Ceramics for Dental Applications: A Review
Abstract
:1. Introduction
| Fabrication technique | Crystalline phase | |
|---|---|---|
| Metal-ceramics | Sintering | Leucite |
| Heat-pressing on metal | Leucite, leucite & fluorapatite | |
| All-ceramics | Sintering | Leucite |
| Heat-pressing | Leucite, lithium disilicate | |
| Dry pressing and sintering | Alumina | |
| Slip-casting & glass infiltration | Alumina, spinel, alumina-zirconia (12Ce-TZP) | |
| Soft machining & glass-infiltration | Alumina, alumina-zirconia (12Ce-TZP) | |
| Soft machining & sintering | Alumina, zirconia (3Y-TZP) | |
| Soft machining, sintering & heat-pressing | Zirconia/fluorapatite-leucite glass-ceramic | |
| Hard machining | Sanidine, leucite | |
| Hard machining & heat treatment | Lithium disilicate |
2. Metal-Ceramics


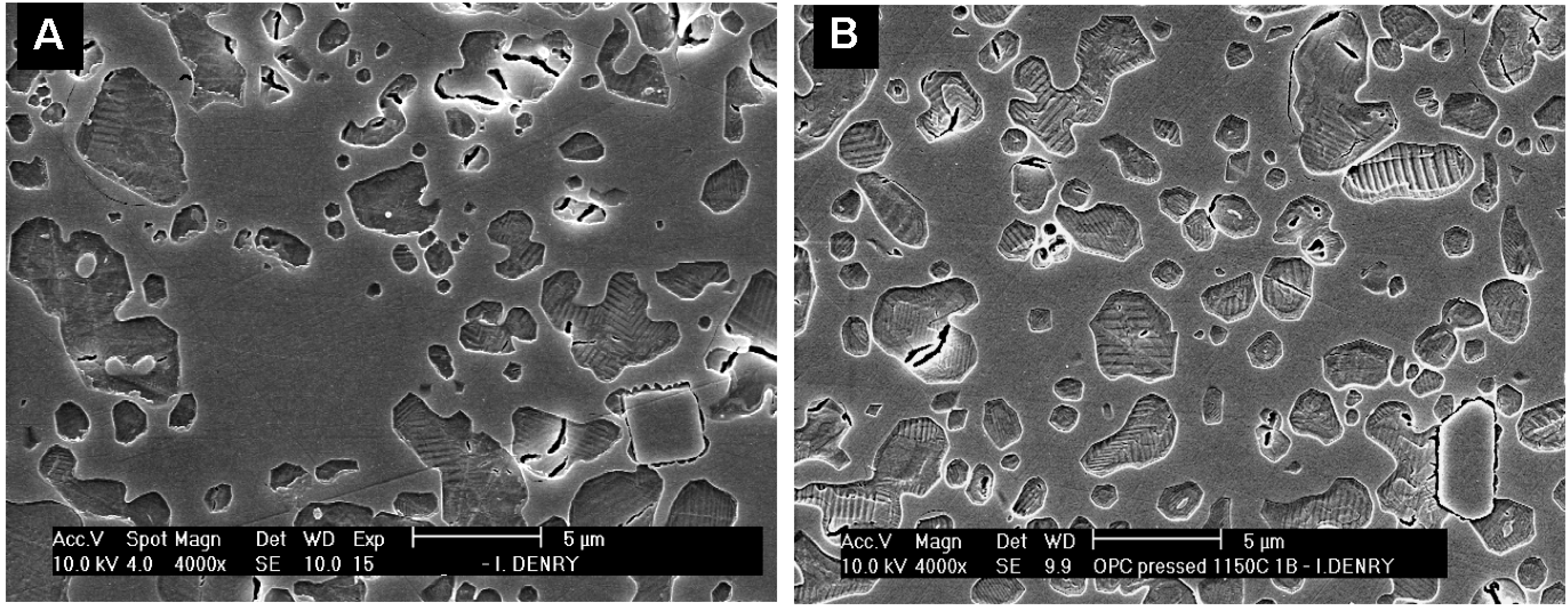
3. All-Ceramic Systems
3.1. Heat-pressed ceramics


3.2. Dry-pressed and sintered ceramics
3.3. Slip-cast ceramics


3.4. Machined ceramics
Hard machining

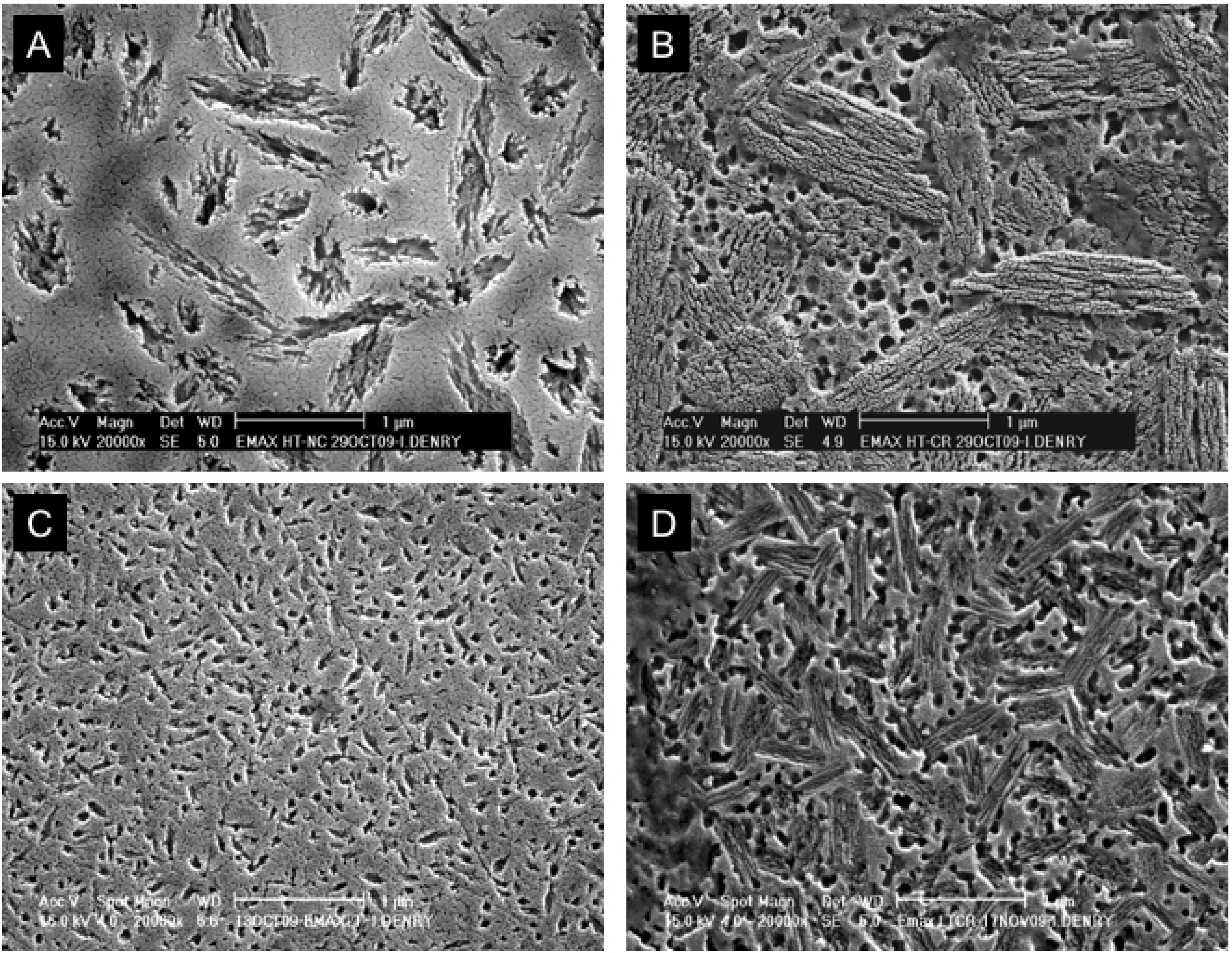
Soft machining

3.5. Manufactured zirconia dental abutments and implants
4. Conclusions
| Processing Method | Crystalline phase | Crystallinity (%) | Strength (MPa) | Brand | Manufacturer |
|---|---|---|---|---|---|
| Sintered on metal substructure | Leucite | 15–25 | 61 ± 5 [16] | Ceramco®3 | Dentsply |
| Heat-pressed | Leucite | ≈ 35 | 106 ± 17 [27] | IPS Empress® | Ivoclar |
| Heat-pressed | Lithium disilicate | 65 | 306 ± 29 [27] | IPS Empress® Eris | Ivoclar |
| Dry-pressed and sintered | Alumina | Highly crystalline | 607 ± 73 [40] | Procera® | Nobel Biocare |
| Slip-cast & glass-infiltrated or soft machined and glass-infiltrated | Alumina | 67–68 | 594 ± 52 [27] | In-Ceram® Alumina | Vident |
| Slip-cast & glass-infiltrated or soft machined and glass-infiltrated | Spinel | 65–68 | 378 ± 65 [26] | In-Ceram® Spinell | Vident |
| Slip-cast & glass-infiltrated or soft machined and glass-infiltrated | 12 Ce-TZP-alumina | 67 | 630 ± 58 [47] | In-Ceram® Zirconia | Vident |
| Soft-machined & sintered | 3Y-TZP | Highly crystalline | 1087 ± 173 [94] | Cercon® | Dentsply |
| Soft-machined & sintered | Alumina | Highly crystalline | 700* | In-Ceram® AL | Vident |
| Hard-machined | Sanidine | ≈ 30 | 122 ± 13 [26] | Vitablocs® Mark II | Vident |
| Hard-machined | Leucite | ≈ 35 | 106 ± 17 [27] | IPS Empress® CAD | Ivoclar |
| Hard-machined & crystallized | Lithium disilicate | 65 | 262 ± 88 [55] | IPS e.max CAD | Ivoclar |
References and Notes
- Weinstein, M.; Weinstein, L.K.; Katz, S.; Weinstein, A.B. Fused porcelain-to-metal teeth. US Patent 3,052,982, 11 September 1962. [Google Scholar]
