2. Experimental Procedure
A 100-nm-thick hydro-generated a-TiO
x film was deposited on silicon substrates (10 × 10 mm
2) by a plasma-enhanced chemical vapor deposition (PECVD) system, using a titanium tetraisopropoxide [Ti(OC
3H
7)
4, TTIP]-oxygen gas mixture. The deposition pressure, power, and gas flow rate of TTIP/O
2 were controlled at 40 Pa, 100 W, and 120/20 sccm, respectively. A detailed system setup and deposition parameters have been described elsewhere [
12]. Incorporating fluorine ions into the nano-textured surface on the a-TiO
x film was achieved by pre-irradiating a UV light for 5 h through an anodic alumina membrane mask and then etching in the diluted hydrofluoric (HF) solution for a different time. The selective fluorination etching (hereafter abbreviated to SFE) has been addressed elsewhere [
13]. A 100-nm-thick a-TiO
x film using PECVD under the same deposition condition was then coated onto the base layer with surface nano-textures to realize the a-TiO
x film surface free of the fluorine ions. In addition, another set of the a-TiO
x film crystallized into anatase structures was prepared by post-annealing the un-treated a-TiO
x film at 500 °C for 30 min under oxygen ambient (hereafter denoted as annealed TiO
x film) as a comparison.
Film thickness of these a-TiOx films with and without the SFE treatment as well as prepared using the two-step deposition was measured using a surface profile system (Dektak 6M, Veeco, New York, NY, USA). Surface roughness was measured using atomic force microscopy (AFM, DI-3100, Veeco, New York, NY, USA) with the tapping mode. The surface and cross-section morphologies were observed by a field emission scanning electron microscope (FE-SEM, JSM-6700F, JEOL, Tokyo, Japan) operated at 3 kV. Fourier transform infrared spectrometry (FTIR, FT/IR-4100, JASCO, Halifax, NS, Canada) and X-ray photoelectron spectroscope (XPS, ULVAC-PHI, Quantera