The Effect of Oxygen Partial Pressure on Microstructure and Properties of Fe40Al Alloy Sintered under Vacuum
Abstract
:1. Introduction
2. Results and Discussion
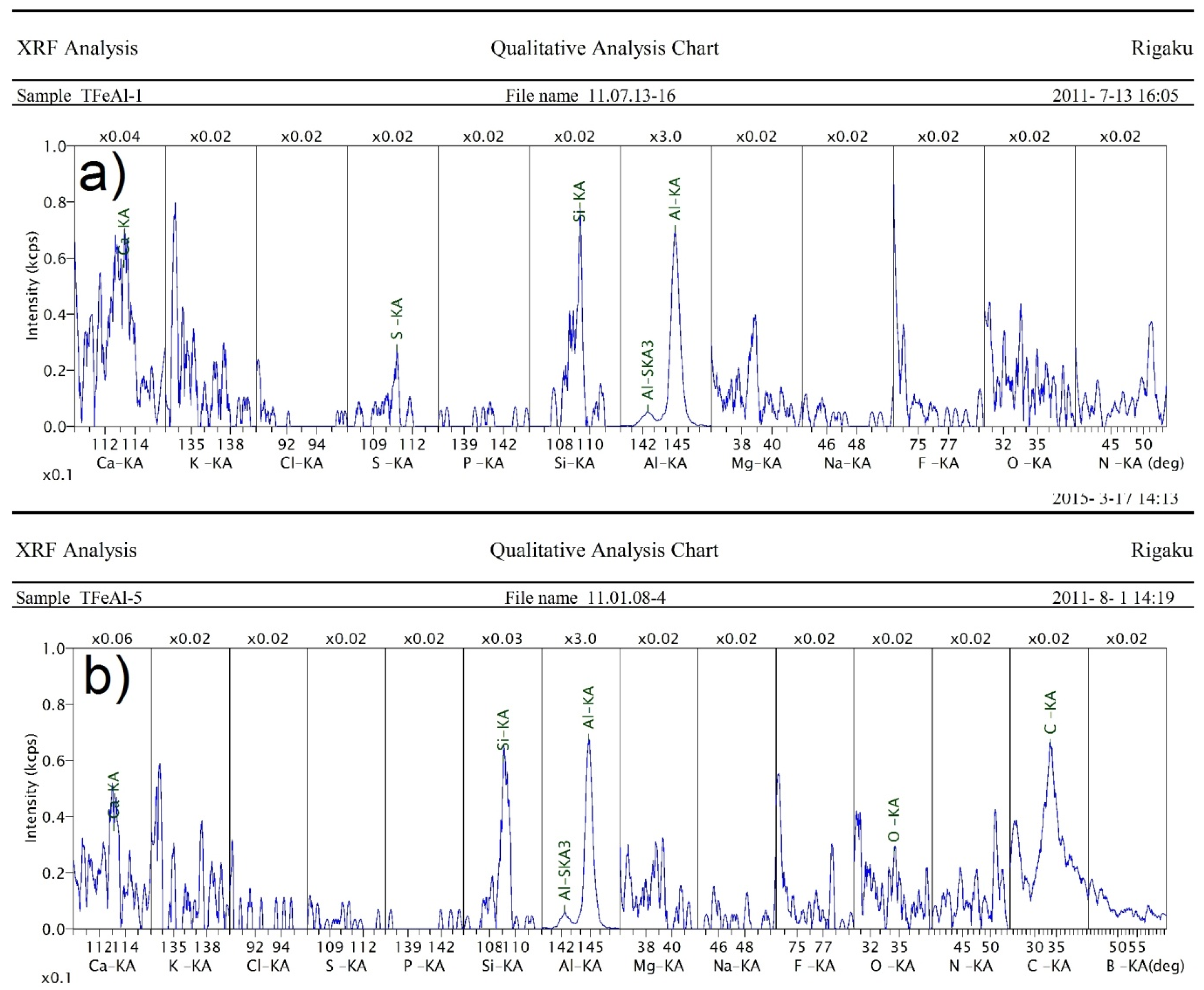
2.1. Characterization of the Iron and Aluminum Powders
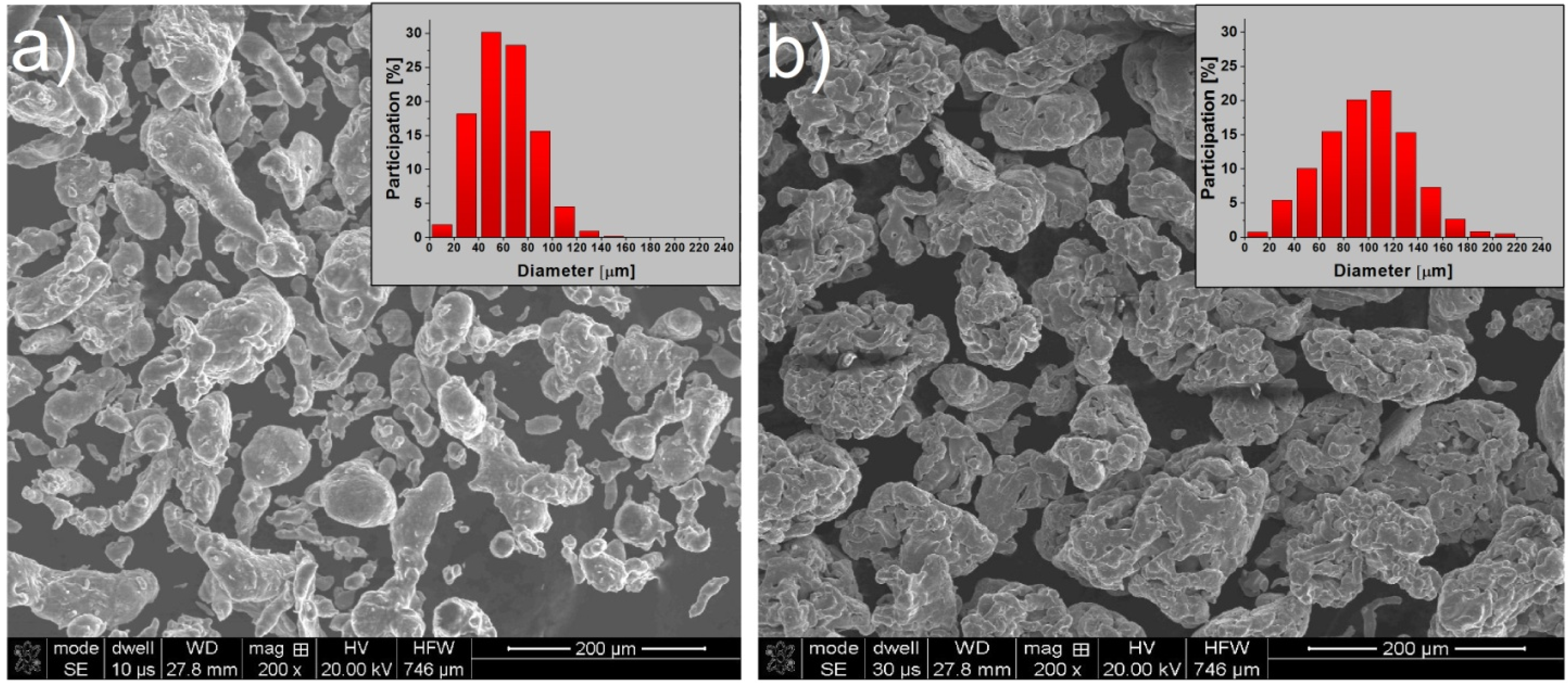
| Method | XRF | EDS | ||
|---|---|---|---|---|
| Power | Mean | Stand. dev. | Mean | Stand. dev. |
| Fe | 2.1% | 0.1 | 1.4% | 0.2 |
| Al | 2.3% | 0.1 | 1.2% | 0.3 |

2.2. Density, Porosity and Hardness of the Samples
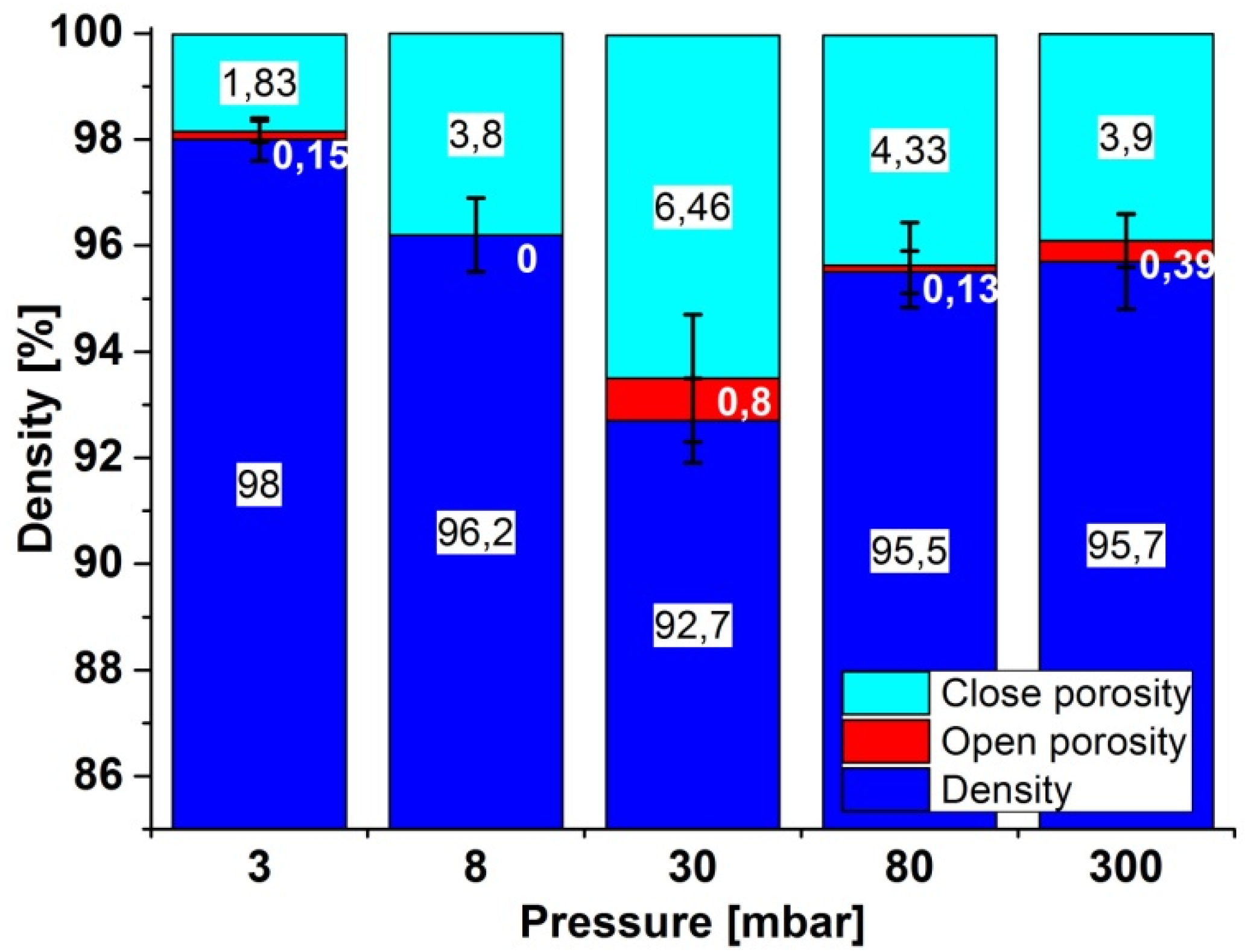
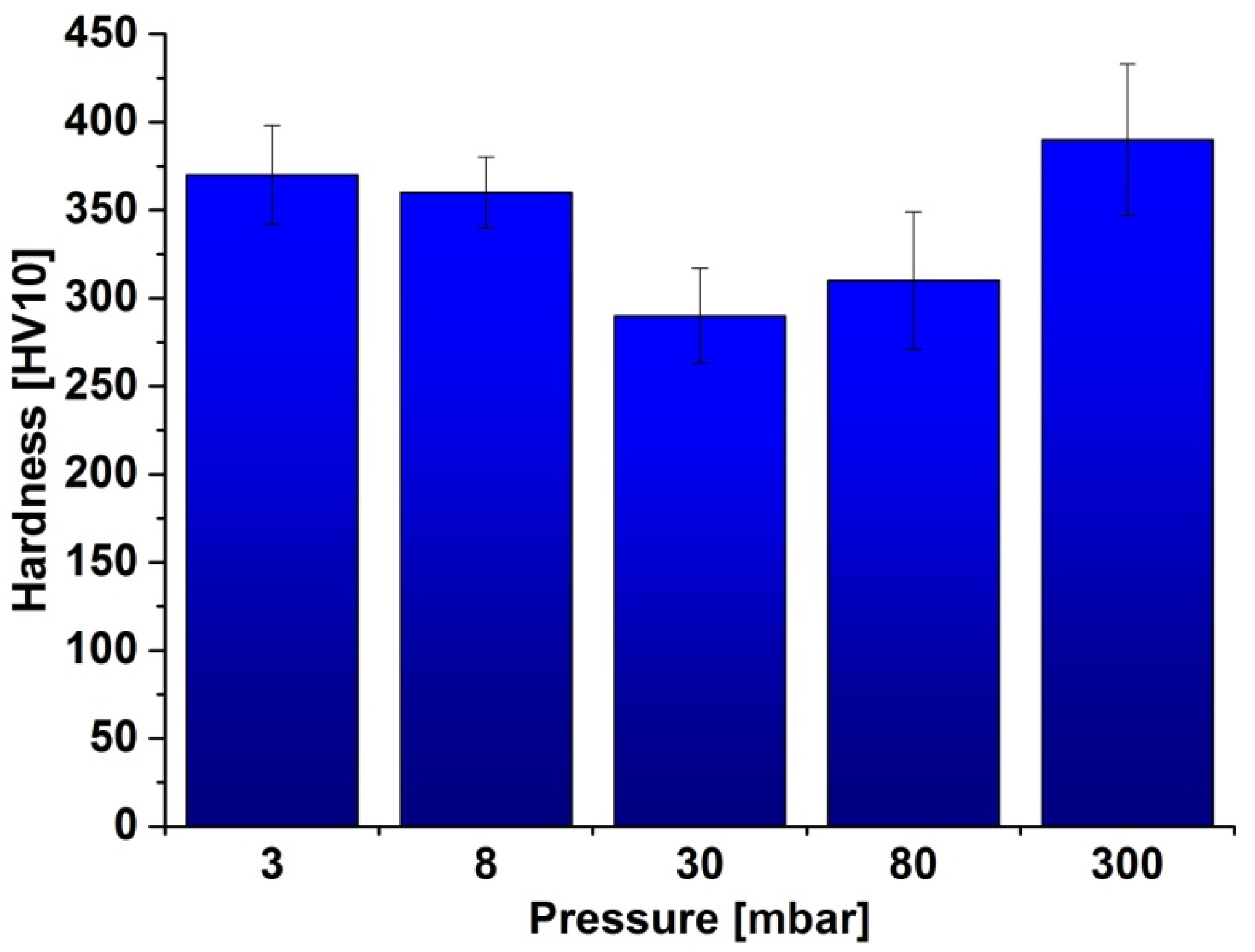
2.3. X-ray Diffraction Analysis
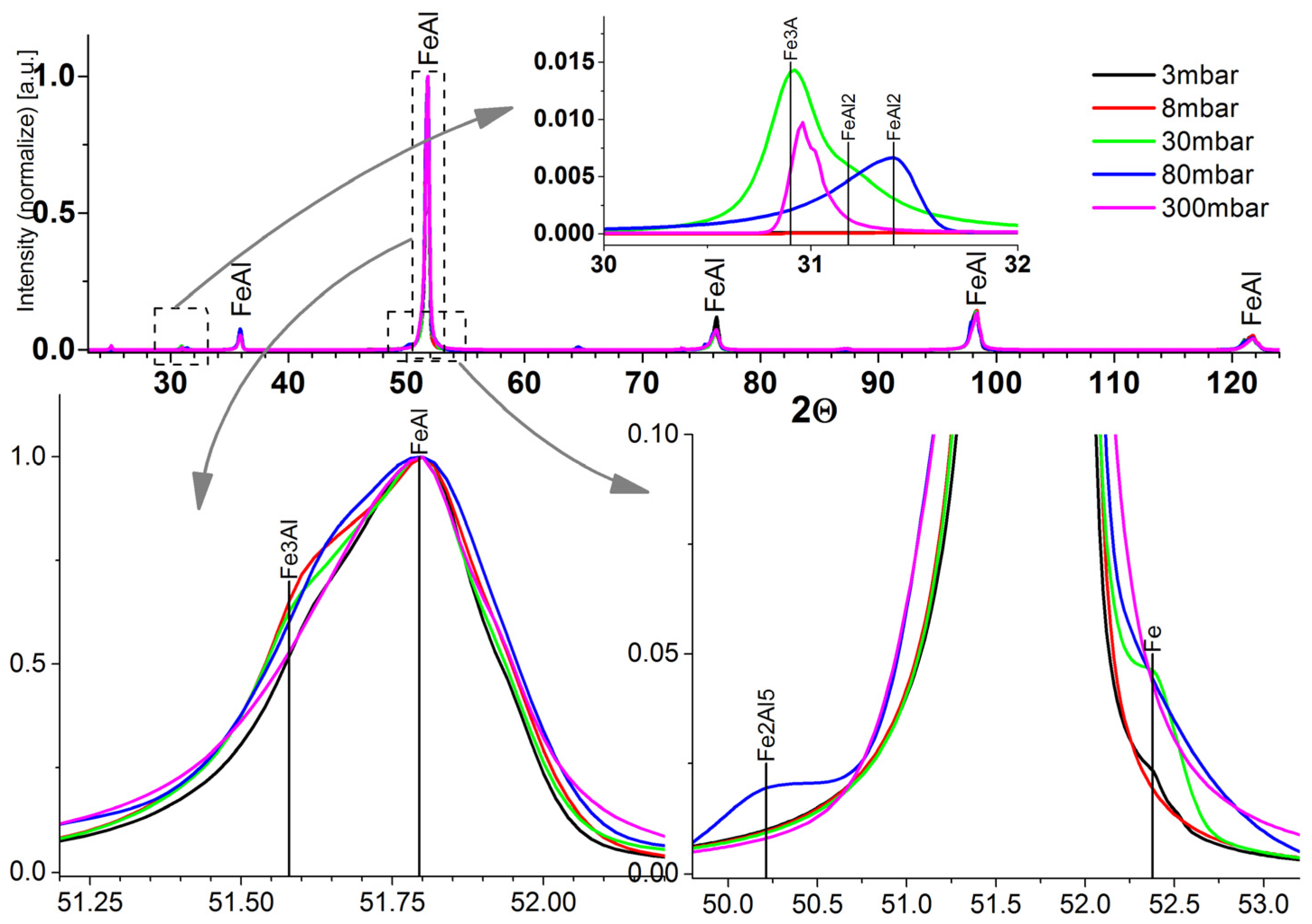
