DNA Barcoding Analysis and Phylogenetic Relation of Mangroves in Guangdong Province, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. DNA Extraction and Sequence Analysis
2.3. Statistical Analysis
3. Results
3.1. Sequence Universality
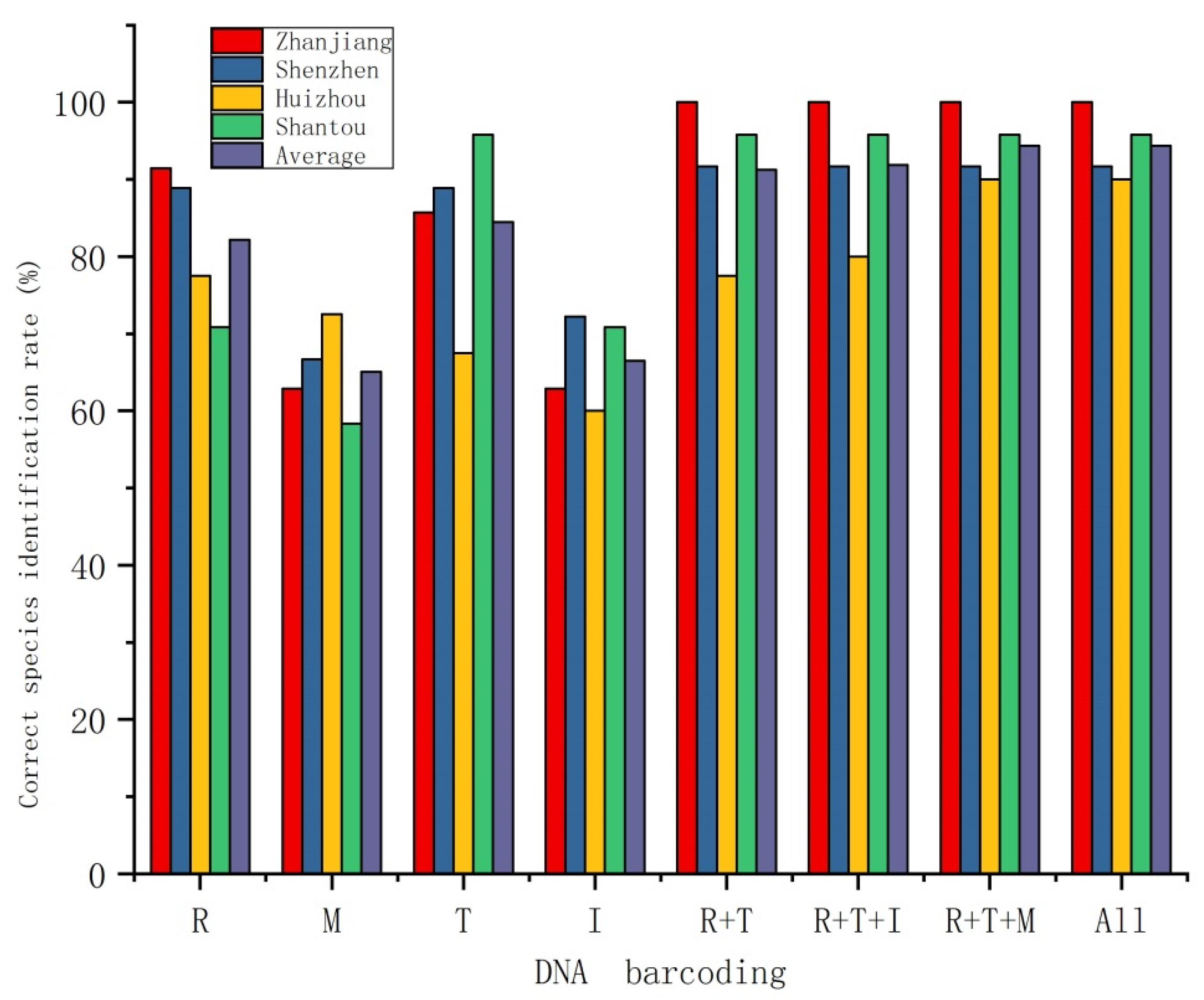
3.2. Species Delimitation
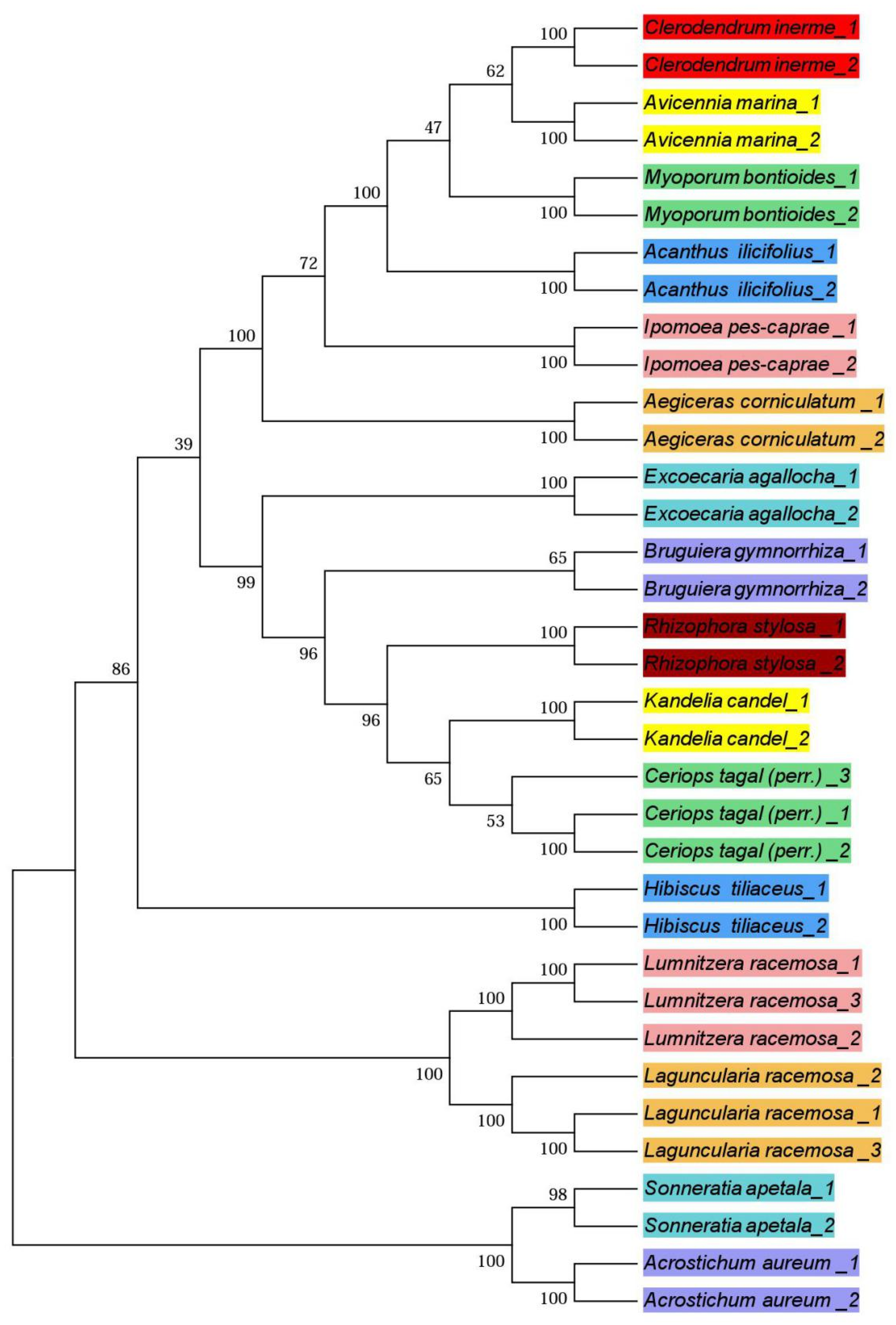
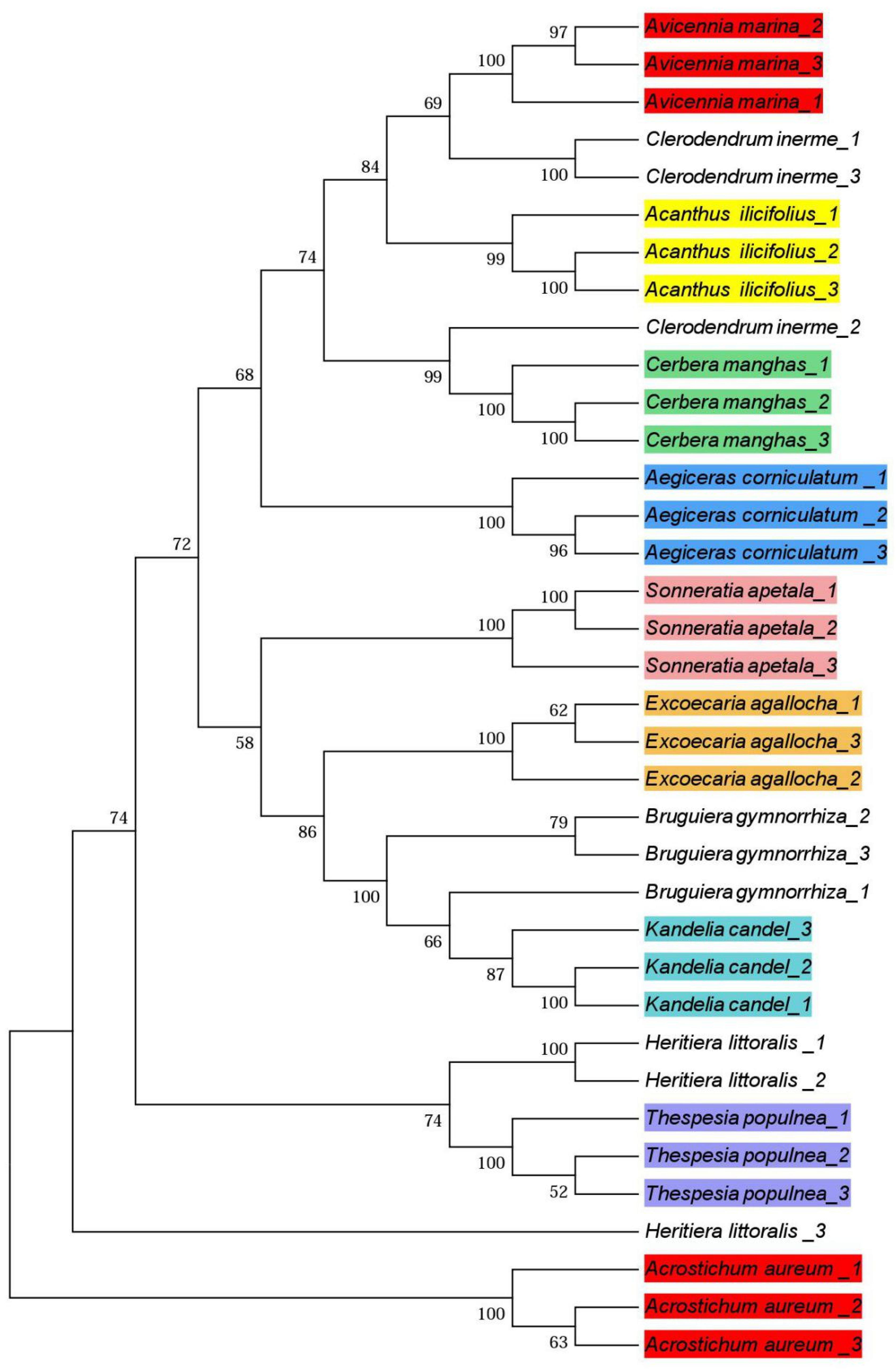
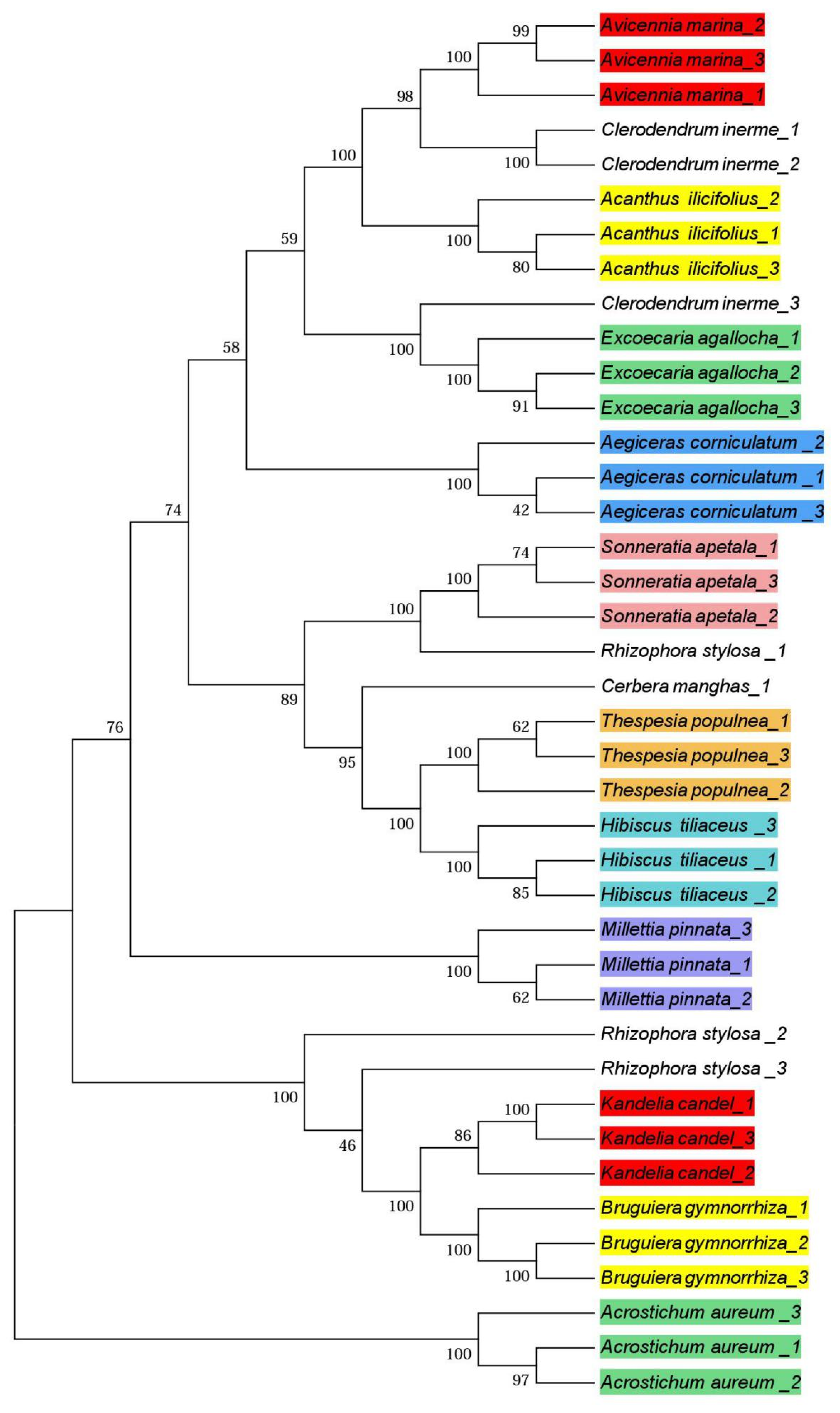
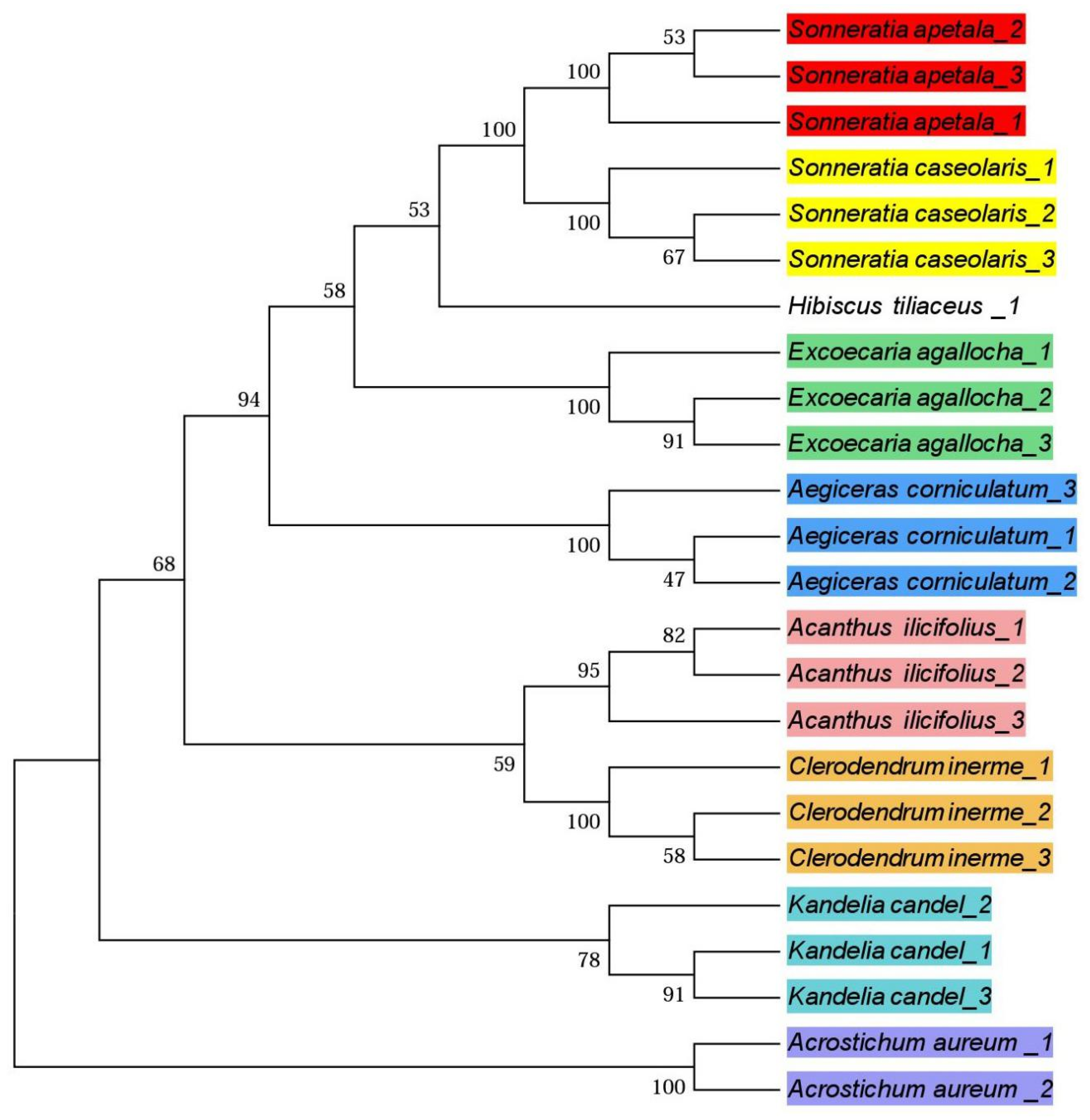
3.3. Phylogenetic Trees
4. Discussion
4.1. The Universality of DNA Barcoding in Mangrove Communities
4.2. Species Identification Ability
4.3. Phylogenetic Trees
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hebert, P.D.; Cywinska, A.; Ball, S.L.; de Waard, J.R. Biological identifications through DNA barcodes. Proc. Biol. Sci. 2003, 270, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Chase, M.W.; Salamin, N.; Wilkinson, M.; Dunwell, J.M.; Kesanakurthi, R.P.; Haider, N.; Savolainen, V. Land plants and DNA barcodes: Short-term and long-term goals. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2005, 360, 1889–1895. [Google Scholar] [CrossRef] [PubMed]
- Kress, W.J.; Wurdack, K.J.; Zimmer, E.A.; Weigt, L.A.; Janzen, D.H. Use of DNA barcodes to identify flowering plants. Proc. Natl. Acad. Sci. USA 2005, 102, 8369–8374. [Google Scholar] [CrossRef] [PubMed]
- CBOL Plant Working Group. A DNA barcode for land plants. Proc. Natl. Acad. Sci. USA 2009, 106, 12794–12797. [Google Scholar] [CrossRef] [PubMed]
- Kress, W.J.; Erickson, D.L. A two-locus global DNA barcode for land plants: The coding rbcL gene complements the non-coding trnH-psbA spacer region. PLoS ONE 2007, 2, e508. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yao, H.; Han, J.; Liu, C.; Song, J.; Shi, L.; Zhu, Y.; Ma, X.; Gao, T.; Pang, X.; et al. Validation of the ITS2 region as a novel DNA barcode for identifying medicinal plant species. PLoS ONE 2010, 5, e8613. [Google Scholar] [CrossRef]
