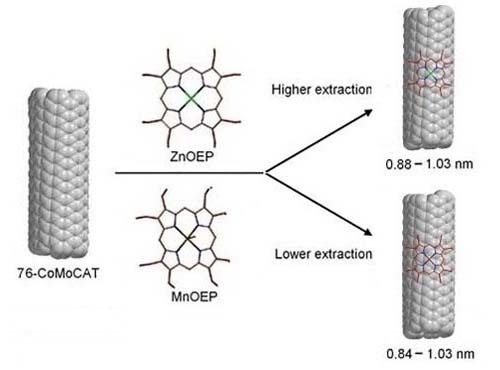
Enrichment of Large-Diameter Single-Walled Carbon Nanotubes (SWNTs) with Metallo-Octaethylporphyrins
Abstract
:1. Introduction
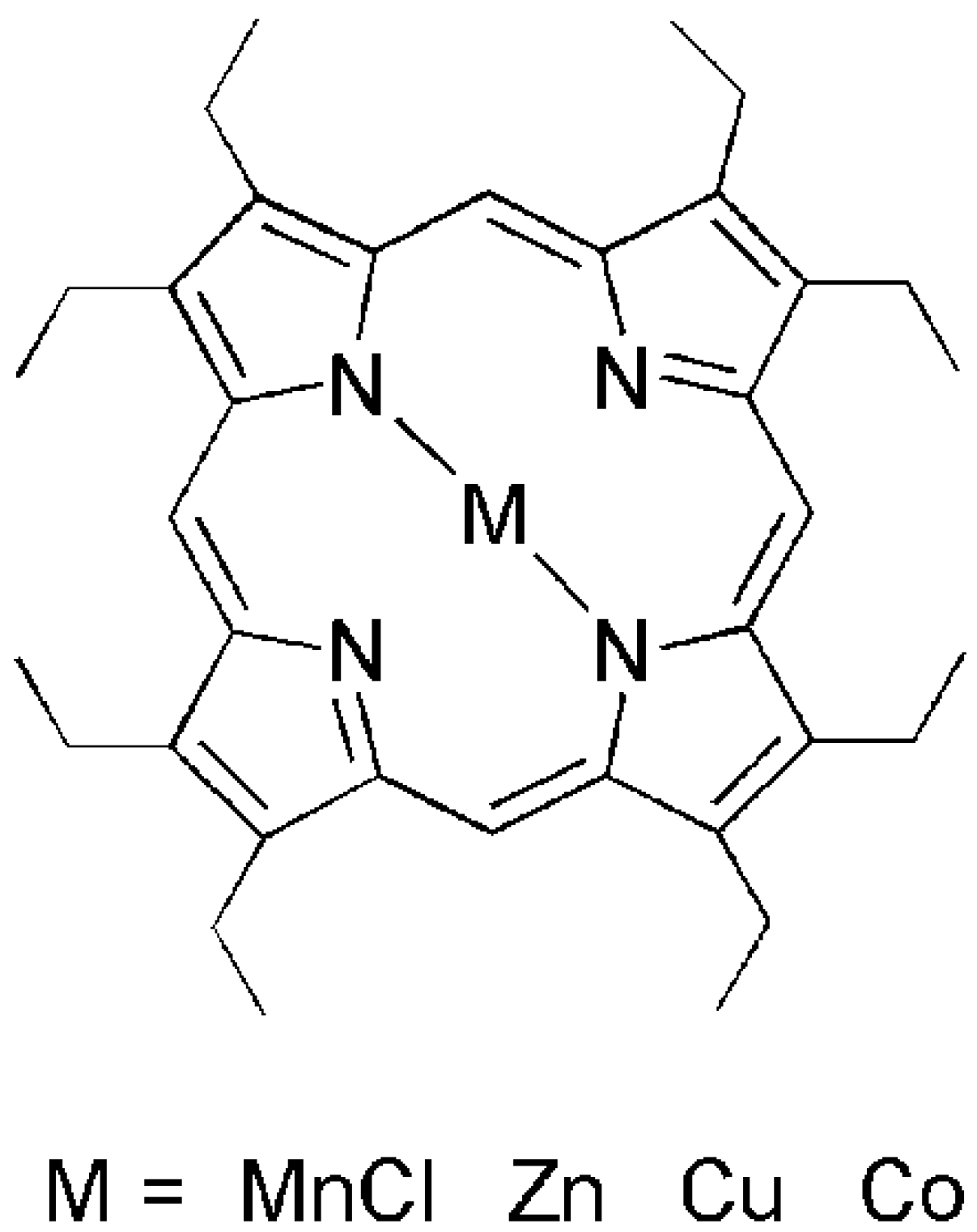
2. Results and Discussion
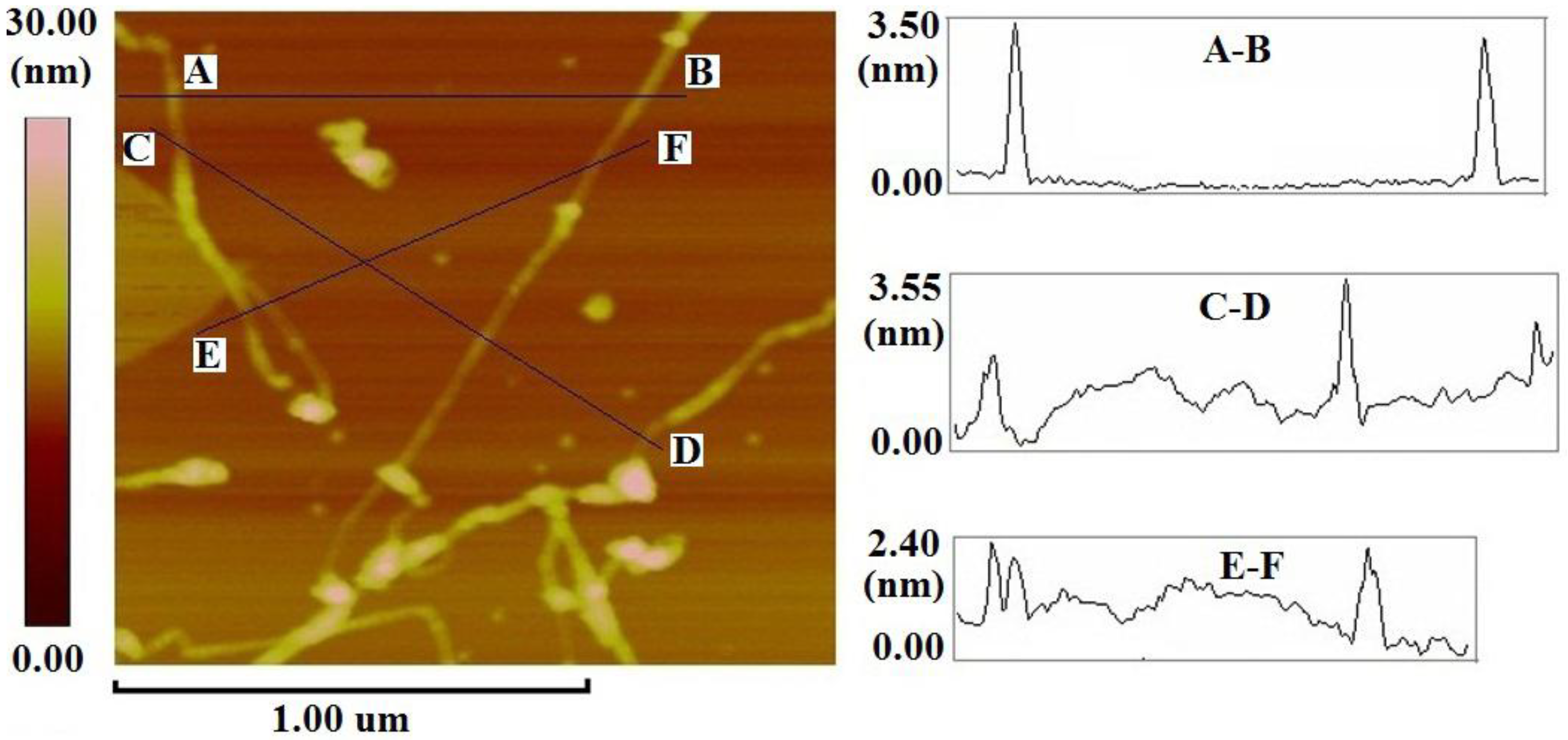
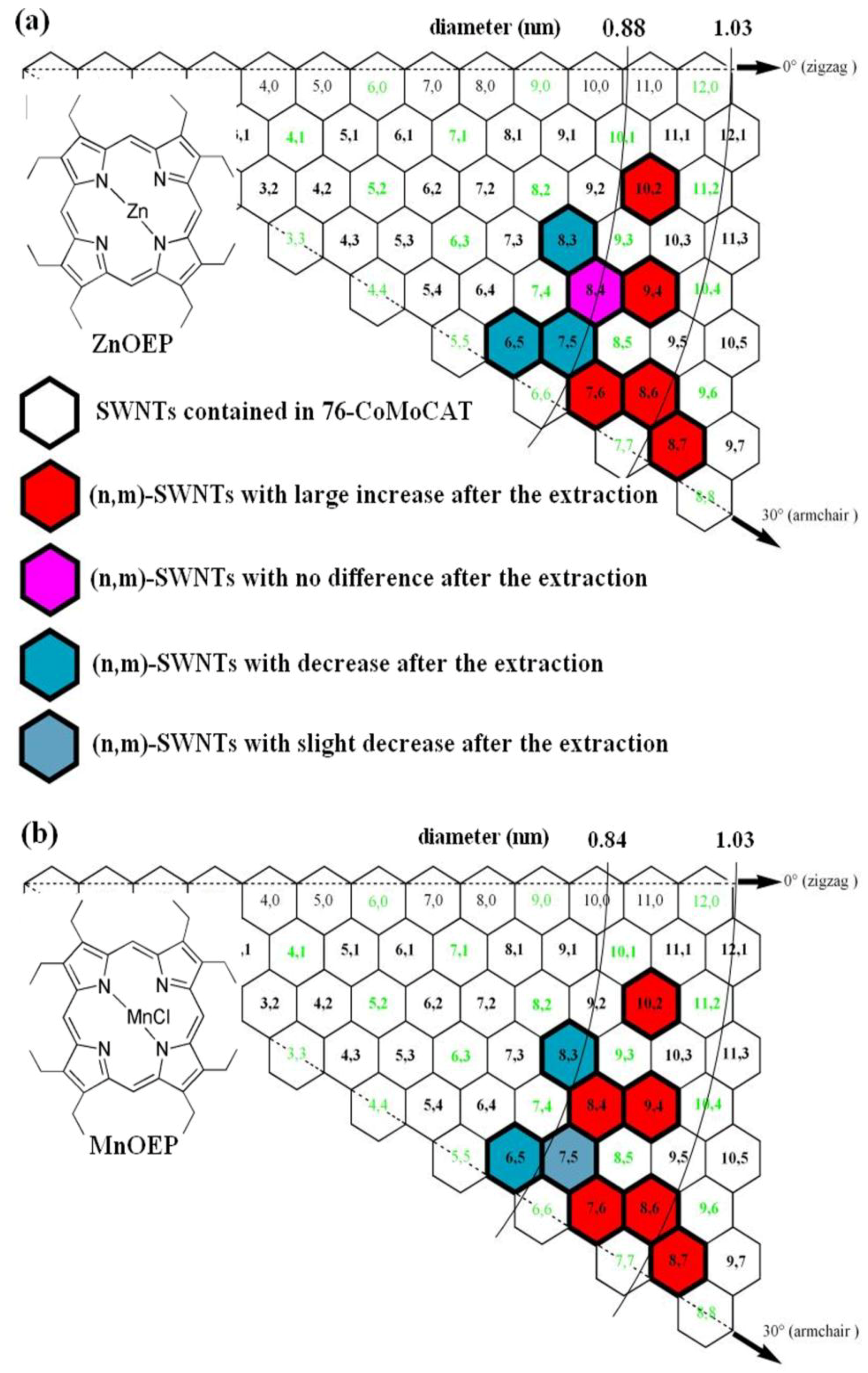
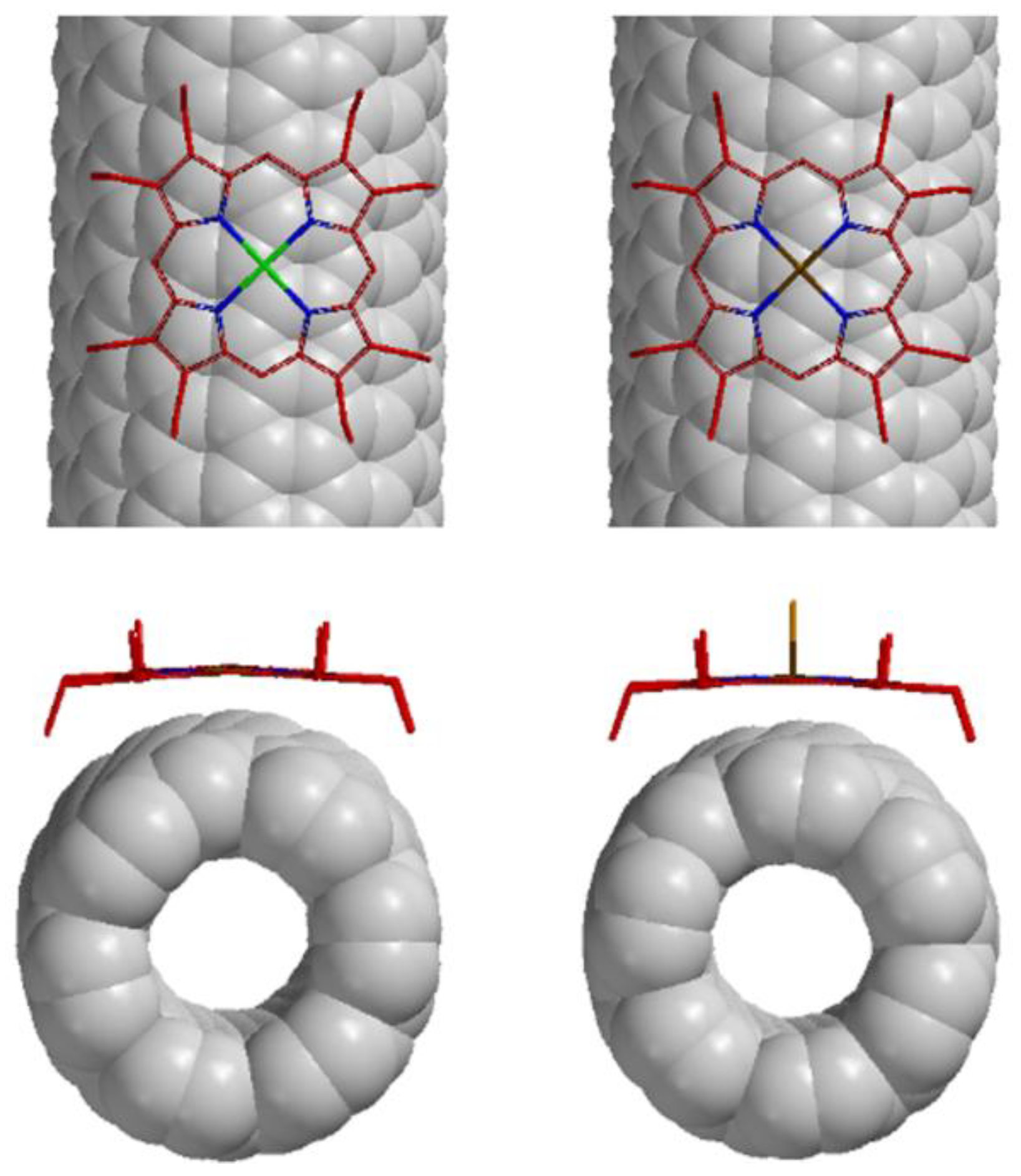
2.1. Supramolecular Interactions of MOEP with 76-CoMoCAT
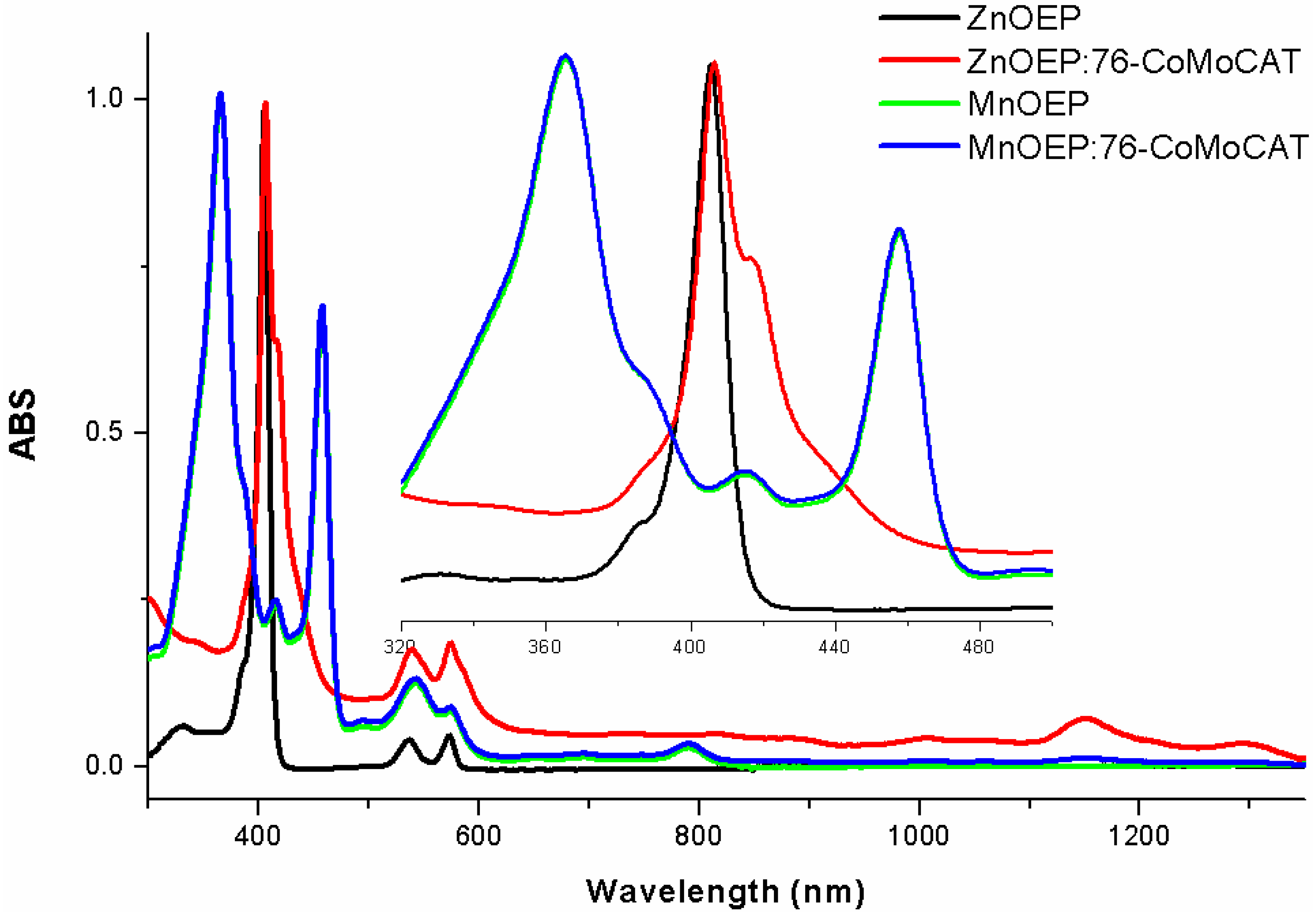
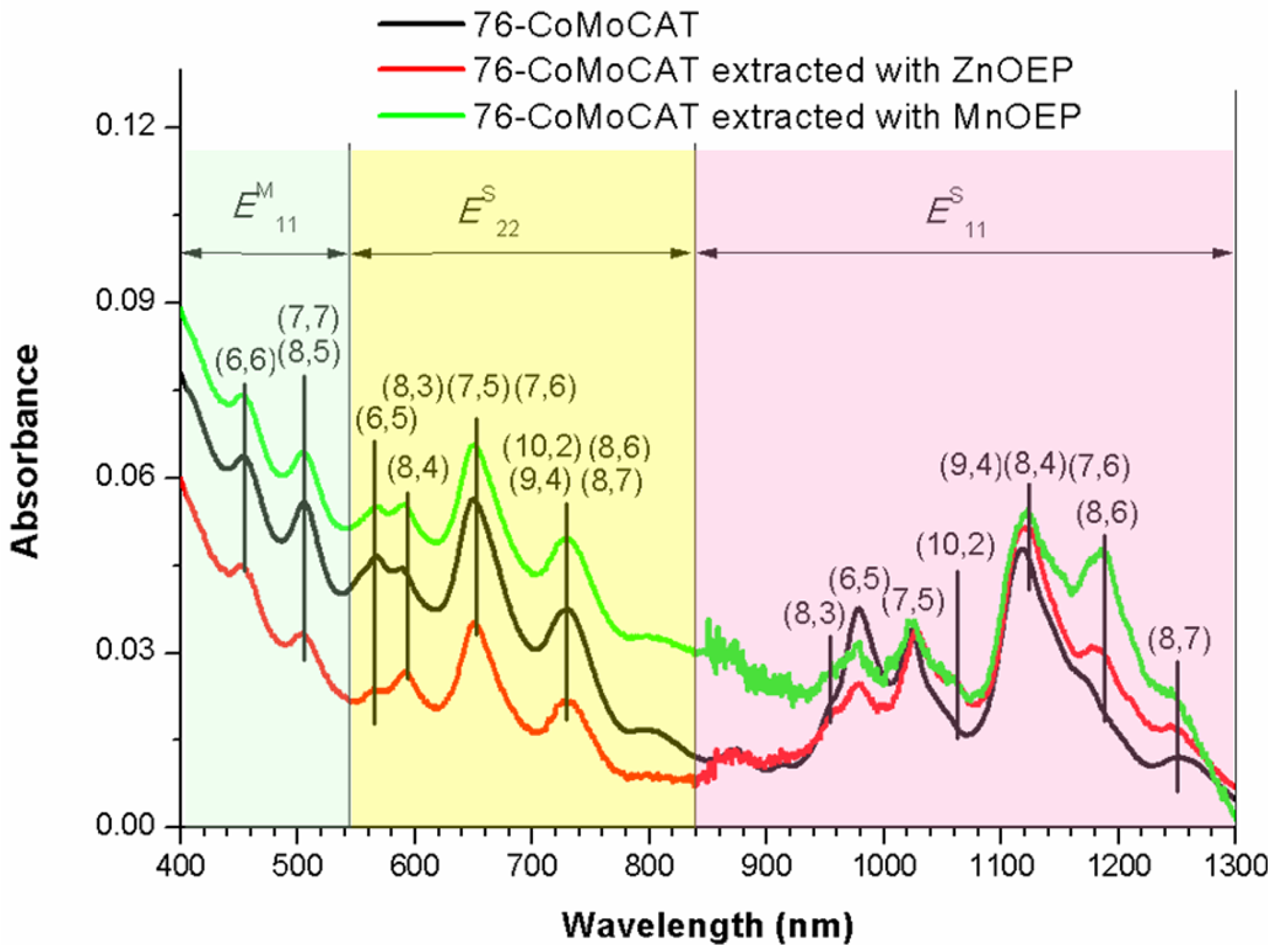
2.2. Absorption Spectra of the Extracts
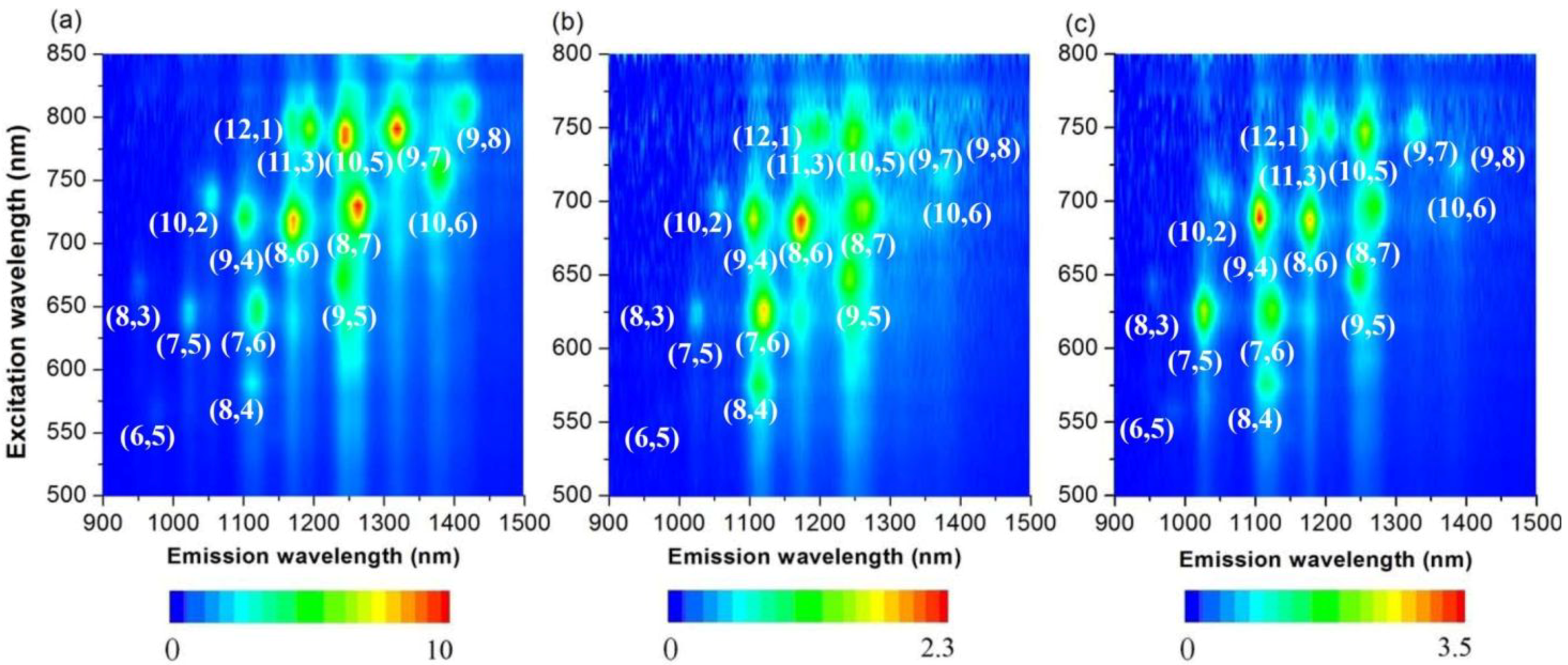
2.3. Large-Diameter Enrichment of 76-CoMoCAT through Extraction

| Major (n,m) components in 76-CoMoCAT | Diameter (nm) | Roll-up angle (degree/°) | Abundance (%) estimated from PL spectra | ||
|---|---|---|---|---|---|
| 76-CoMoCAT | Extraction with ZnOEP | Extraction with MnOEP | |||
| (6,5) | 0.76 | 27.0 | 18 | 5 | 7 |
| (8,3) | 0.78 | 15.3 | 6 | 3 | 3 |
| (7,5) | 0.83 | 24.5 | 25 | 14 | 18 |
| (8,4) | 0.84 | 19.1 | 13 | 13 | 18 |
| (10,2) | 0.88 | 9.0 | 5 | 7 | 5 |
| (7,6) | 0.90 | 27.5 | 13 | 26 | 25 |
| (9,4) | 0.92 | 17.5 | 11 | 19 | 12 |
| (8,6) | 0.97 | 25.3 | 5 | 8 | 8 |
| (8,7) | 1.03 | 27.8 | 4 | 5 | 4 |

| Major ( n,m) components in 76-CoMoCAT | Diameter (nm) | Roll-up angle (degree/°) | Abundance (%) estimated from Raman spectra | ||
|---|---|---|---|---|---|
| 76-CoMoCAT | Extraction with ZnOEP | Extraction with MnOEP | |||
| (8,3) | 0.78 | 15.3 | 5.8 | 1.6 | 2 |
| (7,5) | 0.83 | 24.5 | 57.9 | 48.6 | 27.3 |
| (7,6) | 0.90 | 27.5 | 15.7 | 16.2 | 19.4 |
| (9,4) | 0.92 | 17.5 | 8.4 | 17.3 | 27.2 |
| (10,3) | 0.94 | 12.7 | 12.1 | 16.3 | 24.1 |
3. Experimental Section
3.1. Materials
3.2. Equipment