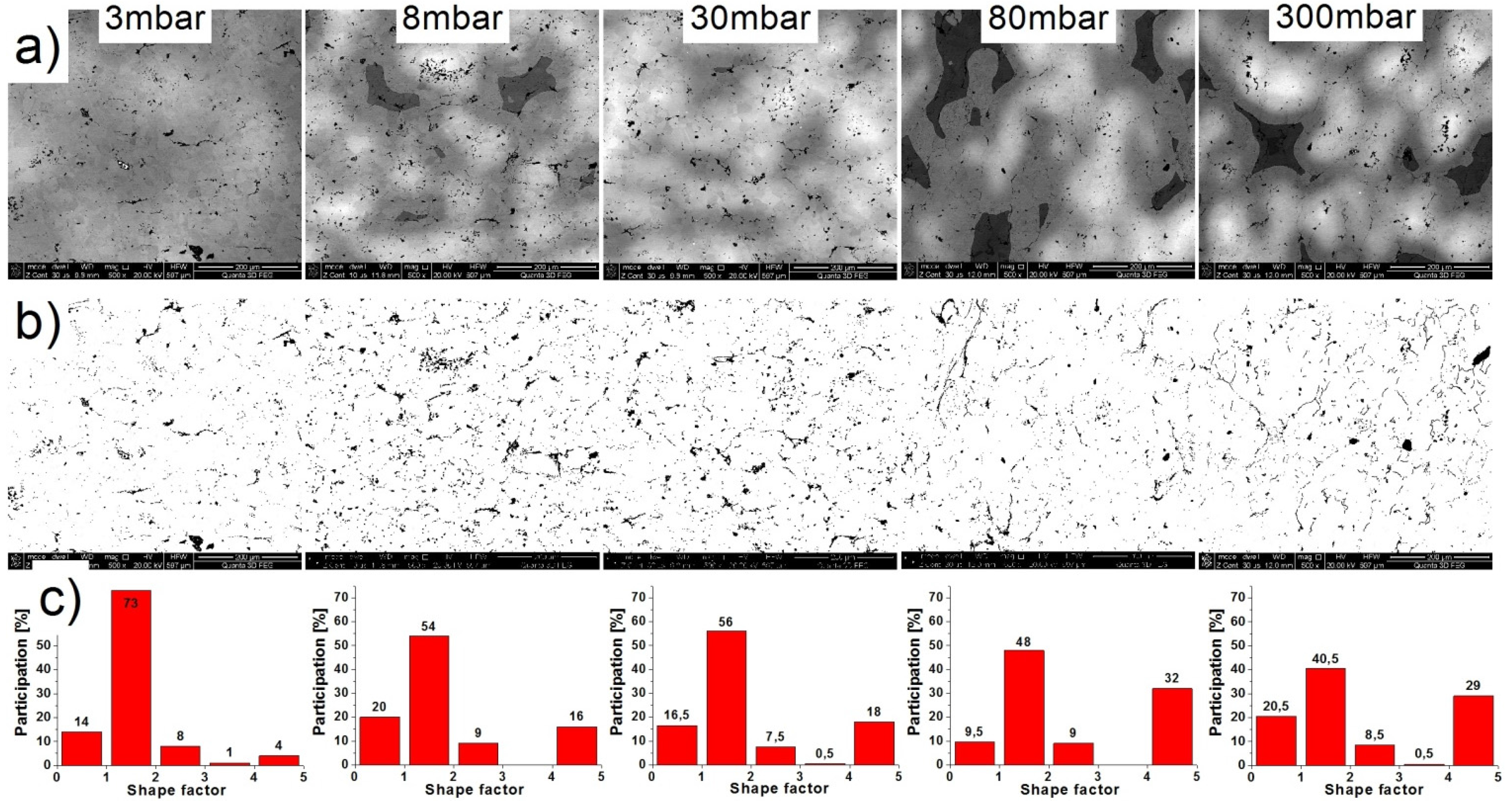
2.4. Microstructure, Chemical Composition, and Oxide Distribution
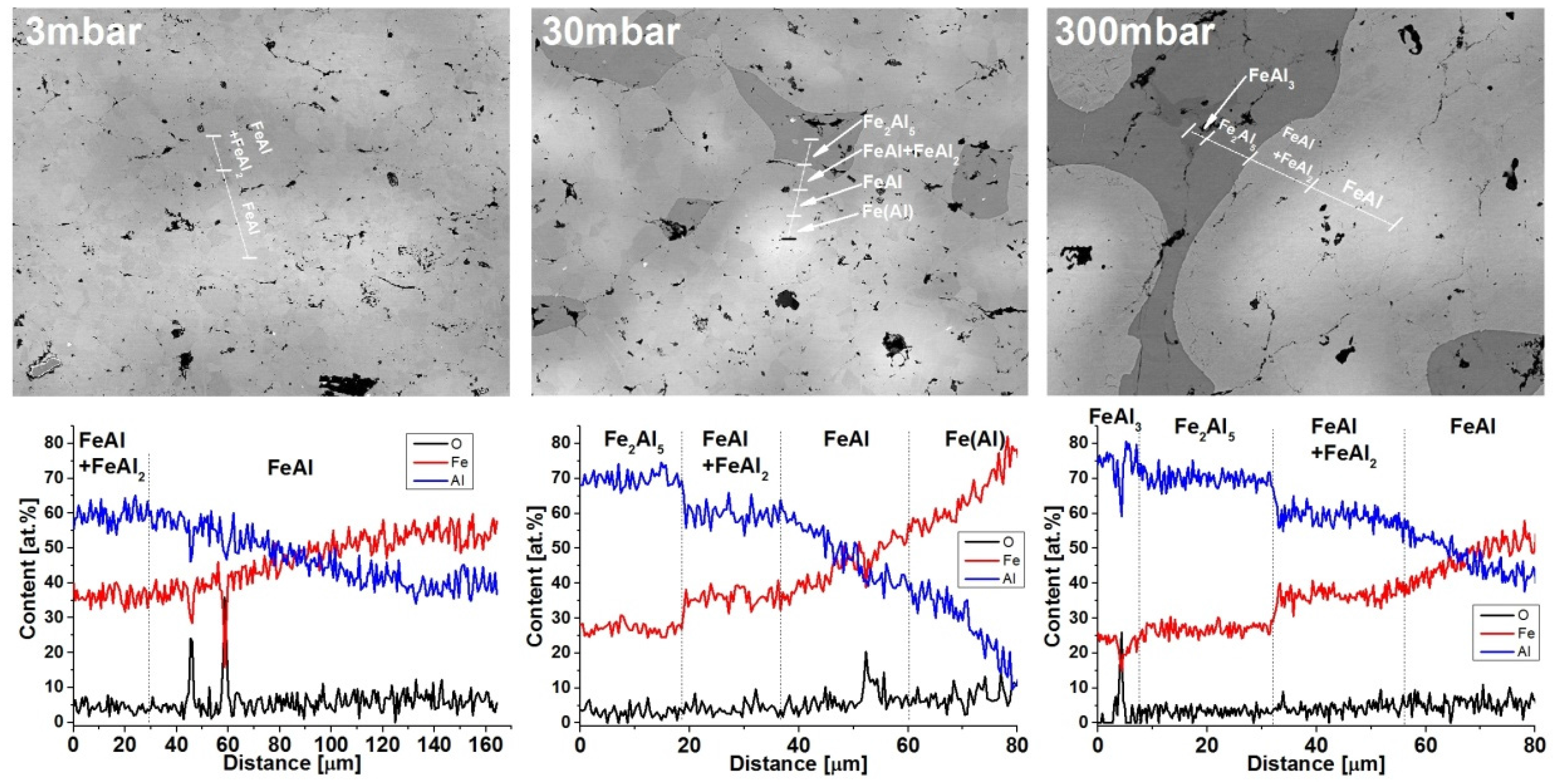
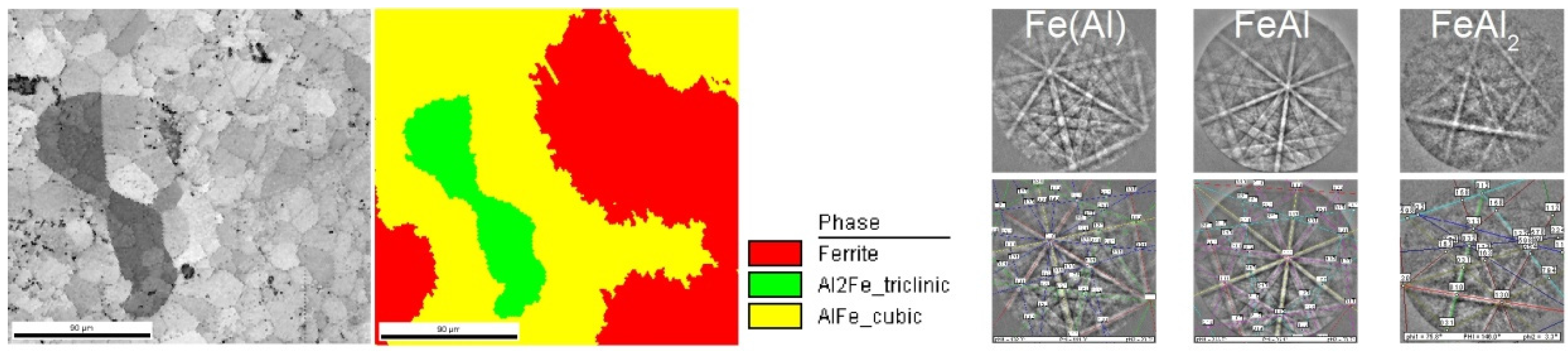
| Pressure | 3 mbar | 8 mbar | 30 mbar | 80 mbar | 300 mbar |
|---|---|---|---|---|---|
| XRF | 2.0% ± 0.4% | 2.3% ± 0.2% | 2.6% ± 0.2% | 1.7% ± 0.2% | 1.6% ± 0.2% |
| EDS | 1.4% ± 0.1% | 1.2% ± 0.05% | 1.3% ± 0.05% | 1.3% ± 0.05% | 1.2% ± 0.05% |
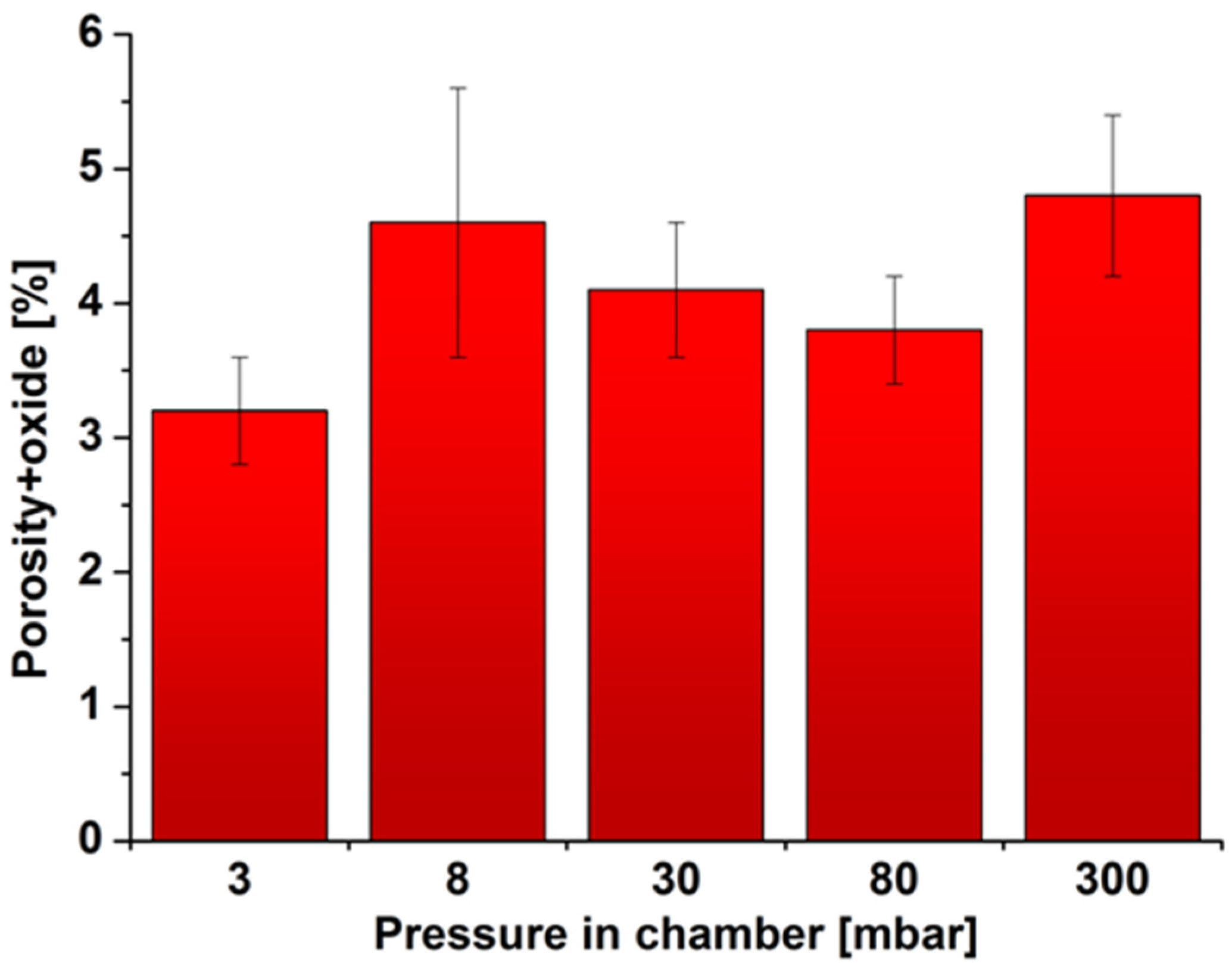
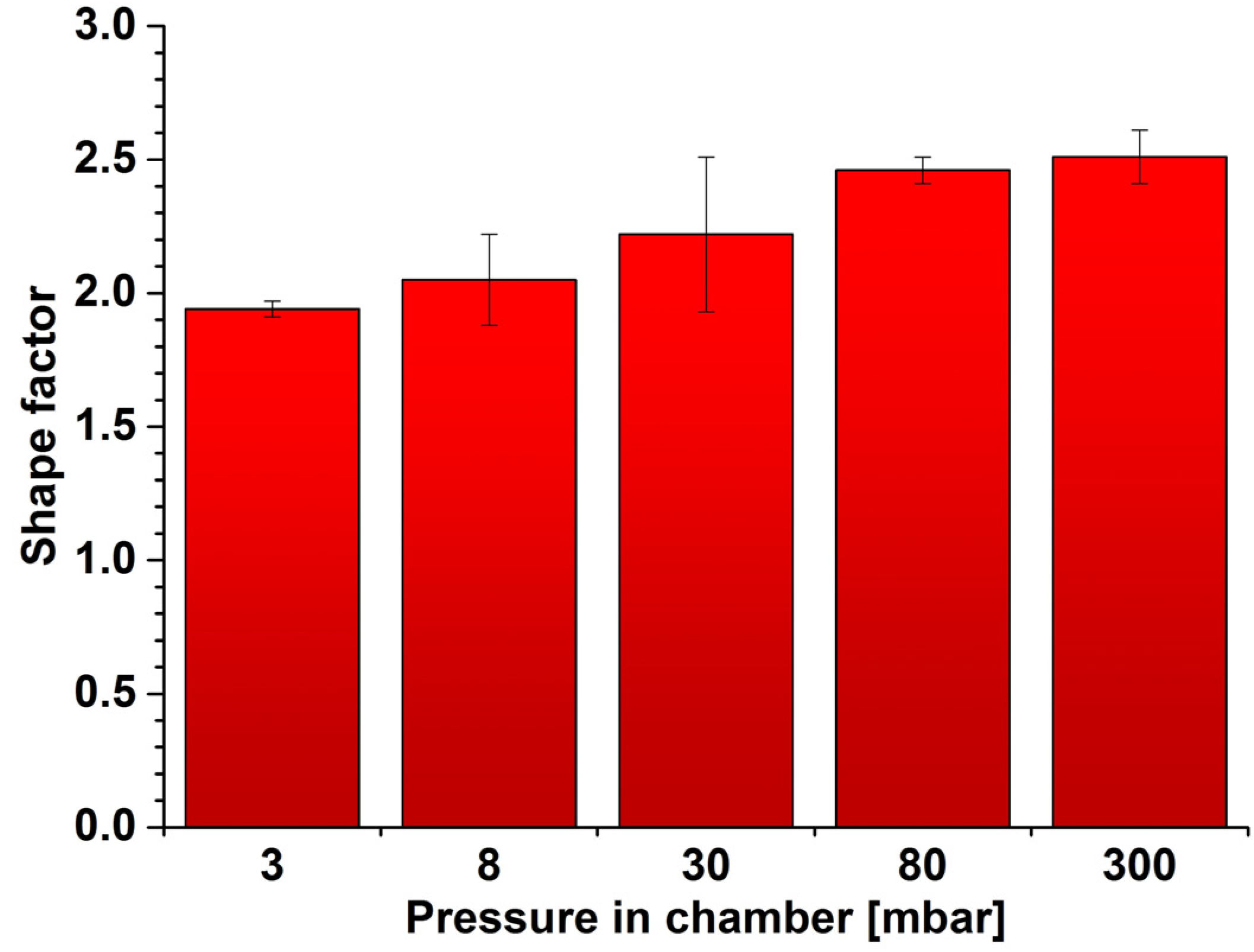

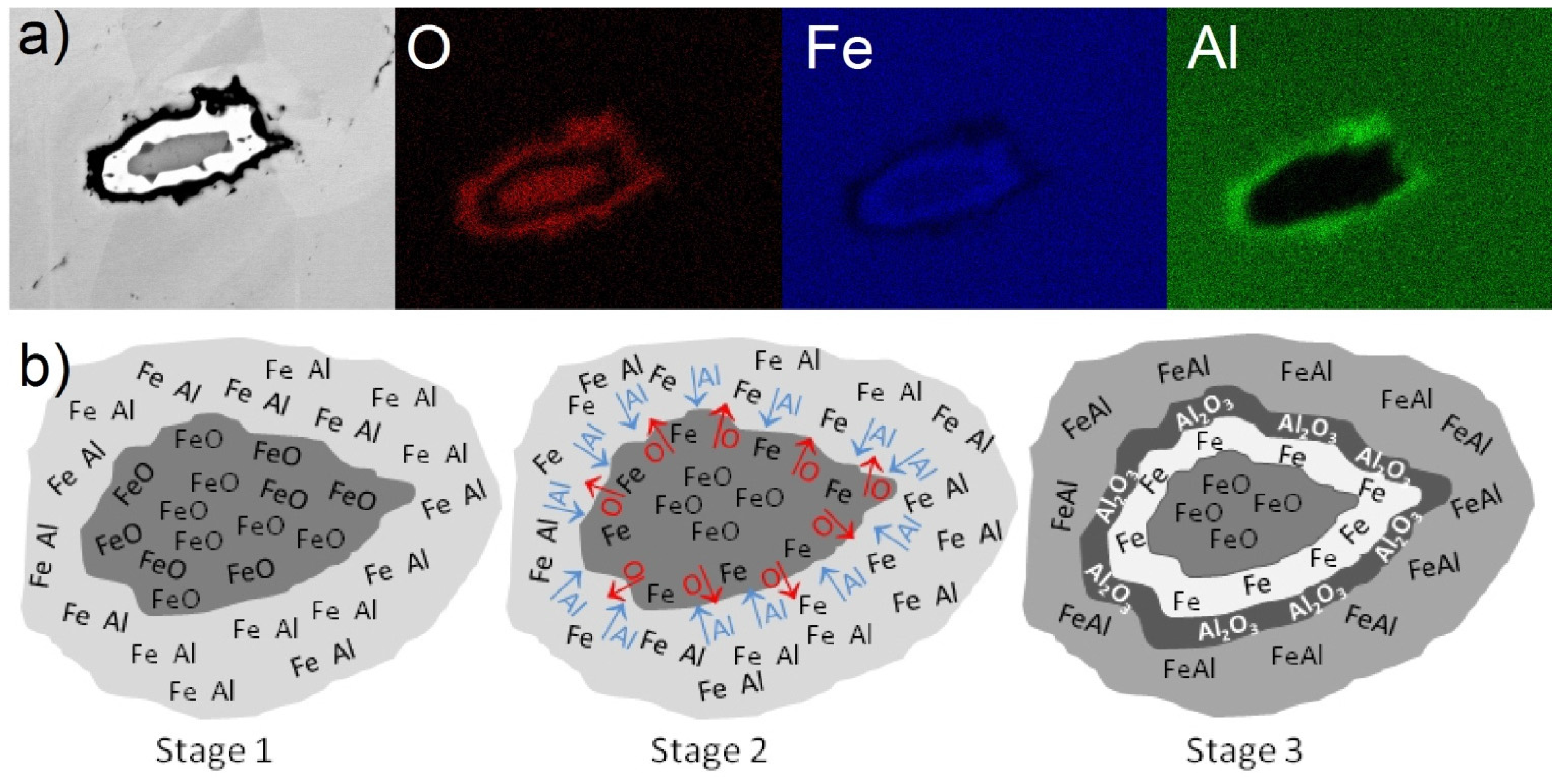
3. Materials and Methods
| Powder | The trade name | Average particle size | Percentile 10% | Percentile 90% | BET surface |
|---|---|---|---|---|---|
