Dopamine-Mediated Sclerotization of Regenerated Chitin in Ionic Liquid
Abstract
:1. Introduction
2. Results and Discussion
2.1. Fabrication of Regenerated Chitin Films
2.2. Quantification of Melanin-Like Compound
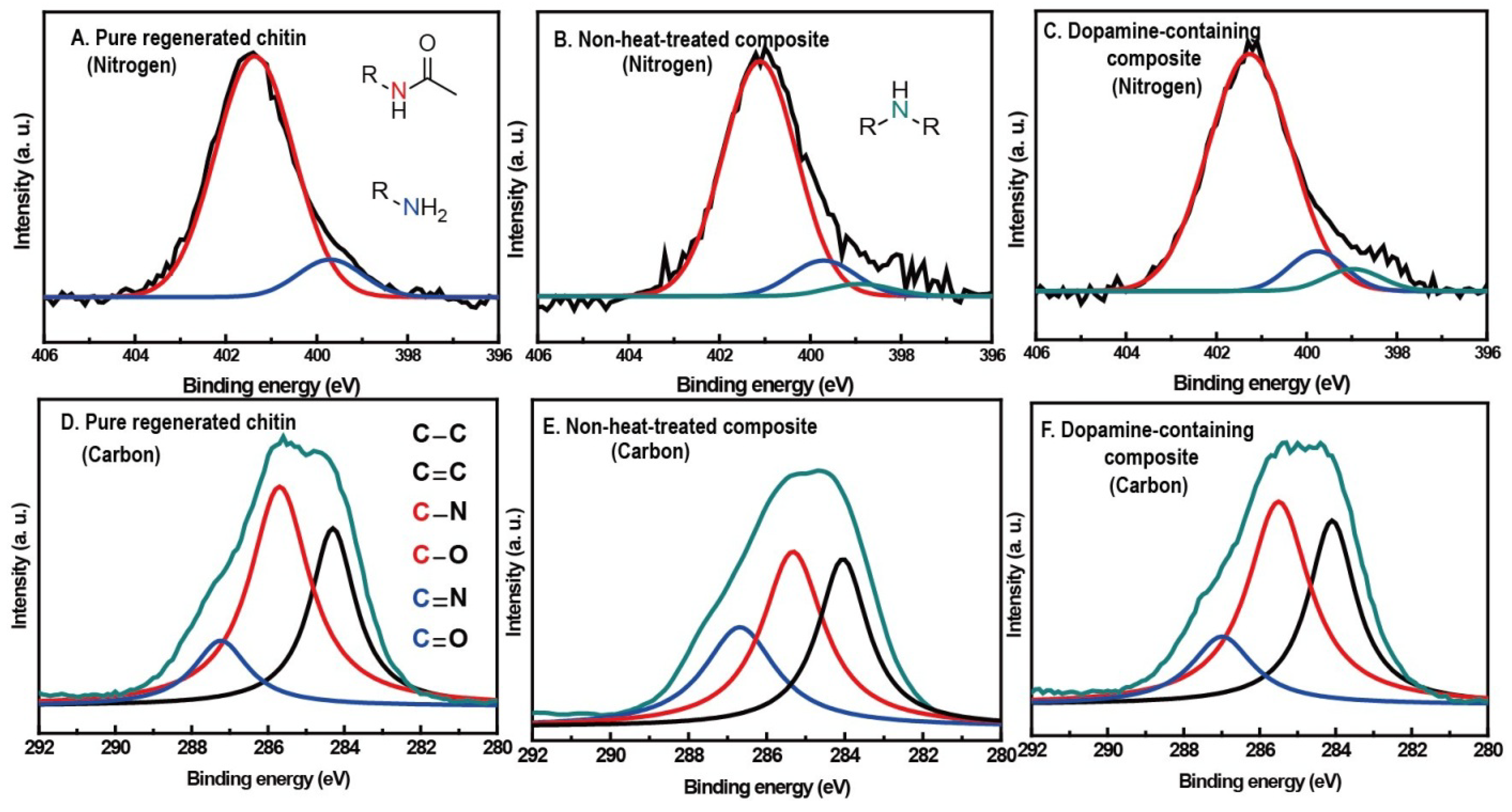
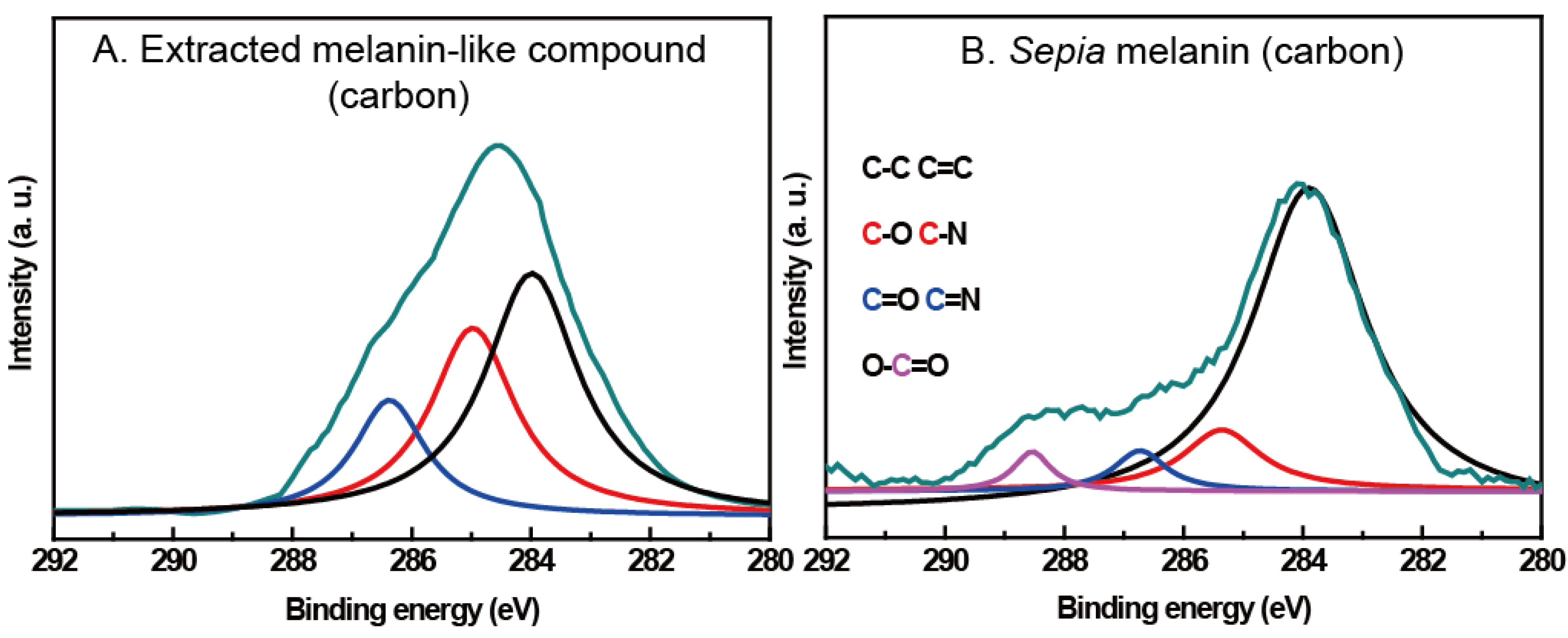
2.3. Spectral Evidences for Dopamine-Meditated Cross-Links and Melanin-Like Compound Formation
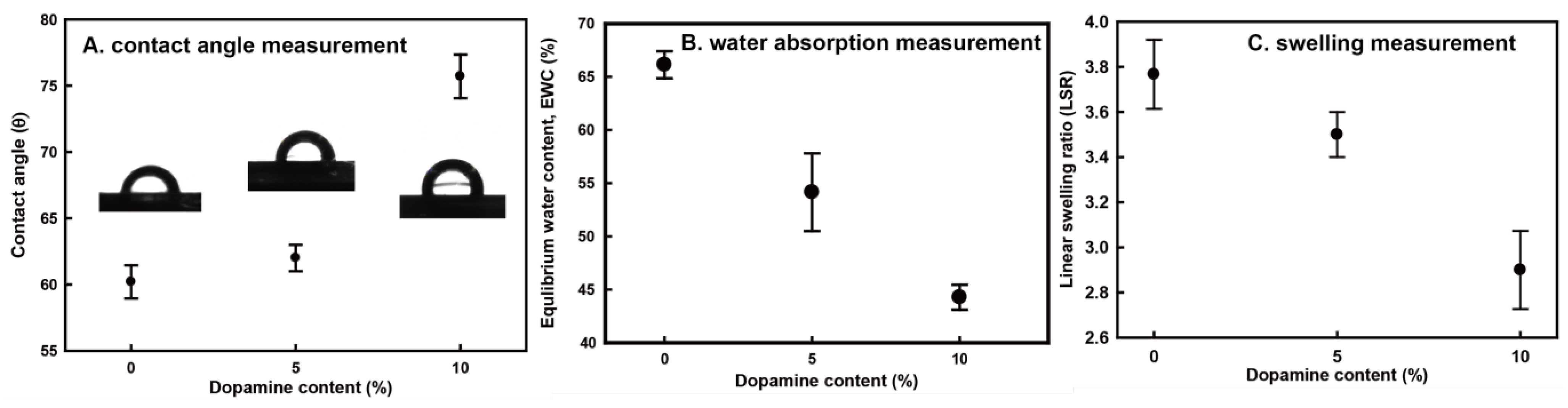
2.4. Contact Angle and Water Absorption
2.5. Crystal Structure and Morphology of Films

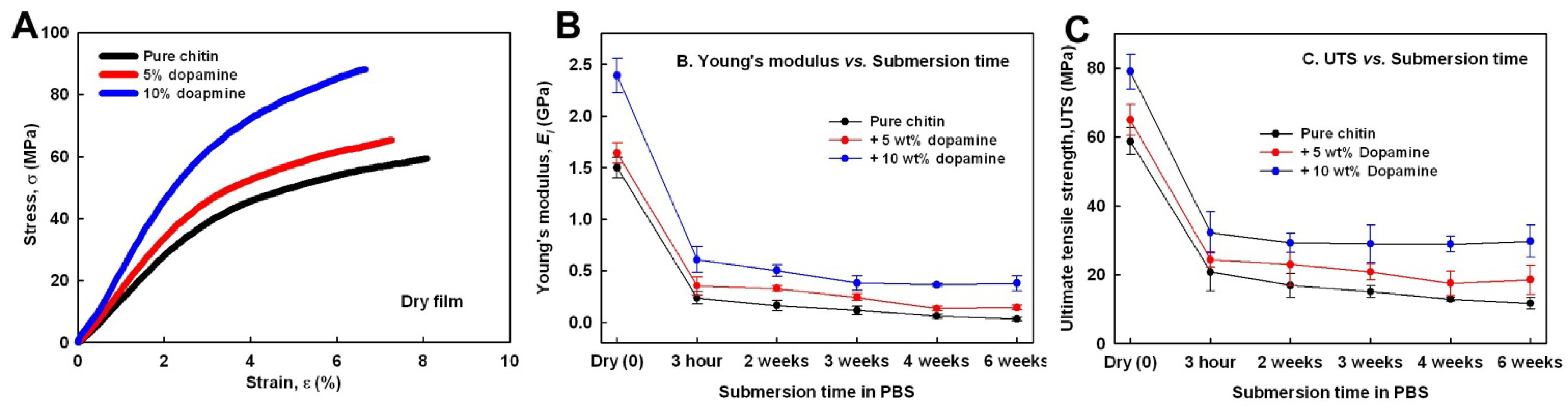
2.6. Mechanical Properties of Chitin and Composites in Wet Conditions
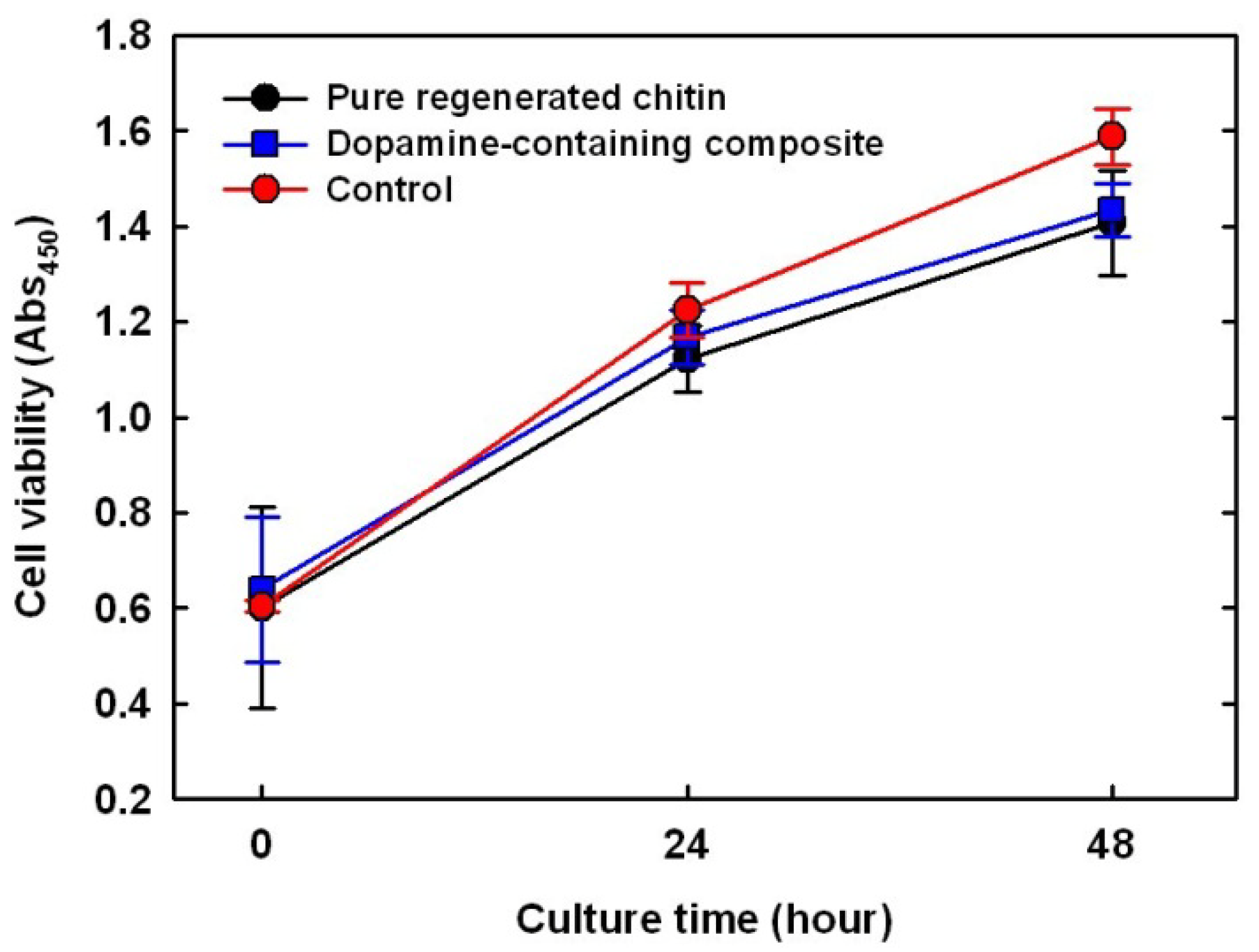
2.7. Cytotoxicity Test on Dopamine-Containing Composite
2.8. Contribution of Dopamine Oxidation to Improve Mechanical Properties and Resistance to Swelling in Wet Conditions
3. Experimental Section
3.1. Material
3.2. Regenerated Films of Pure Chitin and Composites
3.3. Quantification of Melanin-Like Compound and Solubility of Chitin and Composites in DMAc/LiCl Solvent
3.4. Chemistry Characterization on Film Surface
3.5. Characterization of Mechanical Properties
3.6. Characterization of Water Swelling and Hydrophobicity
3.7. Characterization of Morphology and Crystal Structure
3.8. In Vitro Cell Culture Study
4. Conclusions
Acknowledgments
Conflicts of Interest
Appendix

References
- European Chitin Society. Advances in Chitin Science; Universität Potsdam: Potsdam, Germany, 2000. [Google Scholar]
- Nishino, T.; Matsui, R.; Nakamae, K. Elastic modulus of the crystalline regions of chitin and chitosan. J. Polym. Sci. B Polym. Phys. 1999, 37, 1191–1196. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar]
