Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications
Abstract
:1. Introduction
2. Physicochemical Properties of BCP that Influence Bone Formation
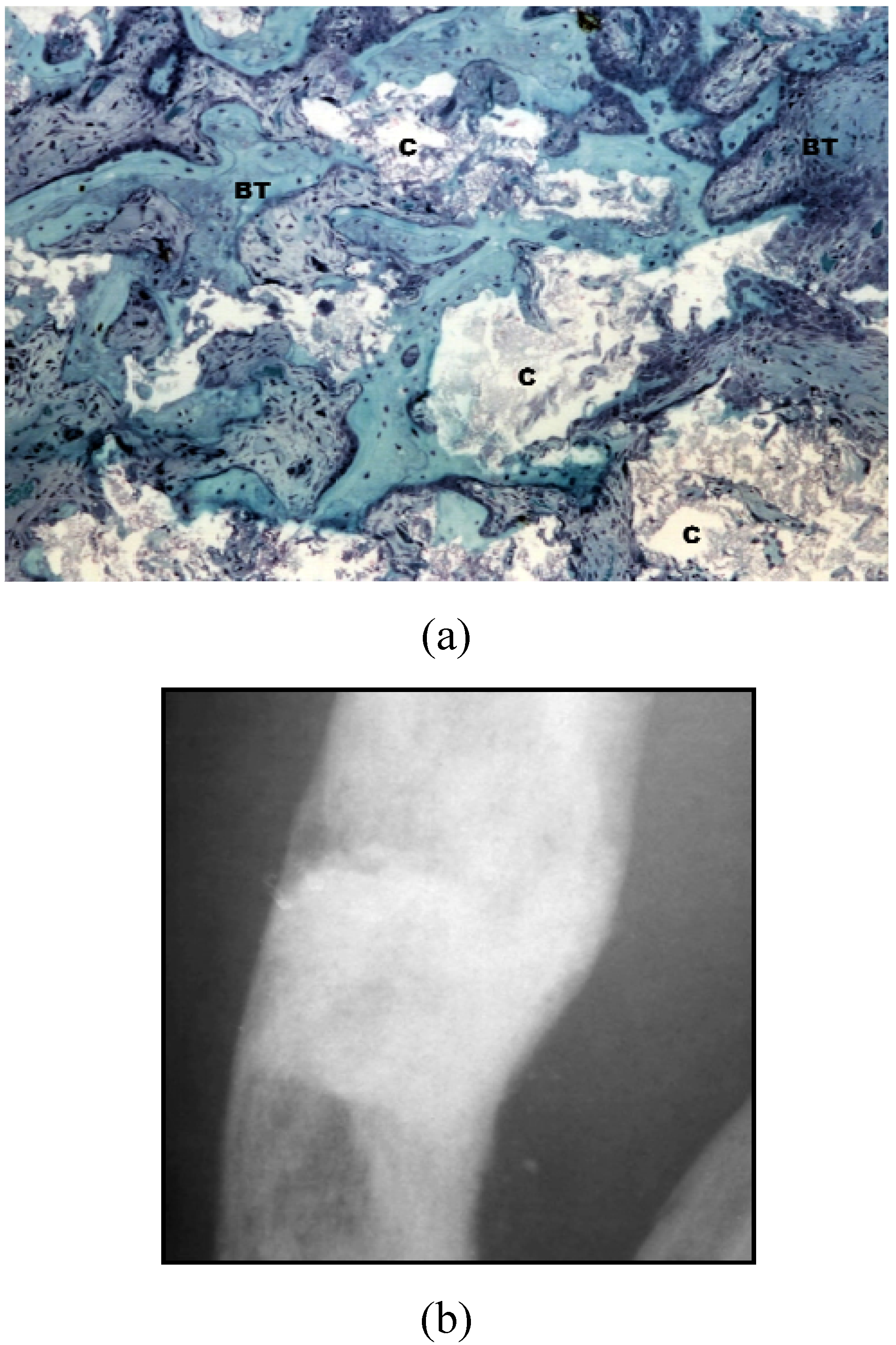

3. BCP Ceramics in Bone Tissue Engineering

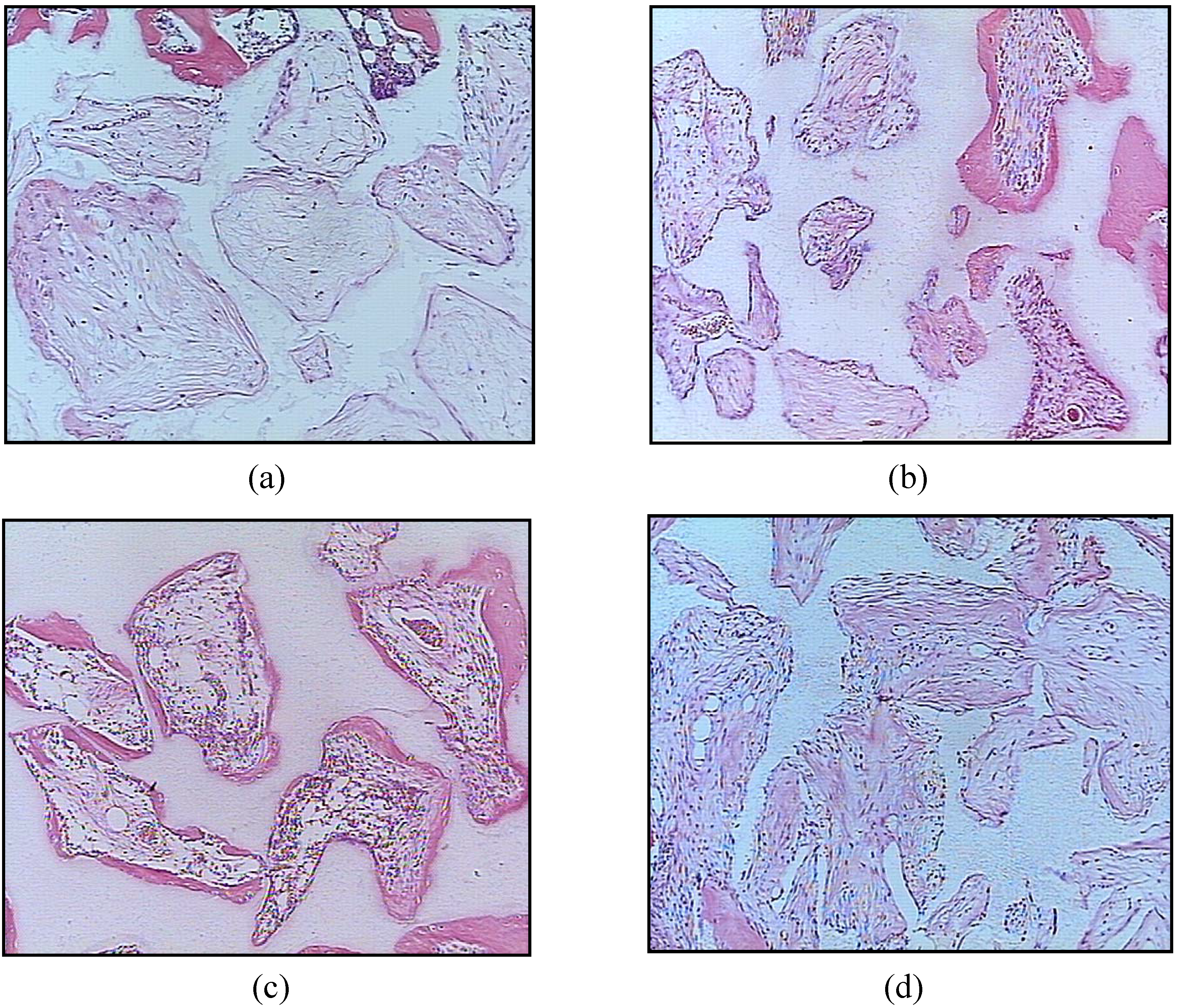
4. Conclusions
References
- Gauthier, O.; Bouler, J.M.; Aguado, E.; LeGeros, R.Z.; Pilet, P.; Daculsi, G. Elaboration conditions influence physicochemical properties and in vivo bioactivity of macroporous biphasic calcium phosphate ceramics. J. Mater. Sci. Mater Med. 1999, 10, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Barrère, F.; van Blitterswijk, C.A.; de Groot, K. Bone regeneration: Molecular and cellular interactions with calcium phosphate ceramics. Int. J. Nanomed. 2006, 1, 317–332. [Google Scholar]
- Le Nihouannen, D.; Duval, L.; Lecomte, A.; Julien, M.; Guicheux, J.; Daculsi, G.; Layrolle, P. Interactions of total bone marrow cells with increasing quantities of macroporous calcium phosphate ceramic granules. J. Mater. Sci. Mater. Med 2007, 18, 1983–1990. [Google Scholar]
- Khan, Y.; Yaszemski, M.J.; Mikos, A.G.; Laurencin, C.T. Tissue engineering of bone: Material and matrix considerations. J. Bone Joint. Surg. Am. 2008, 90, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Schawartz, C.; Liss, P.; Jacquemaire, B.; Lecestre, P.; Frayssinet, P. Biphasic synthetic bone substitute use in orthopaedic and trauma surgery: Clinical, radiological and histological results. J. Mater. Sci. Mater. Med. 1999, 10, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Ransford, A.O.; Morley, T.; Edgar, M.A.; Webb, P.; Passuti, N.; Chopin, D.; Morin, C.; Michel, F.; Garin, C.; Pries, D. Synthetic porous ceramic compared with autograft in scoliosis surgery: A prospective randomized study of 341 patients. J. Bone Join. Surg. Br. 1998, 80, 13–18. [Google Scholar] [CrossRef]
- Cavagna, R.; Daculsi, G.; Bouler, J. Macroporous calcium phosphate ceramic: A prospective study of 106 cases in lumbar spinal fusion. J. Long Term Eff. Med. Implants 1999, 9, 403–412. [Google Scholar] [PubMed]
- Davies, J.E. Bone Engineering; Em Squared Inc: Toronto, Canada, 2000; pp. 516–525. [Google Scholar]
- Bagot D`Arc, M.; Daculsi, G. Micro macroporous biphasic ceramics and fibrin sealant as a moldable material for bone reconstruction in chronic otitis media surgery: A 15 years experience. J. Mater. Sci. Mater. Med 2003, 14, 229–233. [Google Scholar]
- Nich, C.; Bizot, P.; Nizard, R.; Sedel, L. Femoral reconstruction with macroporous biphasic calcium phosphate ceramic in revision hip replacement. Key Engin. Mater. 2003, 240–242, 853–856. [Google Scholar] [CrossRef]
