Development of Hydrogen Storage Tank Systems Based on Complex Metal Hydrides
Abstract
:1. Introduction
2. Thermolysis
2.1. Tank Developments for Thermal Hydrogen Evolution
| System Storage Parameter | Original 2010 Target | Revised 2010 Target | 2017 Target | Ultimate Target |
|---|---|---|---|---|
| Gravimetric capacity (kg·H2·kg−1 system) | 6% | 4.5% | 5.5% | 7.5% |
| Volumetric capacity (g·H2·L−1 system) | 45 | 28 | 40 | 70 |
| Operational cycle life | 1000 | 1000 | 1500 | 1500 |
| Filling time (min for 5 kg) | 3 | 4.2 | 3.3 | 2.5 |
| Min full flow rate (g·H2·s−1/kW) | 0.02 | 0.02 | 0.02 | 0.02 |
| Min delivery pressure at 85 °C PEM fuel cell (atm) | 8 | 5 | 4 | 3 |
| Fuel purity | 99.99% | 99.97% | 99.97% | 99.97% |
2.2. Modeling of Thermolysis Tank Systems
| Material | Reference | Step | g(α) | f(p, peq) | A (s−1) | Ea (kJ·mol−1) |
|---|---|---|---|---|---|---|
| NaAlH4 | [11] | 1des | 1st order | ln(peq/p) | 1.9 × 1011 | 85.6 |
| 2des | 1st order | ln(peq/p) | 2.9 × 1010 | 88.3 | ||
| 1abs | 2nd order | ln(p/peq) | 6.2 × 108 | 61.6 | ||
| 2abs | 1st order | ln(p/peq) | 1.0 × 108 | 56.2 | ||
| [12] | 1abs | JMA n = 1.4 | ln(p/peq)(p/p0)2.7 | 3.6 × 10−1 | 54.7 | |
| 1st order | ln(p/peq)(p/p0)1.5 | 1.9 × 103 | 62.9 | |||
| 2abs | JMA n = 1.4 | (p − peq)/peq | 2.0 × 1011 | 117.5 | ||
| 1st order | ((p − peq)/p)1.5 | 1.2 × 108 | 86.4 | |||
| [13] | 1des | 2nd order | (peq − p)/peq | 4.0 × 105 | 110 | |
| 2des | 1st order | (peq − p)/peq | 6.0 × 1012 | 110 | ||
| 1abs | 2nd order | (p − peq)/peq | 1.0 × 108 | 80 | ||
| 2abs | 1st order | (p − peq)/peq | 1.5 × 105 | 70 | ||
| [14] | 1abs | JMA n = 1.33 | (p − peq)/peq | 1.5 × 109 | 91.5 | |
| 2abs | JMA n = 1.33 | (p − peq)/peq | 2.3 × 108 | 91.7 | ||
| [15] | 1des | 0 order | ((peq − p)/peq)2 + 1.04((peq − p)/peq) | 5.4 × 101° | 105.8 | |
| 2des | 1st order | ((peq − p)/peq)2 + 0.46((peq − p)/peq) | 3.4 × 108 | 91.5 | ||
| Li2Mg(NH)2 | [16] | 1des | JMA n = 1.5 | ln(peq/p) | 2.3 × 1012 | 131.8 |
| 2des | 0 order | 1 − (0.001515/0.33wtmax)(p − 1.1) | 3.0 × 1015 | 161.4 | ||
