What Is the Main Cause of Shrinkage Porosity in AlSi7Mg0.6 Alloy Castings Obtained with an Increased Share of Secondary Materials?
Abstract
1. Introduction
- Perform two series of smeltings with increasing content from 0.3 to 0.8 wt.% Fe and a constant content of approx. 0.1 wt.% Mn and increasing iron and manganese content (Mn/Fe = 1/2) cast by gravity;
- Perform one series of low-pressure smelting (approx. 0.15 MPa) with increasing content from 0.8 wt.% to 1.3 wt.% Fe and a constant content of approx. 0.1 wt.% Mn according to the adopted experiment plan;
- Thermal analysis DTA (Derivative Thermal Analysis) and DSC (Differential Scanning Calorimetry);
- Microstructure testing (including qualitative and quantitative analysis of shrinkage pores);
- Demonstrating the possible relationship between increased iron phase content—due to the higher use of charge scrap—and the development of shrinkage porosity in AlSi7Mg0.6 alloy castings. This applies to castings obtained by both gravity and low-pressure methods;
- Which theory of porosity formation in castings from secondary Al-Si alloys is correct? The one that assumes that shrinkage porosity results primarily from the presence of lamellar β-Fe phases, which hinder the flow of the liquid alloy during crystallization, or the one that assumes that porosity is caused solely by thin, bilayer oxide films?
2. Materials and Methods
2.1. Research Materials and Methodology
2.1.1. Methodology of Melting Alloys
