Synergistic Sintering of Multi-Source Petrochemical Wastes for High-Strength Ceramsite: Process Optimization and Environmental Safety
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material Characterization
2.2. Preparation of the Ceramsite Samples
2.3. Experiment Design
2.3.1. Single-Factor Experiment
2.3.2. Response Surface Optimization
2.4. Performance Characterization of OSPG
2.4.1. Basic Characterization Testing
2.4.2. Composition Characterization of OSPG
2.4.3. Morphology Characterization of OSPG
3. Results
3.1. Thermal Analysis of Raw Materials
3.2. Single-Factor Analysis
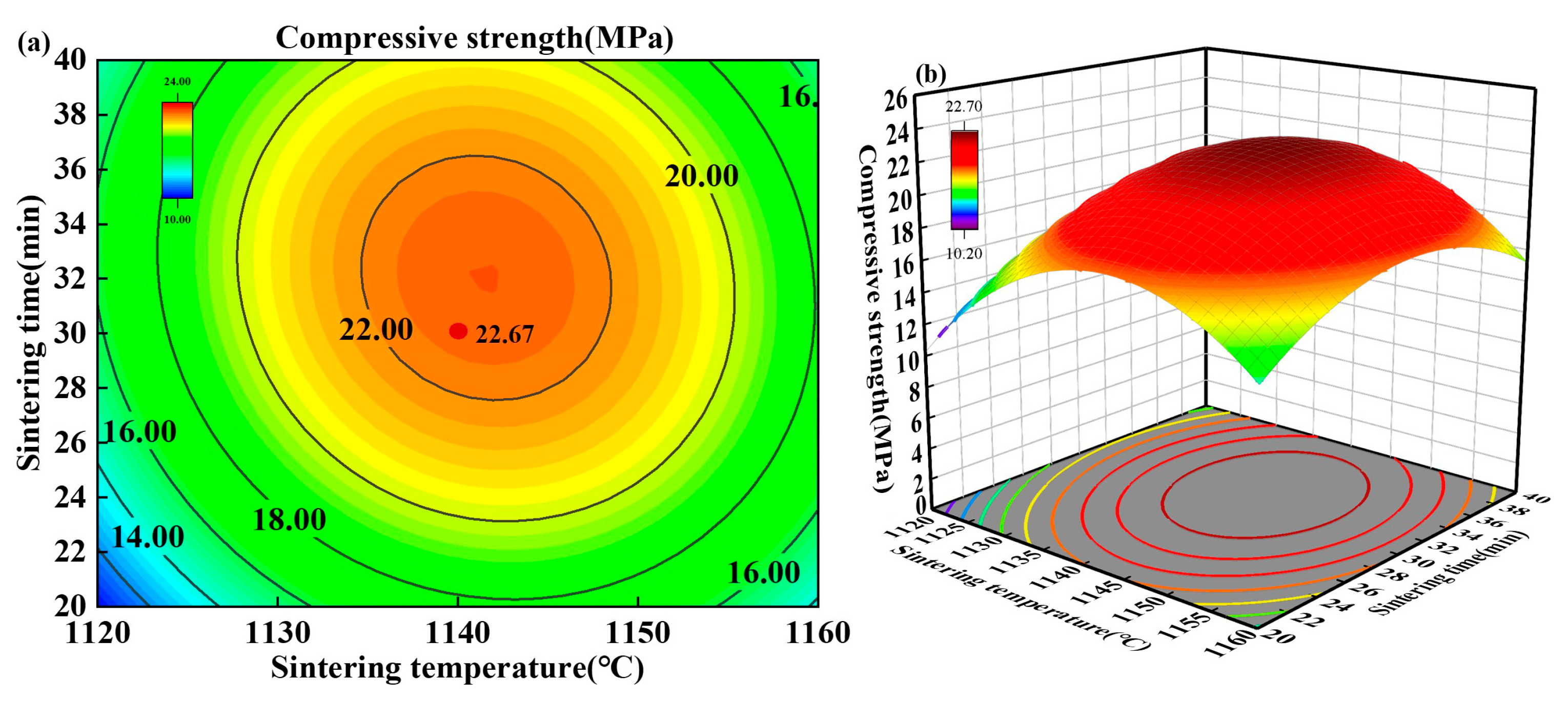
