Three-Dimensional Printing of Multifunctional Composites: Fabrication, Applications, and Biodegradability Assessment
Abstract
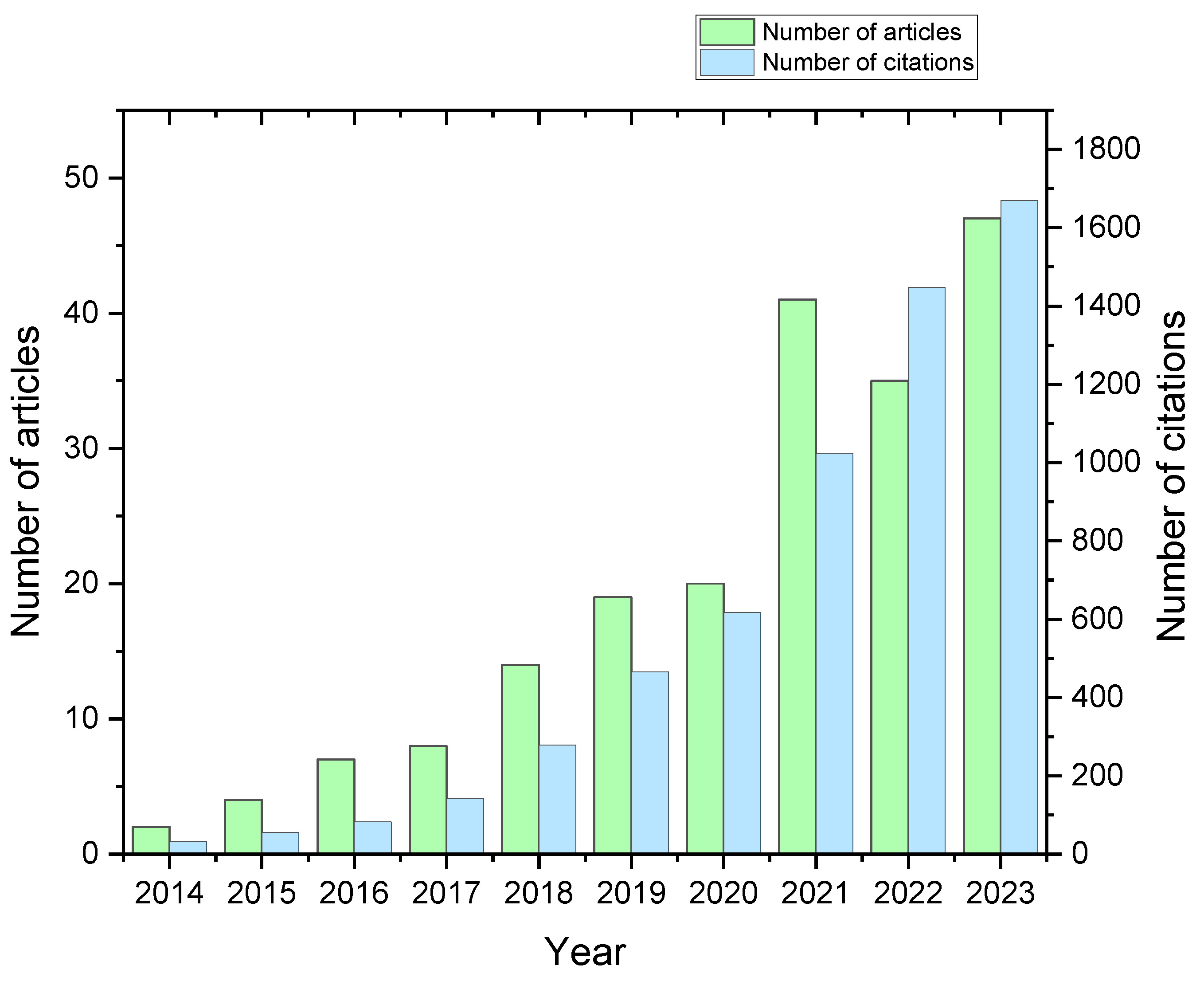
:1. Introduction
2. Multi-Material 3D-Printed Polymer Composites
Three-Dimensional Printing of Composites
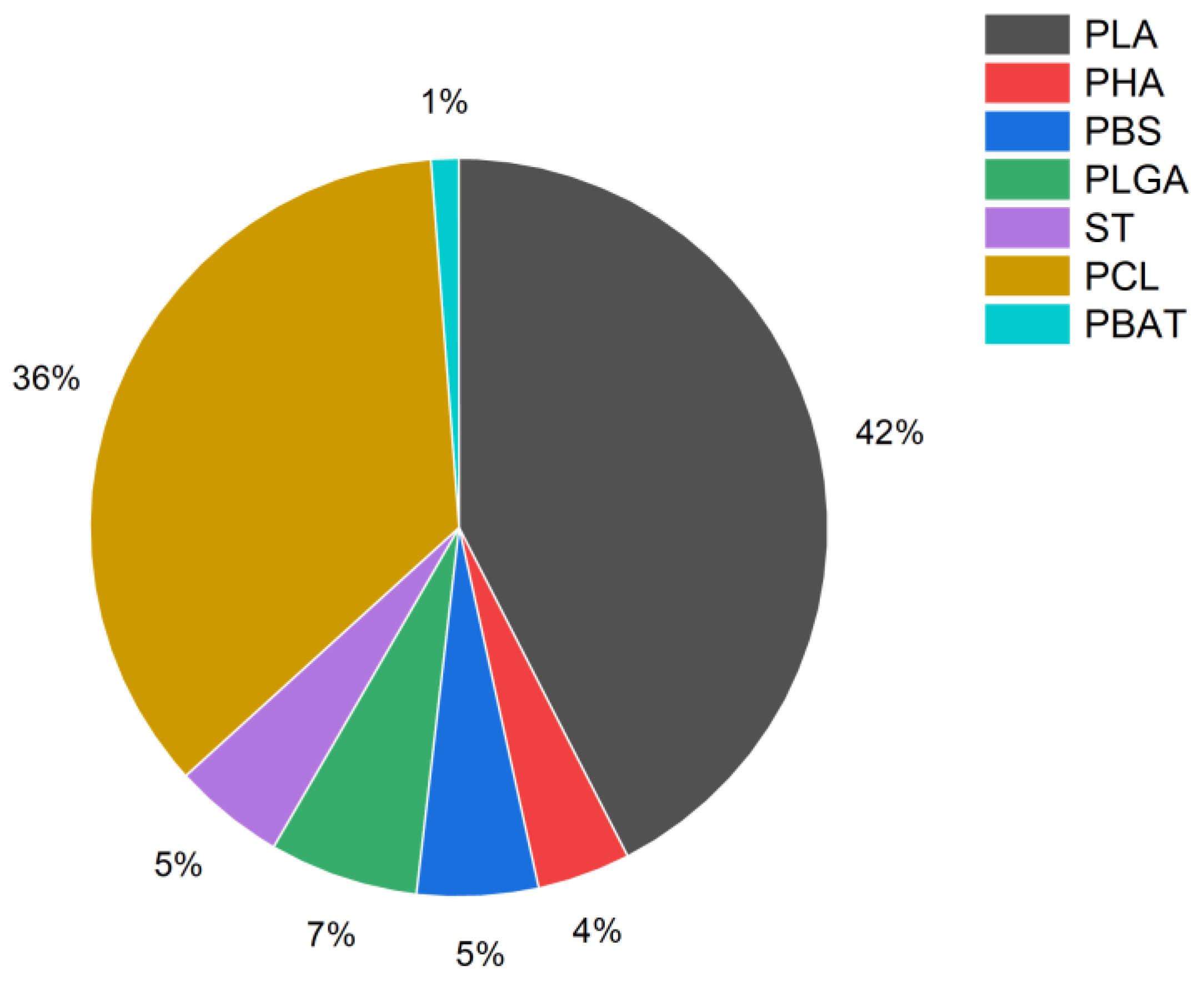
3. Biodegradable Polymers in 3D Printing
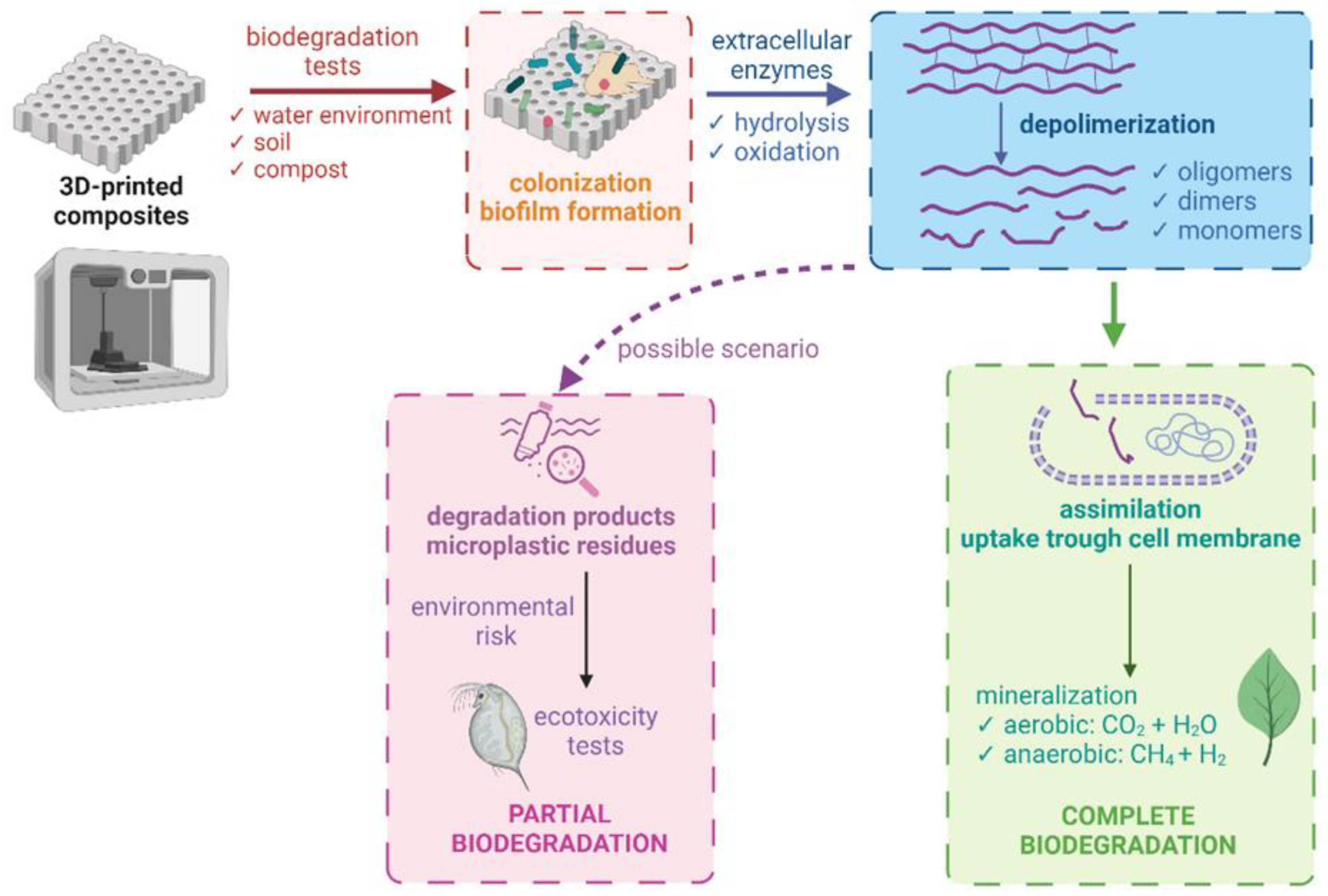
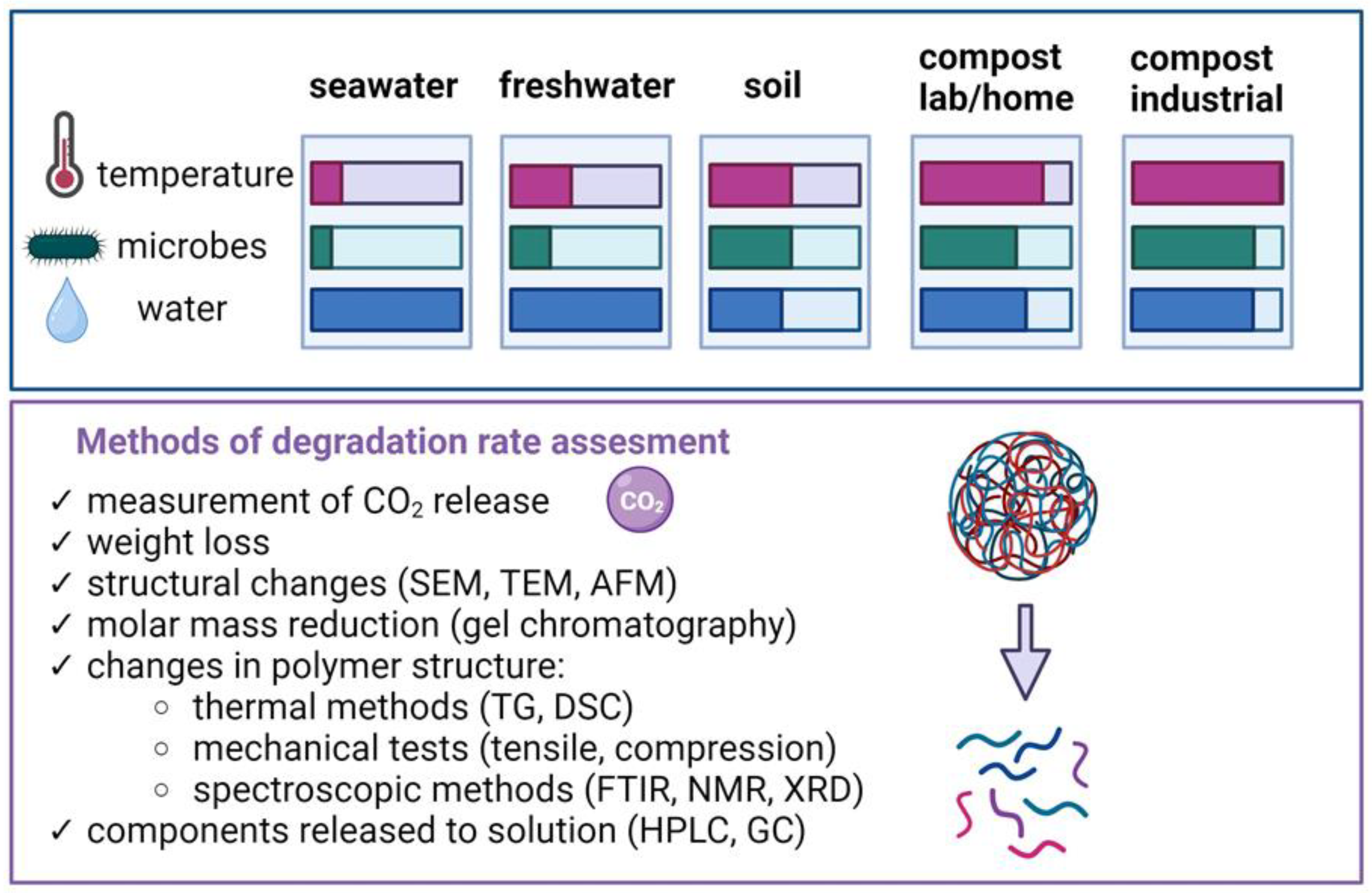
3.1. Methods for Evaluating the Biodegradability of Composites Produced Using 3D Printing Technologies
3.2. Biodegradation of Composites in Aquatic Environments
3.2.1. Biodegradation in Freshwater and Seawater
3.2.2. Degradation Tests in Buffer Solutions for Medical Applications
| Printed Material | AM Technique | T (°C) | Time (Days) | Solution Type /Test Type | Indicators | Biodegradation Level | References |
|---|---|---|---|---|---|---|---|
| PLA, PHB, PLA/PHB | FDM | 25 | 50 | Freshwater aerobic environment, thermophilic microorganisms | CO2 release | PLA 8.7%, PHB 73.3%, and PLA50/PHB50 32.3% | Choe et al. [201] |
| PCL + wool | FDM | 20 | 5 | Marine water collected from Eastern Beach, Geelong, Australia | BS EN 1899-2:1998 [202], CO2 release | n.a. | Haque et al. [194] |
| PCL + wool | FDM | 25 | 5 months | Domestic saltwater fish tank as an established ecosystem | Weight loss | n.a. | Haque et al. [194] |
| PBAT + HAp | FDM | 37 | 30 | Tris-buffer | Weight loss | 6.21 for 3% HAP | Acharya et al. [203] |
| PBAT/Chitosan | FDM | 37 | 168 | ISO 13781:2017 [204] Sorensen buffer solution (0.2 M, pH 7.4) | Weight loss, change in molecular weight | 14.17% | Wang et al. [196] |
| PLA | FDM | n.a. | 21 | PBS + 5% CO2 | Weight loss, morphology changes (scan) | n.a. | Karimipour-Fard et al. [197] |
| PLA/PHB | FDM | 37 | 195 | Saline, PBS, and Hank’s solution | Solutions absorption, microscopic observation, mechanical compressive tests | n.a. | Balogová et al. [205] |
| PLGA | FDM | 37 47 | 56 28 | ISO 13781:2017 [204] PBS | Visual changes, weight loss, thermal properties, molecular weight change, mechanical properties | 56 days at 37 °C: 2.12% mass lost, molecular weight decrease 39.5%; accelerated degradation: 4.38% mass lost, molecular weight decrease 92.4% | Ghosh Dastidar et al. [199] |
| PLGA/HA/CNT | FDM | 37 | 28 | PBS | Weight loss | n.a. | Kaya et al. [135] |
| PLC + fiber yarn | FDM | 37 | 70 | Cell culture medium, Roswell Park Memorial Institute (RPMI) 1640 | Weight loss, visual changes (SEM), mechanical properties | Degradation rate 20 times higher for biomass-reinforced samples | Hedayati et al. [198] |
3.3. Biodegradation of Composites in Soil Environments
| Printed Material | AM Technique | T (°C) | Time | Humidity (%) | Soil Type | Indicators | Biodegradation Level (%) | References |
|---|---|---|---|---|---|---|---|---|
| PLA/PHA-wood fiber | FDM | 30 | 30 days | 80 | Coco peat, red scorched soil, fine sand, charcoal, and microorganisms | Weight loss | 2.47–3.85% | Sekar et al. [209] |
| PLA/PHA-wood fiber | FDM | 30 | 28 days | 80 | Coco peat, red burnt soil, fine sand, charcoal, and microbes | Weight loss | 2.45% | Sekar et al. [209] |
| PLA-TPS-wood | FDM | 30 | 4 months | 85 | Forest soil | Weight loss, thermal properties, FTIR | PLA 0.5% PLA-TPS 1–18% | Lee et al. [207] |
| PLA-TPS-ARP | FDM | Room | 180 days | 17–21.5% | n.a. | Weight loss, surface changes, mechanical properties, thermal stability, and thermal dynamic mechanic testing | 21.40% | Ni et al. [206] |
| PLA/PP | FDM | n.a. | 45 days | n.a. | n.a. | Mechanical parameters (tensile strength) | n.a. | Harris et al. [210] |
| PLA/rice husk | FDM | 21–25 | 90 days | 30% | n.a. | Weight loss | Weight loss up to 40% | Tsou et al. [208] |
3.4. Biodegradation in a Composter
| Printed Material | AM Method | T (°C) | Time (Days) | Compost Type | Indicators | Biodegradation Test Type | Biodegradation Rate | References |
|---|---|---|---|---|---|---|---|---|
| PLA, PHB, PLA/PHB | FDM | 58 | 50 | Thermophilic microorganisms | CO2 release | Laboratory scale ASTM D5338-15 [214], ISO 14852 [215] | PHB—86.4% PLA50/PHB50—85% PLA—21.7% | Choe et al. [201] |
| PLA, PLA/PHA | FDM | 58 | 84 | From a sorting and composting plant | CO2 release | Laboratory scale ASTM D6400 [216] PN-EN 14806:2010 [217] | PLA 21% PLA/PHA 30% | Rydz et al. [212] |
