Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials
Abstract
:1. Introduction
2. Origin and Role of Macrophages
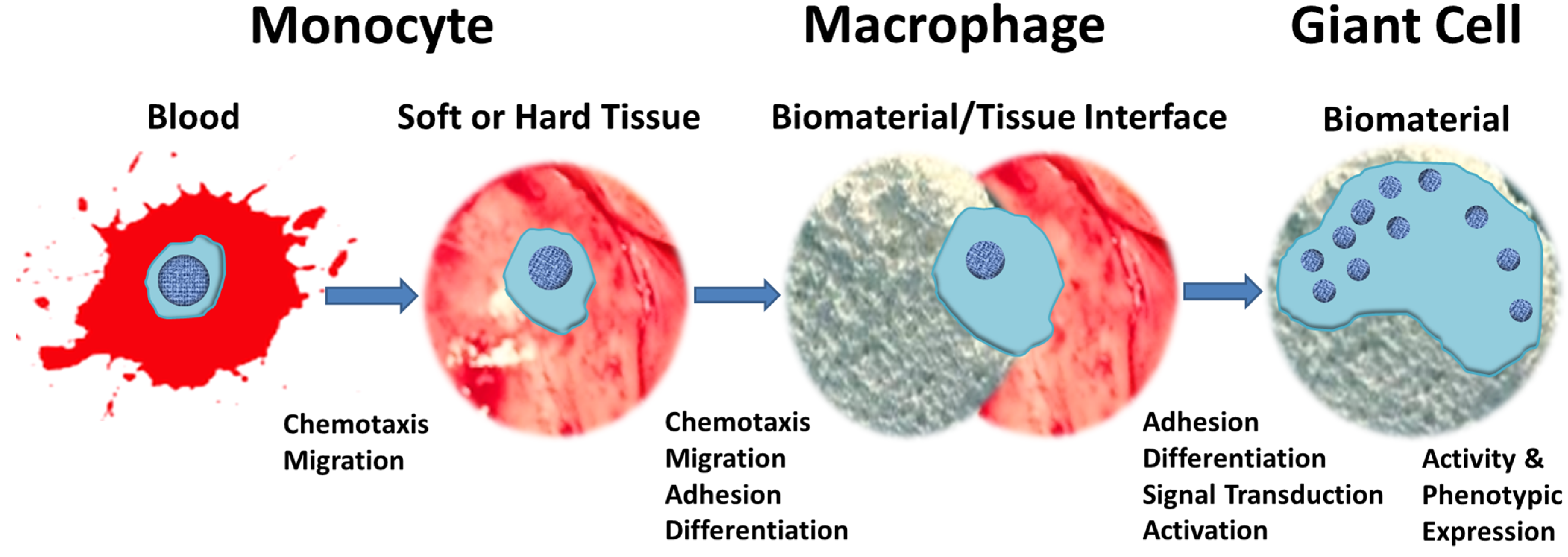
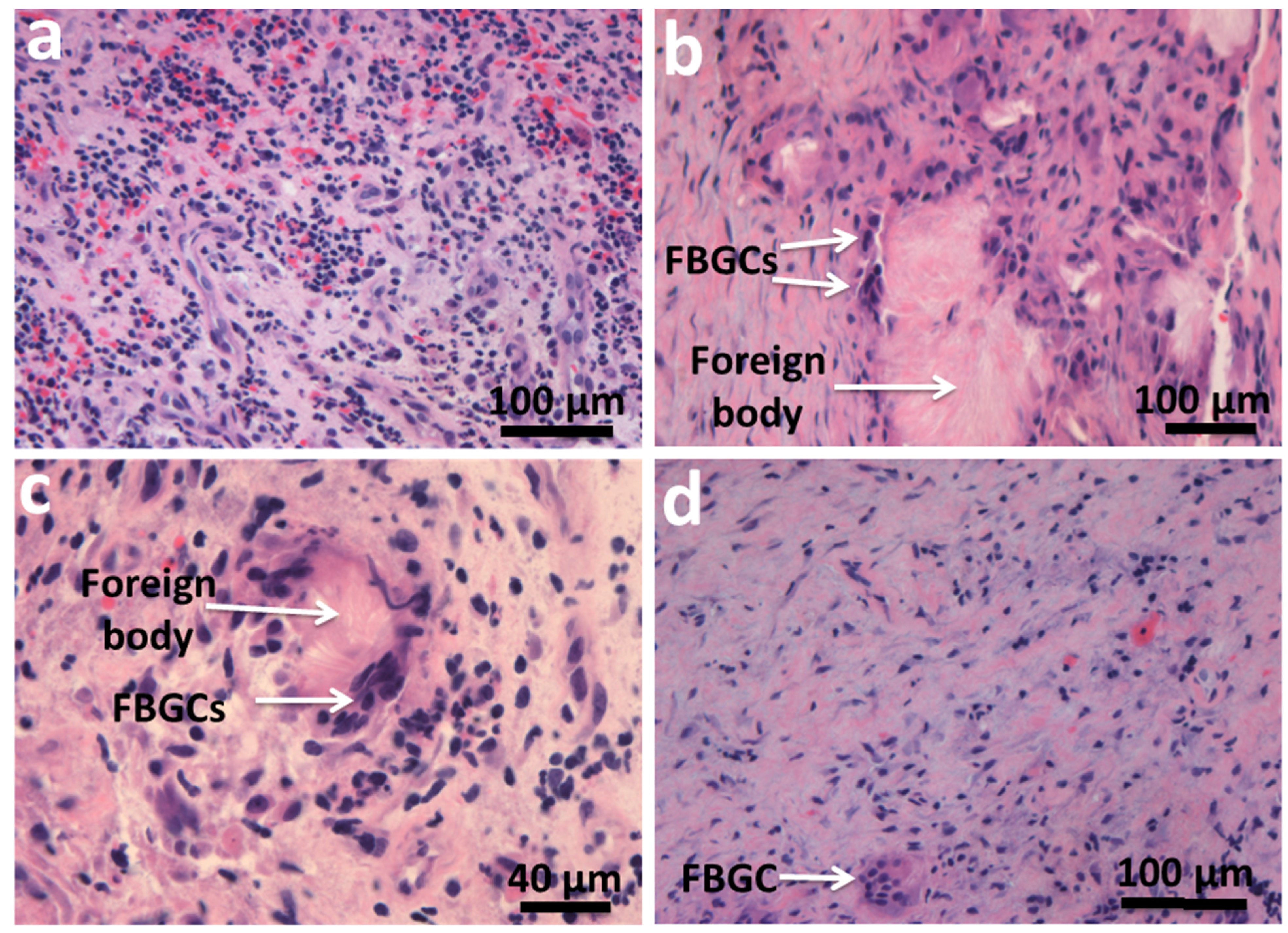
3. Fusion Mechanism for the Formation of MGCs
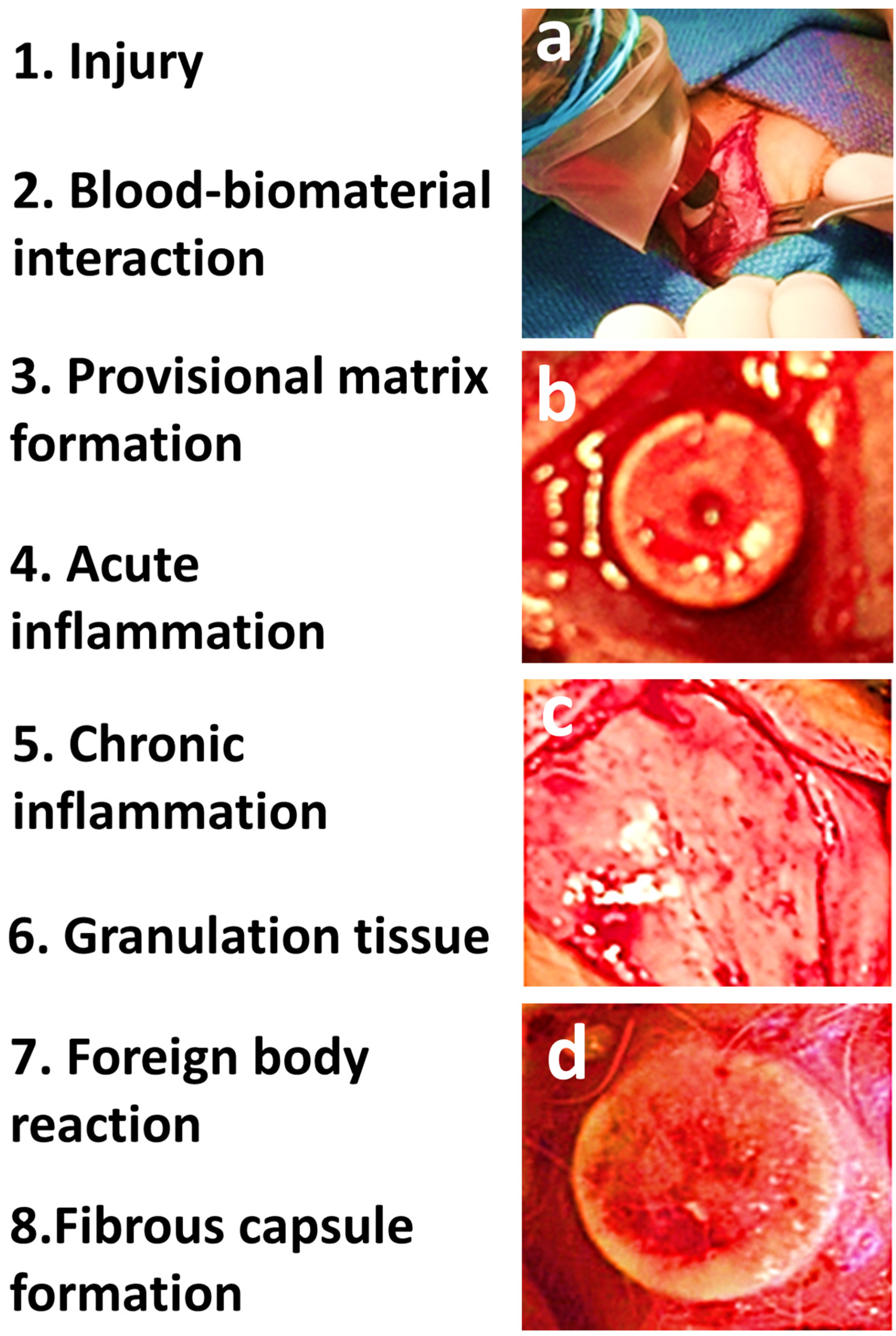
4. Wound Healing and Tissue Response
5. Protein and Cellular Response to Biomaterial Implantation

6. Macrophage Recognition and Attachment to Biomaterials
7. Macrophage-Mediated Phagocytosis of Biomaterials
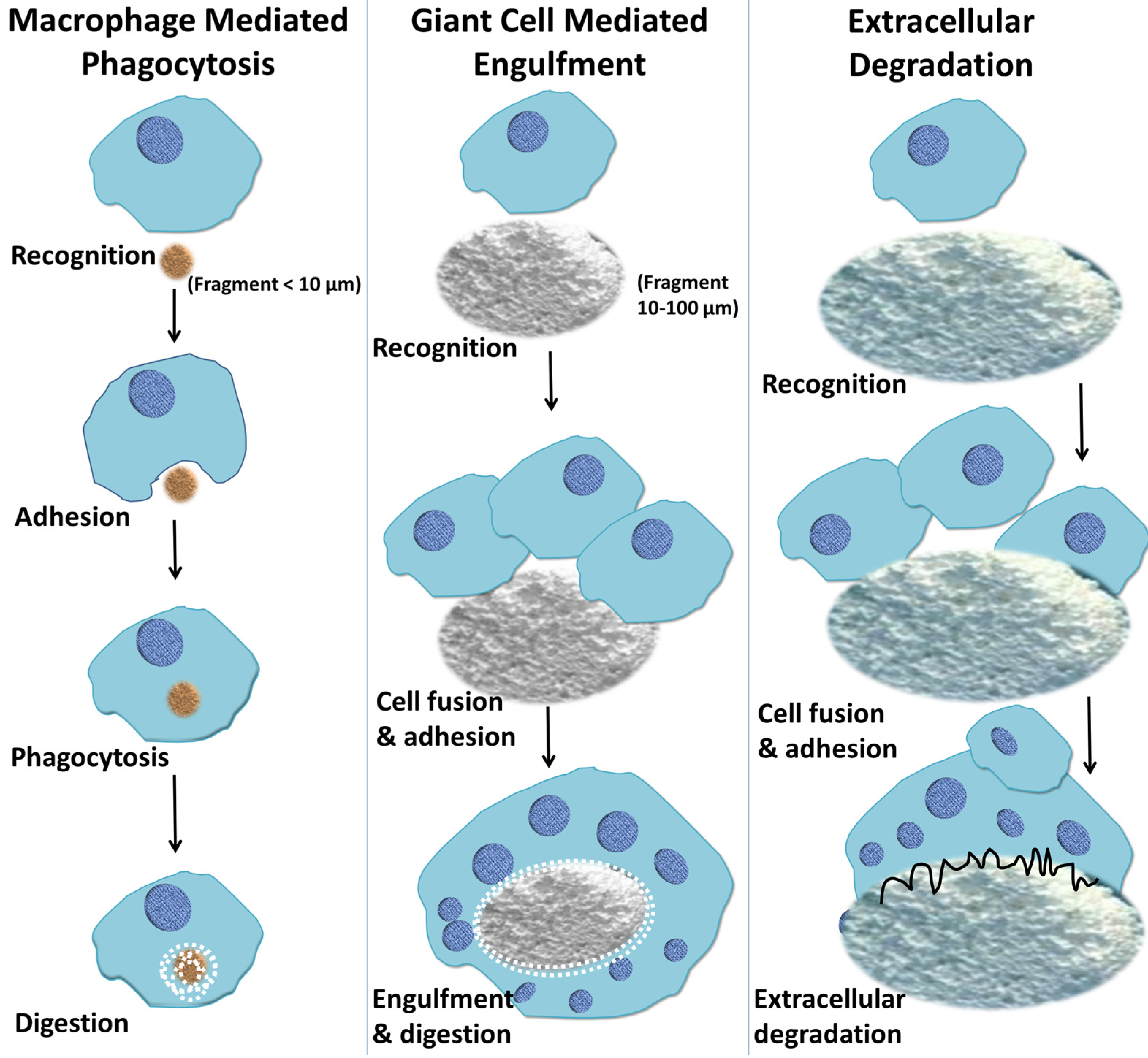
8. Macrophage-Mediated Extracellular Degradation of Biomaterials
9. The Effect of Chemistry and Physical Features of Biomaterial Surface on the Foreign Body Response
10. Conclusions
Author Contributions
Conflicts of Interest
References
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30, 5897–5909. [Google Scholar] [CrossRef] [PubMed]
- Hotaling, N.A.; Tang, L.; Irvine, D.J.; Babensee, J.E. Biomaterial strategies for immunomodulation. Annu. Rev. Biomed. Eng. 2015, 17, 1–16. [Google Scholar]
- Hench, L.L.; Thompson, I. Twenty-first century challenges for biomaterials. J. R. Soc. Interface 2010, 7, S379–S391. [Google Scholar] [CrossRef] [PubMed]
- Hodde, J. Naturally occurring scaffolds for soft tissue repair and regeneration. Tissue Eng. 2002, 8, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Barrere, F.; Mahmood, T.; De Groot, K.; Van Blitterswijk, C. Advanced biomaterials for skeletal tissue regeneration: Instructive and smart functions. Mater. Sci. Eng. R Rep. 2008, 59, 38–71. [Google Scholar] [CrossRef]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomater. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Luttikhuizen, D.T.; Harmsen, M.C.; Luyn, M.J.V. Cellular and molecular dynamics in the foreign body reaction. Tissue Eng. 2006, 12, 1955–1970. [Google Scholar] [CrossRef] [PubMed]
- Gretzer, C.; Emanuelsson, L.; Liljensten, E.; Thomsen, P. The inflammatory cell influx and cytokines changes during transition from acute inflammation to fibrous repair around implanted materials. J. Biomater. Sci. Polym. Ed. 2006, 17, 669–687. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M. Multinucleated giant cells. Curr. Opin. Hematol. 2000, 7, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M. Biological responses to materials. Ann. Rev. Mater. Res. 2001, 31, 81–110. [Google Scholar] [CrossRef]
- Rajesh, P.; Verma, S.; Verma, V.; Balani, K.; Agarwal, A.; Narayan, R. Host response of implanted biomaterials. In Biosurfaces: A Materials Science and Engineering Perspective; John Wiley & Sons, Inc.: Miami, FL, USA, 2015; pp. 106–125. [Google Scholar]
- Latour, R.A. Biomaterials: Protein-surface interactions. Encycl. Biomater. Biomed. Eng. 2005, 28, 1–15. [Google Scholar]
- Wilson, C.J.; Clegg, R.E.; Leavesley, D.I.; Pearcy, M.J. Mediation of biomaterial-cell interactions by adsorbed proteins: A review. Tissue Eng. 2005, 11, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign Body Reaction to Biomaterials. Sem. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Zdolsek, J.; Eaton, J.W.; Tang, L. Histamine release and fibrinogen adsorption mediate acute inflammatory responses to biomaterial implants in humans. J. Transl. Med. 2007, 5, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Jennings, T.A.; Eaton, J.W. Mast cells mediate acute inflammatory responses to implanted biomaterials. Proc. Natl. Acad. Sci. USA 1998, 95, 8841–8846. [Google Scholar] [CrossRef] [PubMed]
- Chomarat, P.; Banchereau, J. Interleukin-4 and lnterleukin-13: Their similarities and discrepancies. Int. Rev. Immunol. 1998, 17, 1–52. [Google Scholar] [CrossRef] [PubMed]
- Murthy, T.; Janaki, K.S.; Nagarjuna, S.; Sangeetha, P.; Sindhura, S. Biological response modifiers. Int. J. PharmTech. Res. 2010, 2, 2152–2160. [Google Scholar]
- Hu, W.J.; Eaton, J.W.; Ugarova, T.P.; Tang, L. Molecular basis of biomaterial-mediated foreign body reactions. Blood 2001, 98, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Kwee, B.J.; Mooney, D.J. Manipulating the intersection of angiogenesis and inflammation. Ann. Biomed. Eng. 2015, 43, 628–640. [Google Scholar] [CrossRef] [PubMed]
- Wooley, P.H.; Hallab, N.J. Wound Healing, Chronic Inflammation, and Immune Responses. In Metal-on-Metal Bearings; Springer: New York, NY, USA, 2014; pp. 109–133. [Google Scholar]
- Williams, D. Tissue-biomaterial interactions. J. Mater. Sci. 1987, 22, 3421–3445. [Google Scholar] [CrossRef]
- Ksander, G.A.; Gray, L. Reduced capsule formation around soft silicone rubber prostheses coated with solid collagen. Ann. Plast. Surg. 1985, 14, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Major, M.R.; Wong, V.W.; Nelson, E.R.; Longaker, M.T.; Gurtner, G.C. The foreign body response: At the interface of surgery and bioengineering. Plast. Reconstr. Surg. 2015, 135, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- DiEgidio, P.; Friedman, H.I.; Gourdie, R.G.; Riley, A.E.; Yost, M.J.; Goodwin, R.L. Biomedical implant capsule formation: Lessons learned and the road ahead. Ann. Plast. Surg. 2014, 73, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Vignery, A. Osteoclasts and giant cells: Macrophage-macrophage fusion mechanism. Int. J. Exp. Pathol. 2000, 81, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, J.A. Colony-stimulating factor in inflammation and autoimmunity. Nat. Rev. Immunol. 2008, 8, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Insogna, K.L.; Sahni, M.; Grey, A.B.; Tanaka, S.; Horne, W.C.; Neff, L.; Mitnick, M.; Levy, J.B.; Baron, R. Colony-stimulating factor-1 induces cytoskeletal reorganization and c-src-dependent tyrosine phosphorylation of selected cellular proteins in rodent osteoclasts. J. Clin. Investig. 1997, 100, 2476–2485. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. The macrophage: Past, present and future. Eur. J. Immunol. 2007, 37, S9–S17. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D.; Ley, K.; Buchmann, K.; Canton, J. Sequential immune responses: The weapons of immunity. J. Innate Immun. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000prime Rep. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Mackaness, G.B. Cellular immunity and the parasite. Adv. Exp. Med. Biol. 1977, 93, 65–73. [Google Scholar] [PubMed]