- Li, D.Z.; Gao, L.M.; Li, H.T.; Wang, H.; Ge, X.J.; Liu, J.Q.; Chen, Z.D.; Zhou, S.L.; Chen, S.L.; Yang, J.B.; et al. Comparative analysis of a large dataset indicates that internal transcribed spacer (its) should be incorporated into the core barcode for seed plants. Proc. Natl. Acad. Sci. USA 2011, 108, 19641–19646. [Google Scholar]
- Kress, W.J.; Erickson, D.L.; Swenson, N.G.; Thompson, J.; Uriarte, M.; Zimmerman, J.K. Advances in the use of DNA barcodes to build a community phylogeny for tropical trees in a Puerto Rican forest dynamics plot. PLoS ONE 2010, 5, e15409. [Google Scholar] [CrossRef]
- Hollingsworth, P.M. Refining the DNA barcode for land plants. Proc. Natl. Acad. Sci. USA 2011, 108, 19451–19452. [Google Scholar] [CrossRef]
- Li, H.Q.; Chen, J.Y.; Wang, S.; Xiong, S.Z. Evaluation of six candidate DNA barcoding loci in Ficus (Moraceae) of China. Mol. Ecol. Resour. 2012, 12, 783–790. [Google Scholar] [CrossRef]
- Braukmann, T.W.; Kuzmina, M.L.; Sills, J.; Zakharov, E.V.; Hebert, P.D. Testing the efficacy of DNA barcodes for identifying the vascular plants of Canada. PLoS ONE 2017, 12, e169515. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.A.; Baraloto, C.; Engel, J.; Mori, S.A.; Petronelli, P.; Riera, B.; Roger, A.; Thebaud, C.; Chave, J. Identification of Amazonian trees with DNA barcodes. PLoS ONE 2009, 4, e7483. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yan, H.F.; Newmaster, S.G.; Pei, N.; Ragupathy, S.; Ge, X.J.; Lowe, A. The use of DNA barcoding as a tool for the conservation biogeography of subtropical forests in China. Divers. Distrib. 2015, 21, 188–199. [Google Scholar] [CrossRef]
