Development and Application of Carbon Deposition State Diagram for H-C-O Systems
Abstract
1. Introduction
2. Carbon Deposition State in the H-C-O System
2.1. Introduction to H-C-O System Mass Balance and Chemical Equilibrium Diagram
2.2. Introduction to Carbon Deposition State Diagram for H-C-O System
- (1)
- The H2/CO molar ratio in H2RG is defined according to the conditions of the dry reforming (DR) process:
- (2)
- The gas phase composition satisfies the normalization condition, with the sum of mole fractions equal to unity:
- (3)
- With the total system pressure specified as a parameter:
3. Verification of “Carbon Deposition State Diagram for H-C-O System”
3.1. Example I for the Prediction and Experimental Verification of Carbon Deposition Behavior of the System Under Different Process Conditions Based on “Carbon Deposition State Diagram for H-C-O System”
3.2. Example II for the Prediction and Experimental Verification of Carbon Deposition Behavior of the System Under Different Process Conditions Based on “Carbon Deposition State Diagram for H-C-O System”
3.3. Example III for the Prediction and Experimental Verification of Carbon Deposition Behavior of the System During the CH4 Reforming Process Based on “Carbon Deposition State Diagram for H-C-O System”
4. Applications of “Carbon Deposition State Diagram for H-C-O System”
4.1. Efforts to Reduce Energy Consumption and Cost of Reforming Unit in Hydrogen Production Process from Natural Gas
4.2. Carbon Deposition Behavior in COG-Based Shaft Furnace Direct Reduction
5. Conclusions
- (1)