3.3. Extraction of 76-CoMoCAT with ZnOEP and MnOEP
4. Conclusions
Supplementary Materials
Acknowledgments
Conflict of Interest
References
- Artukovic, E.; Kaempgen, M.; Hecht, D.S.; Roth, S.; Grüner, G. Transparent and flexible carbon nanotube transistors. Nano Lett. 2005, 5, 757–760. [Google Scholar] [CrossRef] [PubMed]
- Rowell, M.W.; Topinka, M.A.; McGehee, M.D.; Prall, H.-J.; Dennler, G.; Sariciftci, N.S.; Hu, L.; Gruner, G. Organic solar cells with carbon nanotube network electrodes. Appl. Phys. Lett. 2006, 88, 233506:1–233506:3. [Google Scholar] [CrossRef]
- Schuettfort, T.; Nish, A.; Nicholas, R.J. Observation of a type II heterojunction in a highly ordered polymer−carbon nanotube nanohybrid structure. Nano Lett. 2009, 9, 3871–3876. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ryu, K.; Liu, X.; Polikarpov, E.; Ly, J.; Tompson, M.E.; Zhou, C. Transparent, conductive, and flexible carbon nanotube films and their application in organic light-emitting diodes. Nano Lett. 2006, 6, 1880–1886. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Imin, P.; Zhitomirsky, I.; Adronov, A. Amperometric detection of glucose using a conjugated polyelectrolyte complex with single-walled carbon nanotubes. Macromolecules 2010, 43, 10376–10381. [Google Scholar] [CrossRef]
- Wang, F.; Gu, H.; Swager, T.M. Carbon nanotube/polythiophene chemiresistive sensors for chemical warfare agents. J. Am. Chem. Soc. 2008, 130, 5392–5393. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.N.R.; Voggu, R.; Govindaraj, A. Selective generation of single-walled carbon nanotubes with metallic, semiconducting and other unique electronic properties. Nanoscale 2009, 1, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Backes, C.; Mundloch, U.; Ebel, A.; Hauke, F.; Hirsch, A. Dispersion of hipco® and comocat® single-walled nanotubes (swnts) by water soluble pyrene derivatives—depletion of small diameter swnts. Chem. A Eur. J. 2010, 16, 3314–3317. [Google Scholar] [CrossRef]
- Backes, C.; Schmidt, C.D.; Hauke, F.; Hirsch, A. Perylene-based nanotweezers: Enrichment of larger-diameter single-walled carbon nanotubes. Chem. A Eur. J. 2011, 6, 438–444. [Google Scholar]
- Chen, F.; Wang, B.; Chen, Y.; Li, L.-J. Toward the extraction of single species of single-walled carbon nanotubes using fluorene-based polymers. Nano Lett. 2007, 7, 3013–3017. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Bachilo, S.M.; Weisman, R.B. Advanced sorting of single-walled carbon nanotubes by nonlinear density-gradient ultracentrifugation. Nat. Nano 2010, 5, 443–450. [Google Scholar] [CrossRef]
- Hersam, M.C. Progress towards monodisperse single-walled carbon nanotubes. Nat. Nano 2008, 3, 387–394. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Nish, A.; Doig, J.; Douven, S.; Chen, C.-W.; Chen, L.-C.; Nicholas, R.J. Polymer structure and solvent effects on the selective dispersion of single-walled carbon nanotubes. J. Am. Chem. Soc. 2008, 130, 3543–3553. [Google Scholar] [CrossRef] [PubMed]
- Kalbác, M.; Kavan, L.; Dunsch, L. Selective etching of thin single-walled carbon nanotubes. J. Am. Chem. Soc. 2009, 131, 4529–4534. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Niidome, Y.; Nakashima, N. Efficient separation of (6,5) single-walled carbon nanotubes using a “nanometal sinker”. Angew. Chem. Int. Ed. 2009, 48, 5435–5438. [Google Scholar] [CrossRef]