- Parmentier, I.; Duminil, J.; Kuzmina, M.; Philippe, M.; Thomas, D.W.; Kenfack, D.; Chuyong, G.B.; Cruaud, C.; Hardy, O.J. How effective are DNA Barcodes in the identification of African rainforest trees. PLoS ONE 2013, 8, e54921. [Google Scholar] [CrossRef] [PubMed]
- Kress, W.J.; Erickson, D.L. DNA barcodes: Methods and protocols. Methods Mol. Biol. 2012, 858, 3–8. [Google Scholar] [PubMed]
- Li, D.Z.; Zeng, C.X. Prospects for plant DNA barcoding. Biodivers. Sci. 2015, 23, 297–298. [Google Scholar] [CrossRef]
- Kress, W.J.; Garcia-Robledo, C.; Uriarte, M.; Erickson, D.L. DNA barcodes for ecology, evolution, and conservation. Trends Ecol. Evol. 2015, 30, 25–35. [Google Scholar] [CrossRef]
- Pei, N.C.; Chen, B.F.; Kress, W.J. Advances of community-level plant DNA barcoding in China. Front. Plant Sci. 2017, 8, 225. [Google Scholar] [CrossRef]
- Kress, W.J.; Erickson, D.L.; Jones, F.A.; Swenson, N.G.; Perez, R.; Sanjur, O.; Bermingham, E. Plant DNA barcodes and a community phylogeny of a tropical forest dynamics plot in Panama. Proc. Natl. Acad. Sci. USA 2009, 106, 18621–18626. [Google Scholar] [CrossRef]
- Pei, N.C.; Zhang, J.L.; Mi, X.C.; Ge, X.J. Plant DNA barcodes promote the development of phylogenetic community ecology. Biodivers. Sci. 2011, 19, 284–294. (In Chinese) [Google Scholar]
- Valentini, A.; Pompanon, F.; Taberlet, P. DNA barcoding for ecologists. Trends Ecol. Evol. 2009, 24, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.A.; Roger, A.; Courtois, E.A.; Jabot, F.; Norden, N.; Paine, C.E.T.; Baraloto, C.; Thébaud, C.; Chave, J.M. Shifts in species and phylogenetic diversity between sapling and tree communities indicate negative density dependence in a lowland rain forest. J. Ecol. 2010, 98, 137–146. [Google Scholar] [CrossRef]