| 1abs | 0 order | (p − peq)/peq | 2.7 × 1017 | 164.8 | ||
| 2abs | 1st order | (peq − p)/peq | 4.7 × 1014 | 147.8 |
2.3. Simulation of Thermolysis Tank Systems
2.3.1. Simulations on Sodium Alanate
2.3.2. Simulations on Other Complex Hydrides
2.4. Developed Complex Metal Hydride Based Tank Systems
2.4.1. Sodium Alanate Based Tank Systems
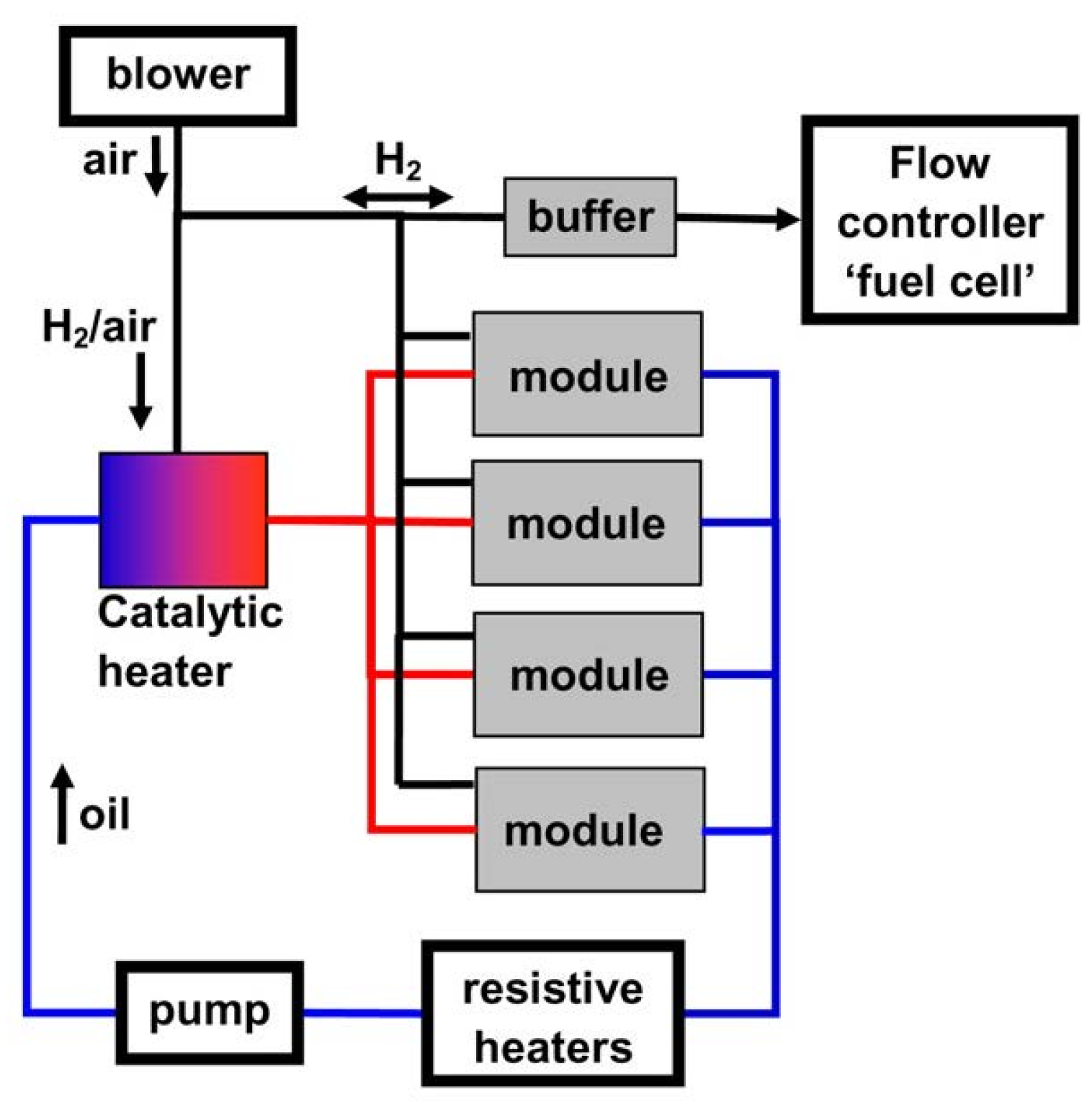


2.4.2. Metal Amide Based Tank Systems
2.4.3. Ammonia Borane and Alane Based Tank Systems
| Storage Material | Weight of Storage Material (kg) | Design | Capacity (wt. % H2) | T, P Conditions Charging/Discharging | Kinetic/Cycles | Purpose |
|---|---|---|---|---|---|---|
| NaAlH4 doped with 2 mol % (TiCl3-0.3AlCl3), 5 mol % carbon | 8 | Tubular reactor with porous sintered metal tube | 3.7 | charging: 125 °C, 10 MPa; discharging: 160–175 °C, 0.02–1 MPa | 1–10 activation cycles after 10 min 80% capacity achieved | Large scale and stationary applications [36]. |
| NaAlH4 doped with 2 mol % CeCl3 | 0.087 | Hydride bed reactor with flow-thru mode | 3.9 | charging: 130 °C, 10 MPa; discharging: 180 °C, 0.13 MPa | 36 experiments; decrease in permeability; increased thermal conductivity λeff = 0.67 W·m−1·K−1 | To investigate an operational principle, changes in heat transfer, permeability and reaction kinetics [39]. |
| NaAlH4, Al, 10 wt % ENG | 4 × 21.5 | Modular system of 12 tubular vessels | 3.2 in 10 min | charging: 120–150 °C, 5.52–6.89 MPa, oil temperature: 120–140 °C | 40 absorption/desorption cycles | To be refueled in 10 min and to deliver H2 up to 2.0 g·s−1 [27]. |
| NaAlH4 doped with 4 mol % TiCl3 | 2.7 | Stainless steel tank with double wounded helical coil heat exchanger | 2.24 | charging: 135 °C, 10 MPa; discharging: 120–180 °C, 0.1 MPa | 7 two or more hour desorption cycles coupled with FC, that supplied 165–240 W power | To couple with HT-PEM and use waste heat from HT-PEM for desorbing H2 from the tank [37]. |
