3.1. Characteristics of PS-Ashes
Chemical compositions of PS-ashes are represented in
Table 1. OTo3 has a usual composition. However, the others have unusual compositions. The high content of Cl in OTs2 would be attributed to refuse derived fuel (RDF), which contains much chlorine and moisture, while the high content of MgO in N45 to recycled plastic fuel (RPF), which contains much talc, commonly used as a filler in plastic appliances. Both RDF and RPF are used widely in Japan as boiler fuels, together with oils.
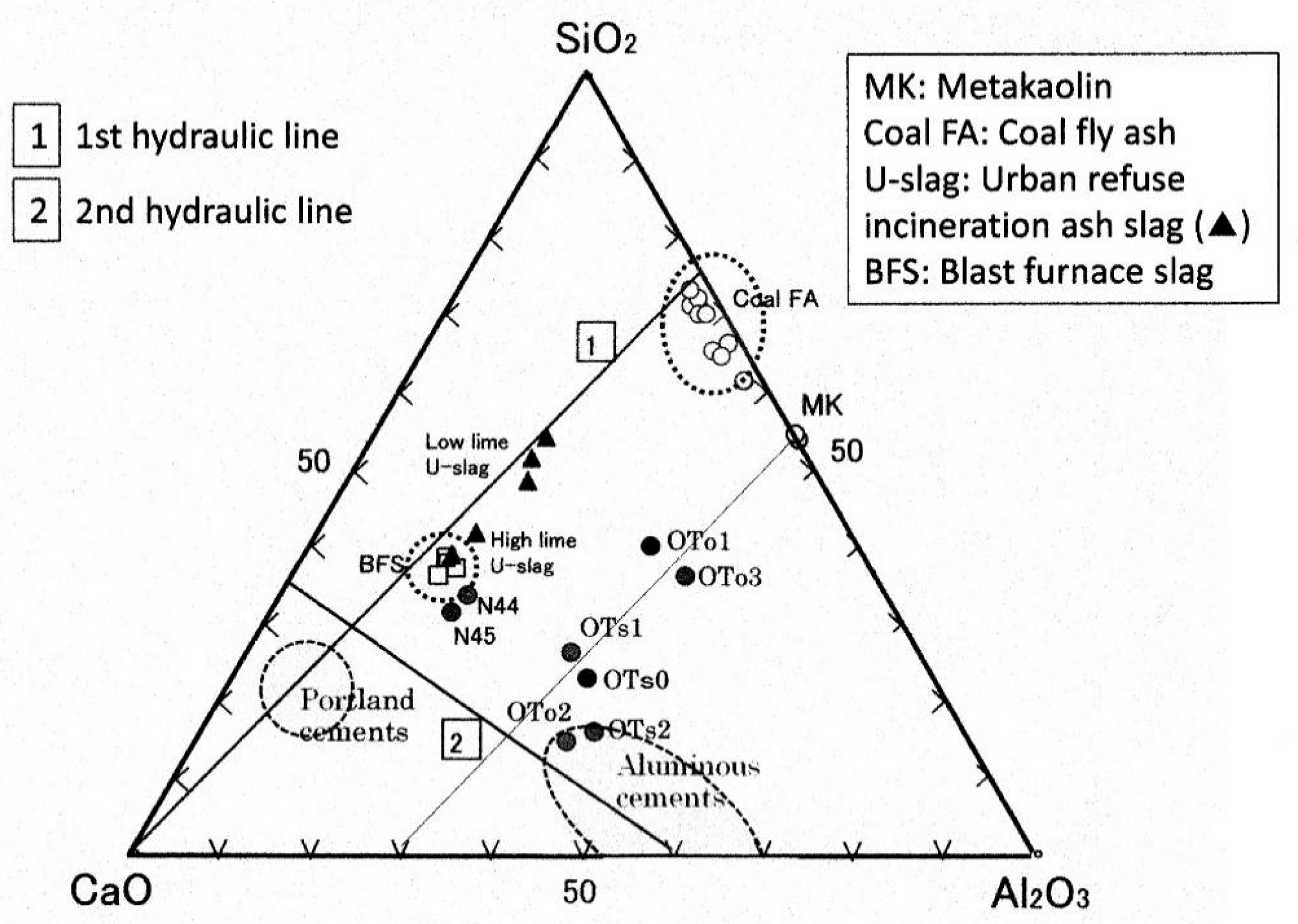
To clearly grasp the features of the PS-ashes, the chemical compositions are plotted on the ternary diagram, as shown in
Figure 1, in terms of CaO-Al
2O
3-SiO
2 mass ratio. The plots encompass a wide range of PS-ashes. OTo/OTs series are plotted alongside with a trend line parallel to the first hydraulic line running through to the metakaolin point. On the other hand, N-series showed different features, plotted in the vicinity of blast furnace slag (BFS), a member of the first hydraulic line. The former group (OTo/OTs series) can be called high-alumina PS-ash, while the latter group (N-series) can be called low-alumina PS-ash. From another point of view, the PS-ashes can be classified into two groups—Ca-rich and Ca-poor. OTo1 and OTo3 are in the latter group, but the others are in the former group. Readers are kindly requested to refer to our past publication for relevant PS-ash compositions [
31].
