Cement and Concrete Nanoscience and Nanotechnology
Abstract
:1. Introduction
2. C-S-H and C-S-H Composites
2.1. Calcium Silicate Hydrate (C-S-H)
2.1.1. Formation and properties
2.1.2. Nanostructural models of C-S-H
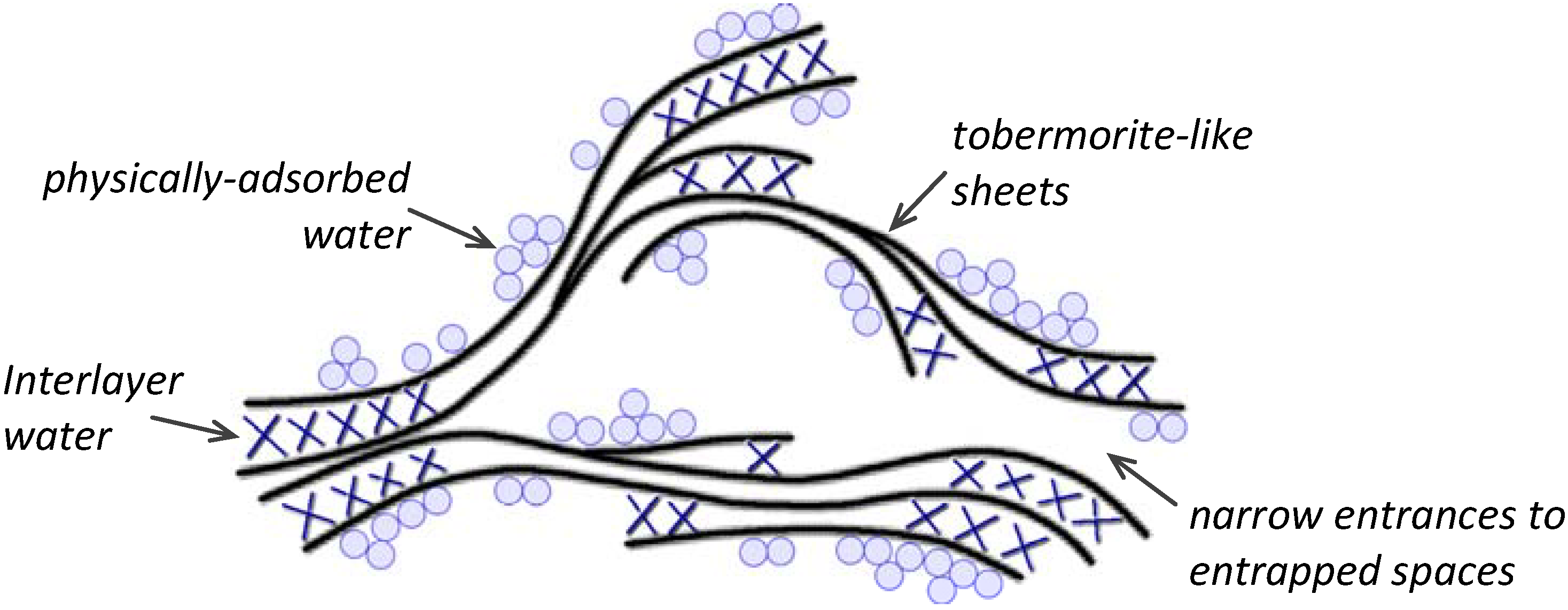
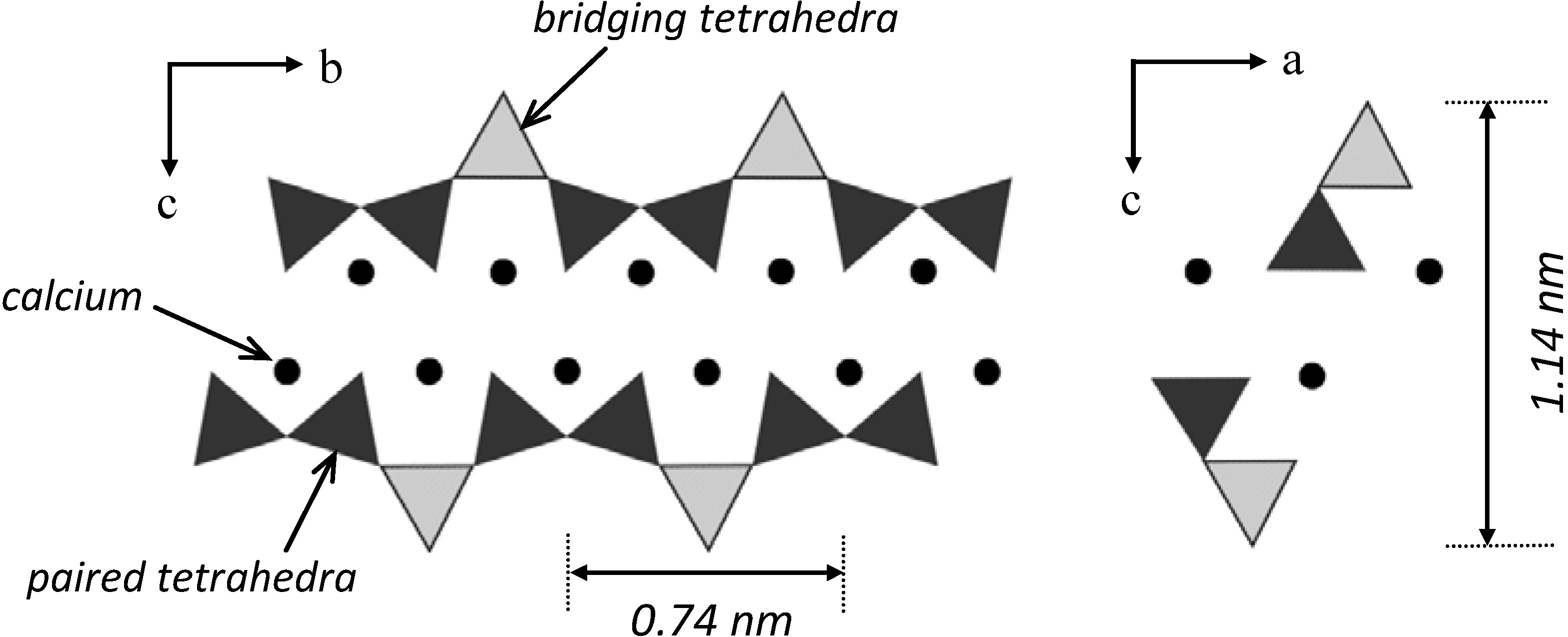
2.1.3. Mechanical properties
2.2. Engineering C-S-H-based Nanocomposites
2.2.1. Background
2.2.2. Surface adsorption and grafting of polymers at defect sites
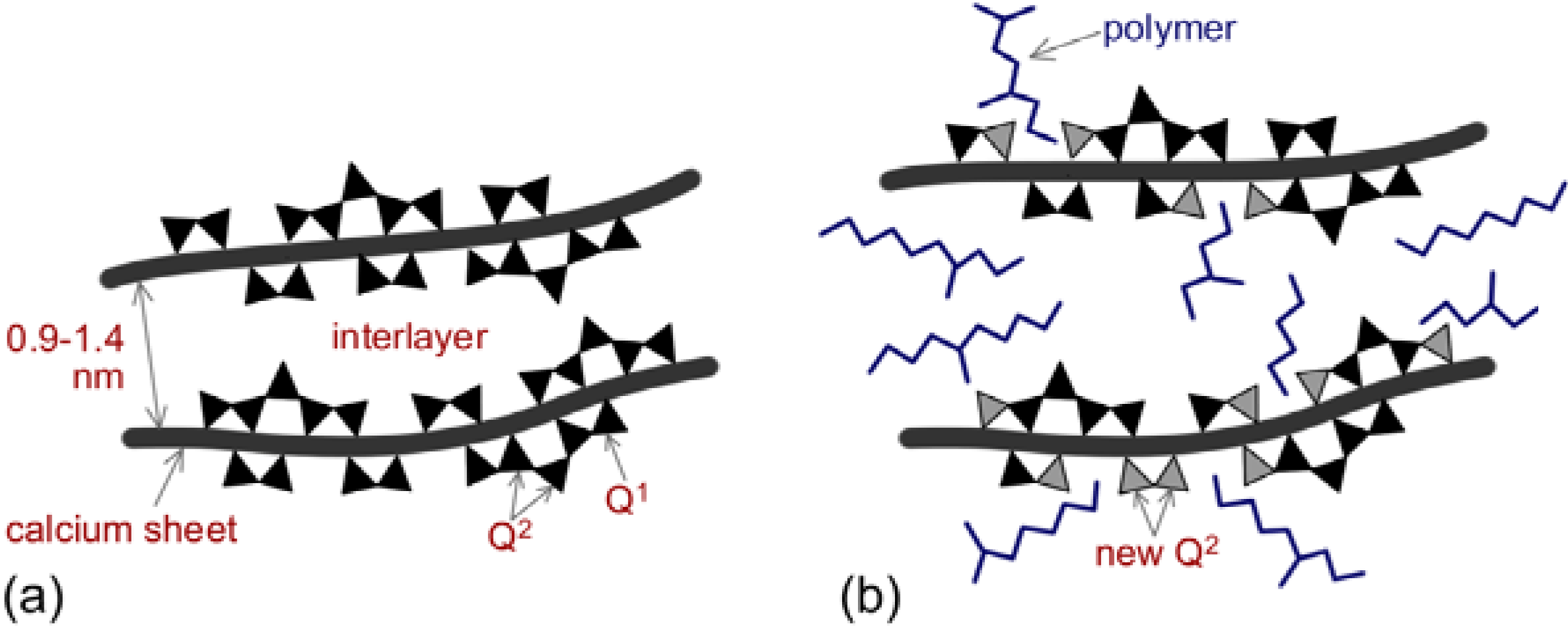
2.2.3. Intercalation of the interlayer spaces between the C-S-H sheets
2.2.4. True hybrids and covalent bonding of polymers with C-S-H
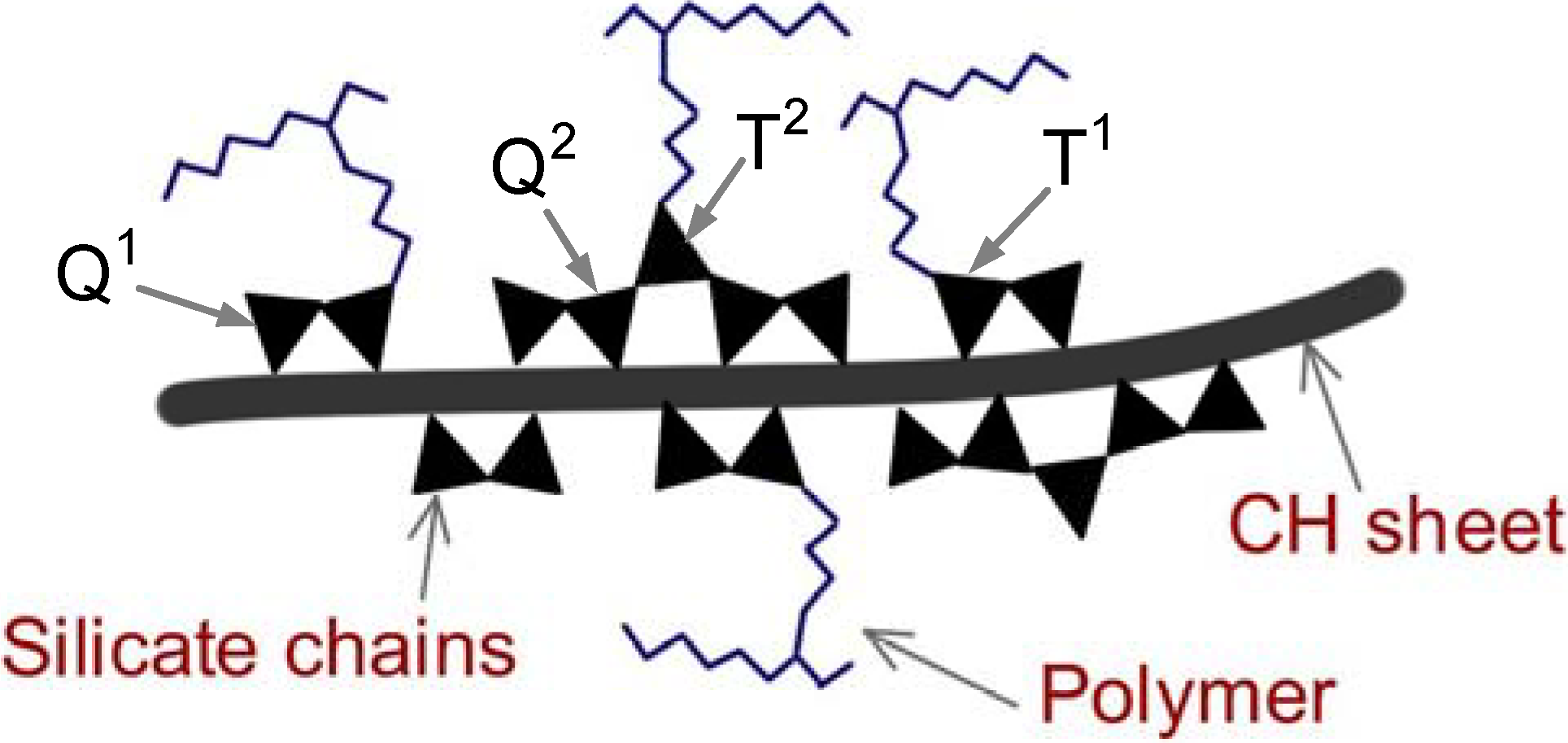
2.2.5. Engineering properties and durability of C-S-H nanocomposites
3. Controlled Release of Admixtures
3.1. Background
3.2. Layered Double Hydroxides (LDHs)
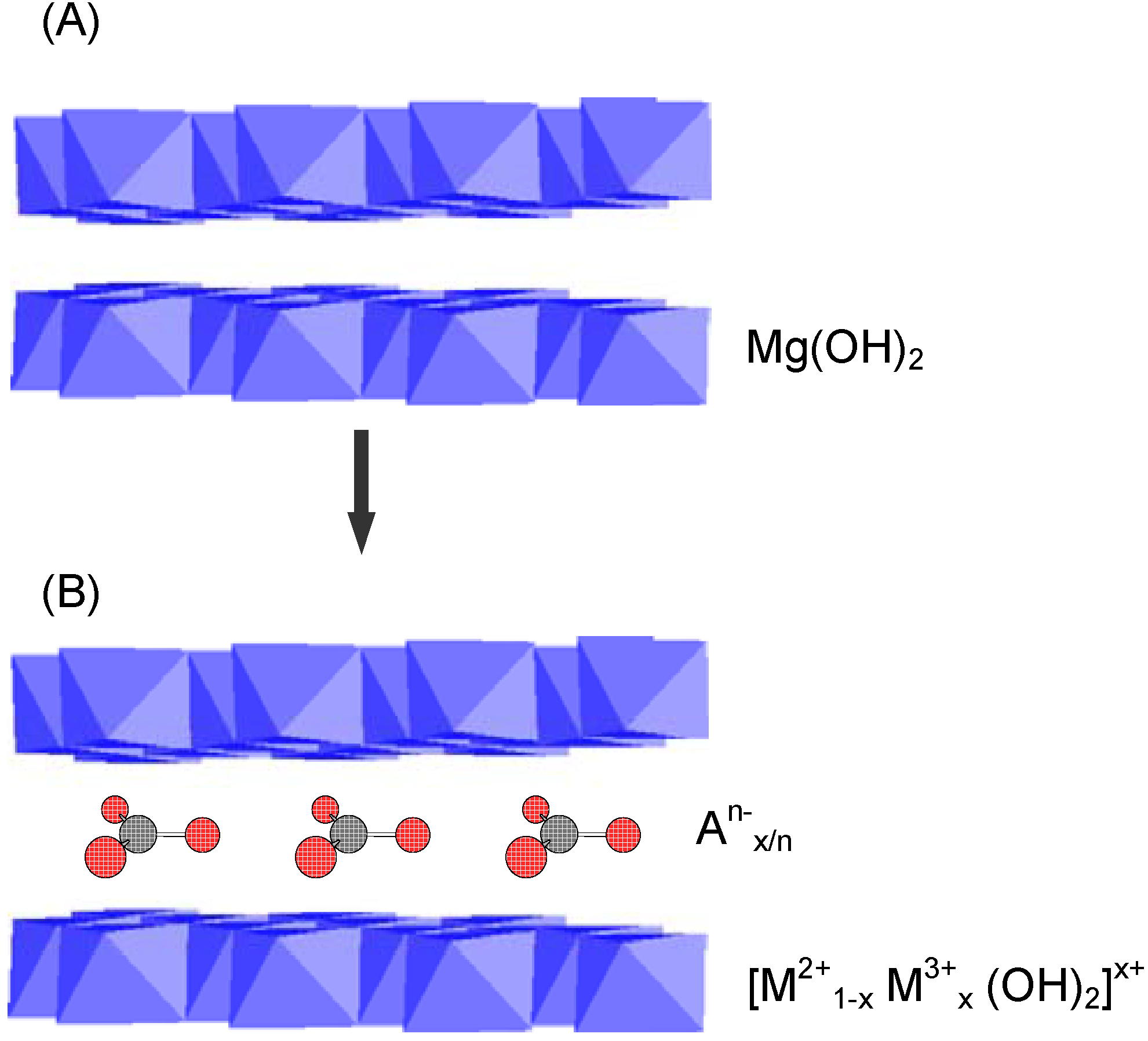
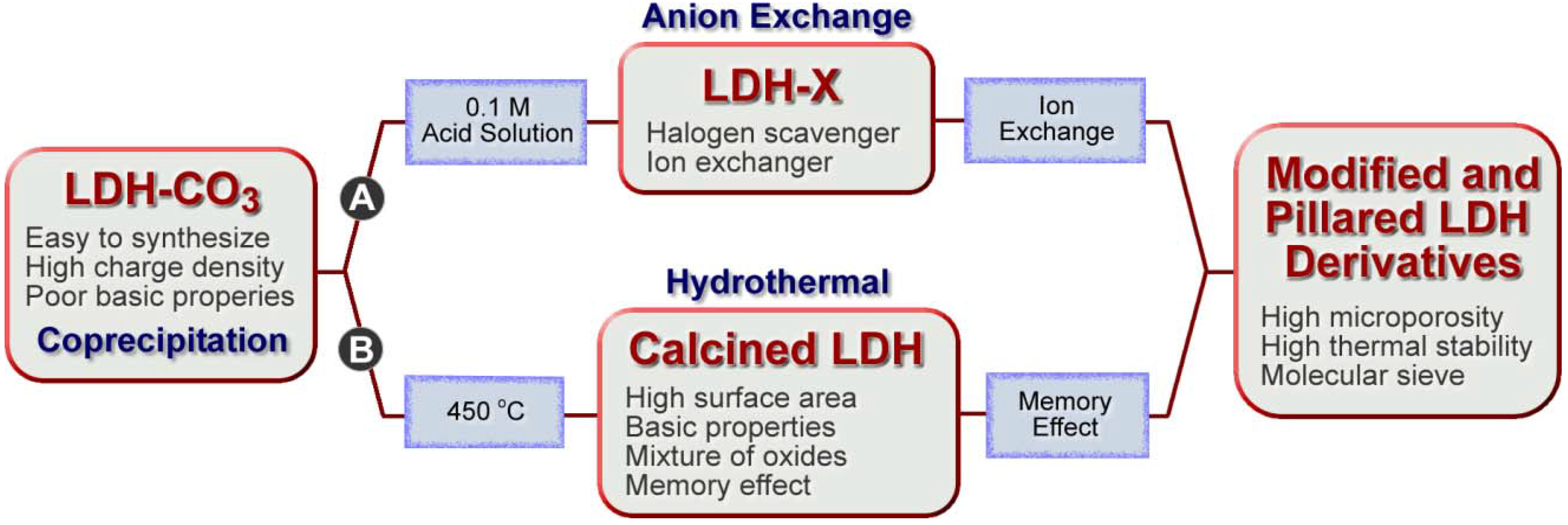
3.3. Controlled Release Technology in Cement and Concrete
3.3.1. Interaction of superplasticizers with cement systems
3.3.2. Controlled release of admixtures
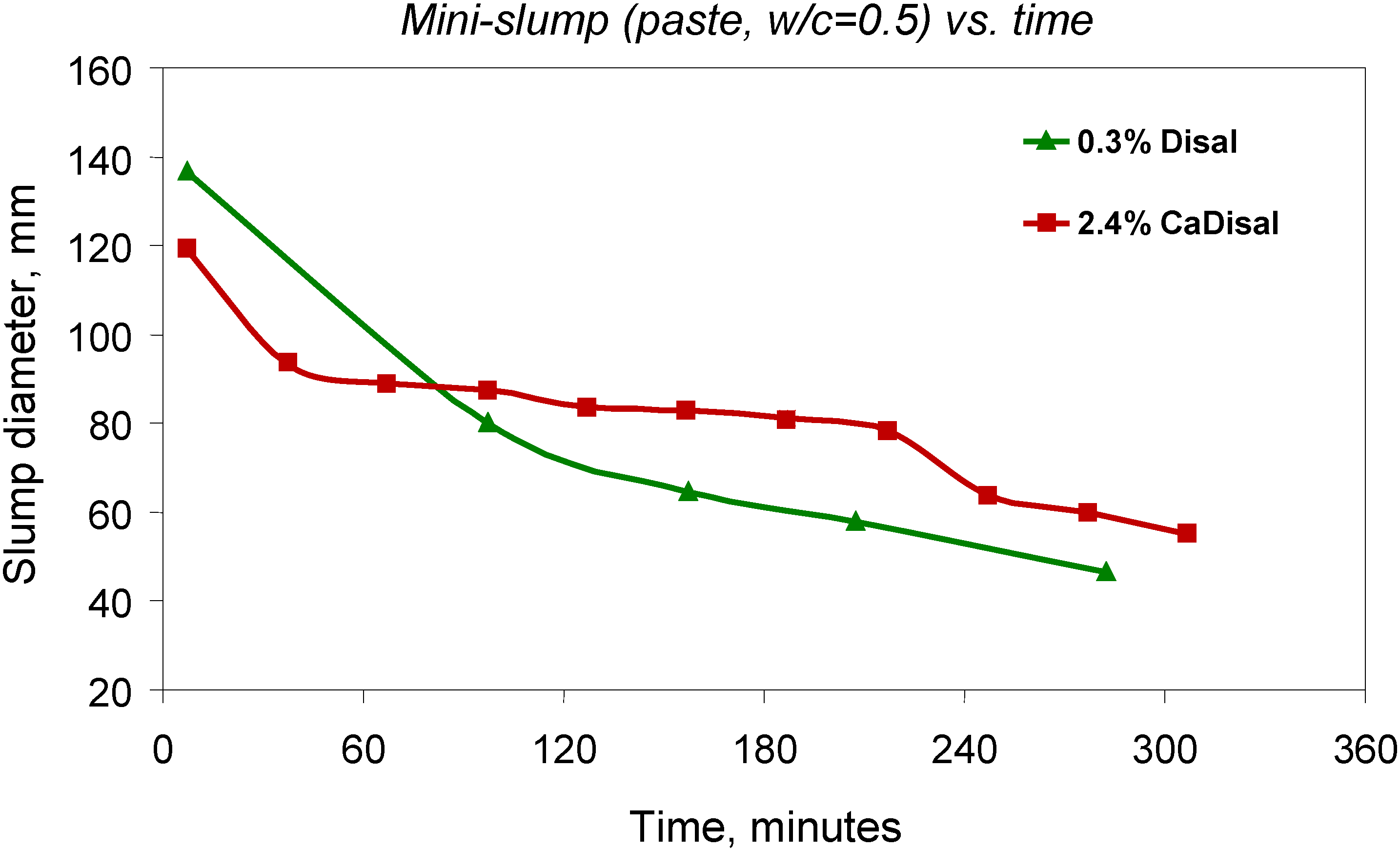
4. Carbon Nanotube/Cement Composite Materials
4.1. Mechanical Properties of Concrete
4.2. Fibers and Nanotube Reinforcements
4.3. Development and Properties of CNT/OPC Composites.
4.3.1. Dispersion of CNT
4.3.2. Composite properties
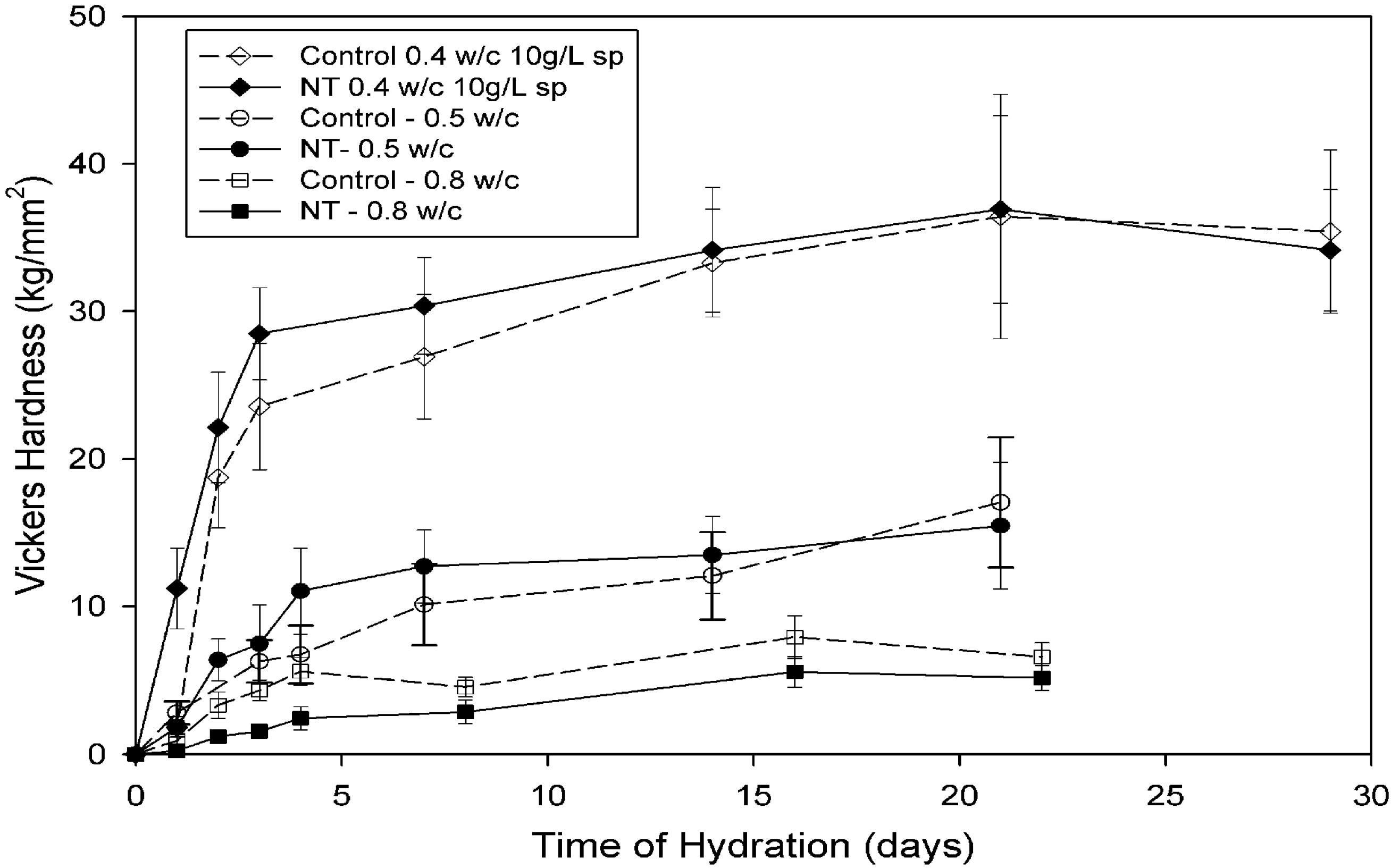


4.3.3. Interactions between OPC and CNT
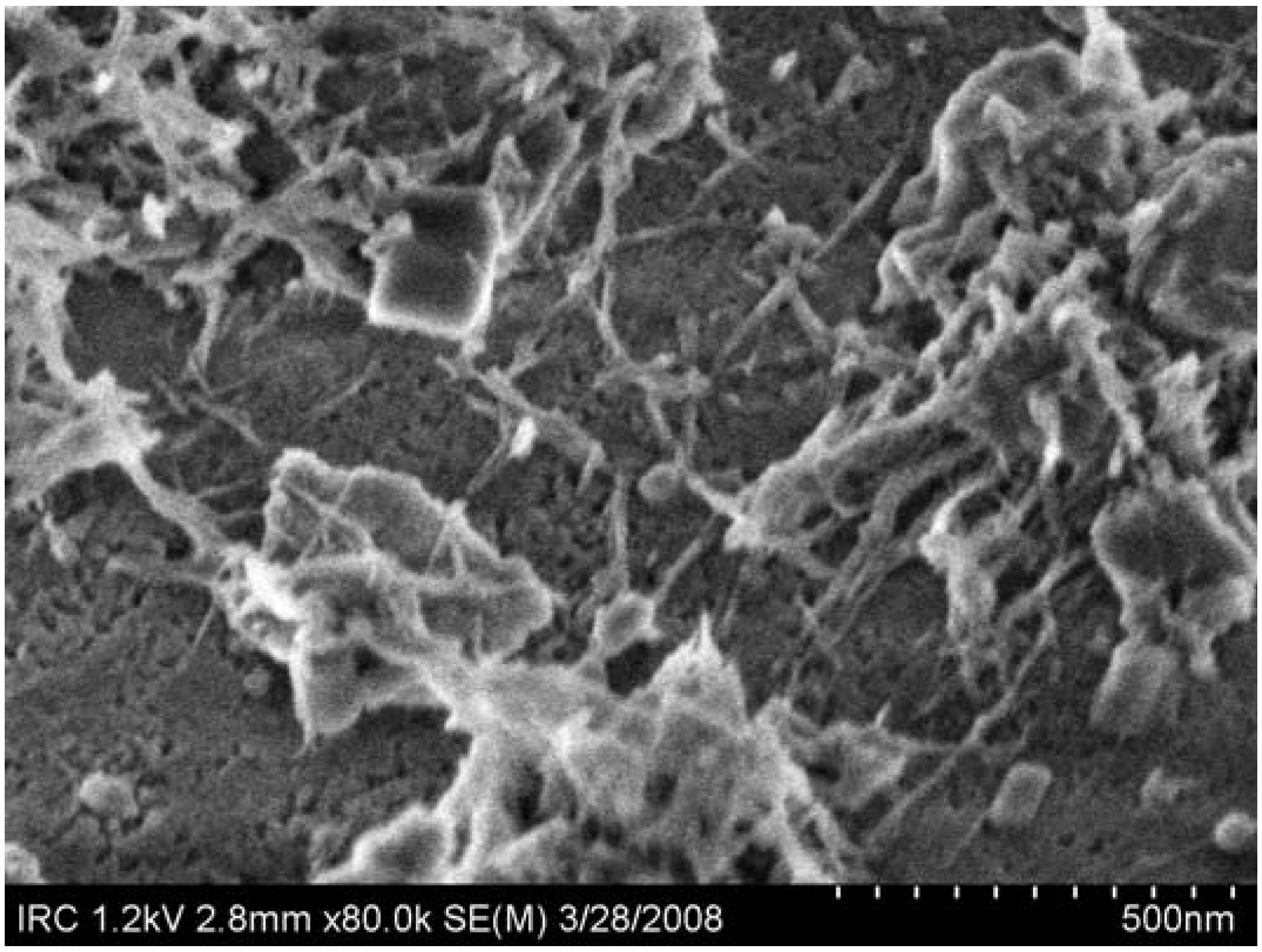
5. Nanoparticles Technology
5.1. Background
5.2. Nanoparticles in Cement and Concrete
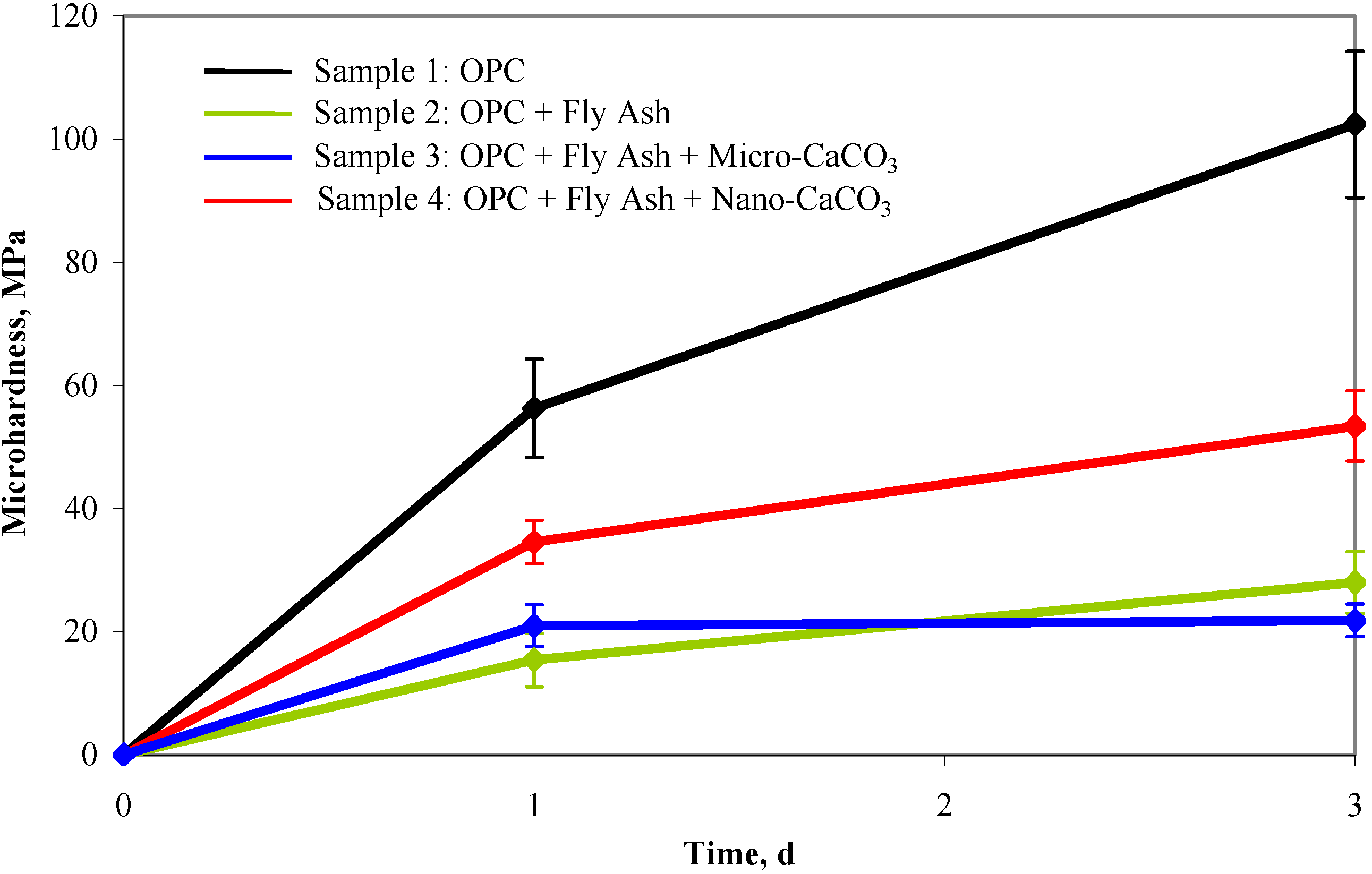
5.3. Research on CaCO3 Nanoparticles—A Case Study


6. Economic and Related Factors Affecting the Application of Nanotechnology to Concrete
6.1. Acceptance of Nanotechnology in the Construction Industry
6.2. Potential Economic Impact of Nanotechnology on the Construction Industry
References
- Richardson, I.G. The nature of C-S-H in hardened cements. Cem. Concr. Res. 1999, 29, 1131–1147. [Google Scholar] [CrossRef]