- Zheng, H.; Zhou, J.; Du, Y.; Zhang, L. Cellulose/chitin films blended in NaOH/urea aqueous solution. J. Appl. Polym. Sci. 2002, 86, 1679–1683. [Google Scholar] [CrossRef]
- Pillai, C.K.S.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, J.; Wang, X.; Huang, Y.; Wang, Y. Dissolution behavior of chitin in ionic liquid. J. Macromol. Sci. B Phys. 2010, 49, 528–541. [Google Scholar] [CrossRef]
- Jaworska, M.M.; Kozlecki, T.; Gorak, A. Review of the application of ionic liquids as solvents for chitin. J. Polym. Eng. 2012, 32, 67–69. [Google Scholar]
- Bochek, A.M.; Murav’ev, A.A.; Novoselov, N.P.; Zaborski, M.; Zabivalova, N.M.; Petrova, V.A.; Vlasova, E.N.; Volchek, B.Z.; Lavrent’ev, V.K. Specific features of cellulose and chitin dissolution in ionic liquids of varied structure and the structural organization of regenerated polysaccharides. Russ. J. Appl. Chem. 2012, 85, 1718–1725. [Google Scholar] [CrossRef]
- Prasad, K.; Murakami, M.A.; Kaneko, Y.; Takada, A.; Nakamura, Y.; Kadokawa, J. Weak gel of chitin with ionic liquid, 1-allyl-3-methylimidazolium bromide. Int. J. Biol. Macromol. 2009, 45, 221–225. [Google Scholar] [CrossRef]
- Qin, Y.; Lu, X.; Sun, N.; Rogers, R.D. Dissolution or extraction of crustacean shells using ionic liquids to obtain high molecular weight purified chitin and direct production of chitin films and fibers. Green Chem. 2010, 12, 968–971. [Google Scholar] [CrossRef]