| PLA, PLA/PHA | FDM | 60 61 | 21 21–84 | BIODEGMA system static composting open-air pile, industrial system | Macroscopic visual evaluation | Industrial scale, sorting and composting plant, Zabrze, Poland | n.a. | Rydz et al. [212] |
| PCL/collagen hydrolysate | FDM | 58 | 30 | Olive mill waste (83 wt.% pomace and 6 wt.% leaves and twigs), waste wool (6 wt.%), wheat straw (3 wt.%), and chicken manure (2 wt.%) | Macroscopic visual evaluation | Composting pile (1 m3) UNI EN ISO 14045 [218] | Complete disintegration in 30 days | Seggiani et al. [219] |
3.5. Ecotoxicity of Composite Degradation Products
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| FDM | Fused deposition modeling |
| PBAT | Polybutylene adipate terephthalate |
| PBS | Phosphate-buffered saline |
| PBS | Polybutylene succinate |
| PCL | Polycaprolactone |
| PE-g-MAH | Maleic anhydride grafted polyethylene |
| PHA | Polyhydroxyalkanoate |
| PLA | Polylactic acid |
| PLGA | Poly lactic-co-glycolic acid |
| PP | Polypropylene |
| HAp | Hydroxyapatite |
References
- Nugroho, W.T.; Dong, Y.; Pramanik, A. 3D printing composite materials: A comprehensive review. In Composite Materials; Elsevier: Amsterdam, The Netherlands, 2021; pp. 65–115. [Google Scholar] [CrossRef]
- Zhou, L.; Fu, J.; He, Y. A Review of 3D Printing Technologies for Soft Polymer Materials. Adv. Funct. Mater. 2020, 30, 28. [Google Scholar] [CrossRef]
- Li, N.; Huang, S.; Zhang, G.; Qin, R.; Liu, W.; Xiong, H.; Shi, G.; Blackburn, J. Progress in additive manufacturing on new materials: A review. J. Mater. Sci. Technol. 2019, 35, 242–269. [Google Scholar] [CrossRef]
- Bhagia, S.; Bornani, K.; Agarwal, R.; Satlewal, A.; Ďurkovič, J.; Lagaňa, R.; Bhagia, M.; Yoo, C.; Zhao, X.; Kunc, V.; et al. Critical review of FDM 3D printing of PLA biocomposites filled with biomass resources, characterization, biodegradability, upcycling and opportunities for biorefineries. Appl. Mater. Today 2021, 24, 101078. [Google Scholar] [CrossRef]
- Chandna, P.; Nain, L.; Singh, S.; Kuhad, R.C. Assessment of bacterial diversity during composting of agricultural byproducts. BMC Microbiol. 2013, 13, 99. [Google Scholar] [CrossRef] [PubMed]
- Bodkhe, S.; Ermanni, P. 3D printing of multifunctional materials for sensing and actuation: Merging piezoelectricity with shape memory. Eur. Polym. J. 2020, 132, 109738. [Google Scholar] [CrossRef]
- Bekas, D.G.; Hou, Y.; Liu, Y.; Panesar, A. 3D printing to enable multifunctionality in polymer-based composites: A review. Compos. B Eng. 2019, 179, 107540. [Google Scholar] [CrossRef]
- Jakus, A.E. An Introduction to 3D Printing—Past, Present, and Future Promise. In 3D Printing in Orthopaedic Surgery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–15. [Google Scholar] [CrossRef]
- Backes, E.H. Engineering 3D printed bioactive composite scaffolds based on the combination of aliphatic polyester and calcium phosphates for bone tissue regeneration. Mater. Sci. Eng. 2021, 122, 111928. [Google Scholar] [CrossRef]
- Ribeiro, J.F.M. Structural monitoring and modeling of the mechanical deformation of three-dimensional printed poly(ε-caprolactone) scaffolds. Biofabrication 2017, 9, 025015. [Google Scholar] [CrossRef]
- Archisman Dasgupta, P.D. A Comprehensive Review on 3D Printing Technology: Current Applications and Challenges. Jordan J. Mech. Ind. Eng. 2022, 16, 529–542. [Google Scholar]
- Tümer, E.H.; Erbil, H.Y. Extrusion-Based 3D Printing Applications of PLA Composites: A Review. Coatings 2021, 11, 390. [Google Scholar] [CrossRef]
- Rong, L. The application of 3D printing technology on starch-based product: A review. Trends Food Sci. Technol. 2023, 134, 149–161. [Google Scholar] [CrossRef]
- Li, Y.; Ren, X.; Zhu, L.; Li, C. Biomass 3D Printing: Principles, Materials, Post-Processing and Applications. Polymers 2023, 15, 2692. [Google Scholar] [CrossRef] [PubMed]
- Nikzad, M.; Masood, S.H.; Sbarski, I. Thermo-mechanical properties of a highly filled polymeric composites for Fused Deposition Modeling. Mater. Des. 2011, 32, 3448–3456. [Google Scholar] [CrossRef]
- ISO/ASTM 52900:2021(en); Additive Manufacturing—General Principles—Fundamentals and Vocabulary. International Organization for Standardization: Geneva, Switzerland, 2015.