- Within the H-C-O elemental framework, hydrogen-rich reducing gas compositions occupy a well-defined thermodynamic domain governed by mass balance and equilibrium constraints. This representation clarifies the position of a given gas mixture within the overall compositional space. Based on this framework, practical adjustment strategies can be formulated. These include adding oxidizing components or coordinating multiple gas sources to reach target compositions while preserving thermodynamic consistency.
- (2)
- Carbon formation in multi-component H-C-O systems does not occur randomly. Instead, it emerges when the gas composition crosses specific boundaries in O/C–H/C space under given temperature, pressure, and H2/CO ratio conditions. These boundaries separate stable operating regions from deposition-prone regimes and offer a coherent thermodynamic interpretation of carbon behavior in gas mixtures typical of direct reduction and hydrogen production processes.
- (3)
- The validity of these thermodynamic boundaries is supported by both experimental observations and industrial operating data. When implemented in a computational tool, the framework becomes straightforward to apply in practice. It helps operators recognize safe operating windows, respond to compositional fluctuations, and maintain stable operation. In this way, the proposed approach contributes to improved energy efficiency, lower operating costs, and enhanced process reliability in natural gas-based hydrogen-related systems.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dong, Y.; Qiao, X.X.; Liu, G.H.; Jia, J.N.; Geng, Z.R.; Zhao, Q.L.; Qi, Q.; Zhang, Y.F. Research Situation of Reduction Gas Used in Gas-based Direct Reduction Iron Technology. Energy Energy Conserv. 2016, 3, 2–4. [Google Scholar]
- Liu, X.K.; Jin, F.; Fu, Y.P.; Chen, S.H.; Cheng, L.; Lin, Q.L.; Yu, C.C. Research on Production Technology of Reducing Gas for Gas-based Direct Reduced. Shanxi Metall. 2023, 46, 109–112. [Google Scholar]
