Impact of Chloride Impurities on the Corrosion Behavior of Stainless Steel in Molten Alkali Carbonate Salts for Concentrated Solar Power Systems
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials Preparation
2.2. Corrosion Experiment
2.3. Characterization Methods
3. Results
3.1. Gravimetric Study
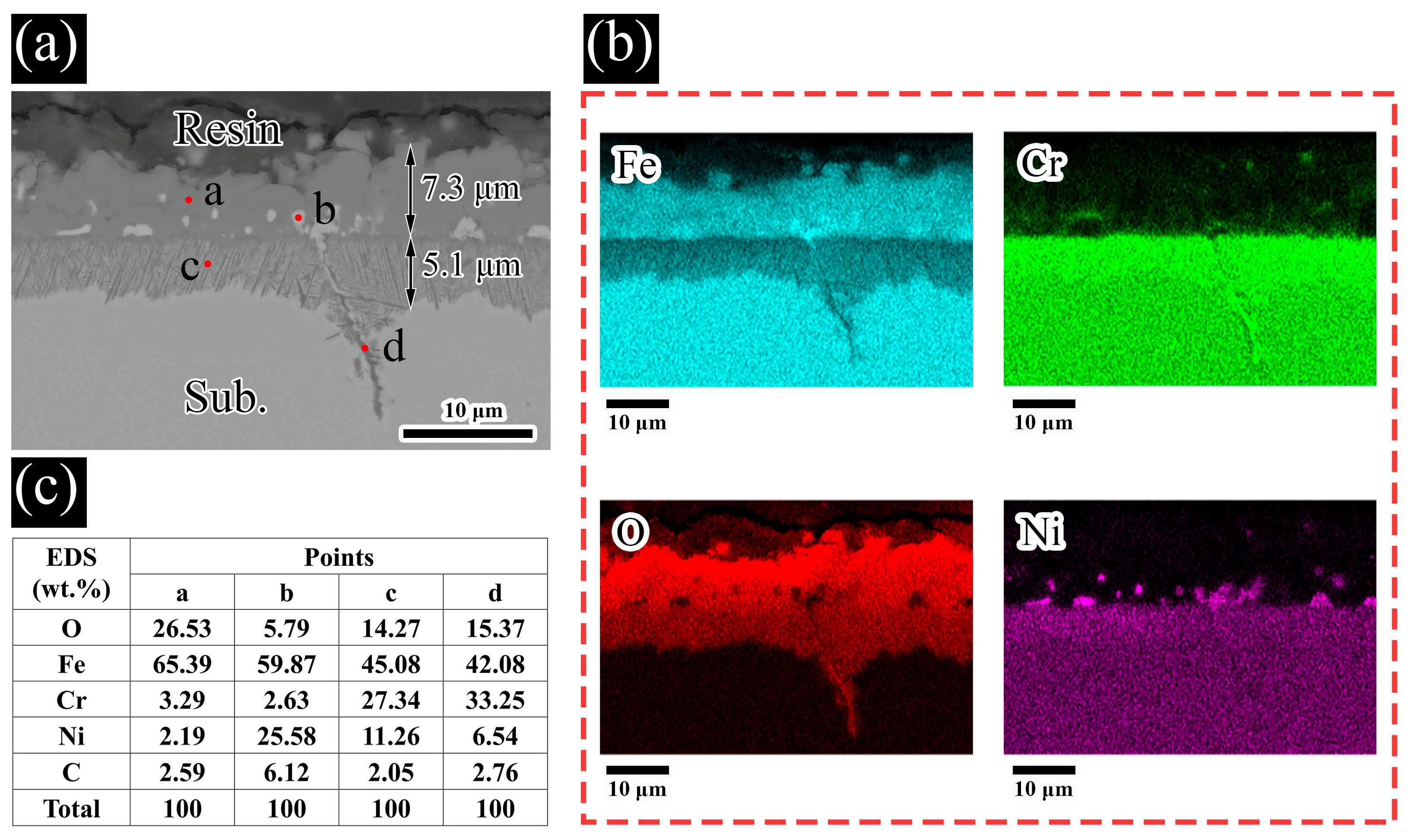
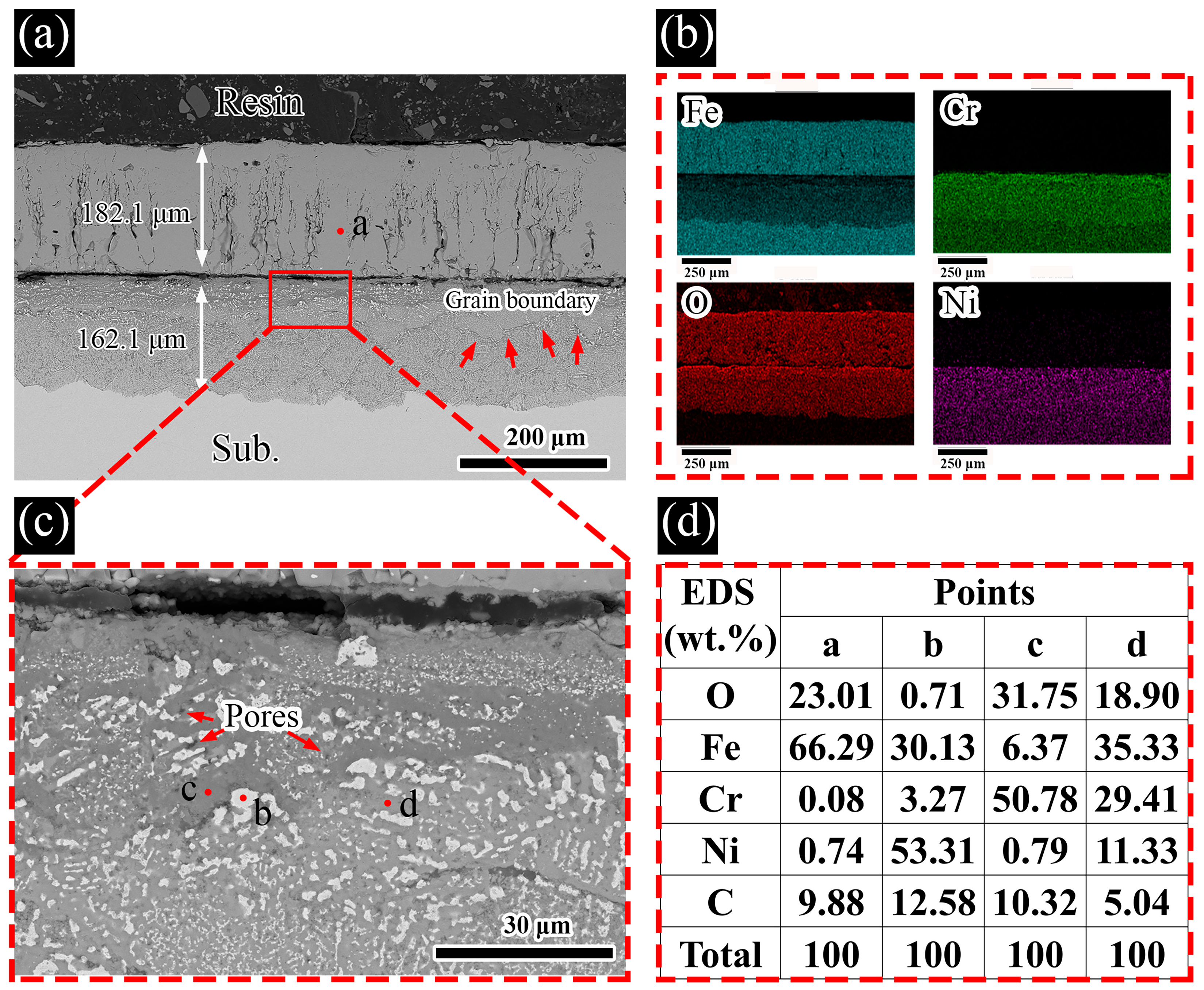
3.2. Sample Characterization
4. Discussion
5. Conclusions
- Exposure of SS316L to molten LiNaK-Cl carbonate salt at 700 °C resulted in substantially faster corrosion rates than those observed in pure LiNaK carbonate salt at the same temperature. Chloride ions were found to strongly promote corrosion progression.
- A distinct two-layered corrosion product scale formed on the SS316L samples immersed in molten LiNaK-Cl carbonate salt. The overall thickness of this bilayer scale grew continuously with increasing corrosion time, reflecting the sustained and accumulating nature of the corrosive attack.
- Transverse cracks along the grain boundaries and micropores were observed in the inner corrosion layer of the SS316L samples immersed in molten LiNaK-Cl carbonate salt at 700 °C. Furthermore, no Cl-rich regions were observed throughout the corroded samples, which is consistent with the proposed mechanism involving transient participation of Cl− during the corrosion process.
- Continuous outward migration of Cl2 and metal chlorides weakens the adhesion of the corrosion layers, resulting in prominent pores, transverse cracks, wrinkling, and partial peeling of the oxide scales, which in turn accelerates corrosion of the underlying substrate.
