nitrogen deposition. Proc. Natl. Acad. Sci. USA 2016, 113, 4086–4091. [Google Scholar] [CrossRef]
- Gilliam, F.S.; Welch, N.T.; Phillips, A.H.; Billmyer, J.H.; Peterjohn, W.T.; Fowler, Z.K.; Walter, C.; Burnham, M.; May, J.D.; Adams, M.B. Twenty-five year response of the herbaceous layer of a temperate hardwood forest to elevated nitrogen deposition. Ecosphere 2016, 7, e01250. [Google Scholar] [CrossRef]
- Gilliam, F.S. Response of the herbaceous layer of forest ecosystems to excess nitrogen deposition. J. Ecol. 2006, 94, 1176–1191. [Google Scholar] [CrossRef]
- Walter, C.A.; Raiff, D.T.; Burnham, M.B.; Gilliam, F.S.; Adams, M.B.; Peterjohn, W.T. Nitrogen fertilization interacts with light to increase Rubus spp. cover in a temperate forest. Plant Ecol. 2016, 217, 421–430. [Google Scholar] [CrossRef]
- Lefsky, M.A.; Cohen, W.B.; Parker, G.G.; Harding, D.J. Lidar remote sensing for ecosystem studies. Bioscience 2002, 52, 19–30. [Google Scholar] [CrossRef]
- Walter, C.A.; Burnham, M.B.; Gilliam, F.S.; Peterjohn, W.T. A reference-based approach for measuring the cover of forest herbs. Environ. Monit. Assess. 2015, 187, 657. [Google Scholar] [CrossRef] [PubMed]
- Gilliam, F.S.; Walter, C.A.; Adams, M.B.; Peterjohn, W.T. Nitrogen (N) dynamics in the mineral soil of a Central Appalachian hardwood forest during a quarter century of whole-watershed N additions. Ecosystems 2018, 21, 1489–1504. [Google Scholar] [CrossRef]
- Parker, G.G.; Harding, D.J.; Berger, M. A portable LIDAR system for rapid determination of forest canopy structure. J. Appl. Ecol. 2004, 41, 755–767. [Google Scholar] [CrossRef]
- MacArthur, R.H.; Horn, H.S. Foliage profiles by vertical measurements. Ecology 1969, 50, 802–804. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Prentice-Hall: Upper Saddle River, NJ, USA, 2009; ISBN 0-13-100846-3. [Google Scholar]
- Pebesma, E.J. Multivariable geostatistics in S: The gstat package. Comput. Geosci. 2004, 30, 683–691. [Google Scholar] [CrossRef]
- Kochenderfer, J.N. Fernow and the Appalachian hardwood region. In The Fernow Watershed Acidification Study; Adams, M.B., DeWalle, D.R., Hom, J.L., Eds.; Springer: Dordrecht, The Netherlands, 2006; Chapter 2; pp. 17–40. ISBN 1-4020-4614-6. [Google Scholar]
- Gilliam, F.S.; Yurish, B.M.; Adams, M.B. Temporal and spatial variation of nitrogen transformations in nitrogen-saturated soils of a Central Appalachian hardwood forest. Can. J. For. Res. 2001, 31, 1768–1785. [Google Scholar] [CrossRef]
- Gilliam, F.S.; May, J.D.; Adams, M.B. Response of foliar nutrients of Rubus allegheniensis to nutrient amendments in a central Appalachian hardwood forest. For. Ecol. Manag. 2018, 411, 101–107. [Google Scholar] [CrossRef]
- Woodland, D.W. Biosystematics of the perennial North American taxa Urtica. II. Taxonomy. Syst. Bot. 1982, 7, 282–290. [Google Scholar] [CrossRef]
- Hoshizaki, B.J.; Wilson, K.A. The cultivated species of the fern genus Dryopteris in the United States. Am. Fern J. 1999, 89, 1–98. [Google Scholar] [CrossRef]
- Roberts, M.R. Response of the herbaceous layer to natural disturbance in North American forests. Can. J. Bot. 2004, 1273–1283. [Google Scholar] [CrossRef]
- Gilliam, F.S. Effects of excess nitrogen deposition on the herbaceous layer of eastern North American forests. In The Herbaceous Layer in Forests of Eastern North America, 2nd ed.; Gilliam, F.S., Ed.; Oxford University Press: New York, NY, USA, 2014; pp. 497–508. ISBN 978-0-19-983765-6. [Google Scholar]
