Real-Time Tumor Motion Tracking in 3D Using Planning 4D CT Images during Image-Guided Radiation Therapy
Abstract
1. Introduction
2. Materials and Methods
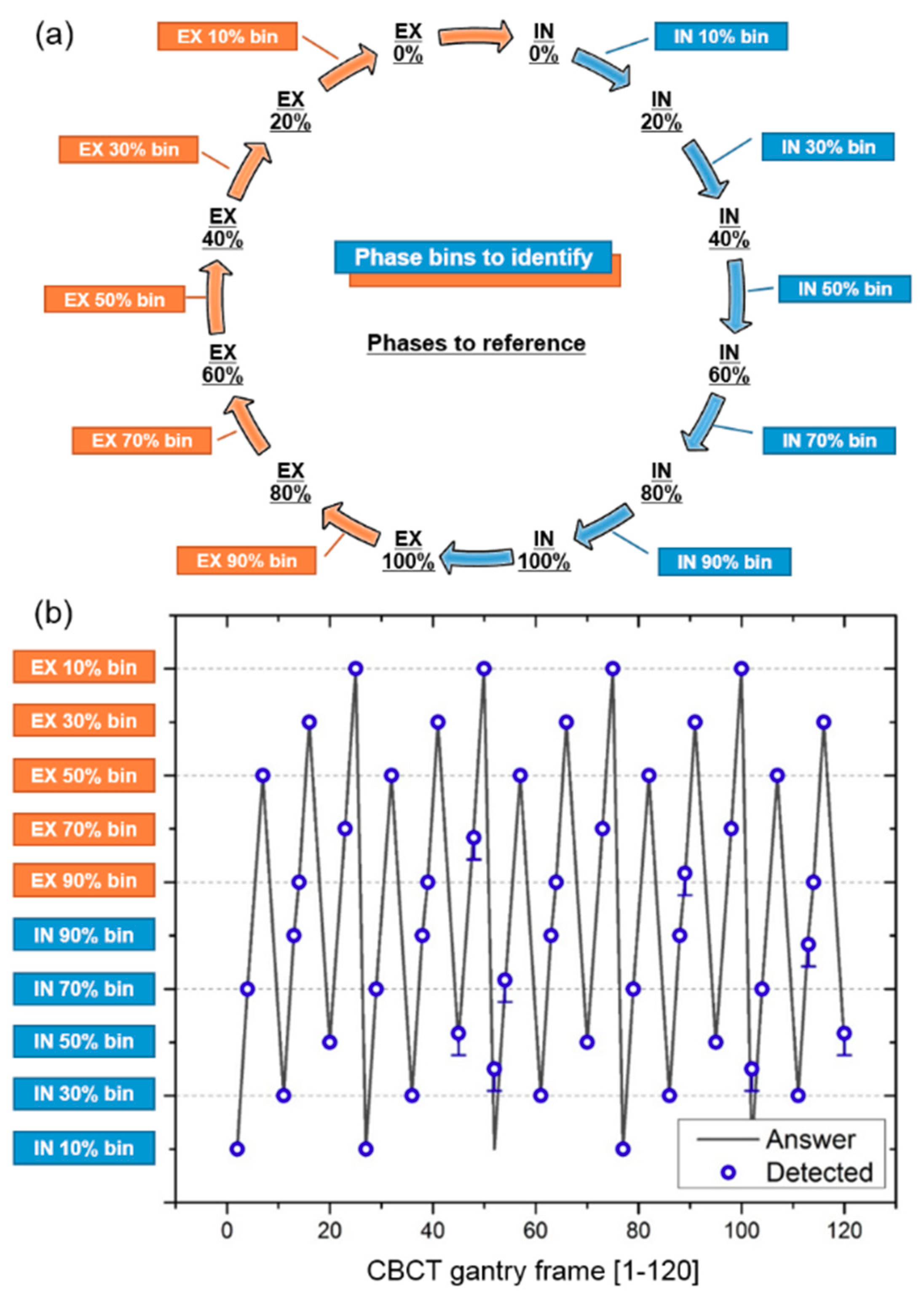
2.1. Pipelines for Respiratory Phase Identification
2.1.1. Pre-processing of Planning and Treatment Images
2.1.2. Structural Similarity-Based Ranking
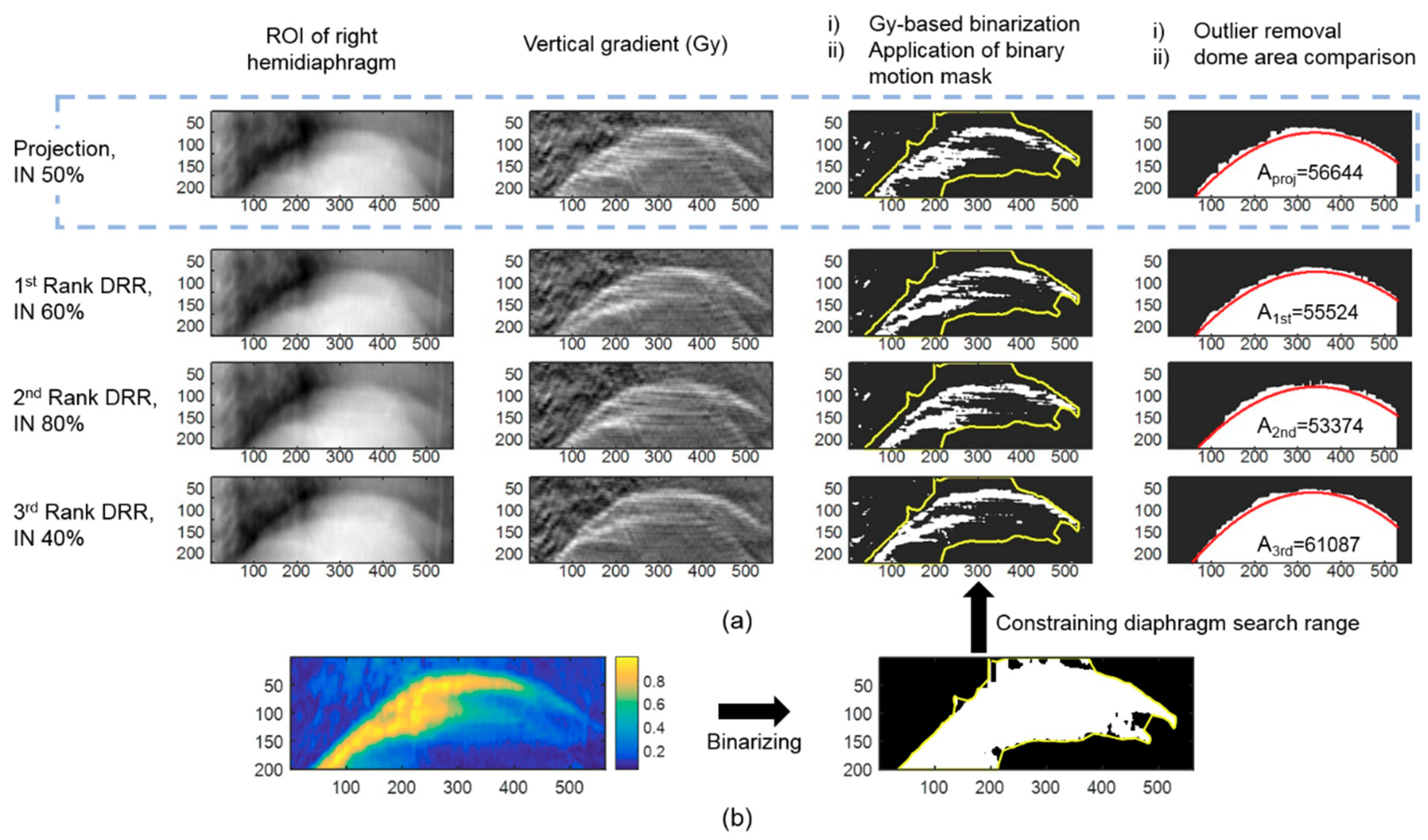
2.1.3. Comparing Hemidiaphragm Area Ranking
2.2. Respiration-Correlated Tracking of Tumor Motion
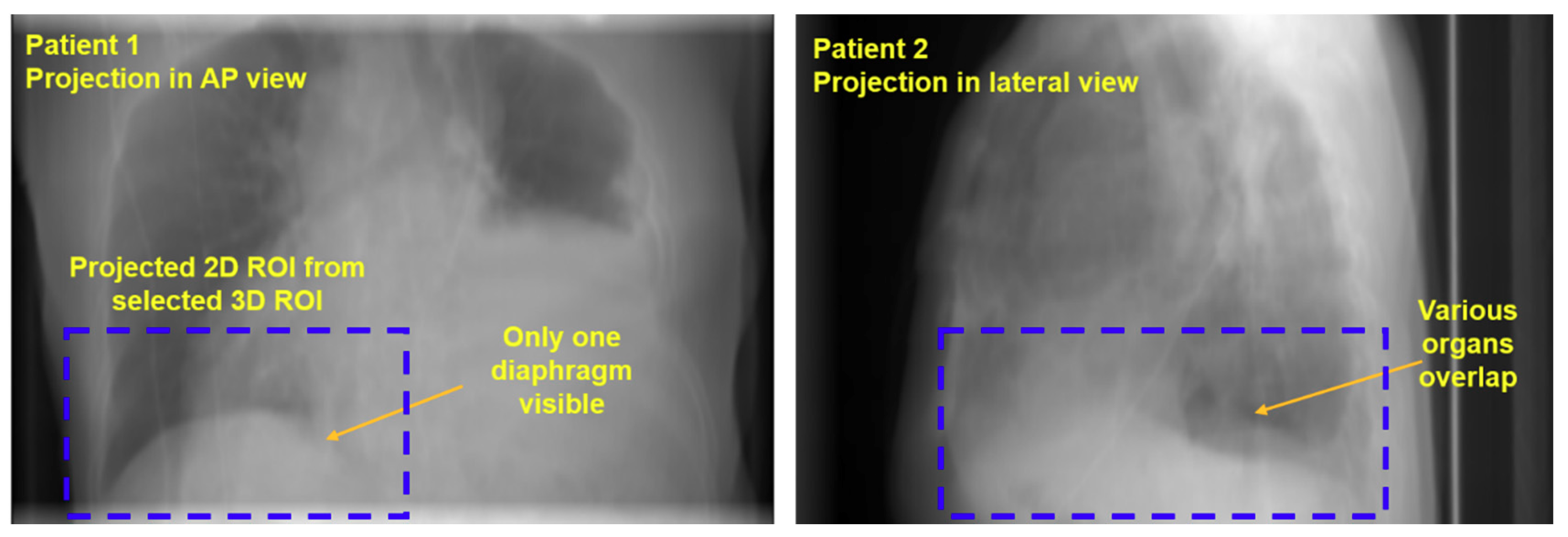
2.3. Patient Data Preparation
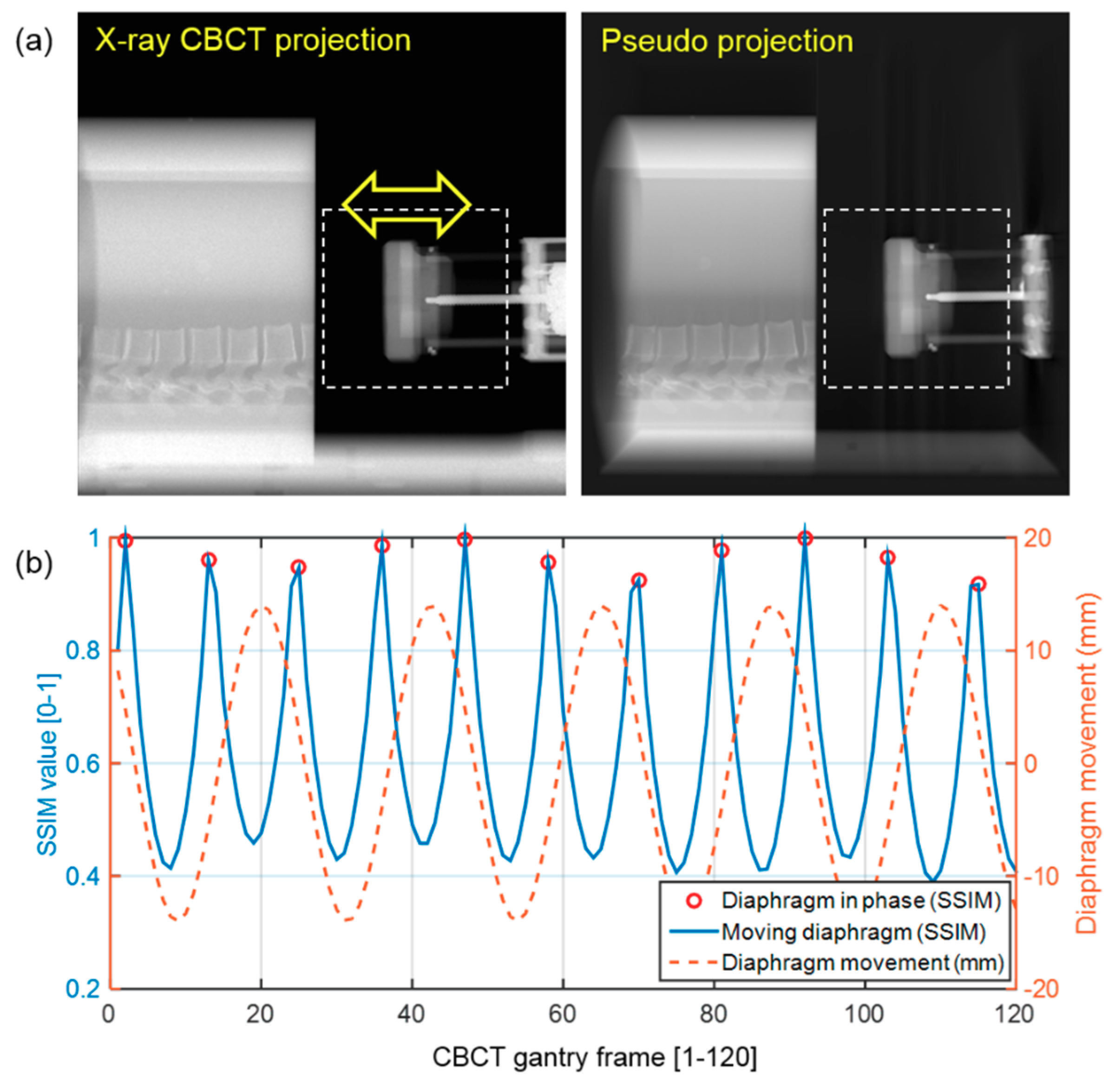
2.4. Evaluation of the Surrogacy of a Pseudo Projection
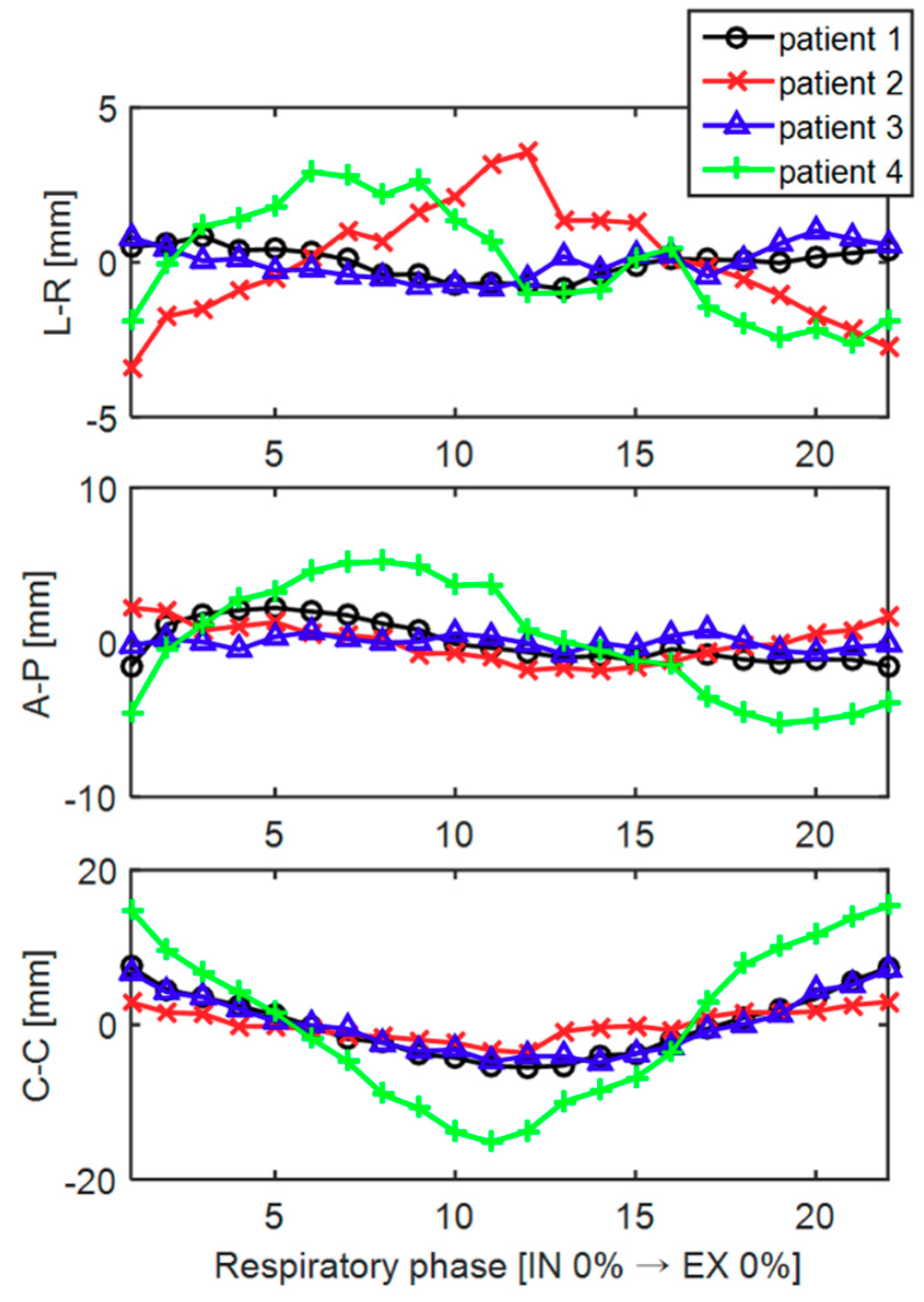
3. Results
3.1. Respiratory Phase Tracking Accuracy
3.2. Tumor Tracking Accuracy
3.3. Phantom Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
References
- Ling, C.C.; Yorke, E.; Amols, H.; Mechalakos, J.; Erdi, Y.; Leibel, S.; Rosenzweig, K.; Jackson, A. High-tech will improve radiotherapy of NSCLC: A hypothesis waiting to be validated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Engelsman, M.; Damen, E.M.; De Jaeger, K.; van Ingen, K.M.; Mijnheer, B.J. The effect of breathing and set-up errors on the cumulative dose to a lung tumor. Radiother. Oncol. 2001, 60, 95–105. [Google Scholar] [CrossRef]
- Pan, T.; Lee, T.Y.; Rietzel, E.; Chen, G.T. 4D-CT imaging of a volume influenced by respiratory motion on multi-slice CT. Med. Phys. 2004, 31, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Lewis, J.H.; Cervino, L.I.; Jiang, S.B. 4D CT sorting based on patient internal anatomy. Phys. Med. Biol. 2009, 54, 4821–4833. [Google Scholar] [CrossRef] [PubMed]
- Furtado, H.; Steiner, E.; Stock, M.; Georg, D.; Birkfellner, W. Real-time 2D/3D registration using kV-MV image pairs for tumor motion tracking in image guided radiotherapy. Acta. Oncol. 2013, 52, 1464–1471. [Google Scholar] [CrossRef] [PubMed]
