The Synthesis, Structure, and Luminescent Properties of TmMgB5O10 Crystals
Abstract
:1. Introduction
2. Materials and Methods
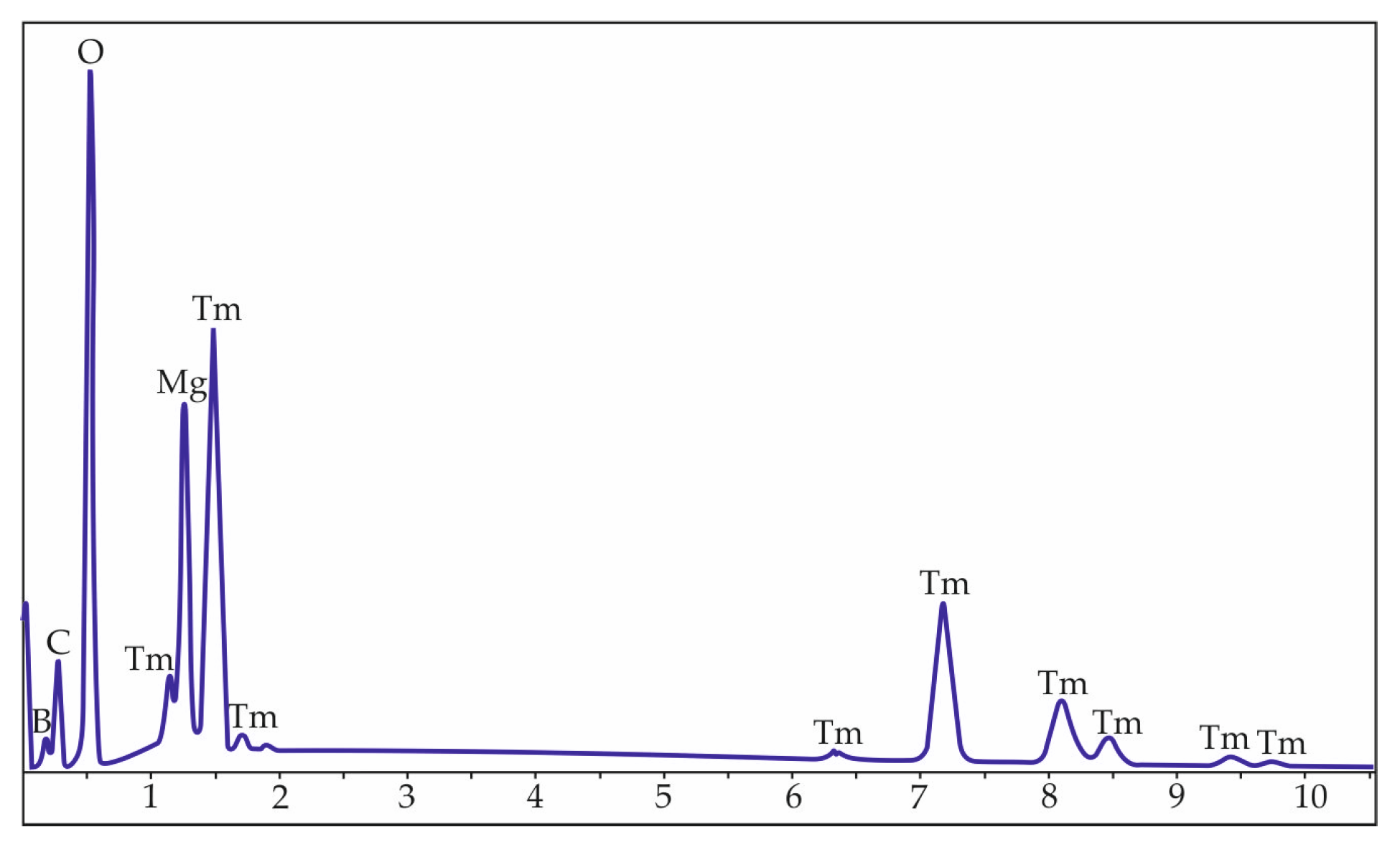
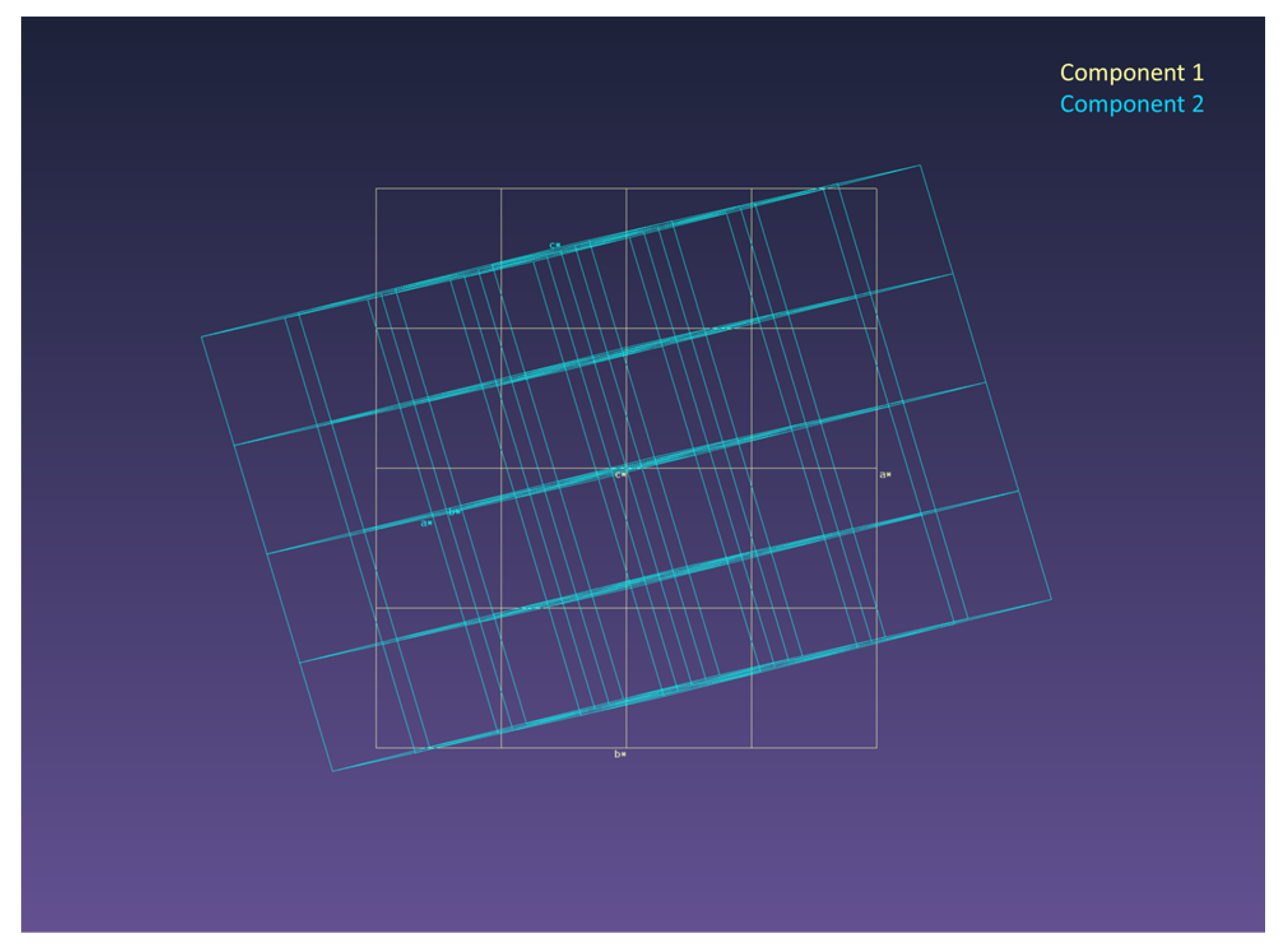