- Lin, P. Chinese Mangrove Ecosystem; Science Press: Beijing, China, 1997; pp. 124–156. ISBN 9787030056924. (In Chinese) [Google Scholar]
- Dan, X.Q.; Liao, B.W.; Wu, Z.B.; Wu, H.J.; Bao, D.M. resources, conservation status and main threats of mangrove wetlands in China. Ecol. Environ. Sci. 2016, 25, 1237–1243. (In Chinese) [Google Scholar]
- Wang, B.J.; Liang, S.C.; Zhang, W.Y.; Zan, Q.J. Mangrove flora of the world. Acta Bot. Sin. 2003, 45, 644–653. (In Chinese) [Google Scholar]
- Liao, B.W.; Zhang, Q.M. Area, distribution and species composition of mangroves in China. Wetl. Sci. 2014, 12, 435–440. (In Chinese) [Google Scholar]
- Murphy, R.W.; Crawford, A.J.; Bauer, A.M.; Che, J.; Donnellan, S.C.; Fritz, U.; Haddad, C.F.B.; Nagy, Z.T.; Poyarkov, N.A.; Vences, M.; et al. Cold Code: The global initiative to DNA barcode amphibians and nonavian reptiles. Mol. Ecol. Resour. 2013, 13, 161–167. [Google Scholar] [CrossRef]
- Shen, Y.Y.; Chen, X.; Murphy, R.W. Assessing DNA barcoding as a tool for species identification and data quality control. PLoS ONE 2013, 8, e57125. [Google Scholar] [CrossRef]
- Martoni, F.; Bulman, S.; Pitman, A.; Taylor, G.; Armstrong, K. DNA barcoding highlights cryptic diversity in the New Zealand Psylloidea (Hemiptera: Sternorrhyncha). Diversity 2018, 10, 50. [Google Scholar] [CrossRef]
- Pfeiler, E. DNA barcoding and taxonomic challenges in describing new putative species: Examples from sootywing and cloudywing butterflies (Lepidoptera: Hesperiidae). Diversity 2018, 10, 111. [Google Scholar] [CrossRef]
- Pei, N.C.; Lian, J.Y.; Erickson, D.L.; Swenson, N.G.; Kress, W.J.; Ye, W.H.; Ge, X.J. Exploring tree-habitat associations in a Chinese subtropical forest plot using a molecular phylogeny generated from DNA barcode Loci. PLoS ONE 2011, 6, e21273. [Google Scholar] [CrossRef]