- Gravitational casting into steel molds with increasing iron content from 0.3 wt.% Fe (G1) to 0.8 wt.% Fe (G6) and constant manganese content (approx. 0.1 wt.%);
- Gravity casting into a steel mold with increasing iron and manganese content (from G1a to G6a);
- Under low pressure from 0.8 wt.% Fe (D1) to 1.3 wt.% Fe (D6) on a Kurtz ND-GM B47 casting machine (Kurtz GmbH & Co. KG, Kreuzwertheim, Germany) with an average pouring speed of approx. 0.04 m·s−1.

2.1.2. Methodology of DTA and DSC Methods
2.1.3. Microstructure Research Methodology
- Properly selected images of transformations of initial pore images into binary measurement;
- Methods for shadow correction (elimination) and image binarization before performing calculations;
- Estimating the necessary number of measurements required to obtain repeatable and reproducible test results;
- Assessment of inhomogeneity, i.e., the size, shape, and distribution of pores.
- Fraction of the image area occupied by pores, given in %. Images with the same surface area were analyzed each time:
- Average pore surface area, μm2,
- Average pore circumference, μm.
- Dimensionless aspect ratio. The closer to unity, the more rounded the shape of an individual pore tends to be, which is a characteristic feature of gas pores. If this coefficient tends to zero, then the shape deviates from roundness, becoming dendritic, which is typical of contractile pores.
3. Results
3.1. Chemical Composition Test Results