| Na3AlH6 doped with 4 mol % TiCl3 | 0.213 | Al-alloy tank with bayonet heat exchanger | 1.7 | charging: 150–170 °C, 2.5 MPa; discharging: 177–180 °C, 0.65 MPa | 10 absorption/desorption cycles | To develop and test lightweight Al-alloy storage tank [38]. |
| Na3AlH6 doped with 4 mol % TiCl3, 8 mol % Al and 8 mol % activated carbon | 1.9 | Al-alloy tank with corrugated heat exchanger | 2.1 | charging: 160 °C, 2.5 MPa; discharging: 180 °C, 1.6 MPa | 31 absorption/desorption cycles | To develop the lightweight Al-tank that is produced by extrusion molding [42]. |
| NaAlH4 doped with 2 mol % (TiCl3-0.3AlCl3) and 5 mol % ENG | 4.4 | Ti-alloy tube-and-shell tank system | 4 | charging: 124 °C, 10 MPa; discharging: 120–170 °C, 9 MPa (constant flow) | 33 cycles, 120 min of absorption with restricted H2 flow of 245 Ln·min−1; 200 min desorption 3.7 L·min−1 | To improve gravimetric and volumetric capacity [43]. |
| Mg(NH2)2-2LiH-0.07KOH with 9 wt % ENG | 0.098 | Cylindrical lab-scale hydrogen storage tank with porous sintered metal tube as H2 supply | N/A | charging: 220 °C, 8 MPa; discharging: 220 °C at constant H2 flow rate of 0.6 L·min−1 | Desorption duration of 79.5 min at 0.6 L·min−1 H2 | To investigate influence of graphite content and compaction pressure on desorption properties [44]. |
| LiNH2-MgH2-LiBH4 3 wt % ZrCoH3 (in annulus) LaNi4.3Al0.4Mn0.3 (in core) | 0.6 | Tubular reactor, two materials separated by a gas permeable layer | N/A | charging: 165–170 °C up to 0.17 MPa; discharging at constant and periodic H2 mass flow | 10% of H2 desorbed in about 30 min; majority of H2 desorbed in about 1 h | To validate a model and study effects of the reactor concept on desorption performance [31]. |
3. Hydrolysis
3.1. Tank Developments for Hydrolysis Based Reactors
3.1.1. Flow Reactors
3.1.2. Batch Reactors
3.1.3. Reactor Modeling of Hydrolysis Tank Systems
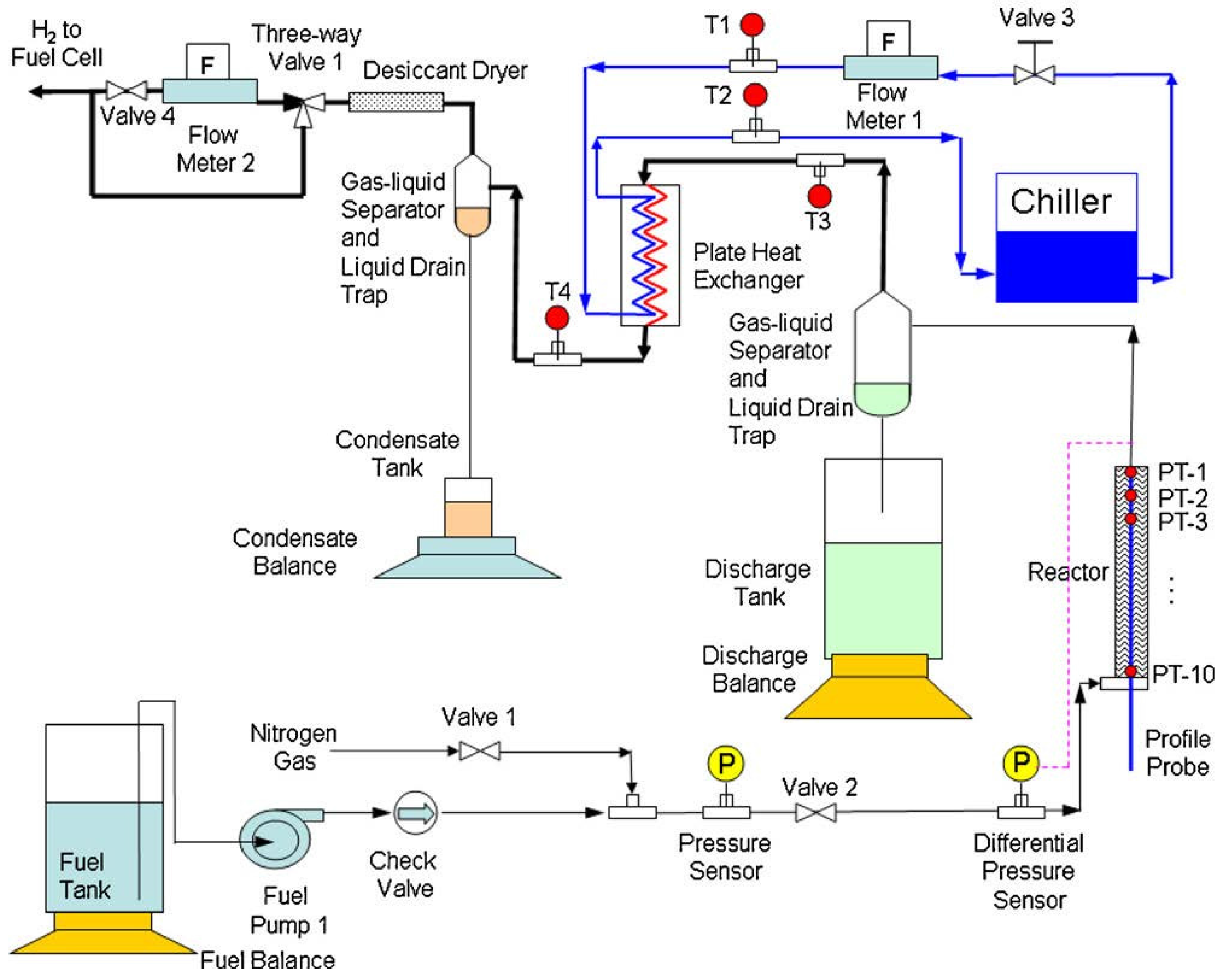
3.1.4. System Modeling for Hydrolysis Setups
3.2. Applications Using Sodium Borohydride
3.2.1. Small-Unmanned Aerial Vehicles
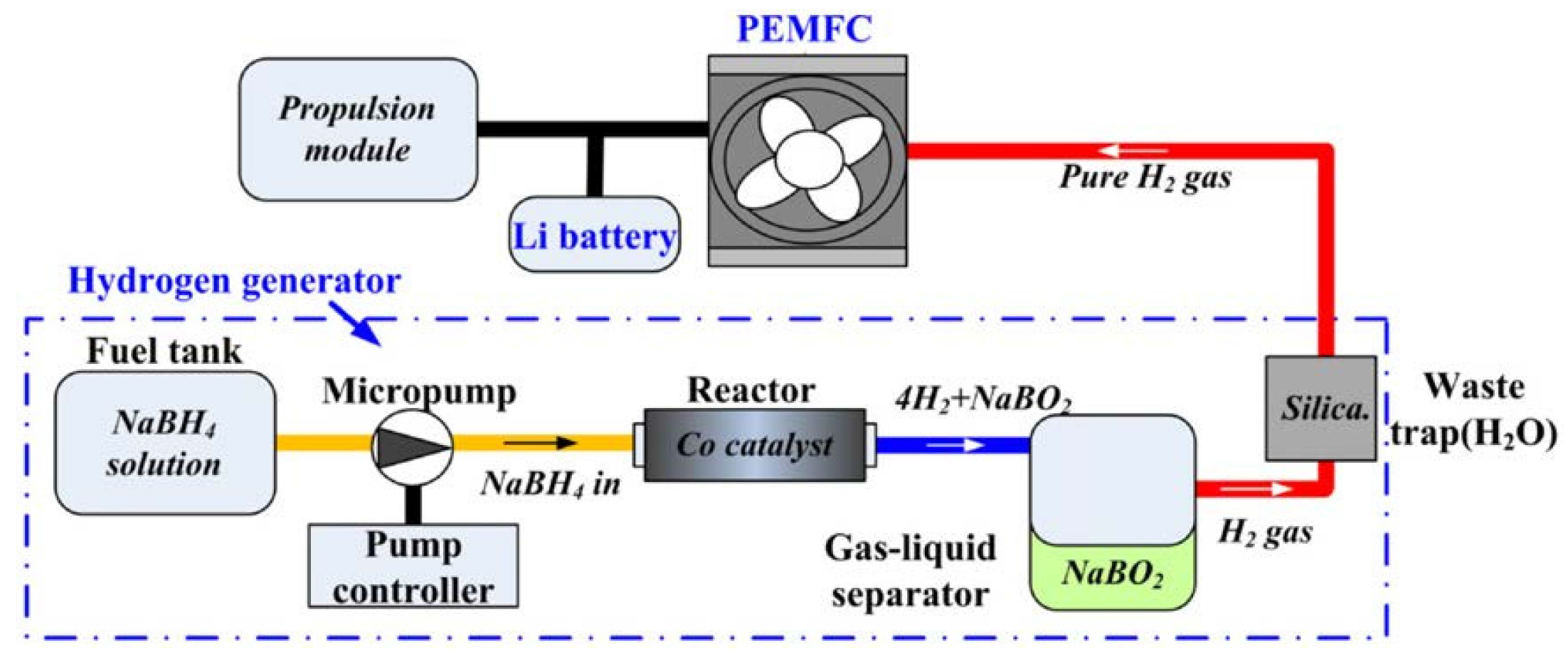
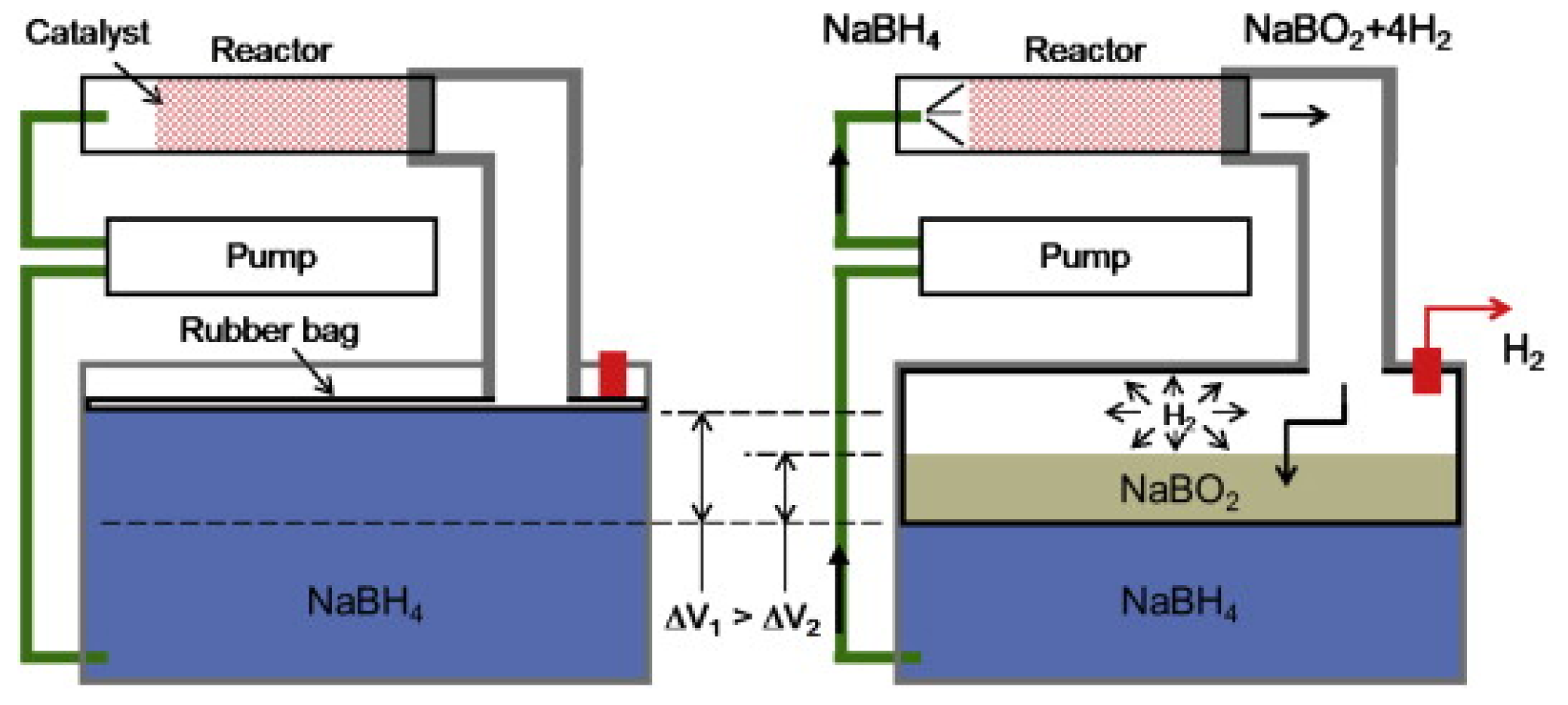