SXM, Kanagawa, Japan) with monochromatic Al Kα radiation were employed to examine the film’s chemical bonding states and surface bond nature. The photocatalytic activity for the a-TiOx and annealed TiOx films illuminated by the UV light with a constant intensity of 1 mW/cm2 was evaluated by the decolorization of a 20 ppm concentrated methylene blue (MB) solution using the UV-Vis spectrophotometer from the absorbance of the resulting solution at 665 nm under atmosphere ambient.
3. Results and Discussion
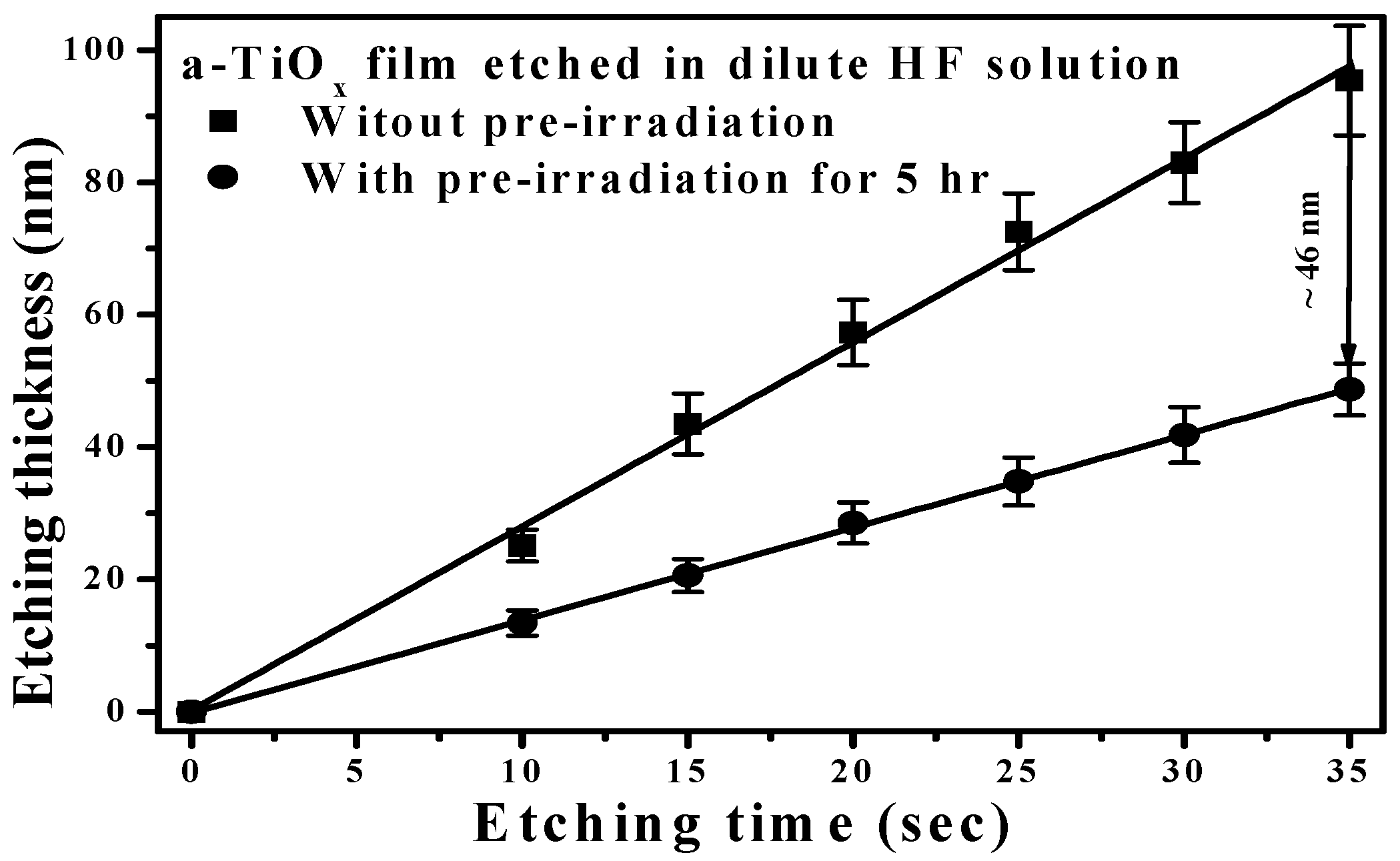
Figure 1 shows the etching thickness of the a-TiO
x film with and without the UV light pre-irradiation for 5 h, and then immersed in the HF etching solution for different etching times. The etching thickness of the a-TiO
x film treated by additive UV light pre-irradiation was apparently lower than the a-TiO
x film directly etched by the HF solution. The less acidic surface of the a-TiO
x film, as a consequence of the Ti(IV)−OH, transformed into a Ti(III)−OH
− group due to the generated electrons under UV light pre-irradiation, was responsible for the alleviation of the sequential etching process [
13]. A large discrepancy occurred in the etching thickness between the a-TiO
x films with and without the UV light pre-irradiation (approximately 46 nm) as the etching time reached 35 s, while at this time the 100-nm-thick a-TiO
x film directly etched by the HF solution was almost completely removed, as shown in
Figure 1. Because an apparent difference in the etching thickness of the a-TiO
x films was obtainable from the fluorination etching with and without UV light pre-irradiation, a selective fluorination etching treatment on the a-TiO
x film to modify its surface morphology was carried out by selectively shadowing the surface through a nano-sized mask with UV light pre-irradiation and then processing the fluorination etching.