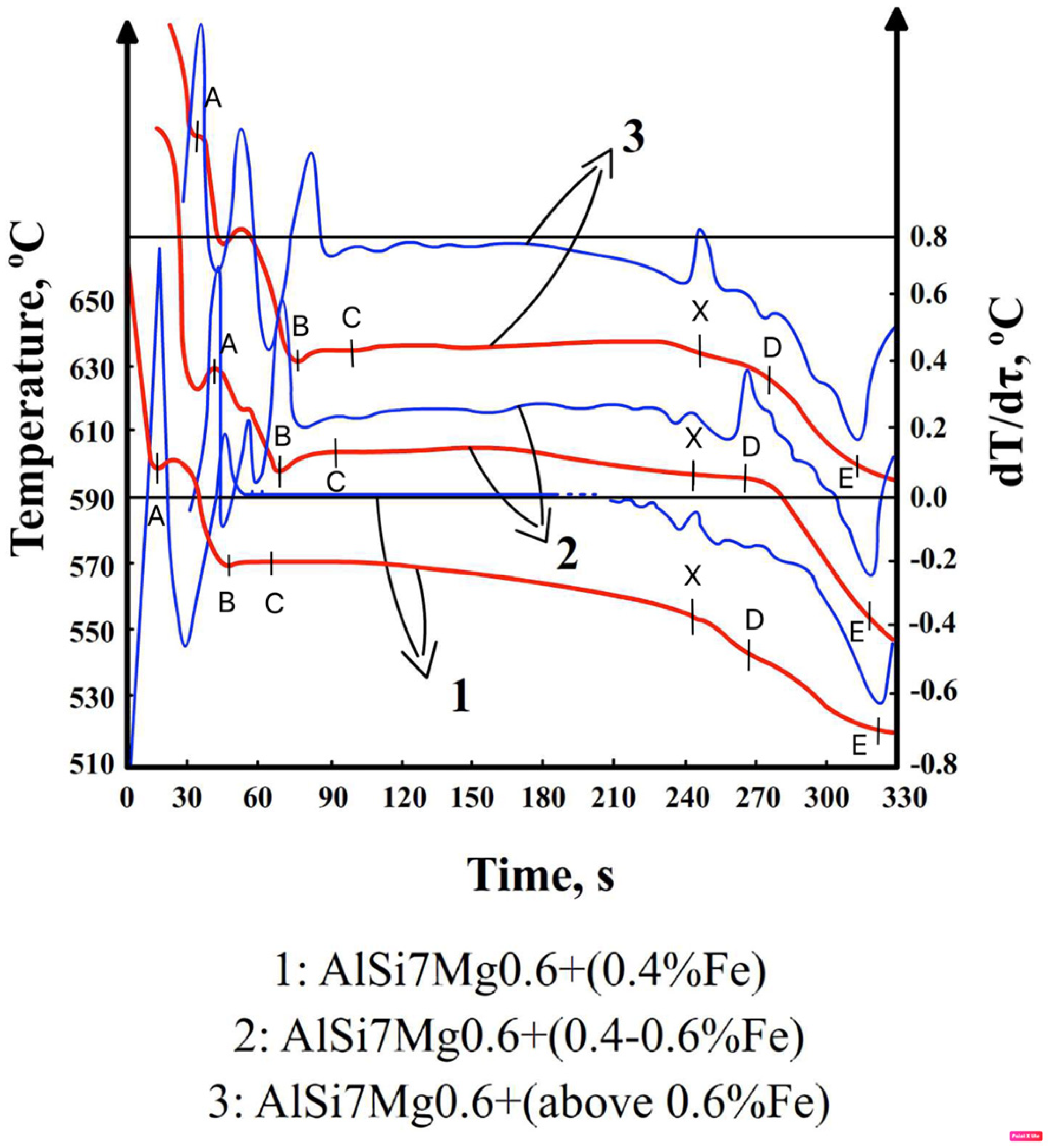
3.2. DTA Test Research
- –
- T = f(τ)—curve of temperature change T over time τ,—the so-called TA curve (Temperature Analysis);
- –
- dT/dτ = f’(τ)—the first derivative of temperature change over time—the DTA (Derivative Thermal Analysis) curve;
- –
- point A—the temperature at which the dendrites of a solid solution begin to crystallize α(Al)—Tliq. = T(α), °C;
- –
- point B—minimum temperature (onset) of eutectic crystallization α(Al) + β(Si),—TEmin(α+β), °C;
- –
- point C—average eutectic crystallization temperature α(Al) + β(Si),—TE(α+β),°C;
- –
- point D—crystallization temperature of magnesium-rich intermetallic phases—TE(Mg), °C;
- –
- point E—end temperature of crystallization of the AlSi7Mg0.6 alloy (Tsol.), °C;
- –
- point X—crystallization temperature of iron-rich intermetallic phases—TFe, °C;
- –
- point Y—crystallization temperature of intermetallic phases rich in iron and manganese—TFe,Mn, °C.
3.3. DSC Test Research
- –
- PαH—thermal effect resulting from the separation of dendrites of the a(Al) solid solution during heating, PαC—during cooling, mW;
- –
- PFeH—thermal effect resulting from the precipitation of the Al5FeSi (β-Fe) phase during heating, PFeC—during cooling, mW;
- –
- Pα+βH—thermal effect resulting from the formation of double eutectic α(Al) + β(Si) during heating, Pα+βC—during cooling, mW;
- –
- PMgH—thermal effect resulting from the release of eutectic containing the Mg2Si phase during heating, PMgC—during cooling, mW;