- Komatsu, N.; Wang, F. A comprehensive review on separation methods and techniques for single-walled carbon nanotubes. Materials 2010, 3, 3818–3844. [Google Scholar] [CrossRef]
- Krupke, R.; Hennrich, F.; Löhneysen, H.V.; Kappes, M.M. Separation of metallic from semiconducting single-walled carbon nanotubes. Science 2003, 301, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Nishide, D.; Tanaka, T.; Kataura, H. Large-scale single-chirality separation of single-wall carbon nanotubes by simple gel chromatography. Nat. Commun. 2011, 2, 309. [Google Scholar] [CrossRef] [PubMed]
- Marquis, R.; Greco, C.; Sadokierska, I.; Lebedkin, S.; Kappes, M.M.; Michel, T.; Alvarez, L.; Sauvajol, J.-L.; Meunier, S.P.; Mioskowski, C. Supramolecular discrimination of carbon nanotubes according to their helicity. Nano Lett. 2008, 8, 1830–1835. [Google Scholar] [CrossRef] [PubMed]
- Marquis, R.; Kulikiewicz, K.; Lebedkin, S.; Kappes, M.M.; Mioskowski, C.; Meunier, S.; Wagner, A. Axially chiral facial amphiphiles with a dihydronaphthopentaphene structure as molecular tweezers for swnts. Chem. A Eur. J. 2009, 15, 11187–11196. [Google Scholar] [CrossRef]
- Nish, A.; Hwang, J.-Y.; Doig, J.; Nicholas, R.J. Highly selective dispersion of single-walled carbon nanotubes using aromatic polymers. Nat. Nano 2007, 2, 640–646. [Google Scholar] [CrossRef]
- Rahman, A.F.M.M.; Wang, F.; Matsuda, K.; Kimura, T.; Komatsu, N. Diameter-based separation of single-walled carbon nanotubes through selective extraction with dipyrene nanotweezers. Chem. Sci. 2011, 2, 862–867. [Google Scholar] [CrossRef]
- Stürzl, N.; Hennrich, F.; Lebedkin, S.; Kappes, M.M. Near monochiral single-walled carbon nanotube dispersions in organic solvents. J. Phys. Chem. C 2009, 113, 14628–14632. [Google Scholar] [CrossRef]