SEM images of the PS-ash are represented in
Figure 2. Sponge-like porous textures peculiar to gel materials [
33] can be seen for all the samples—most clearly for OTo3. Moreover, ettringite prevailed in OTo2 and N45 in acicular to prismatic forms. Particularly in OTs2, some ettringite crystals developed in stout prismatic form.
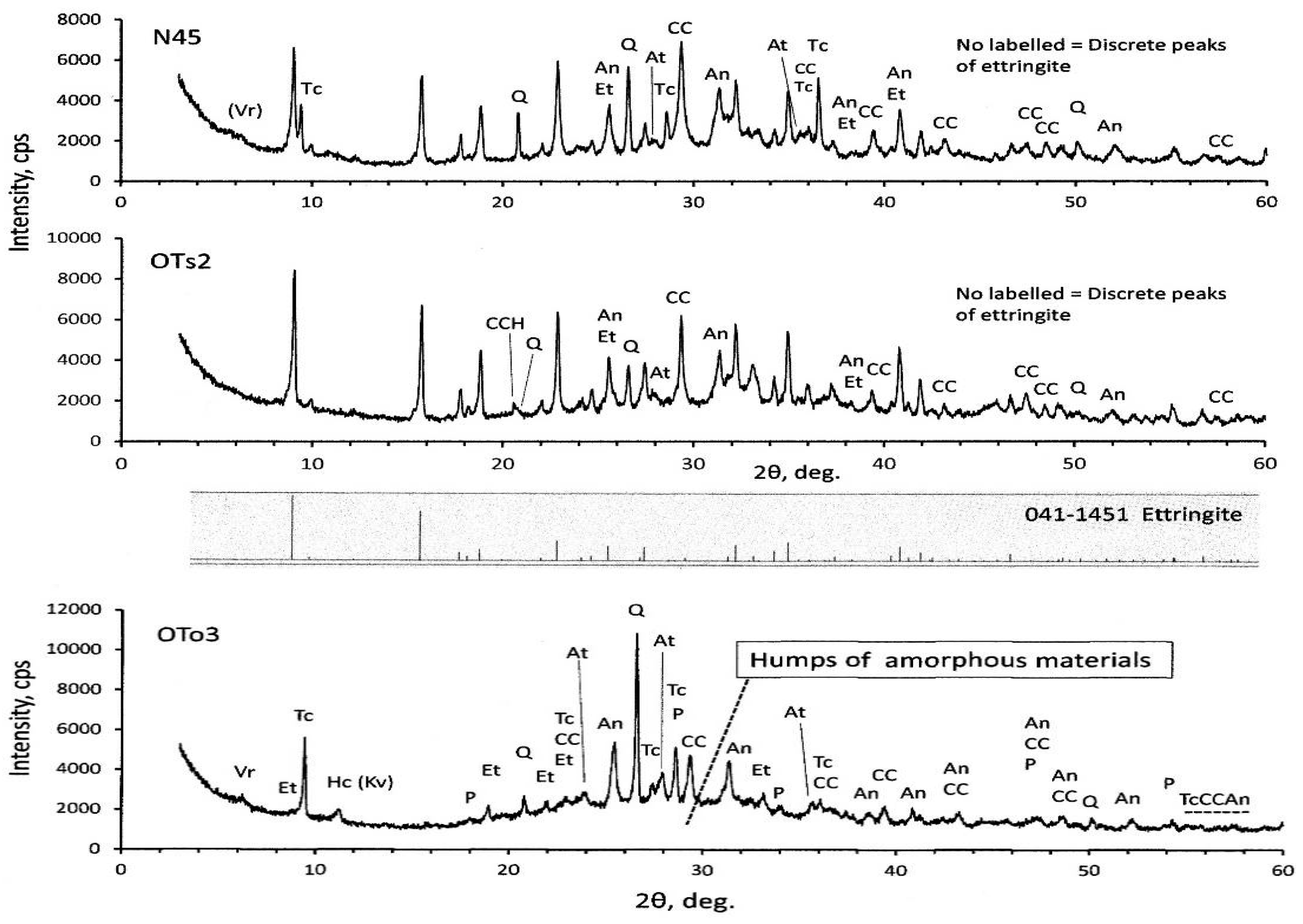
XRD diagrams of the PS-ash are represented in
Figure 3. Calcite, quartz, and anhydrite were identified, irrespective of the PS-ash. In addition, humps are recognized around 2θ = 20°–40°. Particularly, the humps are very clear in OTo3, suggesting the presence of amorphous materials. OTo3 is characterized by less presence of ettringite, while the others (OTs2 and N45) show the pronounced presence of ettringite due to high SO
3 contents, in addition to high LOI and moisture contents, as seen in
Table 1.