- Vergano, P.J.; Hill, D.C.; Uhlmann, D.R. Thermal expansion of feldspar glasses. J. Am. Ceram. Soc. 1967, 50, 59–60. [Google Scholar] [CrossRef]
- Wyart, J. Structure atomique de la leucite. C. R. Acad. Sci., Paris 1941, 282, 356–358. [Google Scholar]
- Wyckoff, R.G.W. Crystal Structures, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1963; Volume 4, pp. 400–401. [Google Scholar]
- Taylor, D.; Henderson, C.M.B. The thermal expansion of the leucite group of minerals. Am. Mineral. 1968, 53, 1476–1489. [Google Scholar]
- Mackert, J.R., Jr.; Butts, M.B.; Morena, R.; Fairhurst, C.W. Phase Changes In a Leucite-Containing Dental Porcelain Frit. J. Am. Ceram. Soc. 1986, 69, C69–C72. [Google Scholar] [CrossRef]
- Mackert, J.R., Jr.; Butts, M.B.; Fairhurst, C.W. The effect of the leucite transformation on dental porcelain expansion. Dent. Mater. 1986, 2, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Schairer, J.F.; Bowen, N.L. The system K2O-Al2O3-SiO2. Am. J. Sci. 1955, 253, 681–746. [Google Scholar] [CrossRef]
- Palmer, D.C.; Putnis, A.; Salje, E.K.H. Twinning in tetragonal leucite. Phys. Chem. Miner. 1988, 16, 298–303. [Google Scholar] [CrossRef]
- Mackert, J.R., Jr. Effect of thermally induced changes on porcelain-metal compatibility. In Perspectives in Dental Ceramics, Proceedings of the Fourth International Symposium on Ceramics; Preston, J.D., Ed.; Quintessence Publishing Co., Inc.: Chicago, IL, USA, 1988; pp. 53–64. [Google Scholar]
- Meyer, J.M.; O'Brien, W.J.; Cu, Y. Sintering of dental porcelain enamels. J. Dent. Res. 1976, 55, 696–699. [Google Scholar] [CrossRef] [PubMed]
- Claus, H.; Rauter, H. The structure and microstructure of dental porcelain in relationship to the firing conditions. Int. J. Prosthodont. 1989, 2, 376–384. [Google Scholar] [PubMed]
- Mackert, J.R., Jr.; Rueggeberg, F.A.; Lockwood, P.E.; Evans, A.L.; Thompson, W.O. Isothermal anneal effect on microcrack density around leucite particles in dental porcelains. J. Dent. Res. 1994, 73, 1221–1227. [Google Scholar] [PubMed]
- Mackert, J.R., Jr.; Evans, A.L. Effect of cooling rate on leucite volume fraction in dental porcelains. J. Dent. Res. 1991, 70, 137. [Google Scholar] [CrossRef] [PubMed]
- Mackert, J.R., Jr.; Evans, A.L. Quantitative X-ray diffraction determination of leucite thermal instability in dental porcelain. J. Am. Ceram. Soc. 1991, 74, 450–453. [Google Scholar] [CrossRef]
- Seghi, R.R.; Daher, T.; Caputo, A. Relative flexural strength of dental restorative ceramics. Dent. Mater. 1990, 6, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Denry, I.L. All-ceramic restorations. In Contemporary Fixed Prosthodontics; Rosenstiel, S.F., Ed.; Mosby Elsevier: St Louis, MO, USA, 2006; p. 777. [Google Scholar]
