Direct Recycling of WC-Co Grinding Chip
Abstract
1. Introduction
1.1. WC-Co Raw Materials
1.2. WC-Co Scraps and Recycling
- -
- direct recycling: scraps are transformed into powders with the same composition. In direct recycling, scrap materials are directly transformed into a product having a similar composition, using physical or low-intensive chemical processing methods (or a combination of both [45,46]). High-purity scraps (i.e., the same composition as the final product and precise size-range) must be sorted and cleaned during the manufacturing process to be further processed. Direct recycling is combined with a minimum of energy consumption, chemical waste generation, and production cost. For those tungsten carbide scrap materials that fail to meet the strict purity standards required by direct processes [27], chemical methods are needed;
- -
- chemical recycling methods include both indirect and semi-indirect processes. Indirect methods involve the conversion of scraps into intermediate products, such as Ammonium Para Tungstate (APT), while semi-indirect methods involve the selective dissolution of one component, typically the binder. These methods require the use of intensive chemical modification techniques, such as the use of acid or alkaline media, to obtain intermediate products that can be further processed. For the sake of readability in this article, we will consider chemical recycling the same as indirect.
2. State of the Art of WC-Co Grinding Sludge Recycling Methods
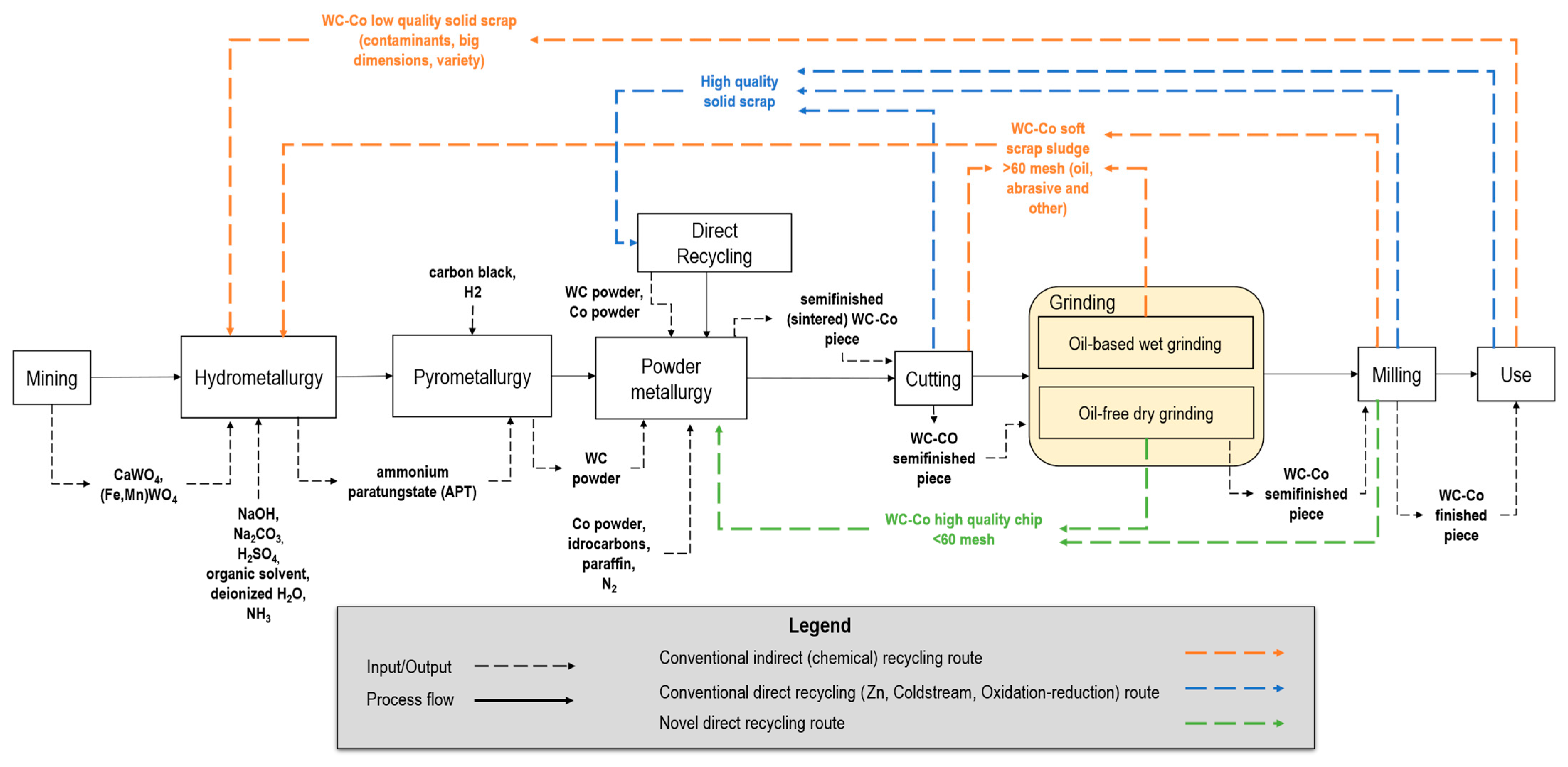
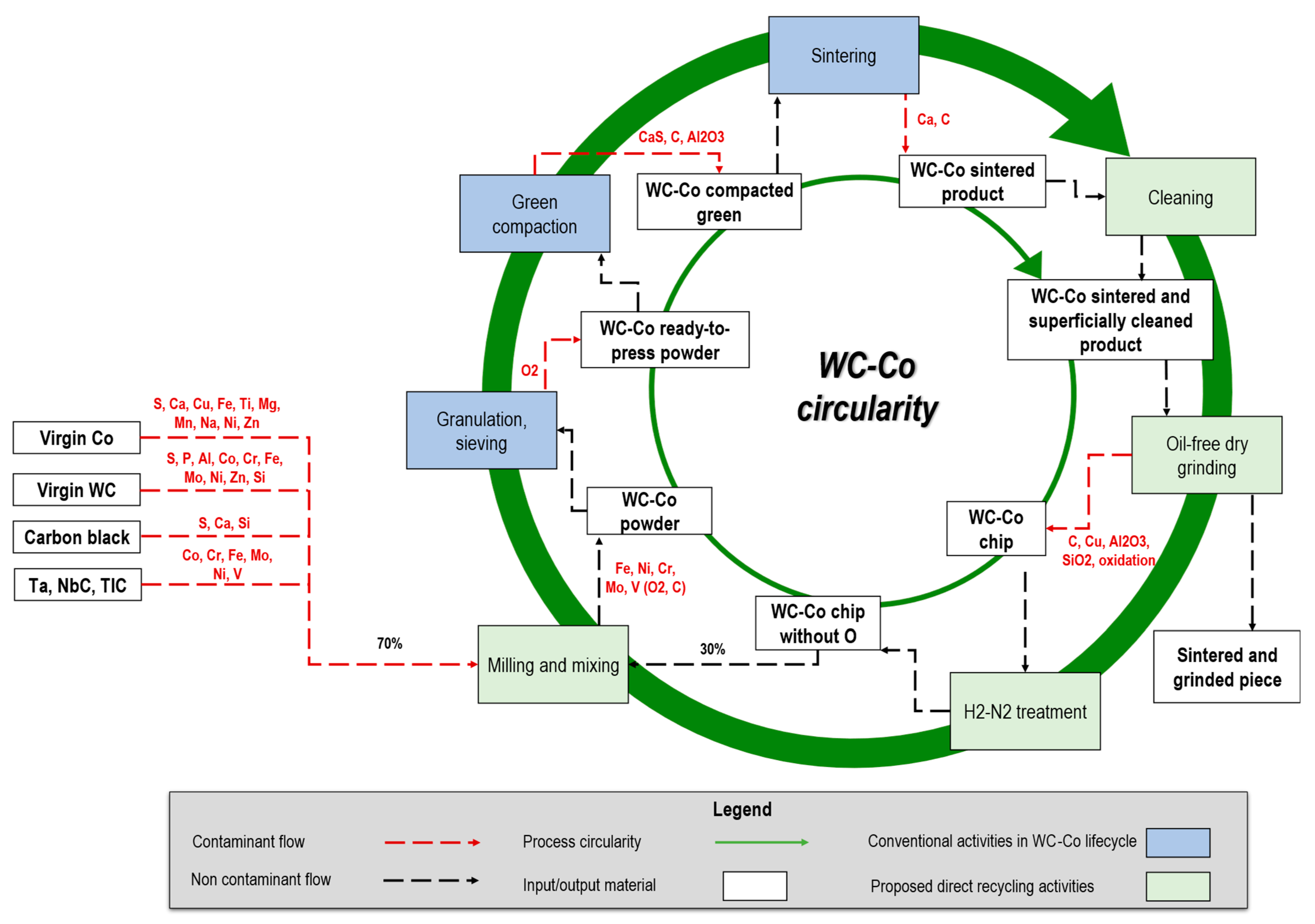
2.1. WC-Co Lifecycle and Processing Overview
- -
- Mining. The first step is the extraction of natural resources through drilling, blasting, and digging. The ore is then crushed and milled [31]. The primary resources, such as wolframite (Fe,Mn)WO4 and scheelite CaWO4, are extracted and transported and then processed further through hydrometallurgy.
- -