- Alizadeh, R.; Beaudoin, J.J.; Ramachandran, V.S.; Raki, L. Applicability of the Hedvall effect to study the reactivity of calcium silicate hydrates. Adv. Cem. Res. 2009, 21, 59–66. [Google Scholar] [CrossRef]
- Nonat, A. The structure and stoichiometry of C-S-H. Cem. Concr. Res. 2004, 34, 1521–1528. [Google Scholar] [CrossRef]
- Ramachandran, V.S.; Feldman, R.F.; Beaudoin, J.J. Concrete Science; Heyden & Son Ltd: London, UK, 1981; pp. 54–89. [Google Scholar]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Thomas Telford: London, UK, 1997; pp. 231–244. [Google Scholar]
- Powers, T.C.; Brownyard, T.L. Studies of the physical properties of hardened Portland cement paste. ACI J. Proc. 1946–1947, 43, pp. 101, 249, 469, 549, 669, 845, 933. [Google Scholar]
- Feldman, R.F.; Sereda, P.J. A model for hydrated Portland cement paste as deduced from sorption-length change and mechanical properties. Matériaux et Construct. 1968, 1, 509–520. [Google Scholar] [CrossRef]
- Feldman, R.F.; Sereda, P.J. A new model for hydrated Portland cement paste and its practical implications. Eng. J. Can. 1970, 53, 53–59. [Google Scholar]
- Jennings, H.M. Refinements to colloid model of C-S-H in cement: CM-II. Cem. Concr. Res. 2008, 38, 275–289. [Google Scholar] [CrossRef]
- Alizadeh, R.; Beaudoin, J.J.; Raki, L. C-S-H (I)-A nanostructural model for the removal of water from hydrated cement paste? J. Am. Ceram. Soc. 2007, 90, 670–672. [Google Scholar] [CrossRef]
- Chen, J.J.; Thomas, J.J.; Taylor, H.F.W.; Jennings, H.M. Solubility and structure of calcium silicate hydrate. Cem. Concr. Res. 2004, 34, 1499–1599. [Google Scholar] [CrossRef]
- Cong, X.; Kirkpatrick, R.J. 29Si and 17O NMR investigation of the structure of some crystalline calcium silicate hydrates. Adv. Cem. Based Mater. 1996, 3, 133–143. [Google Scholar] [CrossRef]
- Beaudoin, J.J.; Feldman, R.F. Dependence of degree of silica polymerization and intrinsic mechanical properties of C-S-H on C/S ratio. In Proceedings of the 8th International Congress of Chemistry of Cement, Rio de Janeiro, Brazil, 22–28 September 1986; Volume 3, pp. 337–342.