- Holand, W.; Schweiger, M.; Rheinberger, V.M.; Kappert, H. Bioceramics and their application for dental restoration. Adv. Appl. Ceram. 2009, 108, 373–380. [Google Scholar] [CrossRef]
- Spear, F.; Holloway, J.A. Which all-ceramic system is optimal for anterior esthetics. J. Am. Dent. Assoc. 2008, 139, 19S–24S. [Google Scholar] [CrossRef] [PubMed]
- Michalske, T.; Freiman, S. A molecular interpretation of stress corrosion in silica. Nature 1982, 295, 511–512. [Google Scholar] [CrossRef]
- Kobayashi, K.; Kuwajima, H.; Masaki, T. Phase change and mechanical properties of ZrO2-Y2O3 solid electrolyte after aging. Solid State Ionics 1981, 3/4, 489–495. [Google Scholar] [CrossRef]
- Chevalier, J.; Calès, B.; Drouin, J.M. Low-temperature aging of Y-TZP ceramics. J. Am. Ceram. Soc. 1999, 82, 2150–2154. [Google Scholar] [CrossRef]
- Lawson, S. Environmental degradation of zirconia ceramics. J. Eur. Ceram. Soc. 1995, 15, 485–502. [Google Scholar] [CrossRef]
- Kelly, J.R. Clinically relevant approach to failure testing of all-ceramic restorations. J. Prosthet. Dent. 1999, 81, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Denry, I.L.; Rosenstiel, S.F. Phase transformations in feldspathic dental porcelains. In Bioceramics: Materials and Applications; Fischman, G., Clare, A., Hench, L., Eds.; The American Ceramic Society: Westerville, OH, USA, 1995; pp. 149–156. [Google Scholar]
- Seghi, R.; Sorensen, J. Relative flexural strength of six new ceramic materials. Int. J. Prosthodont. 1995, 8, 239–246. [Google Scholar] [PubMed]
- Guazzato, M.; Albakry, M.; Ringer, S.P.; Swain, M.V. Strength, fracture toughness and microstructure of a selection of all-ceramic materials. Part I. Pressable and alumina glass-infiltrated ceramics. Dent. Mater. 2004, 20, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Denry, I.L.; Mackert, J.R.J.; Holloway, J.A.; Rosenstiel, S.F. Effect of cubic leucite stabilization on the flexural strength of feldspathic dental porcelain. J. Dent. Res. 1996, 75, 1928–1935. [Google Scholar] [CrossRef] [PubMed]
- Mackert, J.R., Jr.; Williams, A.L. Microcracks in dental porcelains and their behavior during multiple firing. J. Dent. Res. 1996, 75, 1484–1490. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.K.; Luthy, H.; Wohlwend, A. Heat-pressed ceramics: Technology and strength. Int. J. Prosthodont. 1992, 5, 9–16. [Google Scholar]