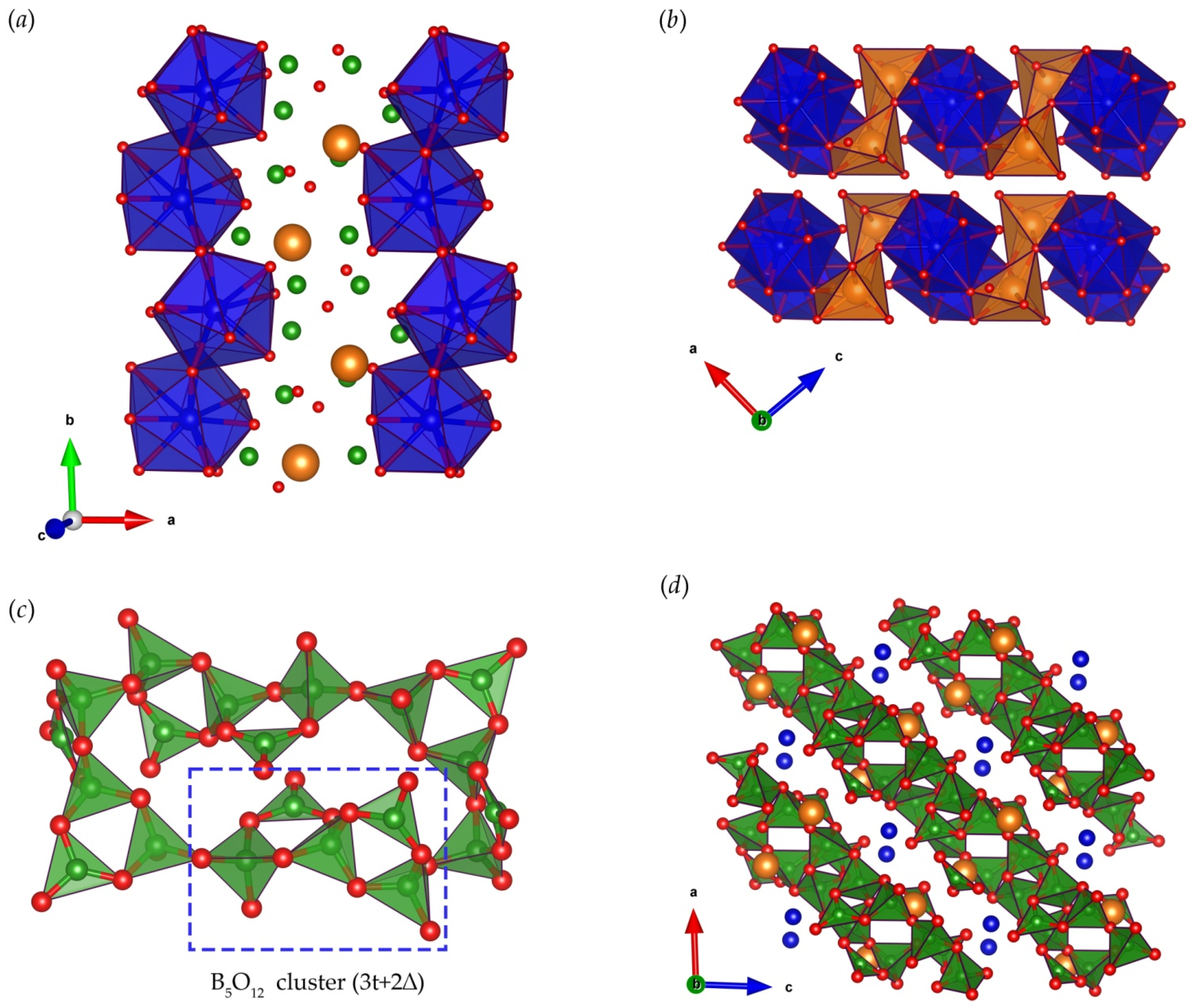