- Hing, K.A.; Wilson, L.F.; Buckland, T. Comparative performance of three ceramic bone graft substitutes. Spine J. 2007, 7, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Adamopoulos, O.; Papadopoulos, T. Nanostructured bioceramics for maxillofacial applications. J. Mater. Sci. Mater. Med. 2007, 18, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Schindler, O.S.; Cannon, S.R.; Briggs, T.W.; Blunn, G.W. Composite ceramic bone graft substitute in the treatment of locally aggressive benign bone tumors. J. Orthop. Surg. 2008, 16, 66–74. [Google Scholar]
- LeGeros, R.Z.; Lin, S.; Rohanizadeh, R.; Mijares, D.; LeGeros, J.P. Biphasic calcium phosphate bioceramics: Preparation, properties and applications. J. Mater. Sci. Mater. Med. 2003, 14, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Daculsi, G.; LeGeros, R. Biphasic calcium phosphate (BCP) bioceramics: Chemical, physical and biological properties. Enc. Biomat. Biomed. Eng. 2006, 1–9. [Google Scholar]
- Habibovic, P.; Sees, T.M.; van den Doel, M.A.; van Blitterswijk, C.A.; de Groot, K. Osteoinduction by biomaterials—physicochemical and structural influences. J. Biomed. Mater. Res. A 2006, 77, 747–762. [Google Scholar] [CrossRef] [PubMed]
- Lobo, S.E.; Wykrota, F.H.; Oliveira, A.C.; Kerkis, I.; Mahecha, G.B.; Alves, H.J. Quantification of bone mass gain in response to the application of biphasic bioceramics and platelet concentrate in critical-size bone defects. J. Mater. Sci. Mater. Med. 2009, 20, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Kurashima, K.; de Bruijn, J.D.; Li, Y.; de Groot, K.; Zhang, X. A preliminary study on osteoinduction of two kinds of calcium phosphate ceramics. Biomaterials 1999, 20, 1799–1806. [Google Scholar] [CrossRef]
- Daculsi, G.; Layrolle, P. Osteoinductive properties of micro macroporous biphasic calcium phosphate bioceramics. Key Eng. Mat. 2004, 254–256, 1005–1008. [Google Scholar] [CrossRef]
- Le Nihouannen, D.; Daculsi, G.; Saffarzadeh, A.; Gauthier, O.; Delplace, S.; Pilet, P.; Layrolle, P. Ectopic bone formation by microporous calcium phosphate ceramic particles in sheep muscles. Bone 2005, 36, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Habibovic, P.; Yuan, H.; van der Valk, C.M.; Meijer, G.; van Blitterswijk, C.A.; de Groot, K. 3D microenvironment as essential element for osteoinduction by biomaterials. Biomaterials 2005, 26, 3565–3575. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, U.; Crooks, J.; Kirkbride, A.N. Sintered porous hydroxyapaptites with intrinsic osteoinductive activity: Geometric induction of bone formation. S. Afr. J. Sci. 1999, 95, 335–343. [Google Scholar]