- Denry, I.L.; Holloway, J.A.; Colijn, H.O. Phase Transformations in a leucite-reinforced pressable dental ceramic. J. Biomed. Mater. Res. 2001, 54, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Borom, M.P.; Turkalo, A.M.; Doremus, R.H. Strength and microstructure in lithium disilicate glass-ceramics. J. Am. Ceram. Soc. 1975, 58, 385–391. [Google Scholar] [CrossRef]
- Ray, C.S.; Day, D.E. Identifying internal and surface crystallization by differential thermal analysis for the glass-to-crystal transformations. Thermochim. Acta 1996, 280, 163–174. [Google Scholar] [CrossRef]
- Ray, C.S.; Day, D.E.; Huang, W.; Narayan, K.L.; Cull, T.S.; Kelton, K.F. Non-isothermal calorimetric studies of the crystallization of lithium disilicate glass. J. Non-Cryst. Solids 1996, 204, 1–12. [Google Scholar] [CrossRef]
- Iqbal, Y.; Lee, W.E.; Holland, D.; James, P.F. Metastable phase formation in the early stage crystallization of lithium disilicate glass. J. Non-Cryst. Solids 1998, 224, 1–16. [Google Scholar] [CrossRef]
- Iqbal, Y.; Lee, W.E.; Holland, D.; James, P.F. Crystal nucleation in P2O5-doped lithium disilicate glasses. J. Mater. Sci. 1999, 34, 4399–4411. [Google Scholar] [CrossRef]
- Höland, W.; Apel, E.; van't Hoen, C.; Rheinberger, V. Studies of crystal phase formations in high-strength lithium disilicate glass-ceramics. J. Non-Cryst. Solids 2006, 352, 4041–4050. [Google Scholar] [CrossRef]
- Albakry, M.; Guazzato, M.; Swain, M.V. Influence of hot pressing on the microstructure and fracture toughness of two pressable dental glass–ceramics. J. Biomed. Mater. Res. 2004, 71B, 99–107. [Google Scholar] [CrossRef]
- Thiel, S.; Schnapp, J.D. Anisotropic crack extension in aligned glass ceramic. J. Non-Cryst. Solids 1998, 242, 189–194. [Google Scholar] [CrossRef]
- Andersson, M.; Oden, A. A new all-ceramic crown. A dense-sintered, high-purity alumina coping with porcelain. Acta Odontol. Scand. 1993, 51, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.; Oden, A.; Rowcliffe, D. Flexure tests on dental ceramics. Int. J. Prosthodont. 1996, 9, 434–439. [Google Scholar] [PubMed]
- Hegenbarth, E.A. Procera aluminum oxide ceramics: A new way to achieve stability, precision, and esthetics in all-ceramic restorations. Quintessence Dent. Technol. 1996, 19, 21–34. [Google Scholar]
- Haag, P.; Andersson, M.; von Steyern, V.P.; Odén, A. 15 Years of clinical experience with Procera® Alumina. Appl. Osseoint Res. 2004, 4, 7–12. [Google Scholar]
- Heffernan, M.J.; Aquilino, S.A.; Diaz-Arnold, A.M.; Haselton, D.R.; Stanford, C.M.; Vargas, M.A. Relative translucency of six all-ceramic systems. Part I: Core materials. J. Prosthet. Dent. 2002, 88, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Magne, P.; Belser, U. Esthetic improvements and in vitro testing of In-Ceram Alumina and Spinell ceramic. Int. J. Prosthodont. 1997, 459–466. [Google Scholar]