- Pawlowski, J.; Audic, S.; Adl, S.; Bass, D.; Belbahri, L.; Berney, C.; Bowser, S.; Cepicka, I.; Decelle, J.; Dunthorn, M.; et al. CBOL protist working group: Barcoding eukaryotic richness beyond the animal, plant, and fungal kingdoms. PLoS Biol. 2012, 10, e1001419. [Google Scholar] [CrossRef] [PubMed]
- Galimberti, A.; de Mattia, F.; Losa, A.; Bruni, I.; Federici, S.; Casiraghi, M.; Martellos, S.; Labra, M. DNA barcoding as a new tool for food traceability. Food Res. Int. 2013, 50, 55–63. [Google Scholar] [CrossRef]
- Gao, L.M.; Liu, J.; Cai, J.; Yang, J.B.; Zhang, T. A synopsis of technical notes on the standards for plant DNA barcoding. Plant Divers. Resour. 2012, 34, 592–606. (In Chinese) [Google Scholar] [CrossRef]
- Doyle, J.; Doyle, J. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Yan, H.F.; Liu, Y.J.; Xie, X.F.; Zhang, C.Y.; Hu, C.M.; Hao, G.; Ge, X.J. DNA barcoding evaluation and its taxonomic implications in the species-rich genus Primula L. in China. PLoS ONE 2015, 10, e122903. [Google Scholar] [CrossRef] [PubMed]
- Liu, J. DNA Barcoding Evaluation of the Dinghusahn Woody Species and the Genus Gentiana L. (Gentianaceae). Ph.D. Thesis, South China Botanical Garden, The Chinese Academy of Sciences, Guangzhou, China, 2014; pp. 27–28. (In Chinese). [Google Scholar]
- Sang, T.; Crawford, D.; Stuessy, T. Chloroplast DNA phylogeny, reticulate evolution, and biogeography of Paeonia (Paeoniaceae). Am. J. Bot. 1997, 84, 1120. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.A.; Simpson, B.B. Paraphyly of Tarasa (Malvaceae) and diverse origins of the polyploid species. Syst. Bot. 2003, 28, 723–737. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Burgess, K.S.; Fazekas, A.J.; Kesanakurti, P.R.; Graham, S.W.; Husband, B.C.; Newmaster, S.G.; Percy, D.M.; Hajibabaei, M.; Barrett, S.C.H. Discriminating plant species in a local temperate flora using the rbcL+matK DNA barcode. Methods Appl. Fluores 2011, 2, 333–340. [Google Scholar] [CrossRef]
- Pei, N.; Erickson, D.L.; Chen, B.; Ge, X.; Mi, X.; Swenson, N.G.; Zhang, J.; Jones, F.A.; Huang, C.; Ye, W.; et al. Closely-related taxa influence woody species discrimination via DNA barcoding: Evidence from global forest dynamics plots. Sci. Rep. 2015, 5, 15127. [Google Scholar] [CrossRef]
- Costion, C.; Ford, A.; Cross, H.; Crayn, D.; Harrington, M.; Lowe, A. Plant DNA barcodes can accurately estimate species richness in poorly known floras. PLoS ONE 2011, 6, e26841. [Google Scholar] [CrossRef]
- Lu, M.M.; Ci, X.Q.; Yang, G.P.; Li, J. DNA barcoding of subtropical forest trees—A study from Ailao mountains nature reserve, Yunnan, China. Plant Divers. Resour. 2013, 35, 733–741. (In Chinese) [Google Scholar]
- Wei, Y.N.; Wang, X.M.; Yao, P.C.; Chen, X.Y.; Li, H.Q. Comparison of species resolution rates of DNA barcoding for Chinese coastal halo-tolerant plants. Biodivers. Sci. 2017, 25, 1095–1104. (In Chinese) [Google Scholar] [CrossRef]
- Tripathi, A.M.; Tyagi, A.; Kumar, A.; Singh, A.; Singh, S.; Chaudhary, L.B.; Roy, S. The internal transcribed spacer (its) region and trnH-psbA [corrected] are suitable candidate loci for DNA barcoding of tropical tree species of India. PLoS ONE 2013, 8, e57934. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Deng, Z.; Zang, R.; Long, W. DNA barcoding analysis and phylogenetic relationships of tree species in tropical cloud forests. Sci. Rep. 2017, 7, 12564. [Google Scholar] [CrossRef] [PubMed]