- Ripamonti, U.; Richter, P.W.; Thomas, M.E. Self-inducing shape memory geometric cues embedded within smart hydroxyapatite-based biomimetic matrices. Plast. Reconstr. Surg. 2007, 120, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Rochet, N.; Loubat, A.; Laugier, J.P.; Hofman, P.; Bouler, J.M.; Daculsi, G.; Carle, G.F.; Rossi, B. Modification of gene expression induced in human osteogenic and osteosarcoma cells by culture on a biphasic calcium phosphate bone substitute. Bone 2003, 32, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.R.; Zhang, Z.R.; Wang, C.Y.; Chen, J.Y.; Zhang, X.D. Dynamic study of calcium phosphate formation on porous HA/TCP ceramics. J. Mater. Sci. Mater. Med. 2004, 15, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Lu, X.; Lu, B.; Wang, J.; Shi, Y.; Zhang, L.; Chen, J.; Li, Y.; Bu, H. A long-term evaluation of osteoinductive HA/β-TCP ceramics in vivo: 4.5 years study in pigs. J. Mater. Sci. Mater. Med. 2007, 18, 2173–2178. [Google Scholar] [CrossRef] [PubMed]
- Daculsi, G.; LeGeros, R.Z.; Nery, E.; Lynch, K.; Kerebel, B. Transformation of biphasic calcium phosphate ceramics in vivo: Ultrastructural and physicochemical characterization. J. Biomed. Mater. Res. 1989, 23, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Daculsi, G.; LeGeros, R.Z.; Heughebaert, M.; Barbieux, I. Formation of carbonate-apatite crystals after implantation of calcium phosphate ceramics. Calcif. Tissue Int. 1990, 46, 20–27. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.S.; Brigado, R.; Madureira, J.F.; Cruz, A.A.; de Mello Filho, F.V.; Alonso, N.; Machado, H.R. Reconstruction of a large complex skull defect in a child: A case report and literature review. Childs Nerv. Syst 2007, 23, 1097–1102. [Google Scholar]
- Hing, K.A. Bioceramic bone graft substitutes: Influence of porosity and chemistry. Int. J. Appl. Ceram. Technol. 2005, 2, 184–199. [Google Scholar] [CrossRef]
- Arcos, D.; Izquierdo-Barba, I.; Vallet-Regí, M. Promising trends of bioceramics in the biomaterials field. J. Mater. Sci. Mater. Med. 2009, 20, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Zyman, Z.; Glushko, V.; Dedukh, N.; Malyshkina, S.; Ashukina, N. Porous calcium phosphate ceramic granules and their behavior in differently loaded areas of skeleton. J. Mater. Sci. Mater. Med. 2008, 19, 2197–2205. [Google Scholar] [CrossRef] [PubMed]
