A Review on Graphene (GN) and Graphene Oxide (GO) Based Biodegradable Polymer Composites and Their Usage as Selective Adsorbents for Heavy Metals in Water
Abstract
1. Introduction
2. Graphene and Graphene Oxide
2.1. Graphene
2.1.1. The Morphology of Graphene
2.1.2. Water Absorption of Graphene
2.1.3. Graphene as an Adsorbent for Heavy Metals in Water
2.2. Graphene Oxide
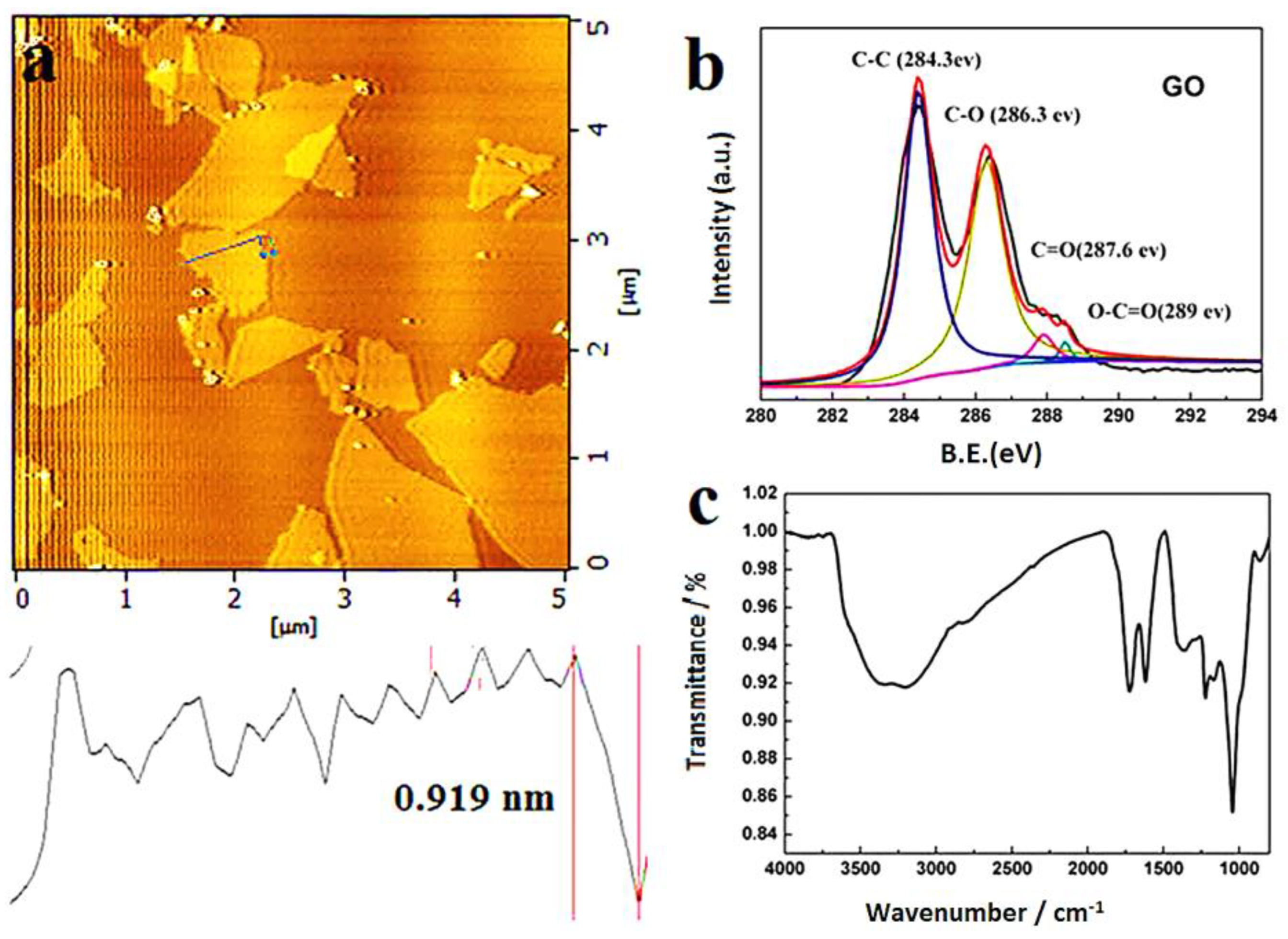
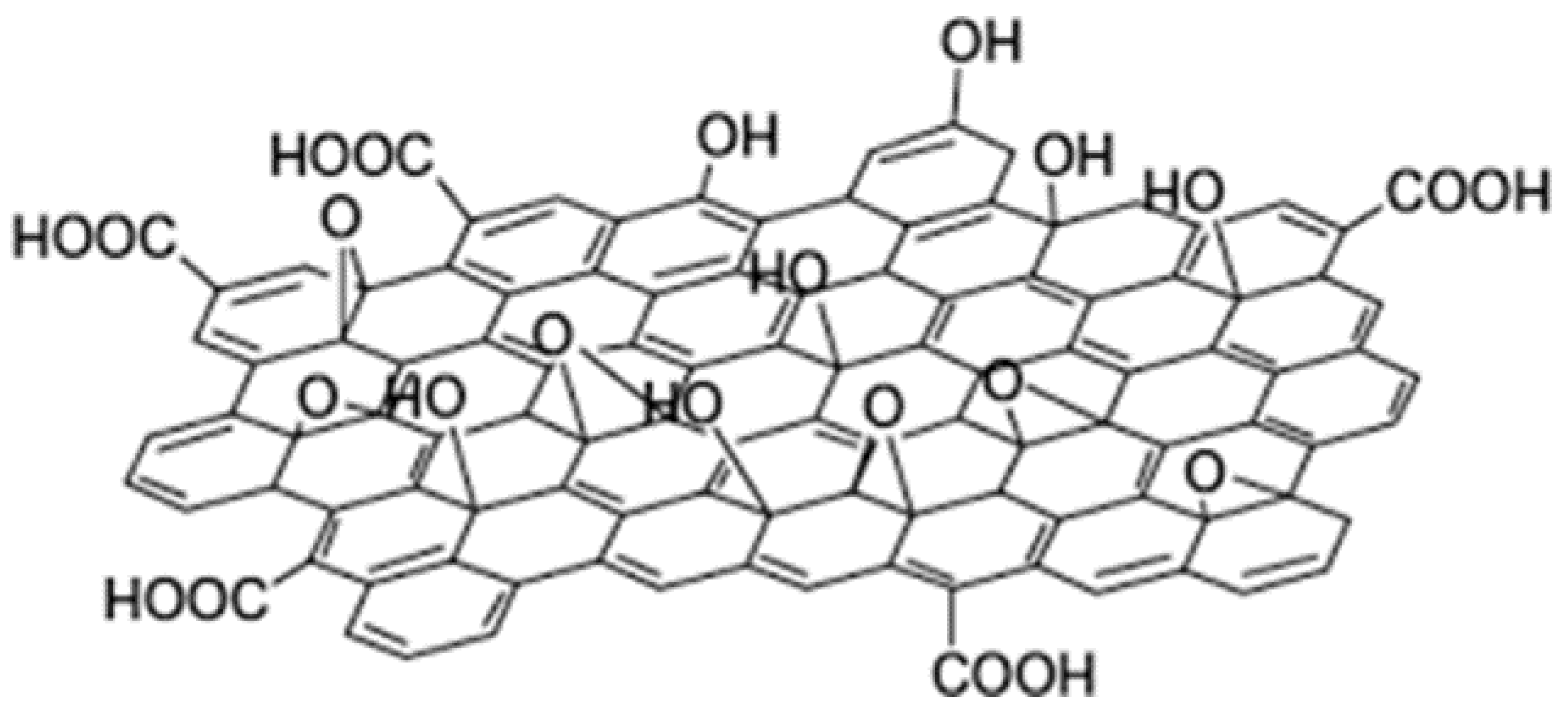
2.2.1. The Morphology of Graphene Oxide
2.2.2. Thermal Properties of Graphene Oxide
2.2.3. Water Absorption Capabilities of Graphene Oxide
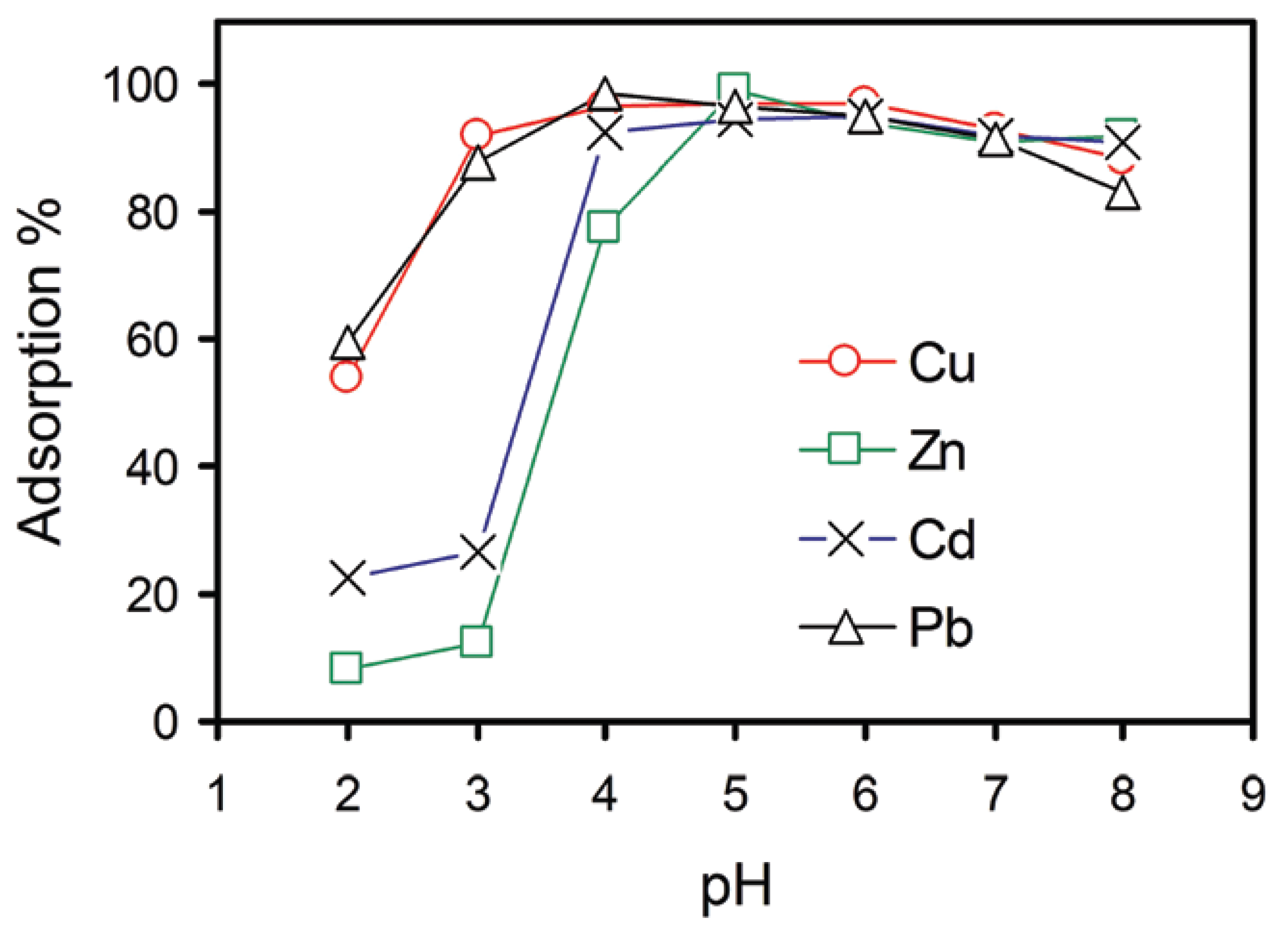
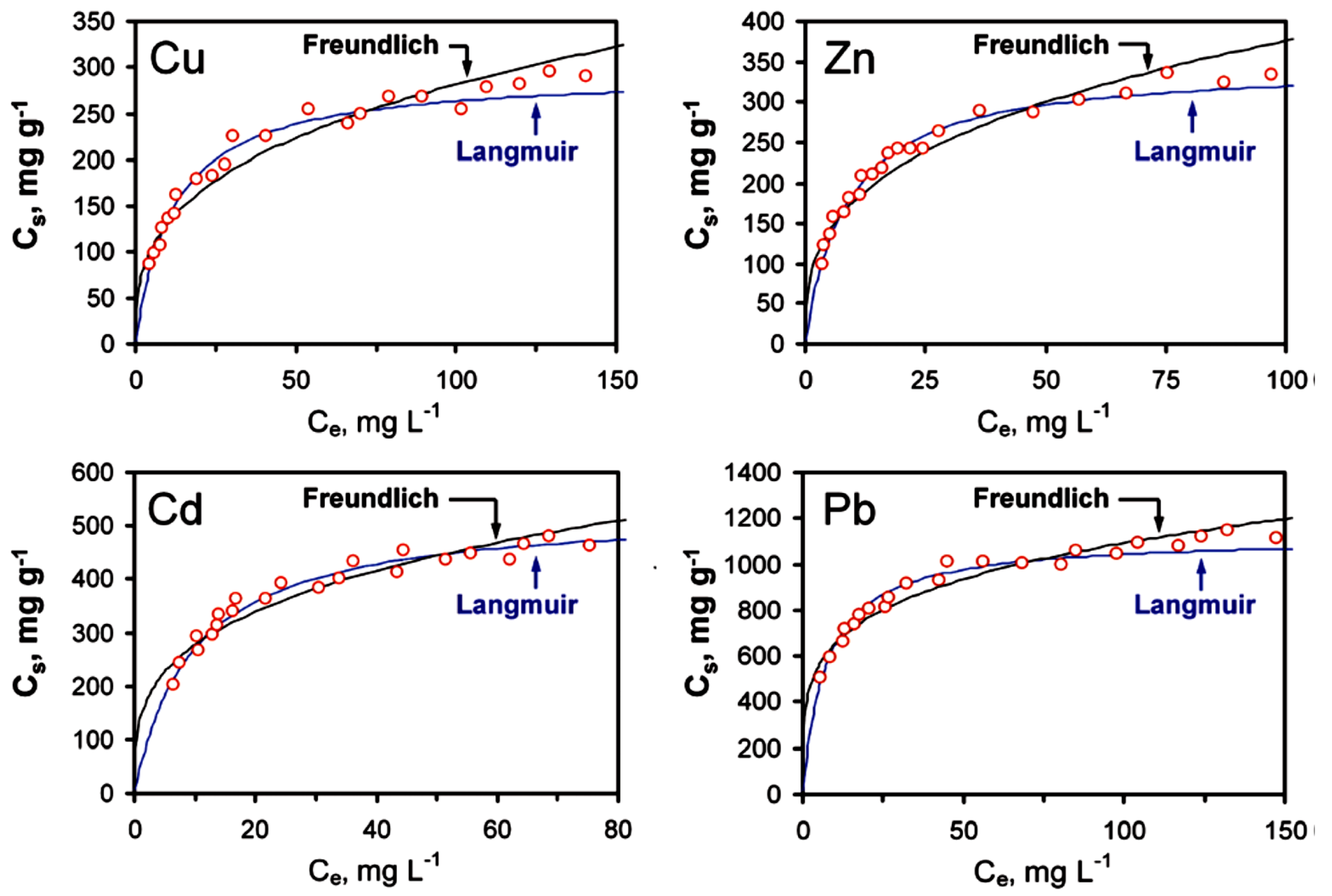
2.2.4. Graphene Oxide as an Adsorbent for Heavy Metals in Water
3. Graphene and Graphene Oxide-Based Polymer Composites
3.1. Graphene/Polymer Composites
3.1.1. The Morphology of Graphene/Polymer Composites
3.1.2. Thermal Properties of Graphene/Polymer Composites
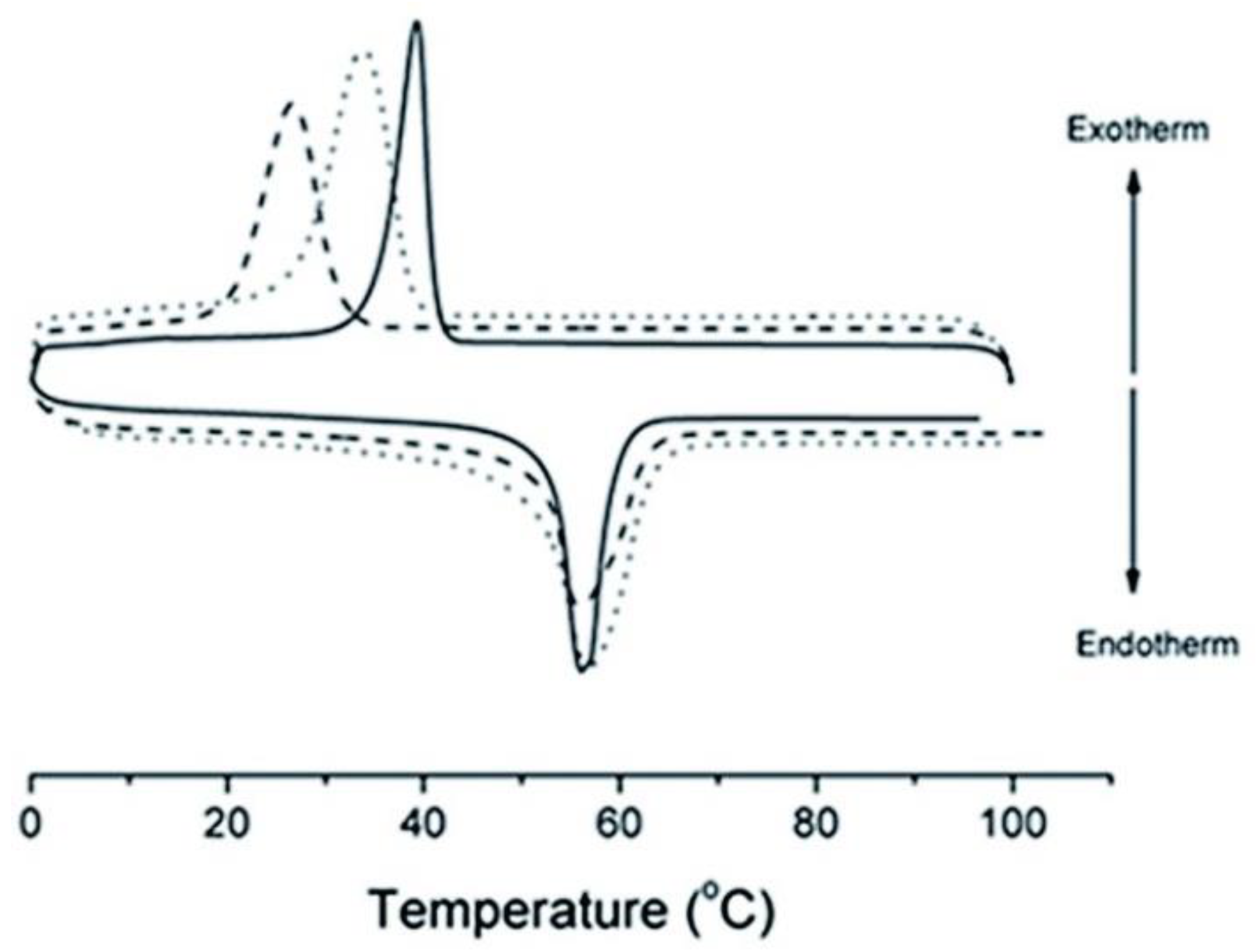
Melting and Crystallization
Thermal Degradation/Stability
3.1.3. Water Absorption Capabilities of Graphene/Polymer Composites
3.1.4. Graphene/Polymer Composites as Adsorbents for Heavy Metals in Water
3.2. Graphene Oxide/Polymer Composites
3.2.1. The Morphology of GO/Polymer Composites
3.2.2. Thermal Properties of GO/Polymer Composites
Melting and Crystallization
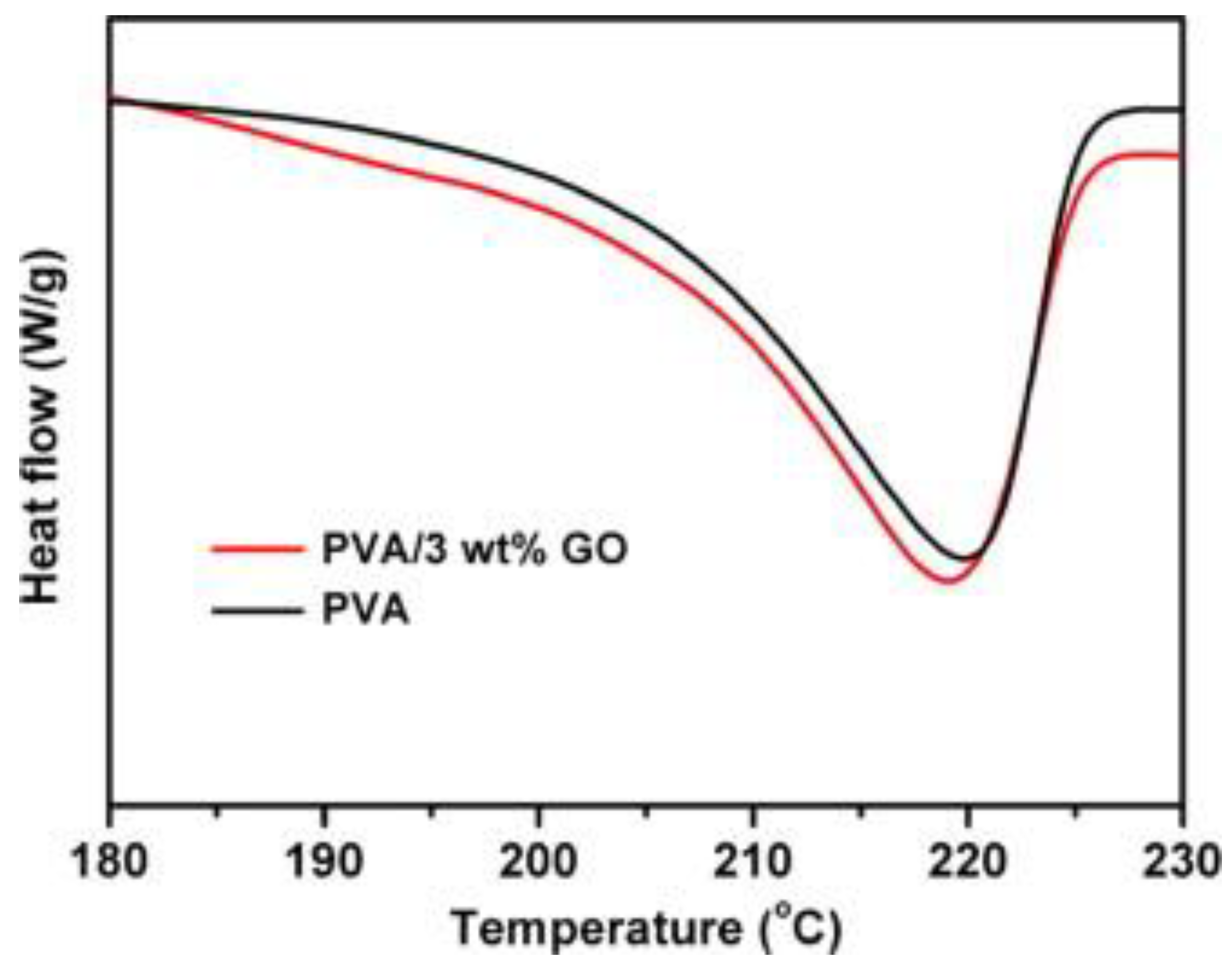
Thermal Degradation/Stability
3.2.3. Water Absorption Capabilities of Graphene Oxide/Polymer Composites
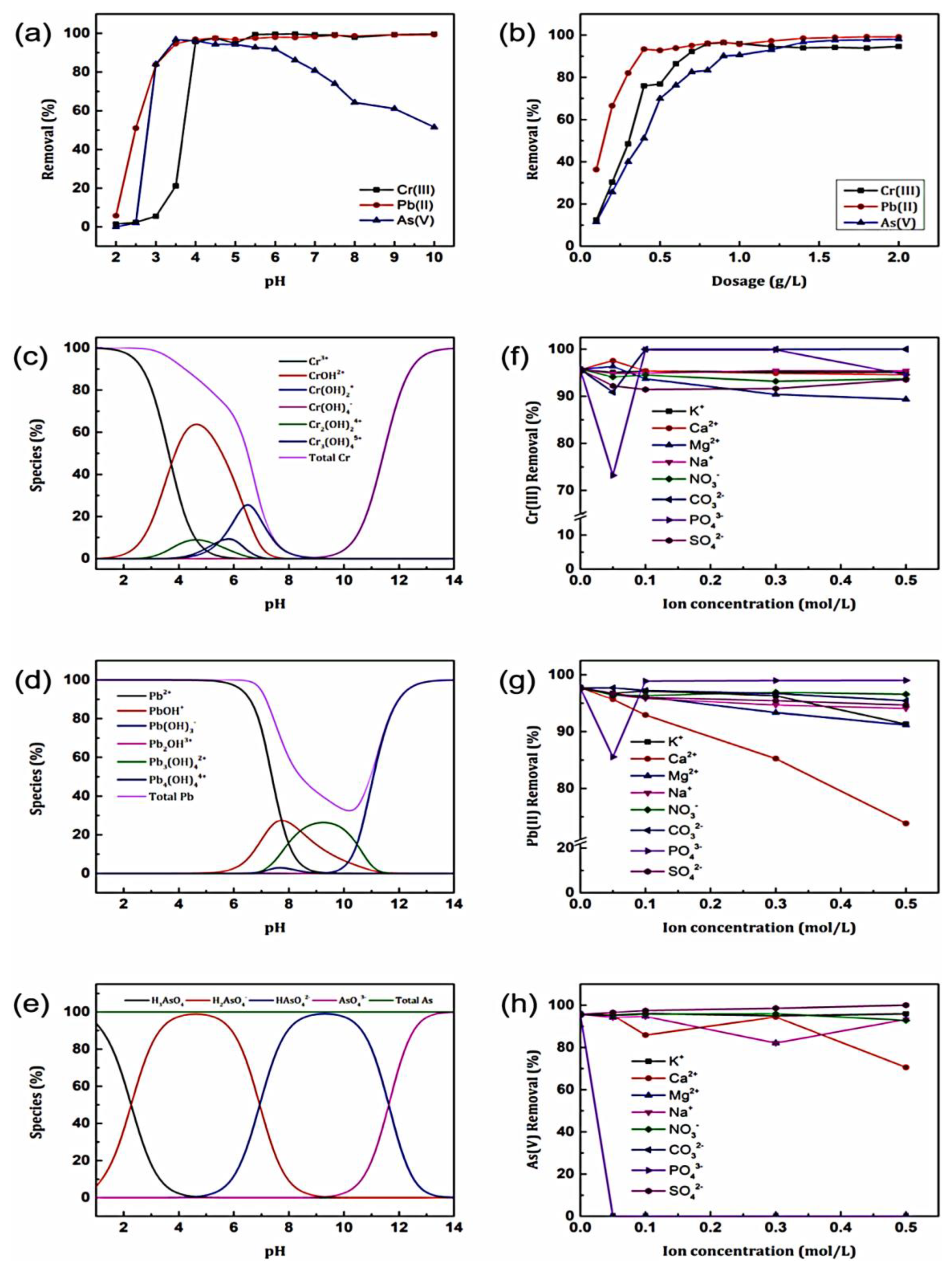
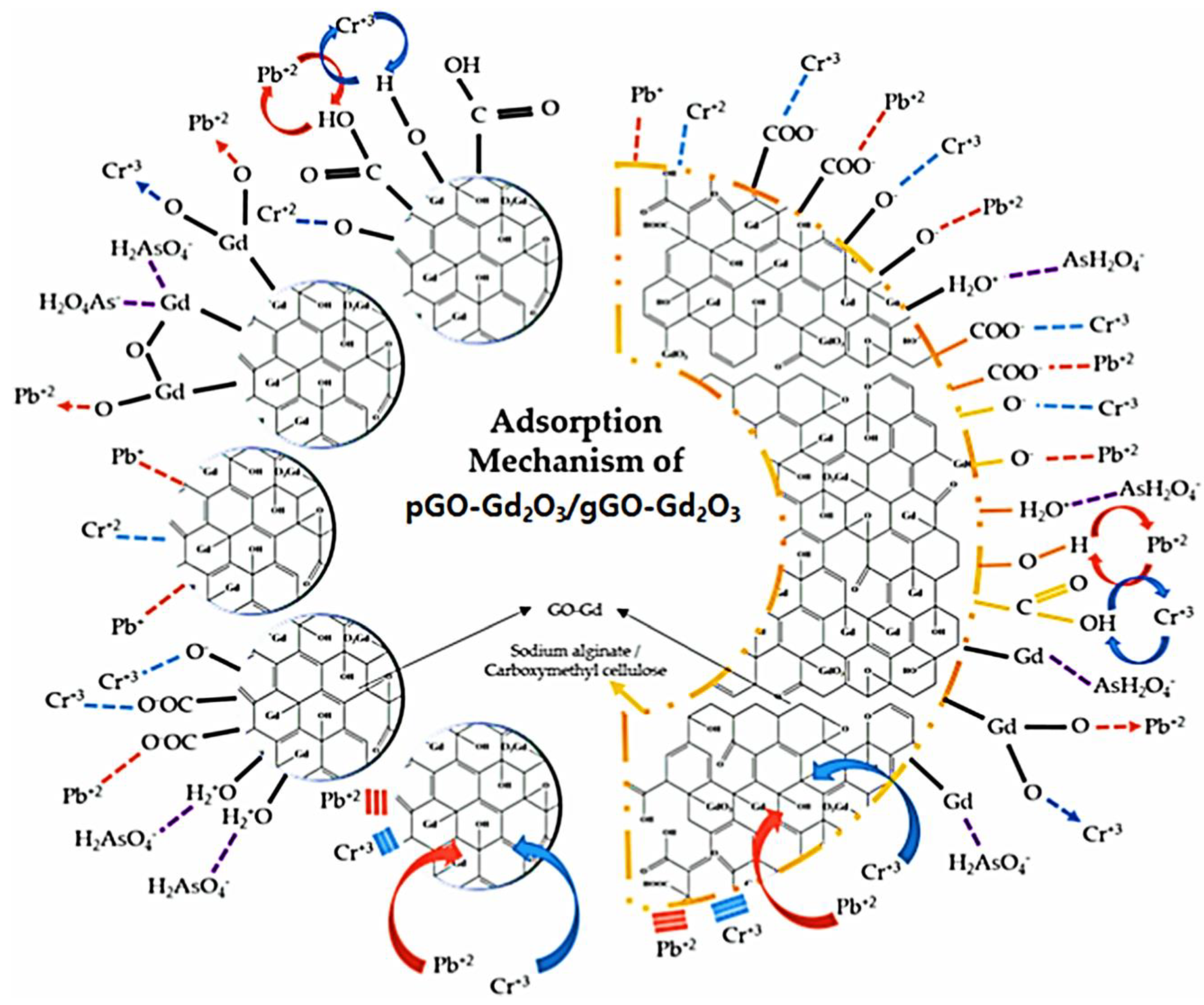
3.2.4. GO/Polymer Composites as Adsorbents for Heavy Metals in Water
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Makhetha, T.A.; Mpitso, K.; Luyt, A.S. Preparation and characterization of EVA/PLA/Sugarcane bagasse composites for water purification. J. Compos. Mater. 2016, 51, 1169–1186. [Google Scholar] [CrossRef]
- Bhushan, B. Design of water harvesting towers and projectors for water collection from fog and condensation. Philos. Trans. R. Soc. 2022, 378, 1–18. [Google Scholar] [CrossRef]
- Akinyemi, B.; Mushunje, A.; Fashogbon, A. Factors explaining household payment for potable water in South Africa. Cogent. Soc. Sci. 2018, 4, 1464379. [Google Scholar] [CrossRef]
- Kusangaya, S.; Jewitt, G. Impacts of climate change on water resources in Southern Africa: A review. Phys. Chem. Earth 2014, 67, 47–54. [Google Scholar] [CrossRef]
- Agoro, A.; Adenji, A.; Adefisoye, M.; Okoh, D. Heavy metals in wastewater and sewage sludge from selected municipal treatment plants in Eastern Cape Province, South Africa. Water 2020, 12, 2746. [Google Scholar] [CrossRef]
- Naidoo, D. Water—Can we do more with less? Water Sanit. Africa 2022, 17, 1–5. Available online: https://journals.co.za/doi/abs/10.10520/ejc-sh_san_v17_n1_a3 (accessed on 19 March 2023).