- Gendrin, C.; Furtado, H.; Weber, C.; Bloch, C.; Figl, M.; Pawiro, S.A.; Bergmann, H.; Stock, M.; Fichtinger, G.; Georg, D.; et al. Monitoring tumor motion by real time 2D/3D registration during radiotherapy. Radiother. Oncol. 2012, 102, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Negoro, Y.; Nagata, Y.; Aoki, T.; Mizowaki, T.; Araki, N.; Takayama, K.; Kokubo, M.; Yano, S.; Koga, S.; Sasai, K.; et al. The effectiveness of an immobilization device in conformal radiotherapy for lung tumor: Reduction of respiratory tumor movement and evaluation of the daily setup accuracy. Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 889–898. [Google Scholar] [CrossRef]
- Simon, L.; Giraud, P.; Servois, V.; Rosenwald, J.C. Comparative study and clinical implementation of two breathing-adapted radiotherapy techniques: Dosimetric benefits for lung cancer treatment. Cancer Radiother. 2006, 10, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Rietzel, E.; Pan, T.; Chen, G.T. Four-dimensional computed tomography: Image formation and clinical protocol. Med. Phys. 2005, 32, 874–889. [Google Scholar] [CrossRef] [PubMed]
- Li, X.A.; Stepaniak, C.; Gore, E. Technical and dosimetric aspects of respiratory gating using a pressure-sensor motion monitoring system. Med. Phys. 2006, 33, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Onishi, H.; Kawakami, H.; Marino, K.; Komiyama, T.; Kuriyama, K.; Araya, M.; Saito, R.; Aoki, S.; Araki, T. A simple respiratory indicator for irradiation during voluntary breath holding: A one-touch device without electronic materials. Radiology 2010, 255, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.; McClelland, J.; Tarte, S.; Lawrence, D.; Ahmad, S.; Hawkes, D.; Landau, D. Assessment of two novel ventilatory surrogates for use in the delivery of gated/tracked radiotherapy for non-small cell lung cancer. Radiother. Oncol. 