- O’Shea, J.J.; Murray, P.J. Cytokine signaling modules in inflammatory responses. Immunity 2008, 28, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sato, S.; Hemmi, H.; Hoshino, K.; Kaisho, T.; Sanjo, H.; Takeuchi, O.; Sugiyama, M.; Okabe, M.; Takeda, K.; et al. Role of adaptor trif in the myd88-independent toll-like receptor signaling pathway. Science 2003, 301, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Langrish, C.L.; Chen, Y.; Blumenschein, W.M.; Mattson, J.; Basham, B.; Sedgwick, J.D.; McClanahan, T.; Kastelein, R.A.; Cua, D.J. Il-23 drives a pathogenic t cell population that induces autoimmune inflammation. J. Exp. Med. 2005, 201, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Veldhoen, M.; Hocking, R.J.; Atkins, C.J.; Locksley, R.M.; Stockinger, B. Tgfβ in the context of an inflammatory cytokine milieu supports de novo differentiation of il-17-producing t cells. Immunity 2006, 24, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Bettelli, E.; Carrier, Y.; Gao, W.; Korn, T.; Strom, T.B.; Oukka, M.; Weiner, H.L.; Kuchroo, V.K. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory t cells. Nature 2006, 441, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Sinha, M.; Datta, S.; Abas, M.; Chaffee, S.; Sen, C.K.; Roy, S. Monocyte and macrophage plasticity in tissue repair and regeneration. Am. J. Pathol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Tarique, A.A.; Logan, J.; Thomas, E.; Holt, P.G.; Sly, P.D.; Fantino, E. Phenotypic, functional and plasticity features of classical and alternatively activated human macrophages. Am. J. Respir. Cell Mol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Loke, P.; Gallagher, I.; Nair, M.G.; Zang, X.; Brombacher, F.; Mohrs, M.; Allison, J.P.; Allen, J.E. Alternative activation is an innate response to injury that requires cd4+ t cells to be sustained during chronic infection. J. Immunol. 2007, 179, 3926–3936. [Google Scholar] [CrossRef] [PubMed]
- Kreider, T.; Anthony, R.M.; Urban, J.F.J.; Gause, W.C. Alternatively activated macrophages in helminth infections. Curr. Opin. Immunol. 2007, 19, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.P.; Zhang, X.; Frauwirth, K.A.; Mosser, D.M. Biochemical and functional characterization of three activated macrophage populations. J. Leukoc. Biol. 2006, 80, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Hesse, M.; Modolell, M.; La Flamme, A.C.; Schito, M.; Fuentes, J.M.; Cheever, A.W.; Pearce, E.J.; Wynn, T.A. Differential regulation of nitric oxide synthase-2 and arginase-1 by type 1/type 2 cytokines in vivo: Granulomatous pathology is shaped by the pattern of l-arginine metabolism. J. Immunol. 2001, 167, 6533–6544. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M. The many faces of macrophage activation. J. Leukoc. Biol. 2003, 73, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Sternberg, E.M. Neural regulation of innate immunity: A coordinated nonspecific host response to pathogens. Nat. Rev. Immunol. 2006, 6, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Fadok, V.A.; Bratton, D.L.; Konowal, A.; Freed, P.W.; Westcott, J.Y.; Henson, P.M. Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving tgf-β, pge2, and paf. J. Clin. Investig. 1998, 101, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.C.; Jenkins, S.J.; Allen, J.E.; Taylor, P.R. Tissue-resident macrophages. Nat. Immunol. 2013, 14, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Palis, J.; Robertson, S.; Kennedy, M.; Wall, C.; Keller, G. Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 1999, 126, 5073–5084. [Google Scholar] [PubMed]
- Bertrand, J.Y.; Jalil, A.; Klaine, M.; Jung, S.; Cumano, A.; Godin, I. Three pathways to mature macrophages in the early mouse yolk sac. Blood 2005, 106, 3004–3011. [Google Scholar] [CrossRef] [PubMed]
- Cumano, A.; Godin, I. Ontogeny of the hematopoietic system. Annu. Rev. Immunol. 2007, 25, 745–785. [Google Scholar] [CrossRef] [PubMed]
- Orkin, S.H.; Zon, L.I. Hematopoiesis: An evolving paradigm for stem cell biology. Cell 2008, 132, 631–644. [Google Scholar] [CrossRef]
- Dittmar, T.; Zanker, K.S. Cell Fusion in Health and Disease; Springer: New York, NY, USA, 2011. [Google Scholar]
- Jeremic, A.; Kelly, M.; Cho, J.A.; Horber, J.K.; Jena, B.P. Calcium drives fusion of snare-apposed bilayers. Cell. Biol. Int. 2004, 28, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Mukai, A.; Kurisaki, T.; Sato, S.B.; Kobayashi, T.; Kondoh, G.; Hashimoto, N. Dynamic clustering and dispersion of lipid rafts contribute to fusion competence of myogenic cells. Exp. Cell. Res. 2009, 315, 3052–3063. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Jena, B.P.; Cho, S.J.; Jeremic, A.; Stromer, M.H.; Abu-Hamdah, R. Structure and compositiion of the fusion pore. Biophys. J. 2003, 84, 1337–1343. [Google Scholar] [CrossRef]
- Sens, K.L.; Zhang, S.; Jin, P.; Duan, R.; Zhang, G.; Luo, F.; Parachini, L.; Chen, E.H. An invasive podosome-like structure promotes fusion pore formation during myoblast fusion. J. Cell. Biol. 2010, 191, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Helming, L.; Gordon, S. Molecular mediators of macrophage fusion. Trends Cell Biol. 2009, 19, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Vignery, A. Macrophage fusion: The making of osteoclasts and giant cells. J. Exp. Med. 2005, 202, 337–340. [Google Scholar] [CrossRef] [PubMed]
- McNally, A.K.; Anderson, J.M. Multinucleated giant cell formation exhibits features of phagocytosis with participation of the endoplasmic reticulum. Exp. Mol. Pathol. 2005, 79, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Dugast, C.; Gaudin, A.; Toujas, L. Generation of multinucleated giant cells by culture of monocyte-derived macrophages with il-4. J. Leukoc. Biol. 1997, 61, 517–521. [Google Scholar] [PubMed]