- Thakur, R.; Harris, G.; Thakur, S.; Onwubu, S. Factors contributing towards high water usage within poor communities in Kwazulu-Natal, South Africa. WIT Trans. Trans. Ecol. Environ. 2019, 239, 1–10. [Google Scholar] [CrossRef]
- Kookana, R.; Williams, M.; Boxall, A.; Larson, D.; Gaw, S.; Choi, K.; Yamamoto, H.; Thatikonda, S.; Zhu, Y.; Carriquiriborde, P. Potential ecological footprints of active pharmaceutical ingredients: An examination of risk factors in low, middle and high income countries. Philos. Trans. R. Soc. 2014, 369, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Tchounwou, P.; Yedjou, C.; Patlolla, A.; Sutton, D. Heavy metals toxicity and the environment. Mol. Clin. Environ. Toxicol. 2012, 101, 133–164. [Google Scholar] [CrossRef]
- Rehman, K.; Fatima, F.; Waheed, I.; Akash, M. Prevalence of exposure of heavy metals and their impact on health consequences. J. Cell. Biochem. 2017, 119, 157–184. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Li, L.; Qu, Z.; Xu, H.; Xu, J.; Yan, N. A novel method for the sequential removal and separation of multiple heavy metals from wastewater. J. Hazard. Mater. 2017, 342, 617–624. [Google Scholar] [CrossRef]
- Tahmasebi, E.; Masomi, M.; Yamimi, Y.; Marsali, A. Application of mechanosynthesized azine-decorated zinc (ii) metal-organic frameworks for highly efficient removal and extraction of some heavy-metal ions from aqueous samples: A comparative study. Inorg. Chem. 2015, 54, 425–433. [Google Scholar] [CrossRef]
- Luo, J.; Luo, X.; Crittenden, J.; Qu, J.; Bai, Y.; Peng, Y.; Li, J. Removal of antimonite (SbC(iii)) an antimonite (Sb(U)) from aqueous solution using carbon nanotubes that are decorated with zirconium oxide ZrO2. Environ. Sci. Technol. 2015, 49, 11115–11124. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Gunatilake, S. Methods of removing heavy metals from industrial wastewater. JMESS 2015, 1, 12–16. [Google Scholar]
- Saravanan, A.; Kumar, P.; Jeevanantham; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.; Reshma, B. Effective water/wastewater treatment methodologies for toxic pollutants removal: Process and applications towards sustainable development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.; Wang, H.; Hu, L.; Zhang, Y.; Xu, P. Treatment of produced water in the Permian basin for hydraulic fracturing: Comparison of different coagulation processes and innovative filter media. Water 2020, 12, 770. [Google Scholar] [CrossRef]
- Shim, H.; Lee, K.; Lee, D.; Jeon, D.; Park, M.; Shin, J.; Lee, Y.; Goo, S.; Chung, D. Application of electrocoagulation and electrolysis on the precipitation of heavy metals and particulate solids in washwater from the soil washing. JACEN 2014, 3, 130–138. [Google Scholar] [CrossRef]
- Golbaz, S.; Jafari, A.; Rafiee, M.; Kalantary, R. Separate and simultaneous removal of phenol, chromium, and cyanide from aqueous solution by coagulation/precipitation: Mechanisms and theory. Chem. Eng. J. 2014, 253, 251–257. [Google Scholar] [CrossRef]