- Hydrometallurgy. This step includes sodium hydroxide NaOH and sodium carbonate Na2CO3 digestion [48], which, after filtration and precipitation to remove silica and molybdenum impurities [49], leads to pure sodium tungstate Na2WO4. Tungsten is extracted from the purified Na2WO4 solution using an organic solvent mixed with sulfuric acid H2SO4. The extract is washed with deionized H2O and then the organic solvent is stripped from the tungsten by the addition of NH3 [49], resulting in an ammonia tungstate ((NH4)2WO4) solution, which is treated to form APT [49,50].
- -
- Pyrometallurgy. At the third step, the APT is transformed to tungsten blue oxide (TBO) WOx via calcination in rotary furnaces [51]. Then, hydrogen H2 reduction converts TBO to tungsten metal powder in push-type furnaces with stoichiometric H2 excess [49]. Finally, carburization is commonly performed by introducing tungsten metal powder blended with carbon black in a furnace with a reducing H2 atmosphere [49] to obtain WC powder.
- -
- Powder metallurgy. WC and Co powders are mixed in variable shares based on the application, along with a solvent (e.g., hydrocarbons [52]) and additives (e.g., paraffin wax, 1–3% share of the mixture). The mixture is then milled to obtain the desired particle size [53,54] and granulation is typically carried out via spray drying [53]. Finally, the ready-to-press powder is compacted into the desired shape (e.g., cylindrical) and sintered until 1450 °C in an inert atmosphere to avoid oxidation.
- -
- Machining (cutting, grinding, milling). Material-removal operations are carried out to cut the cylindrical items, reduce the surface roughness as well as geometrical errors (e.g., run-out) within the required tolerance [55], and reach the final shape of the cutting edges (e.g., helicoidal). As shown in Figure 2, different sources of WC-Co waste are generated during all these subtractive machining steps, including solid swarf and chips (i.e., hard scrap), and very fine chips mixed with fluids and contaminants (i.e., sludges) [56].
- -
- Use. Once the finished cylindrical product is used, at the end-of-life, it must be disposed of and can also enter the recycling route [56].
