Solvothermal Synthesis of Hierarchical Colloidal Nanocrystal Assemblies of ZnFe2O4 and Their Application in Water Treatment
Abstract
:1. Introduction
2. Results
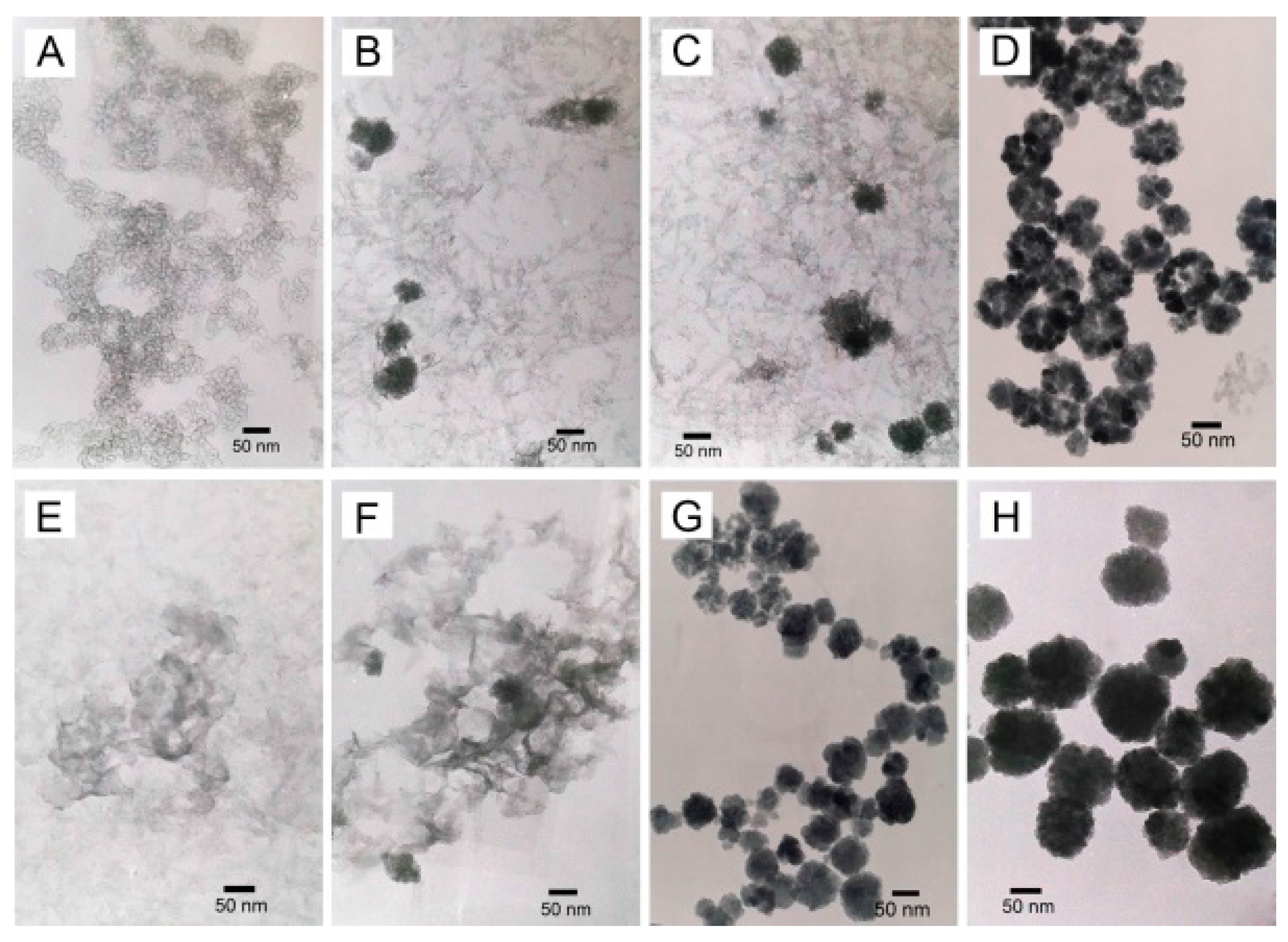
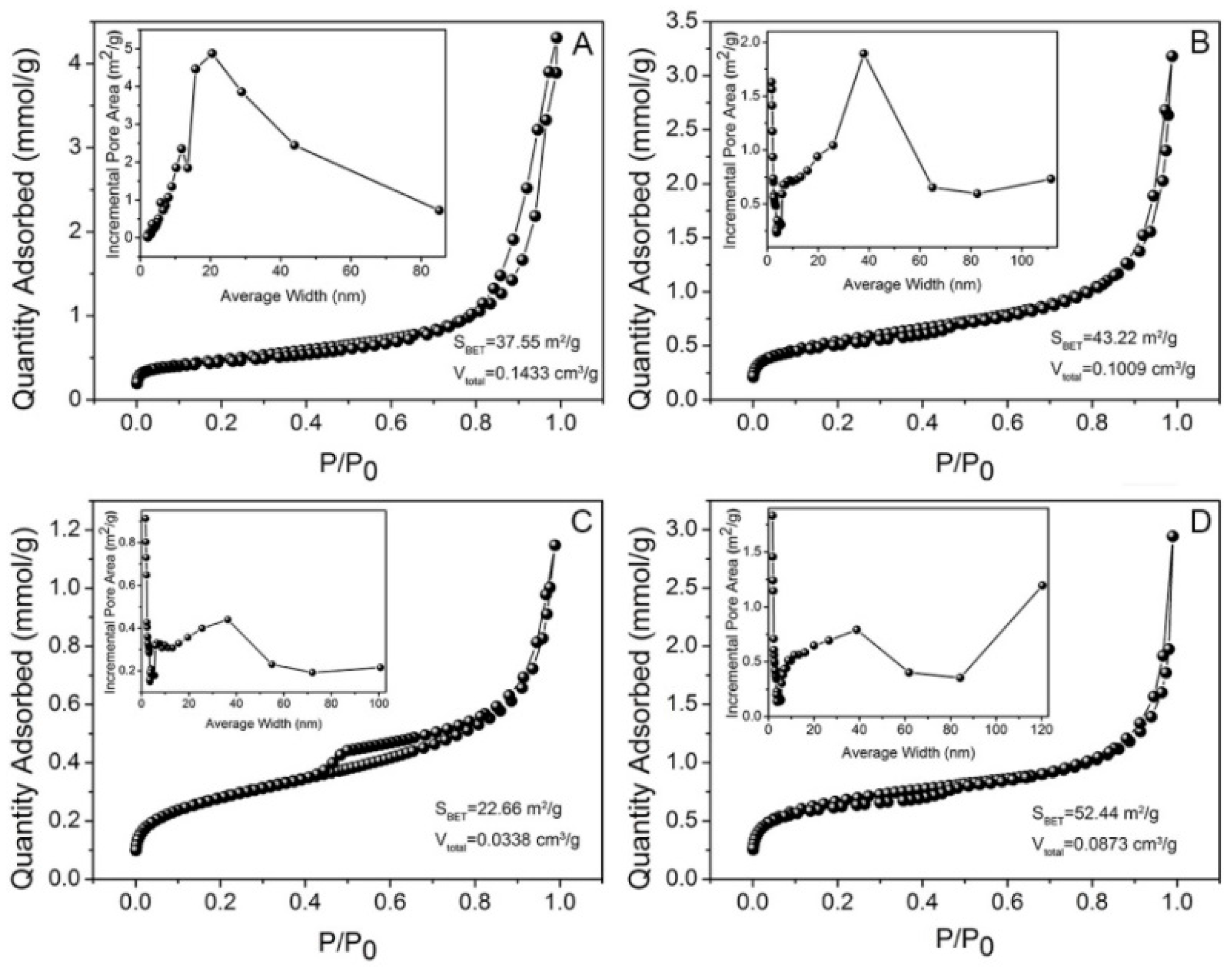
2.1. Morphology and Structure
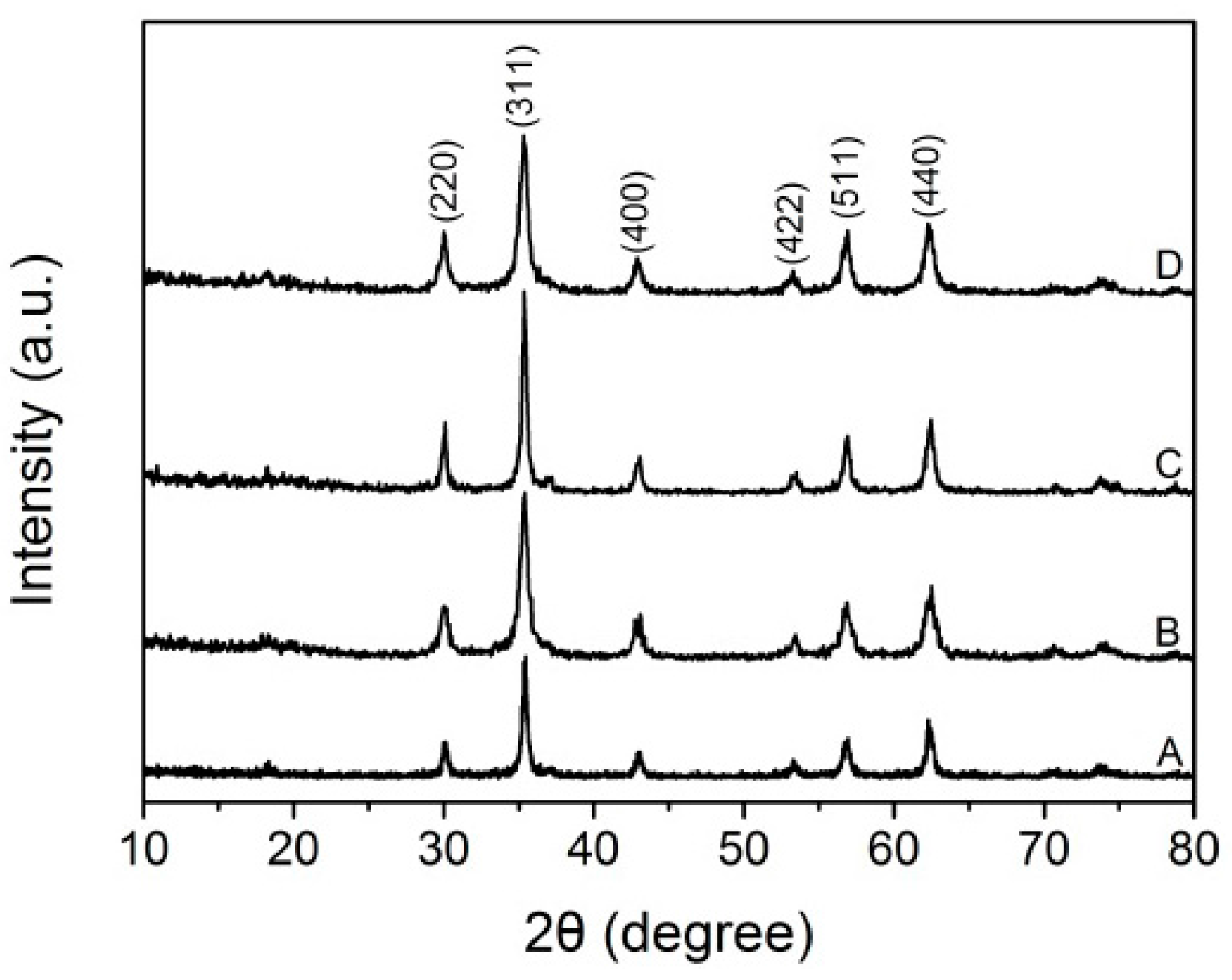
2.2. X-ray Diffraction (XRD) Analysis
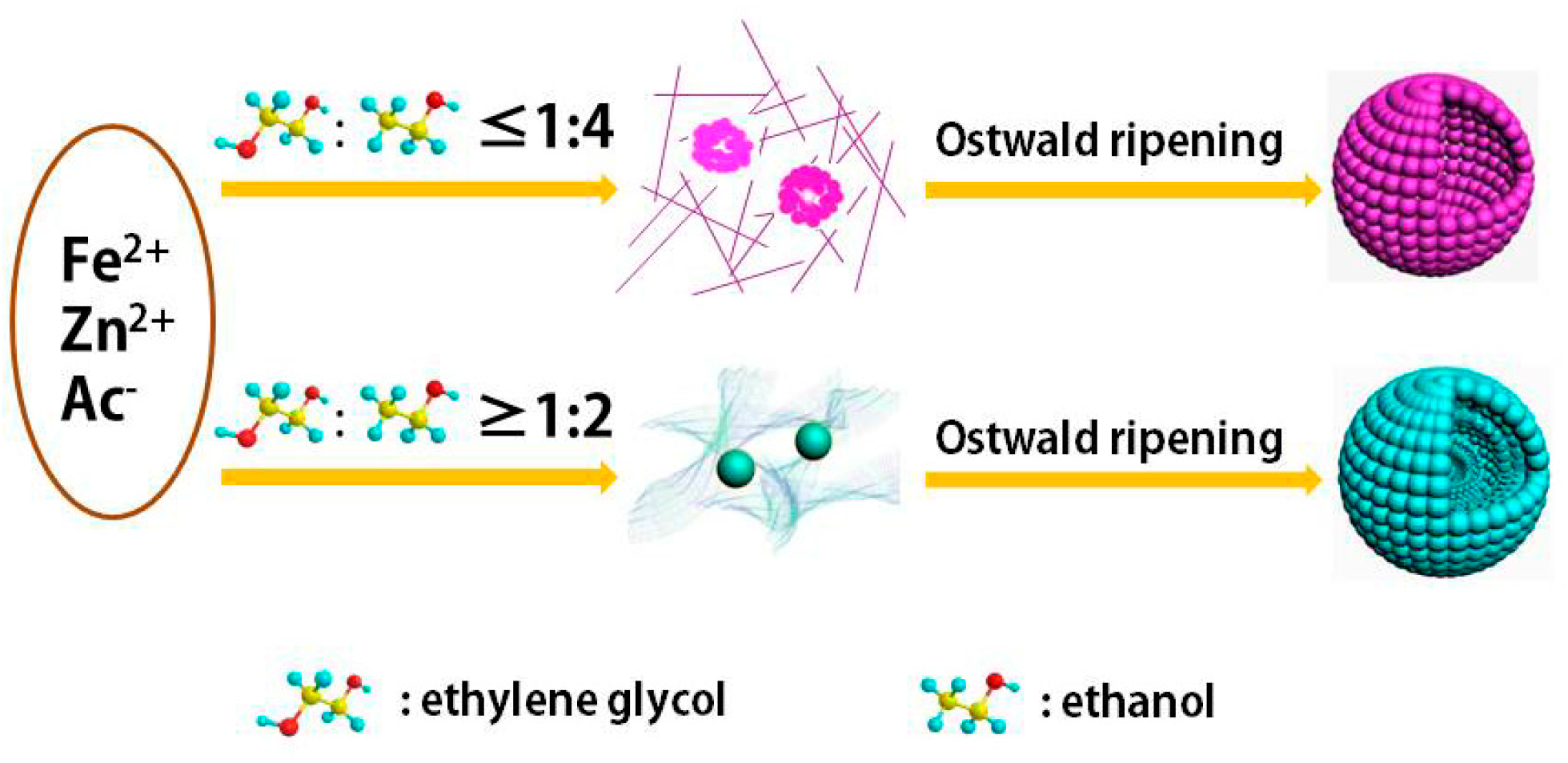
2.3. Evolution of the Intermediates
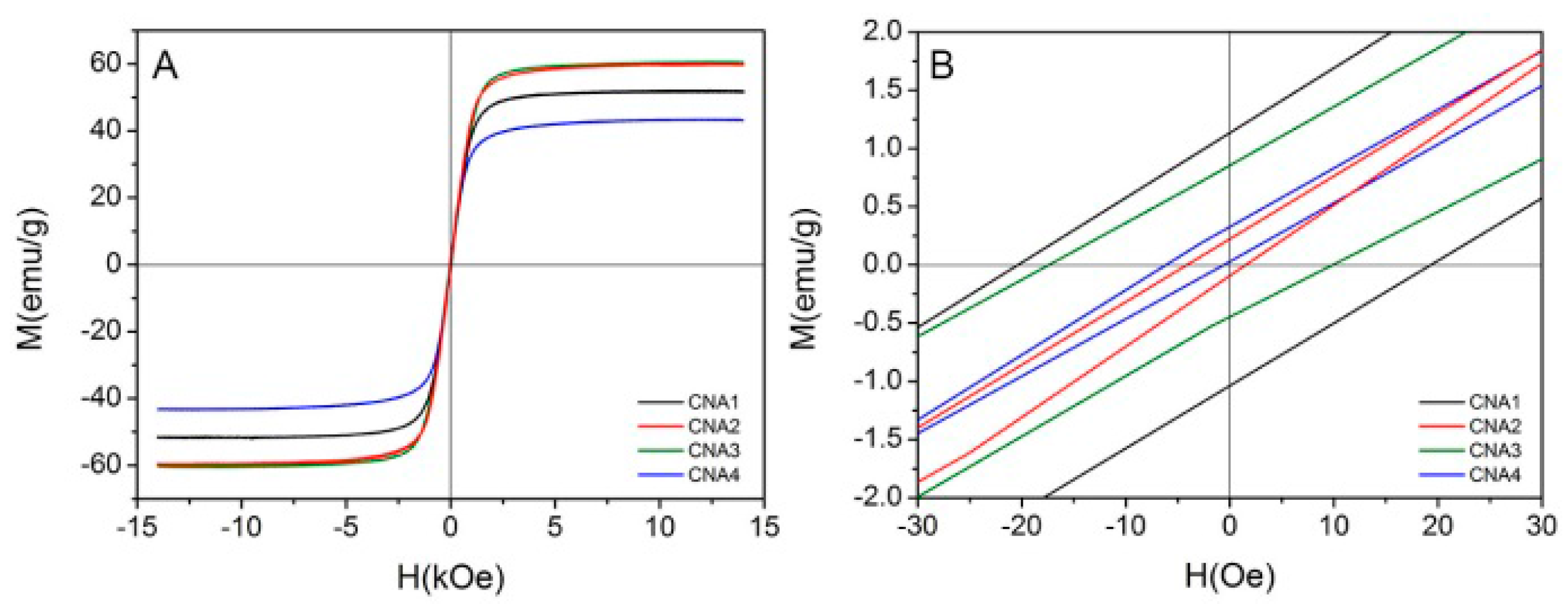
2.4. Magnetic Characterization
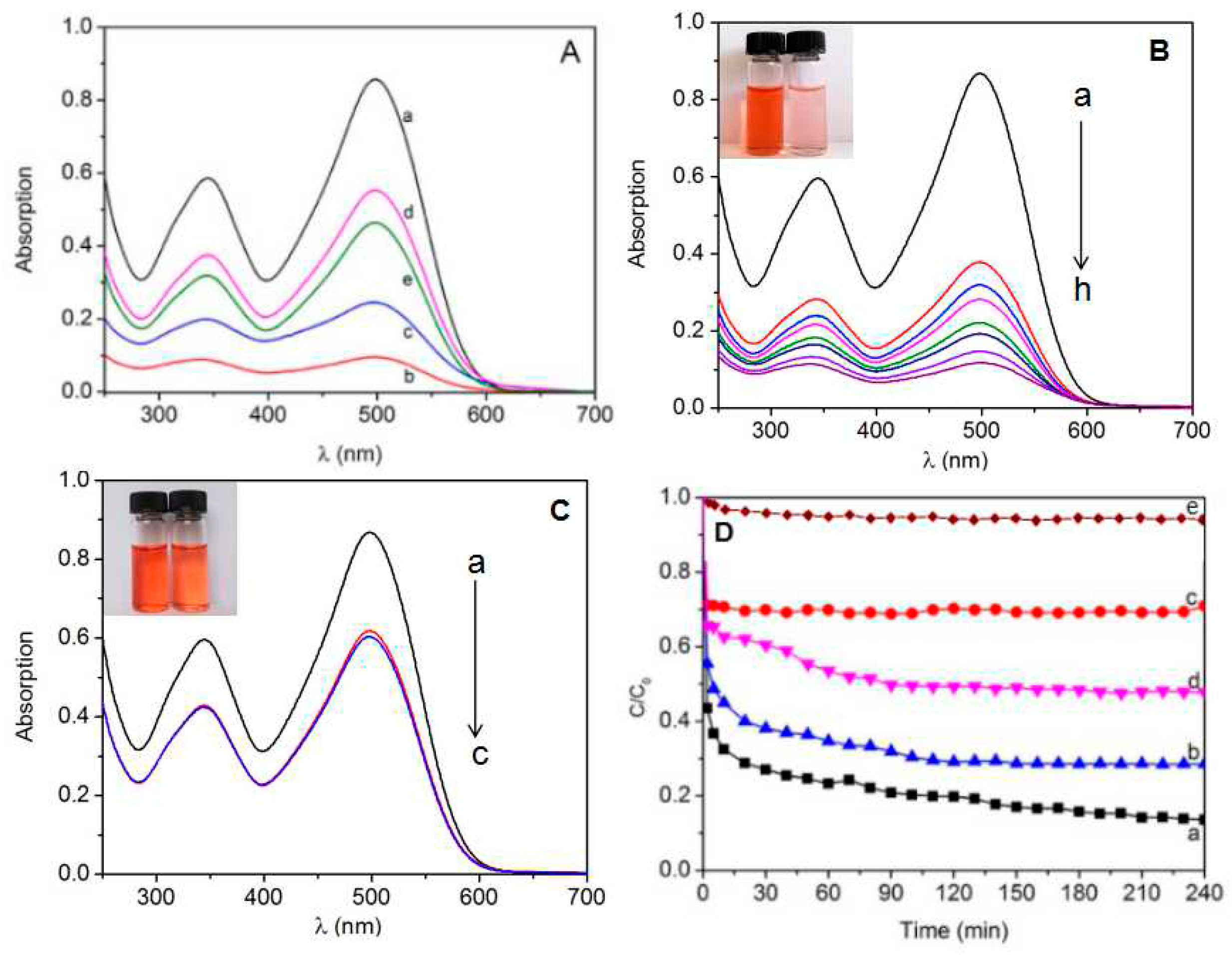
2.5. Adsorption of Congo Red
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Su, L.; Feng, J.; Zhou, X.M.; Ren, C.L.; Li, H.H.; Chen, X.G. Colorimetric detection of urine glucose based ZnFe2O4 magnetic nanoparticles. Anal. Chem. 2012, 84, 5753–5758. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.G.; Liu, X.H.; Yi, R.; Shi, R.R.; Zhang, N.; Qiu, G.Z. Selective synthesis and properties of monodisperse Zn ferrite hollow nanospheres and nanosheets. J. Phys. Chem. C 2008, 112, 8558–8563. [Google Scholar] [CrossRef]
- Sharma, H.B.; Singh, N.B.; Devi, K.N.; Lee, J.H.; Singh, S.B. Structural and optical properties of manganese substituted nanocrystalline bismuth ferrite thin films by sol–gel process. J. Alloy. Compd. 2014, 583, 106–110. [Google Scholar] [CrossRef]