- Vardhan, K.; Kumar, P.; Panda, R. A review on heavy metal pollution, toxicity and remedial measures: Current trends and future perspectives. J. Mol. Liq. 2019, 290, 111197. [Google Scholar] [CrossRef]
- Yousef, R.; Qiblawey, H.; El-Naas, M. Adsoprion as a process for produced water treatment: A review. Processes 2020, 8, 1657. [Google Scholar] [CrossRef]
- Rashid, R.; Shafiq, I.; Akhter, P.; Iqbal, M.; Hussain, M. A state-of-the-art review on wastewater treatment techniques: The effectiveness of adsorption method. Environ. Sci. Pollut. Res. 2021, 28, 9050–9066. [Google Scholar] [CrossRef]
- Rathi, B.; Kumar, P. Application of adsorption process for effective removal of emerging contaminants from water and wastewater. Environ. Pollut. 2021, 280, 116995. [Google Scholar] [CrossRef] [PubMed]
- Bacelo, H.; Pintor, A.; Santos, S.; Boaventura, R.; Botelho, C. Performance and prospects of different adsorbents for phosphorus uptake and recovery from water. Chem. Eng. J. 2020, 381, 122566. [Google Scholar] [CrossRef]
- Mehdizadeh, S.; Sadjadi, S.; Ahmadi, S.; Outokesh, M. Removal of heavy metals from aqueous solution using platinum nanoparticles/Zeolite-4A. J. Environ. Health. Sci. Eng. 2014, 12, 1–7. [Google Scholar] [CrossRef]
- Li, M.; Feng, C.; Zeng, Q. Preparation and Characterization of multi carboxyl-functionalized silica gel for removal of Cu (II), Cd (II), Ni (II) and Zn (II) from aqueous solution. Appl. Surf. Sci. 2014, 314, 1063–1069. [Google Scholar] [CrossRef]
- Nasir, H.; Wee, S.; Aris, S.; Abdullah, L.; Ismail, I. Processing of natural fibre and method improvement for removal of endocrine-disrupting compounds. Chemosphere 2022, 291, 132726. [Google Scholar] [CrossRef]
- Huang, W.; Xiao, S.; Zhong, H.; Yan, M.; Yang, X. Activation of persulfates by carbonaceous materials: A review. Chem. Eng. J. 2021, 418, 129297. [Google Scholar] [CrossRef]
- Jamwal, D.; Hussain, S.; Rohit, R.; Rana, D.; Katoch, A.; Mehta, S. Mixed transition metal oxide nanostructures based supercapacitors. In Encyclopedia of Energy Storage; Elsevier: Amsterdam, The Netherlands, 2022; Volume 4, pp. 566–580. [Google Scholar] [CrossRef]
- Parshetti, G.; Hoekman, S.; Balasubramanian, R. Chemical, structural and combustion characteristics of carbonaceous products obtained by hydrothermal carbonization of palm empty fruit bunches. Bioresour. Technol. 2013, 135, 683–689. [Google Scholar] [CrossRef]
- Sui, Z.; Meng, Q.; Li, J.; Zhu, J.; Cui, Y.; Han, B. High surface area porous carbons produced by steam activation of graphene aerogels. J. Mater. Chem. 2014, 2, 9891–9898. [Google Scholar] [CrossRef]
- Fan, J.; Ran, X.; Ren, Y.; Wang, C.; Yang, J.; Teng, W.; Zou, L.; Sun, Y.; Lu, B.; Deng, Y.; et al. Ordered mesoporous carbonaceous materials with tunable surface property for enrichment of hexachlorobenzene. Langmuir 2016, 39, 9922–9929. [Google Scholar] [CrossRef] [PubMed]