- Hwang, S.; Reyes, E.I.; Moon, K.; Rumpf, R.C.; Kim, N.S. Thermo-mechanical Characterization of Metal/Polymer Composite Filaments and Printing Parameter Study for Fused Deposition Modeling in the 3D Printing Process. J. Electron. Mater. 2015, 44, 771–777. [Google Scholar] [CrossRef]
- Ayrilmis, N.; Kariz, M.; Kwon, J.H.; Kitek Kuzman, M. Effect of printing layer thickness on water absorption and mechanical properties of 3D-printed wood/PLA composite materials. Int. J. Adv. Manuf. Technol. 2019, 102, 2195–2200. [Google Scholar] [CrossRef]
- Boparai, K.; Singh, R.; Singh, H. Comparison of tribological behaviour for Nylon6-Al-Al2O3 and ABS parts fabricated by fused deposition modelling. Virtual Phys. Prototyp. 2015, 10, 59–66. [Google Scholar] [CrossRef]
- Castles, F.; Isakov, D.; Lui, A.; Lei, Q.; Dancer, C.E.J.; Wang, Y.; Janurudin, J.M.; Speller, S.C.; Grovenor, C.R.M.; Grant, P.S. Microwave dielectric characterisation of 3D-printed BaTiO3/ABS polymer composites. Sci. Rep. 2016, 6, 22714. [Google Scholar] [CrossRef] [PubMed]
- Shemelya, C.M.; Rivera, A.; Perez, A.T.; Rocha, C.; Liang, M.; Yu, X.; Kief, C.; Alexander, D.; Stegeman, J.; Xin, H.; et al. Mechanical, Electromagnetic, and X-ray Shielding Characterization of a 3D Printable Tungsten–Polycarbonate Polymer Matrix Composite for Space-Based Applications. J. Electron. Mater. 2015, 44, 2598–2607. [Google Scholar] [CrossRef]
- Torrado Perez, A.R.; Roberson, D.A.; Wicker, R.B. Fracture Surface Analysis of 3D-Printed Tensile Specimens of Novel ABS-Based Materials. J. Fail. Anal. Prev. 2014, 14, 343–353. [Google Scholar] [CrossRef]
- Zhong, W.; Li, F.; Zhang, Z.; Song, L.; Li, Z. Short fiber reinforced composites for fused deposition modeling. Mater. Sci. Eng. 2001, 301, 125–130. [Google Scholar] [CrossRef]
- Tekinalp, H.L.; Kunc, V.; Velez-Garcia, G.M.; Duty, C.E.; Love, L.J.; Naskar, A.K.; Blue, C.A.; Ozcan, S. Highly oriented carbon fiber–polymer composites via additive manufacturing. Compos. Sci. Technol. 2014, 105, 144–150. [Google Scholar] [CrossRef]
- Ning, F.; Cong, W.; Qiu, J.; Wei, J.; Wang, S. Additive manufacturing of carbon fiber reinforced thermoplastic composites using fused deposition modeling. Compos. B Eng. 2015, 80, 369–378. [Google Scholar] [CrossRef]
- Carneiro, O.S.; Silva, A.F.; Gomes, R. Fused deposition modeling with polypropylene. Mater. Des. 2015, 83, 768–776. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Weng, Z.; Senthil, T.; Wu, L. A novel approach to improve mechanical properties of parts fabricated by fused deposition modeling. Mater. Des. 2016, 105, 152–159. [Google Scholar] [CrossRef]
- Shofner, M.L.; Lozano, K.; Rodríguez-Macías, F.J.; Barrera, E.V. Nanofiber-reinforced polymers prepared by fused deposition modeling. J. Appl. Polym. Sci. 2003, 89, 3081–3090. [Google Scholar] [CrossRef]
- Le Duigou, A.; Castro, M.; Bevan, R.; Martin, N. 3D printing of wood fibre biocomposites: From mechanical to actuation functionality. Mater. Des. 2016, 96, 106–114. [Google Scholar] [CrossRef]
- Matsuzaki, R.; Ueda, M.; Namiki, M.; Jeong, T.-K.; Asahara, H.; Horiguchi, K.; Nakamura, T.; Todoroki, A.; Hirano, Y. Three-dimensional printing of continuous-fiber composites by in-nozzle impregnation. Sci. Rep. 2016, 6, 23058. [Google Scholar] [CrossRef]
- Li, N.; Li, Y.; Liu, S. Rapid prototyping of continuous carbon fiber reinforced polylactic acid composites by 3D printing. J. Mater. Process Technol. 2016, 238, 218–225. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Mori, K.; Maeno, T. 3D printing of carbon fibre-reinforced plastic parts. Int. J. Adv. Manuf. Technol. 2017, 91, 2811–2817. [Google Scholar] [CrossRef]
- Zhong, W.; Li, F.; Zhang, Z.; Song, L.; Li, Z. Research on rapid-prototyping/part manufacturing for the continuous fiber reinforced composite. Mater. Manuf. Process. 2001, 16, 17–26. [Google Scholar] [CrossRef]
- Weng, Z.; Wang, J.; Senthil, T.; Wu, L. Mechanical and thermal properties of ABS/montmorillonite nanocomposites for fused deposition modeling 3D printing. Mater. Des. 2016, 102, 276–283. [Google Scholar] [CrossRef]
- Wei, X.; Wei, X.; Li, D.; Jiang, W.; Gu, Z.; Wang, X.; Zhang, Z.; Sun, Z. 3D Printable Graphene Composite. Sci. Rep. 2015, 5, 11181. [Google Scholar] [CrossRef] [PubMed]
- Zein, I.; Hutmacher, D.W.; Tan, K.C.; Teoh, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.J.; Fiore, B.E.; Erb, R.M. Designing bioinspired composite reinforcement architectures via 3D magnetic printing. Nat. Commun. 2015, 6, 8641. [Google Scholar] [CrossRef] [PubMed]
- Kokkinis, D.; Schaffner, M.; Studart, A.R. Multimaterial magnetically assisted 3D printing of composite materials. Nat. Commun. 2015, 6, 8643. [Google Scholar] [CrossRef] [PubMed]
- Compton, B.G.; Lewis, A. 3D-Printing of Lightweight Cellular Composites. Adv. Mater. 2014, 26, 5930–5935. [Google Scholar] [CrossRef] [PubMed]
- Van Der Klift, F.; Koga, Y.; Todoroki, A.; Ueda, M.; Hirano, Y.; Matsuzaki, R. 3D Printing of Continuous Carbon Fibre Reinforced Thermo-Plastic (CFRTP) Tensile Test Specimens. Open J. Compos. Mater. 2016, 6, 18–27. [Google Scholar] [CrossRef]
- Yan, C.; Hao, L.; Xu, L.; Shi, Y. Preparation, characterisation and processing of carbon fibre/polyamide-12 composites for selective laser sintering. Compos. Sci. Technol. 2011, 71, 1834–1841. [Google Scholar] [CrossRef]
- Chung, H.; Das, S. Processing and properties of glass bead particulate-filled functionally graded Nylon-11 composites produced by selective laser sintering. Mater. Sci. Eng. 2006, 437, 226–234. [Google Scholar] [CrossRef]
- Goodridge, R.D.; Shofner, M.L.; Hague, R.J.M.; McClelland, M.; Schlea, M.R.; Johnson, R.B.; Tuck, C.J. Processing of a Polyamide-12/carbon nanofibre composite by laser sintering. Polym. Test. 2011, 30, 94–100. [Google Scholar] [CrossRef]
- Lin, D.; Jin, S.; Zhang, F.; Wang, C.; Wang, Y.; Zhou, C.; Cheng, G.J. 3D stereolithography printing of graphene oxide reinforced complex architectures. Nanotechnology 2015, 26, 434003. [Google Scholar] [CrossRef]
- Yugang, D.; Yuan, Z.; Yiping, T.; Dichen, L. Nano-TiO2-modified photosensitive resin for RP. Rapid Prototyp. J. 2011, 17, 247–252. [Google Scholar] [CrossRef]
- Kim, K.; Zhu, W.; Qu, X.; Aaronson, C.; McCall, W.R.; Chen, S.; Sirbuly, D.J. 3D Optical Printing of Piezoelectric Nanoparticle–Polymer Composite Materials. ACS Nano 2014, 8, 9799–9806. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, H.; Yang, X.; Zhang, T.; Zhu, K.; Si, W.; Liu, Z.; Sun, H. Additive manufacturing of carbon nanotube-photopolymer composite radar absorbing materials. Polym. Compos. 2018, 39, 2. [Google Scholar] [CrossRef]
- Athreya, S.R.; Kalaitzidou, K.; Das, S. Processing and characterization of a carbon black-filled electrically conductive Nylon-12 nanocomposite produced by selective laser sintering. Mater. Sci. Eng. 2010, 527, 2637–2642. [Google Scholar] [CrossRef]
- Zheng, H.; Zhang, J.; Lu, S.; Wang, G.; Xu, Z. Effect of core–shell composite particles on the sintering behavior and properties of nano-Al2O3/polystyrene composite prepared by SLS. Mater. Lett. 2006, 60, 1219–1223. [Google Scholar] [CrossRef]
- Kim, H.C.; Hahn, H.T.; Yang, Y.S. Synthesis of PA12/functionalized GNP nanocomposite powders for the selective laser sintering process. J. Compos. Mater. 2013, 47, 501–509. [Google Scholar] [CrossRef]
- Lin, D.; Richard Liu, C.; Cheng, G.J. Single-layer graphene oxide reinforced metal matrix composites by laser sintering: Microstructure and mechanical property enhancement. Acta Mater. 2014, 80, 183–193. [Google Scholar] [CrossRef]
- Kurimoto, M.; Yamashita, Y.; Ozaki, H.; Kato, T.; Funabashi, T.; Suzuoki, Y. 3D printing of conical insulating spacer using alumina/UV-cured-resin composite. In Proceedings of the IEEE Conference on Electrical Insulation and Dielectric Phenomena (CEIDP), Ann Arbor, MI, USA, 18–21 October 2015; IEEE: New York City, NY, USA, 2015; pp. 463–466. [Google Scholar] [CrossRef]
- Kalsoom, U.; Peristyy, A.; Nesterenko, P.N.; Paull, B. A 3D printable diamond polymer composite: A novel material for fabrication of low cost thermally conducting devices. RSC Adv. 2016, 6, 38140–38147. [Google Scholar] [CrossRef]
- Sandoval, H.J.; Wicker, R.B. Functionalizing stereolithography resins: Effects of dispersed multi-walled carbon nanotubes on physical properties. Rapid Prototyp. J. 2006, 12, 292–303. [Google Scholar] [CrossRef]
- Zanchetta, E.; Cattaldo, M.; Franchin, G.; Schwentenwein, M.; Homa, J.; Brusatin, G.; Colombo, P. Stereolithography of SiOC Ceramic Microcomponents. Adv. Mater. 2016, 28, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Suwanprateeb, J. Improvement in mechanical properties of three-dimensional printing parts made from natural polymers reinforced by acrylate resin for biomedical applications: A double infiltration approach. Polym. Int. 2006, 55, 57–62. [Google Scholar] [CrossRef]
- Guo, S.; Yang, X.; Heuzey, M.-C.; Therriault, D. 3D printing of a multifunctional nanocomposite helical liquid sensor. Nanoscale 2015, 7, 6451–6456. [Google Scholar] [CrossRef] [PubMed]
- Krivec, M.; Roshanghias, A.; Abram, A.; Binder, A. Exploiting the combination of 3D polymer printing and inkjet Ag-nanoparticle printing for advanced packaging. Microelectron. Eng. 2017, 176, 1–5. [Google Scholar] [CrossRef]
- Shao, H.; He, Y.; Fu, J.; He, D.; Yang, X.; Xie, J.; Yao, C.; Ye, J.; Xu, S.; Gou, Z. 3D printing magnesium-doped wollastonite/β-TCP bioceramics scaffolds with high strength and adjustable degradation. J. Eur. Ceram. Soc. 2016, 36, 1495–1503. [Google Scholar] [CrossRef]
- Wu, C.; Luo, Y.; Cuniberti, G.; Xiao, Y.; Gelinsky, M. Three-dimensional printing of hierarchical and tough mesoporous bioactive glass scaffolds with a controllable pore architecture, excellent mechanical strength and mineralization ability. Acta Biomater. 2011, 7, 2644–2650. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, C.; Lindner, M.; Zhang, W.; Koczur, K.; Kirsten, A.; Telle, R.; Fischer, H. 3D printing of bone substitute implants using calcium phosphate and bioactive glasses. J. Eur. Ceram. Soc. 2010, 30, 2563–2567. [Google Scholar] [CrossRef]