2009, 91, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.; Suh, Y.; Dieterich, S.; Keall, P.J. A monoscopic method for real-time tumour tracking using combined occasional x-ray imaging and continuous respiratory monitoring. Phys. Med. Biol. 2008, 53, 2837–2855. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Walston, S.; Welliver, M.X.; Chakravarti, A.; Quick, A.M. Improving intra-fractional target position accuracy using a 3D surface surrogate for left breast irradiation using the respiratory-gated deep-inspiration breath-hold technique. PLoS ONE 2014, 9, e97933. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Yang, Y.; Li, T.; Li, X.; Heron, D.E.; Huq, M.S. Statistical analysis of target motion in gated lung stereotactic body radiation therapy. Phys. Med. Biol. 2011, 56, 1385–1395. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.M. Investigating the robustness of the Anzai respiratory gating system. Master’s Thesis, University of Canterbury, Christchuch, Canterbury, New Zealand, April 2013. [Google Scholar]
- Shirato, H.; Harada, T.; Harabayashi, T.; Hida, K.; Endo, H.; Kitamura, K.; Onimaru, R.; Yamazaki, K.; Kurauchi, N.; Shimizu, T.; et al. Feasibility of insertion/implantation of 2.0-mm-diameter gold internal fiducial markers for precise setup and real-time tumor tracking in radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2003, 56, 240–247. [Google Scholar] [CrossRef]
- Shirato, H.; Oita, M.; Fujita, K.; Watanabe, Y.; Miyasaka, K. Feasibility of synchronization of real-time tumor-tracking radiotherapy and intensity-modulated radiotherapy from viewpoint of excessive dose from fluoroscopy. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.P.; Kupelian, P.A.; Willoughby, T.R.; Langen, K.M.; Meeks, S.L. An evaluation of intrafraction motion of the prostate in the prone and supine positions using electromagnetic tracking. Radiother. Oncol. 2011, 99, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Langen, K.M.; Willoughby, T.R.; Meeks, S.L.; Santhanam, A.; Cunningham, A.; Levine, L.; Kupelian, P.A. Observations on real-time prostate gland motion using electromagnetic tracking. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Shirato, H.; Shimizu, S.; Kitamura, K.; Nishioka, T.; Kagei, K.; Hashimoto, S.; Aoyama, H.; Kunieda, T.; Shinohara, N.; Dosaka-Akita, H.; et al. Four-dimensional treatment planning and fluoroscopic real-time tumor tracking radiotherapy for moving tumor. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 435–442. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Chang, Y.; Liu, H.Y.; Liu, G.; Li, Q. Target margin design for real-time lung tumor tracking stereotactic body radiation therapy using CyberKnife Xsight Lung Tracking System. Sci. Rep. 2017, 7, 10826. [Google Scholar] [CrossRef] [PubMed]
- Ipsen, S.; Bruder, R.; O’Brien, R.; Keall, P.J.; Schweikard, A.; Poulsen, P.R. Online 4D ultrasound guidance for real-time motion compensation by MLC tracking. Med. Phys. 2016, 43, 5695. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Dy, J.G.; Sharp, G.C.; Alexander, B.; Jiang, S.B. Multiple template-based fluoroscopic tracking of lung tumor mass without implanted fiducial markers. Phys. Med. Biol. 2007, 52, 6229–6242. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Cervino, L.I.; Tang, X.; Vasconcelos, N.; Jiang, S.B. Fluoroscopic tumor tracking for image-guided lung cancer radiotherapy. Phys. Med. Biol. 2009, 54, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Hamilton, R.J.; Schowengerdt, R.A.; Alexander, B.; Jiang, S.B. Lung tumor tracking in fluoroscopic video based on optical flow. Med. Phys. 2008, 35, 5351–5359. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Hamilton, R.J.; Schowengerdt, R.A.; Jiang, S.B. A deformable lung tumor tracking method in fluoroscopic video using active shape models: A feasibility study. Phys. Med. Biol. 2007, 52, 5277–5293. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.H.; Li, R.; Watkins, W.T.; Lawson, J.D.; Segars, W.P.; Cervino, L.I.; Song, W.Y.; Jiang, S.B. Markerless lung tumor tracking and trajectory reconstruction using rotational cone-beam projections: A feasibility study. Phys. Med. Biol. 2010, 55, 2505–2522. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Bovik, A.C.; Sheikh, H.R.; Simoncelli, E.P. Image quality assessment: From error visibility to structural similarity. IEEE Trans. Image Process. 2004, 13, 600–612. [Google Scholar] [CrossRef] [PubMed]
- Otsu, N. A Threshold Selection Method from Gray-Level Histograms. IEEE Trans. Syst. Man Cybern. 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Chen, Q.S.; Weinhous, M.S.; Deibel, F.C.; Ciezki, J.P.; Macklis, R.M. Fluoroscopic study of tumor motion due to breathing: Facilitating precise radiation therapy for lung cancer patients. Med. Phys. 2001, 28, 1850–1856. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Hamilton, R.J. A novel respiratory detection method based on automated analysis of ultrasound diaphragm video. Med. Phys. 2006, 33, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Besl, P.J.; McKay, N.D. A method for registration of 3-D shapes. IEEE Trans. Pattern Anal. 1992, 14, 239–256. [Google Scholar] [CrossRef]
- Friedman, J.H.; Bentley, J.L.; Finkel, R.A. An algorithm for finding best matches in logarithmic expected time. ACM Trans. Math. Softw. 1977, 3, 209–226. [Google Scholar] [CrossRef]