- Chai, W.; Cheun, J.; Kumar, P.; Mubashir, M.; Majeed, Z.; Banat, F.; Ho, S.; Show, P. A review on conventional and novel materials toward heavy metal adsorption in wastewater treatment applications. J. Clean. Prod. 2021, 296, 126589. [Google Scholar] [CrossRef]
- Gholami, F.; Tomas, M.; Gholami, Z.; Mirzaei, S.; Vakili, M. Surface characterization of carbonaceous materials using inverse gas chromatography: A review. Electrochem 2020, 4, 367–387. [Google Scholar] [CrossRef]
- Ahsan, A.; Islam, T.; Hernandez, C.; Castro, E.; Katla, S.; Kim, H.; Lin, Y.; Larry, M.; Torresdey, J.; Noveron, J. Biomass conversion of saw dust to a functionalized carbonaceous materials for the removal of Tetracycline, Sulfamethoxazole and Bisphenol A from water. J. Environ. Chem. Eng. 2018, 6, 4329–4338. [Google Scholar] [CrossRef]
- Zheng, B.; Lin, X.; Wu, D.; Matyjaszewski, K. Emerging functional porous polymeric and carbonaceous materials for environmental treatment and energy storage. Adv. Funct. Mater. 2020, 30, 1907006. [Google Scholar] [CrossRef]
- Inagaki, M.; Kang, K. Graphene derivatives: Graphene, fluorographene, graphene oxide, graphyne and graphdiyne. J. Mater Chem. 2014, 2, 13193–13206. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, F.; He, S.; Huang, F.; Peng, Z. Adsorption behaviour of reduced graphene oxide for removal of heavy metal ions. Asian. J. Chem. 2014, 15, 4901–4906. [Google Scholar] [CrossRef]
- Guo, T.; Bulin, C.; Ma, Z.; Li, B.; Zhang, Y.; Zhang, B.; Xing, R.; Ge, X. Mechanism of Cd(II) and Cu(II) adsorption onto few-layered magnetic graphene oxide as an efficient adsorbent. ACS Omega 2021, 25, 16535–16545. [Google Scholar] [CrossRef]
- Hur, J.; Shin, J.; Yoo, J.; Seo, Y. Competitive adsorption of metals onto magnetic graphene oxide: Comparison with other carbonaceous adsorbents. Sci. World. J. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Li, L.; Zhao, L.; Ma, J.; Tian, Y. Preparation of graphene oxide/chitosan complex and its adsorption properties for heavy metal ions. Green Process. Synth. 2020, 9, 294–303. [Google Scholar] [CrossRef]
- Liu, L.; Qing, M.; Wang, Y.; Chen, S. Defects in graphene: Generation, healing, and their effects on the properties of graphene: A review. J. Mater. Sci. Technol. 2015, 31, 599–606. [Google Scholar] [CrossRef]
- Zulaikha, S.; Demon, N.; Kamisan, A.; Abdullah, N.; Noor, S.; Khim, O.; Kasim, N.; Yahya, M.; Manaf, N.; Azmi, A.; et al. Graphene-based materials in gas sensor applications: A review. Sens. Appl. 2020, 32, 759–777. [Google Scholar]
- Rudrapati, R. Graphene: Fabrication methods, properties and applications in modern industries. In Graphene Production and Application, 1st ed.; Ameen, S., Ameen, M., Akhtar, M., Shin, H., Eds.; IntechOpen: London, UK, 2020; Volume 1. [Google Scholar] [CrossRef]