- Lam, C.X.F.; Mo, X.M.; Teoh, S.H.; Hutmacher, D.W. Scaffold development using 3D printing with a starch-based polymer. Mater. Sci. Eng. 2002, 20, 49–56. [Google Scholar] [CrossRef]
- Zhou, Z.; Cunningham, E.; Lennon, A.; McCarthy, H.O.; Buchanan, F.; Dunne, N. Development of three-dimensional printing polymer-ceramic scaffolds with enhanced compressive properties and tuneable resorption. Mater. Sci. Eng. 2018, 93, 975–986. [Google Scholar] [CrossRef]
- Vaezi, M.; Chua, C.K. Effects of layer thickness and binder saturation level parameters on 3D printing process. Int. J. Adv. Manuf. Technol. 2011, 53, 275–284. [Google Scholar] [CrossRef]
- Glasschroeder, J.; Prager, E.; Zaeh, M.F. Powder-bed-based 3D-printing of function integrated parts. Rapid Prototyp. J. 2015, 21, 207–215. [Google Scholar] [CrossRef]
- Hui, D.; Goodridge, R.D.; Scotchford, C.A.; Grant, D.M. Laser sintering of nano-hydroxyapatite coated polyamide 12 powders. Addit. Manuf. 2018, 22, 560–570. [Google Scholar] [CrossRef]
- Schwentenwein, M.; Homa, J. Additive Manufacturing of Dense Alumina Ceramics. Int. J. Appl. Ceram. Technol. 2015, 12, 1–7. [Google Scholar] [CrossRef]
- Salmoria, G.V.; Klauss, P.; Paggi, R.A.; Kanis, L.A.; Lago, A. Structure and mechanical properties of cellulose based scaffolds fabricated by selective laser sintering. Polym. Test. 2009, 28, 648–652. [Google Scholar] [CrossRef]
- Quill, T.J.; Smith, M.K.; Zhou, T.; Baioumy, M.G.S.; Berenguer, J.P.; Cola, B.A.; Kalaitzidou, K.; Bougher, T.L. Thermal and mechanical properties of 3D printed boron nitride—ABS composites. Appl. Compos. Mater. 2018, 25, 1205–1217. [Google Scholar] [CrossRef]
- Liu, J.; Li, W.; Guo, Y.; Zhang, H.; Zhang, Z. Improved thermal conductivity of thermoplastic polyurethane via aligned boron nitride platelets assisted by 3D printing. Compos. Part A Appl. Sci. Manuf. 2019, 20, 140–146. [Google Scholar] [CrossRef]
- Belaid, H.; Nagarajan, S.; Barou, C.; Huon, V.; Bares, J.; Balme, S.; Miele, P.; Cornu, D.; Cavaillès, V.; Teyssier, C.; et al. Boron Nitride Based Nanobiocomposites: Design by 3D Printing for Bone Tissue Engineering. ACS Appl. Bio Mater. 2020, 3, 1865–1874. [Google Scholar] [CrossRef]
- Su, K.-H.; Su, C.-Y.; Shih, W.-L.; Lee, F.-T. Improvement of the Thermal Conductivity and Mechanical Properties of 3D-Printed Polyurethane Composites by Incorporating Hydroxylated Boron Nitride Functional Fillers. Materials 2022, 16, 356. [Google Scholar] [CrossRef]
- Peng, Z.; Lv, Q.; Jing, J.; Pei, H.; Chen, Y.; Ivanov, E. FDM-3D printing LLDPE/BN@GNPs composites with double network structures for high-efficiency thermal conductivity and electromagnetic interference shielding. Compos. B Eng. 2023, 251, 110491. [Google Scholar] [CrossRef]
- Kowalewska, E.; Ficek, M.; Formela, K.; Zieliński, A.; Kunuku, S.; Sawczak, M.; Bogdanowicz, R. Tailoring of Optical Properties of Methacrylate Resins Enriched by HPHT Microdiamond Particles. Nanomaterials 2022, 12, 2604. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, M.; Zhou, Z.; Gou, J.; Hui, D. 3D printing of polymer matrix composites: A review and prospective. Compos. B Eng. 2017, 110, 442–458. [Google Scholar] [CrossRef]
- Bogdanov, K.V.; Zhukovskaya, M.V.; Osipov, V.Y.; Ushakova, E.V.; Baranov, M.A.; Takai, K.; Rampersaud, A.; Baranov, A.V. Highly intensive emission of the NV− centers in synthetic HPHT microdiamonds at low nitrogen doping. APL Mater. 2018, 6, 8. [Google Scholar] [CrossRef]
- Yaragatti, N.; Patnaik, A. A review on additive manufacturing of polymers composites. Mater. Today Proc. 2021, 44, 4150–4157. [Google Scholar] [CrossRef]
- Jariwala, D.; Sangwan, V.K.; Lauhon, L.J.; Marks, T.J.; Hersam, M.C. Carbon nanomaterials for electronics, optoelectronics, photovoltaics, and sensing. Chem. Soc. Rev. 2013, 42, 2824–2860. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, S. Polymer/Carbon Nanotubes (CNT) Nanocomposites Processing Using Additive Manufacturing (Three-Dimensional Printing) Technique: An Overview. Fibers 2017, 5, 40. [Google Scholar] [CrossRef]
- Gnanasekaran, K.; Heijmans, T.; van Bennekom, S.; Woldhuis, H.; Wijnia, S.; de With, G.; Friedrich, H. 3D printing of CNT- and graphene-based conductive polymer nanocomposites by fused deposition modeling. Appl. Mater. Today 2017, 9, 21–28. [Google Scholar] [CrossRef]
- Yang, L.; Li, S.; Zhou, X.; Liu, J.; Li, Y.; Yang, M.; Yuan, Q.; Zhang, W. Effects of carbon nanotube on the thermal, mechanical, and electrical properties of PLA/CNT printed parts in the FDM process. Synth. Met. 2019, 253, 122–130. [Google Scholar] [CrossRef]
- Lage-Rivera, S.; Ares-Pernas, A.; Becerra Permuy, J.C.; Gosset, A.; Abad, M.-J. Enhancement of 3D Printability by FDM and Electrical Conductivity of PLA/MWCNT Filaments Using Lignin as Bio-Dispersant. Polymers 2023, 15, 999. [Google Scholar] [CrossRef]
- Omar, M.H.; Razak, K.A.; Ab Wahab, M.N.; Hamzah, H.H. Recent progress of conductive 3D-printed electrodes based upon polymers/carbon nanomaterials using a fused deposition modelling (FDM) method as emerging electrochemical sensing devices. RSC Adv. 2021, 11, 16557–16571. [Google Scholar] [CrossRef]
- Marconi, S.; Alaimo, G.; Mauri, V.; Torre, M.; Auricchio, F. Impact of graphene reinforcement on mechanical properties of PLA 3D printed materials. In Proceedings of the IEEE MTT-S International Microwave Workshop Series on Advanced Materials and Processes for RF and THz Applications (IMWS-AMP), Pavia, Italy, 20–22 September 2017; IEEE: New York City, NY, USA, 2017; pp. 1–3. [Google Scholar] [CrossRef]
- Martinez, V.; Cicero, S.; Arroyo, B. Effect of graphene on the fracture behaviour of 3D printed PLA SENB specimens. Procedia Struct. Integr. 2021, 33, 89–96. [Google Scholar] [CrossRef]
- Nassar, G.; Daou, E.; Najjar, R.; Bassil, M.; Habchi, R. A review on the current research on graphene-based aerogels and their applications. Carbon Trends 2021, 4, 100065. [Google Scholar] [CrossRef]
- Tandel, R.; Gozen, B.A. Direct-Ink-writing of liquid metal-graphene-based polymer composites: Composition-processing-property relationships. J. Mater. Process Technol. 2022, 302, 117470. [Google Scholar] [CrossRef]
- Haney, R.; Tran, P.; Trigg, E.B.; Koerner, H.; Dickens, T.; Ramakrishnan, S. Printability and performance of 3D conductive graphite structures. Addit. Manuf. 2021, 37, 101618. [Google Scholar] [CrossRef]
- Shao, L.; Shi, L.; Li, X.; Song, N.; Ding, P. Synergistic effect of BN and graphene nanosheets in 3D framework on the enhancement of thermal conductive properties of polymeric composites. Compos. Sci. Technol. 2016, 135, 83–91. [Google Scholar] [CrossRef]
- Tilve-Martinez, D.; Neri, W.; Horaud, D.; Vukadinovic, N.; Berton, B.; Desmedt, A.; Yuan, J.; Poulin, P. Graphene Oxide Based Transparent Resins for Accurate 3D Printing of Conductive Materials. Adv. Funct. Mater. 2023, 33, 21. [Google Scholar] [CrossRef]
- Ajiteru, O.; Sultan, T.; Lee, Y.J.; Seo, Y.B.; Hong, H.; Lee, J.S.; Lee, H.; Suh, Y.J.; Ju, H.W.; Lee, O.J.; et al. A 3D Printable Electroconductive Biocomposite Bioink Based on Silk Fibroin-Conjugated Graphene Oxide. Nano Lett. 2020, 20, 6873–6883. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Huang, X.; Chen, J.; Wu, K.; Wang, J.; Zhang, X. A Review of Conductive Carbon Materials for 3D Printing: Materials, Technologies, Properties, and Applications. Materials 2021, 14, 3911. [Google Scholar] [CrossRef] [PubMed]
- Kidalov, S.; Voznyakovskii, A.; Vozniakovskii, A.; Titova, S.; Auchynnikau, Y. The Effect of Few-Layer Graphene on the Complex of Hardness, Strength, and Thermo Physical Properties of Polymer Composite Materials Produced by Digital Light Processing (DLP) 3D Printing. Materials 2023, 16, 1157. [Google Scholar] [CrossRef]
- Flores, D.; Noboa, J.; Tarapues, M.; Vizuete, K.; Debut, A.; Bejarano, L.; Streitwieser, D.A.; Ponce, S. Simple Preparation of Metal-Impregnated FDM 3D-Printed Structures. Micromachines 2022, 13, 1675. [Google Scholar] [CrossRef]
- Bressan, L.P.; Lima, T.M.; da Silveira, G.D.; da Silva, J.A.F. Low-cost and simple FDM-based 3D-printed microfluidic device for the synthesis of metallic core–shell nanoparticles. SN Appl. Sci. 2020, 2, 984. [Google Scholar] [CrossRef]
- Calamak, S.; Ermis, M. In situ silver nanoparticle synthesis on 3D-printed polylactic acid scaffolds for biomedical applications. J. Mater. Res. 2021, 36, 166–175. [Google Scholar] [CrossRef]
- Raut, N.C.; Al-Shamery, K. Inkjet printing metals on flexible materials for plastic and paper electronics. J. Mater. Chem. C Mater. 2018, 6, 1618–1641. [Google Scholar] [CrossRef]
- Li, W.; Li, L.; Gao, Y.; Hu, D.; Li, C.-F.; Zhang, H.; Jiu, J.; Nagao, S.; Suganuma, K. Highly conductive copper films based on submicron copper particles/copper complex inks for printed electronics: Microstructure, resistivity, oxidation resistance, and long-term stability. J. Alloys Compd. 2018, 732, 240–247. [Google Scholar] [CrossRef]
- Kim, S. Inkjet-Printed Electronics on Paper for RF Identification (RFID) and Sensing. Electronics 2020, 9, 1636. [Google Scholar] [CrossRef]
- Beedasy, V.; Smith, P.J. Printed Electronics as Prepared by Inkjet Printing. Materials 2020, 13, 704. [Google Scholar] [CrossRef] [PubMed]
- Zareei, A.; Gopalakrishnan, S.; Mutlu, Z.; He, Z.; Peana, S.; Wang, H.; Rahimi, R. Highly Conductive Copper–Silver Bimodal Paste for Low-Cost Printed Electronics. ACS Appl. Electron. Mater. 2021, 3, 3352–3364. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, D.-R.; Chen, S.-C. Sampling and characterization of particle emission from the 3D FDM printing. J. Build. Eng. 2022, 52, 104476. [Google Scholar] [CrossRef]
- Afshari, P.; Pavlyuk, M.; Lira, C.; Katnam, K.; Bodaghi, M.; Yazdani Nezhad, H. Mechanical Strain Tailoring via Magnetic Field Assisted 3D Printing of Iron Particles Embedded Polymer Nanocomposites. Macromol. Mater. Eng. 2023, 308, 2300194. [Google Scholar] [CrossRef]
- Deepali, K. 3D-Printable Composites for Magnetic Refrigeration Based on Ni-Mn-In-Co Shape Memory Alloys. Ph.D. Thesis, Universidad Pública de Navarra, Pamplona, Spain, 2023. [Google Scholar] [CrossRef]