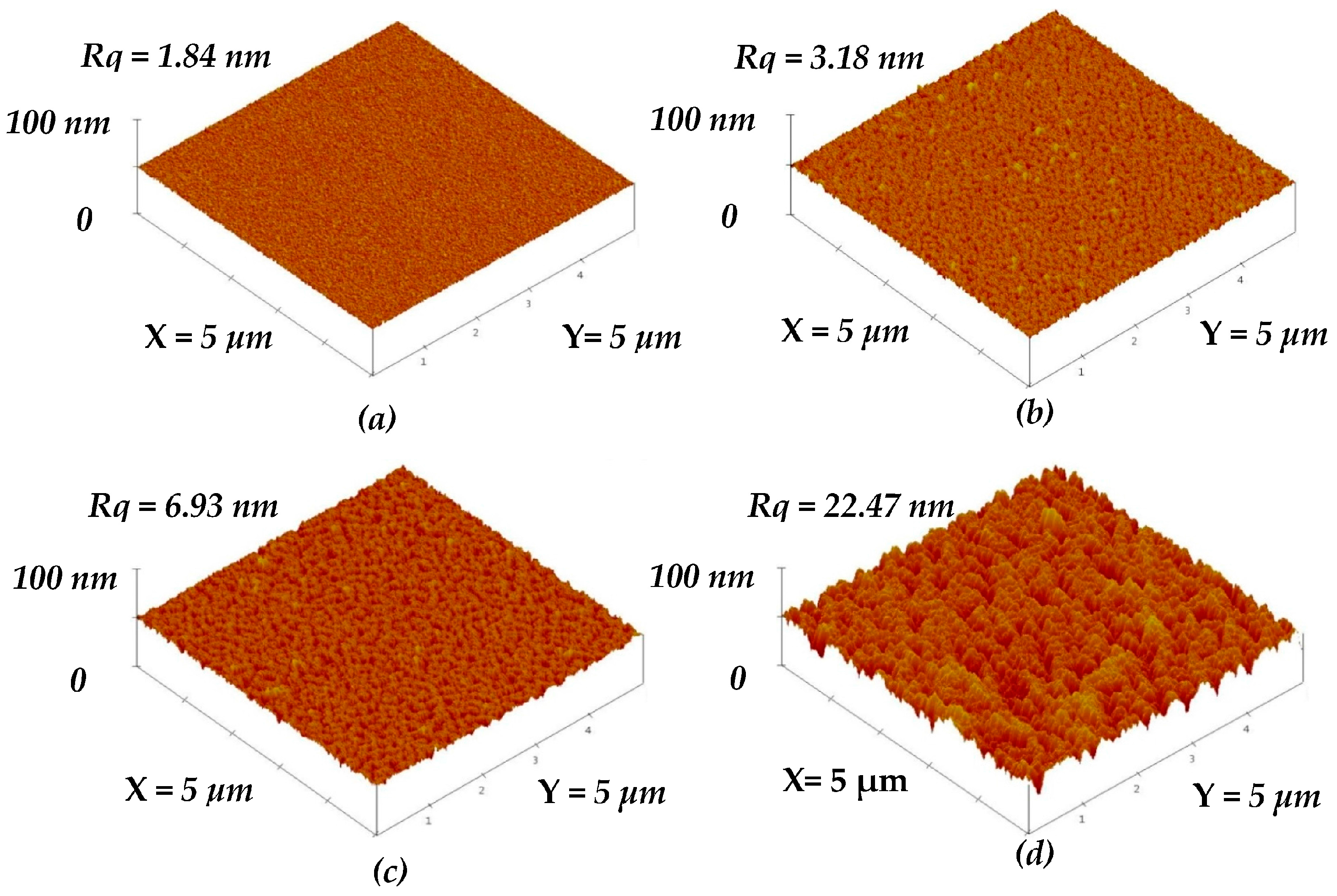
Figure 2b–d show the surface roughness of the a-TiO
x film selectively etched by the HF solution for 10, 20, and 35 s, respectively (the un-treated a-TiO
x film is given in
Figure 2a for comparison). The untreated a-TiO
x film exhibited a smooth surface with a roughness of about 1.84 nm, whereas an increase in the surface roughness was obtained from the a-TiO
x films treated by the additive SFE process. The voids on the a-TiO
x surface gradually became visible as the SFE-treated time increased. Notable pinnacles and valleys were then clearly observed from the surface of the a-TiO
x film treated by the SFE process for 35 s, which corresponded to a very high surface roughness of 22.47 nm.
The surface morphologies of the untreated a-TiO
x film and the films treated by the SFE process for 10, 20, and 35 s, respectively, are shown in
Figure 3a–d. As shown in
Figure 3a, the particles on the untreated a-TiO
x surface are distributed densely and abnormally, whereas these particles observed from the a-TiO
x film surface processed by the SFE treatment for 10 s became separated with a round-like shape. The SFE process also resulted in these particles being uniformly distributed with an average diameter of about 20 nm, which was similar to the porous size of the AAM mask, as shown in the inset figure. Fine particles were then observed from the a-TiO
x film surface as it was further treated by the SFE process for 20 s (
Figure 3b). The sidewall etching that led to the overcut profile was one possible reason why these particles appeared on the surface, evolving from a round-like to a needle-like shape. Meanwhile, the corresponding surface roughness of the a-TiO
x film also increased from 3.18 to 6.93 nm, as measured from
Figure 2c. When the SFE treatment on the a-TiO
x film reached 35 s, the surface morphology also showed definite boundaries in addition to the fine particles. Referring to the etching thickness of the a-TiO
x film given in
Figure 1, the obvious and wide boundaries were attributed to the regions of the a-TiO
x film that was completely removed from the substrate after etching by the HF solution without additive UV light irradiation.