- Guo, P.Z.; Cui, L.J.; Wang, Y.Q.; Lv, M.; Wang, B.Y.; Zhao, X.S. Facile synthesis of ZnFe2O4 nanoparticles with tunable magnetic and sensing sroperties. Langmuir 2013, 29, 8997–9003. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Rout, S.R.; Maiti, S.; Maiti, T.K.; Panda, A.B. Monodisperse mesoporous cobalt ferrite nanoparticles: Synthesis and application in targeted delivery of antitumor drugs. J. Mater. Chem. 2011, 21, 9185–9193. [Google Scholar] [CrossRef]
- Zhang, Q.; Valanoor, N.; Standard, O. Epitaxial (001) BiFeO3 thin-films with excellent ferroelectric properties by chemical solution deposition-the role of gelation. J. Mater. Chem. C 2015, 3, 582–595. [Google Scholar] [CrossRef]
- Yu, S.H.; Yoshimura, M. Ferrite/metal composites fabricated by soft solution processing. Adv. Funct. Mater. 2002, 12, 9–15. [Google Scholar] [CrossRef]
- Pillai, V.; Kumar, P.; Multani, M.S.; Shah, D.O. Structure and magnetic properties of nanoparticles of barium ferrite synthesized using microemulsion processing. Colloids Surf. A 1993, 80, 69–75. [Google Scholar] [CrossRef]
- Ji, R.L.; Cao, C.B.; Chen, Z.; Zhai, H.Z.; Bai, J. Solvothermal synthesis of CoxFe3−xO4 spheres and their microwave absorption properties. J. Mater. Chem. C 2014, 2, 5944–5953. [Google Scholar] [CrossRef]