- Maier, A.; Hofmann, H.G.; Schwemmer, C.; Hornegger, J.; Keil, A.; Fahrig, R. Fast simulation of x-ray projections of spline-based surfaces using an append buffer. Phys. Med. Biol. 2012, 57, 6193–6210. [Google Scholar] [CrossRef] [PubMed]
- Rote, G. Computing the minimum Hausdorff distance between two point sets on a line under translation. Inf. Process. Lett. 1991, 38, 123–127. [Google Scholar] [CrossRef]
- Bral, S.; Van Parijs, H.; Soete, G.; Linthout, N.; Van Moorter, L.; Verellen, D.; Storme, G. A feasibility study of image-guided hypofractionated conformal arc therapy for inoperable patients with localized non-small cell lung cancer. Radiother. Oncol. 2007, 84, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Stock, M.; Kontrisova, K.; Dieckmann, K.; Bogner, J.; Poetter, R.; Georg, D. Development and application of a real-time monitoring and feedback system for deep inspiration breath hold based on external marker tracking. Med. Phys. 2006, 33, 2868–2877. [Google Scholar] [CrossRef] [PubMed]
- Parikh, P.; Hubenschmidt, J.; Dimmer, S.; Vertatschitsch, E.; Eidens, R.; Wright, J.N.; Low, D.A. 4D verification of real-time accuracy of the Calypso system with lung cancer patient trajectory data. Int. J. Radiat. Oncol. 2005, 63, S26–S27. [Google Scholar] [CrossRef]
- Burch, D.; Willoughby, T.; Meeks, S.; Kupelian, P.; Litzenberg, D.; Sandler, H.; Levine, L.; Vertatschitsch, D.; Vertatschitsch, E. Real time prostate translation, rotation, deformation evaluated with Calypso beacon (TM) transponders. Int. J. Radiat. Oncol. 2005, 63, S195. [Google Scholar] [CrossRef]
- Geraghty, P.R.; Kee, S.T.; McFarlane, G.; Razavi, M.K.; Sze, D.Y.; Dake, M.D. CT-guided transthoracic needle aspiration biopsy of pulmonary nodules: Needle size and pneumothorax rate. Radiology 2003, 229, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Whyte, R.I.; Crownover, R.; Murphy, M.J.; Martin, D.P.; Rice, T.W.; DeCamp, M.M., Jr.; Rodebaugh, R.; Weinhous, M.S.; Le, Q.T. Stereotactic radiosurgery for lung tumors: Preliminary report of a phase I trial. Ann. Thorac. Surg. 2003, 75, 1097–1101. [Google Scholar] [CrossRef]