- Tu, X.; Manohar, S.; Jagota, A.; Zheng, M. DNA sequence motifs for structure-specific recognition and separation of carbon nanotubes. Nature 2009, 460, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tu, X.; Welsher, K.; Wang, X.; Zheng, M.; Dai, H. Optical characterizations and electronic devices of nearly pure (10,5) single-walled carbon nanotubes. J. Am. Chem. Soc. 2009, 131, 2454–2455. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Semke, E.D. Enrichment of single chirality carbon nanotubes. J. Am. Chem. Soc. 2007, 129, 6084–6085. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.-L.; Stoddart, J.F. Noncovalent functionalization of single-walled carbon nanotubes. Acc. Chem. Res. 2009, 42, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Zhu, J.; Adronov, A. Supramolecular functionalization of single-walled carbon nanotubes with triply fused porphyrin dimers: A study of structure–property relationships. Chem. Mater. 2011, 23, 3188–3194. [Google Scholar] [CrossRef]
- Guldi, D.M.; Rahman, G.M.A.; Jux, N.; Tagmatarchis, N.; Prato, M. Integrating single-wall carbon nanotubes into donor–acceptor nanohybrids. Angew. Chem. 2004, 116, 5642–5646. [Google Scholar] [CrossRef]
- Rahman, G.M.A.; Guldi, D.M.; Campidelli, S.; Prato, M. Electronically interacting single wall carbon nanotube-porphyrin nanohybrids. J. Mater. Chem. 2006, 16, 62–65. [Google Scholar] [CrossRef]
- Tanaka, H.; Yajima, T.; Matsumoto, T.; Otsuka, Y.; Ogawa, T. Porphyrin molecular nanodevices wired using single-walled carbon nanotubes. Adv. Mater. 2006, 18, 1411–1415. [Google Scholar] [CrossRef]