- Rajamathi, M.; Seshadri, R. Oxide and chalcogenide nanoparticles from hydrothermal/solvothermal reactions. Curr. Opin. Solid. St. Mater. 2002, 6, 337–345. [Google Scholar] [CrossRef]
- Li, S.; Lin, Y.H.; Zhang, B.P.; Wang, Y.; Nan, C.W. Controlled fabrication of BiFeO3 uniform microcrystals and their magnetic and photocatalytic behaviors. J. Phys. Chem. C 2010, 114, 2903–2908. [Google Scholar] [CrossRef]
- Wang, J.; Ren, F.; Yi, R.; Yan, A.G.; Qiu, G.Z.; Liu, X.H. Solvothermal synthesis and magnetic properties of size-controlled nickel ferrite nanoparticles. J. Alloy. Compd. 2009, 479, 791–796. [Google Scholar] [CrossRef]
- Deng, Y.F.; Zhang, Q.M.; Tang, S.D.; Zhang, L.T.; Deng, S.N.; Shi, Z.C.; Chen, G.H. One-pot synthesis of ZnFe2O4/C hollow spheres as superior anode materials for lithium ion batteries. Chem. Coummun. 2011, 47, 6828–6830. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.L.; Gu, X.Y.; Zuo, D.T.; Wang, Z.K.; Wang, N.Y.; Yao, K.H. Microemulsion-based synthesis of porous zinc ferrite nanorods and its application in a room-temperature ethanol sensor. Nanotechnology 2008, 19, 405503. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, X.L.; Yang, Y.; Xiao, W.; Li, Z.W.; Xue, D.S.; Li, F.S.; Ding, J. Synthesis of nonstoichiometric zinc ferrite nanoparticles with extraordinary room temperature magnetism and their diverse applications. J. Mater. Chem. C 2013, 1, 2875–2885. [Google Scholar] [CrossRef]