- Schachtner, H.; Calaminus, S.D.; Thomas, S.G.; Machesky, M.L. Podosomes in adhesion, migration, mechanosensing and matrix remodeling. Cytoskeleton 2013, 70, 572–589. [Google Scholar] [CrossRef] [PubMed]
- Arjonen, A.; Kaukonen, R.; Ivaska, J. Filopodia and adhesion in cancer cell motility. Cell. Adh. Migr. 2011, 5, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Guiet, R.; Verollet, C.; Lamsoul, I.; Cougoule, C.; Poincloux, R.; Labrousse, A.; Calderwood, D.A.; Glogauer, M.; Lutz, P.G.; Maridonneau-Parini, I. Macrophage mesenchymal migration requires podosome stabilization by filamin a. J. Biol. Chem. 2012, 287, 13051–13062. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Reeve, J.L.; Liu, Y.; Teitelbaum, S.L.; Ross, F.P. Dap12 couples c-fms activation to the osteoclast cytoskeleton by recruitment of syk. Mol. Cell 2008, 31, 422–431. [Google Scholar] [CrossRef] [PubMed]
- McNally, A.K.; Anderson, J.M. Interleukin-4 induces foreign body giant cells from human mono-cytes/macrophages: Differential lymphokine regulation of macrophage fusion leads to morphological variants of multinucleated giant cells. Am. J. Pathol. 1995, 147, 1487–1499. [Google Scholar] [PubMed]
- DeFife, K.M.; Jenney, C.R.; Colton, E.; Anderson, J.M. Cytoskeletal and adhesive structural polarizations accompany il-13-induced human macrophage fusion. J. Histochem. Cytochem. 1999, 47, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Defife, K.; McNally, A.; Collier, T.; Jenney, C. Monocyte, macrophage and foreign body giant cell interactions with molecularly engineered surfaces. J. Mater. Sci. 1999, 10, 579–588. [Google Scholar]
- McNally, A.K.; Jones, J.A.; MacEwan, S.R.; Colton, E.; Anderson, J.M. Vitronectin is a critical protein adhesion substrate for il-4-induced foreign body giant cell formation. J. Biomed. Mater. Res. A 2008, 86, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Yagi, M.; Miyamoto, T.; Sawatani, Y.; Iwamoto, K.; Hosogane, N.; Fujita, N.; Morita, K.; Ninomiya, K.; Suzuki, T.; Miyamoto, K.; et al. Dc-stamp is essential for cell-cell fusion in osteoclasts and foreign body giant cells. J. Exp. Med. 2005, 202, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.L.; Mikhailenko, I.; Tondravi, M.M.; Keegan, A.D. Il-4 promotes the formation of multinucleated giant cells from macrophage precursors by a stat6-dependent, homotypic mechanism: Contribution of e-cadherin. J. Leukoc. Biol. 2007, 82, 1542–1553. [Google Scholar] [CrossRef] [PubMed]
- Sterling, H.; Saginario, C.; Vignery, A. Cd44 occupancy prevents macrophage multinucleation. J. Cell. Biol. 1998, 143, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Saginario, C.; Sterling, H.; Beckers, C.; Kobayashi, R.; Solimena, M.; Ullu, E.; Vignery, A. Mfr, a putative receptor mediating the fusion of macrophages. Mol. Cell. Biol. 1998, 18, 6213–6223. [Google Scholar] [PubMed]
- McNally, A.K.; DeFife, K.M.; Anderson, J.M. Interleukin-4-induced macrophage fusion is prevented by inhibitors of mannose receptor activity. Am. J. Pathol. 1996, 149, 975–985. [Google Scholar] [PubMed]
- Burke, B.; Lewis, C.E. The Macrophage; Oxford University Press: Oxford, UK, 2002. [Google Scholar]
- Solheim, E.; Sudmann, B.; Bang, G.; Sudmann, E. Biocompatibility and effect on osteogenesis of poly(orthoester) compared to poly(dl-lactic acid). J. Biomed. Mater. Res. 2000, 49, 257–263. [Google Scholar] [CrossRef]
- Goodman, S.; Aspenberg, P.; Song, Y.; Regula, D.; Lidgren, L. Intermittent micromotion and polyethylene particles inhibit bone ingrowth into titanium chambers in rabbits. J. Appl. Biomater. 1995, 6, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Leibovich, S.J. Inflammatory cells during wound repair: The good, the bad and the ugly. Trends Cell Biol. 2005, 15, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Saurer, L.; Schenk, M.; Dickgreber, N.; Mueller, C. Cx3cr1 defines functionally distinct intestinal mononuclear phagocyte subsets which maintain their respective functions during homeostatic and inflammatory conditions. Eur. J. Immunol. 2011, 41, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, P.; Pellicoro, A.; Vernon, M.A.; Boulter, L.; Aucott, R.L.; Ali, A.; Hartland, S.N.; Snowdon, V.K.; Cappon, A.; Gordon-Walker, T.T.; et al. Differential ly-6c expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. Proc. Natl. Acad. Sci. USA 2012, 109, E3186–E3195. [Google Scholar] [CrossRef] [PubMed]
- Shechter, R.; London, A.; Varol, C.; Raposo, C.; Cusimano, M.; Yovel, G.; Rolls, A.; Mack, M.; Pluchino, S.; Martino, G.; et al. Infiltrating blood-derived macrophages are vital cells playing an anti-inflammatory role in recovery from spinal cord injury in mice. PLoS Med. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Broughton, G.; Janis, J.E.; Attinger, C.E. The basic science of wound healing. Plast. Reconstr. Surg. 2006, 117, S12–S34. [Google Scholar] [CrossRef] [PubMed]
- Nahrendorf, M.; Swirski, F.K.; Aikawa, E.; Stangenberg, L.; Wurdinger, T.; Figueiredo, J.L.; Libby, P.; Weissleder, R.; Pittet, M.J. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 2007, 204, 3037–3047. [Google Scholar] [CrossRef] [PubMed]