- –
- PSPH—thermal effect resulting from the release of sludge particles (SPs) during heating, PSPC—during cooling, mW;
- –
- PFeMnH—thermal effect resulting from the precipitation of the α-Al15(Fe,Mn)3Si2 phase during heating, PFeMnC—during cooling, mW;
- –
- Endo—endothermic reactions;
- –
- Exo—exothermic reactions.
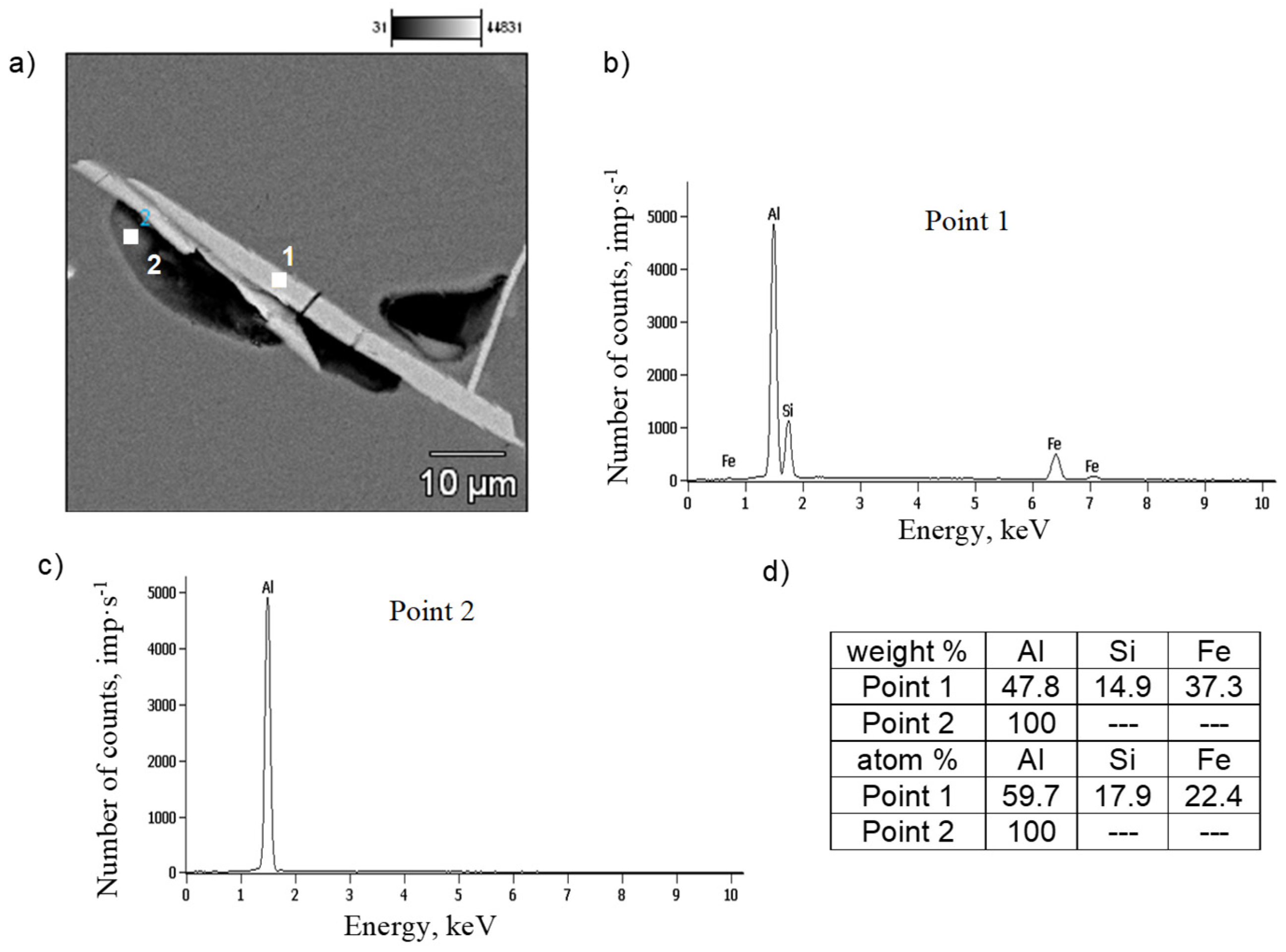
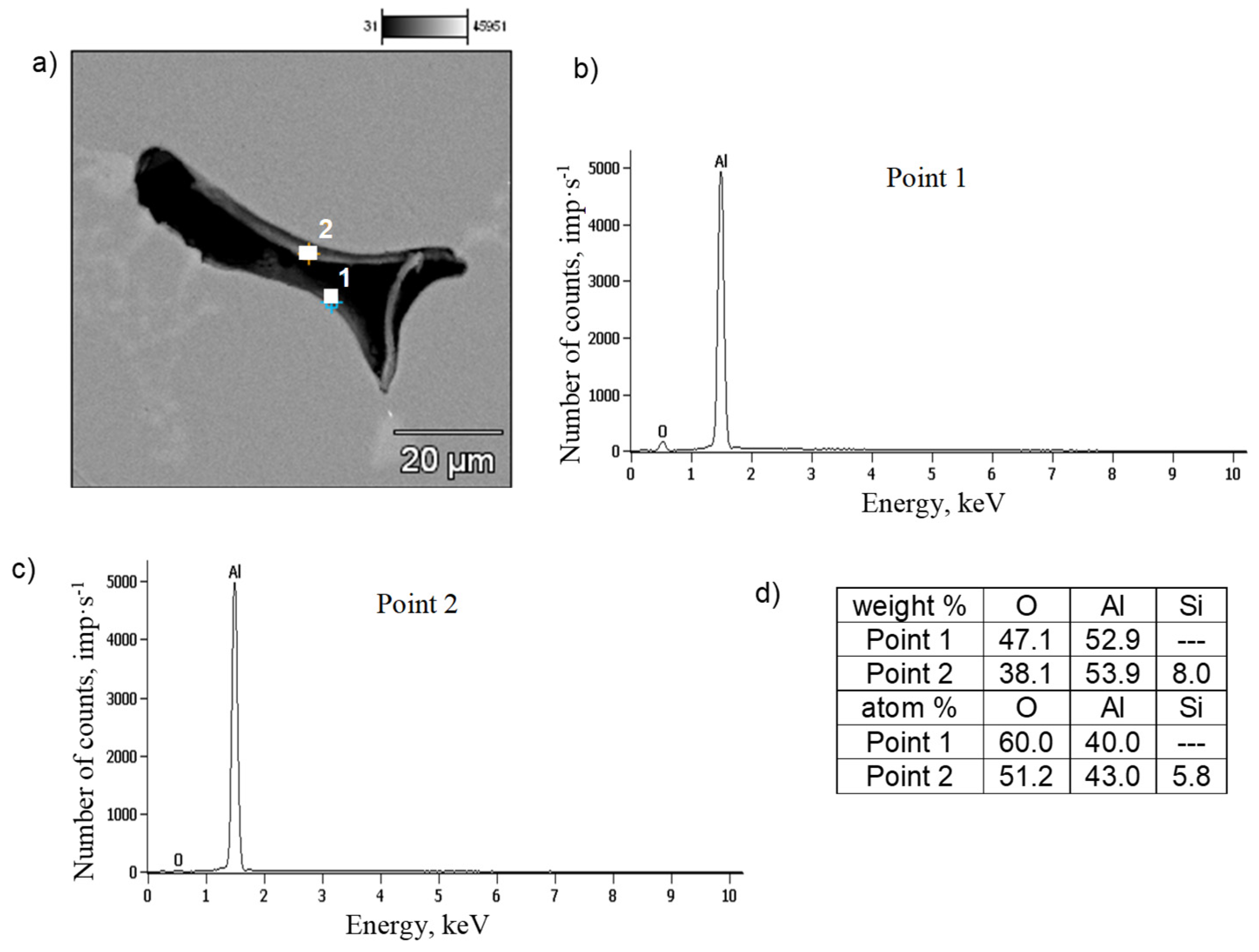
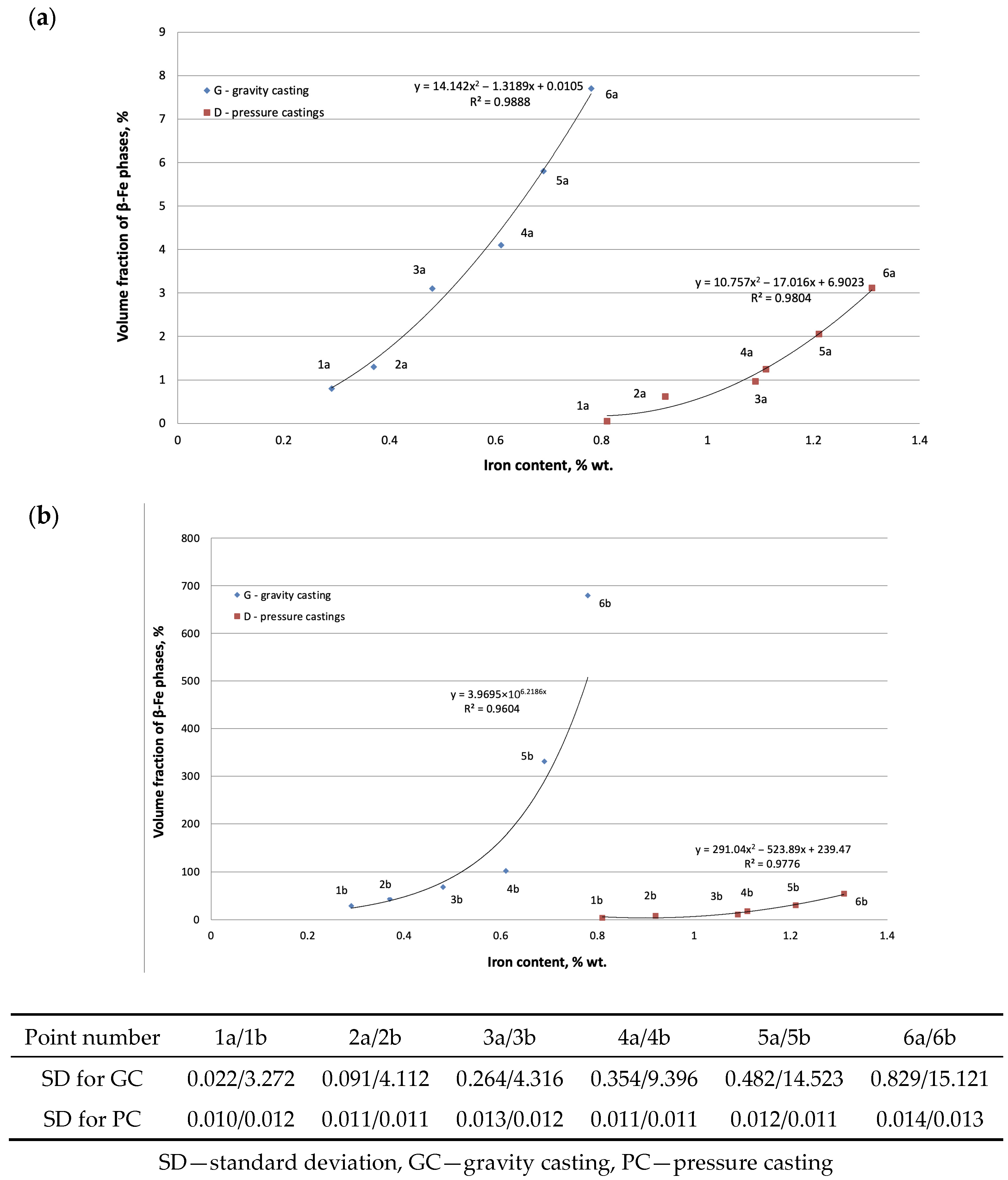
3.4. Microstructure Test Results
4. Discussion
- Gravity with increasing content from 0.3 to 0.8 wt.% Fe and constant content of approx. 0.1 wt.% Mn—sample designation from G1 to G6;
- Gravity with increasing iron content (from 0.3 to 0.8 wt.% Fe) and manganese (Mn/Fe = 1/2)—sample designation from G1a to G6a;
- Under low pressure (approx. 0.15 MPa) with increasing content from 0.8 wt.% to 1.3 wt.% Fe and constant content of approx. 0.1 wt.% Mn—sample designation from D1 to D6.
- Thermal analysis using DTA and DSC methods—for gravity castings;
- Microstructure—for gravity and pressure castings.
- Measurement speed (up to several minutes);
- The possibility of testing under industrial conditions (on the production floor);
- Relatively simple interpretation of results;
- High versatility (determination of phase transformations and composition, metal purity, crystallization range, thermal stability, optimization of heat treatment parameters, structure prediction, etc.).