It is important to classify the identified minerals on the basis of primary and secondary origins, as classified in
Table 3. In OTo3, relict portlandite was detected, indicating that primarily quick lime (CaO) formed in the boiler. Water is generally sprinkled over the ash to prevent the ash from scattering after incineration, or sometimes the ash is pelletized. Accordingly, the quick lime slakes into portlandite and finally turns to calcite, in some cases to vaterite [
31], due to reaction with CO
2 in air. The ashes derived from plants and woods generally comprise K
2O, CaO, and SiO
2 as main components, in addition to MgO, P
2O
5, SO
3, and Na
2O as subordinate components [
34]. Quartz may generate during the incineration in boilers. Anhydrite is a product of desulfurization. Generally, fluidized bed-type boilers are fed by limestone together with fuel coal for desulfurization, but for paper sludge incinerations, limestone is not required, since paper sludge contains much CaO. Moreover, calcite is usually used as filler and coating materials for ordinary grade paper. Contrary, kaolin or kaolin/calcite mixtures are used for high grade paper. Waste papers are so often fed to boilers as fuel, which also act as moisture absorber of wet paper sludge, so the origin of the Al
2O
3 component would be the kaolin. Strictly speaking, the presence of talc is because talc is so widely used as filler materials for paper and plastic appliances, so it remains in the ash of paper sludge, waste paper, and RPF combustions. A small amount of anorthite was identified, of which formation was also reported in literature for the ash from a pressurized fluidized bed-type boiler [
16]. Vermiculite and hydrocalumite are secondary minerals formed during the water sprinkling. The presence of kovdorskite is also suspected other than hydrocalumite, judging from the 2θ positions. OTs2 and N45 show nearly the same XRD patterns—in which ettringite is prevailed—but talc is lacking in OTs2. It should be noted that hydrocalcite was identified in OTs2, of which the main peak is slightly different from the quartz peak, as indicated in
Figure 3. Anorthite is also identified in minor quantities in both OTs2 and N45. Forsterite might be a primary origin, as discussed later in
Section 3.3.
It should be stressed that raw PS-ashes are not completely amorphous, but abundant in crystalline phases—quite different from conventional geopolymer fillers such as metakaolin, water quenched blast furnace slag, coal fly ash, and so on.
3.2. Physical Properties of PS-Ash-Based Geopolymer
As shown in
Table 4, for the #0 and #1 liquors, adequate values of L/F of PS-ash based geopolymer were from 1 to 1.5, depending on filler characters, particularly on specific surface areas and porous textures. Incidentally, a standard L/F is 0.4 for ordinary geopolymers in pastes [
35]. OTs2 showed extremely slow setting with L/F 1.0, taking overnight, presumably due to the presence of so much chlorine, in addition to low specific surface area, so the specimen preparation for OTs2 was skipped in the present experiment. Other PS-ash-based geopolymer mixtures set within 40 min. Retarding effects of chlorine are recently confirmed by the experiment of using NaCl (Ichimiya, K., 2016, personal communication).