3.3. Response Surface Analysis on Strength
3.3.1. Model Construction and Significance Analysis
3.3.2. Analysis of Response Surface Interactions
3.3.3. Model Validation
3.3.4. Experimental Optimization and Verification Characterization
3.4. Leaching Toxicity Analysis
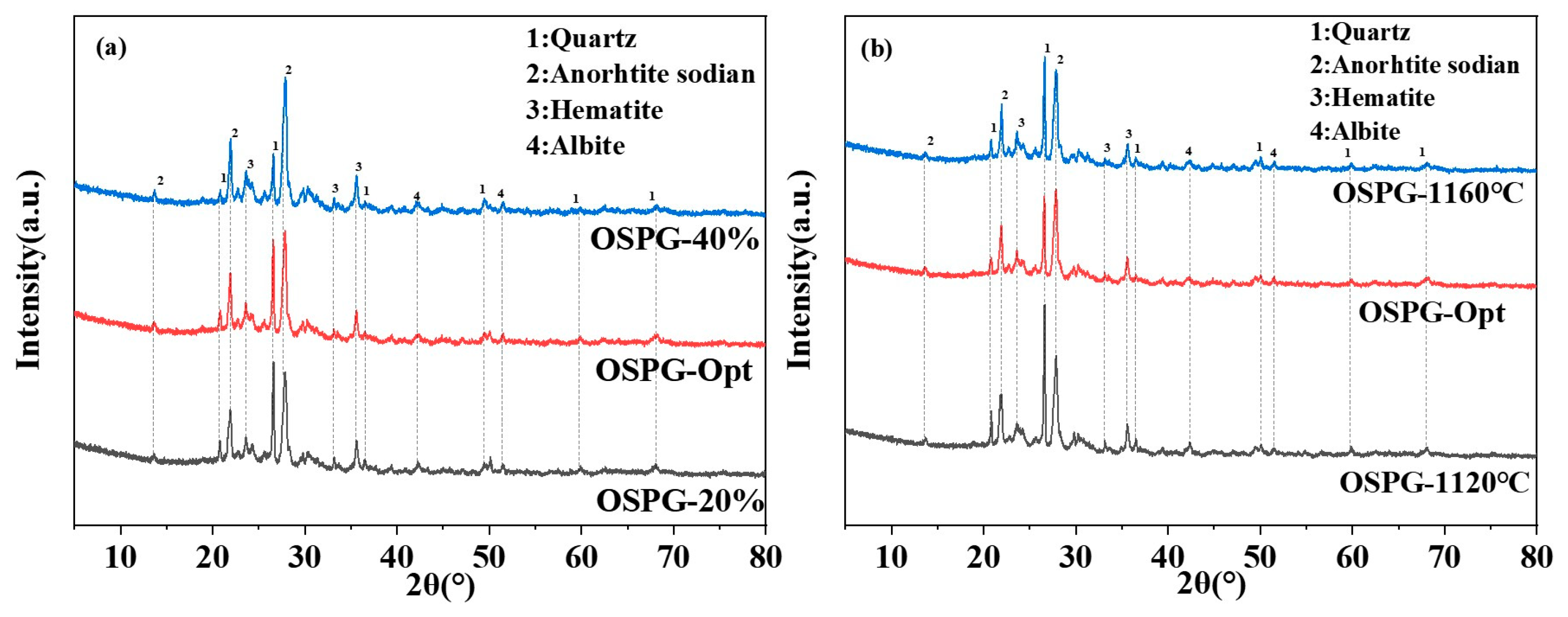
3.5. XRD Analysis
3.6. FTIR Characterization Analysis
3.7. Microstructure Analysis
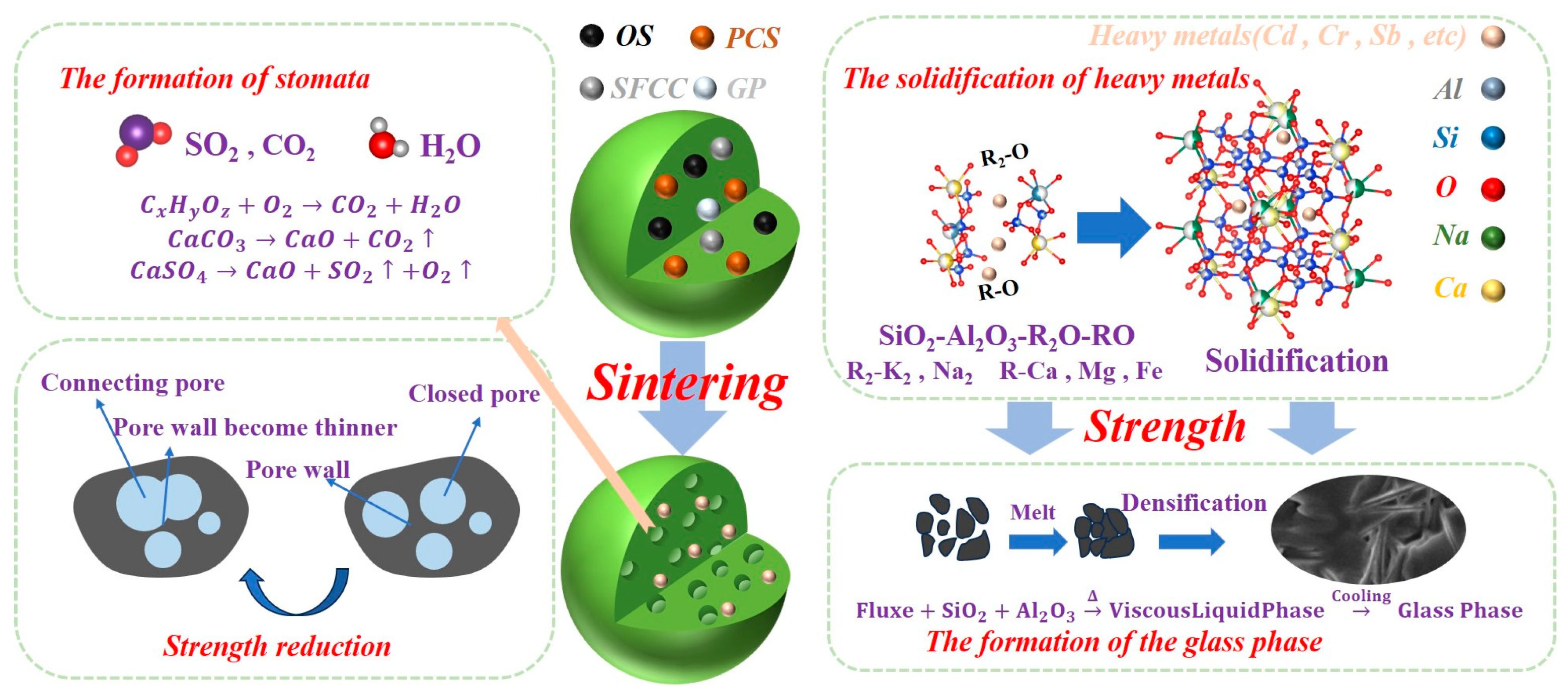
3.8. Mechanism of OSPG Formation
4. Conclusions
- Process optimization based on response surface analysis established that interactions among OS content, sintering temperature, and time critically influence particle strength. Under optimal conditions (30.5% OS, 1142 °C, 32 min), the ceramsite achieved a compressive strength of 23.12 MPa, conforming to the Chinese standard T/CSTM 00548-2022 for high-strength lightweight aggregates from solid waste.
- The ceramsite exhibits excellent environmental compatibility, with heavy metal leaching concentrations below GB 5085.3-2007 limits. Effective immobilization results from the structural incorporation of metals into the anorthite lattice and encapsulation within the dense glass phase, reinforced by a sealing surface glaze.