- Toledo-Antonio, J.A.; Nava, N.; Martinez, M.; Bokhimi, X. Correlation between the magnetism of non-stoichiometric zinc ferrites and their catalytic activity for oxidative dehydrogenation of 1-butene. Appl. Catal. A 2002, 234, 137–144. [Google Scholar] [CrossRef]
- Fan, G.; Gu, Z.; Yang, L.; Li, F. Nanocrystalline zinc ferrite photocatalysts formed using the colloid mill and hydrothermal technique. Chem. Eng. J. 2009, 155, 534–541. [Google Scholar] [CrossRef]
- Ibrahim, I.; Ali, I.O.; Salama, T.M.; Bahgat, A.A.; Mohamed, M.M. Synthesis of magnetically recyclable spinel ferrite (MFe2O4, M = Zn, Co, Mn) nanocrystals engineered by sol gel-hydrothermal technology: High catalytic performances for nitroarenes reduction. Appl. Catal. B Environ. 2016, 181, 389–402. [Google Scholar] [CrossRef]
- Zhang, J.; Song, J.M.; Niu, H.L.; Mao, C.J.; Zhang, S.Y.; Shen, Y.H. ZnFe2O4 nanoparticles: Synthesis, characterization, and enhanced gas sensing property for acetone. Sens. Actuators B Chem. 2015, 221, 55–62. [Google Scholar] [CrossRef]