- Ridolfo, R.; Tavakoli, S.; Junnuthula, V.; Williams, D.; Urtti, A.; Hest, J. Exploring the impact of morphology on the properties of biodegradable nanoparticles and their diffusion in complex biological medium. Biomacromolecules 2021, 22, 126–133. [Google Scholar] [CrossRef]
- Robaiah, M.; Rusop, M.; Abdullah, S.; Khuzaimi, Z.; Azhan, H.; Laila, M.; Salifairus, M.; Asli, N. Morphology and topography study of graphene synthesized from plant oil. AIP Conf. Proc. 2018, 1963, 020045. [Google Scholar] [CrossRef]
- Monti, M.; Rallini, M.; Puglia, D.; Torre, L.; Kenny, J. Morphology and electrical properties of graphene–epoxy nanocomposites obtained by different solvent assisted processing methods. Composites 2012, 46, 166–172. [Google Scholar] [CrossRef]
- Prolongo, S.; Jimenez-Suarez, A.; Moriche, R.; Urena, A. Influence of thickness and lateral size of graphene nanoplatelets on water uptake in epoxy/graphene nanocomposites. Appl. Sci. 2018, 8, 1550. [Google Scholar] [CrossRef]
- Qiang, F.; Hu, L.; Gong, L.; Zhao, L.; Li, S.; Tang, L. Facile synthesis of super-hydrophobic, electrically conductive and mechanically flexible functionalized graphene nanoribbon/polyurethane sponge for efficient oil/water separation at static and dynamic states. Chem. Eng. J. 2018, 334, 2154–2166. [Google Scholar] [CrossRef]
- Ren, X.; Li, J.; Tan, X.; Wang, X. Comparative study of graphene oxide, activated carbon and carbon nanotubes as adsorbents for copper decontamination. Dalton Trans. 2013, 42, 5266–5276. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, P.; delaLuz-Asuncion, M.; Martinez, A.; Velasco-Santos, C. Graphene materials to remove organic pollutants and heavy metals from water: Photocatalysis and adsorption. In Semiconductor Photocatalysis-Materials, Mechanisms and Applications, 1st ed.; Cao, W., Ed.; Intechopen: London, UK, 2016; Volume 1. [Google Scholar] [CrossRef]
- Wang, M.; Duan, X.; Xu, Y.; Duan, X. Functional three-dimensional graphene/polymer composites. ACS Nano 2016, 10, 7231–7247. [Google Scholar] [CrossRef]
- Yu, W.; Sisi, L.; Haiyan, Y.; Jie, L. Progress in the functional modification of graphene/graphene oxide: A review. R. Soc. Chem. 2020, 10, 15328–15345. [Google Scholar] [CrossRef] [PubMed]
- Jiat, L.; Zhang, H.; Chiew, L.; Yee, L.; Suyin, G.; Thangalazhy-Gopakumar, S.; Rigby, S. Review on graphene and its derivatives: Synthesis methods and potential industrial implementation. J. Taiwan. Inst. Chem. Eng. 2019, 98, 163–180. [Google Scholar] [CrossRef]
- Singh, R.; Kumar, R.; Singh, D. Graphene oxide: Strategies for synthesis, reduction and frontier applications. R. Soc. Chem. 2016, 6, 64993–65011. [Google Scholar] [CrossRef]
- Kim, F.; Lote, L.; Huang, J. Graphene oxide: Surface activity and two-dimensional assembly. Adv. Mater. 2010, 22, 1954–1958. [Google Scholar] [CrossRef]