- Microstructural characterization identifies quartz, anorthite solid solution, hematite, and albite as key crystalline phases. Mechanical strength originates from an interlocking architecture between anorthite crystals and the glass matrix, promoted by MgO fluxing from decomposed olivine.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hu, G.; Li, J.; Hou, H. A Combination of Solvent Extraction and Freeze Thaw for Oil Recovery from Petroleum Refinery Wastewater Treatment Pond Sludge. J. Hazard. Mater. 2015, 283, 832–840. [Google Scholar] [CrossRef]
- Wang, B.; Yan, Y.; Zhou, X.; Su, H.; Zhang, H.; Zhang, J.-N.; Xu, J.; Pan, Q.; Yan, W. Mild Activation of Spent Fluid Catalytic Cracking (FCC) Catalysts for the Pilot Synthesis of Zeolite a with Commercial Quality and Excellent Co2+ Removal Ability. Chem. Eng. J. 2024, 490, 151733. [Google Scholar] [CrossRef]
- Yang, T.; Liao, Y.; Wang, M.; Zheng, Z.; Yao, M. Remediation and Resource Utilization of Petroleum-Contaminated Soil by Pyrite-Assisted Pyrolysis as Bifunctional Materials to Adsorb Heavy Metal and Activate Peroxymonosulfate Oxidation. Sci. Total Environ. 2023, 892, 164742. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, Z.; Chen, M.; Yang, J.; Yan, X. Preparation of Sintered Brick from Thermal Desorption Residue of Oily Sludge and Analysis of Environmental Performance. Constr. Build. Mater. 2023, 376, 130923. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Sun, Z. Ecotoxicological Effects of Petroleum-Contaminated Soil on the Earthworm Eisenia Fetida. J. Hazard. Mater. 2020, 393, 122384. [Google Scholar] [CrossRef]
- Chen, T.; Yan, B. Fixation and Partitioning of Heavy Metals in Slag after Incineration of Sewage Sludge. Waste Manag. 2012, 32, 957–964. [Google Scholar] [CrossRef]
- Zubaidy, E.A.H.; Abouelnasr, D.M. Fuel Recovery from Waste Oily Sludge Using Solvent Extraction. Process Saf. Environ. Prot. 2010, 88, 318–326. [Google Scholar] [CrossRef]
- Mi, H.; Yi, L.; Wu, Q.; Xia, J.; Zhang, B. Preparation of High-Strength Ceramsite from Red Mud, Fly Ash, and Bentonite. Ceram. Int. 2021, 47, 18218–18229. [Google Scholar] [CrossRef]
- Xiao, J.; Shen, J.; Bai, M.; Gao, Q.; Wu, Y. Reuse of Construction Spoil in China: Current Status and Future Opportunities. J. Clean. Prod. 2021, 290, 125742. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, X.; Zhang, X.; Zhou, Y.; Wang, T.; Xue, Y. Preparation of High-Strength Ceramsite via Co-Sintering of Shield Tunnel Muck and Steel Slag: Correlation Investigation on Phase Composition and Particle Strength. Constr. Build. Mater. 2024, 439, 137413. [Google Scholar] [CrossRef]
- Jia, G.; Wang, Y.; Yang, F.; Ma, Z. Preparation of CFB Fly Ash/Sewage Sludge Ceramsite and the Morphological Transformation and Release Properties of Sulfur. Constr. Build. Mater. 2023, 373, 130864. [Google Scholar] [CrossRef]
- Xiao, T.; Fan, X.; Zhou, C.; Wang, H.; Wu, K.; Zhou, H. Preparation of Ultra-Lightweight Ceramsite from Waste Materials: Using Phosphate Tailings as Pore-Forming Agent. Ceram. Int. 2024, 50, 15218–15229. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Li, L.H.; Zhang, J.S.; Ma, C.; Wu, X. Solidification of Oily Sludge. Pet. Sci. Technol. 2018, 36, 273–279. [Google Scholar] [CrossRef]