- During the corrosion process, the Cl− participating in the reaction was not consumed, but exerted an autocatalytic effect through the cyclic process of “oxidation-diffusion-reaction-regeneration”. This is also the core reason why no Cl-containing compounds or Cl-rich areas were detected in the corroded samples.
- Given the severe, sustained corrosive attack endured by SS316L in molten LiNaK-Cl carbonate salt, bare SS316L without protective coating is not viable for application as a structural component in CSP molten carbonate salt systems.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pourasl, H.H.; Barenji, R.V.; Khojastehnezhad, V.M. Solar energy status in the world: A comprehensive review. Energy Rep. 2023, 10, 3474–3493. [Google Scholar] [CrossRef]
- Abir, F.M.; Altwarah, Q.; Rana, M.T.; Shin, D. Recent advances in molten salt-based nanofluids as thermal energy storage in concentrated solar power: A comprehensive review. Materials 2024, 17, 955. [Google Scholar] [CrossRef] [PubMed]
- Mohan, G.; Venkataraman, M.; Gomez-Vidal, J.; Coventry, J. Assessment of a novel ternary eutectic chloride salt for next generation high-temperature sensible heat storage. Energy Convers. Manag. 2018, 167, 156–164. [Google Scholar] [CrossRef]
- Prieto, S.A.B.; Sabharwall, P. Technical and economic evaluation of heat transfer fluids for a TES system integrated to an advanced nuclear reactor. Appl. Energy 2024, 360, 122714. [Google Scholar] [CrossRef]
- Wong, W.P.; Walvekar, R.; Vaka, M.; Khalid, M.; Mubarak, N.M. A review on CSP and CPVT technologies: Energy, economic analysis and recycling towards circular economy. ECS J. Solid State Sci. Technol. 2023, 12, 031012. [Google Scholar] [CrossRef]
- Liu, X.; Zhong, Y.; Li, J.L.; Wang, H.Y.; Wang, M. A review of high-temperature molten salt for third-generation concentrating solar power. Energy Sci. Eng. 2025, 13, 456–474. [Google Scholar] [CrossRef]
- Olivares, R.I.; Chen, C.; Wright, S. The thermal stability of molten lithium-sodium-potassium carbonate and the influence of additives on the melting point. J. Sol. Energy Eng. Trans.-ASME 2012, 134, 041002. [Google Scholar] [CrossRef]
- Raud, R.; Jacob, R.; Bruno, F.; Will, G.; Steinberg, T.A. A critical review of eutectic salt property prediction for latent heat energy storage systems. Renew. Sustain. Energy Rev. 2017, 70, 936–944. [Google Scholar] [CrossRef]
- Liu, M.; Saman, W.; Bruno, F. Review on storage materials and thermal performance enhancement techniques for high temperature phase change thermal storage systems. Renew. Sustain. Energy Rev. 2012, 16, 2118–2132. [Google Scholar] [CrossRef]
- Gao, L.J. Progress of research in molten carbonate properties and current applications: A review. Sol. Energy Mater. Sol. Cells 2026, 296, 114022. [Google Scholar] [CrossRef]
- Wang, P.L.; Du, K.F.; Yin, H.Y.; Wang, D.H. Corrosion and protection of metallic materials in molten carbonates for concentrating solar power and molten carbonate electrolysis applications. Corros. Commun. 2023, 11, 58–71. [Google Scholar] [CrossRef]
- White, S.H.; Twardoch, U.M. The solubility and electrochemistry of alkali-metal oxides in the molten eutectic mixture of lithium-carbonate sodium-carbonate potassium carbonate. J. Appl. Electrochem. 1989, 19, 901–910. [Google Scholar] [CrossRef]
- Hsu, H.S.; Devan, J.H.; Howell, M. Solubilities of LiFeO2 and (Li,K)2CrO4 in molten alkali carbonates at 650 °C. J. Electrochem. Soc. 1987, 134, 2146–2150. [Google Scholar] [CrossRef]
- Arcos, C.; Guerra, C.; Ramos-Grez, J.A.; Sancy, M. Ni-Al bronze in molten carbonate manufactured by LPBF: Effect of porosity design on mechanical properties and oxidation. Materials 2023, 16, 3893. [Google Scholar] [CrossRef] [PubMed]
