Progress of the Application of Mesoporous Silica-Supported Heteropolyacids in Heterogeneous Catalysis and Preparation of Nanostructured Metal Oxides
Abstract
:1. Introduction
2. Heterogeneous Catalysis by HPAs Supported on Mesoporous Silicas
2.1. Impregnation of HPA on mesoporous silica
2.1.2. Dehydration reactions
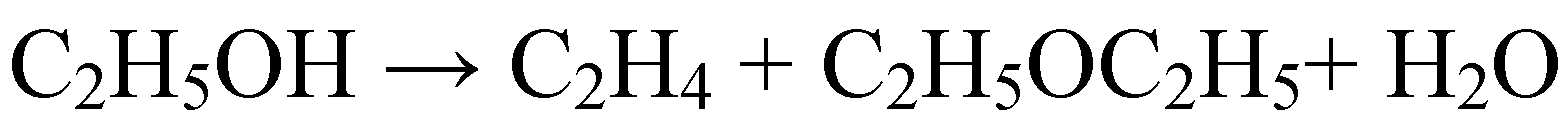
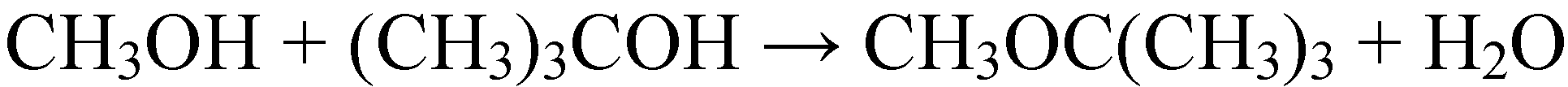
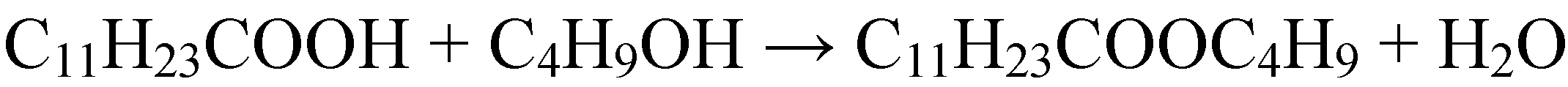
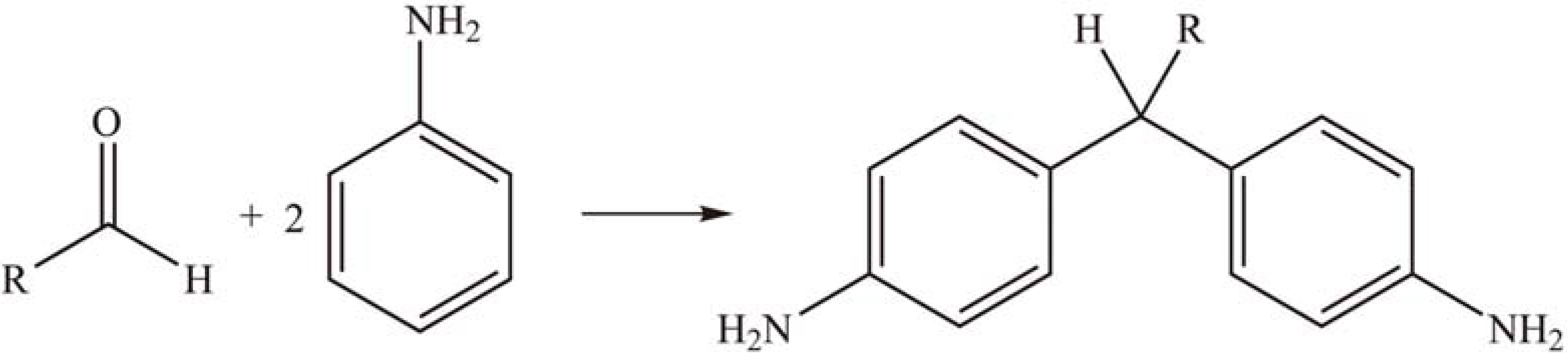
2.1.3. Condensation reactions
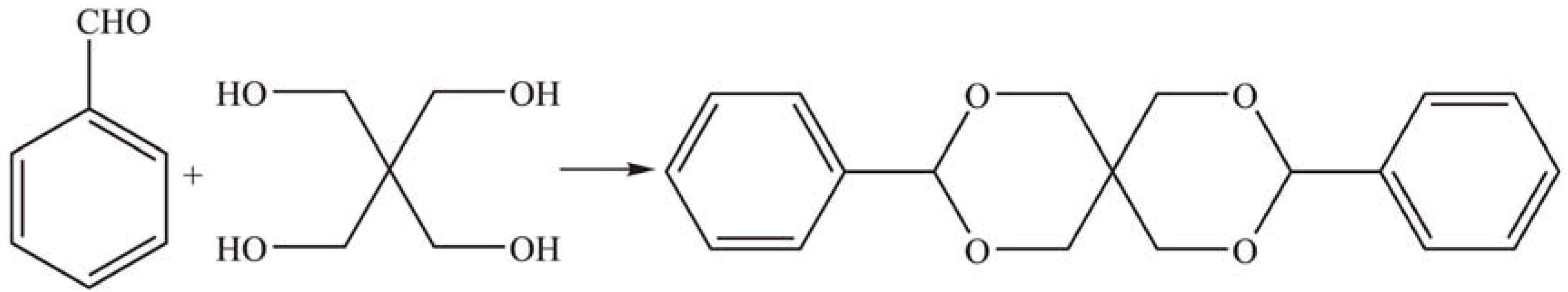
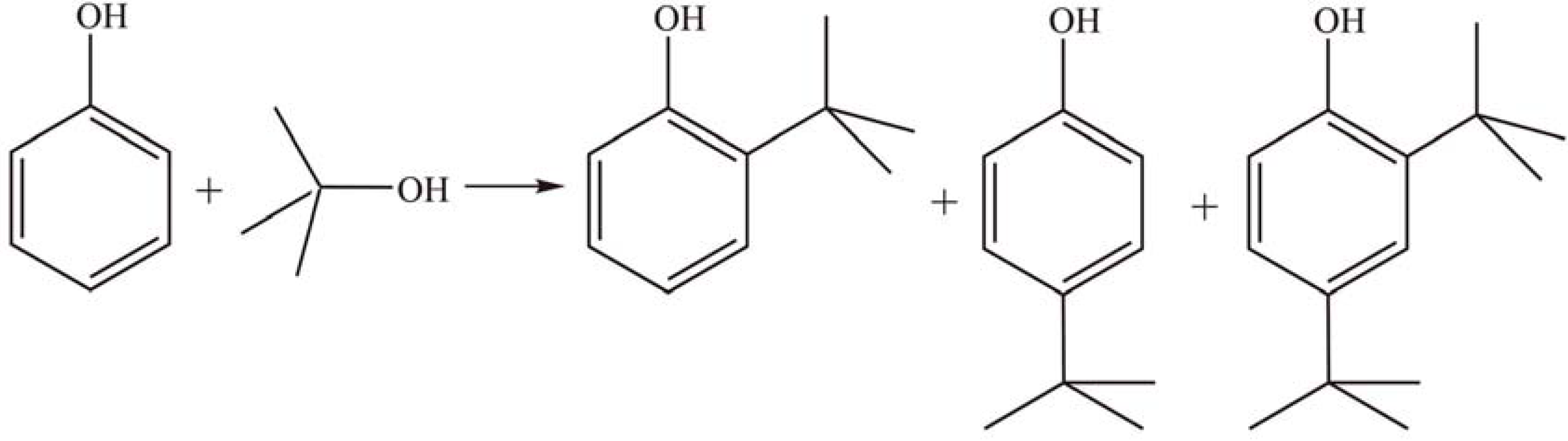
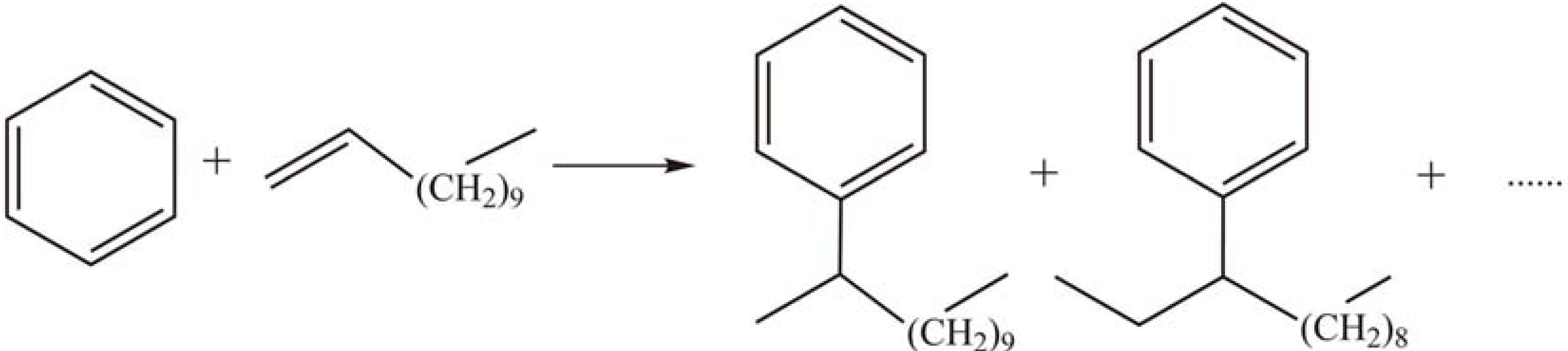
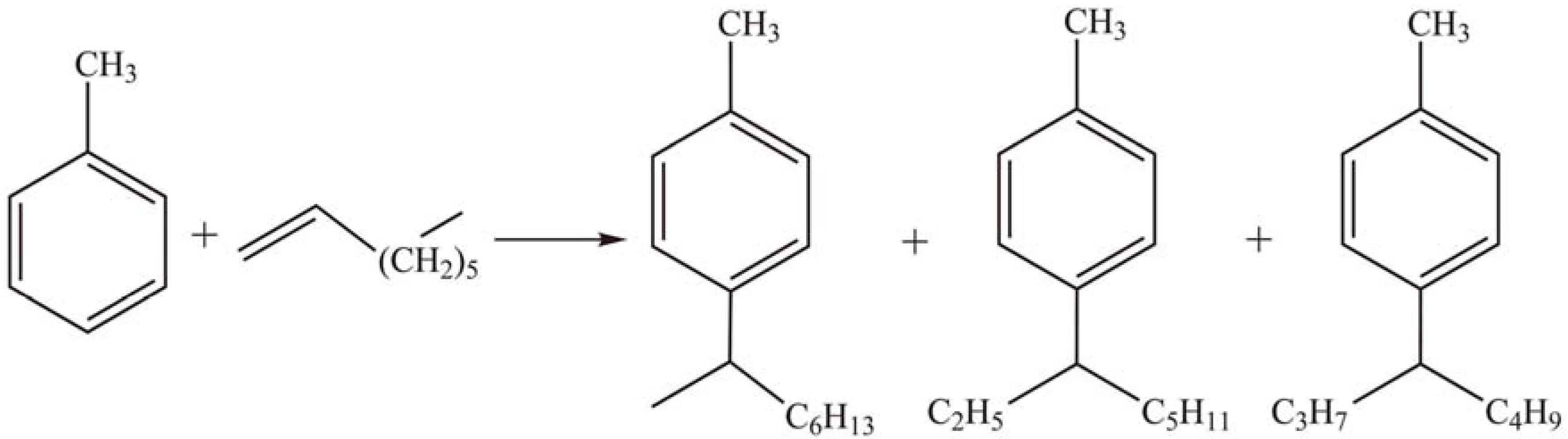
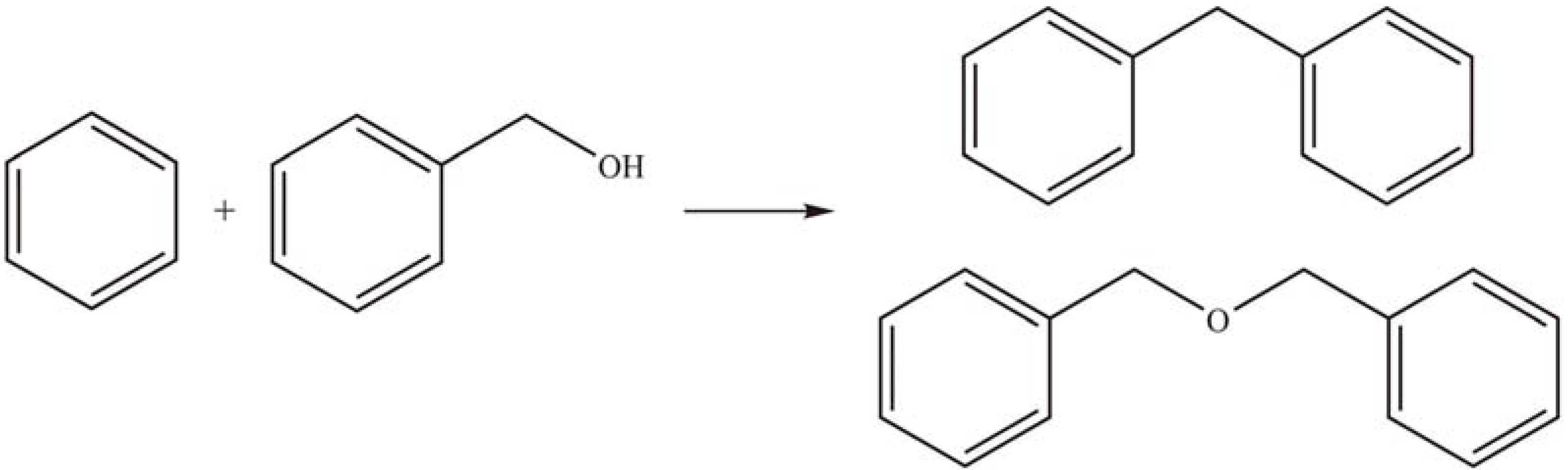
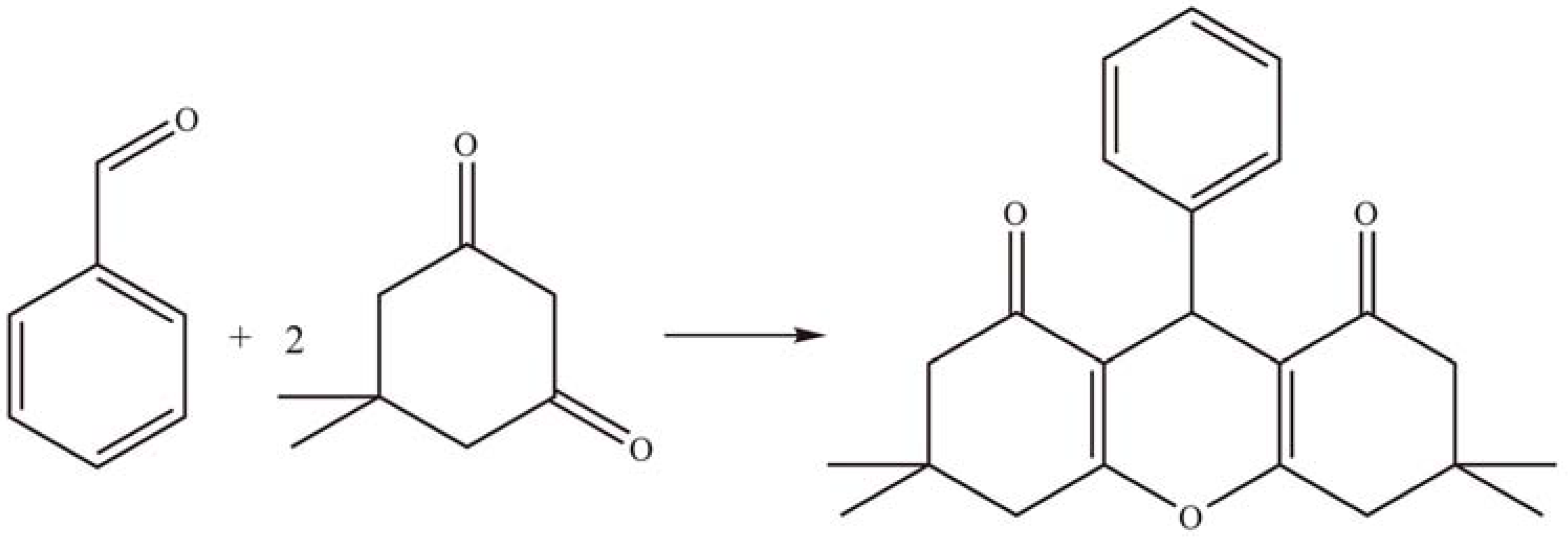
2.1.4. Other reactions
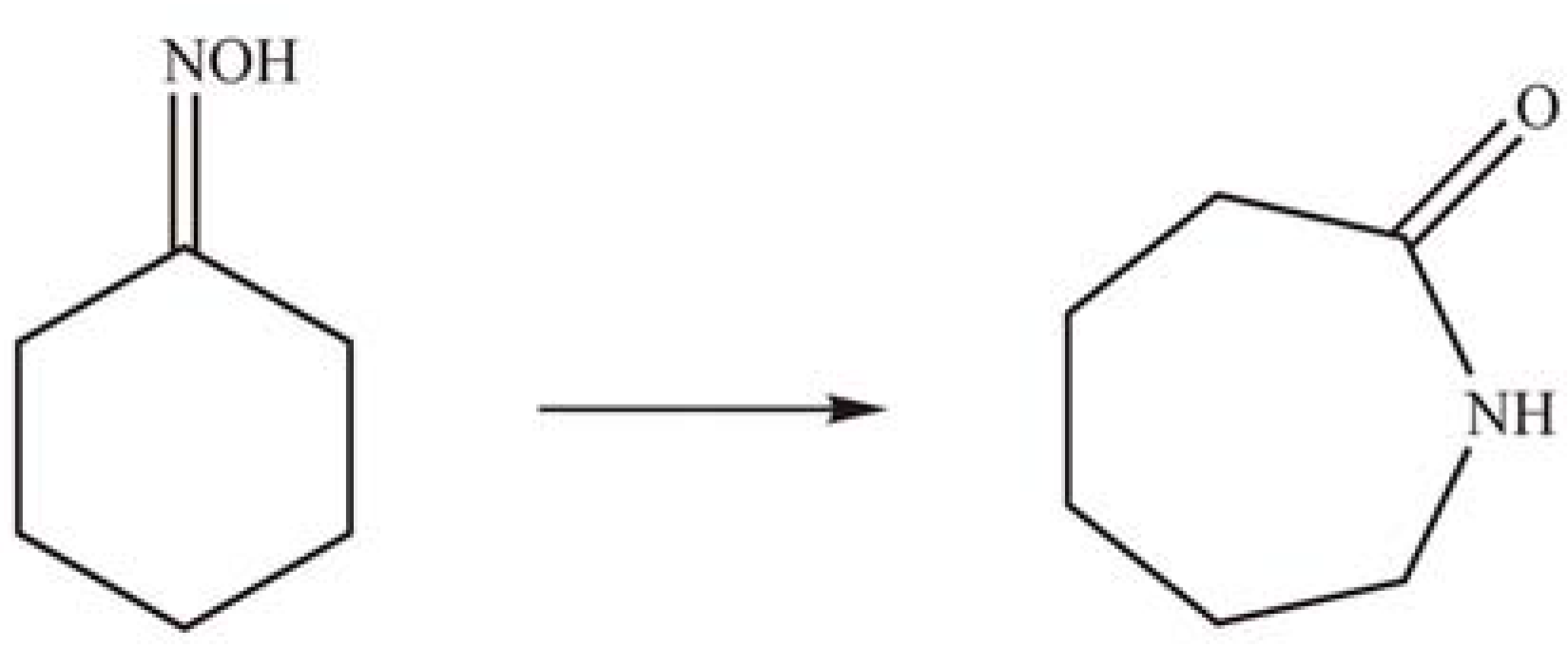
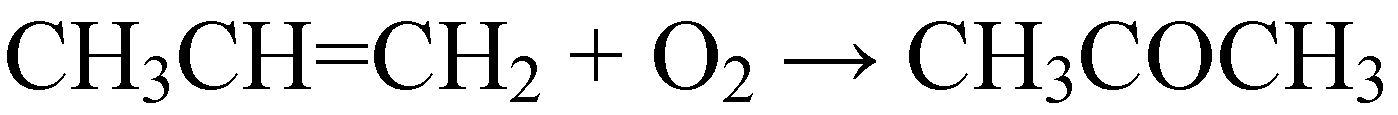
2.2. Grafting of HPAs on amino-functionalized mesoporous silica
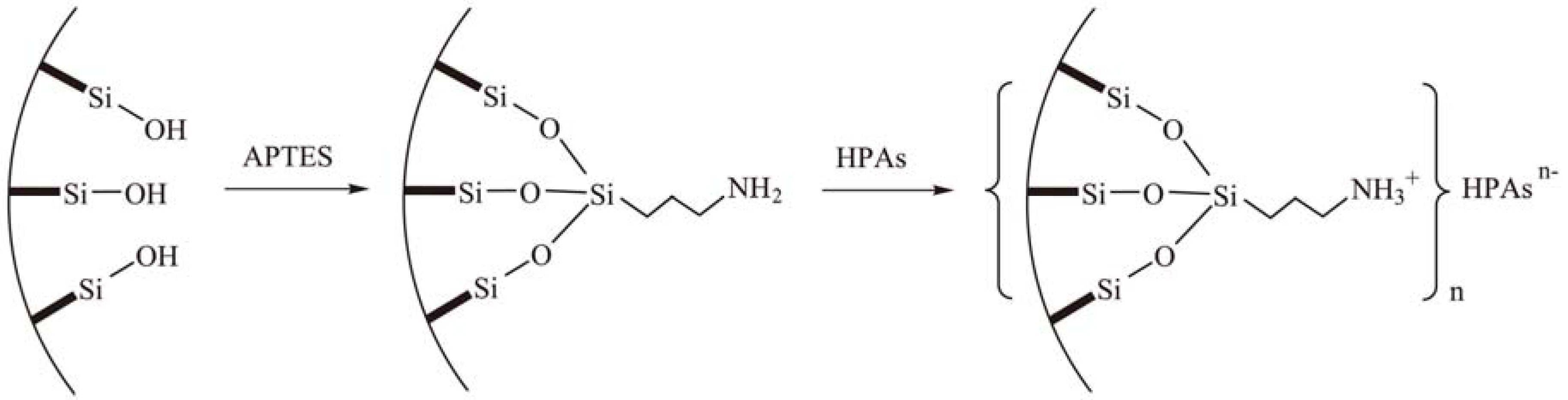
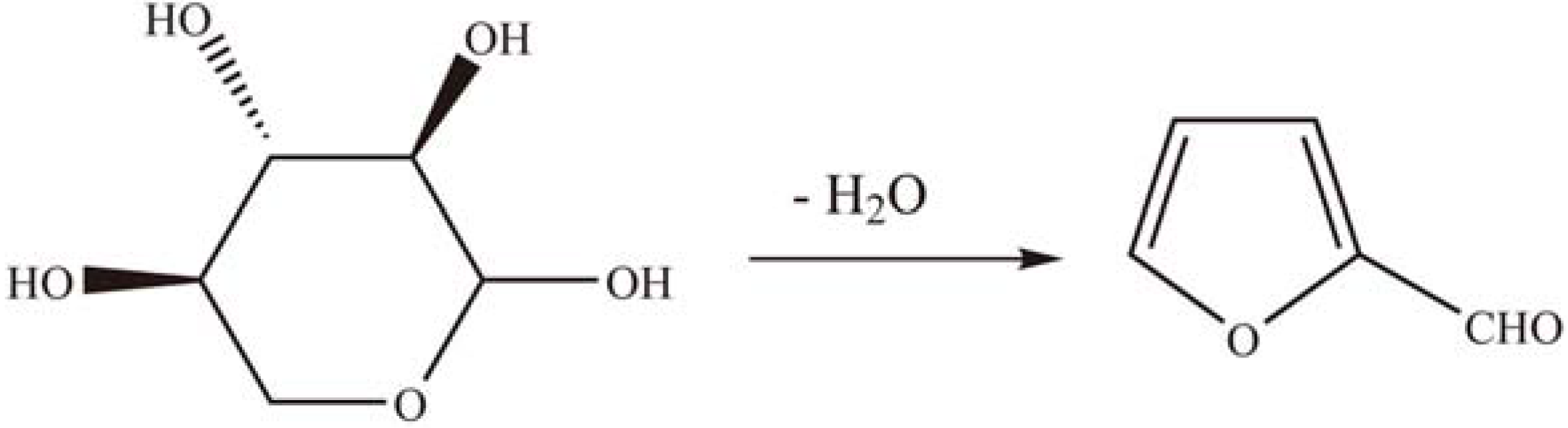
2.2.1. Dehydration reactions
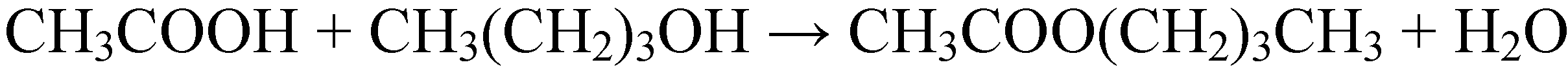
2.2.2. Oxidation reactions
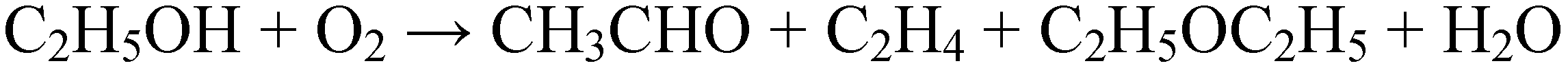
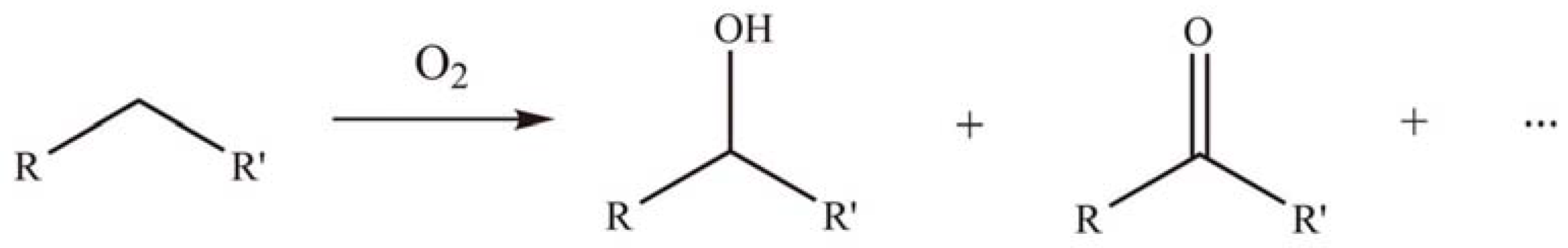
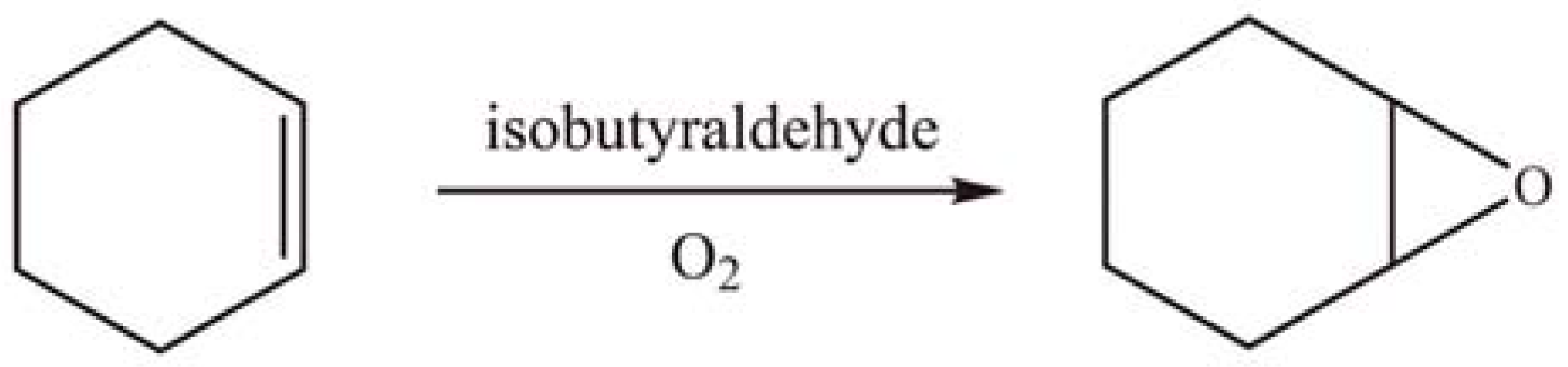
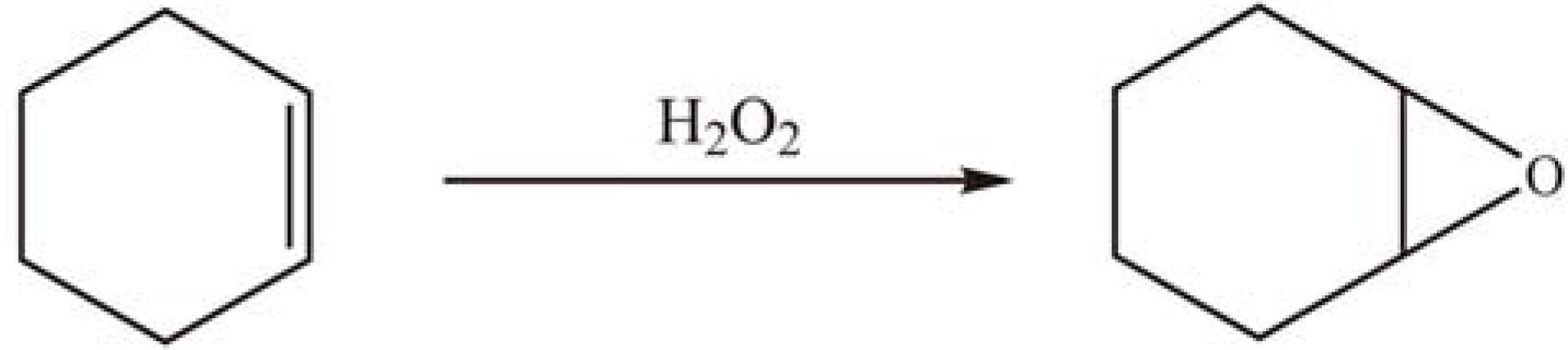
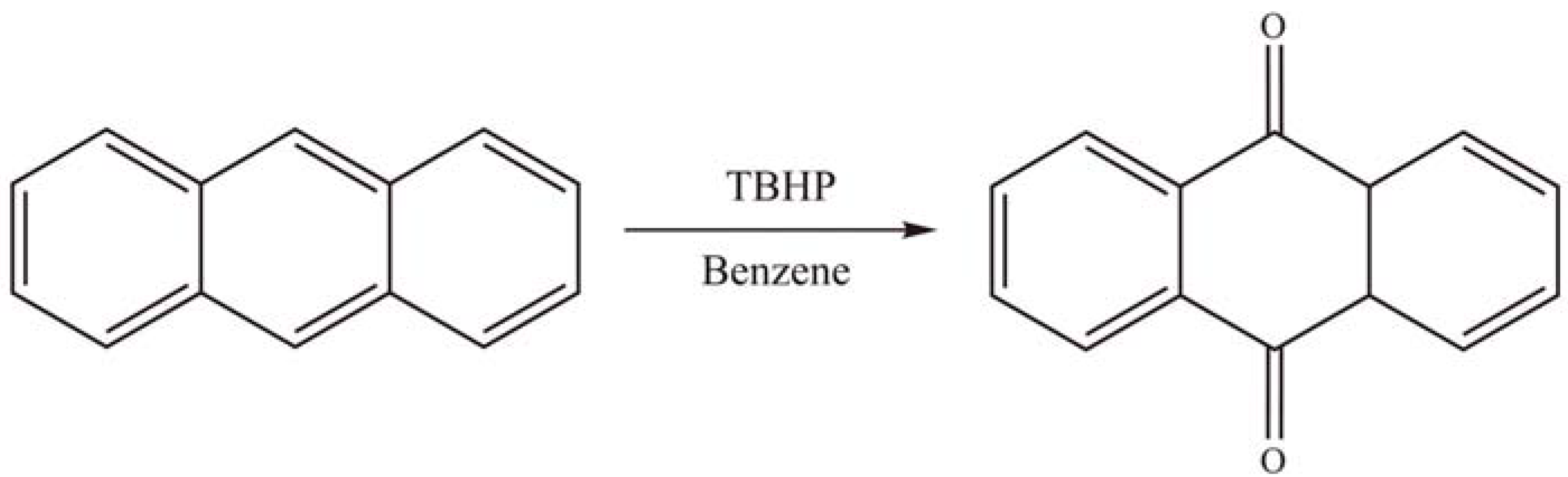
2.2.3. Photocatalytic degradation
2.3. Entrapping of HPAs in porous silica matrix via a sol-gel method
2.3.1. Dehydration reactions
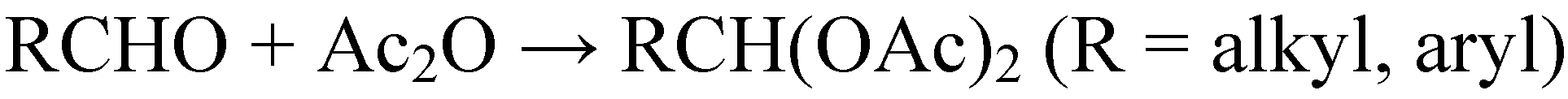
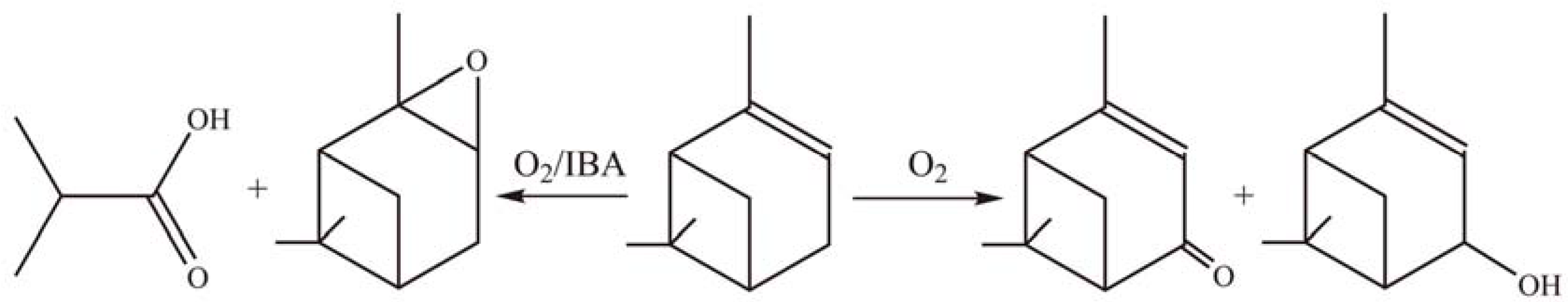
2.3.2. Oxidation reactions
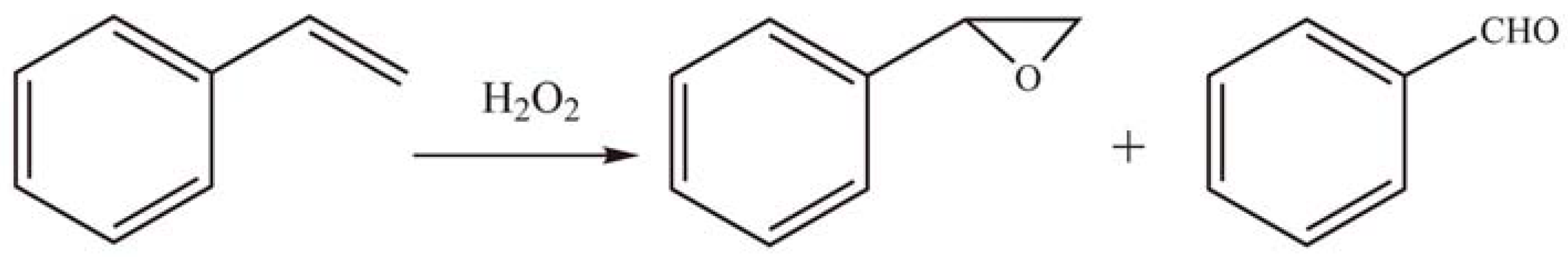
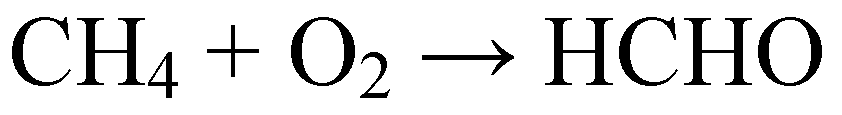
3. Preparation of Nanostructured Metal Oxides Based on HPAs as Precursors and Mesoporous Silicas as Hard Templates
3.1. Preparation of one-dimensional metal oxide nanowires based on HPAs
3.2. Preparation of three-dimensional nanoporous metal oxides based on HPAs


4. Conclusions
Acknowledgements
References and Notes
- Kozhevnikov, I.V. Advances in catalysis by heteropolyacids. Russ. Chem. Rev. 1987, 56, 811–825. [Google Scholar] [CrossRef]
- Kozhevnikov, I.V. Heteropoly acids and related-compounds as catalysts for fine chemical synthesis. Catal. Rev. Sci. Eng. 1995, 37, 311–352. [Google Scholar] [CrossRef]
- Misono, M. Heterogeneous catalysis by heteropoly compounds of molybdenum and tungsten. Catal. Rev. Sci. Eng. 1987, 29, 269–321. [Google Scholar] [CrossRef]
- Misono, M. New catalytic aspects of heteropolyacids and related-compounds - to the molecular design of practical catalysts. Stud. Surf. Sci. Catal. 1993, 75, 69–101. [Google Scholar]
- Misono, M.; Nojiri, N. Recent progress in catalytic technology in Japan. Appl. Catal. 1990, 64, 1–30. [Google Scholar] [CrossRef]
- Okuhara, T.; Mizuno, N.; Misono, M. Catalytic chemistry of heteropoly compounds. Adv. Catal. 1996, 41, 113–252. [Google Scholar]
- Kozhevnikov, I.V. Friedel-Crafts acylation and related reactions catalysed by heteropoly acids. Appl. Catal. A: Gen. 2003, 256, 3–18. [Google Scholar] [CrossRef]
- Kozhevnikov, I.V. Catalysis by heteropoly acids and multicomponent polyoxometalates in liquid-phase reactions. Chem. Rev. 1998, 98, 171–198. [Google Scholar] [CrossRef] [PubMed]