It should be noted that when the #0 liquor was applied, the OTo3-based geopolymer mixtures swelled. Eventually, many pores of foaming origin were observed in the hardened specimens (
Figure 4), resulting in smaller bulk densities, as indicated in
Table 4. Presumably, free Al metal is included in this kind of PS-ash. The origin of the Al metal would be Al-laminated appliances (such as food containers), which might slip into the RDF used in the boiler plant of OTo3. However, at this moment, the Al metal could not be observed, even under the high power of scanning electron microscopy and energy dispersive spectroscopy (SEM-EDS), which is probably disseminated at the nanometer scale in the ash. Hydrogen gas is generated due to the high pH circumstances of the #0 liquor. To the contrary, no swelling was observed for the #1 liquor without mixing the caustic soda solution. Incidentally, this type of swelling is also reported for an RDF-ash [
36] and hydrogen generation is confirmed by gas chromatography (Goda, H., 2012, oral communication).
Flexural strength of this lightweight material was relatively high, compared to the other non-swelled materials. It is postulated that nitride ions suppress the development of strength to a certain degree, due to the pH-lowering action of alkaline liquors [
31,
35]. However, in this study, this phenomenon was restricted to the specimens mixed with the #1 liquor.
3.3. XRD Results of Hardened Geopolymer
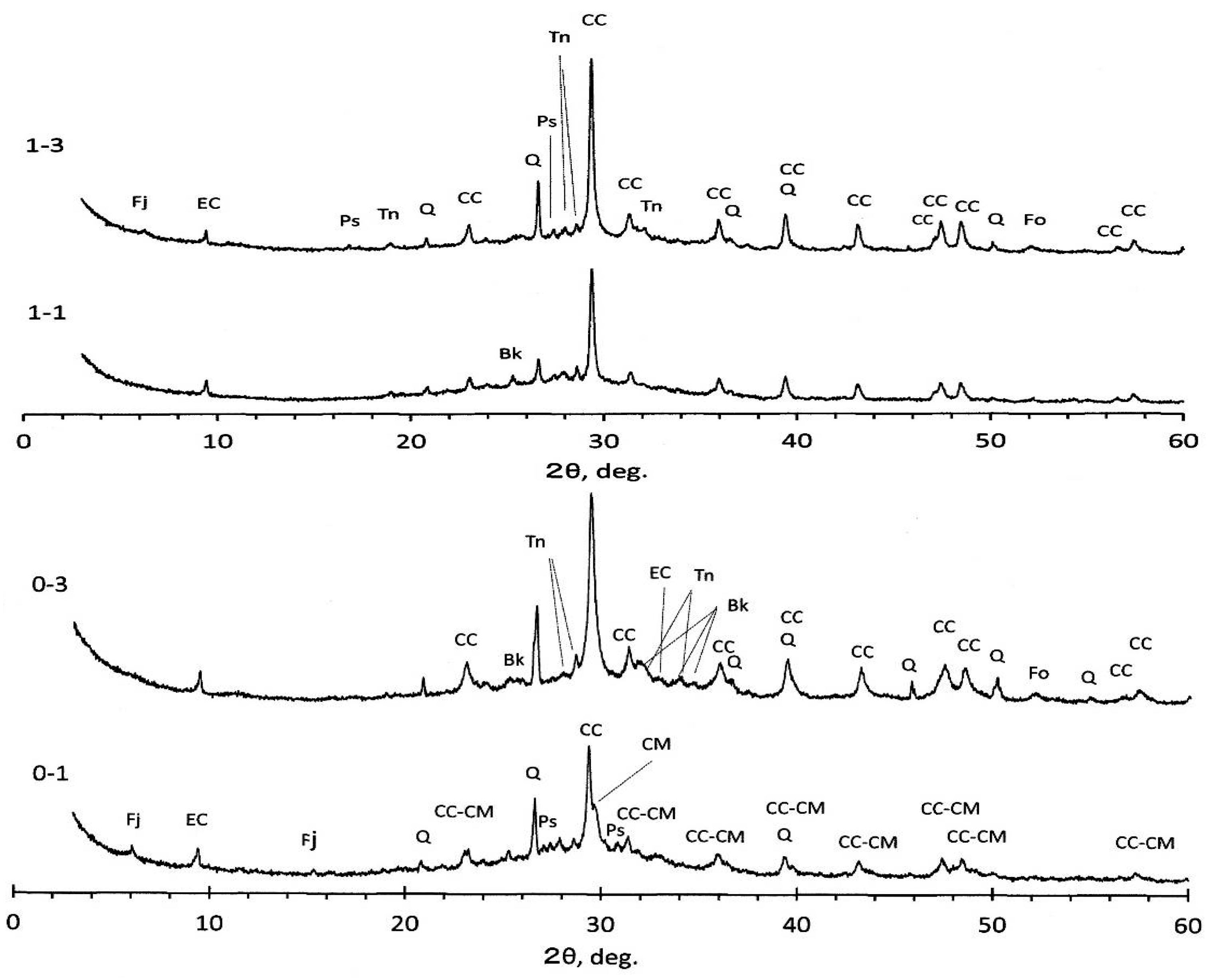
For the plain specimens, XRD diagrams are represented in
Figure 5 for the #0 and #1 liquors, respectively. Quartz and calcite remained more or less intact, irrespective of sample species. However, judging from the variation of peak heights, some part of these minerals were reacted and diminished in highly alkaline circumstances, even for quartz. Quartz decrease was also observed in the literature [
5]. Ettringite and anhydrite were not detected any more, and neither were anorthite and vermuculite. Instead, the formation of carbonate ettringite was observed, irrespective of specimen species. It is noted that magnesian calcite, (Ca, Mg)CO
3, was identified for 0-1 specimen, of which peaks are located just a little bit higher than the 2θ angles of calcite, CaCO
3, as shoulders. Peak patterns of magnesian calcite are quite different from dolomite, Ca, Mg(CO
3)
2. Formation of faujasite, a member of zeolite family, was observed for 0-1 and 1-3 specimens. Other identified minerals are pirssonite, burkeite, and thenerdite, depending on specimens—all formed accompanied with the geopolymerization of mixed alkaline liquors.