- To a content of about 0.4 wt.% Fe:L → α(Al) → α(Al) + β(Si) → [α(Al + Al5FeSi + β(Si)]EFe → [α(Al) + (Mg2Si) + β(Si)]EMg → S
- With a content of about 0.41 to 0.7 wt.% Fe:L → α(Al) → Al5FeSi → α(Al) + β(Si) → [α(Al) + (Mg2Si) + β(Si)]EMg
- With a content of more than 0.70 wt.% Fe:L → Al5FeSi → α(Al) → α(Al) + β(Si) → [α(Al) + (Mg2Si) + β(Si)]E2
5. Conclusions
- Increased risk of crystallization: For Fe contents of 0.4 to 0.7 wt.%, the Al5FeSi phase (with a length of approximately 30 to 200 μm) precipitates along the Al(α) dendrites. Above the basic threshold (~0.7 wt.% Fe), this phase crystallizes primarily (reaching a length of 300 to 700 μm).
- Precipitation of the Al5FeSi phase is inhibited by the properties of the interdendritic regions, which lead to increased shrinkage porosity. This can occur in a dispersed form; in gravity casting, the main cause of shrinkage porosity is Al5FeSi with a lamellar morphology.
- The unfavorable use of the Al5FeSi phase can be introduced by adding manganese. The Al5FeSi cycle is in a smaller, compact Al15(Fe,Mn)3Si2 precipitate. A metallurgical solution.
- During low-pressure feed transfer, the Al5FeSi phase is not as harmful, and the additional content available is higher. Rapid crystallization to the die forms results in a small phase (up to 30 µm) and is not hindered by the liquid alloy at the crystallization front.
- Multi-stage liquid metal transport (furnace → ladle → injection chamber) introduces turbulence and aeration, using double-layer oxides (so-called “bifilms”). Therefore, during pressure transfer, after switching on the comparative feed, the Fe level is of secondary importance.
- A Mn/Fe ratio of ≈1/2 should be used, which will lead to sludge effects. Their presence in Al-Si alloys melted with a separate scrap charge is unfavorable. This applies to the first pressure casting release.
- Identification of oxide films in the microstructure of Al-Si-Mg alloys is very difficult and requires tedious micro-computed tomography studies and a series of cross-sections, subjected to detailed analysis in the micro-areas around the shrinkage pores.
- The presented research results confirm the accepted hypothesis about the influence of oxide films on the development of shrinkage porosity in AlSi7Mg0.6 alloy pressure castings, but do not prove the mechanism of their influence. The detected presence of oxygen does not directly prove the morphology of oxide layers, but only the oxidation of the surface of Al-Si-Mg alloy castings. Therefore, we can only infer the presence of oxide layers in the microstructure of aluminum alloys, not directly observe them.
- For slow-action and quiescent processes (gravity casting), chemical composition control should be considered to preserve the morphology of the Al5FeSi phases. If necessary, manganese addition is required.
- If the method is used (pressure casting), a control process should be considered to prevent turbulence and oxide precipitation.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grandfield, J.F.; Eskin, D.G.; Bainbridge, I.F. (Eds.) Direct-Chill Casting of Light Alloys: Science and Technology, 1st ed.; Wiley: Hoboken, NJ, USA, 2013; ISBN 978-1-118-02265-8. [Google Scholar]
- Bell, S.; Davis, B.; Javaid, A.; Essadiqi, E. Final Report on Effect of Impurities in Aluminum; Government of Canada: Ottawa, ON, Canada, 2006.
- Belov, N.A.; Aksenov, A.A.; Eskin, D.G. Iron in Aluminium Alloys: Impurity and Alloying Element; CRC Press: Boca Raton, FL, USA, 2002; ISBN 978-0-429-18990-6. [Google Scholar]
- Anyalebechi, P.N. Hydrogen-Induced Gas Porosity Formation in Al–4.5 Wt% Cu–1.4 Wt% Mg Alloy. J. Mater. Sci. 2013, 48, 5342–5353. [Google Scholar] [CrossRef]
- Walallawita, R.; Stroh, J.; Sediako, D. The Effect of Hydrogen Embrittlement on Aluminum Alloys Used for Hydrogen-Fueled Internal Combustion Engine. In Proceedings of the 61st Conference of Metallurgists, COM 2022, Montreal, QC, Canada, 21–24 August 2022; Metallurgy and Materials Society of CIM, Ed.; Springer International Publishing: Cham, Switzerland, 2023; pp. 447–450. ISBN 978-3-031-17424-7. [Google Scholar]