- Arslan, S.; Yilmaz, A.; Bayramgurler, B.; Uzman, O.; Nver, E.; Akkaya, E. CT- guided transthoracic fine needle aspiration of pulmonary lesions: Accuracy and complications in 294 patients. Med. Sci. Monit. 2002, 8, CR493–CR497. [Google Scholar] [PubMed]
- Seppenwoolde, Y.; Shirato, H.; Kitamura, K.; Shimizu, S.; van Herk, M.; Lebesque, J.V.; Miyasaka, K. Precise and real-time measurement of 3D tumor motion in lung due to breathing and heartbeat, measured during radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 822–834. [Google Scholar] [CrossRef]
- Ruan, D.; Fessler, J.A.; Balter, J.M.; Berbeco, R.I.; Nishioka, S.; Shirato, H. Inference of hysteretic respiratory tumor motion from external surrogates: A state augmentation approach. Phys. Med. Biol. 2008, 53, 2923–2936. [Google Scholar] [CrossRef] [PubMed]
- Suh, Y.; Dieterich, S.; Cho, B.; Keall, P.J. An analysis of thoracic and abdominal tumour motion for stereotactic body radiotherapy patients. Phys. Med. Biol. 2008, 53, 3623–3640. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.M.; Ten Haken, R.K.; Schipper, M.J.; Sullivan, M.A.; Chen, M.; Lopez, C.; Kalemkerian, G.P.; Hayman, J.A. High-dose radiation improved local tumor control and overall survival in patients with inoperable/unresectable non-small-cell lung cancer: Long-term results of a radiation dose escalation study. Int. J. Radiat. Oncol. Biol. Phys. 2005, 63, 324–333. [Google Scholar] [CrossRef] [PubMed]

| Diaphragm-Elevation Based Method | Proposed Method | |||||
|---|---|---|---|---|---|---|