- London, A.; Itskovich, E.; Benhar, I.; Kalchenko, V.; Mack, M.; Jung, S.; Schwartz, M. Neuroprotection and progenitor cell renewal in the injured adult murine retina requires healing monocyte-derived macrophages. J. Exp. Med. 2011, 208, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Crane, M.J.; Daley, J.M.; van Houtte, O.; Brancato, S.K.; Henry, W.L.; Albina, J.E. The monocyte to macrophage transition in the murine sterile wound. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Niess, J.H.; Brand, S.; Gu, X.; Landsman, L.; Jung, S.; McCormick, B.A.; Vyas, J.M.; Boes, M.; Ploegh, H.L.; Fox, J.G.; et al. Cx3cr1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 2005, 307, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Varol, C.; Vallon-Eberhard, A.; Elinav, E.; Aychek, T.; Shapira, Y.; Luche, H.; Fehling, H.J.; Hardt, W.D.; Shakhar, G.; Jung, S. Intestinal lamina propria dendritic cell subsets have different origin and functions. Immunity 2009, 31, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Bogunovic, M.; Ginhoux, F.; Helft, J.; Shang, L.; Hashimoto, D.; Greter, M.; Liu, K.; Jakubzick, C.; Ingersoll, M.A.; Leboeuf, M.; et al. Origin of the lamina propria dendritic cell network. Immunity 2009, 31, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Fogg, D.K.; Sibon, C.; Miled, C.; Jung, S.; Aucouturier, P.; Littman, D.R.; Cumano, A.; Geissmann, F. A clonogenic bone marrow progenitor specific for macrophages and dendritic cells. Science 2006, 311, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Rakoff-Nahoum, S.; Paglino, J.; Eslami-Varzaneh, F.; Edberg, S.; Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 2004, 118, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Mortha, A.; Chudnovskiy, A.; Hashimoto, D.; Bogunovic, M.; Spencer, S.P.; Belkaid, Y.; Merad, M. Microbiota-dependent crosstalk between macrophages and ilc3 promotes intestinal homeostasis. Science 2014, 343. [Google Scholar] [CrossRef] [PubMed]
- Jenney, C.R.; Anderson, J.M. Adsorbed serum proteins responsible for surface dependent human macrophage behavior. J. Biomed. Mater. Res. 2000, 49, 435–447. [Google Scholar] [CrossRef]
- Brodbeck, W.G.; Colton, E.; Anderson, J. Effects of adsorbed heat labile serum proteins and fibrinogen on adhesion and apoptosis of monocytes/macrophages on biomaterials. J Mater. Sci. Mater. Med. 2003, 14, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Jenney, C.R.; Anderson, J.M. Adsorbed igg: A potent adhesive substrate for human macrophages. Biomed. Mater. Res. 2000, 50, 281–290. [Google Scholar] [CrossRef]
- Milleret, V.; Buzzi, S.; Gehrig, P.; Ziogas, A.; Grossmann, J.; Schilcher, K.; Zinkernagel, A.S.; Zucker, A.; Ehrbar, M. Protein adsorption steers blood contact activation on engineered cobalt chromium alloy oxide layers. Acta Biomater. 2015, 24, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Triffitt, J.T. A review on macrophage responses to biomaterials. Biomed. Mater. 2006, 1. [Google Scholar] [CrossRef] [PubMed]
- Tamimi, F.; Sheikh, Z.; Barralet, J. Dicalcium phosphate cements: Brushite and monetite. Acta Biomater. 2012, 8, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Descamps, M.; Dejou, J.; Koubi, G.; Hardouin, P.; Lemaitre, J.; Proust, J.P. The biodegradation mechanism of calcium phosphate biomaterials in bone. J. Biomed. Mater. Res. 2002, 63, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Takebe, J.; Champagne, C.; Offenbacher, S.; Ishibashi, K.; Cooper, L. Titanium surface topography alters cell shape and modulates bone morphogenetic protein 2 expression in the j774a. 1 macrophage cell line. J. Biomed. Mater. Res. Part A 2003, 64, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Labow, R.S.; Sa, D.; Matheson, L.A.; Santerre, J.P. Polycarbonate-urethane hard segment type influences esterase substrate specificity for human-macrophage-mediated biodegradation. J. Biomatr. Sci. Polym. Ed. 2005, 16, 1167–1177. [Google Scholar] [CrossRef]
- Xia, Z.; Zhu, T.; Du, J.; Zheng, Q.; Wang, L.; Li, S.; Chang, C.; Fang, S. Macrophages in degradation of collagen/hydroxylapatite (cha), beta-tricalcium phosphate ceramics (tcp) artificial bone graft. An in vivo study. Chin. Med. J. 1994, 107, 845–849. [Google Scholar] [PubMed]
- Khouw, I.; van Wachem, P.; De Leij, L.; Van Luyn, M. Inhibition of the tissue reaction to a biodegradable biomaterial by monoclonal antibodies to ifn-γ. J. Biomater. Sci. Polym. Ed. 1998, 41, 202–210. [Google Scholar] [CrossRef]
- Brodbeck, W.G.; MacEwan, M.; Colton, E.; Meyerson, H.; Anderson, J.M. Lymphocytes and the foreign body response: Lymphocyte enhancement of macrophage adhesion and fusion. Biomed. Mater. Res. Part A 2005, 74, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Gwinnett, A.; Tay, F. Early and intermediate time response of the dental pulp to an acid etch technique in vivo. Am. J. Dent. 1998, 11, S35–S44. [Google Scholar] [PubMed]
- Shen, M.; Horbett, T.A. The effects of surface chemistry and adsorbed proteins on monocyte/macrophage adhesion to chemically modified polystyrene surfaces. J. Biomed. Mater. Res. 2001, 57, 336–345. [Google Scholar] [CrossRef]
- Castner, D.G.; Ratner, B.D. Biomedical surface science: Foundations to frontiers. Surf. Sci. 2002, 500, 28–60. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, G.E.; Cho, D.R.; Rubash, H.E.; Goldring, S.R.; Herndon, J.H.; Shanbhag, A.S. Gene expression clustering using self-organizing maps: Analysis of the macrophage response to particulate biomaterials. Biomaterials 2005, 26, 2933–2945. [Google Scholar] [CrossRef] [PubMed]
- Calle, Y.; Burns, S.; Thrasher, A.J.; Jones, G.E. The leukocyte podosome. Eur. J. Cell. Biol. 2006, 85, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Kao, W.J. Evaluation of protein-modulated macrophage behavior on biomaterials: Designing biomimetic materials for cellular engineering. Biomaterials 1999, 20, 2213–2221. [Google Scholar] [CrossRef]
- Phillips, J.M.; Kao, W.J. Macrophage adhesion on gelatin-based interpenetrating networks grafted with pegylated rgd. Tissue Eng. 2005, 11, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Berton, G.; Lowell, C.A. Integrin signalling in neutrophils and macrophages. Cell Signal 1999, 11, 621–635. [Google Scholar] [CrossRef]
- McNally, A.K.; MacEwan, S.R.; Anderson, J.M. Alpha subunit partners to beta1 and beta2 integrins during il-4-induced foreign body giant cell formation. J. Biomed. Mater. Res. A 2007, 82, 568–574. [Google Scholar] [CrossRef] [PubMed]