These experiments demonstrated that the particles’ shape and distribution on the a-TiO
x film surface were modifiable and controllable using the SFE treatment for different times. These base layers with surface nano-textures affecting the subsequent deposited a-TiO
x film were then investigated.
Figure 4b–d give the surface roughness of the a-TiO
x films deposited onto the SFE-treated surface with nano-textures shown in
Figure 2b–d, respectively, as well as the film deposited onto the untreated surface (
Figure 4a). All the a-TiO
x films prepared using this two-step deposition process exhibited a higher surface roughness than that of their base layers shown in
Figure 2a–d. The surface roughness of the a-TiO
x film deposited onto the untreated surface increased slightly to 2.27 nm. In contrast, a marked increase in the surface roughness was measured from the a-TiO
x film deposited onto the SFE-treated surface. The rougher the surface of the base layer obtained, the higher the increase in the roughness of the subsequently deposited a-TiO
x film. In addition, features of the pinnacles and valleys appearing on the surface of the a-TiO
x film deposited onto the surface treated with SFE for 35 s (
Figure 4d) became more visible compared to those observed from the surface of its base layer (
Figure 2d).
The corresponding surface morphologies shown in
Figure 4 conducted from SEM measurements are presented in
Figure 5a–d. The surface morphology of the a-TiO
x film deposited onto the untreated surface (
Figure 5a) was almost identical to the film deposited onto the substrate on which the particles were distributed randomly and densely as presented in
Figure 3a. For the a-TiO
x film deposited onto the surface with round-like particles, as shown in
Figure 3b, the particles appearing on the a-TiO
x film (
Figure 5b) had an average diameter apparently larger than those distributed over the base layer’s surface, although their shape and distribution were quite similar. This suggested that the nuclei of the subsequently deposited a-TiO
x film were prone to forming and growing along the particles distributed over the base layer. The growth of particles on the a-TiO
x film became closer and resulted in the considerable increase in the surface roughness. For the a-TiO
x film deposited onto the surface shown in
Figure 3c, the nuclei growing along the needle-like structure resulted in the particles on the a-TiO
x film surface evolving into a round-like shape with visible boundaries. As the surface morphologies of the two-step a-TiO
x films were deeply relevant to the particles on the base layer, the distributions of the significant channels and clusters observed from the a-TiO
x film shown in
Figure 5d consisted of the growth of the fine particles and the enhancement of the boundaries appearing on the base layer shown in
Figure 3d.
The cross-section micrographs shown in
Figure 6a,b give further insights into the growth of the a-TiO
x film prepared using the two-step deposition process. In
Figure 6a, random and dense fiber-like structures can be seen in both a-TiO
x layers, which had a definite interface, as indicated by arrows. This implied that the untreated base layer would not cause a structural change in the subsequently deposited a-TiO
x film. In contrast, the cross-section of the two-step a-TiO
x film shown in
Figure 6b exhibited the feature of nanorod-like structures with no significant interface, confirming that the growth mechanism of the a-TiO
x film deposited onto the SFE-treated base layer was completely different from the film deposited onto the untreated base layer. The dimension of these nanorod-like structures was found to be widened with the growth of the two-step a-TiO
x film. Combined with their surface morphologies (
Figure 3d and
Figure 5d), the particles on the base layer achieved using the SFE treatment were likely to act as growth seeds for the subsequent a-TiO
x deposition, thereby resulting in the upper a-TiO
x film, demonstrating conformity in the base layer with the increase in the surface roughness.