- Sato, M.; Maeda, M.; Kurosawa, H.; Inoue, Y.; Yamauchi, Y.; Iwase, H. Reconstruction of rabbit Achilles tendon with three bioabsorbable materials: Histological and biomechanical studies. J. Orthop. Sci. 2000, 5, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.F.V.; Wegst, U.G.K. Design and mechanical properties of insect cuticle. Arthropod Struct. Dev. 2004, 33, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Pryor, M.G.M.; Russell, P.B.; Todd, A.R. Phenolic substances concerned in hardening the insect cuticle. Nature 1947, 159, 399–400. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, J.; Kramer, K.J.; Garbow, J.R.; Jacob, G.S.; Stejskal, E.O.; Hopkins, T.L.; Speirs, R.D. Aromatic cross-links in insect cuticle: Detection by solid-state 13C and 15N NMR. Science 1987, 235, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Beament, J.W.L. Wetting properties of insect cuticle. Nature 1960, 186, 408–409. [Google Scholar] [CrossRef]
- Miserez, A.; Li, Y.; Waite, J.H.; Zok, F. Jumbo squid beaks: Inspiration for design of robust organic composites. Acta Biomater. 2007, 3, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Miserez, A.; Schneberk, T.; Sun, C.; Zok, F.W.; Waite, J.H. The transition from stiff to compliant materials in squid beaks. Science 2008, 319, 1816–1819. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, T.L.; Morgan, T.D.; Aso, Y.; Kramer, K.J. N-β-alanyldopamine: Major role in insect cuticle tanning. Science 1982, 217, 364–366. [Google Scholar] [CrossRef] [PubMed]
- Eybl, E.; Griesmacher, A.; Grimm, M.; Wolner, E. Toxic effects of aldehydes released from fixed pericardium on bovine aortic endothelial cells. J. Biomed. Mater. Res. 1989, 23, 1355–1365. [Google Scholar] [CrossRef] [PubMed]