- Kozhevnikov, I.V.; Matveev, K.I. Homogeneous catalysts based on heteropoly acids. Appl. Catal. 1983, 5, 135–150. [Google Scholar] [CrossRef]
- Misono, M.; Okuhara, T. Solid superacid catalysts. Chemtech 1993, 23, 23–29. [Google Scholar]
- Okuhara, T.; Nishimura, T.; Watanabe, H.; Na, K.; Misono, M. Novel catalysis of cesium salt of heteropoly acid and its characterization by solid-state NMR. Stud. Surf. Sci. Catal. 1994, 90, 419–428. [Google Scholar]
- Okuhara, T.; Nishimura, T.; Misono, M. Microporous heteropoly compound as a shape-selective catalyst—Cs2.2H0.8PW12O40. Chem. Lett. 1995, 155–156. [Google Scholar] [CrossRef]
- Bonardet, J.L.; Fraissard, J.; McGarvey, G.B.; Moffat, J.B. A comparative-study of the microporosity of the ammonium and cesium salts of 12-Tungstophosphoric, 12-Molybdophosphoric, and 12-Tungstosilicic acids by Xe-129 NMR. J. Catal. 1995, 151, 147–154. [Google Scholar] [CrossRef]
- Na, K.; Okuhara, T.; Misono, M. Skeletal isomerization of n-butane catalyzed by an acidic cesium salt of 12-tungstophosphoric acid. Chem. Lett. 1993, 1141–1144. [Google Scholar] [CrossRef]
- Izumi, Y.; Hasebe, R.; Urabe, K. Catalysis by heterogeneous supported heteropoly acid. J. Catal. 1983, 84, 402–409. [Google Scholar] [CrossRef]
- Rocchiccioli-Deltcheff, C.; Amirouche, M.; Herve, G.; Fournier, M.; Che, M.; Tatibouet, J.M. Structure and catalytic properties of silica-supported polyoxomolybdates: II. Thermal behavior of unsupported and silica-supported 12-molybdosilic acid catalysts from IR and catalytic reactivity studies. J. Catal. 1990, 126, 591–599. [Google Scholar] [CrossRef]
- Edwards, J.C.; Thiel, C.Y.; Benac, B.; Knifton, J.F. Solid-state NMR and FT-IR investigation of 12-tungstophosphoric acid on TiO2. Catal. Lett. 1998, 51, 77–83. [Google Scholar] [CrossRef]
- Lopez-Salinas, E.; Hernandez-Cortez, J.G.; Schifter, I.; Torres-Garcia, E.; Navarrete, J.; Gutierrez-Carrillo, A.; Lopez, T.; Lottici, P.P.; Bersani, D. Thermal stability of 12-tungstophosphoric acid supported on zirconia. Appl. Catal. A: Gen. 2000, 193, 215–225. [Google Scholar] [CrossRef]
- Devassy, B.M.; Halligudi, S.B.; Hegde, S.G.; Halgeri, A.B.; Lefebvre, F. 12-Tungstophosphoric acid/zirconia - a highly active stable solid acid - comparison with a tungstated zirconia catalyst. Chem. Commun. 2002, 1074–1075. [Google Scholar] [CrossRef]
- Baba, T.; Ono, Y. Heteropolyacids and their salts supported on acidic ion-exchange resin as highly-active solid-acid catalysts. Appl. Catal. 1986, 22, 321–324. [Google Scholar] [CrossRef]
- Izumi, Y.; Urabe, K. Catalysis of heteropoly acids entrapped in activated carbon. Chem. Lett. 1981, 663–666. [Google Scholar] [CrossRef]
- Pizzio, L.R.; Caceres, C.V.; Blanco, M.N. Equilibrium adsorption of 11-tungstophosphate anion on different supports. Appl. Surf. Sci. 1999, 151, 91–101. [Google Scholar] [CrossRef]
- Beck, J.S.; Vartuli, J.C.; Roth, W.J.; Leonowicz, M.E.; Kresge, C.T.; Schmitt, K.D.; Chu, C.T.W.; Olson, D.H.; Sheppard, E.W.; McCullen, S.B.; Higgins, J.B.; Schlenker, J.L. A new family of mesoporous molecular-sieves prepared with liquid-crystal templates. J. Am. Chem. Soc. 1992, 114, 10834–10843. [Google Scholar] [CrossRef]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Zhao, D.Y.; Feng, J.L.; Huo, Q., S.; Nicholas, M.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Nature 1998, 279, 548–552. [Google Scholar]