- Verheyen, K.; Baeten, L.; De Frenne, P.; Bernhardt-Römermann, M.; Brunet, J.; Cornelis, J.; Decocq, G.; Dierschke, H.; Hédl, R.; Heinken, T.; et al. Driving factors behind the eutrophication signal in understorey plant communities of deciduous temperate forests. J. Ecol. 2012, 99, 352–365. [Google Scholar] [CrossRef]
- Dirnböck, T.; Grandin, U.; Bernhardt-Römermann, M.; Beudert, B.; Canullo, R.; Forsius, M.; Grabner, M.-T.; Holmberg, M.; Kleemola, S.; Lundin, L.; et al. Forest floor vegetation response to nitrogen deposition in Europe. Glob. Chang. Biol. 2014, 20, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Roth, T.; Kohli, L.; Rihm, B.; Amrhein, V.; Achermann, B. Nitrogen deposition and multidimensional plant diversity at the landscape scale. R. Soc. Open Sci. 2015, 2, 150017. [Google Scholar] [CrossRef] [PubMed]
- De Schrijver, A.; de Frenne, P.; Ampoorter, E.; van Nevel, L.; Demey, A.; Wuyts, K.; Verheyen, K. Cumulative nitrogen inputs drives species loss in terrestrial ecosystems. Glob. Ecol. Biogeogr. 2011, 20, 803–816. [Google Scholar] [CrossRef]
- Ferretti, M.; Marchetto, A.; Arisci, S.; Bussotti, F.; Calderisi, M.; Carnicelli, S.; Cecchini, G.; Fabbio, G.; Bertini, G.; Matteucci, G.; et al. On the tracks of nitrogen deposition effects on temperate forests at their southern European range—An observational study from Italy. Glob. Chang. Biol. 2014, 20, 3423–3438. [Google Scholar] [CrossRef] [PubMed]
- Walter, C.A.; Adams, M.B.; Gilliam, F.S.; Peterjohn, W.T. Non-random species loss in a forest herbaceous layer following nitrogen addition. Ecology 2017, 98, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Du, E.; de Vries, W.; Galloway, J.N.; Hu, X.; Fang, J. Changes in wet nitrogen deposition in the United States between 1985 and 2012. Environ. Res. Lett. 2014, 9, 095004. [Google Scholar] [CrossRef]
- Du, E. Rise and fall of nitrogen deposition in the United States. Proc. Natl. Acad. Sci. USA 2016, 113, E3594–E3595. [Google Scholar] [CrossRef]
- Gilliam, F.S.; Burns, D.A.; Driscoll, C.T.; Frey, S.D.; Lovett, G.M.; Watmough, S.A. Decreased atmospheric nitrogen deposition in eastern North America: Predicted responses of forest ecosystems. Environ. Pollut. 2019, 244, 560–574. [Google Scholar] [CrossRef] [PubMed]
| Metric | WS4 | WS3 |
|---|---|---|
| S | 0.91 | NS |
| J | NS | −0.81 |
| Hill’s N2 | NS | −0.64 |
| Rubus | 0.86 | 0.98 |
| Watershed | Final Model | r2 |
|---|---|---|
| WS4 | Cover = 60.8 − 5.3NH4+ + 12.6NO3− | 0.85 |
| WS3 | Cover = 408.5 − 58.9CAI + 3.1LOCH − 909.6gapfrac | 0.998 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gilliam, F.S. Excess Nitrogen in Temperate Forest Ecosystems Decreases Herbaceous Layer Diversity and Shifts Control from Soil to Canopy Structure. Forests 2019, 10, 66. https://doi.org/10.3390/f10010066
Gilliam FS. Excess Nitrogen in Temperate Forest Ecosystems Decreases Herbaceous Layer Diversity and Shifts Control from Soil to Canopy Structure. Forests. 2019; 10(1):66. https://doi.org/10.3390/f10010066
Chicago/Turabian StyleGilliam, Frank S. 2019. "Excess Nitrogen in Temperate Forest Ecosystems Decreases Herbaceous Layer Diversity and Shifts Control from Soil to Canopy Structure" Forests 10, no. 1: 66. https://doi.org/10.3390/f10010066
APA StyleGilliam, F. S. (2019). Excess Nitrogen in Temperate Forest Ecosystems Decreases Herbaceous Layer Diversity and Shifts Control from Soil to Canopy Structure. Forests, 10(1), 66. https://doi.org/10.3390/f10010066