- Ehrmann, G.; Blachowicz, T.; Ehrmann, A. Magnetic 3D-Printed Composites—Production and Applications. Polymers 2022, 14, 3895. [Google Scholar] [CrossRef]
- Wang, H.; Lamichhane, T.N.; Paranthaman, M.P. Review of additive manufacturing of permanent magnets for electrical machines: A prospective on wind turbine. Mater. Today Phys. 2022, 24, 100675. [Google Scholar] [CrossRef]
- Vucemilovic, A.; Savary, M.; Espanet, C. 3-D Printing of Multipolar Bonded SmCo Permanent Magnets. IEEE Trans. Magn. 2023, 59, 2101807. [Google Scholar] [CrossRef]
- Li, Z.; Feng, D.; Li, B.; Xie, D.; Mei, Y.; Zeng, T. Fabrication and Properties of Thermoplastic Polyurethane/Silver Parts via Fused Deposition Modeling for Electromagnetic Interference Shielding and Wearable Sensors. Adv. Eng. Mater. 2022, 24, 7. [Google Scholar] [CrossRef]
- Khosravani, M.R.; Reinicke, T. 3D-printed sensors: Current progress and future challenges. Sens. Actuators A Phys. 2020, 305, 111916. [Google Scholar] [CrossRef]
- Li, B.; Zhang, S.; Zhang, L.; Gao, Y.; Xuan, F. Strain sensing behavior of FDM 3D printed carbon black filled TPU with periodic configurations and flexible substrates. J. Manuf. Process 2022, 74, 283–295. [Google Scholar] [CrossRef]
- Nyabadza, A.; Vázquez, M.; Coyle, S.; Fitzpatrick, B.; Brabazon, D. Review of Materials and Fabrication Methods for Flexible Nano and Micro-Scale Physical and Chemical Property Sensors. Appl. Sci. 2021, 11, 8563. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, H.; Han, W.; Lin, H.; Li, R.; Zhu, J.; Huang, W. 3D Printed Flexible Strain Sensors: From Printing to Devices and Signals. Adv. Mater. 2021, 33, 8. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, A.O.; Cachim, P.B.; Costa, P.M.F.J. Self-sensing piezoresistive cement composite loaded with carbon black particles. Cem. Concr. Compos. 2017, 81, 59–65. [Google Scholar] [CrossRef]
- Zhai, Y.; Yu, Y.; Zhou, K.; Yun, Z.; Huang, W.; Liu, H.; Xia, Q.; Dai, K.; Zheng, G.; Liu, C.; et al. Flexible and wearable carbon black/thermoplastic polyurethane foam with a pinnate-veined aligned porous structure for multifunctional piezoresistive sensors. Chem. Eng. J. 2020, 382, 122985. [Google Scholar] [CrossRef]
- Xia, H.; Zhang, D.; Wang, D.; Tang, M.; Zhang, H.; Chen, X.; Mao, R.; Ma, Y.; Cai, H. High Sensitivity, Wide Range Pressure Sensor Based on Layer-by-Layer Self-Assembled MXene/Carbon Black@Polyurethane Sponge for Human Motion Monitoring and Intelligent Vehicle Control. IEEE Sens. J. 2022, 22, 21561–21568. [Google Scholar] [CrossRef]
- Alshammari, A.S.; Alenezi, M.R.; Lai, K.T.; Silva, S.R.P. Inkjet printing of polymer functionalized CNT gas sensor with enhanced sensing properties. Mater. Lett. 2017, 189, 299–302. [Google Scholar] [CrossRef]
- Kuzubasoglu, B.A.; Sayar, E.; Bahadir, S.K. Inkjet-Printed CNT/PEDOT:PSS Temperature Sensor on a Textile Substrate for Wearable Intelligent Systems. IEEE Sens. J. 2021, 21, 13090–13097. [Google Scholar] [CrossRef]
- Yuan, Y.; Tang, X.; Jiang, L.; Yang, Y.; Zhou, Y.; Dong, Y. Convenient CNT-Paper Gas Sensors Prepared by a Household Inkjet Printer. ACS Omega 2020, 5, 32877–32882. [Google Scholar] [CrossRef]
- Platonova, E.; Ponomareva, P.; Lokiaeva, Z.; Pavlov, A.; Nelyub, V.; Polezhaev, A. New Building Blocks for Self-Healing Polymers. Polymers 2022, 14, 5394. [Google Scholar] [CrossRef] [PubMed]
- Almutairi, M.D.; Aria, A.I.; Thakur, V.K.; Khan, M.A. Self-Healing Mechanisms for 3D-Printed Polymeric Structures: From Lab to Reality. Polymers 2020, 12, 1534. [Google Scholar] [CrossRef] [PubMed]
- Snyder, A.D.; Phillips, Z.J.; Turicek, J.S.; Diesendruck, C.E.; Nakshatrala, K.B.; Patrick, J.F. Prolonged in situ self-healing in structural composites via thermo-reversible entanglement. Nat. Commun. 2022, 13, 6511. [Google Scholar] [CrossRef] [PubMed]
- Qamar, I.P.S.; Sottos, N.R.; Trask, R.S. Grand challenges in the design and manufacture of vascular self-healing. Multifunct. Mater. 2020, 3, 013001. [Google Scholar] [CrossRef]
- Shields, Y.; De Belie, N.; Jefferson, A.; Van Tittelboom, K. A review of vascular networks for self-healing applications. Smart Mater. Struct. 2021, 30, 063001. [Google Scholar] [CrossRef]
- Hansen, C.J. Microvascular-based self-healing materials. In Recent Advances in Smart Self-Healing Polymers and Composites; Elsevier: Amsterdam, The Netherlands, 2022; pp. 141–176. [Google Scholar] [CrossRef]
- De Leon, A.C.; Chen, Q.; Palaganas, N.B.; Palaganas, J.O.; Manapat, J.; Advincula, R.C. High performance polymer nanocomposites for additive manufacturing applications. React. Funct. Polym. 2016, 103, 141–155. [Google Scholar] [CrossRef]
- Yan, J.; Zhi, G.; Kong, D.; Wang, H.; Xu, T.; Zang, J.; Shen, W.; Xu, J.; Shi, Y.; Dai, S.; et al. 3D printed rGO/CNT microlattice aerogel for a dendrite-free sodium metal anode. J. Mater. Chem. A Mater. 2020, 8, 19843–19854. [Google Scholar] [CrossRef]
- Agarwala, S.; Goh, G.L.; Goh, G.D.; Dikshit, V.; Yeong, W.Y. 3D and 4D printing of polymer/CNTs-based conductive composites. In 3D and 4D Printing of Polymer Nanocomposite Materials; Elsevier: Amsterdam, The Netherlands, 2020; pp. 297–324. [Google Scholar] [CrossRef]
- Peng, M.; Shi, D.; Sun, Y.; Cheng, J.; Zhao, B.; Xie, Y.; Zhang, J.; Guo, W.; Jia, Z.; Liang, Z.; et al. 3D Printed Mechanically Robust Graphene/CNT Electrodes for Highly Efficient Overall Water Splitting. Adv. Mater. 2020, 32, 23. [Google Scholar] [CrossRef]
- Dul, S.; Gutierrez, B.J.A.; Pegoretti, A.; Alvarez-Quintana, J.; Fambri, L. 3D printing of ABS Nanocomposites. Comparison of processing and effects of multi-wall and single-wall carbon nanotubes on thermal, mechanical and electrical properties. J. Mater. Sci. Technol. 2022, 121, 52–66. [Google Scholar] [CrossRef]
- Mora, A.; Verma, P.; Kumar, S. Electrical conductivity of CNT/polymer composites: 3D printing, measurements and model-ing. Compos. B Eng. 2020, 183, 107600. [Google Scholar] [CrossRef]
- Ding, H.; Barg, S.; Derby, B. Direct 3D printing of graphene using capillary suspensions. Nanoscale 2020, 12, 11440–11447. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Lv, R.; Bai, S. Recent advances on 3D printing graphene-based composites. Nano Mater. Sci. 2019, 1, 101–115. [Google Scholar] [CrossRef]
- Silva, M.; Pinho, I.S.; Covas, J.A.; Alves, N.M.; Paiva, M.C. 3D printing of graphene-based polymeric nanocomposites for biomedical applications. Funct. Compos. Mater. 2021, 2, 8. [Google Scholar] [CrossRef]
- Tang, X.; Zhou, H.; Cai, Z.; Cheng, D.; He, P.; Xie, P.; Zhang, D.; Fan, T. Generalized 3D Printing of Graphene-Based Mixed-Dimensional Hybrid Aerogels. ACS Nano 2018, 12, 3502–3511. [Google Scholar] [CrossRef] [PubMed]
- Kaya, H.; Arıcı, Ş.; Bulut, O.; Bilgili, F.; Ege, D. CNT incorporation improves the resolution and stability of porous 3D printed PLGA/HA/CNT scaffolds for bone regeneration. Biomed. Mater. 2023, 18, 055028. [Google Scholar] [CrossRef] [PubMed]
- Bher, A.; Mayekar, P.C.; Auras, R.A.; Schvezov, C.E. Biodegradation of Biodegradable Polymers in Mesophilic Aerobic Environments. Int. J. Mol. Sci. 2022, 23, 12165. [Google Scholar] [CrossRef]
- Le Duigou, A.; Barbé, A.; Guillou, E.; Castro, M. 3D printing of continuous flax fibre reinforced biocomposites for structural applications. Mater. Des. 2019, 180, 107884. [Google Scholar] [CrossRef]
- Dai, H.; Dai, W.; Hu, Z.; Zhang, W.; Zhang, G.; Guo, R. Advanced Composites Inspired by Biological Structures and Functions in Nature: Architecture Design, Strengthening Mechanisms, and Mechanical-Functional Responses. Adv. Sci. 2023, 10, 14. [Google Scholar] [CrossRef]
- Saroia, J.; Wang, Y.; Wei, Q.; Lei, M.; Li, X.; Guo, Y.; Zhang, K. A review on 3D printed matrix polymer composites: Its potential and future challenges. Int. J. Adv. Manuf. Technol. 2020, 106, 1695–1721. [Google Scholar] [CrossRef]
- Liao, J.; Brosse, N.; Pizzi, A.; Hoppe, S.; Zhou, X.; Du, G. Characterization and 3D printability of poly (lactic acid)/acetylated tannin composites. Ind. Crops Prod. 2020, 149, 112320. [Google Scholar] [CrossRef]
- Bouzidi, K.; Chaussy, D.; Gandini, A.; Bongiovanni, R.; Beneventi, D. 3D printable fully biomass-based composite using poly(furfuryl alcohol) as binder and cellulose as a filler. Carbohydr. Polym. 2022, 293, 119716. [Google Scholar] [CrossRef] [PubMed]
- Tanase-Opedal, M.; Espinosa, E.; Rodríguez, A.; Chinga-Carrasco, G. Lignin: A Biopolymer from Forestry Biomass for Biocomposites and 3D Printing. Materials 2019, 12, 3006. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Li, M.; Zhou, L.; Lee, S.; Mei, C.; Xu, X.; Wu, Q. The influence of grafted cellulose nanofibers and postextrusion annealing treatment on selected properties of poly(lactic acid) filaments for 3D printing. J. Polym. Sci. B Polym. Phys. 2017, 55, 847–855. [Google Scholar] [CrossRef]
- Tekinalp, H.L.; Meng, X.; Lu, Y.; Kunc, V.; Love, L.J.; Peter, W.H.; Ozcan, S. High modulus biocomposites via additive manufacturing: Cellulose nanofibril networks as “microsponges”. Compos. B Eng. 2019, 173, 106817. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, J.; Lu, Y.; Hu, L.; Fan, Y.; Ma, J.; Zhou, X. Preparation of 3D printable micro/nanocellulose-polylactic acid (MNC/PLA) composite wire rods with high MNC constitution. Ind. Crops Prod. 2017, 109, 889–896. [Google Scholar] [CrossRef]
- Xie, G.; Zhang, Y.; Lin, W. Plasticizer Combinations and Performance of Wood Flour–Poly(Lactic Acid) 3D Printing Filaments. Bioresources 2017, 12, 3. [Google Scholar] [CrossRef]
- Xu, W.; Pranovich, A.; Uppstu, P.; Wang, X.; Kronlund, D.; Hemming, J.; Öblom, H.; Moritz, N.; Preis, M.; Sandler, N.; et al. Novel biorenewable composite of wood polysaccharide and polylactic acid for three dimensional printing. Carbohydr. Polym. 2018, 187, 51–58. [Google Scholar] [CrossRef]
- Kariz, M.; Sernek, M.; Obućina, M.; Kuzman, M.K. Effect of wood content in FDM filament on properties of 3D printed parts. Mater. Today Commun. 2018, 14, 135–140. [Google Scholar] [CrossRef]
- Murphy, C.A.; Collins, M.N. Microcrystalline cellulose reinforced polylactic acid biocomposite filaments for 3D printing. Polym. Compos. 2018, 39, 1311–1320. [Google Scholar] [CrossRef]