- Zhao, D.Y.; Huo, Q.S.; Feng, J.L.; Chmelka, B.F.; Stucky, G.D. Nonionic triblock and star diblock copolymer and oligomeric surfactant syntheses of highly ordered, hydrothermally stable, mesoporous silica structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar] [CrossRef]
- Tanev, P.T.; Chibwe, M.; Pinnavaia, T.J. Titanium-containing mesoporous molecular-sieves for catalytic-oxidation of aromatic-compounds. Nature 1994, 368, 321–323. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Z.; Pauly, T.R.; Pinnavaia, T.J. Tailoring the framework and textural mesopores of HMS molecular sieves through an electrically neutral (S0I0) assembly pathway. Chem. Mat. 1997, 9, 2491–2498. [Google Scholar] [CrossRef]
- Zhao, D.Y.; Yang, P.D.; Huo, Q.S.; Chmelka, B.F.; Stucky, G.D. Topological construction of mesoporous materials. Curr. Opin. Solid State Mater. Sci. 1998, 3, 111–121. [Google Scholar] [CrossRef]
- Ren, Y.; Qian, L.P.; Yue, B.; He, H.Y. Synthesis of Ti-containing silica mesoporous molecular sieves with high catalytic activity for epoxidation. Chin. J. Catal. 2003, 24, 947–950. [Google Scholar]
- Liu, S.X.; Yue, B.; Rao, J.; Zhou, Y.; He, H.Y. Template synthesis of one-dimensional nanostructured spinel zinc ferrite. Mater. Lett. 2006, 60, 154–158. [Google Scholar] [CrossRef]
- Kozhevnikov, I.V.; Sinnema, A.; Jansen, R.J.J.; Pamin, K.; Vanbekkum, H. New acid catalyst comprising heteropoly acid on a mesoporous molecular-sieve MCM-41. Catal. Lett. 1995, 30, 241–252. [Google Scholar] [CrossRef]
- Lefebvre, F. P-31 MAS NMR-study of H3PW12O40 supported on silica-formation of (equivalent-to-SiOH2+)(H2PW12O40-). J. Chem. Soc., Chem. Commun. 1992, 756–757. [Google Scholar] [CrossRef]
- Ghanbari-Siahkali, A.; Philippou, A.; Dwyer, J.; Anderson, M.W. The acidity and catalytic activity of heteropoly acid on MCM-41 investigated by MAS NMR, FTIR and catalytic tests. Appl. Catal. A: Gen. 2000, 192, 57–69. [Google Scholar] [CrossRef]
- Kim, W.G.; Kim, M.W.; Kim, J.H.; Seo, G. Dispersion measurement of heteropoly acid supported on KIT-1 mesoporous material. Micropor. Mesopor. Mater. 2003, 57, 113–120. [Google Scholar] [CrossRef]
- Nandhini, K.U.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. t-Butylation of phenol over mesoporous aluminophosphate and heteropolyacid supported aluminophosphate molecular sieves. J. Mol. Catal. A: Chem. 2004, 223, 201–210. [Google Scholar] [CrossRef]
- Nandhini, K.U.; Mabel, J.H.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. The influence of phosphotungstic acid acidity on Al-MCM-41 in t-butylation of phenol with t-butyl alcohol. Micropor. Mesopor. Mater. 2006, 96, 21–28. [Google Scholar] [CrossRef]
- Kumar, G.S.; Vishnuvarthan, M.; Palanichamy, M.; Murugesan, V. SBA-15 supported HPW: Effective catalytic performance in the alkylation of phenol. J. Mol. Catal. A: Chem. 2006, 260, 49–55. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, H.O. Alkylation of 1-dodecene with benzene over H3PW12O40 supported on mesoporous silica SBA-15. Catal. Lett. 2004, 93, 209–212. [Google Scholar] [CrossRef]