- Constantinides, G.; Ulm, F.J. The effect of two types of C-S-H on the elasticity of cement-based materials: Results from nanoindentation and micromechanical modeling. Cem. Concr. Res. 2004, 34, 67–80. [Google Scholar] [CrossRef]
- Alizadeh, R.; Beaudoin, J.J.; Raki, L. Mechanical properties of calcium silicate hydrates. Mater. Struct. 2009. submit. [Google Scholar]
- Pellenq, R.J.M.; Gmira, A.; Van Damme, H. Stability and elastic properties of tobermorite, a model of cement hydrate at the nanoscale. In Proceedings of the Materials Science and Technology Conference, Advances in Cement-Based Materials, Detroit, Michigan, MI, USA, 16–20, September 2007; pp. 1–12.
- Manzano, H.; Dolado, J.S.; Guerrero, A.; Ayuela, A. Mechanical properties of crystalline calcium silicate hydrates: Comparison with cementitious C-S-H gels. Phys. Status Solidi A 2007, 204, 1775–1780. [Google Scholar] [CrossRef]
- Manzano, H.; Dolado, J.S.; Ayuela, A. Elastic properties of the main species present in Portland cement pastes. Acta Mater. 2009, 57, 1666–1674. [Google Scholar]
- Gmira, A.; Zabat, M.; Pellenq, R.J.M.; Van Damme, H. Microscopic physical basis of the poromechanical behavior of cement-based materials. Mater. Struct. 2004, 37, 3–14. [Google Scholar]
- Minet, J.; Abramson, S.; Bresson, B.; Franceschini, A.; Van Damme, H.; Lequeux, N. Organic calcium silicate hydrate hybrids: A new approach to cement-based nanocomposites. J. Mater. Chem. 2006, 16, 1379–1383. [Google Scholar]
- Matsuyama, H.; Young, J.F. Intercalation of polymers in calcium silicate hydrate: A new synthetic approach to biocomposites? Chem. Mater. 1999, 11, 16–19. [Google Scholar]
- Beaudoin, J.J.; Drame, H.; Raki, L.; Alizadeh, R. Formation and characterization of calcium silicate hydrate-hexadecyltrimethlamonium nanostructures. J. Mater. Res. 2008, 23, 2804–2815. [Google Scholar]
- Beaudoin, J.J.; Patarachao, B.; Raki, L.; Alizadeh, R. The interaction of methylene blue dye with calcium-silicate-hydrate. J. Am. Ceram. Soc. 2009, 92, 204–208. [Google Scholar] [CrossRef]
- Beaudoin, J.J.; Drame, H.; Raki, L.; Alizadeh, R. Formation and properties of C-S-H -PEG nano-structures. Mater. Struct. 2009, 42, 1003–1014. [Google Scholar]