3.2.2. Small Portable Applications
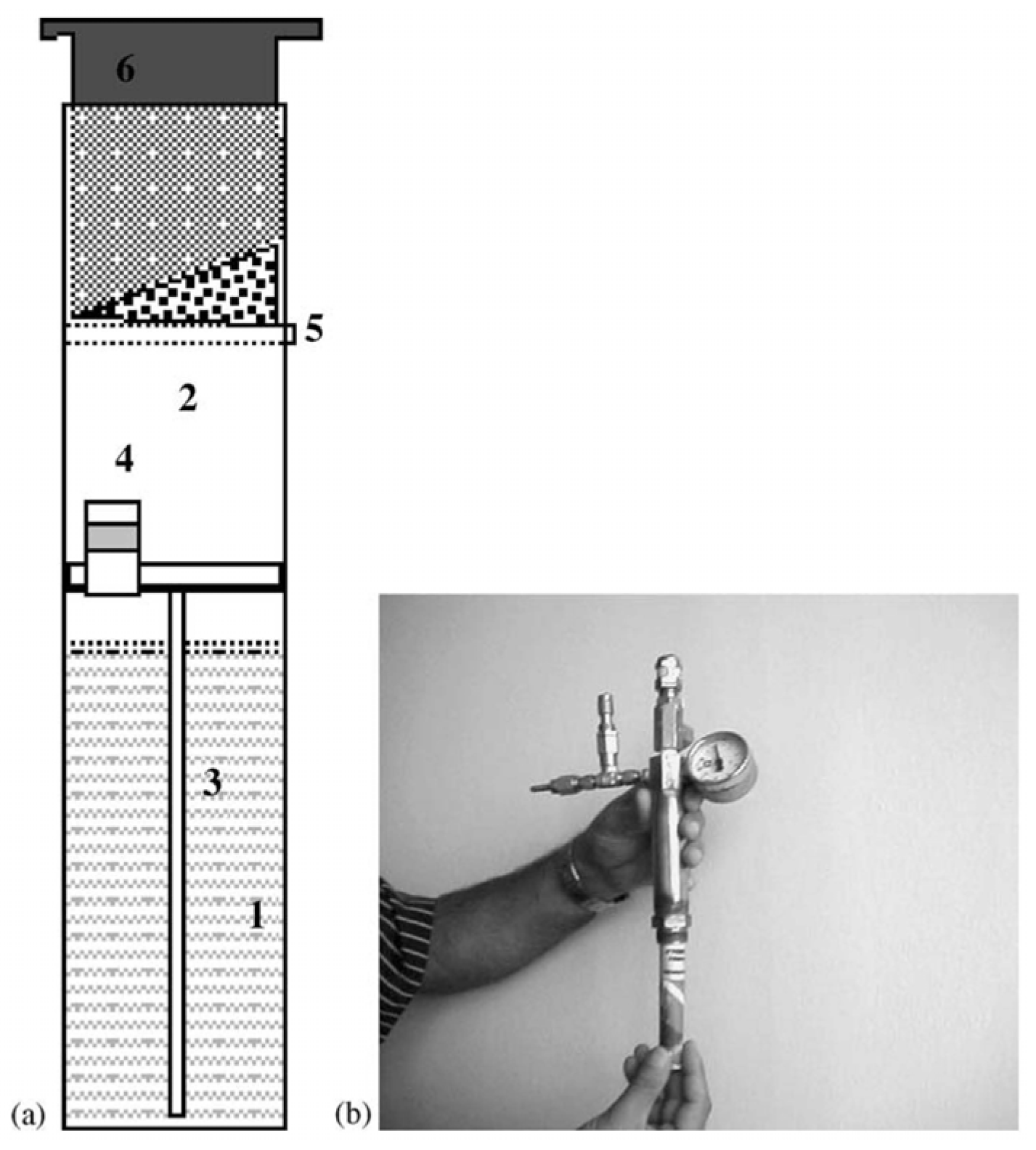
3.2.3. Submarine Applications
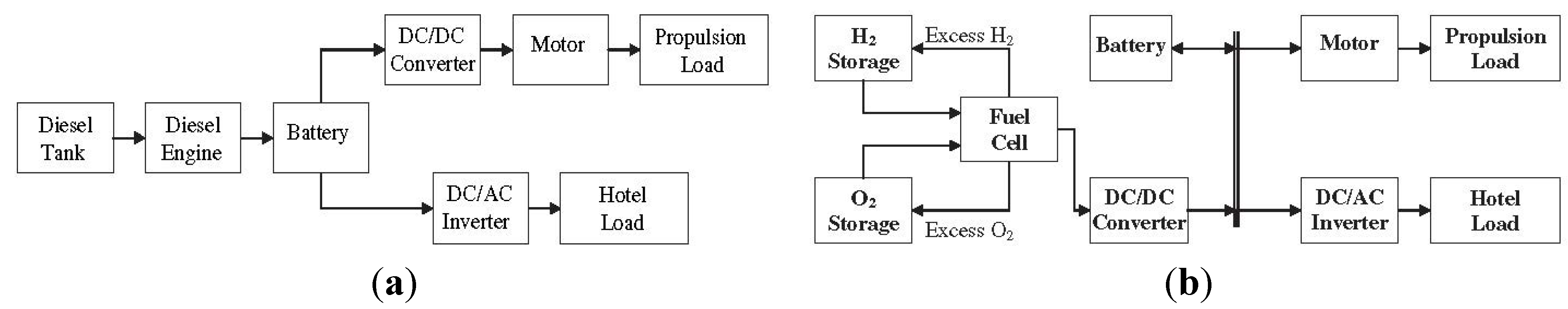
3.3. Recycling of NaBH4 Spent Fuel
3.4. Applications Using Ammonia Borane
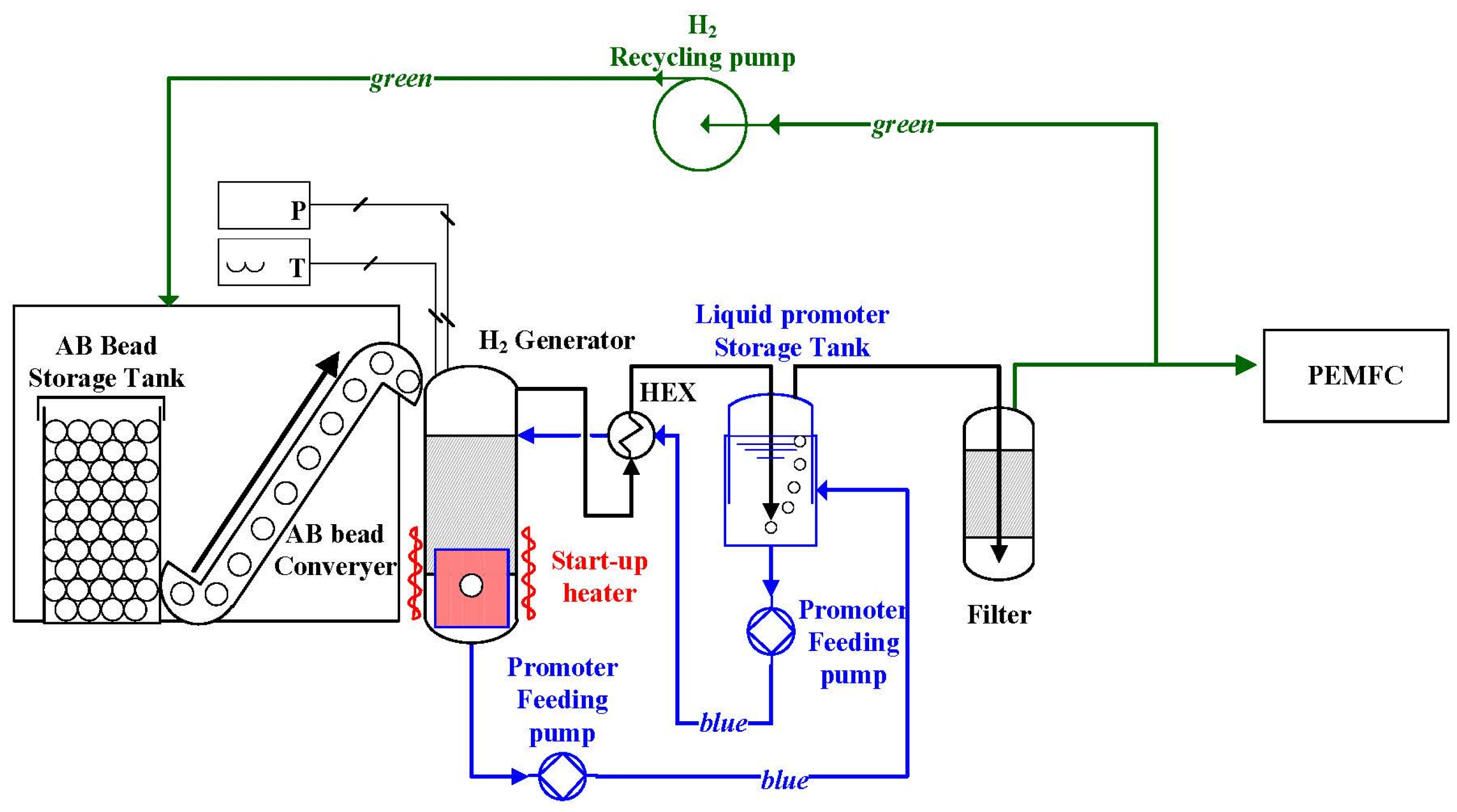
4. Summary and Outlook
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schlapbach, L.; Züttel, A. Hydrogen-storage materials for mobile applications. Nature 2001, 414, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Eberle, U.; Felderhoff, M.; Schüth, F. Chemical and physical solutions for hydrogen storage. Angew. Chem. Int. Ed. 2009, 48, 6608–6630. [Google Scholar] [CrossRef] [PubMed]
- Ley, M.B.; Jepsen, L.H.; Lee, Y.-S.; Cho, Y.W.; Bellosta von Colbe, J.M.; Dornheim, M.; Rokni, M.; Jensen, J.O.; Sloth, M.; Filinchuk, Y.; et al. Complex hydrides for hydrogen storage—New perspectives. Mater. Today 2014, 17, 122–128. [Google Scholar] [CrossRef]
- Bogdanović, B.; Schwickardi, M. Ti-doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage materials. J. Alloys Compd. 1997, 253–254, 1–9. [Google Scholar] [CrossRef]
- Rude, L.H.; Nielsen, T.K.; Ravnsbæk, D.B.; Bösenberg, U.; Ley, M.B.; Richter, B.; Arnbjerg, L.M.; Dornheim, M.; Filinchuk, Y.; Besenbacher, F.; et al. Tailoring properties of borohydrides for hydrogen storage: A review. Phys. Status Solidi A 2011, 208, 1754–1773. [Google Scholar] [CrossRef]
- Mosher, D.A.; Arsenault, S.; Tang, X.; Anton, D.L. Design, fabrication and testing of NaAlH4 based hydrogen storage systems. J. Alloys Compd. 2007, 446–447, 707–712. [Google Scholar] [CrossRef]
- Demirci, U.B. The hydrogen cycle with the hydrolysis of sodium borohydride: A statistical approach for highlighting the scientific/technical issues to prioritize in the field. Int. J. Hydrogen Energy 2015, 40, 2673–2691. [Google Scholar] [CrossRef]
- Technical system targets: Onboard hydrogen storage for light-duty fuel cell vehicles 2012. Available online: http://www1.eere.energy.gov/vehiclesandfuels/pdfs/program/hstt_roadmap_june2013.pdf (accessed on 25 August 2015).