- Oger, L.; Agüero, A.; Audigie, P. Performance study of slurry aluminide coated 347H immersed in Li-Na-K carbonate molten salt for 5000-h at 700 °C. Surf. Coat. Technol. 2025, 517, 132762. [Google Scholar] [CrossRef]
- Luo, J.; Tariq, N.u.H.; Liu, H.H.; Li, N.; Zhao, L.J.; Cui, X.Y.; Xiong, T.Y. Robust corrosion performance of cold sprayed aluminide coating in ternary molten carbonate salt for concentrated solar power plants. Sol. Energy Mater. Sol. Cells 2022, 237, 111573. [Google Scholar] [CrossRef]
- Morales, M.; Gordon, S.; Fernández-Arana, O.; García-Marro, F.; Mateo, A.; Llanes, L.; Fargas, G. Duplex stainless steels for thermal energy storage: Characterization of oxide scales formed in carbonate salts at 500 °C. Metals 2022, 12, 2156. [Google Scholar] [CrossRef]
- Lambrecht, M.; García-Martín, G.; Miguel, M.T.D.; Lasanta, M.I.; Peréz, F.J. Temperature dependence of high-temperature corrosion on nickel-based alloy in molten carbonates for concentrated solar power applications. Corros. Sci. 2023, 220, 111262. [Google Scholar] [CrossRef]
- González-Fernández, L.; Intxaurtieta-Carcedo, M.; Bondarchuk, O.; Grosu, Y. Effect of dynamic conditions on high-temperature corrosion of ternary carbonate salt for thermal energy storage applications. Sol. Energy Mater. Sol. Cells 2022, 240, 111666. [Google Scholar] [CrossRef]
- Meng, Q.; Lai, L.; Rao, W.; Li, A.; Yu, H.C.; La, P. Creep properties and corrosion behavior of TP347H stainless steel with Al in molten carbonate salt. Materials 2024, 17, 6108. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, A.G.; Pineda, F.; Walczak, M.; Cabeza, L.F. Corrosion evaluation of alumina-forming alloys in carbonate molten salt for CSP plants. Renew. Energy 2019, 140, 227–233. [Google Scholar] [CrossRef]
- Dong, J.; Zhao, C.Y. Corrosion behavior of alloys in Li2CO3-K2CO3 eutectic salt under an Air-then-Argon condition. Sol. Energy Mater. Sol. Cells 2025, 290, 113671. [Google Scholar] [CrossRef]
- Li, H.; Feng, X.C.; Wang, X.W.; Yang, X.Y.; Tang, J.Q.; Gong, J.M. Impact of temperature on corrosion behavior of austenitic stainless steels in solar salt for CSP application: An electrochemical study. Sol. Energy Mater. Sol. Cells 2022, 239, 111661. [Google Scholar] [CrossRef]
- Sarvghad, M.; Muransky, O.; Steinberg, T.A.; Hester, J.; Hill, M.R.; Will, G. On the effect of cold-rolling on the corrosion of SS316L alloy in a molten carbonate salt. Sol. Energy Mater. Sol. Cells 2019, 202, 110136. [Google Scholar] [CrossRef]
- Sarvghad, M.; Steinberg, T.A.; Will, G. Corrosion of steel alloys in eutectic NaCl + Na2CO3 at 700 °C and Li2CO3 + K2CO3 + Na2CO3 at 450 °C for thermal energy storage. Sol. Energy Mater. Sol. Cells 2017, 170, 48–59. [Google Scholar] [CrossRef]
- Frangini, S.; Loreti, S. The role of temperature on the corrosion and passivation of type 310S stainless steel in eutectic (Li+K) carbonate melt. J. Power Sources 2006, 160, 800–804. [Google Scholar] [CrossRef]
- Liu, T.; Xu, X.; Liu, W.; Zhuang, X. Corrosion of alloys in high temperature molten-salt heat transfer fluids with air as the cover gas. Sol. Energy 2019, 191, 435–448. [Google Scholar] [CrossRef]
- Zhuang, X.; Liu, W.; Xu, X. Hot corrosion of different alloys in chloride and carbonate molten-salt mixtures under argon atmosphere. Sol. Energy 2019, 189, 254–267. [Google Scholar] [CrossRef]
- Li, N.; Wang, H.; Yin, H.; Liu, Q.; Tang, Z. Effect of temperature and impurity content to control corrosion of 316 stainless steel in molten KCl-MgCl2 salt. Materials 2023, 16, 2025. [Google Scholar] [CrossRef]