2.2. WC-Co Grinding Sludge Recycling
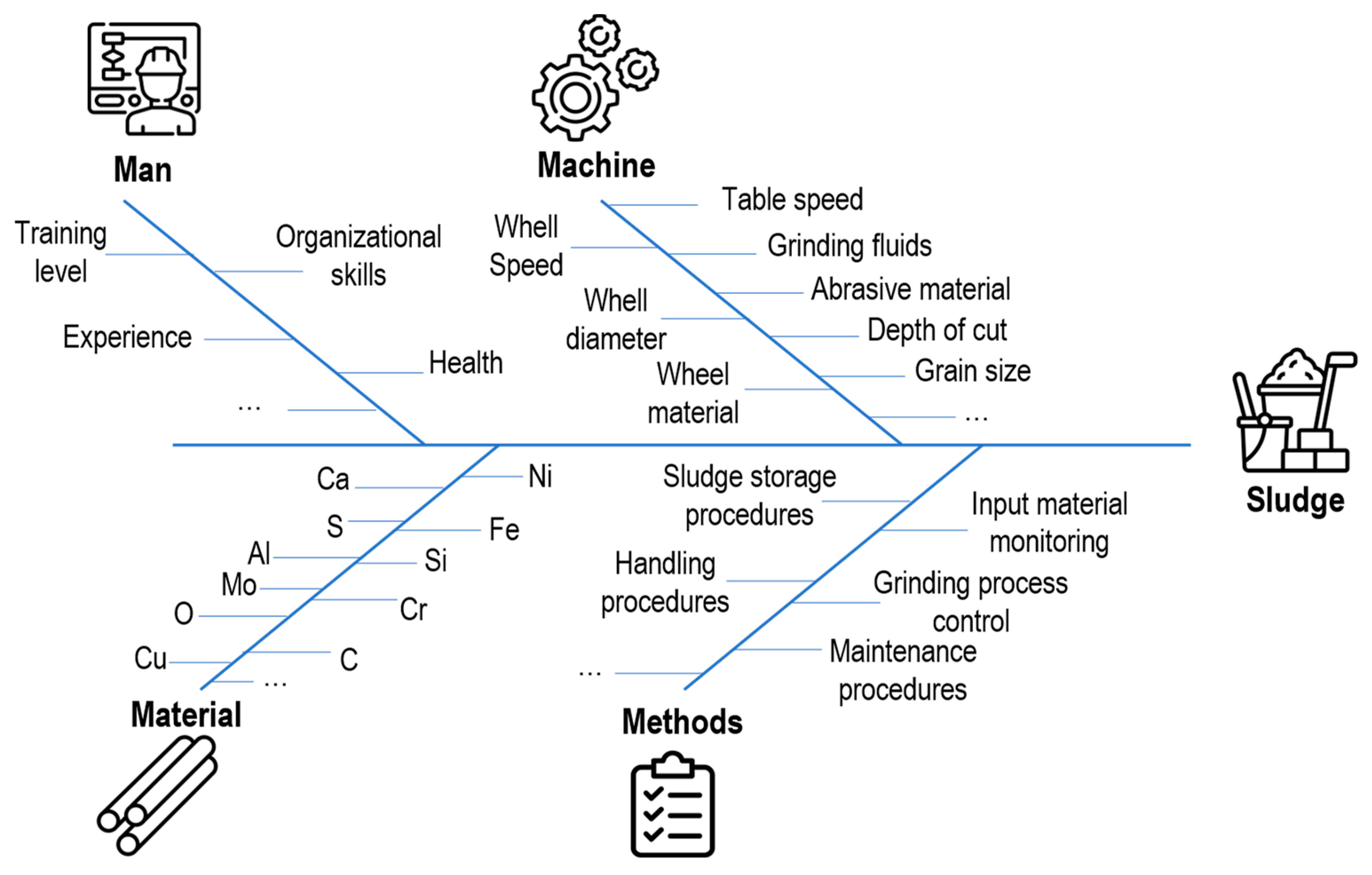
2.3. WC-Co Grinding Sludge Impurity Sources
- -
- Machine: Cemented carbide cylinders are machined using various grinding machines and conditions (such as dry and wet) [16,60]. Conventional grinding often employs a wheel with diamond abrasive grains of varying sizes, ranging from hundreds (roughing) to a few (polishing) μm, which are embedded in a metal or resinoid binder [16]. In combination, a water–oil emulsion is used as a coolant during the grinding process to prevent burnout and thermal damage, lubricate the tools, reduce wheel wear, and remove grits from the grinder [61]. Among several types, there are two significant categories of water-based grinding fluids: mineral oil in water emulsion and semi/synthetic products in water coolants. Both are mixtures of oil and synthetic lubricant with an average dilution rate of 1–5%. Still, considering the machine variables, the major process parameters, such as table speed, abrasive material, grain size, wheel material, diameter, speed, and depth of cut, have an enormous importance in the final product quality as well as the generated sludge purity level.
- -
- Methods: As another variable, quality management methods or procedures greatly influence the sludge’s characteristics. Procedures for the input material control (i.e., incoming quality control), determine the average chemical composition of the input material before the grinding process. Additionally, specific grinding-process control (e.g., process parameters monitoring via sensing techniques), machine and tools monitoring strategy (e.g., wear control), as well as handling and storage, determine the final physical and chemical characteristics of the WC-Co sludge, including the level of organic contaminants and oxidation. Furthermore, production planning and sludge storage affect the level of variation in the output sludge from batch production.
- -
- Material: The characteristics of the output WC-Co sludge will vary depending on the composition of the input WC-Co raw material.
- -
- Man: Based on the operators’ skills, experiences, and training levels as well as according to the previously defined procedure to be followed, the WC-Co sludge presents specific characteristics.
3. Grinding Chip Characterization
- -
- The specific composition of the material and its particle size (fine chips) does not allow for quick X-Ray Fluorescence (XRF) analysis, as matrices and calibration standards for XRF are not readily available.
- -
- The small particle size of the material causes large peaks when using X-Ray Diffraction (XRD), making it difficult to reliably read the spectra as the peaks of Co and W (and of the respective oxides) may overlap.