- Papadimitropoulos, A.; Mastrogiacomo, M.; Peyrin, F.; Molinari, E.; Komlev, V.S.; Rustichelli, F.; Cancedda, R. Kinetics of in vivo bone deposition by bone marrow stromal cells within a resorbable porous calcium phosphate scaffold: An X-ray computed microtomography study. Biotechnol. Bioeng. 2007, 98, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Christophy, C.; Rashid, N.; di Silvio, L.; Hing, K.A. Encouraging nature with ceramics: The roles of surface roughness and physio-chemistry on cell response to substituted apatites. Adv. Sci. Tech. 2008, 57, 22–30. [Google Scholar] [CrossRef]
- Yuan, H.; de Bruijn, J.D.; Li, Y.; Feng, J.; Yang, Z.; de Groot, K.; Zhang, X. Bone formation induced by calcium phosphate ceramics in soft tissue of dogs: A comparative study between porous alpha-TCP and beta-TCP. J. Mater. Sci. Mater. Med 2001, 12, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.F.; Huang, W.; Dunn, L.C.; Miller, T.A.; Wu, B.M. The effect of biomimetic apatite structure on osteoblast viability, proliferation and gene expression. Biomaterials 2005, 26, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.E.M.; Rathod, H.; Gillespie, J.I.; Horrocks, B.R.; Datta, H.K. Scanning electrochemical microscopy at the surface of bone-resorbing osteoclasts: Evidence for steady-state disposal and intracellular functional compartmentalization of calcium. J. Bone Min. Res. 2001, 16, 2092–2102. [Google Scholar] [CrossRef]
- Zaidi, M.; Moonga, B.S.; Huang, C.L. Calcium sensing and cell signaling process in the local regulation os osteoclastic bone resorption. Biol. Rev. Camb. Philos. Soc 2004, 79, 79–100. [Google Scholar]
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Toms, B.B.; Goodisman, J.; Asefa, T. Mesoporosity and functional group dependent endocytosis and cytotoxicity of silica nanomaterials. Chem. Res. Toxicol. 2009, 22, 1869–1880. [Google Scholar] [CrossRef] [PubMed]
- Bhabra, G.; Sood, A.; Fisher, B.; Cartwright, L.; Saunders, M.; Evans, W.H.; Surprenant, A.; Lopez-Castejon, G.; Mann, S.; Davis, S.A.; Hails, L.A.; Ingham, E.; Verkade, P.; Lane, J.; Heesom, K.; Newson, R.; Case, C.P. Nanoparticles can cause DNA damage across a cellular barrier. Nat. Nanotechnol. 2009, 4, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, M.; Casciano, D.A.; Mocan, T.; Iancu, C.; Xu, Y.; Mocan, L.; Iancu, D.T.; Dervishi, E.; Li, Z.; Abdalmuhsen, M.; Biris, A.R.; Ali, N.; Howard, P.; Biris, A.S. Cytotoxicity and biological effects of functional nanomaterials delivered to various cell lines. J. Appl. Toxicol. 2009, 30, 74–83. [Google Scholar] [CrossRef]
- Shi, Z.; Huang, X.; Liu, B.; Tao, H.; Cai, Y.; Tang, R. Biological response of osteosarcoma cells to size-controlled nanostructured hydroxyapatite. J. Biomater. Appl. 2009, in press. [Google Scholar]