| [μm] | [μm] | [μm] | [m2/g] | ||
| Aluminum | AG 90/99.7 | 60.1 | 31.5 | 92.9 | 0.99 |
| Iron | NC 100.24 | 101.2 | 48.9 | 143.5 | 0.07 |
| Sintering temperature [°C] | Load [MPa] | Annealing time [min] | Average heating rate [°C/min] |
|---|---|---|---|
| 1000 | 60 | 5 | 400 |
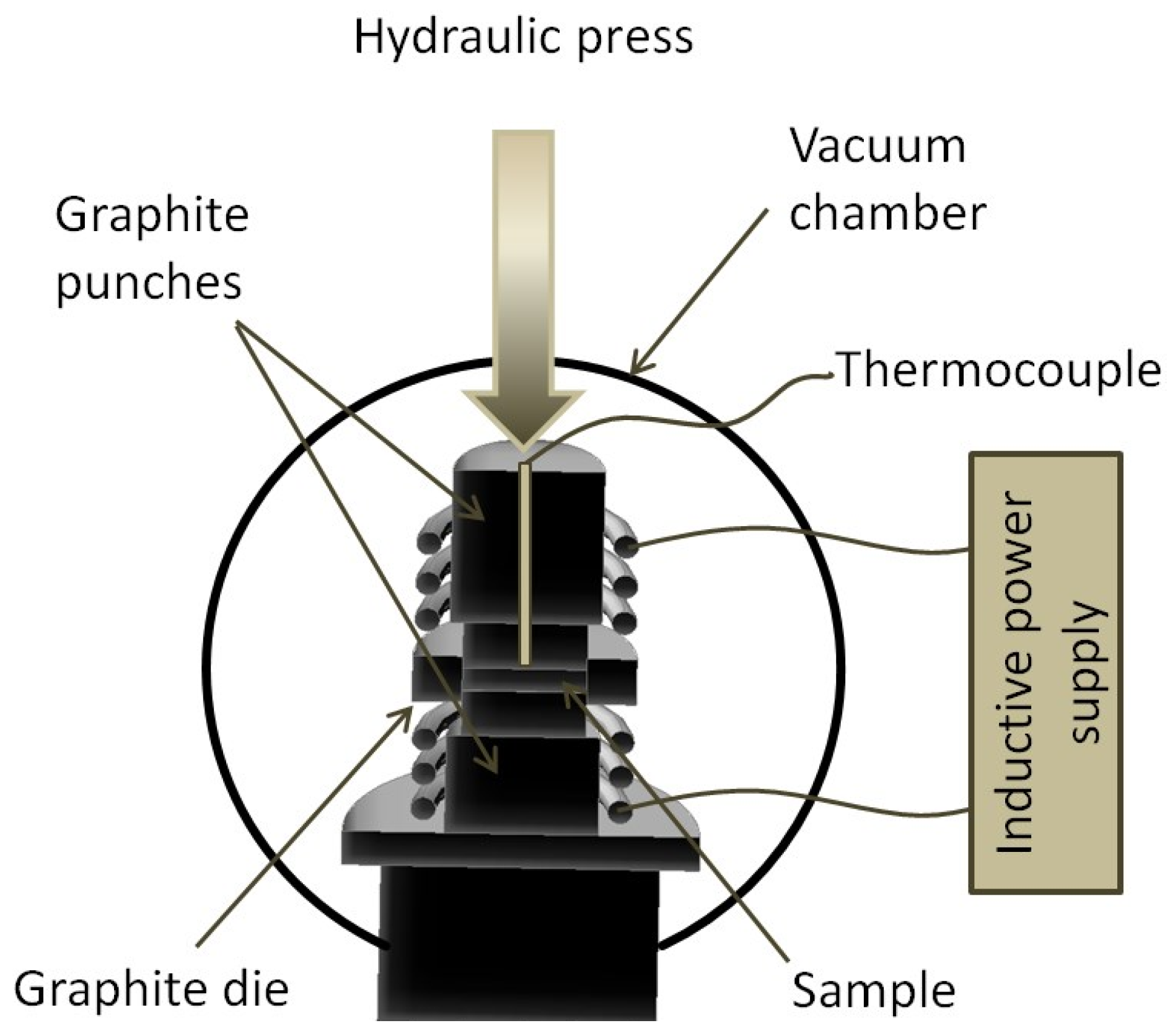
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, C.T.; Stringer, J.; Mundy, J.N.; Horton, L.L.; Angelini, P. Ordered intermetallic alloys: An assessment. Intermetallics 1997, 5, 579–596. [Google Scholar] [CrossRef]
- Deevi, S.C.; Sikka, V.K. Nickel and iron aluminides: An overview on properties, processing, and applications. Intermetallics 1996, 4, 357–375. [Google Scholar] [CrossRef]