- Yoo, J.-S.; Chung, N.T.; Lee, Y.-H.; Kim, Y.-W.; Kim, J.-G. Effect of sulfide and chloride ions on pitting corrosion of type 316 austenitic stainless steel in groundwater conditions using response surface methodology. Materials 2024, 17, 178. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, H.X.; Yin, H.Q.; Li, N.; Wang, W.R.; Li, L.F.; Tang, Z.F.; Qian, Y. Corrosion behaviour of 316 stainless steel in NaCl-KCl-MgCl2 salt vapour at 700 °C. Corros. Sci. 2022, 194, 109921. [Google Scholar] [CrossRef]
- Javier Ruiz-Cabanas, F.; Prieto, C.; Madina, V.; Ines Fernandez, A.; Cabeza, L.F. Materials selection for thermal energy storage systems in parabolic trough collector solar facilities using high chloride content nitrate salts. Sol. Energy Mater. Sol. Cells 2017, 163, 134–147. [Google Scholar] [CrossRef]
- Luo, J.; Deng, C.K.; Tariq, N.u.H.; Li, N.; Han, R.F.; Liu, H.H.; Wang, J.Q.; Cui, X.Y.; Xiong, T.Y. Corrosion behavior of SS316L in ternary Li2CO3-Na2CO3-K2CO3 eutectic mixture salt for concentrated solar power plants. Sol. Energy Mater. Sol. Cells 2020, 217, 110679. [Google Scholar] [CrossRef]
- Dorcheh, A.S.; Durham, R.N.; Galetz, M.C. High temperature corrosion in molten solar salt: The role of chloride impurities. Mater. Corros. 2017, 68, 943–951. [Google Scholar] [CrossRef]
- Bradshaw, R.W.; Clift, W.M. Effect of chloride content of molten nitrate salt on corrosion of A516 carbon steel. In Office of Scientific & Technical Information Technical Reports; Office of Scientific and Technical Information: Washington, DC, USA, 2010. [Google Scholar]
- Grabke, H.J.; Reese, E.; Spiegel, M. The effects of chlorides, hydrogen chloride, and sulfur-dioxide in the oxidation of steels below deposits. Corros. Sci. 1995, 37, 1023–1043. [Google Scholar] [CrossRef]
- Kenisarin, M.M. High-temperature phase change materials for thermal energy storage. Renew. Sustain. Energy Rev. 2010, 14, 955–970. [Google Scholar] [CrossRef]
- Pan, T.J.; Sánchez-Pastén, M.; Li, Y.S. Comparison of hot corrosion behaviour of Fe-Cr and Fe-Al alloys beneath chloride salt melt. High Temp. Mater. Process. 2009, 28, 147–154. [Google Scholar] [CrossRef]
- Li, Y.S.; Spiegel, M.; Shimada, S. Corrosion behaviour of various model alloys with NaCl-KCl coating. Mater. Chem. Phys. 2005, 93, 217–223. [Google Scholar] [CrossRef]







| Salt | Molecular Mass g/mol | Heat of Fusion J/g | Melting Point °C | Purity % | Impurity Level (Max) ppm | |||
|---|---|---|---|---|---|---|---|---|
| Ca | Cl | N(NO3) | SO42− | |||||
| Li2CO3 | 73.9 | 509 | 732 | 98.0 | 300 | 50 | 30(NO3) | 500 |
| Na2CO3 | 106.0 | 165 | 858 | 99.8 | 100 | 20 | 10(N) | 50 |
| K2CO3 | 138.2 | 202 | 900 | 99.0 | 20 | 30 | 10(N) | 30 |
| NaCl | 58.4 | 482 | 801 | 99.5 | 20 | - | 5(N) | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Luo, J.; Li, N.; Tariq, N.u.H.; Xiong, T.; Cui, X. Impact of Chloride Impurities on the Corrosion Behavior of Stainless Steel in Molten Alkali Carbonate Salts for Concentrated Solar Power Systems. Materials 2026, 19, 1312. https://doi.org/10.3390/ma19071312
Luo J, Li N, Tariq NuH, Xiong T, Cui X. Impact of Chloride Impurities on the Corrosion Behavior of Stainless Steel in Molten Alkali Carbonate Salts for Concentrated Solar Power Systems. Materials. 2026; 19(7):1312. https://doi.org/10.3390/ma19071312
Chicago/Turabian StyleLuo, Jing, Ning Li, Naeem ul Haq Tariq, Tianying Xiong, and Xinyu Cui. 2026. "Impact of Chloride Impurities on the Corrosion Behavior of Stainless Steel in Molten Alkali Carbonate Salts for Concentrated Solar Power Systems" Materials 19, no. 7: 1312. https://doi.org/10.3390/ma19071312
APA StyleLuo, J., Li, N., Tariq, N. u. H., Xiong, T., & Cui, X. (2026). Impact of Chloride Impurities on the Corrosion Behavior of Stainless Steel in Molten Alkali Carbonate Salts for Concentrated Solar Power Systems. Materials, 19(7), 1312. https://doi.org/10.3390/ma19071312