- -
- The small particle size and tendency to aggregate negatively impact the success of a particle size analysis by laser diffraction techniques (i.e., granulometry).
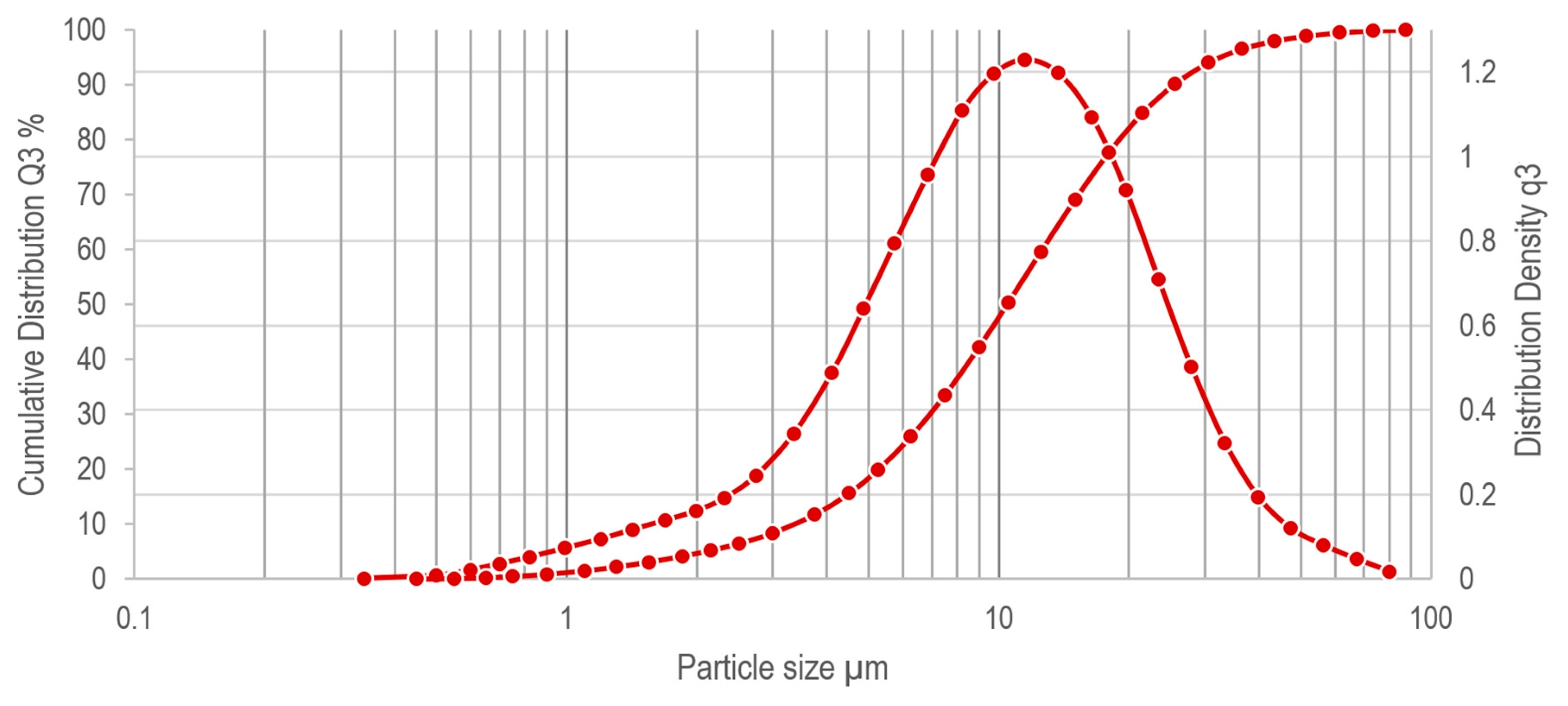
3.1. Chip Morphology, Granulometry and Composition
3.2. Comparison with Commercial Powders
4. The Proposed Direct Recycling Method
- Pre grinding: remove possible sources of superficial contaminants through a simple cleaning of the sintered bar surface. This step is useful to easily remove contaminants such as C, Ca, and others that have been observed and may be present on the bars’ surfaces before grinding.
- Post grinding: reduce the total internal oxygen content of the ground chips to avoid defective power, which in turn leads to a subsequent defective sintered product [68]. Additionally, the content of C must be carefully checked to avoid the formation of a graphite or η-phase in the sintered part (brittle) [69]. In this step, we have compared two alternative methods: treatment in H2 up to 400 °C [70] and treatment in Argon (Ar) up to 900 °C [68]. The former approach (H2) requires longer times but does not modify the C content (which can subsequently be set by adding virgin or recycled W, carbon black, Co powders). The latter approach (Ar) presents faster and more efficiently replicable characteristics at a laboratory scale, but it does not allow precise control of the C content and does not prevent decarburization of the WC.
- Chip milling: reduce the granulometry of the output chips to create a fine powder and mix the virgin W, carbon black, or Co powders to achieve specific compositions, if necessary [54]. The powder has been milled in an MGS Mills planetarium ball mill (440 rpm) for 30 min, loaded with WC-Co balls (3–6 mm) and distilled water as a milling medium. Correction of W, C, and Co can be applied at this stage to improve homogeneity. After milling/mixing, conventional granulation and sieving can generate ready-to-press powder.
5. Direct Recycling Method Validation
6. Discussion
- -
- Cr, V, Nb, and Ta, commonly used as grain growth inhibitors (desired values are defined in [77]), are present in tolerable percentages.
- -
- Fe, Ni, Cr, Mo, and V, deriving from the wear of the steel mills [67], are within limits.
- -
- The oxygen (O) content (due to partial oxidation of the material in air) is above the limits preventing the use of the input material in its current state. The carbon C content also does not fit into the project carbon content range [69].
- -
- Other critical elements are Al, Si, Cu, Ca, and S.