- Liu, J.Z.; Xue, Q.G.; Zhang, S.Y.; She, X.F.; Wang, J.S. Carbon deposition reaction based on heating recycling gas of oxygen blast furnaces. Chin. J. Eng. 2014, 36, 592–596. [Google Scholar]
- Han, Y.H.; Wang, J.S.; Li, Y.Z.; She, X.F.; Kong, L.T.; Xue, Q.G. Comprehensive mathematical model of top gas recycling-oxygen blast furnaces. Chin. J. Eng. 2011, 33, 1280–1286. [Google Scholar]
- Shi, M.L.; Zhao, L.D.; Deng, X.J.; Wang, J.S.; Zuo, H.B.; Xue, Q.G. Research progress on catalytic methane reforming process. CIESC J. 2024, 75, 25–39. [Google Scholar]
- Huang, X.; Lv, Z.G.; Ma, Q.; Wang, H.; Yao, X.; Liu, Y.; Wang, B.; Shuai, Y. The mechanism characterizations of methane steam reforming under coupling condition of temperature and ratio of steam to carbon. Int. J. Hydrogen Energy 2023, 48, 21586. [Google Scholar] [CrossRef]
- Chen, J.; Su, M.; Zhang, X.B.; Ji, C.M.; Zhang, M.S.; Qiao, S. Research progress in preparation of reducing gases for hydrogen metallurgy. China Metallurgy 2023, 33, 24. [Google Scholar]
- De Medeiros, F.G.M.; Lopes, F.W.B.; Rego de Vasconcelos, B. Prospects and Technical Challenges in Hydrogen Production through Dry Reforming of Methane. Catalysts 2022, 12, 363. [Google Scholar] [CrossRef]
- Su, T.M.; Wang, C.S.; Gong, B.; Qin, Z.Z.; Ji, H.B. Research progress in regulation of carbon deposits on Ni-based catalysts for dry reforming of methane. Low-Carbon Chem. Chem. Eng. 2023, 48, 1–9. [Google Scholar]
- Huang, Z.A.; Zhou, Y.X.; Zhang, K.K.; Liu, M.Y.; Yang, M.K.; Zhan, J.J.; Liu, T.; Zhou, Y. Research progress on methane pyrolysis process for hydrogen and carbon materials. Low-Carbon Chem. Chem. Eng. 2024, 49, 1–10. [Google Scholar]