- McNally, A.K.; Anderson, J.M. Complement c3 participation in monocyte adhesion to different surfaces. Proc. Natl. Acad. Sci. USA 1994, 91, 10119–10123. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.A. Recruitment of inflammatory cells into lung: Roles of cytokines, adhesion molecules, and complement. J. Lab. Clin. Med. 1997, 129, 400–404. [Google Scholar] [CrossRef]
- Xia, Z.; Ye, H.; Choong, C.; Ferguson, D.J.; Platt, N.; Cui, Z.; Triffitt, J.T. Macrophagic response to human mesenchymal stem cell and poly(epsilon-caprolactone) implantation in nonobese diabetic/severe combined immunodeficient mice. J. Biomed. Mater. Res. A 2004, 71, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Van der Mei, H.C. Macrophage-Mediated Phagocytosis of Bacteria Adhering on Biomaterial Surfaces. Ph.D. Thesis, Rijksuniversiteit Groningen, the Netherlands, 28 May 2014. [Google Scholar]
- Allen, L.-A.H.; Aderem, A. Mechanisms of phagocytosis. Curr. Opin. Immunol. 1996, 8, 36–40. [Google Scholar] [CrossRef]
- Ogle, O. Tissue response: Biomaterials, dental implants, and compromised osseous tissue. Dent. Clin. N. Am. 2015, 59, 305–315. [Google Scholar]
- Kwiatkowska, K.; Sobota, A. Signaling pathways in phagocytosis. Bioessays 1999, 21, 422–431. [Google Scholar] [CrossRef]
- Tondravi, M.; McKercher, S.; Anderson, K.; Erdmann, J.; Quiroz, M.; Maki, R.; Teitelbaum, S. Osteopetrosis in mice lacking haematopoietic transcription factor PU. 1. Nature 1997, 386, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Jutras, I.; Desjardins, M. Phagocytosis: At the crossroads of innate and adaptive immunity. Ann. Rev. Cell Dev. Biol. 2005, 21, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.; Auger, M.; Burke, B.; Lewis, C. The biology of the macrophage. Macrophage 2002, 2, 16–23. [Google Scholar]
- Henson, P.M. The immunologic release of constituents from neutrophil leukocytes i. The role of antibody and complement on nonphagocytosable surfaces or phagocytosable particles. J. Immunol. 1971, 107, 1535–1546. [Google Scholar] [PubMed]
- Henson, P.M. The immunologic release of constituents from neutrophil leukocytes ii. Mechanisms of release during phagocytosis, and adherence to nonphagocytosable surfaces. J. Immunol. 1971, 107, 1547–1557. [Google Scholar] [PubMed]
- Haas, A. The phagosome: Compartment with a license to kill. Traffic 2007, 8, 311–330. [Google Scholar] [CrossRef] [PubMed]
- Santerre, J.; Woodhouse, K.; Laroche, G.; Labow, R. Understanding the biodegradation of polyurethanes: From classical implants to tissue engineering materials. Biomaterials 2005, 26, 7457–7470. [Google Scholar] [CrossRef] [PubMed]
- Labow, R.S.; Sa, D.; Matheson, L.A.; Dinnes, D.L.M.; Santerre, J.P. The human macrophage response during differentiation and biodegradation on polycarbonate-based polyurethanes: Dependence on hard segment chemistry. Biomaterials 2005, 26, 7357–7366. [Google Scholar] [CrossRef] [PubMed]
- Matheson, L.A.; Labow, R.S.; Santerre, J.P. Biodegradation of polycarbonate—based polyurethanes by the human monocyte—derived macrophage and u937 cell systems. J. Biomed. Mater. Res. 2002, 61, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Labow, R.S.; Tang, Y.; McCloskey, C.B.; Santerre, J.P. The effect of oxidation on the enzyme-catalyzed hydrolytic biodegradation of poly (urethane)s. J. Biomater. Sci. Polym. Ed. 2002, 13, 651–665. [Google Scholar] [CrossRef] [PubMed]
- Labow, R.; Meek, E.; Santerre, J. Hydrolytic degradation of poly (carbonate)-urethanes by monocyte-derived macrophages. Biomaterials 2001, 22, 3025–3033. [Google Scholar] [CrossRef]
- Labow, R.S.; Meek, E.; Matheson, L.A.; Santerre, J.P. Human macrophage-mediated biodegradation of polyurethanes: Assessment of candidate enzyme activities. Biomaterials 2002, 23, 3969–3975. [Google Scholar] [CrossRef]
- Christenson, E.M.; Anderson, J.M.; Hiltner, A. Antioxidant inhibition of poly (carbonate urethane) in vivo biodegradation. J. Biomed. Mater. Res. Part A 2006, 76, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Lamba, N.M.; Woodhouse, K.A.; Cooper, S.L. Polyurethanes in Biomedical Applications; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar]
- Roy, N.; Bruchmann, B.; Lehn, J.-M. Dynamers: Dynamic polymers as self-healing materials. Chem. Soc. Rev. 2015, 44, 3786–3807. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; McNally, A.; Rubin, K.; Renier, M.; Wu, Y.; Rose-Caprara, V.; Anderson, J.; Hiltner, A.; Urbanski, P.; Stokes, K. Human plasma α2—macroglobulin promotes in vitro oxidative stress cracking of pellethane 2363—80a: In vivo and in vitro correlations. J. Biomed. Mater. Res. 1993, 27, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Kao, W.J.; Zhao, Q.H.; Hiltner, A.; Anderson, J.M. Theoretical analysis of in vivo macrophage adhesion and foreign body gaint cell formation on polydimethylsiloxane, low density polyethylene, and polyetherurethanes. J. Biomed. Mater. Res. 1994, 28, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.; Untereker, D. Degradability of polymers for implantable biomedical devices. Int. J. Mol. Sci. 2009, 10, 4033–4065. [Google Scholar] [CrossRef] [PubMed]
- Stokes, K.; Coury, A.; Urbanski, P. Autooxidative degradation of implanted polyether polyurethane devices. J. Biomater. Appl. 1986, 1, 411–448. [Google Scholar] [CrossRef]
- Tokiwa, Y.; Calabia, B.P. Biodegradability and biodegradation of poly (lactide). Appl. Microbiol. Biotechnol. 2006, 72, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Topham, N.; Anderson, J.; Hiltner, A.; Lodoen, G.; Payet, C. Foreign—body giant cells and polyurethane biostability: In vivo correlation of cell adhesion and surface cracking. J. Biomed. Mater. Res. 1991, 25, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.A.; Javaid, MA.; Abdallah, MN. Bone Replacement Graft Materials in Dentistry. In Dental Biomaterials (Principle and Its Application), 2nd ed.; Khurshid, Z., Sheikh, Z., Eds.; Paramount Publishing Enterprise: Karachi, Pakistan, 2013. [Google Scholar]