- Beaudoin, J.J.; Raki, L.; Alizadeh, R. A 29Si MAS NMR study of the modified C-S-H nanostructures. Cem. Concr. Compos. 2009, 31, 585–590. [Google Scholar] [CrossRef]
- Matsuyama, H.; Young, J.F. Synthesis of calcium silicate hydrate/polymer complexes: Part I. Mater. Res. 1999, 14, 3379–3388. [Google Scholar]
- Mojumdar, S.C.; Raki, L. Preparation and properties of calcium silicate hydrate-poly (vinyl alcohol) nanocomposite materials. J. Therm. Anal. Calorim. 2005, 82, 89–95. [Google Scholar] [CrossRef]
- Franceschini, A.; Abramson, S.; Mancini, V.; Bresson, B.; Chassenieux, C.; Lequeux, N. New covalent bonded polymer-calcium silicate hydrate composites. J. Mater. Chem. 2007, 17, 913–922. [Google Scholar]
- Hong, S.I.; Oh, Y.S. Dissolution kinetics and physical characterization of three layered tablet with poly (ethylene) oxide core matrix capped by Carbopol. Int. J. Pharm. 2008, 356, 121–129. [Google Scholar]
- Ai, H.; Jones, S.; Devilliers, M.; Lvov, Y. Nano-encapsulation of furosemide microcrystals for controlled drug release. J. Control. Release 2003, 86, 59–68. [Google Scholar] [CrossRef]
- Kwon, I.C.; Bae, Y.H.; Kim, S.W. Electrically erodible polymer gel for controlled release of drugs. Nature 1991, 354, 291–293. [Google Scholar]
- Del Hoyo, C. Layered double hydroxides and human health: An overview. Appl. Clay Sci. 2007, 36, 103–121. [Google Scholar] [CrossRef]
- Bin Hussein, M.Z.; Zainal, Z.; Yhaya, A.H.; Foo, D.W.V. Controlled release of a plant growth regulator, α-naphthaleneacetate from the lamella of Zn–Al-layered double hydroxide nanocomposite. J. Control. Release 2002, 82, 417–427. [Google Scholar] [CrossRef]
- Deep, C.; Sraf, S. Novel approaches in herbal cosmetics. J. Cosmet Dermatol 2007, 7, 89–95. [Google Scholar]
- Hermann, A.; Debonneville, C.; Laubscher, V.; Aymar, L. Dynamic headspace analysis of the light-induced controlled release of perfumery aldheydes and ketones from alpha-esters in body care and household applications. Flavour Frag. J. 2000, 15, 415–420. [Google Scholar]