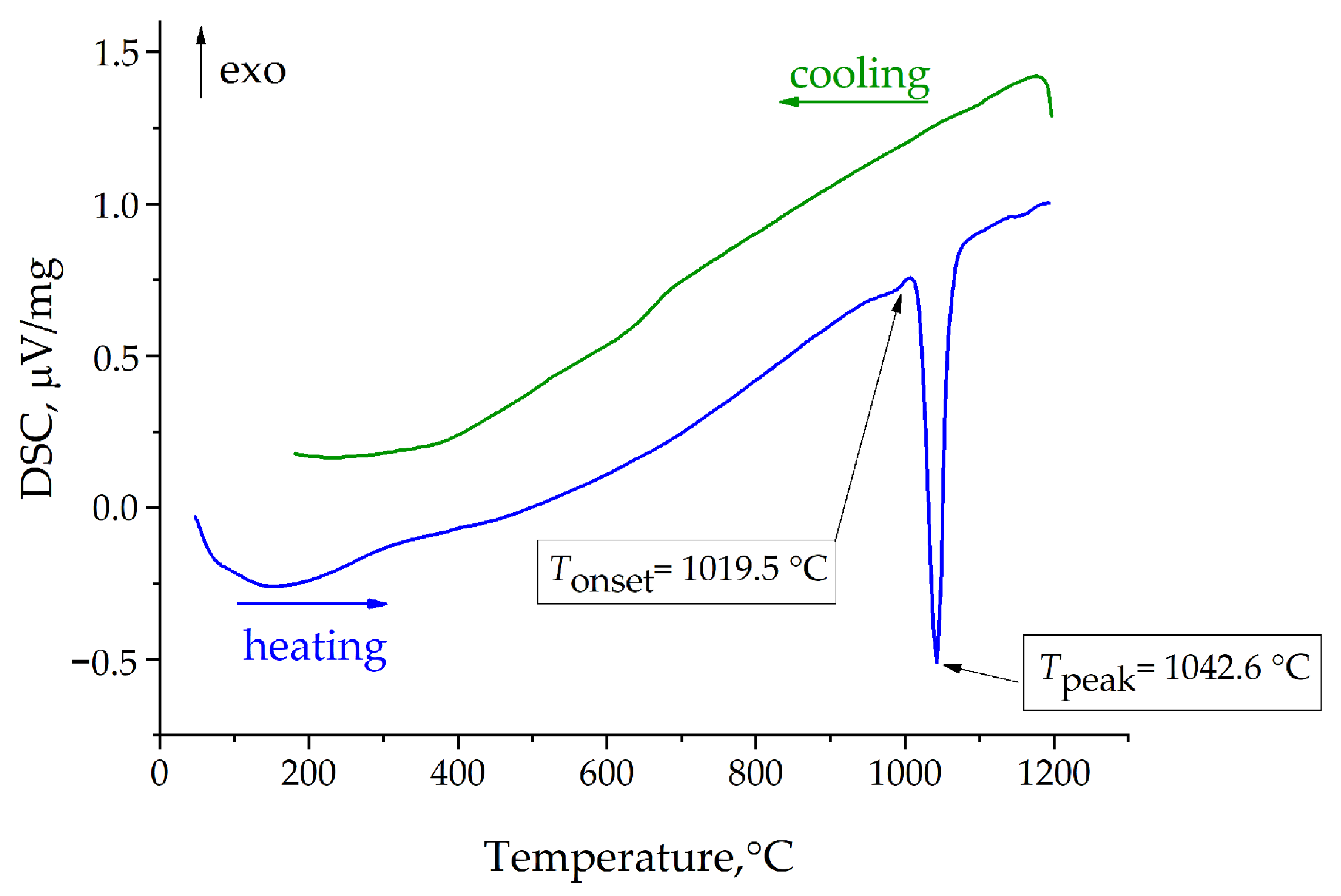
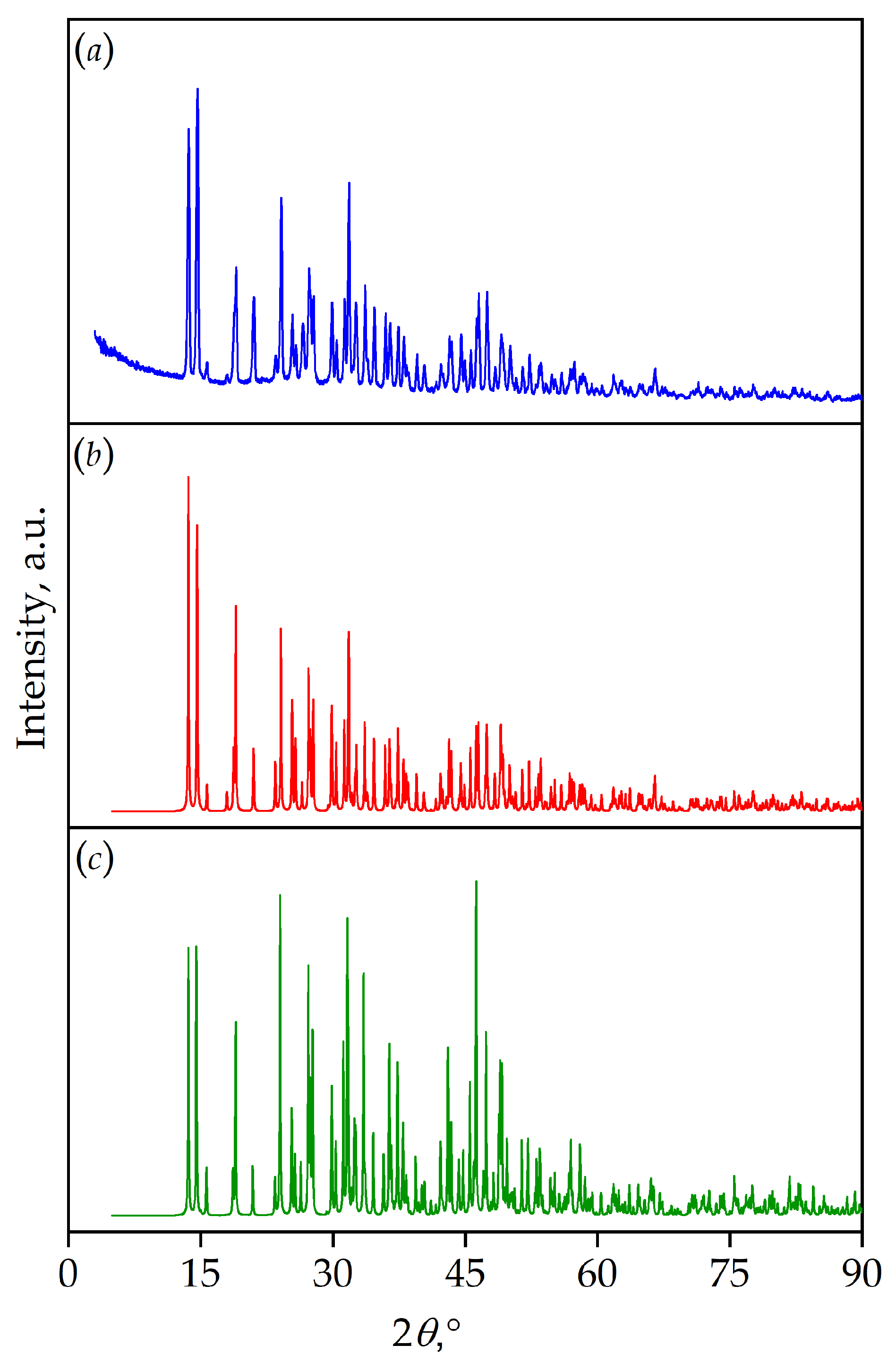