It is more plausible that the forsterite identified for specimens 0-3 and 1-3 might be a primary origin formed in the boiler. This mineral peaks might be concealed with anhydrite peaks in raw PS-ashes, and appeared intact according to the disappearance of anhydrite. Other than the crystalline phases mentioned above, there are amorphous phases more or less depending on specimens as so far postulated, judging from some humps around 2θ = 30°. For SC-GP specimens, no marked differences from plain specimens were observed, probably due to lower-level dosages of the surrogates. However, according to literature using metakaolin filler under higher level presence of nitrates, the formation of sodalite and cancrinite was reported as nitrate-bearing phases in addition to zeolite A and zeolite X, depending on NaOH concentrations [
37].
Talking again about the faujasite, naturally-occurring faujasite generally has the chemical formula (Na
2, Ca, Mg)
3.5(Al
7Si
17O
48)·32H
2O, in which the S/N (SiO
2/Na
2O) molar ratio is 4.86. Incidentally, according to literature, naturally occurring faujasite has an S/N of around 4.5 [
38]. It is well-known that synthetic faujasite is classified by S/N into zeolite X (lower than 3.0) and zeolite Y (higher than 3.0). XRD patterns represented in
Figure 5 show that faujasite patterns are close to Na
2Al
2Si
2.4O
8.8·6.7H
2O, where the S/N is 2.4, so the formation of zeolite X is more plausible, suggesting that the incorporation of Na-Al combination is large in the faujasite structure.
3.4. Reaction Process of PS-Ash-Based Geopolymers
Resultant minerals are summarized in
Table 5, classified into three categories. The first is the intrinsic minerals of primary origin formed in boilers. The second is the extrinsic minerals of secondary origin formed due to the water sprinkling. The third is relevant to the geopolymerization of alkaline liquors with the minerals included in the PS-ashes.
Referring to the hardening process of PS-ashes, the primary and the secondary minerals react with the alkaline liquors so that the recombination takes place, and minerals peculiar to the geopolymerization form. Thereby, forsterite may remain intact, while quartz and calcite may remain partially intact. Other minerals were completely consumed to yield a group of minerals called GP-minerals in
Table 5, which are carbonate ettringite, faujasite, pirssonite, burkeite, and thenerdite, and sometimes magnesian calcite, other than amorphous gels which are truly matrices. The species of GP-minerals are dependent on the PS-ashes and alkaline liquors used. It is noteworthy to refer to the amorphous phases peculiar to geopolymers, which are currently believed to consist of two distinct phases—so called C-A-S-H gels and N-A-S-H gels—where C, A, S, H, and N denote CaO, Al
2O
3, SiO
2, H
2O, and Na
2O, respectively. It is considered that in Ca-rich geopolymer systems, C-A-S-H is stable, while N-A-S-H is unstable, due to the occurrence of an ion exchange of Ca
2+ for Na
+. Accordingly, N-A-S-H turns gradually to C-A-S-H and eventually diminishes [
39,
40,
41,
42]. Strictly speaking, initial C-A-S-H and N-A-S-H may finally become (Ca, N)-A-S-H gels after the ion exchange.