- Cheng, F.; Adronov, A. Noncovalent functionalization and solubilization of carbon nanotubes by using a conjugated Zn–porphyrin polymer. Chem. A Eur. J. 2006, 12, 5053–5059. [Google Scholar] [CrossRef]
- Murakami, H.; Nomura, T.; Nakashima, N. Noncovalent porphyrin-functionalized single-walled carbon nanotubes in solution and the formation of porphyrin–nanotube nanocomposites. Chem. Phys. Lett. 2003, 378, 481–485. [Google Scholar]
- Murakami, H.; Nakamura, G.; Nomura, T.; Miyamoto, T.; Nakashima, N. Noncovalent porphyrin-functionalized single-walled carbon nanotubes: Solubilization and spectral behaviors. J. Porphyr. Phthalocyanines 2007, 11, 418–427. [Google Scholar] [CrossRef]
- Li, H.; Zhou, B.; Lin, Y.; Gu, L.; Wang, W.; Fernando, K.A.S.; Kumar, S.; Allard, L.F.; Sun, Y.-P. Selective interactions of porphyrins with semiconducting single-walled carbon nanotubes. J. Am. Chem. Soc. 2004, 126, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, D.R.; Kuzmych, O.; Star, A. Interactions between single-walled carbon nanotubes and tetraphenyl metalloporphyrins: Correlation between spectroscopic and fet measurements. J. Phys. Chem. C 2007, 111, 3539–3543. [Google Scholar] [CrossRef]
- Zhao, J.-X.; Ding, Y.-H. Functionalization of single-walled carbon nanotubes with metalloporphyrin complexes: A theoretical study. J. Phys. Chem. C 2008, 112, 11130–11134. [Google Scholar] [CrossRef]
- Peng, X.; Komatsu, N.; Bhattacharya, S.; Shimawaki, T.; Aonuma, S.; Kimura, T.; Osuka, A. Optically active single-walled carbon nanotubes. Nat. Nano 2007, 2, 361–365. [Google Scholar] [CrossRef]