- Gendler, E.; Gendler, S.; Nimni, M.E. Toxic reactions evoked by glutaraldehyde-fixed pericardium and cardiac valve tissue bioprosthesis. J. Biomed. Mater. Res. 1984, 18, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Miessner, M.; Peter, M.G.; Vincent, J.F.V. Preparation of insect-cuticle-like biomimetic materials. Biomacromolecules 2001, 2, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.S.G.; Forrester, J.V.; McInnes, C.; Lawrie, F. Adhesion of cells to polystyrene surfaces. J. Cell. Biol. 1983, 97, 1500–1506. [Google Scholar] [CrossRef] [PubMed]
- Ju, K.Y.; Lee, Y.; Lee, S.; Park, S.B.; Lee, J.K. Bioinspired polymerization of dopamine to generate melanin-like nanoparticles having an excellent free-radical-scavenging property. Biomacromolecules 2011, 12, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Moses, D.N.; Harreld, J.H.; Stucky, G.D.; Waite, J.H. Melanin and Glycera jaws: Emerging dark side of a robust biocomposite structure. J. Biol. Chem. 2006, 281, 34826–34832. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Lee, Y.; Hwang, J.W.; Hong, S.; Kim, C.; Park, T.G.; Lee, H.; Hong, S.H. High-strength carbon nanotube fibers fabricated by infiltration and curing of mussel-inspired catecholamine polymer. Adv. Mater. 2011, 23, 1971–1975. [Google Scholar] [CrossRef] [PubMed]
- Pradal, C.; Kithva, P.; Martin, D.; Trau, M.; Grøndahl, L. Improvement of the wet tensile properties of nanostructured hydroxyapatite and chitosan biocomposite films through hydrophobic modification. J. Mater. Chem. 2011, 21, 2330–2337. [Google Scholar] [CrossRef]