- Hou, L.R.; Hua, H.; Lian, L.; Cao, H.; Zhu, S.Q.; Yuan, C.Z. Green template-free synthesis of hierarchical shuttle-shaped mesoporous ZnFe2O4 microrods with enhanced lithium storage for advanced Li-ion batteries. Chem. Eur. J. 2015, 21, 13012–13019. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Hu, Y.; Biasini, M.; Beyermann, W.P.; Yin, Y. Superparamagnetic magnetite colloidal nanocrystal clusters. Angew. Chem. Int. Ed. 2007, 46, 4342–4345. [Google Scholar] [CrossRef] [PubMed]
- Narayanaswamy, A.; Xu, H.; Pradhan, N.; Peng, X. Crystalline nanoflowers with different chemical pompositions and Physical properties grown by limited ligand protection. Angew. Chem. Int. Ed. 2006, 45, 5361–5364. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.Y.; Wang, Q.X.; Shi, Z.P.; Ma, C.; Ding, Y.M.; Huo, N.N.; Zhang, J.; Yang, S.T. Porous hierarchical nitrogen-doped carbon coated ZnFe2O4 composites as high performance anode materials for lithium ion batteries. Electrochim. Acta 2015, 180, 622–628. [Google Scholar] [CrossRef]
- Liu, B.; Zeng, H.C. Symmetric and asymmetric Ostwald ripening in the fabrication of homogeneous core–shell semiconductors. Small 2005, 1, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.G.; Zeng, H.C. Preparation of hollow anatase TiO2 nanospheres via Ostwald ripening. J. Phys. Chem. B 2004, 108, 3492–3495. [Google Scholar] [CrossRef]
- Li, W.; Wang, L.; Li, G.; Xu, Y. Hollow CoFe2O4–Co3Fe7 microspheres applied in electromagnetic absorption. J. Magn. Magn. Mater. 2015, 377, 259–266. [Google Scholar] [CrossRef]