- Llanos, A.; Melo, L.; Avendano, F.; Montes, A.; Brito, J.L. Synthesis and characterization of HPW/MCM-41 (Si) and HPW/MCM-41 (Si/Al) catalysts: Activity for toluene alkylation with 1-dodecene. Catal. Today 2008, 133–135, 20–27. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, L.; Xu, B.B.; Li, Z.K.; Jia, L.P.; Guo, W.H. Toluene alkylation with 1-octene over supported heteropoly acids on MCM-41 catalysts. J. Mol. Catal. A: Chem. 2009, 297, 86–92. [Google Scholar] [CrossRef]
- Kamalakar, G.; Komura, K.; Kubota, Y.; Sugi, Y. Friedel-Crafts benzylation of aromatics with benzyl alcohols catalyzed by heteropoly acids supported on mesoporous silica. J. Chem. Technol. Biotechnol. 2006, 81, 981–988. [Google Scholar] [CrossRef]
- Ajaikumar, S.; Pandurangan, A. HPW and supported HPW catalyzed condensation of aromatic aldehydes with aniline: Synthesis of DATPM derivatives. J. Mol. Catal. A: Chem. 2008, 286, 21–30. [Google Scholar] [CrossRef]
- Liu, Q.Y.; Wu, W.L.; Wang, J.; Ren, X.Q.; Wang, Y.R. Characterization of 12-tungstophosphoric acid impregnated on mesoporous silica SBA-15 and its catalytic performance in isopropylation of naphthalene with isopropanol. Micropor. Mesopor. Mater. 2004, 76, 51–60. [Google Scholar] [CrossRef]
- Varisli, D.; Dogu, T.; Dogu, G. Silicotungstic acid impregnated MCM-41-like mesoporous solid acid catalysts for dehydration of ethanol. Ind. Eng. Chem. Res. 2008, 47, 4071–4076. [Google Scholar] [CrossRef]
- Xia, Q.H.; Hidajat, K.; Kawi, S. Structure, acidity, and catalytic activity of mesoporous acid catalysts for the gas-phase synthesis of MTBE from MeOH and ButOH. J. Catal. 2002, 209, 433–444. [Google Scholar] [CrossRef]
- Juan, J.C.; Zhang, J.C.; Yarmo, M.A. 12-Tungstophosphoric acid supported on MCM-41 for esterification of fatty acid under solvent-free condition. J. Mol. Catal. A: Chem. 2007, 267, 265–271. [Google Scholar] [CrossRef]
- Jermy, B.R.; Pandurangan, A. H3PW12O40 supported on MCM-41 molecular sieves: An effective catalyst for acetal formation. Appl. Catal. A: Gen. 2005, 295, 185–192. [Google Scholar] [CrossRef]
- Karthikeyan, G.; Pandurangan, A. Heteropolyacid (H3PW12O40) supported MCM-41: An efficient solid acid catalyst for the green synthesis of xanthenedione derivatives. J. Mol. Catal. A: Chem. 2009, 311, 36–45. [Google Scholar] [CrossRef]
- Maheswari, R.; Shanthi, K.; Sivakumar, T.; Narayanan, S. Beckmann rearrangement over phosphotungstic acid/SiMCM-41 cyclohexanone oxime to epsilon-caprolactam. Appl. Catal. A: Gen. 2003, 248, 291–301. [Google Scholar] [CrossRef]
- Izumi, Y.; Fujita, T. Iminium salt-catalyzed liquid-phase Beckmann rearrangement of cyclohexanone oxime. J. Mol. Catal. A: Chem. 1996, 106, 43–49. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Murata, K.; Inaba, M.; Mimura, N. Selective oxidation of propylene to acetone by molecular oxygen over Mx/2H5-x[PMo10V2O40]/HMS (M = Cu2+, Co2+, Ni2+). Catal. Commun. 2003, 4, 281–285. [Google Scholar] [CrossRef]
- Verhoef, M.J.; Kooyman, P.J.; Peters, J.A.; van Bekkum, H. A study on the stability of MCM-41-supported heteropoly acids under liquid- and gas-phase esterification conditions. Micropor. Mesopor. Mater. 1999, 27, 365–371. [Google Scholar] [CrossRef]