- Jung, Y.-G.; IM, P.; A, P.; BR., L. Contact damage resistance and strength degradation of glass-infiltrated alumina and spinell ceramics. J. Dent. Res. 1999, 78, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Guazzato, M.; Albakry, M.; Ringer, S.P.; Swain, M.V. Strength, fracture toughness and microstructure of a selection of all-ceramic materials. Part II. Zirconia-based dental ceramics. Dent. Mater. 2004, 20, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Guazzato, M.; Albakry, M.; Swain, M.V.; Ringer, S.P. Microstructure of alumina- and alumina/zirconia-glass infiltrated dental ceramics. In Bioceramics 15; Ben-Nissan, B., Sher, D., Walsh, W., Eds.; Trans Tech Publications: Zurich, Switzerland, 2003; Volume 240–242, pp. 879–882. [Google Scholar]
- Guazzato, M.; Albakry, M.; Swain, M.V.; Ironside, J. Mechanical properties of in-ceram alumina and in-ceram zirconia. Int. J. Prosthodont. 2002, 15, 339–346. [Google Scholar] [PubMed]
- Duret, F.; Blouin, J.L.; Duret, B. CAD-CAM in dentistry. J. Am. Dent. Assoc. 1988, 117, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Grossman, D.G. Machinable glass-ceramic based on tetrasilicic mica. J. Am. Ceram. Soc. 1972, 55, 446–449. [Google Scholar] [CrossRef]
- Denry, I.L.; Abushaban, R.F.; Holloway, J.A. Microstructure, crack patterns and flexural strength of two machinable dental ceramics. J. Dent. Res. 1999, 78, 473. [Google Scholar]
- Tinschert, J.; Zwez, D.; Marx, R.; Anusavice, K.J. Structural reliability of alumina-, feldspar-, leucite-, mica- and zirconia-based ceramics. J. Dent. 2000, 28, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Tinschert, J.; Natt, G.; Hassenpflug, S.; Spiekermann, H. Status of current CAD/CAM technology in dental medicine. Int. J. Comput. Dent. 2004, 7, 25–45. [Google Scholar] [PubMed]
- Lupu, M.; Giordano, R.A. Flexural strength of CAD/CAM ceramic framework materials. J. Dent. Res. 2007, 88, 224. [Google Scholar]
- Filser, F.T. Direct ceramic machining of dental restorations. Ph.D. Thesis, Swiss Federal Institute of Technology, Zurich, Switzerland, 2001. [Google Scholar]
- Filser, F.; Kocher, P.; Gauckler, L.J. Net-shaping of ceramic components by direct ceramic machining. Assembly Autom. 2003, 23, 382–390. [Google Scholar] [CrossRef]