- Li, Z.; Gao, K.; Han, G.; Wang, R.; Li, H.; Zhao, X.S.; Guo, P.Z. Solvothermal synthesis of MnFe2O4 colloidal nanocrystal assemblies and their magnetic and electrocatalytic properties. New J. Chem. 2015, 39, 361–368. [Google Scholar] [CrossRef]
- Greer, J.R.; de Hosson, J.T.M. Plasticity in small-sized metallic systems: Intrinsic versus extrinsic size effect. Prog. Mater. Sci. 2011, 56, 654–724. [Google Scholar] [CrossRef]
- Daou, T.; Greneche, J.; Pourroy, G.; Buathong, S.; Derory, A.; Ulhaq-Bouillet, C.; Donnio, B.; Guillon, D.; Begin-Colin, S. Coupling agent effect on magnetic properties of functionalized magnetite-based nanoparticles. Chem. Mater. 2008, 20, 5869–5875. [Google Scholar] [CrossRef]
- Quickel, T.E.; Le, V.H.; Brezesinski, T.; Tolbert, S.H. On the correlation between nanoscale structure and magnetic properties in ordered mesoporous cobalt ferrite (CoFe2O4) thin films. Nano Lett. 2010, 10, 2982–2988. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.; Wang, Y.; Zhao, L.; Jiang, Q. Adsorption capability for Congo red on nanocrystalline MFe2O4 (M = Mn, Fe, Co, Ni) spinel ferrites. Chem. Eng. J. 2012, 181, 72–79. [Google Scholar] [CrossRef]