- Lozano, G.A.; Bellosta von Colbe, J.M.; Klassen, T.; Dornheim, M. Transport phenomena versus intrinsic kinetics: Hydrogen sorption limiting sub-process in metal hydride beds. Int. J. Hydrogen Energy 2014, 39, 18952–18957. [Google Scholar] [CrossRef]
- Vyazovkin, S.; Burnham, A.K.; Criado, J.M.; Pérez-Maqueda, L.A.; Popescu, C.; Sbirrazzuoli, N. Ictac kinetics committee recommendations for performing kinetic computations on thermal analysis data. Thermochim. Acta 2011, 520, 1–19. [Google Scholar] [CrossRef]
- Luo, W.; Gross, K.J. A kinetics model of hydrogen absorption and desorption in Ti-doped NaAlH4. J. Alloys Compd. 2004, 385, 224–231. [Google Scholar] [CrossRef]
- Franzen, J. Modellierung und Simulation eines Wasserstoffspeichers auf der Basis von Natriumalanat; Hamburg, Techn. Univ.: Hamburg, Germany, 2009. [Google Scholar]
- Hardy, B.J. Geometry, heat removal and kinetic scoping models for hydrogen storage systems. Available online: http://www1.eere.energy.gov/hydrogenandfuelcells/pdfs/bruce_hardy_srnl-2007-0043_part1.pdf (accessed on 25 February 2015).
- Lozano, G.A.; Ranong, C.N.; Bellosta von Colbe, J.M.; Bormann, R.; Fieg, G.; Hapke, J.; Dornheim, M. Empirical kinetic model of sodium alanate reacting system (I). Hydrogen absorption. Int. J. Hydrogen Energy 2010, 35, 6763–6772. [Google Scholar] [CrossRef]
- Lozano, G.A.; Ranong, C.N.; Bellosta von Colbe, J.M.; Bormann, R.; Fieg, G.; Hapke, J.; Dornheim, M. Empirical kinetic model of sodium alanate reacting system (II). Hydrogen desorption. Int. J. Hydrogen Energy 2010, 35, 7539–7546. [Google Scholar] [CrossRef]
- Bürger, I.; Hu, J.J.; Vitillo, J.G.; Kalantzopoulos, G.N.; Deledda, S.; Fichtner, M.; Baricco, M.; Linder, M. Material properties and empirical rate equations for hydrogen sorption reactions in 2LiNH2–1.1MgH2–0.1LiBH4–3wt % ZrCoH3. Int. J. Hydrogen Energy 2014, 39, 8283–8292. [Google Scholar] [CrossRef]
- Na Ranong, C.; Höhne, M.; Franzen, J.; Hapke, J.; Fieg, G.; Dornheim, M.; Eigen, N.; Bellosta von Colbe, J.M.; Metz, O. Concept, design and manufacture of a prototype hydrogen storage tank based on sodium alanate. Chem. Eng. Technol. 2009, 32, 1154–1163. [Google Scholar] [CrossRef]
- Hardy, B.J.; Anton, D.L. Hierarchical methodology for modeling hydrogen storage systems. Part i: Scoping models. Int. J. Hydrogen Energy 2009, 34, 2269–2277. [Google Scholar] [CrossRef]
- Hardy, B.J.; Anton, D.L. Hierarchical methodology for modeling hydrogen storage systems. Part ii: Detailed models. Int. J. Hydrogen Energy 2009, 34, 29923004. [Google Scholar] [CrossRef]
- Dedrick, D.E.; Kanouff, M.P.; Larson, R.S.; Johnson, T.A.; Jorgensen, S.W. Heat and mass transport in metal hydride based hydrogen storage systems. In Proceeding of the ASME Summer Heat Transfer Conference, San Francisco, CA, USA, 19–23 July 2009; pp. 249–260.
- Mosher, D.A.; Tang, X.; Arsenault, S.; Laube, B.; Cao, M.; Brown, R. High density hydrogen storage system demonstration using NaAlH4 complex compound hydrides. Available online: http://www.hydrogen.energy.gov/pdfs/progress05/vi_a_2_anton.pdf (accessed on 22 February 2015).
- Pfeifer, P.; Wall, C.; Jensen, O.; Hahn, H.; Fichtner, M. Thermal coupling of a high temperature pem fuel cell with a complex hydride tank. Int. J. Hydrogen Energy 2009, 34, 3457–3466. [Google Scholar] [CrossRef]
- Bhouri, M.; Goyette, J.; Hardy, B.J.; Anton, D.L. Sensitivity study of alanate hydride storage system. Int. J. Hydrogen Energy 2011, 36, 621–633. [Google Scholar] [CrossRef]
- Bhouri, M.; Goyette, J.; Hardy, B.J.; Anton, D.L. Numerical modeling and performance evaluation of multi-tubular sodium alanate hydride finned reactor. Int. J. Hydrogen Energy 2012, 37, 1551–1567. [Google Scholar] [CrossRef]
- Raju, M.; Kumar, S. System simulation modeling and heat transfer in sodium alanate based hydrogen storage systems. Int. J. Hydrogen Energy 2011, 36, 1578–1591. [Google Scholar] [CrossRef]
- Lozano, G.A.; Ranong, C.N.; Bellosta von Colbe, J.M.; Bormann, R.; Hapke, J.; Fieg, G.; Klassen, T.; Dornheim, M. Optimization of hydrogen storage tubular tanks based on light weight hydrides. Int. J. Hydrogen Energy 2012, 37, 2825–2834. [Google Scholar] [CrossRef]
- Johnson, T.A.; Kanouff, M.P.; Dedrick, D.E.; Evans, G.H.; Jorgensen, S.W. Model-based design of an automotive-scale, metal hydride hydrogen storage system. Int. J. Hydrogen Energy 2012, 37, 2835–2849. [Google Scholar] [CrossRef]
- Devarakonda, M.; Brooks, K.; Rönnebro, E.; Rassat, S. Systems modeling, simulation and material operating requirements for chemical hydride based hydrogen storage. Int. J. Hydrogen Energy 2012, 37, 2779–2793. [Google Scholar] [CrossRef]
- Semelsberger, T.A.; Brooks, K.P. Chemical hydrogen storage material property guidelines for automotive applications. J. Power Sources 2015, 279, 593–609. [Google Scholar] [CrossRef]
- Bürger, I.; Komogowski, L.; Linder, M. Advanced reactor concept for complex hydrides: Hydrogen absorption from room temperature. Int. J. Hydrogen Energy 2014, 39, 7030–7041. [Google Scholar] [CrossRef]
- Bürger, I.; Luetto, C.; Linder, M. Advanced reactor concept for complex hydrides: Hydrogen desorption at fuel cell relevant boundary conditions. Int. J. Hydrogen Energy 2014, 39, 7346–7355. [Google Scholar] [CrossRef]
- Bhouri, M.; Bürger, I.; Linder, M. Optimization of hydrogen charging process parameters for an advanced complex hydride reactor concept. Int. J. Hydrogen Energy 2014, 39, 17726–17739. [Google Scholar] [CrossRef]
- Van Hassel, B.A.; Mosher, D.; Pasini, J.M.; Gorbounov, M.; Holowczak, J.; Tang, X.; Brown, R.; Laube, B.; Pryor, L. Engineering improvement of NaAlH4 system. Int. J. Hydrogen Energy 2012, 37, 2756–2766. [Google Scholar] [CrossRef]
- Sulic, M.; Cai, M.; Kumar, S. Cycling and engineering properties of highly compacted sodium alanate pellets. Int. J. Hydrogen Energy 2012, 37, 15187–15195. [Google Scholar] [CrossRef]
- Brooks, K.P.; Semelsberger, T.A.; Simmons, K.L.; van Hassel, B. Slurry-based chemical hydrogen storage systems for automotive fuel cell applications. J. Power Sources 2014, 268, 950–959. [Google Scholar] [CrossRef]
- Bellosta von Colbe, J.M.; Metz, O.; Lozano, G.A.; Pranzas, P.K.; Schmitz, H.W.; Beckmann, F.; Schreyer, A.; Klassen, T.; Dornheim, M. Behavior of scaled-up sodium alanate hydrogen storage tanks during sorption. Int. J. Hydrogen Energy 2012, 37, 2807–2811. [Google Scholar] [CrossRef]
- Urbanczyk, R.; Peil, S.; Bathen, D.; Heßke, C.; Burfeind, J.; Hauschild, K.; Felderhoff, M.; Schüth, F. HT-PEM fuel cell system with integrated complex metal hydride storage tank. Fuel Cells 2011, 11, 911–920. [Google Scholar] [CrossRef]