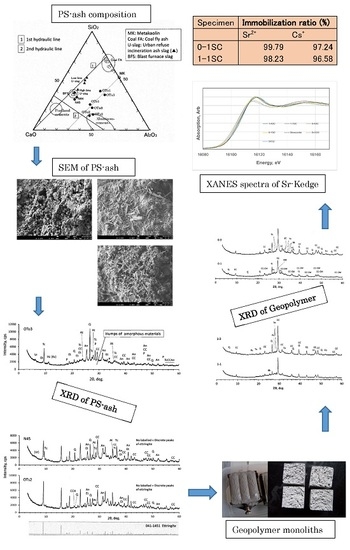
3.5. Immobiization Ratios of Sr2+ and Cs+
Results of the 4 + 2 week age samples are tabulated in
Table 6, together with relevant data for calculations. High immobilization ratios of Sr
2+ and Cs
+ were reached for 0-1SC and 1-1SC, both using OTo3 as filler, whereas Cs
+ immobilization was relatively low for 0-3SC and 1-3SC, both using N45 as filler. It should be noted that specimen 1-3SC showed over-scale, indicating poor immobilization of Sr
2+, so more detailed measurements of ICP were skipped for this specimen. Cs
+ will be mentioned later.
Regarding the immobilization mechanisms of Sr
2+ and Cs
+, two possibilities are considered. One is that these ions are incorporated into faujasite and the other is into geopolymer gels (GP-gels). Faujasite would accommodate both Sr
2+ and Cs
+, since it consists of sodalite cages with openings of 0.74 nm in diameter. Incidentally, according to Shannon and Prewitt [
43], ionic radii of Sr
2+ and Cs
+ are 0.118–0.144 nm and 0.167–0.188 nm, respectively, depending on coordination numbers from 6-hold to 12-hold. Gobbinsite, (Na,Ca
0.5)
6(Al
6Si
10)O
32·12H
2O—a zeolite family mineral—was also identified as a candidate mineral in a previous study [
31]. However, judging from XRD peak intensities, the formation of faujasite is in very small quantities, and the formation of GP-gels is prevalent due to the polycondensation of applied alkaline liquors.
It is postulated that there are two kinds of GP-gels—C-A-S-H and N-A-S-H [
39,
40,
41,
42]. N-A-S-H is considered to be unstable under the co-presence of C-A-S-H, due to the ion exchange of Ca
2+ for Na
+. Sr
2+ may be preferentially incorporated into C-A-S-H. We considered at the moment that the high MgO content of specimens 1-3SC (
Table 1) may interfere the stable formation of C-A-S-H in young ages, so 1-3SC showed over-scale for Sr
2+. Incidentally, the stability and instability of GP-gels are also called “a complex multistep reaction process”, leading to a phase-mixed product in the case of N-A-S-H [
44]. That is, a non-equilibrium-to-equilibrium transition of GP-gels. However, little has been studied in the case of C-A-S-H.
For Cs
+ incorporation, N-A-S-H and faujasite may play an important role. However, it is predictive that N-A-S-H becomes unstable with elapsed time. Therefore, the immobilization ratio of Cs
+ may decrease with the ion exchange, resulting in some Cs
+ depletion. In the long run, however, with the formation of new C-A-S-H phase, (C, N)-A-S-H, Cs
+ may be again incorporated. In order to confirm this hypothesis, immobilization ratios of prolonged ages were measured up to 24 weeks, nearly 6 months. The results are shown in
Table 7.
For Sr2+, higher immobilization ratios were generally maintained, irrespective of material age. However, 1-3SC showed unstable immobilization ratios. Strangely, we found that it turned dramatically up to 91.73% at 12 week of age, but it turned down again at 24 weeks of age, suggesting unstable incorporation of Sr2+ in this mixture. As mentioned, the high MgO content may be a cause of this instability, and there also may be some transitional stages in the evolution of C-A-S-H gels with elapsed time.