- Oliveira, L.C.; Petkowicz, D.I.; Smaniotto, A.; Pergher, S.B. Magnetic zeolites: A new adsorbent for removal of metallic contaminants from water. Water Res. 2004, 38, 3699–3704. [Google Scholar] [CrossRef] [PubMed]
- Konicki, W.; Sibera, D.; Mijowska, E.; Lendzion-Bieluń, Z.; Narkiewicz, U. Equilibrium and kinetic studies on acid dye Acid Red 88 adsorption by magnetic ZnFe2O4 spinel ferrite nanoparticles. J. Colloid Interf. Sci. 2013, 398, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.S.; Hu, J.S.; Liang, H.P.; Cao, A.M.; Song, W.G.; Wan, L.J. Self-assembled 3D flowerlike iron oxide nanostructures and their application in water treatment. Adv. Mater. 2006, 18, 2426–2431. [Google Scholar] [CrossRef]
- Guo, P.Z.; Zhang, G.L.; Yu, J.Q.; Li, H.L.; Zhao, X.S. Controlled synthesis, magnetic and photocatalytic properties of hollow spheres and colloidal nanocrystal clusters of manganese ferrite. Colloids Surf. A Physicochem. Eng. Asp. 2012, 395, 168–174. [Google Scholar] [CrossRef]
- Zeng, S.; Duan, S.; Tang, R.; Li, L.; Liu, C.; Sun, D. Magnetically separable Ni0.6Fe2.4O4 nanoparticles as an effective adsorbent for dye removal: Synthesis and study on the kinetic and thermodynamic behaviors for dye adsorption. Chem. Eng. J. 2014, 258, 218–228. [Google Scholar] [CrossRef]