- VanDerVoort, P.; Vansant, E.F. Silylation of the silica surface a review. J. Liq. Chromatogr. Related Technol. 1996, 19, 2723–2752. [Google Scholar] [CrossRef]
- Kaleta, W.; Nowinska, K. Immobilisation of heteropoly anions in Si-MCM-41 channels by means of chemical bonding to aminosilane groups. Chem. Commun. 2001, 535–536. [Google Scholar] [CrossRef]
- Tarlani, A.; Abedini, M.; Nemati, A.; Khabaz, M.; Amini, M.M. Immobilization of Keggin and Preyssler tungsten heteropolyacids on various functionalized silica. J. Colloid Inter. Sci. 2006, 303, 32–38. [Google Scholar] [CrossRef]
- Wu, S.S.; Wang, J.; Zhang, W.H.; Ren, X.Q. Preparation of Keggin and Preyssler heteropolyacid catalysts on amine-modified SBA-15 and their catalytic performances in esterification of n-butanol with acetic acid. Catal. Lett. 2008, 125, 308–314. [Google Scholar] [CrossRef]
- Wu, S.S.; Liu, P.; Leng, Y.; Wang, J. Heteropolyacids anchored on amino-functionalized MCM-41 via condensation as reusable catalysts for esterification. Catal. Lett. 2009, 132, 500–505. [Google Scholar] [CrossRef]
- Dias, A.S.; Pillinger, M.; Valente, A.A. Mesoporous silica-supported 12-tungstophosphoric acid catalysts for the liquid phase dehydration of D-xylose. Micropor. Mesopor. Mater. 2006, 94, 214–225. [Google Scholar] [CrossRef]
- Kim, H.; Jung, J.C.; Kim, P.; Yeom, S.H.; Lee, K.Y.; Song, I.K. Preparation of H3PMo12O40 catalyst immobilized on surface modified mesostructured cellular foam (SM-MCF) silica and its application to the ethanol conversion reaction. J. Mol. Catal. A: Chem. 2006, 259, 150–155. [Google Scholar] [CrossRef]
- Kim, H.; Jung, J.C.; Song, I.K. Chemical immobilization of heteropolyacid catalyst on inorganic mesoporous material for use as an oxidation catalyst. Catal. Surv. Asia 2007, 11, 114–122. [Google Scholar] [CrossRef]
- Chen, L.F.; Zhu, K.; Bi, L.H.; Suchopar, A.; Reicke, M.; Mathys, G.; Jaensch, H.; Kortz, U.; Richards, R.M. Solvent-free aerobic oxidation of n-alkane by iron(III)-substituted polyoxotungstates immobilized on SBA-15. Inorg. Chem. 2007, 46, 8457–8459. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.F.; Hu, J.C.; Mal, S.S.; Kortz, U.; Jaensch, H.; Mathys, G.; Richards, R.M. Heterogeneous wheel-shaped Cu20-polyoxotungstate [Cu20Cl(OH)24(H2O)12(P8W48O184)]25- catalyst for solvent-free aerobic oxidation of n-hexadecane. Chem. Eur. J. 2009, 15, 7490–7497. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.J.S.; Stein, A. Surface modification of mesoporous macroporous and amorphous silica with catalytically active polyoxometalate clusters. Inorg. Chem. 2001, 40, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Bao, R.L.; Yue, B.; Gu, M.; Pei, S.P.; He, H.Y. Synthesis, characterization and catalytic application of SBA-15 immobilized rare earth metal sandwiched polyoxometalates. J. Mol. Catal. A: Chem. 2007, 270, 50–55. [Google Scholar] [CrossRef]
- Zhou, Y.; Yue, B.; Bao, R.L.; Gu, M.; He, H.Y. Surface aminosilylated mesoporous SBA-15 with rare earth metal sandwiched polyoxometalates as heterogeneous catalyst. Stud. Surf. Sci. Catal. 2007, 165, 479–482. [Google Scholar]