- Winter, A.; Mundigler, N.; Holzweber, J.; Veigel, S.; Müller, U.; Kovalcik, A.; Gindl-Altmutter, W. Residual wood polymers facilitate compounding of microfibrillated cellulose with poly(lactic acid) for 3D printer filaments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2018, 376, 20170046. [Google Scholar] [CrossRef] [PubMed]
- Le Guen, M.-J.; Hill, S.; Smith, D.; Theobald, B.; Gaugler, E.; Barakat, A.; Mayer-Laigle, C. Influence of Rice Husk and Wood Biomass Properties on the Manufacture of Filaments for Fused Deposition Modeling. Front. Chem. 2019, 7, 735. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, Y.; Yu, T.; Wang, N.; Wang, C.; Wang, H. Preparation of polylactic acid/TEMPO-oxidized bacterial cellulose nanocomposites for 3D printing via Pickering emulsion approach. Compos. Commun. 2019, 16, 162–167. [Google Scholar] [CrossRef]
- Lin, W.; Xie, G.; Qiu, Z. Effects of ultraviolet aging on properties of wood flour–poly(lactic acid) 3D printing filaments. Bioresources 2019, 14, 8689–8700. [Google Scholar] [CrossRef]
- Liu, L.; Lin, M.; Xu, Z.; Lin, M. Polylactic acid-based wood-plastic 3D printing composite and its properties. Bioresources 2019, 14, 8484–8498. [Google Scholar] [CrossRef]
- Mimini, V.; Sykacek, E.; Hashim, S.N.A.S.; Holzweber, J.; Hettegger, H.; Fackler, K.; Potthast, A.; Mundigler, N.; Rosenau, T. Compatibility of Kraft Lignin, Organosolv Lignin and Lignosulfonate with PLA in 3D Printing. J. Wood Chem. Technol. 2019, 39, 14–30. [Google Scholar] [CrossRef]
- Xiao, X.; Chevali, V.S.; Song, P.; He, D.; Wang, H. Polylactide/hemp hurd biocomposites as sustainable 3D printing feedstock. Compos. Sci. Technol. 2019, 184, 107887. [Google Scholar] [CrossRef]
- Zhao, X.; Tekinalp, H.; Meng, X.; Ker, D.; Benson, B.; Pu, Y.; Ragauskas, A.J.; Wang, Y.; Li, K.; Webb, E.; et al. Poplar as Biofiber Reinforcement in Composites for Large-Scale 3D Printing. ACS Appl. Bio Mater. 2019, 2, 4557–4570. [Google Scholar] [CrossRef]
- Liu, H.; He, H.; Peng, X.; Huang, B.; Li, J. Three-dimensional printing of poly(lactic acid) bio-based composites with sugarcane bagasse fiber: Effect of printing orientation on tensile performance. Polym. Adv. Technol. 2019, 30, 910–922. [Google Scholar] [CrossRef]
- Bhagia, S.; Lowden, R.R.; Erdman, D., III; Rodriguez, M., Jr.; Haga, B.; Solano, A.I.R.M.; Gallego, N.C.; Pu, Y.; Muchero, W.; Kunc, V.; et al. Tensile properties of 3D-printed wood-filled PLA materials using poplar trees. Appl. Mater. Today 2020, 21, 100832. [Google Scholar] [CrossRef]
- Cisneros-López, E.O.; Pal, A.K.; Rodriguez, A.U.; Wu, F.; Misra, M.; Mielewski, D.F.; Kiziltas, A.; Mohanty, A.K. Recycled poly(lactic acid)–based 3D printed sustainable biocomposites: A comparative study with injection molding. Mater. Today Sustain. 2020, 7–8, 100027. [Google Scholar] [CrossRef]
- Dong, Y.; Milentis, J.; Pramanik, A. Additive manufacturing of mechanical testing samples based on virgin poly (lactic acid) (PLA) and PLA/wood fibre composites. Adv. Manuf. 2018, 6, 71–82. [Google Scholar] [CrossRef]
- Guessasma, S.; Belhabib, S.; Nouri, H. Microstructure and Mechanical Performance of 3D Printed Wood-PLA/PHA Using Fused Deposition Modelling: Effect of Printing Temperature. Polymers 2019, 11, 1778. [Google Scholar] [CrossRef] [PubMed]
- Figueroa-Velarde, V.; Diaz-Vidal, T.; Cisneros-López, E.O.; Robledo-Ortiz, J.R.; López-Naranjo, E.J.; Ortega-Gudiño, P.; Rosales-Rivera, L.C. Mechanical and Physicochemical Properties of 3D-Printed Agave Fibers/Poly(lactic) Acid Biocomposites. Materials 2021, 14, 3111. [Google Scholar] [CrossRef] [PubMed]
- Shahar, F.S.; Sultan, M.T.H.; Safri, S.N.A.; Jawaid, M.; Talib, A.R.A.; Basri, A.A.; Shah, A.U.M. Physical, thermal and tensile behaviour of 3D printed kenaf/PLA to suggest its usability for ankle–foot orthosis—A preliminary study. Rapid Prototyp. J. 2022, 28, 1573–1588. [Google Scholar] [CrossRef]
- Jamadi, A.H.; Razali, N.; Petrů, M.; Taha, M.M.; Muhammad, N.; Ilyas, R.A. Effect of Chemically Treated Kenaf Fibre on Mechanical and Thermal Properties of PLA Composites Prepared through Fused Deposition Modeling (FDM). Polymers 2021, 13, 3299. [Google Scholar] [CrossRef]
- Aumnate, C.; Soatthiyanon, N.; Makmoon, T.; Potiyaraj, P. Polylactic acid/kenaf cellulose biocomposite filaments for melt extrusion based-3D printing. Cellulose 2021, 28, 8509–8525. [Google Scholar] [CrossRef]
- Yu, W.; Shi, J.; Sun, L.; Lei, W. Effects of Printing Parameters on Properties of FDM 3D Printed Residue of Astragalus/Polylactic Acid Biomass Composites. Molecules 2022, 27, 7373. [Google Scholar] [CrossRef]
- Fekete, I.; Ronkay, F.; Lendvai, L. Highly toughened blends of poly(lactic acid) (PLA) and natural rubber (NR) for FDM-based 3D printing applications: The effect of composition and infill pattern. Polym. Test. 2021, 99, 107205. [Google Scholar] [CrossRef]
- Depuydt, D.; Balthazar, M.; Hendrickx, K.; Six, W.; Ferraris, E.; Desplentere, F.; Ivens, J.; Vuure, A.W.V. Production and characterization of bamboo and flax fiber reinforced polylactic acid filaments for fused deposition modeling (FDM). Polym. Compos. 2019, 40, 1951–1963. [Google Scholar] [CrossRef]
- Dey, A.; Rahman, M.M.; Yodo, N.; Grewell, D. Development of biocomposite filament for fused filament fabrication from soy hulls and soy protein isolate. Mater. Today Commun. 2023, 34, 105316. [Google Scholar] [CrossRef]
- Shi, G.; Peng, X.; Zeng, J.; Zhong, L.; Sun, Y.; Yang, W.; Zhong, Y.L.; Zhu, Y.; Zou, R.; Admassie, S.; et al. A Liquid Metal Microdroplets Initialized Hemicellulose Composite for 3D Printing Anode Host in Zn-Ion Battery. Adv. Mater. 2023, 35, 25. [Google Scholar] [CrossRef] [PubMed]
- Long, H. Mechanical and thermal properties of bamboo fiber reinforced polypropylene/polylactic acid composites for 3D printing. Polym. Eng. Sci. 2019, 59, 2. [Google Scholar] [CrossRef]
- Yu, W.; Dong, L.; Lei, W.; Zhou, Y.; Pu, Y.; Zhang, X. Effects of Rice Straw Powder (RSP) Size and Pretreatment on Properties of FDM 3D-Printed RSP/Poly(lactic acid) Biocomposites. Molecules 2021, 26, 3234. [Google Scholar] [CrossRef] [PubMed]
- Ambone, T.; Torris, A.; Shanmuganathan, K. Enhancing the mechanical properties of 3D printed polylactic acid using nanocellulose. Polym. Eng. Sci. 2020, 60, 1842–1855. [Google Scholar] [CrossRef]
- Ryu, J.-A.; Lee, J.M.; Eom, T.-J. Comparison of 3D Printer Application and Strength Property Using Polylactic Acid Filaments with Lignin-free and -rich MFC. J. Korea Tech. Assoc. Pulp Pap. Ind. 2019, 51, 68–76. [Google Scholar] [CrossRef]
- Yang, T.-C.; Yeh, C.-H. Morphology and Mechanical Properties of 3D Printed Wood Fiber/Polylactic Acid Composite Parts Using Fused Deposition Modeling (FDM): The Effects of Printing Speed. Polymers 2020, 12, 1334. [Google Scholar] [CrossRef]
- Rahim, T.N.A.T.; Abdullah, A.M.; Akil, H.M. Recent Developments in Fused Deposition Modeling-Based 3D Printing of Polymers and Their Composites. Polym. Rev. 2019, 59, 589–624. [Google Scholar] [CrossRef]
- Taşcıoğlu, E.; Kıtay, Ö.; Keskin, A.Ö.; Kaynak, Y. Effect of printing parameters and post-process on surface roughness and dimensional deviation of PLA parts fabricated by extrusion-based 3D printing. J. Braz. Soc. Mech. Sci. Eng. 2022, 44, 139. [Google Scholar] [CrossRef]
- Fico, D.; Rizzo, D.; Casciaro, R.; Corcione, C.E. A Review of Polymer-Based Materials for Fused Filament Fabrication (FFF): Focus on Sustainability and Recycled Materials. Polymers 2022, 14, 465. [Google Scholar] [CrossRef] [PubMed]
- Cano-Vicent, A.; Tambuwala, M.M.S.; Hassan, S.; Barh, D.; Aljabali, A.A.A.; Birkett, M.; Arjunan, A.; Serrano-Aroca, Á. Fused deposition modelling: Current status, methodology, applications and future prospects. Addit. Manuf. 2021, 47, 102378. [Google Scholar] [CrossRef]
- Baechle-Clayton, M.; Loos, E.; Taheri, M.; Taheri, H. Failures and Flaws in Fused Deposition Modeling (FDM) Additively Manufactured Polymers and Composites. J. Compos. Sci. 2022, 6, 202. [Google Scholar] [CrossRef]
- Müller, M.; Jirků, P.; Šleger, V.; Mishra, R.K.; Hromasová, M.; Novotný, J. Effect of Infill Density in FDM 3D Printing on Low-Cycle Stress of Bamboo-Filled PLA-Based Material. Polymers 2022, 14, 4930. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Dong, L.; Lei, W.; Shi, J. Rice straw powder/polylactic acid biocomposites for three-dimensional printing. Adv. Compos. Lett. 2020, 29, 2633366X2096736. [Google Scholar] [CrossRef]
- Zheng, L.; Liu, J.; Liu, R.; Xing, Y.; Jiang, H. 3D printing performance of gels from wheat starch, flour and whole meal. Food Chem. 2021, 356, 129546. [Google Scholar] [CrossRef] [PubMed]
- Travitzky, N.; Windsheimer, H.; Fey, T.; Greil, P. Preceramic Paper-Derived Ceramics. J. Am. Ceram. Soc. 2008, 91, 3477–3492. [Google Scholar] [CrossRef]
- Rosenthal, M.; Henneberger, C.; Gutkes, A.; Bues, C.-T. Liquid Deposition Modeling: A promising approach for 3D printing of wood. Eur. J. Wood Wood Prod. 2018, 76, 797–799. [Google Scholar] [CrossRef]
- Rosa, R.P.; Rosace, G.; Arrigo, R.; Malucelli, G. Preparation and characterization of a fully biobased resin system for 3d-printing, suitable for replacing fossil-based acrylates. J. Polym. Res. 2023, 30, 139. [Google Scholar] [CrossRef]
- Sutton, J.T.; Rajan, K.; Harper, D.P.; Chmely, S.C. Lignin-Containing Photoactive Resins for 3D Printing by Stereolithography. ACS Appl. Mater. Interfaces 2018, 10, 36456–36463. [Google Scholar] [CrossRef]
- Zhang, Y.; Fang, J.; Li, J.; Guo, Y.; Wang, Q. The Effect of Carbon Nanotubes on the Mechanical Properties of Wood Plastic Composites by Selective Laser Sintering. Polymers 2017, 9, 728. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.; Kim, Y.; Won, Y.; Myung, J. Bridging Three Gaps in Biodegradable Plastics: Misconceptions and Truths About Biodegradation. Front. Chem. 2021, 9, 671750. [Google Scholar] [CrossRef] [PubMed]
- ISO 18830:2016; Plastics Determination of Aerobic Biodegradation of Non-Floating Plastic Materials in a Seawater/Sandy Sediment Interface Method by Measuring the Oxygen Demand in Closed Respirometer. International Organization for Standardization: Geneva, Switzerland, 2016.