For Cs+, decreasing immobilization ratios were found at 12 weeks of age, as expected, and this phenomenon is extremely remarkable for the #0 liquor. However, these ratios again rose up at 24 weeks of age, as expected.
As a whole, 1-1SC applying #1 liquor exhibited stable immobilization ratios, irrespective of material ages. To prevent the fluctuation of immobilization ratios, one of the measures would be high-temperature curing, which accelerates the polycondensation reactions to yield stable GP-gels in a short period of time. Incidentally, high immobilization ratios were also reported for Cs
+ incorporated into alkali-activated fly ash matrices cured at 85 °C and 120 °C, reaching 98.56%–99.78% immobilization ratios that were analyzed by toxic characteristic leaching procedure (TCLP), using the pH 2.88 leaching solution of glacial acetic acid [
35]. Retentions of 95.2% and 81.4% are reported, respectively, for Sr
2+ and Cs
+ in 0.01 M NaOH solution, using magnetic zeolite nanocomposites [
45].
Finally, we tentatively tried to convert the hardened SC-GP into slags to reduce volumes. Fusion temperature and duration were 1400 °C-1 h for 0-1SC and 1-1SC, and 1300 °C-1 h for 0-3SC and 1-3SC, respectively. Moistures of GP-liquor origin as well as evaporates represented as LOI in
Table 1 were taken into account in calculations. Results were tabulated at the right-bottom corner of
Table 6, showing high immobilization ratios for both Sr
2+ and Cs
+. Therefore, slag conversion would be one of the options for eternal burials as proposed in a previous study [
31].
3.6. XANES
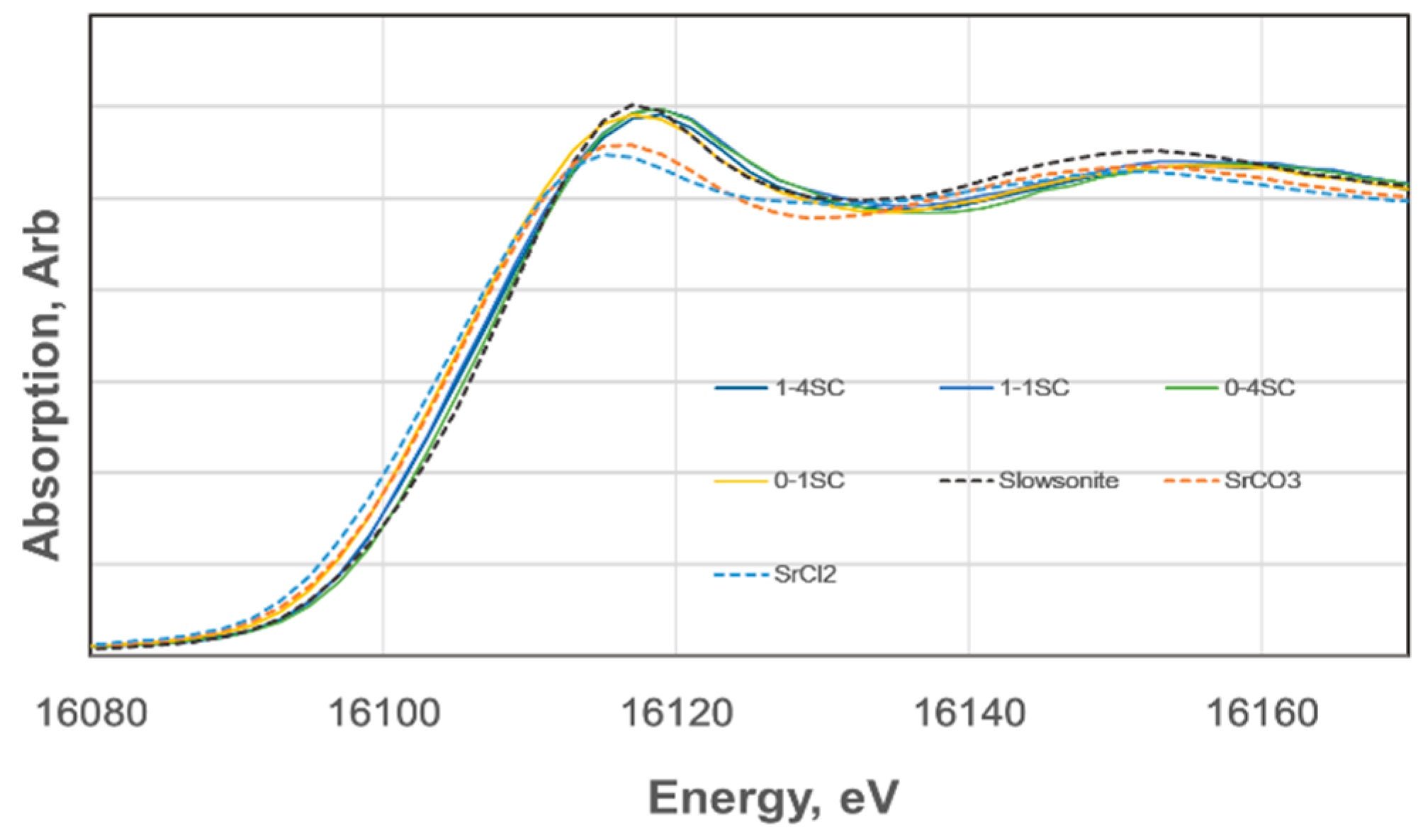
XANES spectra of the K absorption edge of strontium for SC-GP specimens of 4 + 2 week age were compared with the spectra of the standard samples of SrCl
2, SrCO
3, and slawsonite, (Sr, Ca)Al
2Si
2O
8). The former two are reagents, and slawsonite is a naturally-occurring mineral involving strontium in the structure from Sarusaka, Kochi, Japan. As shown in
Figure 6, spectra are relatively well-separated from each other for the standard samples, indicating that the chemical states of Sr
2+ were different among SrCl
2, SrCO
3, and slawsonite. On the other hand, XANES spectra of strontium in the SC-GP specimens are almost in a bundle, indicating that chemical states of Sr