- Zhou, Y.; Yue, B.; Bao, R.L.; Liu, S.X.; He, H.Y. Catalytic aerobic oxidation of acetaldehyde over Keggin-type molybdovanadophosphoric acid/SBA-15 under ambient condition. Chin. J. Chem. 2006, 24, 1001–1005. [Google Scholar] [CrossRef]
- Raj, N.K.K.; Deshpande, S.S.; Ingle, R.H.; Raja, T.; Manikandan, P. Heterogenized molybdovanadophosphoric acid on amine-functionalized SBA-15 for selective oxidation of alkenes. Catal. Lett. 2004, 98, 217–223. [Google Scholar] [CrossRef]
- Bordoloi, A.; Lefebvre, E.; Halligudi, S.B. Selective oxidation of anthracene using inorganic-organic hybrid materials based on molybdovanadophosphoric acids. J. Catal. 2007, 247, 166–175. [Google Scholar] [CrossRef]
- Maksimchuk, N.V.; Meigunov, M.S.; Chesalov, Y.A.; Mrowlec-Bialon, J.; Jarzebski, A.B.; Kholdeeva, O.A. Aerobic oxidations of alpha-pinene over cobalt-substituted polyoxometalate supported on amino-modified mesoporous silicates. J. Catal. 2007, 246, 241–248. [Google Scholar] [CrossRef]
- Li, L.; Liu, C.M.; Geng, A.F.; Jiang, C.J.; Guo, Y.H.; Hu, C.W. Preparation, characterization and photocatalytic applications of amine-functionalized mesoporous silica impregnated with transition-metal-monosubstituted polyoxometalates. Mater. Res. Bull. 2006, 41, 319–332. [Google Scholar] [CrossRef]
- Guo, Y.H.; Hu, C.W.; Jiang, C.J.; Yang, Y.; Jiang, S.C.; Li, X.L.; Wang, E.B. Preparation and heterogeneous photocatalytic behaviors of the surface-modified porous silica materials impregnated with monosubstituted keggin units. J. Catal. 2003, 217, 141–151. [Google Scholar]
- Molnar, A.; Keresszegi, C.; Torok, B. Heteropoly acids immobilized into a silica matrix: Characterization and catalytic applications. Appl. Catal. A: Gen. 1999, 189, 217–224. [Google Scholar] [CrossRef]
- Yang, L.; Qi, Y.T.; Yuan, X.D.; Shen, H.; Kim, J. Direct synthesis, characterization and catalytic application of SBA-15 containing heteropolyacid H3PW12O40. J. Mol. Catal. A: Chem. 2005, 229, 199–205. [Google Scholar] [CrossRef]
- He, N.Y.; Woo, C.S.; Kim, H.G.; Lee, H.I. Catalytic formation of acetic anhydride over tungstophosphoric acid modified SBA-15 mesoporous materials. Appl. Catal. A: Gen. 2005, 281, 167–178. [Google Scholar] [CrossRef]
- Shi, C.F.; Wang, R.W.; Zhu, G.S.; Qiu, S.L.; Long, J. Synthesis, characterization, and catalytic properties of SiPW-X mesoporous silica with heteropolyacid encapsulated into their framework. Eur. J. Inorg. Chem. 2005, 4801–4807. [Google Scholar] [CrossRef]
- Shi, C.F.; Wang, R.W.; Zhu, G.S.; Qiu, S.L.; Long, J. In situ synthesis, characterization of SiPMo-X, and different catalytic properties of SiPMo-X and SiPW-X. Eur. J. Inorg. Chem. 2006, 3054–3060. [Google Scholar] [CrossRef]
- Wang, J.M.; Yan, L.; Qian, G.; Yang, K.L.; Liu, H.T.; Wang, X.L. Heteropolyacid encapsulated into mesoporous silica framework for an efficient preparation of 1,1-diacetates from aldehydes under a solvent-free condition. Tetrahedron Lett. 2006, 47, 8309–8312. [Google Scholar] [CrossRef]
- Caetano, C.S.; Fonseca, I.M.; Ramos, A.M.; Vital, J.; Castanheiro, J.E. Esterification of free fatty acids with methanol using heteropolyacids immobilized on silica. Catal. Commun. 2008, 9, 1996–1999. [Google Scholar] [CrossRef]