- ISO 19679:2020; Plastics—Determination of Aerobic Biodegradation of Non-Floating Plastic Materials in a Seawater/Sediment Interface—Method by Analysis of Evolved Carbon Dioxide. International Organization for Standardization: Geneva, Switzerland, 2020.
- Pires, J.R.A.; Souza, V.G.L.; Fuciños, P.; Pastrana, L.; Fernando, A.L. Methodologies to Assess the Biodegradability of Bio-Based Polymers—Current Knowledge and Existing Gaps. Polymers 2022, 14, 1359. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.N.M.A.; Naebe, M. Material Extrusion of Wool Waste/Polycaprolactone with Improved Tensile Strength and Biodegradation. Polymers 2023, 15, 3439. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993-13:2010; Biological Evaluation of Medical Devices Part 13: Identification and Quantification of Degradation Products from Polymeric Medical Devices. International Organization for Standardization: Geneva, Switzerland, 2010.
- Wang, S.; Xing, Q. Preparation and in vitro biocompatibility of PBAT and chitosan composites for novel biodegradable cardiac occluders. e-Polymers 2022, 22, 705–718. [Google Scholar] [CrossRef]
- Karimipour-Fard, P.; Pop-Iliev, R.; Jones-Taggart, H.; Rizvi, G. Design of 3D scaffold geometries for optimal biodegradation of poly(lactic acid)-based bone tissue. AIP Conf. Proc. 2020, 2205, 020062. [Google Scholar] [CrossRef]
- Hedayati, S.K.; Behravesh, A.H.; Hasannia, S.; Bagheri Saed, A.; Akhoundi, B. 3D printed PCL scaffold reinforced with continuous biodegradable fiber yarn: A study on mechanical and cell viability properties. Polym. Test. 2020, 83, 106347. [Google Scholar] [CrossRef]
- Ghosh Dastidar, A.; Clarke, S.A.; Larrañeta, E.; Buchanan, F.; Manda, K. In Vitro Degradation of 3D-Printed Poly(L-lactide-co-glycolic acid) Scaffolds for Tissue Engineering Applications. Polymers 2023, 15, 3714. [Google Scholar] [CrossRef]
- Ausejo, J.G.; Rydz, J.; Musioł, M.; Sikorska, W.; Sobota, M.; Włodarczyk, J.; Adamus, G.; Janeczek, H.; Kwiecień, I.; Hercog, A.; et al. A comparative study of three-dimensional printing directions: The degradation and toxicological profile of a PLA/PHA blend. Polym. Degrad. Stab. 2018, 152, 191–207. [Google Scholar] [CrossRef]
- Choe, S.; Kim, Y.; Park, G.; Lee, D.H.; Park, J.; Mossisa, A.T.; Lee, S.; Myung, J. Biodegradation of 3D-Printed Biodegradable/Non-biodegradable Plastic Blends. ACS Appl. Polym. Mater. 2022, 4, 5077–5090. [Google Scholar] [CrossRef]
- BS EN 1899-2:1998; Water Quality. Determination of Biochemical Oxygen Demand after n Days (BODn) Method for Undiluted Samples. International Organization for Standardization: London, UK, 1998.
- Acharya, A.; Puri, R.; Giri, J.; Malla, K.P.; Khatiwada, L.N.; Sharma, K.P.; Maruyama, T.; Adhikari, R. Preparation of Hydroxyapatite from Buffalo Bone and its Biodegradable Nanocomposite with Poly(Butylene Adipate-co-Terephthalate). Macromol. Symp. 2023, 408, 2200169. [Google Scholar] [CrossRef]
- ISO 13781:2017; Implants for Surgery Homopolymers, Copolymers and Blends on Poly(Lactide) In Vitro Degradation Testing. International Organization for Standardization: Geneva, Switzerland, 2017.
- Balogová, A.F.; Trebuňová, M.; Ižaríková, G.; Kaščák, Ľ.; Mitrík, L.; Klímová, J.; Feranc, J.; Modrák, M.; Hudák, R.; Živčák, J. In Vitro Degradation of Specimens Produced from PLA/PHB by Additive Manufacturing in Simulated Conditions. Polymers 2021, 13, 1542. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Shi, J.; Li, M.; Lei, W.; Yu, W. FDM 3D Printing and Soil-Burial-Degradation Behaviors of Residue of Astragalus Particles/Thermoplastic Starch/Poly(lactic acid) Biocomposites. Polymers 2023, 15, 2382. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Sun, Y.; Youe, W.J.; Gwon, J.; Cheng, H.N.; Wu, Q. 3D-printed wood-polylactic acid-thermoplastic starch composites: Performance features in relation to biodegradation treatment. J. Appl. Polym. Sci. 2021, 138, 50914. [Google Scholar] [CrossRef]
- Tsou, C.-H.; Yao, W.-H.; Wu, C.-S.; Tsou, C.-Y.; Hung, W.-S.; Chen, J.-C.; Guo, J.; Yuan, S.; Wen, E.; Wang, R.-Y.; et al. Preparation and characterization of renewable composites from Polylactide and Rice husk for 3D printing applications. J. Polym. Res. 2019, 26, 227. [Google Scholar] [CrossRef]
- Sekar, V.; Putra, A.; Palaniyappan, S.; Eh Noum, S.Y.; Sivanesan, S.; Jiun, Y.L. Additive manufactured acoustic absorbers made of wood-fiber composites with modified infill patterns. Wood Mater. Sci. Eng. 2023. [Google Scholar] [CrossRef]
- Harris, M.; Harris, M.; Mohsin, H.; Potgieter, J.; Ishfaq, K.; Archer, R.; Chen, Q.; De Silva, K.; Le Guen, M.-J.; Wilson, R.; et al. Partial Biodegradable Blend with High Stability against Biodegradation for Fused Deposition Modeling. Polymers 2022, 14, 1541. [Google Scholar] [CrossRef]
- Sekar, V.; Palaniyappan, S.; Noum, S.Y.E.; Putra, A.; Sivanesan, S.; Sheng, D.D.C.V. Acoustic absorbers made of wood fiber composites developed by compression molding and additive manufacturing. Wood Res. 2023, 68, 68–82. [Google Scholar] [CrossRef]
- Rydz, J.; Sikorska, W.; Musioł, M.; Janeczek, H.; Włodarczyk, J.; Misiurska-Marczak, M.; Łęczycka, J.; Kowalczuk, M. 3D-Printed Polyester-Based Prototypes for Cosmetic Applications—Future Directions at the Forensic Engineering of Advanced Polymeric Materials. Materials 2019, 12, 6. [Google Scholar] [CrossRef]
- Seggiani, M.; Altieri, R.; Puccini, M.; Stefanelli, E.; Esposito, A.; Castellani, F.; Stanzione, V.; Vitolo, S. Polycaprolactone-collagen hydrolysate thermoplastic blends: Processability and biodegradability/compostability. Polym. Degrad. Stab. 2018, 150, 13–24. [Google Scholar] [CrossRef]
- Dey, A.; Rahman, M.M.; Gupta, A.; Yodo, N.; Lee, C.W. A Performance Study on 3D-Printed Bioplastic Pots from Soybean By-Products. Sustainability 2023, 15, 10535. [Google Scholar] [CrossRef]
- ASTM D5338-15; Standard Test Method for Determining Aerobic Biodegradation of Plastic MaterialsUnder Controlled Composting Conditions. ASTM International: West Conshohocken, PA, USA, 2021.
- PN-EN-ISO-14852:2007; Oznaczanie Całkowitej Biodegradacji Tlenowej Materiałów Polimerowych w Środowisku Wodnym|Metoda Oznaczania Wydzielonego Ditlenku Węgla, Genplast. Polish Committee for Standardization: Warsaw, Poland, 2007.
- ASTM D6400; Standard Specification for Labeling of Plastics Designed to be Aerobically Composted in Municipal or Industrial Facilities. ASTM International: West Conshohocken, PA, USA, 2023.
- PN EN 14806:2010; Packaging—Preliminary Evaluation of the Disintegration of Packaging Materials under Simulated Composting Conditions in a Laboratory Scale Test. Polish Committee for Standardization: Warsaw, Poland, 2010.
- ISO 14045:2012(en); Environmental Management—Eco-Efficiency Assessment of Product Systems—Principles, Requirements and Guidelines. International Organization for Standardization: Geneva, Switzerland, 2012.
- PN-EN 13432:2002; Packaging Requirements for Packaging Suitable for Recovery through Composting and Biodegradation Testing Program and Evaluation Criteria for Final Approval of Packaging. Polish Committee for Standardization: Warsaw, Poland, 2002.