- Denry, I.; Kelly, J.R. State of the art of zirconia for dental applications. Dent. Mater. 2008, 24, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, E.C. Zirconia-an overview. In Science and Technology of Zirconia; Heuer, A.H., Hobbs, L.W., Eds.; The American Ceramic Society: Columbus, OH, USA, 1981; Volume 3, pp. 1–24. [Google Scholar]
- Piconi, C.; Macauro, G. Zirconia as a ceramic biomaterial. Biomaterials 1999, 20, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Hannink, R.H.J.; Kelly, P.M.; Muddle, B.C. Transformation toughening in zirconia-containing ceramics. J. Am. Ceram. Soc. 2000, 83, 461–487. [Google Scholar] [CrossRef]
- Garvie, R.C.; Hannink, R.H.; Pascoe, R.T. Ceramic steel? Nature 1975, 258, 703–704. [Google Scholar] [CrossRef]
- Porter, D.l.; Heuer, A.H. Mechanisms of toughening partially stabilized zirconia (PSZ). J. Am. Ceram. Soc. 1977, 60, 183–184. [Google Scholar] [CrossRef]
- Green, D.; Hannink, R.; Swain, M. Transformation Toughening of Ceramics; CRC Press: Boca Raton, FL, USA, 1989; pp. 137–144. [Google Scholar]
- Ruiz, L.; Readey, M.J. Effect of heat-treatment on grain size, phase assemblage, and mechanical properties of 3 mol% Y-TZP. J. Am. Ceram. Soc. 1996, 79, 2331–2340. [Google Scholar] [CrossRef]
- Heuer, A.H.; Claussen, N.; Kriven, W.M.; Ruhle, M. Stability of tetragonal ZrO2 particles in ceramic matrices. J. Am. Ceram. Soc. 1982, 65, 642–650. [Google Scholar] [CrossRef]
- Heuer, A.H.; Lange, F.F.; Swain, M.V.; Evans, A.G. Transformation toughening: An overview. J. Am. Ceram. Soc. 1986, 69, i–iv. [Google Scholar] [CrossRef]
- Von Steyern, P.V. All-ceramic fixed partial dentures. Studies on aluminum oxide- and zirconium dioxide-based ceramic systems. Swed. Dent. J. Suppl. 2005, 173, 1–69. [Google Scholar] [PubMed]
- von Steyern, P.V.; Carlson, P.; Nilner, K. All-ceramic fixed partial dentures designed according to the DC-Zircon® technique. A 2-year clinical study. J. Oral Rehabil. 2005, 32, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Larsson, C.; von Steyern, P.V.; Sunzel, B.; Nilner, K. All-ceramic two- and five-unit implant-supported reconstructions. A randomized, prospective clinical trial. Swed. Dent. J. 2006, 30, 45–53. [Google Scholar] [PubMed]
- Raigrodski, A.J.; Chiche, G.J.; Potiket, N.; Hochstedler, J.L.; Mohamed, S.E.; Billiot, S.; Mercante, D.E. The efficacy of posterior three-unit zirconium-oxide-based ceramic fixed partial dental prostheses: A prospective clinical pilot study. J. Prosthet. Dent. 2006, 96, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Coelho, P.G.; Bonfante, E.A.; Silva, N.R.F.; Rekow, E.D.; Thompson, V.P. Laboratory Simulation of Y-TZP All-ceramic Crown Clinical Failures. J. Dent. Res. 2009, 88, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Tholey, M.J.; Swain, M.V.; Thiel, N. SEM observations of porcelain Y-TZP interface. Dent. Mater. 