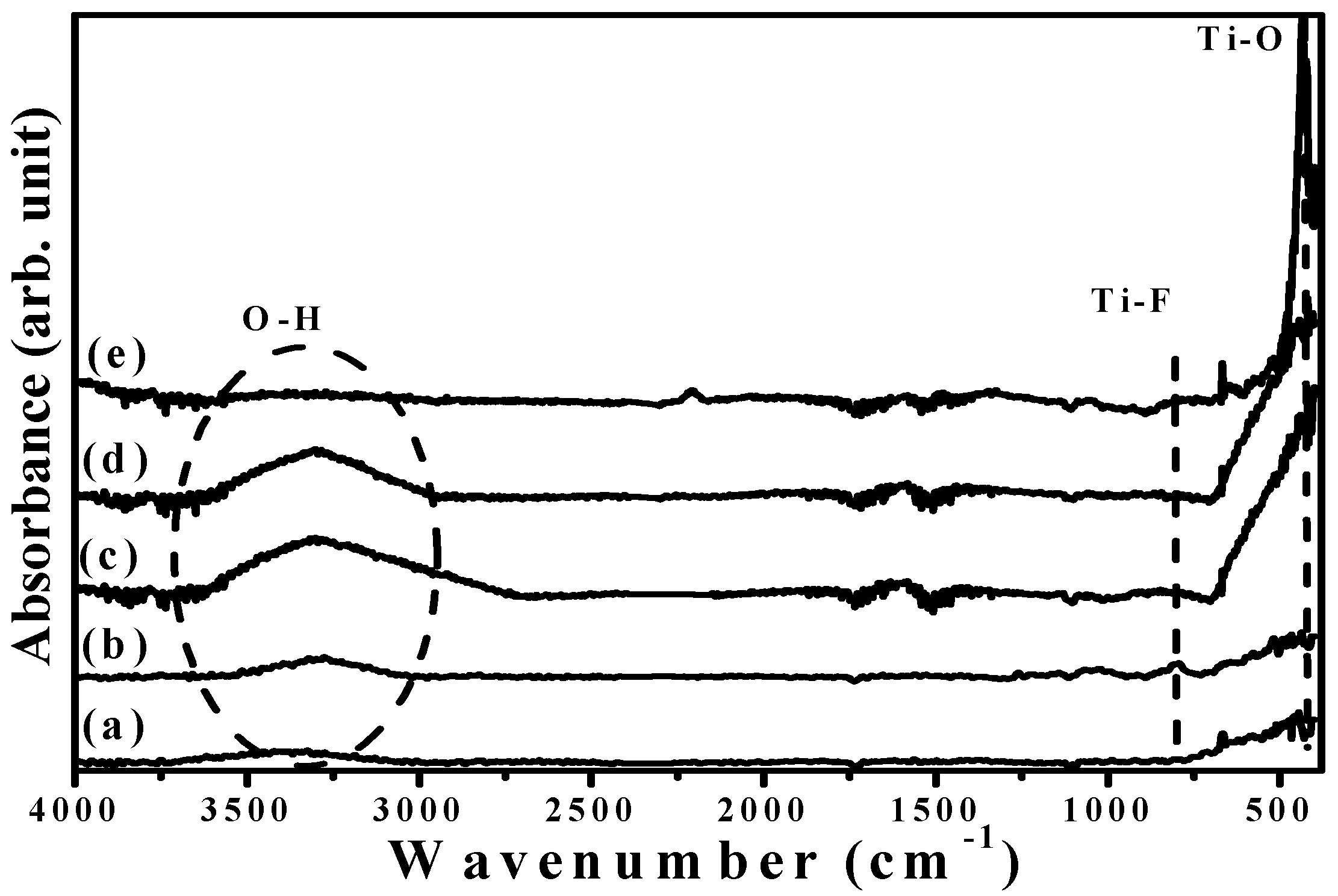
FTIR spectra of the base layer with and without an additive SFE treatment and the a-TiO
x films prepared using two-step deposition are illustrated in
Figure 7a–d, respectively (
Figure 7e) also shows the FTIR spectrum of the annealed TiO
x film for comparison). Both the untreated and SFE-treated base layers, as shown in
Figure 7a,b, respectively, consisted of the fingerprint peak of the Ti–O bond around 400–800 cm
−1 with the functional group of hydroxyl (OH) around high wavenumbers (2800–3700 cm
−1) [
12]. The SFE treatment also caused the base layer to emit an additive signal at about 820 cm
−1, which was associated with the Ti–F vibration mode [
13]. In addition, the incorporation of the fluorine ions into the base layer led to a shift of the O-H bond from 3450 to 3275 cm
−1 due to an increase in the surface acidity. When the a-TiO
x film was deposited onto the SFE-treated base layer, the Ti–F bond was hardly observed in the FTIR spectrum (
Figure 7d). Compared with the FTIR spectrum of the a-TiO
x film deposited onto the untreated base layer (curve c), both spectra only featured Ti–O and O–H bonds with almost the same peak position, except for a higher relative O-H bond intensity obtained from the a-TiO
x film deposited onto the SFE-treated base layer. Since the porous structures distributed in the low-temperature deposited oxidation film were the main reason responsible for the formation of the O–H groups [
15,
16], the reinforcement in the O–H bond obtained from the two-step deposition of the a-TiO
x coated onto the SFE-treated base layer thus implied the increase in the amounts of the pores. In addition, a sharp and intense Ti–O bond with the disappearance of the O–H bond as a consequence of the anatase crystallization was obtained from the FTIR spectrum of the annealed TiO
x film.