- -
- Cu, which is not usually present in virgin powders, is present in high percentages.
6.1. Sustainability of the Direct Recycling Method
6.2. Future Directions
- -
- Various stakeholders, including manufacturers, users, and recyclers, can be identified from a research and industrial perspective with long-term, well-defined relationships between the value chain actors that may cover the separate activities shown in Figure 8. Such relationships can be enhanced by vertical integration, or by long-term contractual agreements [22].
- -
- Scaling up the proposed methodology for the direct recycling of WC-Co chips from laboratory to industrial context. This analysis entails a disruptive change in the business model of WC-Co bars production and finishing for greener manufacturing [78].
- -
- Exploring additive manufacturing and other powder metallurgy processes using the directly recycled WC-Co powder in the interest of energy, resources, and sustainable development of alternative sintering methods. WC-Co hard metal parts are usually produced by casting and powder metallurgy, which are limited by complex geometries and require post-processing such as conventional and non-conventional machining [37,79].
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lupi, F.; Mabkhot, M.M.; Finžgar, M.; Minetola, P.; Stadnicka, D.; Maffei, A.; Litwin, P.; Boffa, E.; Ferreira, P.; Podržaj, P.; et al. Toward a sustainable educational engineer archetype through Industry 4.0. Comput. Ind. 2021, 134, 103543. [Google Scholar] [CrossRef]
- Mabkhot, M.M.; Ferreira, P.; Maffei, A.; Podržaj, P.; Mądziel, M.; Antonelli, D.; Lanzetta, M.; Barata, J.; Boffa, E.; Finžgar, M.; et al. Mapping Industry 4.0 Enabling Technologies into United Nations Sustainability Development Goals. Sustainability 2021, 13, 2560. [Google Scholar] [CrossRef]
- ISO. ISO 14001:2015—Environmental Management Systems—Requirements with Guidance for Use. n.d. Available online: https://www.iso.org/standard/60857.html (accessed on 14 December 2022).
- Bjørnbet, M.M.; Skaar, C.; Fet, A.M.; Schulte, K.Ø. Circular economy in manufacturing companies: A review of case study literature. J. Clean. Prod. 2021, 294, 126268. [Google Scholar] [CrossRef]
- Jawahir, I.; Bradley, R. Technological Elements of Circular Economy and the Principles of 6R-Based Closed-loop Material Flow in Sustainable Manufacturing. Procedia CIRP 2016, 40, 103–108. [Google Scholar] [CrossRef]
- Botelho, A.B., Jr.; Espinosa, D.C.R.; Tenório, J.A.S. Extraction of Scandium from Critical Elements-Bearing Mining Waste: Silica Gel Avoiding in Leaching Reaction of Bauxite Residue. J. Sustain. Met. 2021, 7, 1627–1642. [Google Scholar] [CrossRef]
- Dos Santos, D.M.; Buzzi, D.C.; Botelho, A.B., Jr.; Espinosa, D.C.R. Recycling of printed circuit boards: Ultrasound-assisted comminution and leaching for metals recovery. J. Mater. Cycles Waste Manag. 2022, 24, 1991–2001. [Google Scholar] [CrossRef]
- Larouche, F.; Tedjar, F.; Amouzegar, K.; Houlachi, G.; Bouchard, P.; Demopoulos, G.P.; Zaghib, K. Progress and Status of Hydrometallurgical and Direct Recycling of Li-Ion Batteries and Beyond. Materials 2020, 13, 801. [Google Scholar] [CrossRef]
- Zhang, C.; Hu, M.; Yang, X.; Miranda-Xicotencatl, B.; Sprecher, B.; Di Maio, F.; Zhong, X.; Tukker, A. Upgrading construction and demolition waste management from downcycling to recycling in the Netherlands. J. Clean. Prod. 2020, 266, 121718. [Google Scholar] [CrossRef]
- Pacheco, J.; de Brito, J. Recycled Aggregates Produced from Construction and Demolition Waste for Structural Concrete: Constituents, Properties and Production. Materials 2021, 14, 5748. [Google Scholar] [CrossRef]
- Barreto, E.D.S.; Stafanato, K.V.; Marvila, M.T.; de Azevedo, A.R.G.; Ali, M.; Pereira, R.M.L.; Monteiro, S.N. Clay Ceramic Waste as Pozzolan Constituent in Cement for Structural Concrete. Materials 2021, 14, 2917. [Google Scholar] [CrossRef]
- Fazli, A.; Rodrigue, D. Waste Rubber Recycling: A Review on the Evolution and Properties of Thermoplastic Elastomers. Materials 2020, 13, 782. [Google Scholar] [CrossRef] [PubMed]
- Borek, K.; Czapik, P.; Dachowski, R. Recycled Glass as a Substitute for Quartz Sand in Silicate Products. Materials 2020, 13, 1030. [Google Scholar] [CrossRef] [PubMed]
- Butenegro, J.A.; Bahrami, M.; Abenojar, J.; Martínez, M. Recent Progress in Carbon Fiber Reinforced Polymers Recycling: A Review of Recycling Methods and Reuse of Carbon Fibers. Materials 2021, 14, 6401. [Google Scholar] [CrossRef] [PubMed]
- Critical Raw Materials. n.d. Available online: https://single-market-economy.ec.europa.eu/sectors/raw-materials/areas-specific-interest/critical-raw-materials_en (accessed on 14 December 2022).