- Fellah, B.H.; Josselin, N.; Chappard, D.; Weiss, P.; Layrolle, P. Inflammatory reaction in rats muscle after implantation of biphasic calcium phosphate micro particles. J. Mater. Sci. Mater. Med. 2007, 18, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Saldaňa, L.; Sánchez-Salcedo, S.; Izquierdo-Barba, I.; Bensiamar, F.; Munuera, L.; Vallet-Regí, M.; Vilaboa, N. Calcium phosphate-based particles influence osteogenic maturation of human mesenchymal stem cells. Acta. Biomater. 2009, 5, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Logeart-Avramoglou, D.; Anagnostou, F.; Bizios, R.; Petite, H. Engineering bone: challenges and obstacles. J. Cell Mol. Med 2005, 9, 72–84. [Google Scholar]
- Graziano, A.; d`Aquino, R.; Cusella-de Angelis, M.G.; de Francesco, F.; Giordano, A.; Laino, G.; Piattelli, A.; Traini, T.; de Rosa, A.; papaccio, G. Scaffold’s surface geometry significantly affects human stem cell bone tissue engineering. J. Cell. Physiol. 2008, 214, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Hing, K.A.; Annaz, B.; Saeed, S.; Revell, P.A.; Buckland, T. Microporosity enhances bioactivity of synthetic bone graft substitutes. J. Mater. Sci. Mater. Med. 2005, 16, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Mastrogiacomo, M.; Scaglione, S.; Martinetti, R.; Dolcini, L.; Beltrame, F.; Cancedda, R.; Quarto, R. Role of scaffold internal structure on in vivo bone formation in macroporous calcium phosphate bioceramics. Biomaterials 2006, 27, 3230–3237. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, U. Osteoinduction in porous hydroxyapatite implanted in heterotopic sites of different animal models. Biomaterials 1996, 17, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Giannoni, P.; Mastrogiacomo, M.; Alini, M.; Pearce, S.G.; Corsi, A.; Santolini, F.; Muraglia, A.; Bianco, P.; Cancedda, R. Regeneration of large bone defects in sheep using bone marrow stromal cells. J. Tissue Eng. Regen. Med. 2008, 2, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Petite, H.; Viateau, V.; Bensaid, W.; Meunier, A.; de Pollak, C.; Bourguignon, M.; Oudina, K.; Sedel, L.; Guillemin, G. Tissue-engineered bone regeneration. Nat. Biotechnol. 2000, 18, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; de Bruijn, J.D.; Zhang, X.; van Blitterswijk, C.A.; de Groot, K. Use of an osteoinductive biomaterial as a bone morphogenetic protein carrier. J. Mater. Sci. Mater. Med. 2001, 12, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Arinzeh, T.L.; Tran, T.; Mcalary, J.; Daculsi, G. A comparative study of biphasic calcium phosphate ceramics for human mesenchymal stem-cell-induced bone formation. Biomaterials 2005, 26, 3631–3638. [Google Scholar] [CrossRef] [PubMed]