- Kuang, Y.; Ou, J. Self-repairing performance of concrete beams strengthened using super elastic SMA wires in combination with adhesives released from hollow fibers. Smart Mater. Struct. 2008, 17, 1–7. [Google Scholar]
- Cavani, F.; Trifirio, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Nyambo, C.; Kandare, E.; Wang, D.; Wilkie, C.A. Flame-retarded polystyrene: Investigating chemical interactions between ammonium polyphosphate and MgAl layered double hydroxide. Polym. Degrad. Stab. 2008, 93, 1656–1663. [Google Scholar]
- Lv, L.; Sun, P.; Gu, Z.; Du, H.; Pang, X.; Tao, X.; Xu, R.; Xu, L. Removal of chloride ion from aqueous solution by ZnAl-NO3 layered double hydroxides as anion exchanger. J. Hazard. Mater. 2009, 161, 1444–1449. [Google Scholar] [CrossRef] [PubMed]
- Trikeriotis, M.; Ghanotakis, D.F. Intercalation of hydrophilic and hydrophobic antibiotics in layered double hydroxides. Int. J. Pharm. 2007, 332, 176–184. [Google Scholar]
- Kwak, S.Y.; Jeong, Y.J.; Park, J.S.; Choy, J.H. Bio-LDH nanohybrid for gene therapy. Solid State Ionics 2002, 151, 229–234. [Google Scholar]
- Gallop, R.S.; Taylor, H.F.W. Microstructural and microanalytical studies of sulfate attack. V. Comparison of different slag blends. Cem. Concr. Res. 1996, 26, 1029–1044. [Google Scholar] [CrossRef]
- Gallop, R.S.; Taylor, H.F.W. Microstructural and microanalytical studies of sulfate attack. II. Sulfate-resisting Portland cement: Ferrite composition and hydration chemistry. Cem. Concr. Res. 1994, 24, 1347–1358. [Google Scholar] [CrossRef]
- Pérez, M.R.; Pavlovic, I.; Barriga, C.; Cornejo, J.; Hermosín, M.C.; Ulibarri, M.A. Uptake of Cu2+, Cd2+ and Pb2+ on Zn–Al layered double hydroxide intercalated with EDTA. Appl. Clay Sci. 2006, 32, 245–251. [Google Scholar]
- Dadwhal, M.; Ostwal, M.M.; Liu, P.-K.T.; Sahimi, M.; Tsotsis, T.T. Adsorption of arsenic on conditioned layered double hydroxides: Column experiments and modeling. Ind. Eng. Chem. Res. 2009, 48, 2076–2084. [Google Scholar] [CrossRef]
- Uchikawa, H.; Sawaki, D.; Hanehara, S. Influence of kind and added timing of organic admixture on the composition, structure and property of fresh cement paste. Cem. Concr. Res. 1995, 25, 353–364. [Google Scholar] [CrossRef]