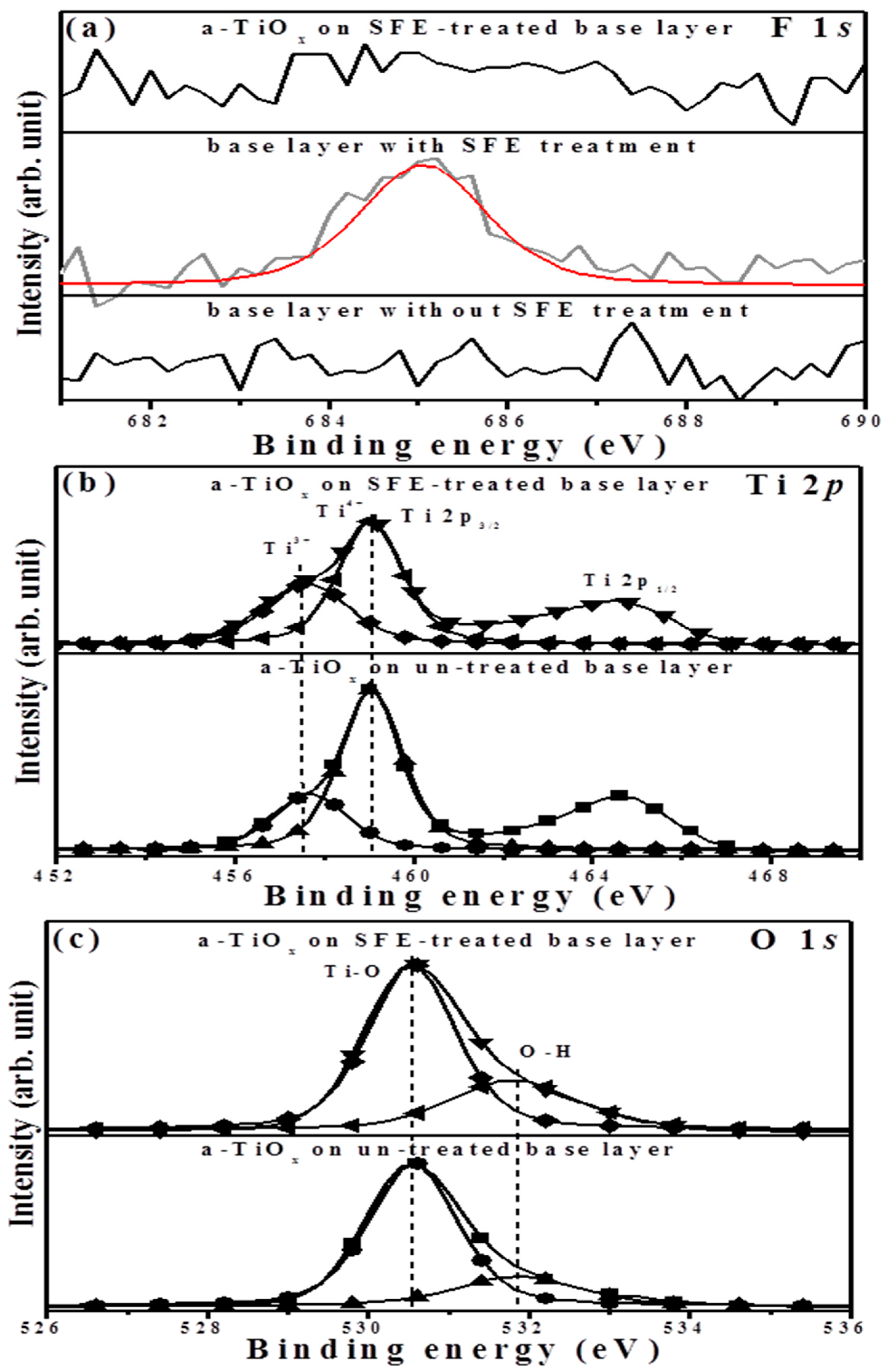
The binding energies related to the F 1
s, Ti 2
p, and O 1
s core levels measured from the surface of the two-step deposited a-TiO
x film as well as the base layer are respectively illustrated in
Figure 8a–c. Although a significant fluorine signal on the surface of the base layer emerged at about 684.9 eV, which was assigned to the Ti–F chemical bond due to the SFE treatment [
17], this signal was absent in the surface of the subsequently deposited a-TiO
x film, indicating the achievement of the fluorine-free surface. In the Ti 2
p spectrum (
Figure 8b), both the peaks of the binding energies for the Ti 2
p1/2 and Ti 2
p3/2 occurred at approximately 464.6 and 458.9 eV, respectively, with a binding energy difference of 5.7 eV. A broad signal with a distinct satellite peak was observed from the binding energy of Ti 2
p3/2 for the a-TiO
x film deposited onto the SFE-treated base layer, while this peak became sharp with a tail extending to the low binding energy in the spectrum of the a-TiO
x film deposited onto the untreated base layer. The peak of Ti 2
p3/2 could be deconvoluted into two species of Ti
4+ and Ti
3+ ion states at 457.6 and 459.0 eV, respectively [
18]. The composition of the Ti
3+ to Ti
4+ state (in the area of Ti
3+/(Ti
4+ + Ti
3+)), which was associated with the deficiency in the oxygen atoms on the surface, apparently increased from 0.29 to 0.39 as the a-TiO
x film was deposited onto the SFE-treated base layer. Regarding the O 1
s spectra (
Figure 8c), both surfaces of these two samples displayed an intense peak with a long tail extending to a high binding energy that could be deconvoluted into two feature peaks. The binding energy peak located at 530.6 eV was related to the Ti–O chemical bond, where the peak at the high binding energy of 531.8 eV emerged from the hydroxyl group (O–H) [
19,
20]. As determined from previous papers [
21,
22], the presence of the O-H bond indicated the termination of the chemical bond and/or contaminants and, thus, was responsible for the structural voids and boundaries. Accordingly, marked boundaries and voids observed from the surface and cross-section morphologies of the a-TiO
x film deposited onto the SFE-treated base layer were hydroxylated more intensely in the binding energy of O 1
s (approximately 0.31, in the area of O–H/(Ti–O + O–H)) than those of the film deposited onto the untreated base layer (approximately 0.24).