- Marcacci, M.; Kon, E.; Mouhachev, V.; Lavroukov, A.; Kutepov, S.; Quarto, R.; Mastrogiacomo, M.; Cancedda, R. Stem cells associated with macroporous bioceramics for long bone repair: 6- to 7-year outcome of a pilot clinical study. Tissue Eng. 2007, 13, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Arinzeh, T.L. Mesenchymal stem cells for bone repair: preclinical studies and potential clinical applications. Foot Ankle Clinics 2005, 10, 651–665. [Google Scholar] [CrossRef] [PubMed]
- Eniwumide, J.O.; Yuan, H.; Cartmell, S.H.; Meijer, G.J.; de Bruijn, J.D. Ectopic bone formation in bone marrow stem cell seeded calcium phosphate scaffolds as compared to autograft and (cell seeded) allograft. Eur. Cell. Mater. 2007, 14, 30–38; discussion 39. [Google Scholar] [PubMed]
- Sun, H.; Ye, F.; Wang, J.; Shi, Y.; Tu, Z.; Bao, J.; Qin, M.; Bu, H.; Li, Y. The upregulation of osteoblast marker genes in mesenchymal stem cells prove the osteoinductivity of hydroxyapatite/tricalcium phosphate biomaterial. Transplant. Proc. 2008, 40, 2645–2648. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, A.; Kotobuki, N.; Tadokoro, M.; Kawate, K.; Yajima, H.; Takakura, Y.; Ohgushi, H. In vivo osteogenic capability of human mesenchymal cells cultured on hydroxyapatite and on beta-tricalcium phosphate. Artif. Organs. 2009, 33, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Viateau, V.; Guillemin, G.; Bousson, V.; Oudina, K.; Hannouche, D.; Sedel, L.; Logeart-Avramoglou, D.; Petite, H. Long-bone critical-size defects treated with tissue-engineered grafts: A study on sheep. J. Orthop. Res. 2007, 25, 741–749. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lobo, S.E.; Livingston Arinzeh, T. Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications. Materials 2010, 3, 815-826. https://doi.org/10.3390/ma3020815
Lobo SE, Livingston Arinzeh T. Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications. Materials. 2010; 3(2):815-826. https://doi.org/10.3390/ma3020815
Chicago/Turabian StyleLobo, Sonja Ellen, and Treena Livingston Arinzeh. 2010. "Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications" Materials 3, no. 2: 815-826. https://doi.org/10.3390/ma3020815
APA StyleLobo, S. E., & Livingston Arinzeh, T. (2010). Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications. Materials, 3(2), 815-826. https://doi.org/10.3390/ma3020815