- Flatt, R.J.; Houst, Y.F. A simplified view on chemical effects perturbing the action of superplasticizers. Cem. Concr. Res. 2001, 31, 1169–1176. [Google Scholar] [CrossRef]
- Ramachandran, V.S.; Beaudoin, J.J.; Paroli, R.M. The effect of nitrobenzoic and aminobenzoic acids on the hydration of tricalcium silicate: A conduction calorimetric study. Thermochim. Acta 1991, 190, 325–333. [Google Scholar] [CrossRef]
- Chandra, S.; Bjornstrom, J. Influence of superplasticizers type and dosage on the slump loss of Portland cement mortars-Part II. Cem. Concr. Res. 2002, 32, 1613–1619. [Google Scholar] [CrossRef]
- Ergogdu, S. Effect of retemping with superplasticizer admixture on slump loss and compressive strength of concrete subjected to prolonged mixing. Cem. Concr. Res. 2005, 35, 907–912. [Google Scholar] [CrossRef]
- Dry, C.M.A. Time release technique for corrosion prevention. Cem. Concr. Res. 1998, 28, 1133–1140. [Google Scholar] [CrossRef]
- Dry, C.M. Alteration of matrix permeability and associated pore and crack structure by timed release of internal chemicals. Ceram. Trans. 1991, 16, 729–768. [Google Scholar]
- Reddy, B.R.; Crook, R.J.; Chatterji, J.; King, B.J.; Gray, D.W.; Fitzgerald, R.M.; Powell, R.J.; Todd, B.L. Controlling the release of chemical additives in well treating fluids. US Pat. 6,209, 646, 2001. [Google Scholar]
- Tatematsu, H.; Nakamura, T.; Koshimuzu, H.; Morishita, T.; Kotaki, H. Cement-additive for inhibiting concrete-deterioration. US Pat. 5,435, 848, 1995. [Google Scholar]
- Raki, L.; Beaudoin, J.J.; Mitchell, L.D. Layered double hydroxide-like materials: Nanocomposites for use in concrete. Cem. Concr. Res. 2004, 34, 1717–1724. [Google Scholar] [CrossRef]
- Raki, L.; Beaudoin, J.J. Release Formulation of Chemical Admixtures in Cement and Concrete. Canadian Patent Application CA 2554347. US Patent Application US 2007/0022916 a1, 2007. [Google Scholar]
- Kantro, D.L. Influence of water reducing admixtures on properties of cement pastes-A miniature slump test. Cem. Concr. Aggregates 1980, 2, 95–102. [Google Scholar]