2009, 25, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, J. What future for zirconia as a biomaterial? Biomaterials 2006, 27, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.R.; Denry, I. Stabilized zirconia as a structural material. Dent. Mater. 2008, 24, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, J.; Grémillard, L.; Virkar, A.V.; Clarke, D.R. The Tetragonal-monoclinic transformation in zirconia: Lessons learned and future trends. J. Am. Ceram. Soc. 2009, 92, 1901–1920. [Google Scholar] [CrossRef]
- Sailer, I.; Feher, A.; Filser, F.; Gauckler, L.J.; Luthy, H.; Hammerle, C.H.F. Five-year clinical results of zirconia frameworks for posterior fixed partial dentures. Int. J. Prosthodont. 2007, 20, 383–388. [Google Scholar] [PubMed]
- Sailer, I.; Feher, A.; Filser, F.; Luthy, H.; Gauckler, L.J.; Scharer, P.; Hammerie, C.H.F. Prospective clinical study of zirconia posterior fixed partial dentures: 3-year follow-up. Quintessence Int. 2006, 37, 685–693. [Google Scholar] [PubMed]
- Sailer, I.; Pjetursson, B.E.; Zwahlen, M.; Hammerle, C.H.F. A systematic review of the survival and complication rates of all-ceramic and metal-ceramic reconstructions after an observation period of at least 3 years. Part II: Fixed dental prostheses. Clin. Oral Implan. Res. 2007, 18, 86–96. [Google Scholar] [CrossRef]
- Andreiotelli, M.; Wenz, H.J.; Kohal, R.J. Are ceramic implants a viable alternative to titanium implants? A systematic literature review. Clin. Oral Implan. Res. 2009, 20, 32–47. [Google Scholar] [CrossRef]
- Calès, B.; Stefani, Y.; Lilley, E. Long-term in vivo and in vitro aging of a zirconia ceramic used in orthopaedy. J. Biomed. Mater. Res. 1994, 28, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Deville, S.; Grémillard, L.; Chevalier, J.; Fantozzi, G. A critical comparison of methods for the determination of the aging sensitivity in biomedical grade yttria-stabilized zirconia. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 72B, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Drummond, J.L. In vitro aging of yttria-stabilized zirconia. J. Am. Ceram. Soc. 1989, 72, 675–676. [Google Scholar] [CrossRef]
- Fernandez-Fairen, M.; Blanco, A.; Murcia, A.; Sevilla, P.; Gil, F.J. Aging of retrieved zirconia femoral heads. Clin. Orthop. Related Res. 2007, 122–129. [Google Scholar] [CrossRef]
- Guo, X. On the degradation of zirconia ceramics during low-temperature annealing in water or water vapor. J. Phys. Chem. Solids 1999, 60, 539–546. [Google Scholar] [CrossRef]