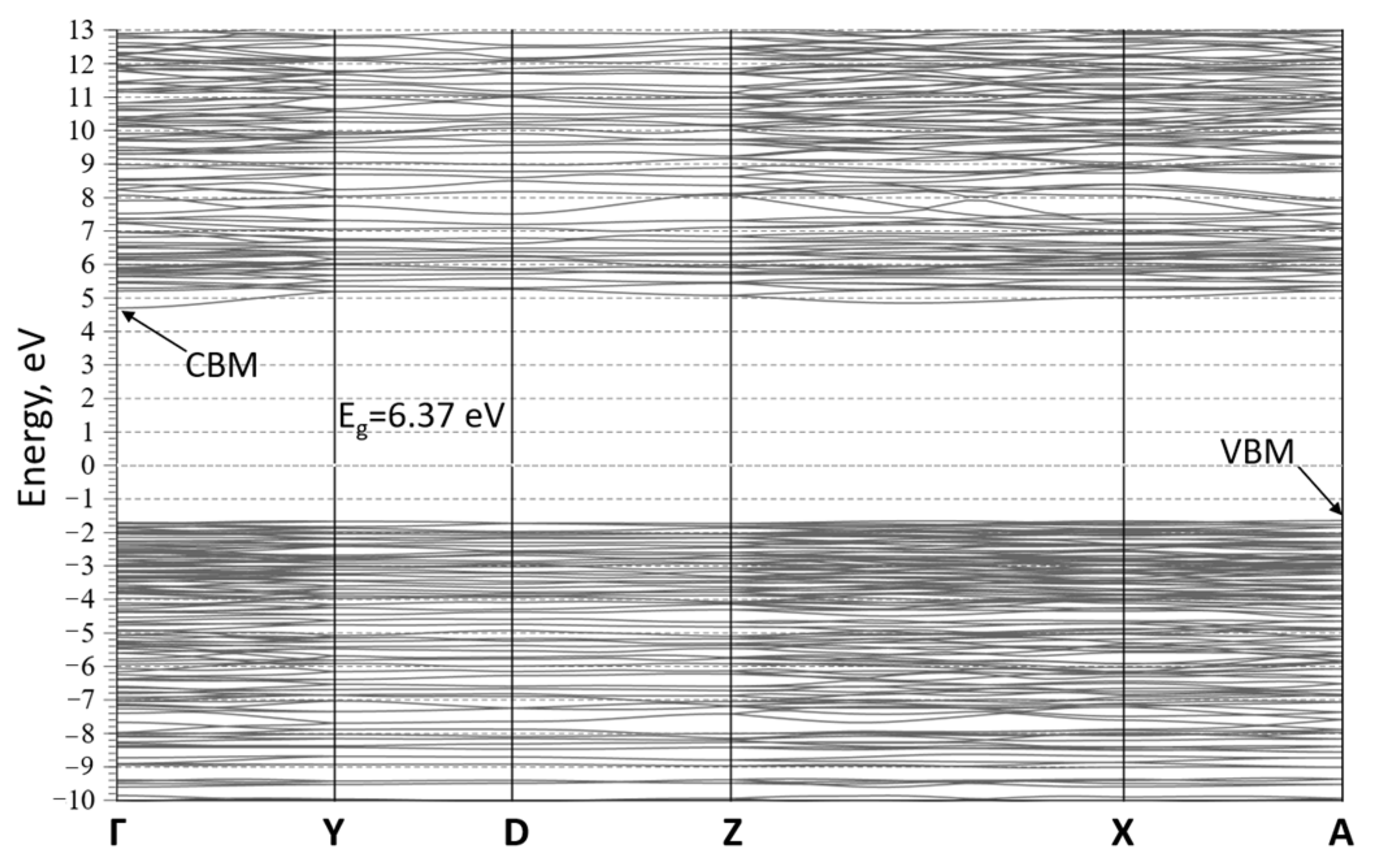