- Polettini, A.; Pomi, R.; Trinci, L.; Muntoni, A.; Lo Mastro, S. Engineering and Environmental Properties of Thermally Treated Mixtures Containing MSWI Fly Ash and Low-Cost Additives. Chemosphere 2004, 56, 901–910. [Google Scholar] [CrossRef]
- Guan, H.; Yu, J.; Umuhuza Kibugenza, A.S.; Sun, Q. Preparation of Coal Gangue Ceramsite High-Strength Concrete and Investigation of Its Physico-Mechanical Properties. Sci. Rep. 2022, 12, 16369. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, W.; Pan, J.; Zhu, D.; Dong, T.; Guo, Z.; Yang, C.; Li, S. Synergistic Mechanism for Preparation of High-Performance Ceramsite from Coal Gangue and Iron Ore Tailings. Process Saf. Environ. Prot. 2025, 201, 107591. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, H.; Lin, X.; Zhang, H.; Zhang, Z.; Zhang, S. Investigations on the Preparation of Ceramsite from Petrochemical Excess Sludge. Front. Chem. 2022, 10, 1008884. [Google Scholar] [CrossRef]
- Jiang, J.; Chen, S.; Jin, C.; Wang, G.; Liu, T.; Xu, T.; Lei, L.; Dang, W.; Yang, X.; Ding, T.; et al. Preparation and Properties of High-Strength Lightweight Aggregate Ceramsite from Nepheline Tailings. Constr. Build. Mater. 2023, 368, 130458. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Li, Y.; Jiao, S.-Q.; Chou, K.-C.; Zhang, G.-H. Preparation and Properties of Diopside-Based CaO-MgO-Al2O3-SiO2 Glass-Ceramic Using Biomass Fly Ash. J. Non-Cryst. Solids 2023, 600, 122015. [Google Scholar] [CrossRef]
- Long, Y.; Pu, K.; Yang, Y.; Huang, H.; Fang, H.; Shen, D.; Geng, H.; Ruan, J.; Gu, F. Preparation of High-Strength Ceramsite from Municipal Solid Waste Incineration Fly Ash and Clay Based on CaO-SiO2-Al2O3 System. Constr. Build. Mater. 2023, 368, 130492. [Google Scholar] [CrossRef]
- Matos, S.R.C.; Matos, P.R.D.; Andrade Neto, J.S.; Campos, C.E.M.; Kirchheim, A.P.; Gonçalves, J.P. Synthesis and Hydration of Ye’elimite-Containing Cement (YCC) Produced with Spent Fluid Catalytic Cracking Catalyst. Constr. Build. Mater. 2022, 359, 129364. [Google Scholar] [CrossRef]
- Ma, W.; Lv, Y.; Li, S.; He, C.; Gong, Z.; Wen, X.; Dan, J.; Li, X. Study on Thermal Behavior and Gas Pollutant Emission Control during the Co-Combustion of Waste Tire-Modified Oily Sludge and Coal. Fuel 2025, 391, 134758. [Google Scholar] [CrossRef]
- Riley, C.M. Relation of Chemical Properties to the Bloating of Clays. J. Am. Ceram. Soc. 1951, 34, 121–128. [Google Scholar] [CrossRef]
- T/CSTM 00548-2022; Light Aggregate with High Strength Prepared from Solid Waste. Zhongguancun Materials Testing Technology Alliance (CSTM): Beijing, China, 2022.
- Li, C.; Zhang, G.; Liu, D.; Wang, M. Preparation of Lightweight Ceramsite from Solid Waste Lithium Slag and Fly Ash. Constr. Build. Mater. 2023, 398, 132419. [Google Scholar] [CrossRef]
- Yashima, S.; Kanda, Y.; Sano, S. Relationships between Particle Size and Fracture Energy or Impact Velocity Required to Fracture as Estimated from Single Particle Crushing. Powder Technol. 1987, 51, 277–282. [Google Scholar] [CrossRef]
- Song, C.; Zhang, H.; Dong, Y.; Pei, L.; Liu, H.; Jiang, J.; Xu, H. Investigation on the Fabrication of Lightweight Aggregate with Acid-Leaching Tailings of Vanadium-Bearing Stone Coal Minerals and Red Mud. Chin. J. Chem. Eng. 2021, 32, 353–359. [Google Scholar] [CrossRef]