- Che, Y.M.; Cao, L.X.; Liu, J.Z. Large scale hydrogen production and its application and prospect in iron and steel industry. China Metallurgy 2022, 32, 1–7. [Google Scholar]
- Wang, X.Y.; Li, J.X.; Zhou, G.B.; Wang, H.Z.; Zhang, J.; Pan, L.W. Preparation of Ni/γ-Al2O3/Monolith catalyst and its capability for methane autothermal reforming. Petrochem. Technol. 2023, 52, 615–621. [Google Scholar]
- Shi, K.Y.; Xu, H.Y.; Fan, Y.M.; Shang, Y.C.; Xu, G.L. Study on the Conversion of Natural Gas and Carbon Dioxide to Syngas. Part V: Characteristics of Methane Dehydrogenation Carbon Deposition Reaction. J. Mol. Catal. 1996, 10, 41–47. [Google Scholar]
- Yu, C.C.; Li, R.J.; Wang, W.; Liu, Q.J. Study of carbon deposition controlling over CO2/CH4 dry reforming catalyst. Petrochem. Technol. 2020, 49, 925–930. [Google Scholar]
- Zeng, Q.S. Thermodyn. Minim. Steam—Carbon Ratio Steam Reforming Light Hydrocarbons. Chem. Eng. 1994, 22, 43–45. [Google Scholar]
- Qian, H.L.; Ran, J.L.; He, A.B.; Zhang, A.H.; Liu, Z.H.; Du, J.; Tao, C.Y. Thermodynamic analysis of carbon dioxide-methane dry reforming and its carbon deposition control. Low-Carbon Chem. Chem. Eng. 2023, 48, 55–61. [Google Scholar]
- Challiwala, M.S.; Ghouri, M.M.; Linke, P.; El-Halwagi, M.M.; Elbashir, N.O. A combined thermo-kinetic analysis of various methane reforming technologies: Comparison with dry reforming. J. CO2 Util. 2017, 17, 99–111. [Google Scholar] [CrossRef]
- Chaudhary, P.K.; Deo, G. Process and catalyst improvements for the dry reforming of methane. Chem. Eng. Sci. 2023, 276, 118767. [Google Scholar] [CrossRef]
- Li, Z.Q.; Wang, H.; Li, K.Z. Research progress on hydrogen production technology from coke oven gas. Clean Coal Technol. 2023, 29, 31–42. [Google Scholar]