- Mei, Q.; Liu, B.; Han, G.; Liu, R.; Han, M.; Zhang, Z. Graphene oxide: From tunable structures to diverse luminescence behaviours. Adv. Sci. 2019, 6, 1900855. [Google Scholar] [CrossRef]
- Shang, Y.; Zhang, D. Preparation and thermal properties of graphene-oxide-microencapsulated phase change materials. Nanoscale Microscale Thermophys. Eng. 2016, 2, 147–157. [Google Scholar] [CrossRef]
- Ghosh, T.; Gope, S.; Mondal, D.; Bhowmik, B.; Mollick, M.; Maity, D.; Roy, I.; Sarkar, G.; Sadhukhan, S.; Rana, D.; et al. Assessment of morphology and property of graphene-hydroxypropylmethylcellulose nanocomposite films. Int. J. Biol. Macromol. 2014, 66, 338–345. [Google Scholar] [CrossRef]
- Xia, Z.; Maccaferri, G.; Zanardi, C.; Christian, M.; Ortolani, L.; Morandi, V.; Bellani, V.; Kovtun, A.; Dell’Elce, S.; Candini, A.; et al. Dispersion stability and surface morphology study of electrochemically exfoliated bilayer grapheme oxide. J. Phys. Chem. 2019, 123, 15122–15130. [Google Scholar] [CrossRef]
- Jaafar, E.; Kashif, M.; Sahari, S.; Ngaini, Z. Study on morphological, optical and electrical properties of graphene oxide (GO) and reduced grapehene oxide (rGO). Mater. Sci. Forum. 2018, 917, 112–116. [Google Scholar] [CrossRef]
- Castilla-Cortaza, I.; Vidaurre, A.; Mari, B.; Campillo-Fernandez, A. Morphology, crystallinity and molecular weight of poly (ε-caprolactone)/grapheme oxide hybrids. Polymers 2019, 11, 1099. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Babaei, F.; Fakhri, P.; Jaleh, B. Synthesis, characterization, structural, optical properties ad catalytic activity of reduced graphene oxide/copper nanocomposites. R. Soc. Chem. 2015, 5, 10782–10789. [Google Scholar] [CrossRef]
- Abid, M.; Hou, X.; Zheng, W.; Hussain, R. High temperature and residual properties of reactive powder concrete- A review. Constr. Build. Mater. 2017, 147, 339–351. [Google Scholar] [CrossRef]
- Alhassan, S.; Qutubuddin, S.; Schiraldi, D.; Agag, T.; Ishida, H. Preparation and thermal properties of graphene oxide/main chain benzoxazine polymer. Eur. Polym. J. 2013, 49, 3825–3833. [Google Scholar] [CrossRef]
- Bhawal, P.; Ganguly, S.; Chaki, T.; Das, N. Synthesis and characterization of graphene oxide filled ethylene acrylate hybrid nanocomposites. R. Soc. Chem. 2016, 6, 20781–20790. [Google Scholar] [CrossRef]
- Liu, R.; Gong, T.; Zhong, K.; Lee, C. Graphene oxide papers with high water adsorption capacity for air dehumidification. Sci. Rep. 2017, 7, 9761. [Google Scholar] [CrossRef]
- Lian, B.; De Luca, S.; You, Y.; Alwarappan, S.; Yoshimura, M.; Sahajwalla, V.; Smith, S.; Leslie, G.; Joshi, R. Extraordinary water adsorption characteristics of graphene oxide. Chem. Sci. 2018, 9, 5106–5111. [Google Scholar] [CrossRef]
- Sitko, R.; Turek, E.; Zawisza, B.; Malicka, E.; Talik, E.; Heiman, J.; Gagor, A.; Feist, B.; Wizalik, R. Adsorption of divalent metal ions from aqueous solutions using graphene oxide. Dalton Trans. 2013, 42, 5682–5689. [Google Scholar] [CrossRef]