- AASHTO-AGC-ARTBA Joint Committee, Task Force 36. The use and state-of-the-practice of fiber reinforced concrete. American Association of State Highway and Transportation Officials: Washington, D.C., USA, 2001. [Google Scholar]
- Beaudoin, J.J. Handbook of Fibre Reinforced Concrete: Principles, Properties, Developments and Applications; Noyes Pub: Park Ridge, NJ, USA, 1990; p. 332. [Google Scholar]
- Yu, M.-F.; Lourie, O.; Dyer, M.J.; Moloni, K.; Kelly, T.F.; and Ruoff, R.S. Strength and breaking mechanism of multiwalled carbon nanotubes under tensile load. Science 2000, 287, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Salvetat, J.-P.; Bonard, J.-M.; Thomson, N.H.; Kulik, A.J.; Forró, L.; Benoit, W.; Zuppiroli, L. Mechanical properties of carbon nanotubes. Appl. Phys. A Mat. Sci. Process. 1999, 69, 255–260. [Google Scholar] [CrossRef]
- Walters, D.A.; Ericson, L.M.; Casavant, M.J.; Liu, J.; Colbert, D.T.; Smith, K.A.; Smalley, R.E. Elastic strain of freely suspended single-wall carbon nanotube ropes. Appl. Phys. Lett. 1999, 74, 3803–3805. [Google Scholar] [CrossRef]
- Sinnott, S.B.; Andrews, R. Carbon nanotubes: Synthesis, properties and applications. Crit. Rev. Sol. St. Mat. Sci. 2001, 26, 145–249. [Google Scholar] [CrossRef]
- Berber, S.; Kwon, Y.-K.; Tomanek, D. Unusually high thermal conductivity of carbon nanotubes. Phys. Rev. Lett. 2000, 84, 4613–4616. [Google Scholar] [CrossRef] [PubMed]
- Li, G.Y.; Wang, P.M.; Zhao, X. Mechanical behavior and microstructure of cement composites incorporating surface-treated multi-walled carbon nanotubes. Carbon 2005, 43, 1239–1245. [Google Scholar] [CrossRef]
- De Ibarra, Y.S.; Gaitero, J.J.; Campillo, I. Atomic force microscopy and nanoindentation of cement pastes with nanotube dispersions. Phys. Status Solidi A 2006, 203, 1076–1081. [Google Scholar] [CrossRef]
- Xiang, X.J.; Torwald, T.L.; Staedler, T.; Trettin, R.H.F. Carbon Nanotubes as a new reinforcement material for modern cement-based binders. In Proceedings of 2nd International Symposium on Nanotechnology in Construction, NICOM2, Bilbao, Spain, 13–16 November 2005; pp. 209–214.