- De Vere, N.; Rich, T.C.; Ford, C.R.; Trinder, S.A.; Long, C.; Moore, C.W.; Satterthwaite, D.; Davies, H.; Allainguillaume, J.; Ronca, S.; et al. DNA barcoding the native flowering plants and conifers of Wales. PLoS ONE 2012, 7, e37945. [Google Scholar] [CrossRef] [PubMed]
- Sass, C.; Little, D.P.; Stevenson, D.W.; Specht, C.D. DNA barcoding in the cycadales: Testing the potential of proposed barcoding markers for species identification of cycads. PLoS ONE 2007, 2, e1154. [Google Scholar] [CrossRef] [PubMed]
- Pei, N.C. Building a subtropical forest community phylogeny based on plant DNA barcodes from Dinghushan plot. Plant Divers. Resour. 2012, 34, 263–270. (In Chinese) [Google Scholar] [CrossRef]
| Site | Collection Places | Longitude | Latitude | Numbers of Tree Species |
|---|---|---|---|---|
| Zhanjiang | Baitoupo | 109°45′38″ | 21°34′03″ | 16 |
| Fengdi | 109°46′21″ | 21°32′36″ | ||
| Shenzhen | Mangrove reserve | 114°00′33″ | 22°31′33″ | 12 |
| Huizhou | Yanzhou island | 114°56′17″ | 22°43′47″ | 14 |
| Xiezhouwan | 114°47′00″ | 22°49′40″ | ||
| Haozhaolou | 114°52′02″ | 22°45′58″ | ||
| Shantou | Su aiwan | 116°43′36″ | 23°19′16″ | 9 |
| Waishahe | 116°48′11″ | 23°23′09″ | ||
| Yifengxi | 116°53′11″ | 23°32′45″ |
| DNA Fragment | Primers | Sequences (5′–3′) | References | Amplification Protocol |
|---|---|---|---|---|
| rbcL | rbcLa_F | ATGTCACCACAAACAGAGACTAAAGC | Kress [19] | 94 °C 3 min; 94 °C 30 s, 55 °C 1 min, 72 °C 1 min, 35 cycles; 72 °C 7 min |
| rbcLa_R | GTAAAATCAAGTCCACCRCG | |||
| matK | Kim_3F | CGTACAGTACTTTTGTGTTTACGAG | Kim, unpublished | 94 °C 3 min; 94 °C 45 s, 51 °C 45 s, 72 °C 1 min, 35 cycles; 72 °C 7 min |
| Kim_1R | ACCCAGTCCATCTGGAAATCTTGGTTC | |||
| trnH-psbA | psbA3 | GTTATGCATGAACGTAATGCTC | Sang [38] | 94 °C 3 min; 94 °C 30 s, 55 °C 1 min, 72 °C 1 min,35 cycles; 72 °C 7 min |