- Sheikh, Z.; Javaid, M.A.; Hamdan, N.; Hashmi, R. Bone regeneration using bone morphogenetic proteins and various biomaterial carriers. Materials 2015, 8, 1778–1816. [Google Scholar] [CrossRef]
- Sheikh, Z.; Sima, C.; Glogauer, M. Bone replacement materials and techniques used for achieving vertical alveolar bone augmentation. Materials 2015, 8, 2953–2993. [Google Scholar] [CrossRef]
- Tamimi, F.; Le Nihouannen, D.; Eimar, H.; Sheikh, Z.; Komarova, S.; Barralet, J. The effect of autoclaving on the physical and biological properties of dicalcium phosphate dihydrate bioceramics: Brushite vs. Monetite. Acta Biomater. 2012, 8, 3161–3169. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Geffers, M.; Christel, T.; Barralet, J.E.; Gbureck, U. Chelate setting of alkali ion substituted calcium phosphates. Ceram. Int. 2015, 41, 10010–10017. [Google Scholar] [CrossRef]
- Sheikh, Z.; Glogauer, M. Successful ridge augmentation: The challenge of periodontal tissue engineering. EC Dent. Sci. 2015, 2, 216–218. [Google Scholar]
- Rae, T. The macrophage response to implant materials-with special reference to those used in orthopedics. CRC Crit. Rev. Biocompat. 1986, 2, 97–126. [Google Scholar]
- Hannink, G.; Arts, J.C. Bioresorbability, porosity and mechanical strength of bone substitutes: What is optimal for bone regeneration? Injury 2011, 42, S22–S25. [Google Scholar] [CrossRef] [PubMed]
- Heymann, D.; Pradal, G.; Benahmed, M. Cellular mechanisms of calcium phosphate ceramic degradation. Histol. Histopathol. 1999, 14, 871–877. [Google Scholar] [PubMed]
- Thevenot, P.; Hu, W.; Tang, L. Surface chemistry influence implant biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [Google Scholar] [PubMed]
- Paul, N.E.; Skazik, C.; Harwardt, M.; Bartneck, M.; Denecke, B.; Klee, D.; Salber, J.; Zwadlo-Klarwasser, G. Topographical control of human macrophages by a regularly microstructured polyvinylidene fluoride surface. Biomaterials 2008, 29, 4056–4064. [Google Scholar] [CrossRef] [PubMed]
- Vardaxis, N.; Ruijgrok, J.; Rietveld, D.; Marres, E.; Boon, M. Chemical and physical properties of collagen implants influence their fate in vivo as evaluated by light and confocal microscopy. J. Biomed. Mater. Res. 1994, 28, 1013–1025. [Google Scholar] [CrossRef] [PubMed]
- Mitragotri, S.; Lahann, J. Physical approaches to biomaterial design. Nat. Mater. 2009, 8, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.; Qiu, Y.; Sang, H.; Mei, H.; Zhu, A.; Shen, J.; Lin, S. Various approaches to modify biomaterial surfaces for improving hemocompatibility. Adv. Colloid Interface Sci. 2004, 110, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Kim, K.-H.; Ong, J.L. A review on calcium phosphate coatings produced using a sputtering process—An alternative to plasma spraying. Biomaterials 2005, 26, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Hu, W. Molecular determinants of biocompatibility. Exp. Rev. Med. Dev. 2005, 2, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Craddock, P.R.; Fehr, J.; Dalmasso, A.; Brighan, K.; Jacob, H. Hemodialysis leukopenia. Pulmonary vascular leukostasis resulting from complement activation by dialyzer cellophane membranes. J Clin Invest 1977, 59, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Ward, W.K.; Slobodzian, E.P.; Tiekotter, K.L.; Wood, M.D. The effect of microgeometry, implant thickness and polyurethane chemistry on the foreign body response to subcutaneous implants. Biomaterials 2002, 23, 4185–4192. [Google Scholar] [CrossRef]
- Green, R.; Davies, M.; Roberts, C.; Tendler, S. A surface plasmon resonance study of albumin adsorption to peo–ppo–peo triblock copolymers. J. Biomed. Mater. Res. 1998, 42, 165–171. [Google Scholar] [CrossRef]
- Gombotz, W.R.; Guanghui, W.; Horbett, T.A.; Hoffman, A.S. Protein adsorption to poly (ethylene oxide) surfaces. J. Biomed. Mater. Res. 1991, 25, 1547–1562. [Google Scholar] [CrossRef] [PubMed]
- Jenney, C.R.; Anderson, J.M. Effects of surface—coupled polyethylene oxide on human macrophage adhesion and foreign body giant cell formation in vitro. J. Biomed. Mater. Res. 1999, 44, 206–216. [Google Scholar] [CrossRef]
- Rodgers, K.E.; Schwartz, H.E.; Roda, N.; Thornton, M.; Kobak, W. Effect of oxiplex* films (peo/cmc) on adhesion formation and reformation in rabbit models and on peritoneal infection in a rat model. Fertil. Steril. 2000, 73, 831–838. [Google Scholar] [CrossRef]
- Anderson, J.; Bonfield, T.; Ziats, N. Protein adsorption and cellular adhesion and activation on biomedical polymers. Int. J. Artif. Org. 1990, 13, 375–382. [Google Scholar]
- Nath, N.; Hyun, J.; Ma, H.; Chilkoti, A. Surface engineering strategies for control of protein and cell interactions. Surf. Sci. 2004, 570, 98–110. [Google Scholar] [CrossRef]
- Evans-Nguyen, K.M.; Fuierer, R.R.; Fitchett, B.D.; Tolles, L.R.; Conboy, J.C.; Schoenfisch, M.H. Changes in adsorbed fibrinogen upon conversion to fibrin. Langmuir 2006, 22, 5115–5121. [Google Scholar] [CrossRef] [PubMed]
- Heuberger, M.; Drobek, T.; Spencer, N.D. Interaction forces and morphology of a protein-resistant poly (ethylene glycol) layer. Biophys. J. 2005, 88, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Collier, T.; Anderson, J. Protein and surface effects on monocyte and macrophage adhesion, maturation, and survival. J. Biomed. Mater. Res. 2002, 60, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Dadsetan, M.; Jones, J.A.; Hiltner, A.; Anderson, J.M. Surface chemistry mediates adhesive structure, cytoskeletal organization, and fusion of macrophages. J. Biomed. Mater. Res. Part A 2004, 71, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.D.; Hlady, V.L.; van Wagenen, R.A. Effects of plasma protein adsorption on protein conformation and activity. Pure Appl. Chem. 1984, 56, 1345–1350. [Google Scholar] [CrossRef]