- Peng, X.; Komatsu, N.; Kimura, T.; Osuka, A. Improved optical enrichment of swnts through extraction with chiral nanotweezers of 2,6-pyridylene-bridged diporphyrins. J. Am. Chem. Soc. 2007, 129, 15947–15953. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Wang, F.; Kimura, T.; Komatsu, N.; Osuka, A. Optical resolution and diameter-based enrichment of single-walled carbon nanotubes through simultaneous recognition of their helicity and diameter with chiral monoporphyrin. J. Phys. Chem. C 2009, 113, 9108–9113. [Google Scholar] [CrossRef]
- Wang, F.; Matsuda, K.; Rahman, A.F.M.M.; Kimura, T.; Komatsu, N. Improved selectivity in discriminating handedness and diameter of single-walled carbon nanotubes with n-substituted 3,6-carbazolylene-bridged chiral diporphyrin nanotweezers. Nanoscale 2011, 3, 4117–4124. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Matsuda, K.; Rahman, A.F.M.M.; Peng, X.; Kimura, T.; Komatsu, N. Simultaneous discrimination of handedness and diameter of single-walled carbon nanotubes (swnts) with chiral diporphyrin nanotweezers leading to enrichment of a single enantiomer of (6,5)-swnts. J. Am. Chem. Soc. 2010, 132, 10876–10881. [Google Scholar] [CrossRef] [PubMed]
- Tu, W.; Lei, J.; Ju, H. Functionalization of carbon nanotubes with water-insoluble porphyrin in ionic liquid: Direct electrochemistry and highly sensitive amperometric biosensing for trichloroacetic acid. Chem. A Eur. J. 2009, 15, 779–784. [Google Scholar] [CrossRef]
- Guldi, D.M.; Rahman, G.M.A.; Prato, M.; Jux, N.; Qin, S.; Ford, W. Single-wall carbon nanotubes as integrative building blocks for solar-energy conversion. Angew. Chem. 2005, 117, 2051–2054. [Google Scholar] [CrossRef]
- Wei, L.; Wang, B.; Wang, Q.; Li, L.-J.; Yang, Y.; Chen, Y. Effect of centrifugation on the purity of single-walled carbon nanotubes from MCM-41 containing cobalt. J. Phys. Chem. C 2008, 112, 17567–17575. [Google Scholar] [CrossRef]