- Carlson, K.D.; Lin, Z.; Beckermann, C. Modeling the Effect of Finite-Rate Hydrogen Diffusion on Porosity Formation in Aluminum Alloys. Metall. Mater. Trans. B 2007, 38, 541–555. [Google Scholar] [CrossRef]
- Park, J.M. Behaviours of Bifilms in A356 Alloy During Solidification: Developing Observation Techniques with 3-D Micro X-Ray Tomography. Master’s Thesis, The University of Birmingham, Birmingham, UK, 2009. [Google Scholar]
- Bahreinian, F.; Boutorabi, S.M.A.; Campbell, J. Critical Gate Velocity for Magnesium Casting Alloy (ZK51A). Int. J. Cast Met. Res. 2006, 19, 45–51. [Google Scholar] [CrossRef]
- Campbell, J. Entrainment Defects. Mater. Sci. Technol. 2006, 22, 127–145. [Google Scholar] [CrossRef]
- Campbell, J. Complete Casting Handbook; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 978-0-444-63509-9. [Google Scholar]
- Raiszadeh, R.; Griffiths, W.D. A Method to Study the History of a Double Oxide Film Defect in Liquid Aluminum Alloys. Metall. Mater. Trans. B 2006, 37, 865–871. [Google Scholar] [CrossRef]
- Cao, X.; Campbell, J. The Nucleation of Fe-Rich Phases on Oxide Films in Al-11.5Si-0.4Mg Cast Alloys. Metall. Mater. Trans. A 2003, 34, 1409–1420. [Google Scholar] [CrossRef]
- Kammer, C. Aluminium Handbook, Tom 1 Aluminium Handbook; American Society for Testing & Materials: West Conshohocken, PA, USA, 1999; Volume 1, ISBN 3-87017-261-4. [Google Scholar]
- Mbuya, T.O.; Odera, B.O.; Ng’ang’a, S.P. Influence of Iron on Castability and Properties of Aluminium Silicon Alloys: Literature Review. Int. J. Cast Met. Res. 2003, 16, 451–465. [Google Scholar] [CrossRef]
- Ferraro, S.; Fabrizi, A.; Timelli, G. Evolution of Sludge Particles in Secondary Die-Cast Aluminum Alloys as Function of Fe, Mn and Cr Contents. Mater. Chem. Phys. 2015, 153, 168–179. [Google Scholar] [CrossRef]
- Seifeddine, S.; Svensson, I.L. The Influence of Fe and Mn Content and Cooling Rate on the Microstructure and Mechanical Properties of A380-Die Casting Alloys. Metall. Sci. Technol. 2009, 27, 11–20. [Google Scholar]
- Mahta, M.; Emamy, M.; Cao, X.; Campbell, J. Overview of Beta-Al5FeSi Phase in Al-Si Cast Alloys. In Materials Science Research Trends; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2008; pp. 251–271. [Google Scholar]
- Ebhota, W.S.; Jen, T.-C. Intermetallics Formation and Their Effect on Mechanical Properties of Al-Si-X Alloys. In Intermetallic Compounds—Formation and Applications; Aliofkhazraei, M., Ed.; InTech: Vienna, Austria, 2018; ISBN 978-1-78923-178-6. [Google Scholar]
- Khalifa, W.; Samuel, F.H.; Gruzleski, J.E. Iron Intermetallic Phases in the Al Corner of the Al-Si-Fe System. Metall. Mater. Trans. A 2003, 34, 807–825. [Google Scholar] [CrossRef]
- Mikołajczak, P.; Ratke, L. Three Dimensional Morphology of β-Al5FeSi Intermetallics in AlSi Alloys. Arch. Foundry Eng. 2015, 15, 47–50. [Google Scholar] [CrossRef]
- Lu, L.; Dahle, A.K. Iron-Rich Intermetallic Phases and Their Role in Casting Defect Formation in Hypoeutectic Al−Si Alloys. Metall. Mater. Trans. A 2005, 36, 819–835. [Google Scholar] [CrossRef]
- Taylor, J.A.; Schaffer, G.B.; StJohn, D.H. The Role of Iron in the Formation of Porosity in Al-Si-Cu-Based Casting Alloys: Part I. Initial Experimental Observations. Metall. Mater. Trans. A 1999, 30, 1643–1650. [Google Scholar] [CrossRef]
- Mohamed, A.M.A.; Samuel, E.; Zedan, Y.; Samuel, A.M.; Doty, H.W.; Samuel, F.H. Intermetallics Formation during Solidification of Al-Si-Cu-Mg Cast Alloys. Materials 2022, 15, 1335. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Y.; Wu, Y.; Wei, Z.; Liu, X. Supportive Strengthening Role of Cr-Rich Phase on Al–Si Multicomponent Piston Alloy at Elevated Temperature. Mater. Sci. Eng. A 2011, 528, 4427–4430. [Google Scholar] [CrossRef]