- Uribe-Echeverría, T.; Beiras, R. Acute toxicity of bioplastic leachates to Paracentrotus lividus sea urchin larvae. Mar. Environ. Res. 2022, 176, 105605. [Google Scholar] [CrossRef]
| 3D Printing Method | Type of Materials | References |
|---|---|---|
| FDM | Copper/ABS, iron/ABS | Nikzad et al., (2011) [15] Hwang et al., (2015) [17] |
| Wood/PLA | Ayrilmis et al., (2019) [18] | |
| Al and Al2O3/Nylon-6 | Boparai et al., (2015) [19] | |
| BaTiO3/ABS | Castles et al., (2016) [20] | |
| Tungsten/PC | Shemelya et al.(2015) [21] | |
| TPE/ABS | Perez ART et al., (2014) [22] | |
| Short glass fiber/ABS | Zhong et al., (2001) [23] | |
| Short carbon fiber/AB | Tekinalp et al., (2014) [24] | |
| Glass fiber/PP | Ning et al., (2015) [25] | |
| Microspheres/polywax | Carneiro et al., (2015) [26] | |
| VGCFs/ABS | Wang et al., (2016) [27] | |
| SWNTs/ABS | Shofner et al., (2003) [28] | |
| Recycled wood fibers/PLA and PHA matrix carbon fibers/PLA, natural jute fibers/PLA | Le Duigou et al., (2016) [29] Matsuzaki et al., (2016) [30] | |
| Continuous carbon fiber/PLA | Li et al., (2016) [31] | |
| Carbon fibers/ABS | Nakagawa et al., (2017) [32] | |
| Continuous carbon fiber/ABS/resin | Zhong et al., (2001) [33] | |
| Montmorillonite/ABS | Weng et al., (2016) [34] | |
| Graphene/ABS | Wei et al., (2015) [35] | |
| poly epsilon-caprolactone (PCL) | Zein et al., (2002) [36] | |
| Martin et al. [37] | ||
| DLP | Alumina/UV-sensitive resin | Kokkinis et al., (2015) [38] |
| Direct writing with magnetic assistance | Alumina/polyurethane acrylate | Compton et al., (2014) [39] |
| Direct write | Short carbon fiber/SiC whisker/epoxy | Van Der Klift et al., (2016) [40] |
| DDM | Continuous carbon fiber/nylon | Yan et al., (2011) [41] |
| SLS | PA12+nanokrzemionka | Chung et al., (2006) [42] |
| Glass bead/Nylon-11 | Goodridge et al., (2011) [43] | |
| Carbon nanofibre-polyamide-12 | Lin et al., (2015) [44] | |
| Graphene oxide/photopolymer | Yugang et al., (2011) [45] | |
| TiO2/epoxy acrylate | Kim et al., (2014) [46] | |
| BaTiO3/PEGDA | Zhang et al., (2018) [47] | |
| CNT/acrylic ester | Athreya et al., (2010) [48] | |
| Carbon black/nylon-12 | Zheng et al., (2006) [49] | |
| Al2O3/polystyrene | Kim et al., (2013) [50] | |
| Cont. carbon-TiO2/nylon-12 and graphite/nylon-12 | Lin et al., (2014) [51] | |
| Graphene oxide (GO)/iron (Fe) | Kurimoto et al., (2015) [52] | |
| SLA | Al2O3/UV-cured resin | Kalsoom et al., (2016) [53] |
| Diamond microparticle/acrylate resins | Hector et al., (2006) [54] | |
| CNT/epoxy | Zanchetta et al., (2016) [55] | |
| Silicon oxycarbide (SiOC) (PDCs) | Suwanprateeb et al., (2006) [56] | |
| Starch-based polymer powders (starch/cellulose fiber/sucrose sugar/maltodextrin) | Guo et al., (2015) [57] | |
| Solvent-cast direct writing | CNT/PLA | Krivec et al., (2017) [58] |
| Inkjet printing | Ag/photopolymer | Shao et al., (2016) [59] |
| CSi-Mg/TCP | Wu et al., (2011) [60] | |
| MBG powder (Si/Ca/P)/PVA | Bergmann et al., (2010) [61] | |
| Bioactive glass/β-TCP | Lam et al., (2002) [62] | |
| PLP (3DP) | Starch-based polymer powders (cornstarch/dextran/gelatin) | Zhou et al., (2018) [63] |
| HA/CaSO4 powder with PCL infiltration | Vaezi et al., (2011) [64] | |
| Powder (ZP102)/binder (Zb56) | Glasschroeder et al., (2015) [65] | |
| PMMA/screw nuts and PMMA/carbon fibers | Hui et al., (2018) [66] | |
| LS | nHA/PA12 | Schwentenwein et al., (2015) [67] |
| LCM | Alumina ceramics | Nikzad et al., (2011) [15] Hwang et al., (2015) [17] |
| Property Type | 3D Printing Method | Additional Material | Intentions | Application | Reference |
|---|---|---|---|---|---|
| Thermal properties | FDM | Boron nitride | Dispersion quality | Heat exchangers | Quill et al. [69] Liu et al. [70] Belaid et al. [71] Su et al. [72] Peng et al. [73] |
| FEAM | Synthetic microdiamonds | Performance | Heat sinks | Kowalewska et al. [74] Wang et al. [75] Bogdanov et al. [76] Yaragatti et al. [77] | |
| Conductive properties | FDM | CNT | Viscosity | Electrical conductors | Jariwala et al. [78] Ghoshal [79] Gnanasekaran et al. [80] Yang et al. [81] Lage-Rivera et al. [82] Omar et al. [83] |
| DIW | Graphene | Dispersion quality | Self-sensing composites | Marconi et al. [84] Martinez et al. [85] Nassar et al. [86] Tandel et al. [87] Haney et al. [88] Shao et al. [89] | |
| DLP | GO | Percolation threshold | Lin et al. [44] Tilve-Martinez et al. [90] Ajiteru et al. [91] Zheng et al. [92] | ||
| Embedded circuitry | FDM | Silver particles | Sintering temperature | Electrical devices | Kidalov et al. [93] Flores et al. [94] Bressan et al. [95] Calamak et al. [96] |
| Inkjet | Copper particles | Multiprocessing | Photovoltaics | Raut et al. [97] Li et al. [98] Kim [99] Beedasy et al. [100] Zareei et al. [101] | |
| Magnetic properties | FDM | Iron particles | Viscosity | Magnetic sensors | Zhang et al. [102] Afshari et al. [103] |
| MnAlC particles | Dispersion quality | EMIf shields | Bekas et al. [7,104] Ehrmann et al. [105] Wang et al. [106] Vucemilovic et al. [107] | ||
| Sensing | FDM | Silver particles | Repeatability | Damage detection | Li et al. [108] Khosravani et al. [109] Omar et al. [83] Li et al. [110] Nyabadza et al. [111] Liu et al. [112] |
| TEAM | Carbon black | Accuracy | Structural health | Monteiro et al. [113] Zhai et al. [114] Xia et al. [115] | |
| Inkjet | CNT | Performance | Monitoring | Alshammari et al. [116] Kuzubasoglu et al. [117] Yuan et al. [118] | |
| Self-healing | FDM | Re-mendable polymer | Capsule development | Autonomous structures | Platonova et al. [119] Almutairi et al. [120] Snyder et al. [121] |
| DIW | Vascule development | Qamar et al. [122] Shields et al. [123] Hansen [124] |
| 3D Printing Method | Fibers | References |
|---|---|---|
| FDM | Cellulose | Dong et al. [143] Tekinalp et al. [144] |
| Microcellulose and PEG 6000 | Wang et al. [145] | |
| Poplar/glycerol/tributyl citrate | Xie et al. [146] | |
| Galactomannan(GM) from spruce thermomechanical pulp | Xu et al. [147] | |
| Beechwood | Kariz et al. [148] | |
| Microcrystalline cellulose | Murphy et al. [149] | |
| Native and partially delignified fibrillated beechwood | Winter et al. [150] | |
| Rice husk flour, pine wood flour | Le Guen et al. [151] | |
| TEMPO-oxidized bacterial cellulose | Chen et al. [152] | |
| Poplar wood flour + tributyl citrate | Lin et al. [153] | |
| Pulp, wood, or kraft lignin < 300 mesh | Liu et al. [154] | |
| Pine kraft lignin, beech organosolv lignin, or beech lignosulfonate | Mimini et al. [155] | |
| PBAT + hemp + EGMA (Lotader AX8900) + lubricant, antioxidant and anti-hydrolysis agent | Xiao et al. [156] | |
| Poplar | Zhao et al. [157] | |
| Alkaline spruce lignin | Tanase-Opedal et al. [142] | |
| Sugarcane bagasse and cellulose extracted from bagasse | Liu et al. [158] | |
| Ball-milled poplar | Bhagia et al. [159] | |
| Acetylated tannin | Liao et al. [140] | |
| Recycled—PLA + microcrystalline cellulose and Joncryl chain extender | Cisneros-López et al. [160] | |
| PHA + wood (commercial filament) | Le Duigou et al. [29] | |
| Cellulose (silanized) + PEG6000 | Wang et al. [145] | |
| Poplar + glycerol + tributyl citrate | Xie et al. [146] | |
| Galactomannan | Pranovich et al. [147] | |
| Wood | Dong et al. [161] | |
| Beechwood | Kariz et al. [148] | |
| 1% native and partially delignified fibrillated beechwood | Winter et al. [150] | |
| PLA/PHA + pinewood | Guessasma et al. [162] | |
| Rice husk flour or pine wood flour | Le Guen et al. [151] | |
| Pulp, wood, or lignin silanized with KH550 silicone oil | Liu et al. [158] | |
| Pine kraft lignin, beech organosolv lignin, or beech lignosulfonate | Mimini et al. [155] | |
| PLA + PBAT (2003F) + hemp + EGMA (Lotader AX8900 + lubricant, antioxidant and anti-hydrolysis agent | Xiao et al. [156] | |
| Alkali spruce lignin | Tanase-Opedal et al. [142] | |
| Sugarcane bagasse or cellulose extracted from bagasse | Liu et al. [158] | |
| Acetylated mimosa tannins | Liao et al. [140] | |
| Agave fibers | Figueroa et al. [163] | |
| Kenaf | Shahar et al. [164] Jamadi et al. [165] Aumnate et al. [166] | |
| Kenaf cellulose | Liu et al. [158] | |
| Astragalus | Yu et al. [167] | |
| Natural rubber | Fekete et al. [168] | |
| Bamboo and flax fiber | Depuydt et al. [169] | |
| Wood-flour-filled fiber | Tao et al. [47] | |
| Soy hulls and soy protein | Dey et al. [170] | |
| Hemicellulose composite | Shi et al. [171] | |
| Bamboo compounded fiber | Long et al. [172] | |
| Compound of wood with plastic | Kariz et al. [148] | |
| Compound of straw with plastic | Yu et al. [173] | |
| Compound of cellulose | Ambone et al. [174] | |
| Lignin with plastic | Ryu et al. [175] | |
| Wood plastic wire | Yang et al. [176] | |
| Wood plastic composite | Liu et al. [154], Rahim et al. [177] Tascioglu et al. [178], Fico et al. [179], Cano-Vicent et al. [180], Baechle-Clayton et al. [181] | |
| Bamboo wood | Muller et al. [182] | |
| Straw | Yu et al. [183] | |
| Wheat | Zheng et al. [184] | |
| Corn | Paggi et al. [68] | |
| Galactoglucomannan | Xu et al. [147] | |
| Paper | Travitzky et al. [185] | |
| Wood chips | Rosenthal et al. [186] | |
| SLA | Epoxy acrylate soybean oil (AESO) | Rosa et al. [187] |
| Lignin-based photosensitive resins | Sutton et al. [188] | |
| SLS | Wood plastic pellets | Zhang et al. [189] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anwajler, B.; Witek-Krowiak, A. Three-Dimensional Printing of Multifunctional Composites: Fabrication, Applications, and Biodegradability Assessment. Materials 2023, 16, 7531. https://doi.org/10.3390/ma16247531
Anwajler B, Witek-Krowiak A. Three-Dimensional Printing of Multifunctional Composites: Fabrication, Applications, and Biodegradability Assessment. Materials. 2023; 16(24):7531. https://doi.org/10.3390/ma16247531
Chicago/Turabian StyleAnwajler, Beata, and Anna Witek-Krowiak. 2023. "Three-Dimensional Printing of Multifunctional Composites: Fabrication, Applications, and Biodegradability Assessment" Materials 16, no. 24: 7531. https://doi.org/10.3390/ma16247531
APA StyleAnwajler, B., & Witek-Krowiak, A. (2023). Three-Dimensional Printing of Multifunctional Composites: Fabrication, Applications, and Biodegradability Assessment. Materials, 16(24), 7531. https://doi.org/10.3390/ma16247531