- Zhao, G.; Ren, X.; Ghao, X.; Tan, X.; Li, J.; Chen, C.; Huang, Y.; Wang, X. Removal of Pb(II) ions from solutions on few-layered graphene oxide nanosheets. Dalton Trans. 2011, 40, 10945–10952. [Google Scholar] [CrossRef]
- Gao, Y.; Picot, O.; Bilotti, E.; Peijs, T. Influence of filler size on the properties of poly(lactic acid) (PLA)/graphene nanoplatelet (GNP) nanocomposites. Eur. Polym. J. 2017, 86, 117–131. [Google Scholar] [CrossRef]
- Wu, D.; Cheng, Y.; Feng, S.; Yao, Z.; Zhang, M. Crystallization behaviour of polylactide/graphene composites. Ind. Eng. Chem. Res. 2013, 52, 6731–6739. [Google Scholar] [CrossRef]
- Murray, E.; Sayyar, S.; Thompson, B.; Gorkini, R.; Officer, D.; Wallace, G. A bio-friendly, green route to processable, biocompatible graphene/polymer composites. R. Soc. Chem. 2015, 5, 45284–45290. [Google Scholar] [CrossRef]
- Villa-Rodil, S.; Paredes, J.; Martinez-Alonso, A.; Tascon, J. Preparation of graphene dispersions and graphene–polymer composites in organic media. J. Mater. Chem. 2009, 19, 3591–3593. [Google Scholar] [CrossRef]
- Du, J.; Cheng, H. The fabrication, properties, and uses of graphene/polymer composites. Macromol. Chem. Phys. 2012, 213, 1060–1077. [Google Scholar] [CrossRef]
- Pinto, A.M.; Cabral, J.; Tanaka, D.A.; Mendes, A.M.; Magalhaes, F.D. Effect of incorporation of graphene oxide and graphene nanoplatelets on mechanical and gas permeability properties of poly (lactic acid) films. Polym. Int. 2012, 62, 33–40. [Google Scholar] [CrossRef]
- Theophile, N.; Jeong, H.K. Electrochemical properties of poly (vinylalcohol) and graphene oxide composite for supercapacitor applications. Chem. Phys. 2017, 669, 125–129. [Google Scholar] [CrossRef]
- Chen, Y.; Qi, Y.; Tai, Z.; Yan, X.; Zhu, F.; Xue, Q. Preparation, mechanical properties and biocompatibility of graphene oxide/ultrahigh molecular weight polyethylene composites. Eur. Polym. J. 2012, 48, 1026–1033. [Google Scholar] [CrossRef]
- Xu, Y.; Hong, W.; Bai, H.; Hi, C.; Shi, G. Strong and ductile poly (vinyl alcohol)/graphene oxide composite films with a layered structure. Carbon 2009, 47, 3538–3543. [Google Scholar] [CrossRef]
- Zhou, J.; Yao, Z.; Chen, Y.; Wei, D.; Wu, Y.; Xu, T. Mechanical and thermal properties of graphene oxide/phenolic resin composite. Polym. Compos. 2013, 34, 125–1249. [Google Scholar] [CrossRef]
- Mindivian, F. The synthesis, thermal and structural characterization of polyvinylchloride/grapheme oxide (PVC/GO) composites. Mater. Sci. Non-Equilib. Phase Transform. 2015, 1, 33–36. [Google Scholar]
- Gavin, F.; Cousin, P.; Robert, M. Effect of modified graphene oxide on the mechanical, thermal and barrier properties of vinylester. J. Compos. Mater. 2018, 52, 3853–3864. [Google Scholar] [CrossRef]
- Lee, S.; Lingamdinne, L.; Koduru, J.; Chang, Y.; Naushad, M. Biopolymer mixture-entrapped modified graphene oxide for sustainable treatment of heavy metal contaminated real surface water. J. Water Process. Eng. 2022, 46, 10263. [Google Scholar] [CrossRef]