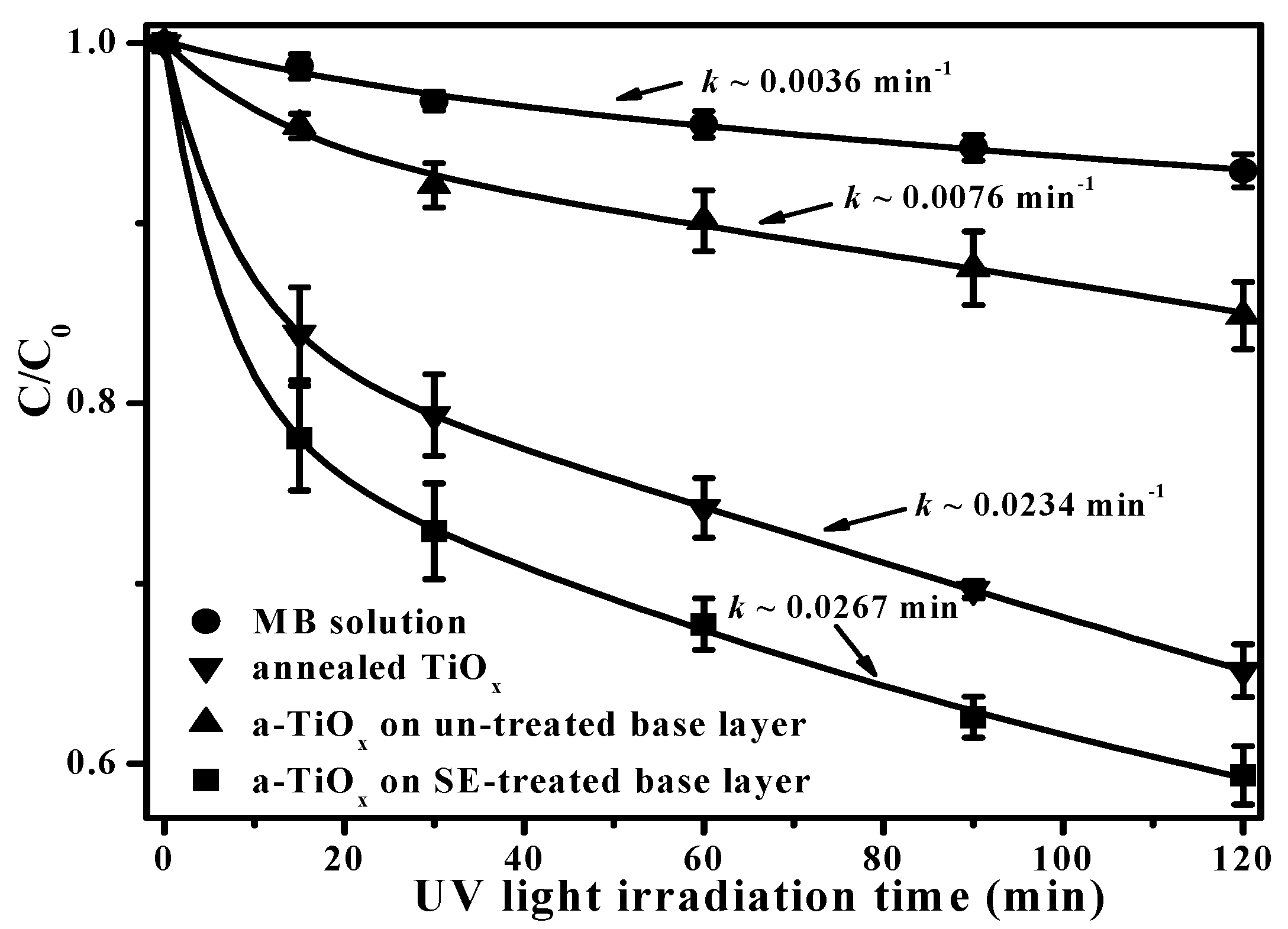
In addition, because the hydroxyl groups are beneficial for trapping hole carriers to suppress the recombination of the photo-generated electron-hole pairs, as demonstrated in previous studies [
23,
24], the a-TiO
x film deposited onto the SFE-treated base layer to degrade the MB solution, as depicted in
Figure 9, is greatly improved compared to the same film deposited onto the untreated base layer. It is also worth noting that the a-TiO
x film with a surface free of the fluorine ions achieved using the two-step deposition even exhibited better efficiency in decomposing the MB solution than the annealed TiO
x film with anatase structures. The rate constant,
k, which represents the quality of the photocatalytic activity of the film can be evaluated by fitting the curves shown in
Figure 9, using the following relationship [
25]:
where
C and
C0 are the concentrations of the MB solution at a UV light irradiation time of
t = 0 and
t, respectively. The
k value evaluated from each curve is denoted in
Figure 9. Clearly, the a-TiO
x film deposited onto the SFE-treated base layer corresponded to a rate constant of 0.0267 min
−1, which was three times higher than the film deposited onto the untreated base layer (0.0076 min
−1) as well as a little higher than that of the annealed TiO
x film (0.0234 min
−1). According to the investigations into the morphologies and the analysis of the chemical bond configurations of the two-step deposited a-TiO
x film, the apparent roughening and nano-textured surface of the upper a-TiO
x film without the incorporation of the fluorine ions that grew by conforming along the particle seeds on the base layer surface, which was achieved using an additive SFE treatment, was the mechanism responsible for the great enhancement in the resulting photocatalytic activity.