- Liu, C.T.; George, E.P.; Maziasz, P.J.; Schneibel, J.H. Recent advances in B2 iron aluminide alloys: Deformation, fracture and alloy desing. Mater. Sci. Eng. A 1998, 258, 84–98. [Google Scholar] [CrossRef]
- Sikka, V.K.; Wilkening, D.; Liebetrau, J.; Mackey, B. Melting and casting of FeAl-based cast alloy. Mater. Sci. Eng. A 1998, 258, 229–235. [Google Scholar] [CrossRef]
- Sundar, R.S.; Baligidad, R.G.; Prasad, Y.V.R.K.; Sastry, D.H. Processing of iron aluminides. Mater. Sci. Eng. A 1998, 258, 219–228. [Google Scholar] [CrossRef]
- Josh, D.L.; Easton, D.S.; Liu, C.T.; Babu, S.S.; David, S.A. Processing of Fe3Al and FeAl alloys by reaction synthesis. Intermetallics 1995, 3, 467–481. [Google Scholar] [CrossRef]
- Durejko, T.; Bojar, Z. Materials based on intermetallic phases of the Fe-Al obtained by modified compression method at elevated temperature. Composites 2002, 2, 323–327. [Google Scholar]
- Jóźwiak, S.; Karczewski, K.; Bojar, Z. The effect of loading mode changes during the sintering process on the mechanical properties of FeAl intermetallic sinters. Intermetallics 2013, 33, 99–104. [Google Scholar] [CrossRef]
- Grabke, H.J. Oxidation of NiAl and FeAl. Intermetallics 1999, 7, 1153–1158. [Google Scholar] [CrossRef]