- Urbanczyk, R.; Peinecke, K.; Felderhoff, M.; Hauschild, K.; Kersten, W.; Peil, S.; Bathen, D. Aluminium alloy based hydrogen storage tank operated with sodium aluminium hexahydride Na3AlH6. Int. J. Hydrogen Energy 2014, 39, 17118–17128. [Google Scholar] [CrossRef]
- Utz, I.; Linder, M.; Schmidt, N.; Hu, J.J.; Fichtner, M.; Wörner, A. Experimental study of powder bed behavior of sodium alanate in a lab-scale H2 storage tank with flow-through mode. Int. J. Hydrogen Energy 2012, 37, 7645–7653. [Google Scholar] [CrossRef]
- Yan, M.-Y.; Sun, F.; Liu, X.-P.; Ye, J.-H. Effects of compaction pressure and graphite content on hydrogen storage properties of Mg(NH2)2–2LiH hydride. Int. J. Hydrogen Energy 2014, 39, 19656–19661. [Google Scholar] [CrossRef]
- Eigen, N.; Keller, C.; Dornheim, M.; Klassen, T.; Bormann, R. Industrial production of light metal hydrides for hydrogen storage. Scr. Mater. 2007, 56, 847–851. [Google Scholar] [CrossRef]
- Urbanczyk, R.; Peinecke, K.; Meggouh, M.; Felderhoff, M.; Peil, S.; Bathen, D. Solid hydrogen storage realized by doped Na3AlH6 in kg scale charged and operated in an Al alloy tank. 2015; to be published. [Google Scholar]
- Bellosta von Colbe, J.M.; Lozano, G.; Metz, O.; Bücherl, T.; Bormann, R.; Klassen, T.; Dornheim, M. Design, sorption behaviour and energy management in a sodium alanate-based lightweight hydrogen storage tank. Int. J. Hydrogen Energy 2015, 40, 2984–2988. [Google Scholar] [CrossRef]
- Yan, M.-Y.; Sun, F.; Liu, X.-P.; Ye, J.-H.; Wang, S.-M.; Jiang, L.-J. Effects of graphite content and compaction pressure on hydrogen desorption properties of Mg(NH2)2–2LiH based tank. J. Alloys Compd. 2015, 628, 63–67. [Google Scholar] [CrossRef]
- Ismail, I.M.K.; Hawkins, T. Kinetics of thermal decomposition of aluminium hydride: I-non-isothermal decomposition under vacuum and in inert atmosphere (argon). Thermochim. Acta 2005, 439, 32–43. [Google Scholar] [CrossRef]
- Nakagawa, T.; Burrell, A.K.; Del Sesto, R.E.; Janicke, M.T.; Nekimken, A.L.; Purdy, G.M.; Paik, B.; Zhong, R.-Q.; Semelsberger, T.A.; Davis, B.L. Physical, structural, and dehydrogenation properties of ammonia borane in ionic liquids. RSC Adv. 2014, 4, 21681–21687. [Google Scholar] [CrossRef]
- Sankir, M.; Serin, R.B.; Semiz, L.; Sankir, N.D. Unusual behavior of dynamic hydrogen generation from sodium borohydride. Int. J. Hydrogen Energy 2014, 39, 2608–2613. [Google Scholar] [CrossRef]
- Amendola, S.C.; Sharp-Goldman, S.L.; Janjua, M.S.; Kelly, M.T.; Petillo, P.J.; Binder, M. An ultrasafe hydrogen generator: Aqueous, alkaline borohydride solutions and Ru catalyst. J. Power Sources 2000, 85, 186–189. [Google Scholar] [CrossRef]
- Liu, B.H.; Li, Z.P. A review: Hydrogen generation from borohydride hydrolysis reaction. J. Power Sources 2009, 187, 527–534. [Google Scholar] [CrossRef]
- Demirci, U.B.; Akdim, O.; Andrieux, J.; Hannauer, J.; Chamoun, R.; Miele, P. Sodium borohydride hydrolysis as hydrogen generator: Issues, state of the art and applicability upstream from a fuel cell. Fuel Cells 2010, 10, 335–350. [Google Scholar] [CrossRef]
- Schlesinger, H.I.; Brown, H.C.; Hoekstra, H.R.; Rapp, L.R. Reactions of diborane with alkali metal hydrides and their addition compounds. New syntheses of borohydrides. Sodium and potassium borohydrides. J. Am. Chem. Soc. 1953, 75, 199–204. [Google Scholar] [CrossRef]
- Demirci, U.B.; Miele, P. Sodium borohydride versus ammonia borane, in hydrogen storage and direct fuel cell applications. Energ. Environ. Sci. 2009, 2, 627–637. [Google Scholar] [CrossRef]
- Weidenthaler, C.; Felderhoff, M. Complex hydrides. In Handbook of Hydrogen Storage; Hirscher, M., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2010. [Google Scholar]
- Metin, Ö.; Mazumder, V.; Özkar, S.; Sun, S. Monodisperse nickel nanoparticles and their catalysis in hydrolytic dehydrogenation of ammonia borane. J. Am. Chem. Soc. 2010, 132, 1468–1469. [Google Scholar]
- Sun, D.; Mazumder, V.; Metin, Ö.; Sun, S. Catalytic hydrolysis of ammonia borane via cobalt palladium nanoparticles. ACS Nano 2011, 5, 6458–6464. [Google Scholar] [CrossRef] [PubMed]
- Demirci, U.B.; Miele, P. Cobalt-based catalysts for the hydrolysis of NaBH4 and NH3BH3. Phys. Chem. Chem. Phys. 2014, 16, 6872–6885. [Google Scholar] [CrossRef] [PubMed]
- Aiello, R.; Sharp, J.H.; Matthews, M.A. Production of hydrogen from chemical hydrides via hydrolysis with steam. Int. J. Hydrogen Energy 1999, 24, 1123–1130. [Google Scholar] [CrossRef]
- Chen, J.; Fu, H.; Xiong, Y.; Xu, J.; Zheng, J.; Li, X. MgCl2 promoted hydrolysis of MgH2 nanoparticles for highly efficient H2 generation. Nano Energy 2014, 10, 337–343. [Google Scholar] [CrossRef]
- Kojima, Y.; Kawai, Y.; Kimbara, M.; Nakanishi, H.; Matsumoto, S. Hydrogen generation by hydrolysis reaction of lithium borohydride. Int. J. Hydrogen Energy 2004, 29, 1213–1217. [Google Scholar] [CrossRef]
- Kojima, Y.; Suzuki, K.-I.; Kawai, Y. Hydrogen generation by hydrolysis reaction of magnesium hydride. J. Mater. Sci. 2004, 39, 2227–2229. [Google Scholar] [CrossRef]
- Kong, V.; Foulkes, F.; Kirk, D.; Hinatsu, J. Development of hydrogen storage for fuel cell generators I: Hydrogen generation using hydrolysis hydrides. Int. J. Hydrogen Energy 1999, 24, 665–675. [Google Scholar] [CrossRef]
- Kong, V.; Kirk, D.; Foulkes, F.; Hinatsu, J. Development of hydrogen storage for fuel cell generators II: Utilization of calcium hydride and lithium hydride. Int. J. Hydrogen Energy 2003, 28, 205–214. [Google Scholar] [CrossRef]
- Laversenne, L.; Goutaudier, C.; Chiriac, R.; Sigala, C.; Bonnetot, B. Hydrogen storage in borohydrides comparison of hydrolysis conditions of LiBH4, NaBH4 and KBH4. J. Therm. Anal. Calorim. 2008, 94, 785–790. [Google Scholar] [CrossRef]
- Ferreira, M.J.F.; Coelho, F.; Rangel, C.M.; Pinto, A.M.F.R. Batch sodium borohydride hydrolysis systems: Effect of sudden valve opening on hydrogen generation rate. Int. J. Hydrogen Energy 2012, 37, 1947–1953. [Google Scholar] [CrossRef]
- Ferreira, M.J.F.; Fernandes, V.R.; Gales, L.; Rangel, C.M.; Pinto, A.M.F.R. Effects of the addition of an organic polymer on the hydrolysis of sodium tetrahydroborate in batch reactors. Int. J. Hydrogen Energy 2010, 35, 11456–11469. [Google Scholar] [CrossRef]
- Pinto, A.; Falcao, D.; Silva, R.; Rangel, C. Hydrogen generation and storage from hydrolysis of sodium borohydride in batch reactors. Int. J. Hydrogen Energy 2006, 31, 1341–1347. [Google Scholar] [CrossRef]
- Pinto, A.M.F.R.; Ferreira, M.J.F.; Fernandes, V.R.; Rangel, C.M. Durability and reutilization capabilities of a Ni–Ru catalyst for the hydrolysis of sodium borohydride in batch reactors. Catal. Today 2011, 170, 40–49. [Google Scholar] [CrossRef]