- Beaucamp, A.; Kirsch, B.; Zhu, W. Advances in grinding tools and abrasives. CIRP Ann. 2022, 71, 623–646. [Google Scholar] [CrossRef]
- German, R.; Suri, P.; Park, S. Review: Liquid phase sintering. J. Mater. Sci. 2009, 44, 1–39. [Google Scholar] [CrossRef]
- Katiyar, P.K.; Randhawa, N.S. A comprehensive review on recycling methods for cemented tungsten carbide scraps highlighting the electrochemical techniques. Int. J. Refract. Met. Hard Mater. 2020, 90, 105251. [Google Scholar] [CrossRef]
- Katiyar, P.K.; Randhawa, N.S. Corrosion behavior of WC-Co tool bits in simulated (concrete, soil, and mine) solutions with and without chloride additions. Int. J. Refract. Met. Hard Mater. 2019, 85, 105062. [Google Scholar] [CrossRef]
- Løvik, A.N.; Hagelüken, C.; Wäger, P. Improving supply security of critical metals: Current developments and research in the EU. Sustain. Mater. Technol. 2018, 15, 9–18. [Google Scholar] [CrossRef]
- Hofmann, M.; Hofmann, H.; Hagelüken, C.; Hool, A. Critical raw materials: A perspective from the materials science community. Sustain. Mater. Technol. 2018, 17, e00074. [Google Scholar] [CrossRef]
- Hool, A.; Schrijvers, D.; van Nielen, S.; Clifton, A.; Ganzeboom, S.; Hagelueken, C.; Harada, Y.; Kim, H.; Ku, A.; Meese-Marktscheffel, J.; et al. How companies improve critical raw material circularity: 5 use cases: Findings from the International Round Table on Materials Criticality. Miner. Econ. 2022, 35, 325–335. [Google Scholar] [CrossRef]
- Andersson, P. Chinese assessments of “critical” and “strategic” raw materials: Concepts, categories, policies, and implications. Extr. Ind. Soc. 2020, 7, 127–137. [Google Scholar] [CrossRef]
- Interior Seeks Public Comment on Draft List of 35 Minerals Deemed Critical to U.S. National Security and the Economy|U.S. Department of the Interior. n.d. Available online: https://www.doi.gov/pressreleases/interior-seeks-public-comment-draft-list-35-minerals-deemed-critical-us-national (accessed on 14 December 2022).
- Critical Minerals at Geoscience Australia|Geoscience Australia. n.d. Available online: https://www.ga.gov.au/scientific-topics/minerals/critical-minerals (accessed on 17 January 2023).
- Critical Materials Brazil. n.d. Available online: https://www.in.gov.br/web/dou/-/resolucao-n-2-de-18-de-junho-de-2021-327352416 (accessed on 17 January 2023).
- Shemi, A.; Magumise, A.; Ndlovu, S.; Sacks, N. Recycling of tungsten carbide scrap metal: A review of recycling methods and future prospects. Miner. Eng. 2018, 122, 195–205. [Google Scholar] [CrossRef]
- Han, Z.; Golev, A.; Edraki, M. A Review of Tungsten Resources and Potential Extraction from Mine Waste. Minerals 2021, 11, 701. [Google Scholar] [CrossRef]
- Exner, H.E. Physical and chemical nature of cemented carbides. Int. Met. Rev. 1979, 24, 149–173. [Google Scholar] [CrossRef]
- Eso, O.; Fang, Z.; Griffo, A. Liquid phase sintering of functionally graded WC–Co composites. Int. J. Refract. Met. Hard Mater. 2005, 23, 233–241. [Google Scholar] [CrossRef]
- Leal-Ayala, D.R.; Allwood, J.M.; Petavratzi, E.; Brown, T.J.; Gunn, G. Mapping the global flow of tungsten to identify key material efficiency and supply security opportunities. Resour. Conserv. Recycl. 2015, 103, 19–28. [Google Scholar] [CrossRef]
- Fan, K.; Jiang, W.; Luzin, V.; Gong, T.; Feng, W.; Ruiz-Hervias, J.; Yao, P. Influence of WC Particle Size on the Mechanical Properties and Residual Stress of HVOF Thermally Sprayed WC–10Co–4Cr Coatings. Materials 2022, 15, 5537. [Google Scholar] [CrossRef]
- Kapusta, J.P.T. Cobalt production and markets: A brief overview. Jom 2006, 58, 33–36. [Google Scholar] [CrossRef]
- Sun, X.; Hao, H.; Liu, Z.; Zhao, F.; Song, J. Tracing global cobalt flow: 1995–2015. Resour. Conserv. Recycl. 2019, 149, 45–55. [Google Scholar] [CrossRef]
- Wu, C.-C.; Chang, S.-H.; Tang, T.-P.; Peng, K.-Y.; Chang, W.-C. Study on the properties of WC-10Co alloys adding Cr 3 C 2 powder via various vacuum sintering temperatures. J. Alloys Compd. 2016, 686, 810–815. [Google Scholar] [CrossRef]
- Mégret, A.; Vitry, V.; Delaunois, F. Study of the Processing of a Recycled WC–Co Powder: Can It Compete with Conventional WC–Co Powders? J. Sustain. Met. 2021, 7, 448–458. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, C.; Wang, D.; Nie, L.; Wellmann, D.; Tian, Y. Additive manufacturing of WC-Co hardmetals: A review. Int. J. Adv. Manuf. Technol. 2020, 108, 1653–1673. [Google Scholar] [CrossRef]
- Pinna, E.G.; Toro, N.; Gallegos, S.; Rodriguez, M.H. A Novel Recycling Route for Spent Li-Ion Batteries. Materials 2021, 15, 44. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Liu, J.; Tang, H.; Ma, X.; Zhao, W. Investigation on the mechanical properties of WC–Fe–Cu hard alloys. J. Alloys Compd. 2015, 632, 729–734. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, J.; Tang, H.; Ma, X.; Zhao, W. Effect of Mo addition on the microstructure and properties of WC–Ni–Fe hard alloys. J. Alloys Compd. 2015, 646, 155–160. [Google Scholar] [CrossRef]