- Minh, D.P.; Pham, X.H.; Siang, T.J.; Vo, N. Review on the catalytic tri-reforming of methane—Part I: Impact of operating conditions, catalyst deactivation and regeneration. Appl. Catal. A Gen. 2021, 621, 118–202. [Google Scholar]
- Shen, F.M.; Zheng, A.J.; Zheng, H.Y.; Wang, X.A.; Li, Y.; Zhang, W.L. Thoughts on preparation of hydrogen-based reduction gas and process of direct reduction iron. Iron Steel 2022, 57, 10–15. [Google Scholar]
- Shen, F.M. Development of H-C-O system mass balance and chemical equilibrium diagram. Iron Steel 2023, 58, 12–17. [Google Scholar]
- Shen, F.M.; Long, F.; Zhang, W.L.; Wang, S.; Zheng, H.Y.; Shen, Y.S. Thermodynamic analysis of carbon deposition in the preparation of hydrogen-rich reducing gas for direct reduction processes. Fuel 2023, 349, 128739. [Google Scholar] [CrossRef]
- Shen, M. A Method for Determining Process Parameters for Preparing Hydrogen-Based Reducing Gas. China Patent ZL202110727435.6, 29 June 2021. [Google Scholar]
- Shen, M. Determination of Process Parameters for Preparing Hydrogen-Based Reducing Gas based on the Control of the Carbon Deposition. China Patent CN202210932708.5, 4 August 2022. [Google Scholar]
- Shen, F.M.; Zheng, H.Y.; Nie, X.S.; Ding, Z.M.; Zhang, Y.; Wang, S.; Pan, X.Y.; Zhang, G.P. Method, Apparatus, Storage Medium, and Computer Device for Determining Carbon Deposition State for H-C-O System. China Patent CN202410517733.6, 4 August 2024. [Google Scholar]
- Wang, S.; Shen, F.M.; Zhang, Y.; Pan, X.Y.; Zhang, W.L.; Zheng, H.Y.; Shen, Y.S. Gas composition and carbon deposition in a shaft furnace using coke oven Gas: A Lab experimental study. Fuel Part D 2026, 406, 137131. [Google Scholar] [CrossRef]
- H-C-O system carbon deposition state calculation special software (V1.2). Softw. Copyr. 2024, 2024SR1164467.