- Liu, Z.; Guo, R.; Pan, T.; Yan, Y.; Fu, C. Preparation of Ceramsite from Low-Silicon Red Mud (LSRM): Effects of Si–Al Ratio and Sintering Temperature. Ceram. Int. 2023, 49, 34191–34204. [Google Scholar] [CrossRef]
- Heide, K.; Földvari, M. High Temperature Mass Spectrometric Gas-Release Studies of Kaolinite Al2[Si2O5(OH)4] Decomposition. Thermochim. Acta 2006, 446, 106–112. [Google Scholar] [CrossRef]
- Li, P.; Luo, S.; Zhang, L.; Wang, Q.; Huang, X.; Zhang, Y.; Liu, X.; Liang, J.; Duan, X. Study on Preparation and Performance of Iron Tailings-Based Porous Ceramsite Filter Materials for Water Treatment. Sep. Purif. Technol. 2021, 276, 119380. [Google Scholar] [CrossRef]
- Wang, J.; Ma, Y.; Li, J.; Wan, X.; Zhang, M.; Zhao, Y.; Zhang, B. Preparation of Egg-Structured Ceramsites from Molybdenum Tailings with Improved Properties. Case Stud. Constr. Mater. 2024, 20, e03303. [Google Scholar] [CrossRef]
- Ouyang, J.; Zhou, Z.; Zhang, Y.; Yang, H. High Morphological Stability and Structural Transition of Halloysite (Hunan, China) in Heat Treatment. Appl. Clay Sci. 2014, 101, 16–22. [Google Scholar] [CrossRef]
- Li, G.; Zeng, J.; Luo, J.; Liu, M.; Jiang, T.; Qiu, G. Thermal Transformation of Pyrophyllite and Alkali Dissolution Behavior of Silicon. Appl. Clay Sci. 2014, 99, 282–288. [Google Scholar] [CrossRef]
- Wu, X.; Gu, F.; Su, C.; Wang, W.; Pu, K.; Shen, D.; Long, Y. Preparing High-Strength Ceramsite from Ferronickel Slag and Municipal Solid Waste Incineration Fly Ash. Ceram. Int. 2022, 48, 34265–34272. [Google Scholar] [CrossRef]
- Tong, L.; Ji, J.; Yang, J.; Qian, X.; Li, X.; Wang, H.; Zhou, S.; Wu, Y.; Zhao, Y.; Yuan, X. Sludge-Based Ceramsite for Environmental Remediation and Architecture Ingredients. J. Clean. Prod. 2024, 448, 141556. [Google Scholar] [CrossRef]
- GB 5085.3-2007; Identification Standards for Hazardous Wastes—Identification for Extraction Toxicity. State Environmental Protection Administration, General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China: Beijing, China, 2007.
- Geng, J.; Niu, S.; Han, K.; Wang, Y.; Zhu, J.; Yang, Z.; Liu, J.; Zhang, H.; Sun, X.; Liang, B.; et al. Properties of Artificial Lightweight Aggregates Prepared from Coal and Biomass Co-Fired Fly Ashes and Sewage Sludge Fly Ash. Ceram. Int. 2024, 50, 28609–28618. [Google Scholar] [CrossRef]
- Sun, X.; Niu, S.; Cheng, S.; Li, X.; Geng, J.; Liu, S.; Liu, J. Preparation of Sintered Ceramsite by Synergising Different Types of Expansion Agents with Multiple Bulk Solid Wastes. Constr. Build. Mater. 2025, 484, 141873. [Google Scholar] [CrossRef]
- Zhao, H.; Mao, Y.; Jiang, M.; Dong, X.; Wang, T.; Xue, Y. Sustainable Production of Lightweight Ceramics from Shield Tunnel Muck and Phosphorus Tailings Enhanced by CaO/SiO2 Ratios. Constr. Build. Mater. 2025, 482, 141738. [Google Scholar] [CrossRef]
- Mohaček-Grošev, V.; Furić, K.; Vujnović, V. Raman Study of Water Deposited in Solid Argon Matrix. Spectrochim. Acta Part A 2022, 269, 120770. [Google Scholar] [CrossRef]
- Pei, J.; Pan, X.; Qi, Y.; Yu, H.; Tu, G. Preparation and Characterization of Ultra-Lightweight Ceramsite Using Non-Expanded Clay and Waste Sawdust. Constr. Build. Mater. 2022, 346, 128410. [Google Scholar] [CrossRef]