- Zhang, Y.; Thomas, Y.; Kim, E.; Payne, G.F. pH- and voltage-responsive chitosan hydrogel through covalent cross-linking with catechol. J. Phys. Chem. B 2012, 116, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Lieder, R.; Darai, M.; Thor, M.B.; Ng, C.H.; Einarsson, J.M.; Gudmundsson, S.; Helgason, B.; Gaware, V.S.; Másson, M.; Gíslason, J.; et al. In vitro bioactivity of different degree of deacetylation chitosan, a potential coating material for titanium implants. J. Biomed. Mater. Res. A 2012, 100, 3392–3399. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.D.; Saito, T.; Fan, Y.; Isogai, A. Multifunctional coating films by layer-by-layer deposition of cellulose and chitin nanofibrils. Biomacromolecules 2012, 13, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, J.; Zhang, J.; Li, H.; Chen, P.; Gu, Q.; Wang, Z. Thermo-mechanical properties of the composite made of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) and acetylated chitin nanocrystals. Carbohydr. Polym. 2013, 95, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.X.; Hwang, D.S. A biomimetic chitosan composite with improved mechanical properties in wet conditions. Biotechnol. Prog. 2013, 29, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, L.; Xu, T. Novel aromatic proton-exchange polyelectrolytes via polyacylation of pre-sulfonated monomers. J. Mater. Chem. 2012, 22, 13996–14000. [Google Scholar] [CrossRef]
- Wu, Y.; Sasaki, T.; Irie, S.; Sakurai, K. A novel biomass-ionic liquid platform for the utilization of native chitin. Polymer 2008, 49, 2321–2327. [Google Scholar] [CrossRef]