- Parker, J.; Walboomers, X.; von den Hoff, J.; Maltha, J.; Jansen, J. Soft-tissue response to silicone and poly-l-lactic acid implants with a periodic or random surface micropattern. J. Biomed. Mater. Res. 2002, 61, 91–98. [Google Scholar] [CrossRef] [PubMed]
- DeFife, K.M.; Colton, E.; Nakayama, Y.; Matsuda, T.; Anderson, J.M. Spatial regulation and surface chemistry control of monocyte/macrophage adhesion and foreign body giant cell formation by photochemically micropatterned surfaces. J. Biomed. Mater. Res. 1999, 45, 148–154. [Google Scholar] [CrossRef]
- Rice, J.; Hunt, J.; Gallagher, J.; Hanarp, P.; Sutherland, D.; Gold, J. Quantitative assessment of the response of primary derived human osteoblasts and macrophages to a range of nanotopography surfaces in a single culture model in vitro. Biomaterials 2003, 24, 4799–4818. [Google Scholar] [CrossRef]
- Wójciak-Stothard, B.; Curtis, A.; Monaghan, W.; Macdonald, K.; Wilkinson, C. Guidance and activation of murine macrophages by nanometric scale topography. Exp. Cell Res. 1996, 223, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Popat, K.C.; Leoni, L.; Grimes, C.A.; Desai, T.A. Influence of engineered titania nanotubular surfaces on bone cells. Biomaterials 2007, 28, 3188–3197. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.J.; Marshall, G.E.; Johnstone, H.J.; Affrossman, S.; Riehle, M.O. Interactions of human blood and tissue cell types with 95-nm-high nanotopography. IEEE Transa. NanoBiosci. 2002, 1, 18–23. [Google Scholar] [CrossRef]
- Champion, J.A.; Mitragotri, S. Role of target geometry in phagocytosis. Proc. Natl. Acad. Sci. USA 2006, 103, 4930–4934. [Google Scholar] [CrossRef] [PubMed]
- Gratton, S.E.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The effect of particle design on cellular internalization pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Dalhaimer, P.; Cai, S.; Tsai, R.; Tewari, M.; Minko, T.; Discher, D.E. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat. Nanotechnol. 2007, 2, 249–255. [Google Scholar] [CrossRef]
- Dalby, M.J.; Riehle, M.O.; Sutherland, D.S.; Agheli, H.; Curtis, A.S. Use of nanotopography to study mechanotransduction in fibroblasts–methods and perspectives. Eur. J. Cell Biol. 2004, 83, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Hulbert, S.; Morrison, S.; Klawitter, J. Tissue reaction to three ceramics of porous and non-porous structures. J. Biomed. Mater. Res. 1972, 6, 347–374. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.; Childs, S.; Riehle, M.; Johnstone, H.; Affrossman, S.; Curtis, A. Fibroblast reaction to island topography: Changes in cytoskeleton and morphology with time. Biomaterials 2003, 24, 927–935. [Google Scholar] [CrossRef]
- Chen, C.S.; Mrksich, M.; Huang, S.; Whitesides, G.M.; Ingber, D.E. Geometric control of cell life and death. Science 1997, 276, 1425–1428. [Google Scholar] [CrossRef] [PubMed]
- Sanders, J.; Bale, S.; Neumann, T. Tissue response to microfibers of different polymers: Polyester, polyethylene, polylactic acid, and polyurethane. J. Biomed. Mater. Res. 2002, 62, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Urban, R.M.; Jacobs, J.J.; Gilbert, J.L.; Galante, J.O. Migration of corrosion products from modular hip prostheses. Particle microanalysis and histopathological findings. J. Bone Joint Surg. 1994, 76, 1345–1359. [Google Scholar] [PubMed]
- Blac, J. Systemic effects of biomaterials. Biomaterials 1984, 5, 11–18. [Google Scholar] [CrossRef]
- Yang, J.; Merritt, K. Production of monoclonal antibodies to study corrosion products of co-cr biomaterials. J. Biomed. Mater. Res. 1996, 31, 71–80. [Google Scholar] [CrossRef]
- Yang, J.; Merritt, K. Detection of antibodies against corrosion products in patients after Co-Cr total joint replacements. J. Biomed. Mater. Res. 1994, 28, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Flatebø, R.S.; Johannessen, A.C.; Grønningsæter, A.G.; Bøe, O.E.; Gjerdet, N.R.; Grung, B.; Leknes, K.N. Host response to titanium dental implant placement evaluated in a human oral model. J. Periodontol. 2006, 77, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Niinomi, M.; Boehlert, C.J. Titanium Alloys for Biomedical Applications. In Advances in Metallic Biomaterials; Springer: New York, NY, USA, 2015; pp. 179–213. [Google Scholar]
- Mitchelson, A.J.; Wilson, C.J.; Mihalko, W.M.; Grupp, T.M.; Manning, B.T.; Dennis, D.A.; Goodman, S.B.; Tzeng, T.H.; Vasdev, S.; Saleh, K.J. Biomaterial hypersensitivity: Is it real? Supportive evidence and approach considerations for metal allergic patients following total knee arthroplasty. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; von der Helm, C.; Schopf, C.; Mazoochian, F.; Frommelt, L.; Gollwitzer, H.; Schneider, J.; Flaig, M.; Krenn, V.; Thomas, B. Patients with intolerance reactions to total knee replacement: Combined assessment of allergy diagnostics, periprosthetic histology, and peri-implant cytokine expression pattern. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P. Allergic reactions to implant materials. Der Orthopade 2003, 32, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.; Merritt, K.; Jacobs, J.J. Metal sensitivity in patients with orthopaedic implants. J. Bone Joint Surg. Am. 2001, 83, 428–437. [Google Scholar] [PubMed]
- Gross, U. Biocompatibility-The interaction of biomaterials and host response. J. dent. Educ. 1988, 52, 798–803. [Google Scholar]
- Cochran, D.L. A comparison of endosseous dental implant surfaces. J. Periodontol 1999, 70, 1523–1539. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Brånemark, P.-I.; Hansson, H.-A.; Lindström, J. Osseointegrated titanium implants: Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop. 1981, 52, 155–170. [Google Scholar] [CrossRef]
- Hunt, J.A.; Shoichet, M. Biomaterials: Surface interactions. Curr. Opin. Solid State Mater. Sci. 2001, 5, 161–162. [Google Scholar] [CrossRef]