- Luo, Z.; Pfefferle, L.D.; Haller, G.L.; Papadimitrakopoulos, F. (n,m) abundance evaluation of single-walled carbon nanotubes by fluorescence and absorption spectroscopy. J. Am. Chem. Soc. 2006, 128, 15511–15516. [Google Scholar] [CrossRef] [PubMed]
- Bachilo, S.M.; Balzano, L.; Herrera, J.E.; Pompeo, F.; Resasco, D.E.; Weisman, R.B. Narrow (n,m)-distribution of single-walled carbon nanotubes grown using a solid supported catalyst. J. Am. Chem. Soc. 2003, 125, 11186–11187. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, Y.; Rahman, A.F.M.M.; Liu, G.; Xiong, Z.; Koezuka, K.; Xu, Z.; Komatsu, N.; Wang, F. Enrichment of Large-Diameter Single-Walled Carbon Nanotubes (SWNTs) with Metallo-Octaethylporphyrins. Materials 2013, 6, 3064-3078. https://doi.org/10.3390/ma6083064
Li Y, Rahman AFMM, Liu G, Xiong Z, Koezuka K, Xu Z, Komatsu N, Wang F. Enrichment of Large-Diameter Single-Walled Carbon Nanotubes (SWNTs) with Metallo-Octaethylporphyrins. Materials. 2013; 6(8):3064-3078. https://doi.org/10.3390/ma6083064
Chicago/Turabian StyleLi, Yuda, A. F. M. Mustafizur Rahman, Gang Liu, Zichao Xiong, Kenji Koezuka, Zhigao Xu, Naoki Komatsu, and Feng Wang. 2013. "Enrichment of Large-Diameter Single-Walled Carbon Nanotubes (SWNTs) with Metallo-Octaethylporphyrins" Materials 6, no. 8: 3064-3078. https://doi.org/10.3390/ma6083064
APA StyleLi, Y., Rahman, A. F. M. M., Liu, G., Xiong, Z., Koezuka, K., Xu, Z., Komatsu, N., & Wang, F. (2013). Enrichment of Large-Diameter Single-Walled Carbon Nanotubes (SWNTs) with Metallo-Octaethylporphyrins. Materials, 6(8), 3064-3078. https://doi.org/10.3390/ma6083064