- Malyshev, V.V.; Gab, A.I. Resource-saving methods for recycling waste tungsten carbide-cobalt cermets and extraction of tungsten from tungsten concentrates. Theor. Found. Chem. Eng. 2007, 41, 436–441. [Google Scholar] [CrossRef]
- Martins, F.; Castro, H. Significance ranking method applied to some EU critical raw materials in a circular economy—Priorities for achieving sustainability. Procedia CIRP 2019, 84, 1059–1062. [Google Scholar] [CrossRef]
- Zeiler, B.; Bartl, A.; Schubert, W.-D. Recycling of tungsten: Current share, economic limitations, technologies and future potential. Int. J. Refract. Met. Hard Mater. 2021, 98, 105546. [Google Scholar] [CrossRef]
- Bhosale, S.; Mookherjee, S.; Pardeshi, R. Current Practices in Tungsten Extraction and Recovery. High Temp. Mater. Process. 1990, 9, 147–162. [Google Scholar] [CrossRef]
- Srivastava, R.R.; Lee, J.-C.; Bae, M.; Kumar, V. Reclamation of tungsten from carbide scraps and spent materials. J. Mater. Sci. 2018, 54, 83–107. [Google Scholar] [CrossRef]
- Mishra, D.; Sinha, S.; Sahu, K.; Agrawal, A.; Kumar, R. Recycling of Secondary Tungsten Resources. Trans. Indian Inst. Met. 2017, 70, 479–485. [Google Scholar] [CrossRef]
- Furberg, A.; Arvidsson, R.; Molander, S. Environmental life cycle assessment of cemented carbide (WC-Co) production. J. Clean. Prod. 2018, 209, 1126–1138. [Google Scholar] [CrossRef]
- Gaur, R.P.S. Modern hydrometallurgical production methods for tungsten. Jom 2006, 58, 45–49. [Google Scholar] [CrossRef]
- Wolfe, T.A.; Jewett, T.J.; Gaur, R.P.S. Powder Synthesis. Compr. Hard Mater. 2014, 1, 185–212. [Google Scholar] [CrossRef]
- Lassner, E. From Tungsten Concentrates and Scrap to Highly Pure Ammonium Paratungstate (APT). In The Chemistry of Non-Sag Tungsten; Elsevier: Amsterdam, The Netherlands, 1995; pp. 35–44. [Google Scholar] [CrossRef]
- Naboychenko, S.S.; Murashova, I.B.; Neikov, O.D. Production of Refractory Metal Powders. In Handbook of Non-Ferrous Metal Powders; Elsevier: Amsterdam, The Netherlands, 2009; pp. 436–484. [Google Scholar] [CrossRef]
- Soares, E.; Malheiros, L.F.; Sacramento, J.; Valente, M.A.; Oliveira, F.J. Ethanol and Water Processing of Submicrometer Cemented Carbide Powders. J. Am. Ceram. Soc. 2010, 94, 84–91. [Google Scholar] [CrossRef]
- Mehrotra, P.K. Powder Processing and Green Shaping. Compr. Hard Mater. 2014, 1, 213–235. [Google Scholar] [CrossRef]
- Zhang, F.; Zhu, M.; Wang, C. Parameters optimization in the planetary ball milling of nanostructured tungsten carbide/cobalt powder. Int. J. Refract. Met. Hard Mater. 2008, 26, 329–333. [Google Scholar] [CrossRef]
- ISO. ISO 14638:2015—Geometrical Product Specifications (GPS)—Matrix Model. n.d. Available online: https://www.iso.org/standard/57054.html (accessed on 14 December 2022).
- Santero, N.; Hendry, J. Harmonization of LCA methodologies for the metal and mining industry. Int. J. Life Cycle Assess. 2016, 21, 1543–1553. [Google Scholar] [CrossRef]
- Lassner, E.; Schubert, W.-D. Tungsten Scrap Recycling. In Tungsten; Springer: Boston, MA, USA, 1999; pp. 377–385. [Google Scholar] [CrossRef]
- Lee, J.-C.; Kim, E.-Y.; Kim, J.-H.; Kim, W.; Kim, B.-S.; Pandey, B.D. Recycling of WC–Co hardmetal sludge by a new hydrometallurgical route. Int. J. Refract. Met. Hard Mater. 2011, 29, 365–371. [Google Scholar] [CrossRef]
- Pacana, A.; Siwiec, D. Universal Model to Support the Quality Improvement of Industrial Products. Materials 2021, 14, 7872. [Google Scholar] [CrossRef]
- Zheng, H.; Cai, G.; Wang, S.; Yuan, S. An Experimental Study on Mechanism of Cermet Grinding. CIRP Ann. 1989, 38, 335–338. [Google Scholar] [CrossRef]
- Hegeman, J.; De Hosson, J.; de With, G. Grinding of WC–Co hardmetals. Wear 2001, 248, 187–196. [Google Scholar] [CrossRef]
- EP1681137B2—Grinding Device and Use of Same Grinding Device for Rectifying Cylindrical Items, Apparatus and Method for Rectifying Cylindrical Items—Google Patents. n.d. Available online: https://patents.google.com/patent/EP1681137B2/en?inventor=Giuseppe+Niesi (accessed on 14 December 2022).
- Gille, G.; Szesny, B.; Dreyer, K.; Berg, H.V.D.; Schmidt, J.; Gestrich, T.; Leitner, G. Submicron and ultrafine grained hardmetals for microdrills and metal cutting inserts. Int. J. Refract. Met. Hard Mater. 2002, 20, 3–22. [Google Scholar] [CrossRef]
- García, J.; Ciprés, V.C.; Blomqvist, A.; Kaplan, B. Cemented carbide microstructures: A review. Int. J. Refract. Met. Hard Mater. 2019, 80, 40–68. [Google Scholar] [CrossRef]
- Zinc Reclaimed Powders|Global Tungsten & Powders Corp. n.d. Available online: https://www.globaltungsten.com/en/products/zinc-reclaimed-powders.html (accessed on 14 December 2022).