- Zhang, Q.; Smith, G.; Wu, Y. Catalytic hydrolysis of sodium borohydride in an integrated reactor for hydrogen generation. Int. J. Hydrogen Energy 2007, 32, 4731–4735. [Google Scholar] [CrossRef]
- Zhang, Q.; Smith, G.; Wu, Y.; Mohring, R. Catalytic hydrolysis of sodium borohydride in an auto-thermal fixed-bed reactor. Int. J. Hydrogen Energy 2006, 31, 961–965. [Google Scholar] [CrossRef]
- Ferreira, M.J.F.; Rangel, C.M.; Pinto, A.M.F.R. Water handling challenge on hydrolysis of sodium borohydride in batch reactors. Int. J. Hydrogen Energy 2012, 37, 6985–6994. [Google Scholar] [CrossRef]
- Beaird, A.M.; Davis, T.A.; Matthews, M.A. Deliquescence in the hydrolysis of sodium borohydride by water vapor. Ind. Eng. Chem. Res. 2010, 49, 9596–9599. [Google Scholar] [CrossRef]
- Yu, L.; Matthews, M.A. A reactor model for hydrogen generation from sodium borohydride and water vapor. Int. J. Hydrogen Energy 2014, 39, 3830–3836. [Google Scholar] [CrossRef]
- Gervasio, D.; Tasic, S.; Zenhausern, F. Room temperature micro-hydrogen-generator. J. Power Sources 2005, 149, 15–21. [Google Scholar] [CrossRef]
- Amendola, S.C.; Binder, M.; Sharp-Goldman, S.L.; Kelly, M.T.; Petillo, P.J. System for hydrogen generation. US Patent 2003. [Google Scholar]
- Pozio, A.; Defrancesco, M.; Monteleone, G.; Oronzio, R.; Galli, S.; Dangelo, C.; Marrucci, M. Apparatus for the production of hydrogen from sodium borohydride in alkaline solution. Int. J. Hydrogen Energy 2008, 33, 51–56. [Google Scholar] [CrossRef]
- Oronzio, R.; Monteleone, G.; Pozio, A.; De Francesco, M.; Galli, S. New reactor design for catalytic sodium borohydride hydrolysis. Int. J. Hydrogen Energy 2009, 34, 4555–4560. [Google Scholar] [CrossRef]
- Galli, S.; De Francesco, M.; Monteleone, G.; Oronzio, R.; Pozio, A. Development of a compact hydrogen generator from sodium borohydride. Int. J. Hydrogen Energy 2010, 35, 7344–7349. [Google Scholar] [CrossRef]
- Li, H.-Y.; Chen, Y.-T.; Lu, M.-T.; Lai, Y.-H.; Yang, J.-T. Design and testing of a novel catalytic reactor to generate hydrogen. Int. J. Hydrogen Energy 2014, 39, 11945–11954. [Google Scholar] [CrossRef]
- Kim, T. Hydrogen generation from sodium borohydride using microreactor for micro fuel cells. Int. J. Hydrogen Energy 2011, 36, 1404–1410. [Google Scholar] [CrossRef]
- Kim, T. Fully-integrated micro PEM fuel cell system with NaBH4 hydrogen generator. Int. J. Hydrogen Energy 2012, 37, 2440–2446. [Google Scholar] [CrossRef]
- Zhu, L.; Meng, D.D.; Kroodsma, N.; Yeom, J.; Shannon, M.A. An integrated microfluidic self-regulating and self-circulating hydrogen generator for fuel cells. In Proceedings of International Solid-State Sensors, Actuators and Microsystems Conference, Denver, CO, USA, 21–25 June 2009; pp. 652–655.
- Zhu, L.; Kroodsma, N.; Yeom, J.; Haan, J.L.; Shannon, M.A.; Meng, D.D. An on-demand microfluidic hydrogen generator with self-regulated gas generation and self-circulated reactant exchange with a rechargeable reservoir. Microfluid Nanofluidics 2011, 11, 569–578. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, Y.; Gore, J.P.; Fisher, T.S. 1 kWe sodium borohydride hydrogen generation system: Part I: Experimental study. J. Power Sources 2007, 165, 844–853. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, Y.; Gore, J.P.; Mudawar, I.; Fisher, T.S. 1 kWe sodium borohydride hydrogen generation system: Part II: Reactor modeling. J. Power Sources 2007, 170, 150–159. [Google Scholar] [CrossRef]
- Sousa, T.; Fernandes, V.R.; Pinto, P.J.R.; Slavkov, Y.; Bosukov, L.; Rangel, C.M. A sodium borohydride hydrogen generation reactor for stationary applications: Experimental and reactor simulation studies. Chem. Eng. Sci. 2012, 84, 70–79. [Google Scholar] [CrossRef]
- Sousa, T.; Rangel, C.M. A dynamic two phase flow model for a pilot scale sodium borohydride hydrogen generation reactor. Int. J. Hydrogen Energy 2014, 39, 5291–5300. [Google Scholar] [CrossRef]
- Pinto, P.J.R.; Sousa, T.; Fernandes, V.R.; Pinto, A.M.F.R.; Rangel, C.M. Simulation of a stand-alone residential PEMFC power system with sodium borohydride as hydrogen source. Int. J. Electr. Power Energy Syst. 2013, 49, 57–65. [Google Scholar] [CrossRef]
- Lee, C.J.; Kim, T. Hydrogen supply system employing direct decomposition of solid-state NaBH4. Int. J. Hydrogen Energy 2015, 40, 2274–2282. [Google Scholar] [CrossRef]
- Ma, J.; Choudhury, N.A.; Sahai, Y. A comprehensive review of direct borohydride fuel cells. Renew. Sustain. Energy Rev. 2010, 14, 183–199. [Google Scholar] [CrossRef]
- Merino-Jiménez, I.; de León Ponce, C.; Shah, A.A.; Walsh, F.C. Developments in direct borohydride fuel cells and remaining challenges. J. Power Sources 2012, 219, 339–357. [Google Scholar] [CrossRef]
- Jamard, R.; Salomon, J.; Martinent-Beaumont, A.; Coutanceau, C. Life time test in direct borohydride fuel cell system. J. Power Sources 2009, 193, 779–787. [Google Scholar] [CrossRef]
- Kim, T.; Kwon, S. Design and development of a fuel cell-powered small unmanned aircraft. Int. J. Hydrogen Energy 2012, 37, 615–622. [Google Scholar] [CrossRef]
- Bradley, T.H.; Moffitt, B.A.; Mavris, D.N.; Parekh, D.E. Development and experimental characterization of a fuel cell powered aircraft. J. Power Sources 2007, 171, 793–801. [Google Scholar] [CrossRef]
- McConnell, V.P. Military UAVs claiming the skies with fuel cell power. Fuel Cells Bull. 2007, 2007, 12–15. [Google Scholar] [CrossRef]
- Stroman, R.O.; Jackson, G.S. Modeling the performance of an ideal NaBH4–H2O2 direct borohydride fuel cell. J. Power Sources 2014, 247, 756–769. [Google Scholar] [CrossRef]
- Stroman, R.O.; Jackson, G.S.; Garsany, Y.; Swider-Lyons, K. A calibrated hydrogen-peroxide direct-borohydride fuel cell model. J. Power Sources 2014, 271, 421–430. [Google Scholar] [CrossRef]
- Stroman, R.O.; Schuette, M.W.; Swider-Lyons, K.; Rodgers, J.A.; Edwards, D.J. Liquid hydrogen fuel system design and demonstration in a small long endurance air vehicle. Int. J. Hydrogen Energy 2014, 39, 11279–11290. [Google Scholar] [CrossRef]
- Kim, K.; Kim, T.; Lee, K.; Kwon, S. Fuel cell system with sodium borohydride as hydrogen source for unmanned aerial vehicles. J. Power Sources 2011, 196, 9069–9075. [Google Scholar] [CrossRef]
- AeroVironment. Aerovironment Puma small UAS achieves record flight of over nine hours using fuel cell battery hybrid system. Available online: https://www.avinc.com/public-safety/news-events/article/aerovironment_puma_small_uas_achieves_record_flight (accessed on 25 August 2015).