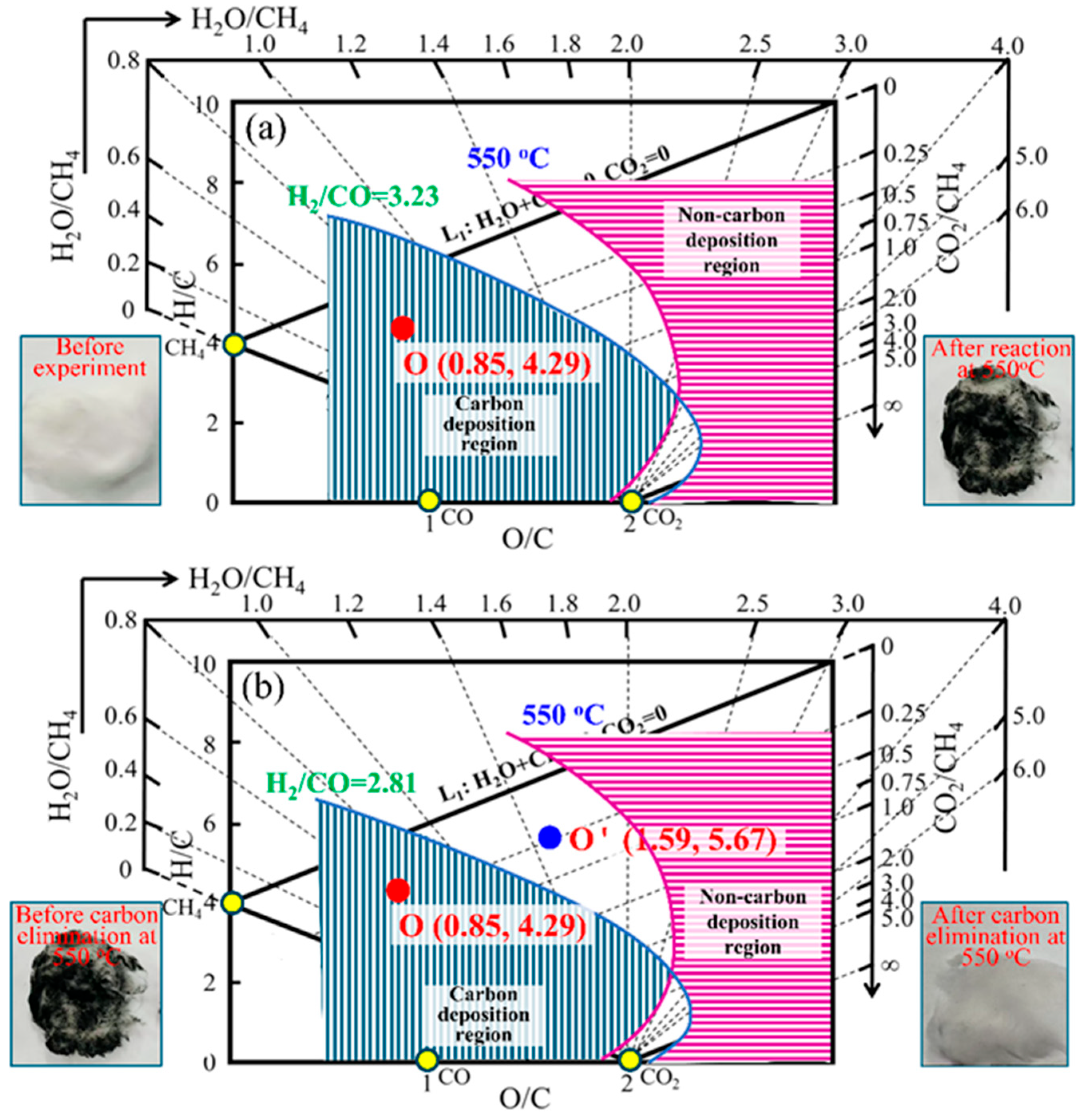
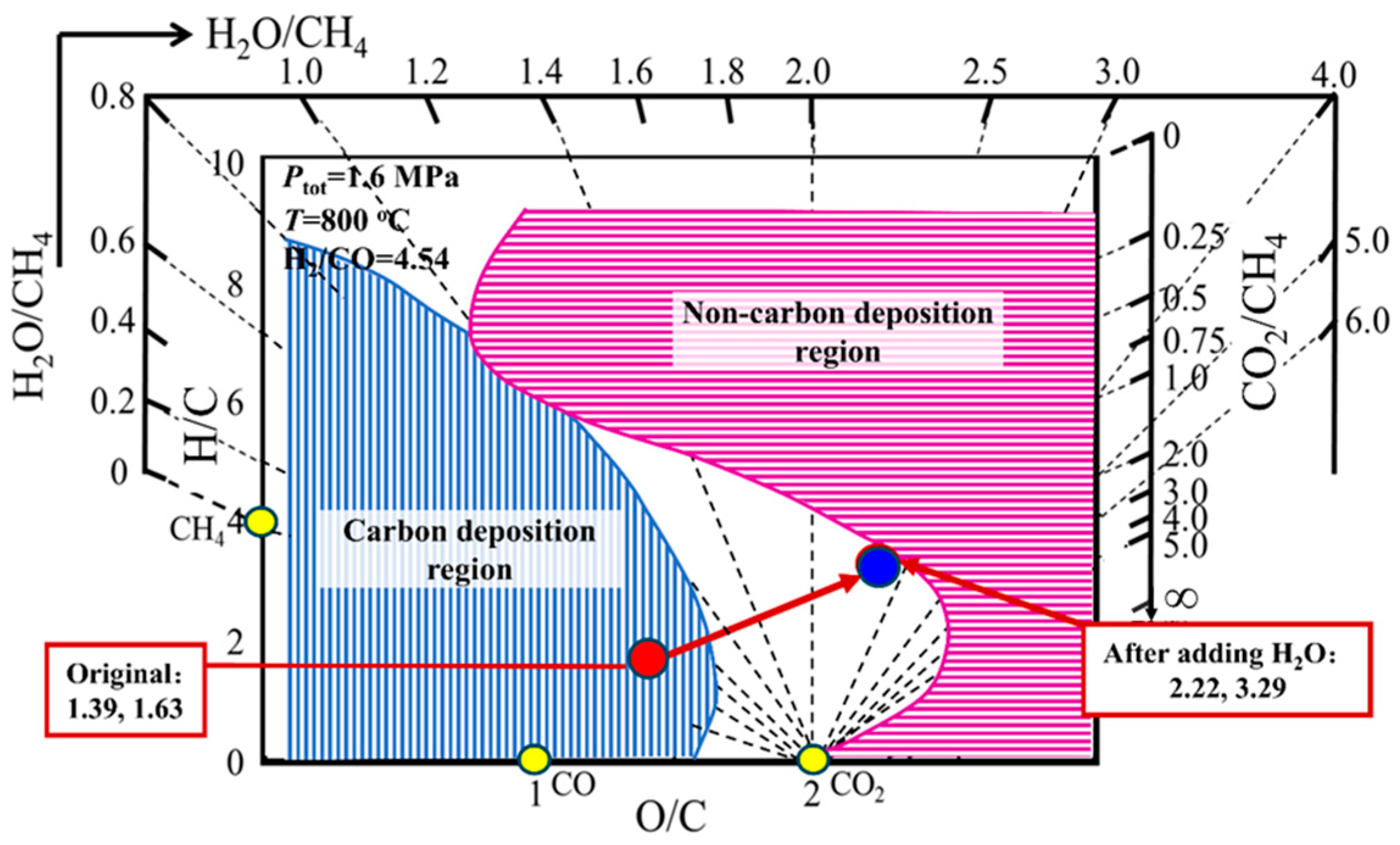
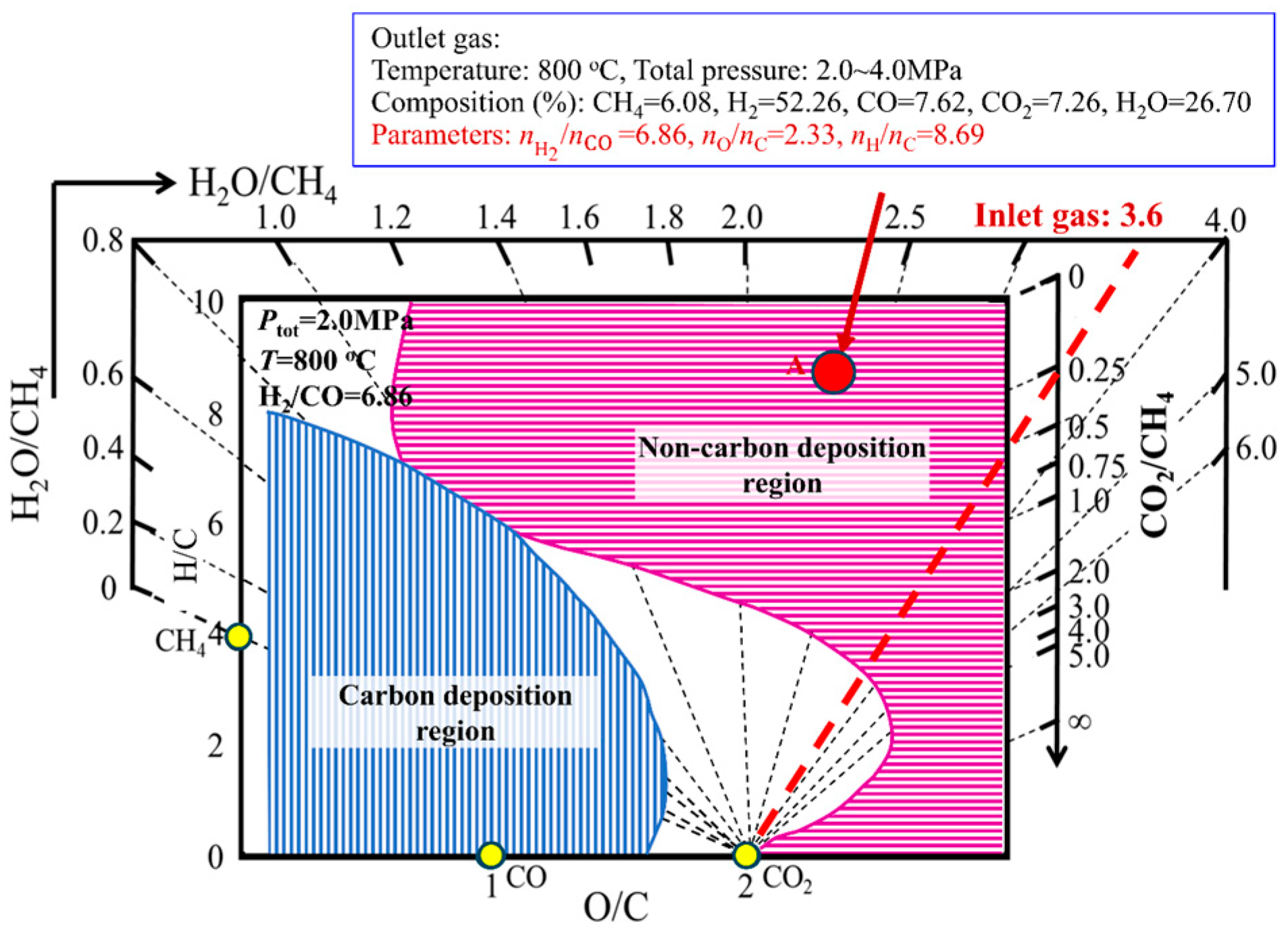

| Composition, % | Pressure, MPa | Temperature, °C | O/C, - | H/C | H2/CO | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO | CH4 | N2 | CO2 | H2O | Content | ||||||
| Mixture (Inlet) | 20.22 | 4.45 | 21.58 | 2 | 51.76 | 0.00 | 100.01 | 1.6 | 800~900 | 1.39 | 1.63 | 4.54 |
| Mixture (H2O/CH4 = 3) | 20.22 | 4.45 | 21.58 | 2 | 51.76 | 64.74 | 164.75 | 1.6 | 800~900 | 2.22 | 3.29 | 4.54 |
| Composition | H2, % | CH4, % | CO, % | CO2, % | H2O, % | N2, % | Sum, % | O/C | H/C | H2/CO |
|---|---|---|---|---|---|---|---|---|---|---|
| Inlet gas | 46.8 | 20.2 | 9.2 | 2.4 | 5.1 | 16.2 | 99.9 | 0.60 | 5.81 | 5.09 |
| Outlet gas | 37.4 | 12.8 | 8.4 | 6.1 | 20.4 | 14.7 | 99.8 | 1.50 | 6.11 | 4.45 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ding, Z.; Pan, X.; Zhang, Y.; Wang, S.; Zheng, H.; Shen, F. Development and Application of Carbon Deposition State Diagram for H-C-O Systems. Materials 2026, 19, 648. https://doi.org/10.3390/ma19040648
Ding Z, Pan X, Zhang Y, Wang S, Zheng H, Shen F. Development and Application of Carbon Deposition State Diagram for H-C-O Systems. Materials. 2026; 19(4):648. https://doi.org/10.3390/ma19040648
Chicago/Turabian StyleDing, Zhimin, Xiangyang Pan, Yan Zhang, Shuo Wang, Haiyan Zheng, and Fengman Shen. 2026. "Development and Application of Carbon Deposition State Diagram for H-C-O Systems" Materials 19, no. 4: 648. https://doi.org/10.3390/ma19040648
APA StyleDing, Z., Pan, X., Zhang, Y., Wang, S., Zheng, H., & Shen, F. (2026). Development and Application of Carbon Deposition State Diagram for H-C-O Systems. Materials, 19(4), 648. https://doi.org/10.3390/ma19040648