| Inhale/Exhale Identification * (%) | Phase Deviation † (#) | Accuracy (%) | Inhale/Exhale Identification * (%) | Phase Deviation † (#) | Accuracy (%) | |
| Patient 1 | 68.8 | 4 | 60.4 | 100 | 1 | 97.9 |
| Patient 2 | 41.7 | 24 | 52.1 | 100 | 2 | 95.8 |
| Patient 3 | 39.6 | 18 | 47.9 | 100 | 0 | 100 |
| Patient 4 | 50 | 9 | 41.7 | 100 | 0 | 100 |
| Patient 5 | 41.7 | 24 | 20.8 | 100 | 2 | 95.8 |
| Patient 6 | 47.9 | 49 | 18.8 | 95.8 | 3 | 93.8 |
| Total | 48.3 | 21.3 | 40.3 ± 17.0 | 99.3 | 1.3 | 97.2 ± 2.5 |
| Amplitude (mm) | SD (mm) | Tumor Tracking Error in 3D (mm) | ||||||
|---|---|---|---|---|---|---|---|---|
| L–R * | A–P * | C–C * | L–R | A–P | C–C | Surface Distance | Center of Mass Distance | |
| Patient 1 | 1.7 | 3.8 | 13.0 | 0.5 | 1.3 | 4.1 | 0.9 (± 0.4) | 0.6 (± 0.5) |
| Patient 2 | 7.0 | 4.1 | 6.6 | 1.8 | 1.2 | 1.9 | 0.7 (± 0.3) | 0.7 (± 0.4) |
| Patient 3 | 1.9 | 1.5 | 12.0 | 0.5 | 0.4 | 3.8 | 0.8 (± 0.4) | 0.8 (± 0.2) |
| Patient 4 | 5.5 | 10.4 | 30.5 | 1.8 | 3.8 | 10.2 | 1.0 (± 0.5) | 1.0 (± 0.4) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.-H.; Lee, S. Real-Time Tumor Motion Tracking in 3D Using Planning 4D CT Images during Image-Guided Radiation Therapy. Algorithms 2018, 11, 155. https://doi.org/10.3390/a11100155
Choi J-H, Lee S. Real-Time Tumor Motion Tracking in 3D Using Planning 4D CT Images during Image-Guided Radiation Therapy. Algorithms. 2018; 11(10):155. https://doi.org/10.3390/a11100155
Chicago/Turabian StyleChoi, Jang-Hwan, and Sooyeul Lee. 2018. "Real-Time Tumor Motion Tracking in 3D Using Planning 4D CT Images during Image-Guided Radiation Therapy" Algorithms 11, no. 10: 155. https://doi.org/10.3390/a11100155
APA StyleChoi, J.-H., & Lee, S. (2018). Real-Time Tumor Motion Tracking in 3D Using Planning 4D CT Images during Image-Guided Radiation Therapy. Algorithms, 11(10), 155. https://doi.org/10.3390/a11100155