- Dufaud, V.; Lefebvre, F.; Niccolai, G.P.; Aouine, M. New insights into the encapsulation and stabilization of heteropolyacids inside the pore walls of mesostructured silica materials. J. Mater. Chem. 2009, 19, 1142–1150. [Google Scholar] [CrossRef]
- Pei, S.P.; Yue, B.; Qian, L.P.; Yan, S.R.; Cheng, J.F.; Zhou, Y.; Xie, S.H.; He, H.Y. Preparation and characterization of P-Mo-V mixed oxide-incorporating mesoporous silica catalysts for selective oxidation of methane to formaldehyde. Appl. Catal. A: Gen. 2007, 329, 148–155. [Google Scholar] [CrossRef]
- Yu, X.D.; Xu, L.L.; Yang, X.; Guo, Y.N.; Li, K.X.; Hu, J.L.; Li, W.; Ma, F.Y.; Guo, Y.H. Preparation of periodic mesoporous silica-included divacant Keggin units for the catalytic oxidation of styrene to synthesize styrene oxide. Appl. Surf. Sci. 2008, 254, 4444–4451. [Google Scholar] [CrossRef]
- Guo, H.Q.; Li, D.B.; Xiao, H.C.; Zhang, J.L.; Li, W.H.; Sun, Y.H. Methanol selective oxidation to dimethoxymethane on H3PMo12O40/SBA-15 supported catalysts. Korean J. Chem. Eng. 2009, 26, 902–906. [Google Scholar] [CrossRef]
- Jing, G.H.; Li, J.H.; Yang, D.; Hao, J.M. Promotional mechanism of tungstation on selective catalytic reduction of NOx by methane over In/WO3/ZrO2. Appl. Catal. B: Environ. 2009, 91, 123–134. [Google Scholar] [CrossRef]
- Reddy, B.M.; Lee, S.C.; Han, D.S.; Park, S.E. Utilization of carbon dioxide as soft oxidant for oxydehydrogenation of ethylbenzene to styrene over V2O5-CeO2/TiO2-ZrO2 catalyst. Appl. Catal. B: Environ. 2009, 87, 230–238. [Google Scholar] [CrossRef]
- Umbarkar, S.B.; Kotbagi, T.V.; Biradar, A.V.; Pasricha, R.; Chanale, J.; Dongare, M.K.; Mamede, A.S.; Lancelot, C.; Payen, E. Acetalization of glycerol using mesoporous MoO3/SiO2 solid acid catalyst. J. Mol. Catal. A: Chem. 2009, 310, 150–158. [Google Scholar] [CrossRef]
- Ryoo, R.; Joo, S.H.; Jun, S. Synthesis of highly ordered carbon molecular sieves via template-mediated structural transformation. J. Phys. Chem. B 1999, 103, 7743–7746. [Google Scholar] [CrossRef]
- Liu, X.Y.; Tian, B.Z.; Yu, C.Z.; Gao, F.; Xie, S.H.; Tu, B.; Che, R.C.; Peng, L.M.; Zhao, D.Y. Room-temperature synthesis in acidic media of large-pore three-dimensional bicontinuous mesoporous silica with Ia3d symmetry. Angew. Chem. Int. Ed. 2002, 41, 3876–3878. [Google Scholar] [CrossRef]
- Kleitz, F.; Choi, S.H.; Ryoo, R. Cubic Ia3d large mesoporous silica: Synthesis and replication to platinum nanowires, carbon nanorods and carbon nanotubes. Chem. Commun. 2003, 2136–2137. [Google Scholar] [CrossRef]
- Yang, C.M.; Sheu, H.S.; Chao, K.J. Templated synthesis and structural study of densely packed metal nanostructures in MCM-41 and MCM-48. Adv. Funct. Mater. 2002, 12, 143–148. [Google Scholar] [CrossRef]
- Adhyapak, P.V.; Karandikar, P.; Vijayamohanan, K.; Athawale, A.A.; Chandwadkar, A.J. Synthesis of silver nanowires inside mesoporous MCM-41 host. Mater. Lett. 2004, 58, 1168–1171. [Google Scholar] [CrossRef]
- Huang, M.H.; Choudrey, A.; Yang, P.D. Ag nanowire formation within mesoporous silica. Chem. Commun. 2000, 1063–1064. [Google Scholar] [CrossRef]
- Wang, D.H.; Kou, R.; Gil, M.P.; Jakobson, H.P.; Tang, J.; Yu, D.H.; Lu, Y.F. Templated synthesis, characterization, and sensing application of macroscopic platinum nanowire network electrodes. J. Nanosci. Nanotech. 2005, 5, 1904–1909. [Google Scholar] [CrossRef]