- Denry, I.L.; Holloway, J.A. Microstructural and crystallographic surface changes after grinding zirconia-based dental ceramics. J. Biomed. Mater. Res. B-Appl. Biomater. 2006, 76B, 440–448. [Google Scholar] [CrossRef] [PubMed]
- ISO-13356 International Standard - Implants for surgery - Ceramic materials based on yttria-stabilized tetragonal zirconia (Y-TZP). ISO: Geneva, Switzerland, 2008; pp. 1–13.
- Deville, S.; Chevalier, J.; Grémillard, L. Influence of surface finish and residual stresses on the ageing sensitivity of biomedical grade zirconia. Biomaterials 2006, 27, 2186–2192. [Google Scholar] [CrossRef] [PubMed]
- Deville, S.; Chevalier, J.; Fantozzi, G.; Bartolome, J.F.; Requena, J.; Moya, J.S.; Torrecillas, R.; Diaz, L.A. Development of advanced zirconia-toughened alumina nanocomposites for orthopaedic applications. In Euro Ceramics Viii, Pts 1–3; Trans Tech Publications: Zurich, Switzerland, 2004; Volume 264–268, pp. 2013–2016. [Google Scholar]
- Nawa, M.; Bamba, N.; Sekino, T.; Niihara, K. The effect of TiO2 addition on strengthening and toughening in intragranular type of 12Ce-TZP/Al2O3 nanocomposites. J. Eur. Ceram. Soc. 1998, 18, 209–219. [Google Scholar] [CrossRef]
- Nawa, M.; Nakamoto, S.; Sekino, T.; Niihara, K. Tough and strong Ce-TZP/Alumina nanocomposites doped with titania. Ceram. Int. 1998, 24, 497–506. [Google Scholar] [CrossRef]
- Ban, S. Reliability and properties of core materials for all-ceramic dental restorations. Jpn. Dent. Sci. Rev. 2008, 44, 3–21. [Google Scholar] [CrossRef]
- Ban, S.; Nawa, M. Application of zirconia/alumina composite to all ceramic crown. Dent. Mater. J. 2005, 24, 70. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.; Holloway, J.A.; Denry, I.L. Effect of coloring with various metal oxides on the microstructure, color, and flexural strength of 3Y-TZP. J. Biomed. Mater. Res.: Appl. Biomater. 2008, 87B, 329–337. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an-open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Denry, I.; Holloway, J.A. Ceramics for Dental Applications: A Review. Materials 2010, 3, 351-368. https://doi.org/10.3390/ma3010351
Denry I, Holloway JA. Ceramics for Dental Applications: A Review. Materials. 2010; 3(1):351-368. https://doi.org/10.3390/ma3010351
Chicago/Turabian StyleDenry, Isabelle, and Julie A. Holloway. 2010. "Ceramics for Dental Applications: A Review" Materials 3, no. 1: 351-368. https://doi.org/10.3390/ma3010351
APA StyleDenry, I., & Holloway, J. A. (2010). Ceramics for Dental Applications: A Review. Materials, 3(1), 351-368. https://doi.org/10.3390/ma3010351