- Peng, W.; Li, H.; Liu, X.; Song, S. A review on heavy metal ions adsorption from water by graphene oxide and its composites. J. Mol. Liq. 2017, 230, 496–504. [Google Scholar] [CrossRef]

| Sample | Weight% | ||
|---|---|---|---|
| Nickel | Carbon | Oxygen | |
| Nickel substrate | 100 | 0 | 0 |
| RCO | 82.95 | 0.96 | 16.09 |
| RPO | 83.12 | 0.91 | 15.97 |
| WCPO | 77.33 | 1.54 | 21.13 |
| Sample | Ra (nm) |
|---|---|
| Nickel substrate | 20.099 |
| Refined corn oil (RCO) | 54.794 |
| Refined palm oil (RPO) | 61.493 |
| Waste cooking palm oil (WCPO) | 69.098 |
| Langmuir | Freundlich | |||||
|---|---|---|---|---|---|---|
| qmax (mg·g−1) | KL (L·mg−1) | RL | KF (mg·g−1)(L·mg−1)1/n | n | RF | |
| Cu | 294 ± 18 | 0.09 ± 0.01 | 0.990 | 61 ± 8 | 3.0 ± 0.3 | 0.974 |
| Zn | 345 ± 24 | 0.12 ± 0.02 | 0.986 | 83 ± 11 | 3.1 ± 0.4 | 0.965 |
| Cd | 530 ± 26 | 0.10 ± 0.01 | 0.986 | 141 ± 20 | 3.4 ± 0.5 | 0.955 |
| Pd | 1119 ± 41 | 0.14 ± 0.02 | 0.983 | 390 ± 37 | 4.5 ± 0.5 | 0.973 |
| Sample | Tg (°C) | Tcc (°C) | ΔHcc (°C) | Tm1 (°C) | Tm2 (°C) | Xc (°C) |
|---|---|---|---|---|---|---|
| PLA | 56.3 | 101.2 | 26.25 | 145.8 | 154.4 | 28.44 |
| PLA/G | 55.8 | 103.8 | 27.28 | 146.0 | 154.3 | 29.23 |
| Sample | Water Absorption (%) | Contact Angle (°) |
|---|---|---|
| Pure PVA | 105.2 | 36 |
| 0.5 wt.% graphene/PVA | 59.8 | 93 |
| 1 wt.% graphene/PVA | 48.8 | 97 |
| Samples | Glass Transition Temperature (Tg) (°C) | Melting Temperature (Tm) (°C) | Enthalpy (ΔH) (J/g) |
|---|---|---|---|
| PVC | 59 | 302 | 58.66 |
| PVC/GO-0.1 | 56 | 299 | 62.9 |
| PVC/GO-0.3 | 53 | 303 | 57.24 |
| PVC/GO-0.5 | 54 | 294 | 55.41 |
| PVC/GO-1 | 55 | 303 | 29.81 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mokoena, L.S.; Mofokeng, J.P. A Review on Graphene (GN) and Graphene Oxide (GO) Based Biodegradable Polymer Composites and Their Usage as Selective Adsorbents for Heavy Metals in Water. Materials 2023, 16, 2527. https://doi.org/10.3390/ma16062527
Mokoena LS, Mofokeng JP. A Review on Graphene (GN) and Graphene Oxide (GO) Based Biodegradable Polymer Composites and Their Usage as Selective Adsorbents for Heavy Metals in Water. Materials. 2023; 16(6):2527. https://doi.org/10.3390/ma16062527
Chicago/Turabian StyleMokoena, Lesia Sydney, and Julia Puseletso Mofokeng. 2023. "A Review on Graphene (GN) and Graphene Oxide (GO) Based Biodegradable Polymer Composites and Their Usage as Selective Adsorbents for Heavy Metals in Water" Materials 16, no. 6: 2527. https://doi.org/10.3390/ma16062527
APA StyleMokoena, L. S., & Mofokeng, J. P. (2023). A Review on Graphene (GN) and Graphene Oxide (GO) Based Biodegradable Polymer Composites and Their Usage as Selective Adsorbents for Heavy Metals in Water. Materials, 16(6), 2527. https://doi.org/10.3390/ma16062527