- Tian, B.Z.; Liu, X.Y.; Yang, H.F.; Xie, S.H.; Yu, C.Z.; Tu, B.; Zhao, D.Y. General synthesis of ordered crystallized metal oxide nanoarrays replicated by microwave-digested mesoporous silica. Adv. Mater. 2003, 15, 1370–1374. [Google Scholar] [CrossRef]
- Zhu, K.K.; Bin, Y.; Xie, S.H.; Zhang, S.Y.; Biao, Z.; Jin, S.L.; He, H.Y. Preparation and characterization of divanadium pentoxide nanowires inside SBA-15 channels. Chin. J. Chem. 2004, 22, 33–37. [Google Scholar] [CrossRef]
- Yue, B.; Tan, D.J.; Yan, S.R.; Zhou, Y.; Zhu, K.K.; Pan, J.F.; Zhuang, J.H.; He, H.Y. Preparation of MoO3-V2O5 nanowires with controllable Mo/V ratios inside SBA-15 channels using a chemical approach with heteropoly acid. Chin. J. Chem. 2005, 23, 32–36. [Google Scholar] [CrossRef]
- Zhu, K.K.; He, H.Y.; Xie, S.H.; Zhang, X.; Zhou, W.Z.; Jin, S.L.; Yue, B. Crystalline WO3 nanowires synthesized by templating method. Chem. Phys. Lett. 2003, 377, 317–321. [Google Scholar] [CrossRef]
- Filowitz, M.; Ho, R.K.C.; Klemperer, W.G.; Shum, W. 17O nuclear magnetic resonance spectroscopy of polyoxometalates .1. sensitivity and resolution. Inorg. Chem. 1979, 18, 93–103. [Google Scholar] [CrossRef]
- Zhu, K.K.; Yue, B.; Zhou, W.Z.; He, H.Y. Preparation of three-dimensional chromium oxide porous single crystals templated by SBA-15. Chem. Commun. 2003, 98–99. [Google Scholar] [CrossRef]
- Shin, H.J.; Ryoo, R.; Kruk, M.; Jaroniec, M. Modification of SBA-15 pore connectivity by high-temperature calcination investigated by carbon inverse replication. Chem. Commun. 2001, 349–350. [Google Scholar] [CrossRef]
- Fan, J.; Yu, C.Z.; Wang, L.M.; Tu, B.; Zhao, D.Y.; Sakamoto, Y.; Terasaki, O. Mesotunnels on the silica wall of ordered SBA-15 to generate three-dimensional large-pore mesoporous networks. J. Am. Chem. Soc. 2001, 123, 12113–12114. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.; Tang, H.L.; Kong, Z.P.; Zhu, K.; Dickinson, C.; Zhou, W.Z.; He, H.Y. Preparation and characterization of three-dimensional mesoporous crystals of tungsten oxide. Chem. Phys. Lett. 2005, 407, 83–86. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ren, Y.; Yue, B.; Gu, M.; He, H. Progress of the Application of Mesoporous Silica-Supported Heteropolyacids in Heterogeneous Catalysis and Preparation of Nanostructured Metal Oxides. Materials 2010, 3, 764-785. https://doi.org/10.3390/ma3020764
Ren Y, Yue B, Gu M, He H. Progress of the Application of Mesoporous Silica-Supported Heteropolyacids in Heterogeneous Catalysis and Preparation of Nanostructured Metal Oxides. Materials. 2010; 3(2):764-785. https://doi.org/10.3390/ma3020764
Chicago/Turabian StyleRen, Yuanhang, Bin Yue, Min Gu, and Heyong He. 2010. "Progress of the Application of Mesoporous Silica-Supported Heteropolyacids in Heterogeneous Catalysis and Preparation of Nanostructured Metal Oxides" Materials 3, no. 2: 764-785. https://doi.org/10.3390/ma3020764
APA StyleRen, Y., Yue, B., Gu, M., & He, H. (2010). Progress of the Application of Mesoporous Silica-Supported Heteropolyacids in Heterogeneous Catalysis and Preparation of Nanostructured Metal Oxides. Materials, 3(2), 764-785. https://doi.org/10.3390/ma3020764