- Cwirzen, A.; Habermehl-Cwirzen, K.; Penttala, V. Surface decoration of carbon nanotubes and mechanical properties of cement/carbon nanotube composites. Adv. Cem. Res. 2008, 20, 65–73. [Google Scholar] [CrossRef]
- Makar, J.M.; Margeson, J.; Luh, J. Carbon nanotube/cement composites-early results and potential applications, Construction Materials. In Proceedings of ConMat’05 and Mindess Symposium, Vancouver, Canada, 22–24 August 2005; p. 32.
- Makar, J.M.; Chan, G.W. Growth of cement hydration products on single walled carbon nanotubes. J. Am. Ceram. Soc. 2009, 92, 1303–1310. [Google Scholar] [CrossRef]
- Li, G.Y.; Wang, P.M.; Zhao, X. Pressure-sensitive properties and microstructure of carbon nanotube reinforced cement composites. Cem. Concr. Comp. 2007, 29, 377–382. [Google Scholar] [CrossRef]
- Wansom, S.; Kidner, N.J.; Woo, L.Y.; Mason, T.O. AC-impedance response of multi-walled carbon nanotube/cement composites. Cem. Concr. Comp. 2006, 28, 509–519. [Google Scholar] [CrossRef]
- Gengying, L. Properties of high-volume fly ash concrete incorporating nano-SiO2. Cem. Concr. Res. 2004, 34, 1043–1049. [Google Scholar] [CrossRef]
- Qing, Y.; Zenan, Z.; Deyu, K.; Rongshen, C. Influence of nano-SiO2 addition on properties of hardened cement paste as compared with silica fume. Constr. Build. Mater. 2005, 21, 539–545. [Google Scholar] [CrossRef]
- Korpa, A.; Trettin, R. Pyrogene nano-oxides in modern cement-based composites. In Proceedings of 2nd International Symposium on Nanotechnology in Construction, Bilbao, Spain, 13–16 November 2005; pp. 313–320.
- Lin, K.L.; Chang, W.C.; Lin, D.F.; Luo, H.L.; Tsai, M.C. Effects of nano-SiO2 and different ash particle sizes on sludge ash–cement mortar. J. Environ. Manag. 2008, 88, 708–714. [Google Scholar] [CrossRef]
- Li, H.; Xiao, H.; Yuan, J.; Ou, J. Microstructure of cement mortar with nano-particles. Compos. B: Eng. 2004, 35, 185–159. [Google Scholar] [CrossRef]
- Li, H.; Zhang, M.; Ou, J. Abrasion resistance of concrete containing nano-particles for pavement. Wear 2006, 260, 1262–1266. [Google Scholar] [CrossRef]
- Han, B.; Guan, X.; Ou, J. Specific resistance and pressure-sensitivity of cement paste admixing with nano-TiO2 and carbon fiber. J. Chin. Ceram. Soc. 2004, 32, 884–887. [Google Scholar]
- Xiong, G.; Deng, M.; Xu, L.; Tang, M. Properties of cement-based composites by doping nano-TiO2. J. Chin. Ceram. Soc. 2006, 34, 1158–1161. [Google Scholar]
- Sato, T.; Beaudoin, J.J.; Ramachandran, V.S.; Mitchell, L.D.; Tumidajski, P.J. Thermal decomposition of nanoparticulate Ca(OH)2–anomalous effects. Adv. Cem. Res. 2007, 19, 1–7. [Google Scholar] [CrossRef]
- Colston, S.L.; O'Connor, D.; Barnes, P.; Mayes, E.L.; Mann, S.; Freimuth, H.; Ehrfeld, W. Functional micro-concrete: The incorporation of zeolites and inorganic nano-particles into cement micro-structures. J. Mater. Sci. Lett. 2000, 19, 1085–1088. [Google Scholar] [CrossRef]
- Lee, B.Y.; Thomas, J.J.; Treager, M.; Kurtis, K.E. Influence of TiO2 Nanoparticles on Early C3S Hydration. Nanotechnology of Concrete: The Next Big Thing is Small. In ACI Convention, New Orleans, LA, USA, 8–12 November 2009; CD-ROM (SP-267). pp. 35–44.
- Cassar, L. Nanotechnology and photocalatalysis in cementitious materials. In Proceedings of 2nd International Symposium on Nanotechnology in Construction, NICOM2, Bilbao, Spain, 13–16 November 2007; pp. 277–283.
- Alizadeh, R.; Raki, L.; Makar, J.M.; Beaudoin, J.J.; Moudrakovski, I. Hydration of tricalcium silicate in the presence of synthetic calcium silicate hydrate. J. Mater. Chem. 2009, 19, 7937–7946. [Google Scholar] [CrossRef]
- Sato, T.; Beaudoin, J.J. The Effect of nano-sized CaCO3 addition on the hydration of cement paste containing high volumes of fly ash. In Proceedings of the 12th International Congress on the Chemistry of Cement, Montreal, Canada, 8–13 July 2007; pp. 1–12.
- Sato, T.; Beaudoin, J.J. The Effect of nano-sized CaCO3 addition on the hydration of OPC containing high volumes of ground granulated blast-furnace slag. In Proceedings of the 2nd International RILEM Symposium on Advances in Concrete Through Science and Engineering, Québec City, Canada, 11–13 September 2006; pp. 355–366.
- Ramachandran, V.S.; Zhang, C. Influence of CaCO3 on hydration and microstructural characteristics of tricalcium silicate. Il- Cemento 1986, 83, 29–152. [Google Scholar]
- Péra, J.; Husson, S.; Guilhot, B. Influence of finely ground limestone on cement hydration. Cem. Concr. Compos. 1999, 21, 99–105. [Google Scholar] [CrossRef]
- Van Oss, H.G. Cement, Minerals Commodity Survey; United States Geological Survey: Washington, D.C., USA, 2008; pp. 44–45. http://minerals.usgs.gov/minerals/pubs/commodity/ (accessed 1 February 2010).
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Raki, L.; Beaudoin, J.; Alizadeh, R.; Makar, J.; Sato, T. Cement and Concrete Nanoscience and Nanotechnology. Materials 2010, 3, 918-942. https://doi.org/10.3390/ma3020918
Raki L, Beaudoin J, Alizadeh R, Makar J, Sato T. Cement and Concrete Nanoscience and Nanotechnology. Materials. 2010; 3(2):918-942. https://doi.org/10.3390/ma3020918
Chicago/Turabian StyleRaki, Laila, James Beaudoin, Rouhollah Alizadeh, Jon Makar, and Taijiro Sato. 2010. "Cement and Concrete Nanoscience and Nanotechnology" Materials 3, no. 2: 918-942. https://doi.org/10.3390/ma3020918
APA StyleRaki, L., Beaudoin, J., Alizadeh, R., Makar, J., & Sato, T. (2010). Cement and Concrete Nanoscience and Nanotechnology. Materials, 3(2), 918-942. https://doi.org/10.3390/ma3020918