- Soskolne, W.A.; Cohen, S.; Shapira, L.; Sennerby, L.; Wennerberg, A. The effect of titanium surface roughness on the adhesion of monocytes and their secretion of tnf-α and pge2. Clin. Oral Implants Res. 2002, 13, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Kieswetter, K.; Schwartz, Z.; Hummert, T.; Cochran, D.; Simpson, J.; Dean, D.; Boyan, B. Surface roughness modulates the local production of growth factors and cytokines by osteoblast-like mg-63 cells. J. Biomed. Mater. Res. 1996, 32, 55–63. [Google Scholar] [CrossRef]
- Brunette, D. Effects of surface topography of implant materials on cell behavior in vitro and in vivo. Nanofabr. Biosyst. Integr. Mater. Sci. Eng. Biol. 1996, 335–355. [Google Scholar]
- Sennerby, L.; Thomsen, P.; Ericson, L. Early tissue response to titanium implants inserted in rabbit cortical bone. J. Mater. Sci. Mater. Med. 1993, 4, 240–250. [Google Scholar] [CrossRef]
- Futami, T.; Fujii, N.; Ohnishi, H.; Taguchi, N.; Kusakari, H.; Ohshima, H.; Maeda, T. Tissue response to titanium implants in the rat maxilla: Ultrastructural and histochemical observations of the bone-titanium interface. J. Periodontol. 2000, 71, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Miller, K.M. Biomaterial biocompatibility and the macrophage. Biomaterials 1984, 5, 5–10. [Google Scholar] [CrossRef]
- Ziats, N.P.; Miller, K.M.; Anderson, J.M. In vitro and in vivo interactions of cells with biomaterials. Biomaterials 1988, 9, 5–13. [Google Scholar] [CrossRef]
- Rich, A.; Harris, A.K. Anomalous preferences of cultured macrophages for hydrophobic and roughened substrata. J. Cell Sci. 1981, 50, 1–7. [Google Scholar] [PubMed]
- Salthouse, T.N. Some aspects of macrophage behavior at the implant interface. J. Biomed. Mater. Res. 1984, 18, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Wójciak-Stothard, B.; Madeja, Z.; Korohoda, W.; Curtis, A.; Wilkinson, C. Activation of macrophage-like cells by multiple grooved substrata. Topographical control of cell behaviour. Cell Biol. Int. 1995, 19, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Refai, A.K.; Textor, M.; Brunette, D.M.; Waterfield, J.D. Effect of titanium surface topography on macrophage activation and secretion of proinflammatory cytokines and chemokines. J. Biomed. Mater. Res. Part A 2004, 70, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Schwartz, Z.; Hummert, T.; Schraub, D.; Simpson, J.; Lankford, J.; Dean, D.; Cochran, D.; Boyan, B. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (mg63). J. Biomed. Mater. Res. 1995, 29, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Shanbhag, A.S.; Jacobs, J.J.; Black, J.; Galante, J.O.; Glant, T.T. Macrophage/particle interactions: Effect of size, composition and surface area. J. Biomed. Mater. Res. 1994, 28, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Pearson, B.; Klebe, R.; Boyan, B.; Moskowicz, D. Comments on the clinical application of fibronectin in dentistry. J. Dent. Res. 1988, 67, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Champagne, C.; Takebe, J.; Offenbacher, S.; Cooper, L. Macrophage cell lines produce osteoinductive signals that include bone morphogenetic protein-2. Bone 2002, 30, 26–31. [Google Scholar] [CrossRef]
- Jones, K.S. Effects of biomaterial-induced inflammation on fibrosis and rejection. Semin. Immunol. 2008, 20, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Donath, K.; Laaß, M.; Günzl, H.-J. The histopathology of different foreign-body reactions in oral soft tissue and bone tissue. Virchows. Archiv. A 1992, 420, 131–137. [Google Scholar] [CrossRef]
- Albrektsson, T.; Dahlin, C.; Jemt, T.; Sennerby, L.; Turri, A.; Wennerberg, A. Is marginal bone loss around oral implants the result of a provoked foreign body reaction? Clin. Implant Dent. Relat. Res. 2014, 16, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Trindade, R.; Albrektsson, T.; Tengvall, P.; Wennerberg, A. Foreign body reaction to biomaterials: On mechanisms for buildup and breakdown of osseointegration. Clin. Implant Dent. Relat. Res. 2014. [Google Scholar] [CrossRef] [PubMed]
- Trindade, R.; Albrektsson, T.; Wennerberg, A. Current concepts for the biological basis of dental implants: Foreign body equilibrium and osseointegration dynamics. Oral Maxillofac. Surg. Clin. North Am. 2015, 27, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Ciuti, G.; Ricotti, L.; Menciassi, A.; Dario, P. Mems sensor technologies for human centred applications in healthcare, physical activities, safety and environmental sensing: A review on research activities in Italy. Sensors 2015, 15, 6441–6468. [Google Scholar] [CrossRef] [PubMed]
- Wujcik, E.K.; Monty, C.N. Nanotechnology for implantable sensors: Carbon nanotubes and graphene in medicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2013, 5, 233–249. [Google Scholar] [CrossRef] [PubMed]
- Thompson, B.C.; Murray, E.; Wallace, G.G. Graphite oxide to graphene. Biomaterials to bionics. Adv. Mater. 2015. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Lizu, M.; Stewart, M.; Zygula, K.; Lu, Y.; Chauhan, R.; Yan, X.; Guo, Z.; Wujcik, E.K.; Wei, S. Multifunctional nanofibers towards active biomedical therapeutics. Polymers 2015, 7, 186–219. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sheikh, Z.; Brooks, P.J.; Barzilay, O.; Fine, N.; Glogauer, M. Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials. Materials 2015, 8, 5671-5701. https://doi.org/10.3390/ma8095269
Sheikh Z, Brooks PJ, Barzilay O, Fine N, Glogauer M. Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials. Materials. 2015; 8(9):5671-5701. https://doi.org/10.3390/ma8095269
Chicago/Turabian StyleSheikh, Zeeshan, Patricia J. Brooks, Oriyah Barzilay, Noah Fine, and Michael Glogauer. 2015. "Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials" Materials 8, no. 9: 5671-5701. https://doi.org/10.3390/ma8095269
APA StyleSheikh, Z., Brooks, P. J., Barzilay, O., Fine, N., & Glogauer, M. (2015). Macrophages, Foreign Body Giant Cells and Their Response to Implantable Biomaterials. Materials, 8(9), 5671-5701. https://doi.org/10.3390/ma8095269