- Timelli, G.; Bonollo, F. The Influence of Cr Content on the Microstructure and Mechanical Properties of AlSi9Cu3(Fe) Die-Casting Alloys. Mater. Sci. Eng. A 2010, 528, 273–282. [Google Scholar] [CrossRef]
- Mahta, M.; Emamy, M.; Daman, A.; Keyvani, A.; Campbell, J. Precipitation of Fe Rich Intermetallics in Cr- and Co-Modified A413 Alloy. Int. J. Cast Met. Res. 2005, 18, 73–79. [Google Scholar] [CrossRef]
- De Moraes, H.L.; De Oliveira, J.R.; Espinosa, D.C.R.; Tenório, J.A.S. Removal of Iron from Molten Recycled Aluminum through Intermediate Phase Filtration. Mater. Trans. 2006, 47, 1731–1736. [Google Scholar] [CrossRef]
- Samuel, A.M.; Doty, H.W.; Valtierra, S.; Samuel, F.H. Beta Al5FeSi Phase Platelets-Porosity Formation Relationship in A319.2 Type Alloys. Int. J. Met. 2018, 12, 55–70. [Google Scholar] [CrossRef]
- EuRIC aisbl. Metal Recycling Factsheet. Europe Aluminium Market Overview 2024–2028; EuRIC aisbl: Schaerbeek, Belgium, 2020; pp. 1–8. [Google Scholar]
- Guo, H.-M.; Yang, X.-J.; Wang, J.-X. Pressurized Solidification of Semi-Solid Aluminum Die Casting Alloy A356. J. Alloys Compd. 2009, 485, 812–816. [Google Scholar] [CrossRef]
- Piątkowski, J.; Roskosz, S.; Stach, S.; Górny, M. Crystallisation and Microstructure of Sludge Particles in AlSi7Mg Secondary Alloys with Increased Iron Content. Materials 2025, 18, 4921. [Google Scholar] [CrossRef]
- Piatkowski, J.; Nowinska, K.; Matula, T.; Siwiec, G.; Szucki, M.; Oleksiak, B. Microstructure and Mechanical Properties of AlSi10MnMg Alloy with Increased Content of Recycled Scrap. Materials 2025, 18, 1119. [Google Scholar] [CrossRef]
- Samuel, A.M.; Samuel, E.; Songmene, V.; Samuel, F.H. A Review on Porosity Formation in Aluminum-Based Alloys. Materials 2023, 16, 2047. [Google Scholar] [CrossRef]
- Piatkowski, J.; Matula, T.; Chowaniec, L. Influence of Iron and Manganese on the Microstructure and Mechanical Properties of AlSi9Cu3(Fe) Alloy Castings. Mach. Technol. Mater. 2025, 19, 191–194. [Google Scholar]
- Fox, S.; Campbell, J. Visualisation of Oxide Film Defects during Solidification of Aluminium Alloys. Scr. Mater. 2000, 43, 881–886. [Google Scholar] [CrossRef]
- Dispinar, D.; Campbell, J. Porosity, Hydrogen and Bifilm Content in Al Alloy Castings. Mater. Sci. Eng. A 2011, 528, 3860–3865. [Google Scholar] [CrossRef]
- Lee, P.D.; Wang, J. Modeling of Porosity Formation during Solidification. In Metals Process Simulation; Furrer, D.U., Semiatin, S.L., Eds.; ASM International: Almere, The Netherlands, 2010; pp. 253–263. ISBN 978-1-62708-197-9. [Google Scholar]
- Piątkowski, J.; Roskosz, S.; Stach, S. The Influence of Selected High—Pressure Die Casting Parameters on the Porosity ofEN AB-46000 Alloy Castings. Adv. Sci. Technol. Res. J. 2024, 18, 361–371. [Google Scholar] [CrossRef] [PubMed]






| Sample No. | Element Content, wt.% | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Si | Mg | Fe 1 | Fe 2 | Mn | Cu | Ni | Zn | Ti | Al | |
| G1 | 6.93 | 0.57 | 0.3 | 0.29 | 0.08 | 0.05 | 0.04 | ≤0.08 | 0.12 | rest |
| G2 | 7.11 | 0.62 | 0.4 | 0.37 | 0.06 | 0.07 | 0.03 | ≤0.08 | 0.15 | rest |
| G3 | 7.08 | 0.60 | 0.5 | 0.48 | 0.06 | 0.03 | 0.04 | ≤0.09 | 0.13 | rest |
| G4 | 6.96 | 0.59 | 0.6 | 0.61 | 0.07 | 0.03 | 0.02 | ≤0.10 | 0.13 | rest |
| G5 | 6.97 | 0.61 | 0.7 | 0.69 | 0.08 | 0.05 | 0.02 | ≤0.06 | 0.15 | rest |
| G6 | 7.08 | 0.61 | 0.8 | 0.78 | 0.09 | 0.04 | 0.02 | ≤0.10 | 0.18 | rest |
| G1a | 7.01 | 0.60 | 0.3 | 0.27 | 0.16 | 0.03 | 0.03 | ≤0.08 | 0.17 | rest |
| G2a | 6.96 | 0.59 | 0.4 | 0.40 | 0.21 | 0.05 | 0.04 | ≤0.09 | 0.15 | rest |
| G3a | 6.68 | 0.61 | 0.5 | 0.49 | 0.26 | 0.04 | 0.02 | ≤0.10 | 0.13 | rest |
| G4a | 7.02 | 0.60 | 0.6 | 0.58 | 0.31 | 0.03 | 0.03 | ≤0.10 | 0.16 | rest |
| G5a | 7.02 | 0.59 | 0.7 | 0.69 | 0.35 | 0.05 | 0.04 | ≤0.08 | 0.15 | rest |
| G6a | 6.96 | 0.61 | 0.8 | 0.81 | 0.41 | 0.04 | 0.02 | ≤0.09 | 0.13 | rest |
| D1 | 7.05 | 0.62 | 0.8 | 0.81 | 0.40 | 0.04 | 0.04 | ≤0.10 | 0.15 | rest |