- Kim, T. NaBH4 (sodium borohydride) hydrogen generator with a volume-exchange fuel tank for small unmanned aerial vehicles powered by a PEM (proton exchange membrane) fuel cell. Energy 2014, 69, 721–727. [Google Scholar] [CrossRef]
- Liu, J.G.; Zhao, T.S.; Chen, R.; Wong, C.W. The effect of methanol concentration on the performance of a passive DMFC. Electrochem. Commun. 2005, 7, 288–294. [Google Scholar] [CrossRef]
- Akiyama, K.; Matsumoto, S.; Miyasaka, A.; Shodai, T. An air-breathing single cell small proton exchange membrane fuel cell system with AB5-type metal hydride and an ultra-low voltage input boost converter. J. Power Sources 2009, 186, 37–44. [Google Scholar] [CrossRef]
- Han, H.S.; Cho, C.; Kim, S.Y.; Hyun, J.M. Performance evaluation of a polymer electrolyte membrane fuel cell system for powering portable freezer. Appl. Energy 2013, 105, 125–137. [Google Scholar] [CrossRef]
- Kim, S.H.; Miesse, C.M.; Lee, H.B.; Chang, I.W.; Hwang, Y.S.; Jang, J.H.; Cha, S.W. Ultra compact direct hydrogen fuel cell prototype using a metal hydride hydrogen storage tank for a mobile phone. Appl. Energy 2014, 134, 382–391. [Google Scholar] [CrossRef]
- Prosini, P.P.; Gislon, P. A hydrogen refill for cellular phone. J. Power Sources 2006, 161, 290–293. [Google Scholar] [CrossRef]
- McConnell, V.P. Now, voyager? The increasing marine use of fuel cells. Fuel Cells Bull. 2010, 2010, 12–17. [Google Scholar] [CrossRef]
- Ghosh, P.C.; Vasudeva, U. Analysis of 3000 t class submarines equipped with polymer electrolyte fuel cells. Energy 2011, 36, 3138–3147. [Google Scholar] [CrossRef]
- Psoma, A.; Sattler, G. Fuel cell systems for submarines: From the first idea to serial production. J. Power Sources 2002, 106, 381–383. [Google Scholar] [CrossRef]
- Lakeman, J.B.; Rose, A.; Pointon, K.D.; Browning, D.J.; Lovell, K.V.; Waring, S.C.; Horsfall, J.A. The direct borohydride fuel cell for uuv propulsion power. J. Power Sources 2006, 162, 765–772. [Google Scholar] [CrossRef]
- Swider-Lyons, K.E.; Carlin, R.T.; Rosenfeld, R.L.; Nowak, R.J. Technical issues and opportunities for fuel cell development for autonomous underwater vehicles. In Proceedings of the 2002 Workshop on Autonomous Underwater Vehicles, San Antonio, TX, USA, 20–21 June 2002; pp. 61–64.
- Luo, N.; Miley, G.H.; Kim, K.-J.; Burton, R.; Huang, X. NaBH4/H2O2 fuel cells for air independent power systems. J. Power Sources 2008, 185, 685–690. [Google Scholar] [CrossRef]
- Santos, D.M.F.; Sequeira, C.A.C. Sodium borohydride as a fuel for the future. Renew. Sustain. Energy Rev. 2011, 15, 3980–4001. [Google Scholar] [CrossRef]
- Miley, G.H.; Kim, K.J.; Luo, N.; Shrestha, P.J. Advanced NaBH4/H2O2 fuel cell for space applications. AIP Conf. Proc. 2009, 1103, 157–163. [Google Scholar]
- Protonex. Underwater applications. Available online: http://www.protonex.com/solutions/underwater-applications/ (accessed on 25 August 2015).
- Kojima, Y.; Haga, T. Recycling process of sodium metaborate to sodium borohydride. Int. J. Hydrogen Energy 2003, 28, 989–993. [Google Scholar] [CrossRef]
- Seo, J.-E.; Kim, Y.; Kim, Y.; Kim, K.; Lee, J.H.; Lee, D.H.; Kim, Y.; Shin, S.J.; Kim, D.-M.; Kim, S.-Y.; et al. Portable ammonia-borane-based H2 power-pack for unmanned aerial vehicles. J. Power Sources 2014, 254, 329–337. [Google Scholar] [CrossRef]
- Nakagawa, T.; Paik, B.; Davis, B.; Semelsberger, T.; Baker, T.; Sneddon, L. Annual merit review, May 17th Los Alamos National Laboratory la-ur 12-20427. Available online: http://www.hydrogen.energy.gov/pdfs/review12/st040_davis_2012_o.pdf (accessed on 21 August 2015).
- Semelsberger, T.A.; Davis, B.L.; Rekken, B.D.; Paik, B.; Tafoya, J.I. Chemical hydrogen rate modeling, validation, and system demonstration. In Proceedings of DOE Fuel Cell Technologies Program Annual Merit Review, EERE: Hydrogen, Fuel Cells and Infrastructure Technologies Program, Washington, DC, USA, 13–17 May 2013.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ley, M.B.; Meggouh, M.; Moury, R.; Peinecke, K.; Felderhoff, M. Development of Hydrogen Storage Tank Systems Based on Complex Metal Hydrides. Materials 2015, 8, 5891-5921. https://doi.org/10.3390/ma8095280
Ley MB, Meggouh M, Moury R, Peinecke K, Felderhoff M. Development of Hydrogen Storage Tank Systems Based on Complex Metal Hydrides. Materials. 2015; 8(9):5891-5921. https://doi.org/10.3390/ma8095280
Chicago/Turabian StyleLey, Morten B., Mariem Meggouh, Romain Moury, Kateryna Peinecke, and Michael Felderhoff. 2015. "Development of Hydrogen Storage Tank Systems Based on Complex Metal Hydrides" Materials 8, no. 9: 5891-5921. https://doi.org/10.3390/ma8095280
APA StyleLey, M. B., Meggouh, M., Moury, R., Peinecke, K., & Felderhoff, M. (2015). Development of Hydrogen Storage Tank Systems Based on Complex Metal Hydrides. Materials, 8(9), 5891-5921. https://doi.org/10.3390/ma8095280