2+ in these specimens are nearly identical. The obtained results indicate that the chemical states of Sr
2+ in SC-GP specimens resemble that in slawsonite. Incidentally, in slawsonite, Sr
2+ is surrounded by O
2− in 7-hold coordination with a mean M-O distance of 0.263 nm [
46]. Deducting the ionic radius of O
2−, which is 0.126 nm, after Shannon and Prewitt [
43], 0.137 nm is obtained as the net ionic radius of Sr
2+. However, this ionic radius of Sr
2+ corresponds to 8-hold coordination, according to [
47].
It should be stressed again that 1-3SC showed less capability of Sr
2+ at 4 + 2 and 24 weeks of age. However, it was found that the immobilization ratio became enhanced to 91.73% at the intermediate age of 12 weeks. XANES measurements were performed by using 4 + 2 week age specimens, but there is about one month time lag for the actual measurements, due to time sharing schedules of the facility. Therefore, it is difficult, at the moment, to tell the spectra of 1-3SC, whether coming from embryo stage of C-A-S-H gels due to slow coagulation or coming from well-developed and matured C-A-S-H or (C, N)-A-S-H gels. According to literature [
48,
49], Sr
2+ incorporations are unstable in metakaolin-based geopolymers, in which N-A-S-H gels form preferentially. In other words, depletion of Sr
2+ takes place to yield SrCO
3 in air, but no depletion of Cs
+ occurs, meaning 100% incorporation of Cs
+ into N-A-S-H. Formation of Sr(OH)
2 and as SrCO
3 is also reported in the metakaolin-based geopolymer, where the clinoptilolite zeolite encapsulating Sr
2+ and Cs
+ is mixed [
50].
At the moment, no spectral profiles corresponding to SrCO3 were observed in the XANES spectra, even for 1-3SC, which showed highly fluctuating immobilization ratios of Sr2+ with elapsed time, as mentioned. Therefore, we concluded that no depletion of Sr2+ took place in our SC-GP specimens and that the fluctuations may be attributed to the transitional GP-gel structures of C-A-S-H or (C, N)-A-S-H. That is, for instance, differences in degrees of polymerization and/or differences in tetrahedral order–disorder configurations, which would be sensitive to acidic conditions of the leaching test. The XANES technique will become a powerful measure to clarify the bonding and coordination circumstances of matrix gels in geopolymers.