- Yamauchi, A.; Tsunekane, M.; Kurokawa, K.; Hanada, S.; Yoshimi, K. Influence of vacuum annealing conditions on the surface oxidation and vacancy condensation in the surface of an FeAl single crystal. Intermetallics 2010, 18, 412–416. [Google Scholar] [CrossRef]
- Hou, P.Y.; Niu, Y.; van Lienden, C. Analysis of pore formation at oxide–alloy interfaces—I: Experimental results on FeAl. Oxid. Metals 2003, 59, 41–61. [Google Scholar] [CrossRef]
- Siemiaszko, D.; Jóźwiak, S.; Czarnecki, M.; Bojar, Z. Influence of temperature during pressure-assisted induction sintering (PAIS) on structure and properties of the Fe40Al intermetallic phase. Intermetallics 2013, 41, 16–21. [Google Scholar] [CrossRef]
- Siemiaszko, D.; Szykut, M. Influence of eccentric arrangement of the die for the temperature distribution in the sample during the pressure-assisted induction sintering (PAIS). Key Eng. Mater. 2014, 514, 622–623. [Google Scholar]
- Gedevanishvili, S.; Deevi, S.C. Processing of iron aluminides by pressureless sintering through Fe+Al elemental route. Mater. Sci. Eng. A 2002, 325, 163–176. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siemiaszko, D.; Kowalska, B.; Jóźwik, P.; Kwiatkowska, M. The Effect of Oxygen Partial Pressure on Microstructure and Properties of Fe40Al Alloy Sintered under Vacuum. Materials 2015, 8, 1513-1525. https://doi.org/10.3390/ma8041513
Siemiaszko D, Kowalska B, Jóźwik P, Kwiatkowska M. The Effect of Oxygen Partial Pressure on Microstructure and Properties of Fe40Al Alloy Sintered under Vacuum. Materials. 2015; 8(4):1513-1525. https://doi.org/10.3390/ma8041513
Chicago/Turabian StyleSiemiaszko, Dariusz, Beata Kowalska, Paweł Jóźwik, and Monika Kwiatkowska. 2015. "The Effect of Oxygen Partial Pressure on Microstructure and Properties of Fe40Al Alloy Sintered under Vacuum" Materials 8, no. 4: 1513-1525. https://doi.org/10.3390/ma8041513
APA StyleSiemiaszko, D., Kowalska, B., Jóźwik, P., & Kwiatkowska, M. (2015). The Effect of Oxygen Partial Pressure on Microstructure and Properties of Fe40Al Alloy Sintered under Vacuum. Materials, 8(4), 1513-1525. https://doi.org/10.3390/ma8041513