- Mao, W.; Ma, H.; Wang, B. Mild Ring-Opening Coupling of Liquid-Phase Cyclohexane to Diesel Components Using Sulfated Metal Oxides. J. Hazard. Mater. 2010, 176, 361–366. [Google Scholar] [CrossRef]
- Wu, Q.; Song, Y.; Wang, Q.; Liu, W.; Hao, L.; Wang, Z.; Wang, C. Combination of Magnetic Solid-Phase Extraction and HPLC-UV for Simultaneous Determination of Four Phthalate Esters in Plastic Bottled Juice. Food Chem. 2021, 339, 127855. [Google Scholar] [CrossRef]
- Zhang, Z.; Qiao, X.; Yu, J. Microwave Selective Heating-Enhanced Reaction Rates for Mullite Preparation from Kaolinite. RSC Adv. 2014, 4, 2640–2647. [Google Scholar] [CrossRef]
- Espinosa-Navarro, A.T.; Del Díaz-Nava, M.C.; Alvarado-Pérez, Y.; Muro-Urista, C.; Sánchez, J.J.G. Polymer-Cellulosic-Clay and Polymer-Cellulosic-Zeolitic Biocomposites for the Removal of Methylene Blue in Aqueous Solution. MRS Adv. 2019, 4, 3417–3421. [Google Scholar] [CrossRef]
- Khan, I.; Kuzmann, E.; Nomura, K.; Ali, A.S.; Akiyama, K.; Homonnay, Z.; Sinkó, K.; Pavić, L.; Kubuki, S. Structural Characterization, Electrical and Photocatalytic Properties of A−and γ-Fe2O3 Nanoparticles Dispersed in Iron Aluminosilicate Glass. J. Non-Cryst. Solids 2021, 561, 120756. [Google Scholar] [CrossRef]
- Zhang, L.; He, Y.; Lü, P.; Peng, J.; Li, S.; Chen, K.; Yin, S.; Zhang, L. Comparison of Microwave and Conventional Heating Routes for Kaolin Thermal Activation. J. Cent. South Univ. 2020, 27, 2494–2506. [Google Scholar] [CrossRef]
- Peng, C.; Dai, G.; Wang, Y.; Yang, J.; Wang, C.; Jiao, S.; Chen, L.; Duan, C.; Li, P. Preparation of High-Strength Ceramsite from Coal Gangue and Printing and Dyeing Sludge: Design Strategy and Modelling Mechanism. Ceram. Int. 2024, 50, 19963–19970. [Google Scholar] [CrossRef]
- Wang, C.; Wang, Y.; Sun, W.; Wang, L.; Huang, D. Thermodynamic Assessment of Iron and the Expansion Behavior of Porous Ceramsite in Pure Mineral Systems Relating to Solid Waste Components. Ceram. Int. 2022, 48, 32867–32876. [Google Scholar] [CrossRef]
- Shi, Y.; Guo, W.; Jia, Y.; Xue, C.; Qiu, Y.; Zhao, Q.; Wang, D. Preparation of Non-Sintered Lightweight Aggregate Ceramsite Based on Red Mud-Carbide Slag-Fly Ash: Strength and Curing Method Optimization. J. Clean. Prod. 2022, 372, 133788. [Google Scholar] [CrossRef]
- Zhao, S.; Wu, J.; Xu, J.; Li, S.; Qin, J.; Liao, D.; Weng, R. Structure and Properties of High-Strength Lightweight Ceramsite Prepared by Synergistic Sintering of Iron Tailings, Inorganic Sludge, and Recycled Micro-Powder. Mater. Today Commun. 2025, 48, 113598. [Google Scholar] [CrossRef]
- Cheng, G.; Li, Q.; Su, Z.; Sheng, S.; Fu, J. Preparation, Optimization, and Application of Sustainable Ceramsite Substrate from Coal Fly Ash/Waterworks Sludge/Oyster Shell for Phosphorus Immobilization in Constructed Wetlands. J. Clean. Prod. 2018, 175, 572–581. [Google Scholar] [CrossRef]











| Chemical Composition (wt.%) | OS | PCS | SFCC | GP |
|---|---|---|---|---|
| SiO2 | 24.31 | 56.91 | 37.61 | 71.57 |
| Al2O3 | 21.81 | 23.20 | 54.73 | 1.66 |
| Fe2O3 | 15.01 | 3.47 | 0.47 | 0.12 |
| Na2O | 1.26 | 1.84 | 0.30 | 14.96 |
| K2O | 0.69 | 4.49 | 0.16 | 0.21 |
| CaO | 15.75 | 2.26 | 0.16 | 7.46 |
| P2O5 | 2.04 | 0.13 | 0.56 | 0.02 |
| MgO | 1.88 | 0.71 | 0.94 | 3.59 |
| SO3 | 13.48 | / | 0.22 | 0.21 |