- Zhu, H.; Fu, Y.; Jiang, R.; Yao, J.; Liu, L.; Chen, Y.; Xiao, L.; Zeng, G. Preparation, characterization and adsorption properties of chitosan modified magnetic graphitized multi-walled carbon nanotubes for highly effective removal of a carcinogenic dye from aqueous solution. Appl. Surf. Sci. 2013, 285, 865–873. [Google Scholar] [CrossRef]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, P.; Lv, M.; Han, G.; Wen, C.; Wang, Q.; Li, H.; Zhao, X. Solvothermal Synthesis of Hierarchical Colloidal Nanocrystal Assemblies of ZnFe2O4 and Their Application in Water Treatment. Materials 2016, 9, 806. https://doi.org/10.3390/ma9100806
Guo P, Lv M, Han G, Wen C, Wang Q, Li H, Zhao X. Solvothermal Synthesis of Hierarchical Colloidal Nanocrystal Assemblies of ZnFe2O4 and Their Application in Water Treatment. Materials. 2016; 9(10):806. https://doi.org/10.3390/ma9100806
Chicago/Turabian StyleGuo, Peizhi, Meng Lv, Guangting Han, Changna Wen, Qianbin Wang, Hongliang Li, and Xiusong Zhao. 2016. "Solvothermal Synthesis of Hierarchical Colloidal Nanocrystal Assemblies of ZnFe2O4 and Their Application in Water Treatment" Materials 9, no. 10: 806. https://doi.org/10.3390/ma9100806
APA StyleGuo, P., Lv, M., Han, G., Wen, C., Wang, Q., Li, H., & Zhao, X. (2016). Solvothermal Synthesis of Hierarchical Colloidal Nanocrystal Assemblies of ZnFe2O4 and Their Application in Water Treatment. Materials, 9(10), 806. https://doi.org/10.3390/ma9100806