- Freemantle, C.; Sacks, N.; Topic, M.; Pineda-Vargas, C. Impurity characterization of zinc-recycled WC-6wt.% Co cemented carbides. Int. J. Refract. Met. Hard Mater. 2014, 44, 94–102. [Google Scholar] [CrossRef]
- The Influence of Trace Elements on the Properties of Hard Metals—Publications Office of the EU. n.d. Available online: https://op.europa.eu/de/publication-detail/-/publication/9ea6173d-04ff-41d5-a07f-45f4afe54bb3 (accessed on 14 December 2022).
- Markoulides, D.; Koursaris, A.; Rafferty, M. Important factors which affect the removal of organic residues during the presintering of WC-Co. Int. J. Refract. Met. Hard Mater. 1997, 15, 123–131. [Google Scholar] [CrossRef]
- Fernandes, C.; Senos, A. Cemented carbide phase diagrams: A review. Int. J. Refract. Met. Hard Mater. 2011, 29, 405–418. [Google Scholar] [CrossRef]
- Roulon, Z.; Missiaen, J.-M.; Lay, S. Shrinkage and microstructure evolution during sintering of cemented carbides with alternative binders. Int. J. Refract. Met. Hard Mater. 2021, 101, 105665. [Google Scholar] [CrossRef]
- ISO. ISO 3369:2006—Impermeable Sintered Metal Materials and Hardmetals—Determination of Density. n.d. Available online: https://www.iso.org/standard/44435.html (accessed on 18 January 2023).
- ISO. ISO 4499-2:2020—Hardmetals—Metallographic Determination of Microstructure—Part 2: Measurement of WC Grain Size. n.d. Available online: https://www.iso.org/standard/74884.html (accessed on 18 January 2023).
- ISO. ISO 3738-1:1982—Hardmetals—Rockwell Hardness Test (scale A)—Part 1: Test Method. n.d. Available online: https://www.iso.org/standard/9225.html (accessed on 18 January 2023).
- ISO. ISO 3738-2:1988—Hardmetals—Rockwell Hardness Test (Scale A)—Part 2: Preparation and Calibration of Standard Test Blocks. n.d. Available online: https://www.iso.org/standard/9226.html (accessed on 18 January 2023).
- ISO. ISO 6507-1:2018. Metallic Materials—Vickers Hardness Test—Part 1: Test Method. Available online: https://www.iso.org/standard/64065.html (accessed on 18 January 2023).
- ISO. ISO 4499-4:2016(en), Hardmetals—Metallographic Determination of Microstructure—Part 4: Characterisation of Porosity, Carbon Defects and Eta-Phase Content. n.d. Available online: https://www.iso.org/obp/ui/fr/#iso:std:iso:4499:-4:ed-1:v1:en (accessed on 14 December 2022).
- Azcona, I.; Ordóñez, A.; Sanchez, J.M.; Castro, F. Hot isostatic pressing of ultrafine tungsten carbide-cobalt hardmetals. J. Mater. Sci. 2002, 37, 4189–4195. [Google Scholar] [CrossRef]
- Pang, R.; Zhang, X. Achieving environmental sustainability in manufacture: A 28-year bibliometric cartography of green manufacturing research. J. Clean. Prod. 2019, 233, 84–99. [Google Scholar] [CrossRef]
- Rossi, A.; Lanzetta, M. Integration of hybrid additive/subtractive manufacturing planning and scheduling by metaheuristics. Comput. Ind. Eng. 2020, 144, 106428. [Google Scholar] [CrossRef]



| Main Contaminants | Input Chips | Recycled Powder | Upper Limit [65,66,67] |
|---|---|---|---|
| C | 5.92 | 5.83 | Carbon content range 1 |
| O | 3.60 | 0.80 | 0.50 |
| Cr | 0.46 | 0.41 | 0.60 |
| Al | 0.10 | 0.25 | 0.05 |
| Si | 0.10 | 0.15 | 0.10 |
| Mo | 0.10 | 0.03 | 0.10 |
| V | 0.09 | 0.11 | 0.20 |
| Ni | 0.16 | 0.08 | 0.50 |
| Fe | 0.06 | 0.12 | 0.30 |
| Cu | 0.02 | 0.02 | 0.03 |
| Ca | 0.06 | 0.04 | 0.01 |
| S | 0.06 | 0.05 | 0.01 |
| Properties | Reference ISO Standard | Recycled Powder Sintered | Commercial “Virgin” Powder Sintered [65] |
|---|---|---|---|
| Density (g/cm3) | ISO 3369 [71] | 14 | 14.4 |
| Grain size (µm) | ISO 4499-2 [72] | 3 (medium) | 1–3 (medium-fine) |
| Hardness (HRA) | ISO 3738-1/2 [73,74] | 90.7 | 90.7 |
| Hardness (HV30) | ISO 6507-1 [75] | 1430 | 1435 |
| Porosity (grade A-B-C) | ISO4499-4 [76] | A04-06 | A02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacini, A.; Lupi, F.; Rossi, A.; Seggiani, M.; Lanzetta, M. Direct Recycling of WC-Co Grinding Chip. Materials 2023, 16, 1347. https://doi.org/10.3390/ma16041347
Pacini A, Lupi F, Rossi A, Seggiani M, Lanzetta M. Direct Recycling of WC-Co Grinding Chip. Materials. 2023; 16(4):1347. https://doi.org/10.3390/ma16041347
Chicago/Turabian StylePacini, Alessio, Francesco Lupi, Andrea Rossi, Maurizia Seggiani, and Michele Lanzetta. 2023. "Direct Recycling of WC-Co Grinding Chip" Materials 16, no. 4: 1347. https://doi.org/10.3390/ma16041347
APA StylePacini, A., Lupi, F., Rossi, A., Seggiani, M., & Lanzetta, M. (2023). Direct Recycling of WC-Co Grinding Chip. Materials, 16(4), 1347. https://doi.org/10.3390/ma16041347