| Sample | OS (%) | PCS (%) | SFCC (%) | GP (%) | H2O (%) | SiO2 (wt.%) | Al2O3 (wt.%) |
|---|---|---|---|---|---|---|---|
| A1 | 20 | 50 | 10 | 20 | 30 | 51.39 | 21.77 |
| A2 | 30 | 40 | 10 | 20 | 27 | 48.13 | 21.63 |
| A3 | 40 | 30 | 10 | 20 | 24 | 44.87 | 21.49 |
| A4 | 50 | 20 | 10 | 20 | 21 | 41.61 | 21.35 |
| A5 | 60 | 10 | 10 | 20 | 18 | 38.35 | 21.21 |
| Encoding | Design Variables | Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| A | OS concentration (%) | 20 | 30 | 40 |
| B | Sintering temperature (°C) | 1120 | 1140 | 1160 |
| C | Sintering time (min) | 20 | 30 | 40 |
| Test Number | OS Concentration (%) | Sintering Temperature (°C) | Sintering Time (min) | Compressive Strength (MPa) |
|---|---|---|---|---|
| A | B | C | ||
| C1 | 20 | 1120 | 30 | 10.16 |
| C2 | 30 | 1140 | 30 | 22.57 |
| C3 | 30 | 1160 | 40 | 15.83 |
| C4 | 30 | 1140 | 30 | 23.22 |
| C5 | 40 | 1160 | 30 | 13.25 |
| C6 | 20 | 1140 | 20 | 12.60 |
| C7 | 30 | 1140 | 30 | 22.75 |
| C8 | 40 | 1140 | 40 | 15.85 |
| C9 | 30 | 1160 | 20 | 13.89 |
| C10 | 20 | 1160 | 30 | 13.09 |
| C11 | 30 | 1140 | 30 | 22.42 |
| C12 | 30 | 1140 | 30 | 22.32 |
| C13 | 40 | 1120 | 30 | 12.88 |
| C14 | 40 | 1140 | 20 | 14.22 |
| C15 | 30 | 1120 | 40 | 14.79 |
| C16 | 30 | 1120 | 20 | 9.93 |
| C17 | 20 | 1140 | 40 | 15.70 |
| Source | F-Value | p-Value | |
|---|---|---|---|
| Model | 168.82 | <0.0001 | significant |
| A | 12.18 | 0.0101 | |
| B | 38.79 | 0.0004 | |
| C | 74.86 | <0.0001 | |
| AB | 7.38 | 0.0299 | |
| AC | 2.43 | 0.1627 | |
| BC | 9.60 | 0.0173 | |
| A2 | 412.67 | <0.0001 | |
| B2 | 604.84 | <0.0001 | |
| C2 | 219.18 | <0.0001 | |
| Lack of Fit | 2.79 | 0.1738 | not significant |
| Factor | A | B | C | Compressive Strength (MPa) |
|---|---|---|---|---|
| Predicted | 30.5% | 1142 °C | 32 min | 22.85 |
| Actual | 23.12 | |||
| Relative deviation (%) | - | - | - | 1.18 |
| Indexes | OSPG-Opt | Limit Value |
|---|---|---|
| Cylinder compressive strength (MPa) | 15.58 | ≥10.5 |
| Bulk density (kg/m3) | 1012.50 | ≤1200 |
| Apparent density (kg/m3) | 2089.02 | / |
| Water absorption (%) | 1.61 | ≤10 |
| Softening coefficient | 0.83 | ≥0.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, Y.; Wang, T.; Dang, J.; Liu, S.; Hu, J.; Xue, Y. Synergistic Sintering of Multi-Source Petrochemical Wastes for High-Strength Ceramsite: Process Optimization and Environmental Safety. Materials 2026, 19, 787. https://doi.org/10.3390/ma19040787
Liu Y, Wang T, Dang J, Liu S, Hu J, Xue Y. Synergistic Sintering of Multi-Source Petrochemical Wastes for High-Strength Ceramsite: Process Optimization and Environmental Safety. Materials. 2026; 19(4):787. https://doi.org/10.3390/ma19040787
Chicago/Turabian StyleLiu, Yang, Teng Wang, Jiayan Dang, Siwei Liu, Jiawei Hu, and Yongjie Xue. 2026. "Synergistic Sintering of Multi-Source Petrochemical Wastes for High-Strength Ceramsite: Process Optimization and Environmental Safety" Materials 19, no. 4: 787. https://doi.org/10.3390/ma19040787
APA StyleLiu, Y., Wang, T., Dang, J., Liu, S., Hu, J., & Xue, Y. (2026). Synergistic Sintering of Multi-Source Petrochemical Wastes for High-Strength Ceramsite: Process Optimization and Environmental Safety. Materials, 19(4), 787. https://doi.org/10.3390/ma19040787