- Chandrashekar, N.; Mansouri, H.; Slauterbeck, J.; Hashemi, J. Sex-based differences in the tensile properties of the human anterior cruciate ligament. J. Biomech. 2006, 39, 2943–2950. [Google Scholar] [CrossRef] [PubMed]
- Fratzl, P.; Weinkamer, R. Nature’s hierarchical materials. Prog. Mater. Sci. 2007, 52, 1263–1334. [Google Scholar] [CrossRef]
- Kadokawa, J.I.; Takegawa, A.; Mine, S.; Prasad, K. Preparation of chitin nanowhiskers using an ionic liquid and their composite materials with poly(vinyl alcohol). Carbohydr. Polym. 2011, 84, 1408–1412. [Google Scholar] [CrossRef]
- Cheng, S.T.; Chen, Z.F.; Chen, G.Q. The expression of cross-linked elastin by rabbit blood vessel smooth muscle cells cultured in polyhydroxyalkanoate scaffolds. Biomaterials 2008, 29, 4187–4194. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oh, D.X.; Shin, S.; Lim, C.; Hwang, D.S. Dopamine-Mediated Sclerotization of Regenerated Chitin in Ionic Liquid. Materials 2013, 6, 3826-3839. https://doi.org/10.3390/ma6093826
Oh DX, Shin S, Lim C, Hwang DS. Dopamine-Mediated Sclerotization of Regenerated Chitin in Ionic Liquid. Materials. 2013; 6(9):3826-3839. https://doi.org/10.3390/ma6093826
Chicago/Turabian StyleOh, Dongyeop X., Sara Shin, Chanoong Lim, and Dong Soo Hwang. 2013. "Dopamine-Mediated Sclerotization of Regenerated Chitin in Ionic Liquid" Materials 6, no. 9: 3826-3839. https://doi.org/10.3390/ma6093826
APA StyleOh, D. X., Shin, S., Lim, C., & Hwang, D. S. (2013). Dopamine-Mediated Sclerotization of Regenerated Chitin in Ionic Liquid. Materials, 6(9), 3826-3839. https://doi.org/10.3390/ma6093826