| trnH05 | CGCGCATGGTGGATTCACAATCC | Tate [39] | ||
| ITS | ITS-leu1 | GTCCACTGAACCTTATCATTTAG | Urbatsh | 94 °C 3 min; 94 °C 30 s, 55 °C 1 min, 72 °C 1 min, 35 cycles; 72 °C 7 min |
| ITS4 | TCCTCCGCTTATTGATATGC | White [40] |
| Plot | DNA Fragment | PCR Amplification Success Rate (%) | Sequencing Success Rate (%) | Individuals Resolution Rate |
|---|---|---|---|---|
| Zhanjiang | rbcL | 100 | 100 | 35 |
| matK | 71.43 | 65.71 | 23 | |
| trnH-psbA | 100 | 88.57 | 31 | |
| ITS | 94.29 | 82.86 | 29 | |
| Shenzhen | rbcL | 100 | 100 | 36 |
| matK | 80.56 | 77.78 | 28 | |
| trnH-psbA | 100 | 97.22 | 35 | |
| ITS | 94.44 | 88.89 | 32 | |
| Huizhou | rbcL | 100 | 100 | 40 |
| matK | 77.5 | 77.5 | 31 | |
| trnH-psbA | 97.5 | 92.5 | 37 | |
| ITS | 100 | 82.5 | 33 | |
| Shantou | rbcL | 100 | 100 | 24 |
| matK | 91.67 | 79.17 | 19 | |
| trnH-psbA | 100 | 100 | 24 | |
| ITS | 100 | 79.17 | 19 | |
| Total | 476 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, F.; Li, M.; Liao, B.; Shi, X.; Xu, Y. DNA Barcoding Analysis and Phylogenetic Relation of Mangroves in Guangdong Province, China. Forests 2019, 10, 56. https://doi.org/10.3390/f10010056
Wu F, Li M, Liao B, Shi X, Xu Y. DNA Barcoding Analysis and Phylogenetic Relation of Mangroves in Guangdong Province, China. Forests. 2019; 10(1):56. https://doi.org/10.3390/f10010056
Chicago/Turabian StyleWu, Feng, Mei Li, Baowen Liao, Xin Shi, and Yong Xu. 2019. "DNA Barcoding Analysis and Phylogenetic Relation of Mangroves in Guangdong Province, China" Forests 10, no. 1: 56. https://doi.org/10.3390/f10010056
APA StyleWu, F., Li, M., Liao, B., Shi, X., & Xu, Y. (2019). DNA Barcoding Analysis and Phylogenetic Relation of Mangroves in Guangdong Province, China. Forests, 10(1), 56. https://doi.org/10.3390/f10010056