| D2 | 7.12 | 0.60 | 0.9 | 0.92 | 0.45 | 0.03 | 0.03 | ≤0.06 | 0.18 | rest |
| D3 | 7.20 | 0.59 | 1.0 | 1.09 | 0.49 | 0.04 | 0.02 | ≤0.10 | 0.15 | rest |
| D4 | 7.18 | 0.62 | 1.1 | 1.11 | 0.51 | 0.03 | 0.01 | ≤0.08 | 0.13 | rest |
| D5 | 6.98 | 0.61 | 1.2 | 1.21 | 0.60 | 0.05 | 0.03 | ≤0.09 | 0.16 | rest |
| D6 | 7.02 | 0.59 | 1.3 | 1.31 | 0.65 | 0.04 | 0.01 | ≤0.10 | 0.16 | rest |
| Sample | Name of the Alloy and the Share of Iron and Manganese, wt.% | Points and Their Corresponding Crystallization Temperatures, °C | ||||||
|---|---|---|---|---|---|---|---|---|
| A Tliq. = T(α) | B TEmin(α+β) | C TE(α+β) | D TE(Mg) | E Tsol. | X TFe | Y TFe,Mn | ||
| G1 | AlSi7Mg0.6 + 0.3% Fe + 0.08% Mn | 604 | 575 | 578 | 546 | 499 | 558 | --- |
| G2 | AlSi7Mg0.6 + 0.4% Fe + 0.06% Mn | 606 | 576 | 577 | 545 | 502 | 569 | --- |
| G3 | AlSi7Mg0.6 + 0.5% Fe + 0.06% Mn | 608 | 575 | 577 | 544 | 498 | 587 | --- |
| G4 | AlSi7Mg0.6 + 0.6% Fe + 0.07% Mn | 603 | 576 | 578 | 545 | 500 | 594 | --- |
| G5 | AlSi7Mg0.6 + 0.7% Fe + 0.08% Mn | 607 | 574 | 579 | 543 | 501 | 612 | --- |
| G6 | AlSi7Mg0.6 + 0.8% Fe + 0.09% Mn | 605 | 576 | 577 | 546 | 500 | 619 | --- |
| G1a | AlSi7Mg0.6 + 0.3% Fe + 0.16% Mn | 607 | 576 | 578 | 543 | 499 | --- | 581 |
| G2a | AlSi7Mg0.6 + 0.4% Fe + 0.21% Mn | 604 | 575 | 578 | 543 | 503 | --- | 589 |
| G3a | AlSi7Mg0.6 + 0.5% Fe + 0.26% Mn | 608 | 576 | 579 | 544 | 502 | --- | 592 |
| G4a | AlSi7Mg0.6 + 0.6% Fe + 0.31% Mn | 604 | 574 | 578 | 543 | 500 | --- | 596 |
| G5a | AlSi7Mg0.6 + 0.7% Fe + 0.35% Mn | 606 | 575 | 577 | 546 | 502 | --- | 599 |
| G6a | AlSi7Mg0.6 + 0.8% Fe + 0.41% Mn | 609 | 574 | 577 | 545 | 501 | --- | 600 |
| Sample | Heating | Cooling | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T, °C | E(Mg) | α + β | FeMn | α(Al) | Fe | SP | SP | Fe | α(Al) | FeMn | α + β | E(Mg) | |
| G2 | Tstart | 542 | 561 | --- | 608 | --- | --- | --- | --- | 610 | --- | 560 | 546 |
| Tend | 550 | 568 | --- | 618 | --- | --- | --- | --- | 621 | --- | 567 | 551 | |
| G4 | Tstart | 540 | 560 | --- | 607 | 588 | --- | --- | 585 | 609 | --- | 560 | 541 |
| Tend | 552 | 570 | --- | 612 | 597 | --- | --- | 599 | 614 | --- | 569 | 553 | |
| G6 | Tstart | 540 | 563 | --- | 614 | 608 | --- | --- | 610 | 617 | --- | 559 | 542 |
| Tend | 553 | 569 | --- | 620 | 618 | --- | --- | 619 | 624 | --- | 569 | 550 | |
| G2a | Tstart | 540 | 562 | 573 | 608 | --- | --- | --- | --- | 610 | 575 | 568 | 543 |
| Tend | 551 | 570 | 582 | 614 | --- | --- | --- | --- | 618 | 580 | 571 | 549 | |
| G4a | Tstart | 539 | 563 | 578 | 607 | --- | 618 | 616 | --- | 609 | 577 | 561 | 540 |
| Tend | 549 | 570 | 594 | 613 | --- | 627 | 630 | --- | 616 | 591 | 565 | 549 | |
| G6a | Tstart | 541 | 560 | 581 | 609 | --- | 623 | 620 | --- | 610 | 582 | 560 | 542 |
| Tend | 548 | 569 | 603 | 616 | --- | 640 | 639 | --- | 615 | 601 | 565 | 550 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Piatkowski, J.; Nowinska, K.; Matula, T.; Nowrot, A. What Is the Main Cause of Shrinkage Porosity in AlSi7Mg0.6 Alloy Castings Obtained with an Increased Share of Secondary Materials? Materials 2026, 19, 910. https://doi.org/10.3390/ma19050910
Piatkowski J, Nowinska K, Matula T, Nowrot A. What Is the Main Cause of Shrinkage Porosity in AlSi7Mg0.6 Alloy Castings Obtained with an Increased Share of Secondary Materials? Materials. 2026; 19(5):910. https://doi.org/10.3390/ma19050910
Chicago/Turabian StylePiatkowski, Jaroslaw, Katarzyna Nowinska, Tomasz Matula, and Andrzej Nowrot. 2026. "What Is the Main Cause of Shrinkage Porosity in AlSi7Mg0.6 Alloy Castings Obtained with an Increased Share of Secondary Materials?" Materials 19, no. 5: 910. https://doi.org/10.3390/ma19050910
APA StylePiatkowski, J., Nowinska, K., Matula, T., & Nowrot, A. (2026). What Is the Main Cause of Shrinkage Porosity in AlSi7Mg0.6 Alloy Castings Obtained with an Increased Share of Secondary Materials? Materials, 19(5), 910. https://doi.org/10.3390/ma19050910