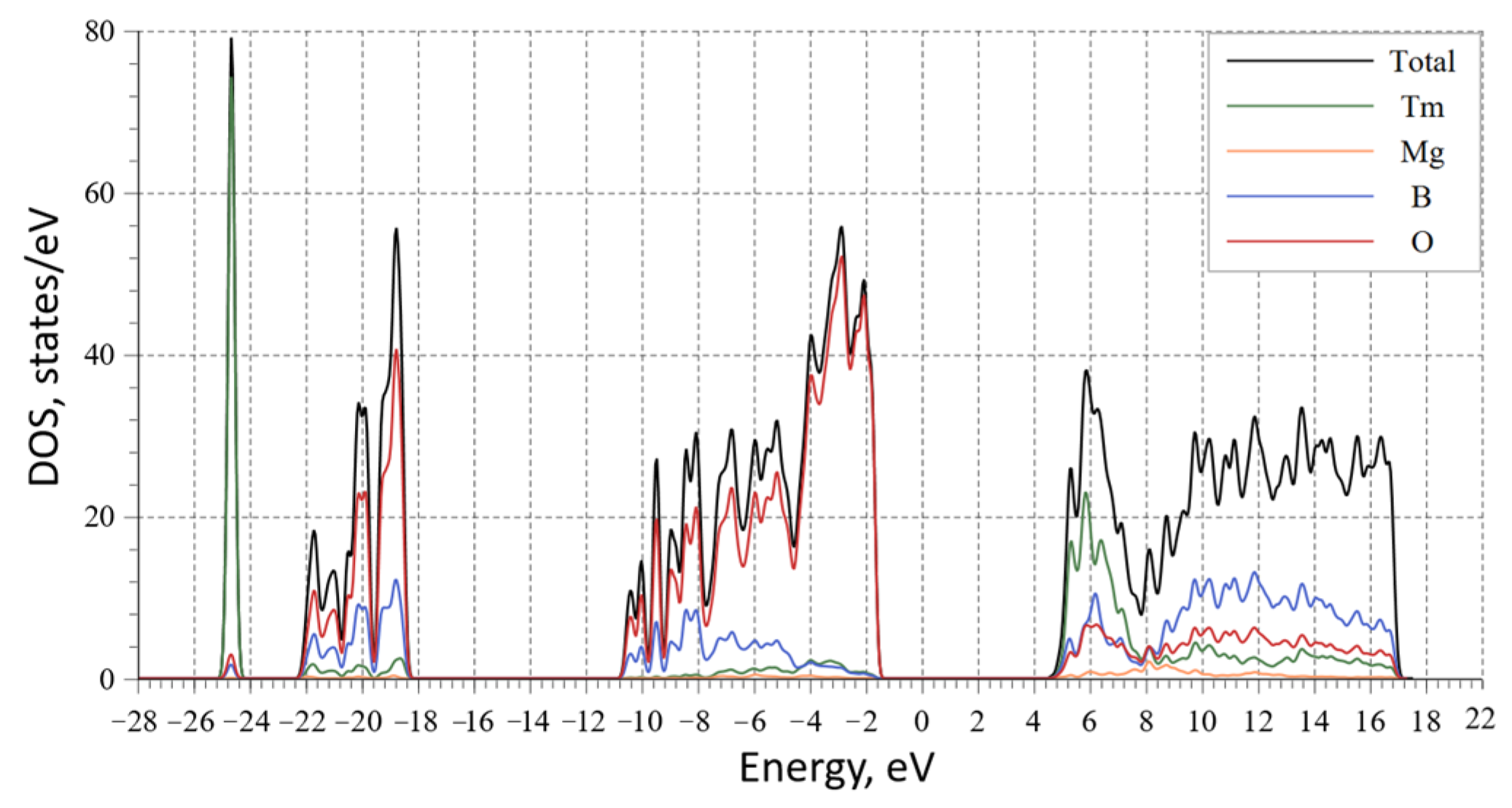
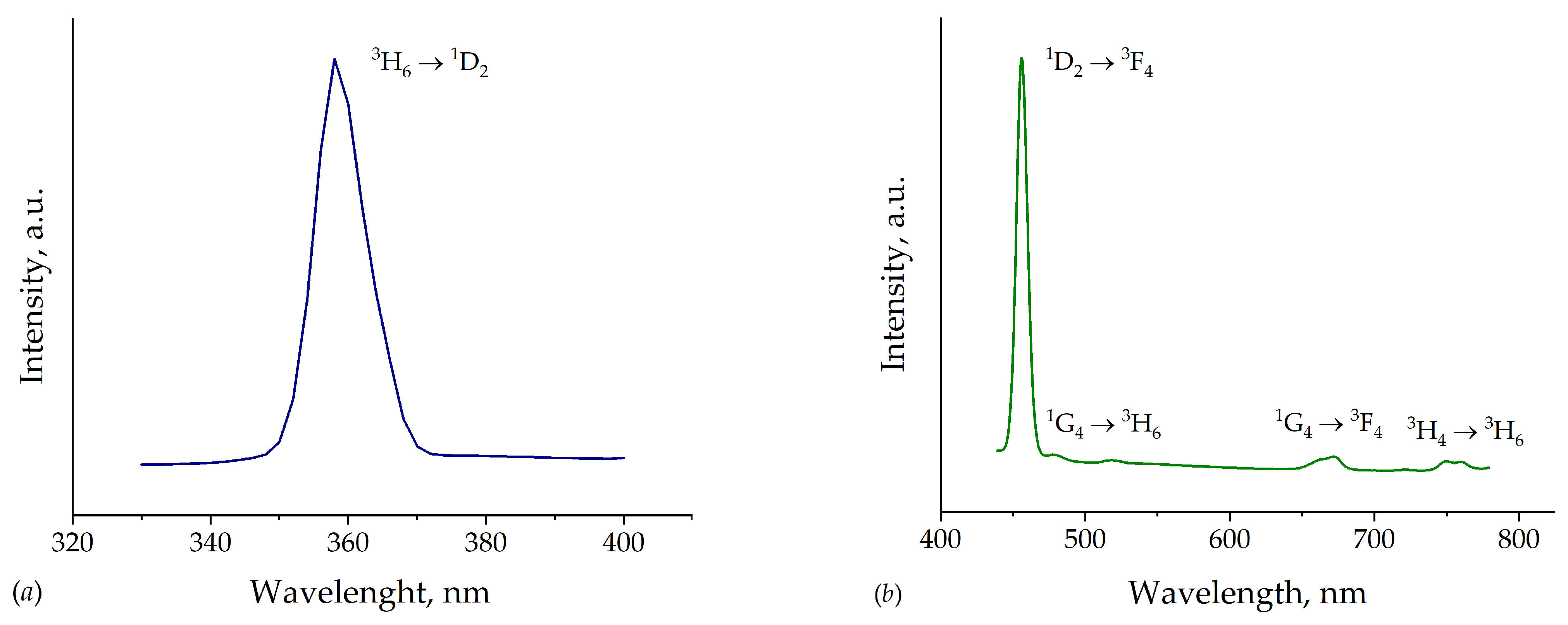
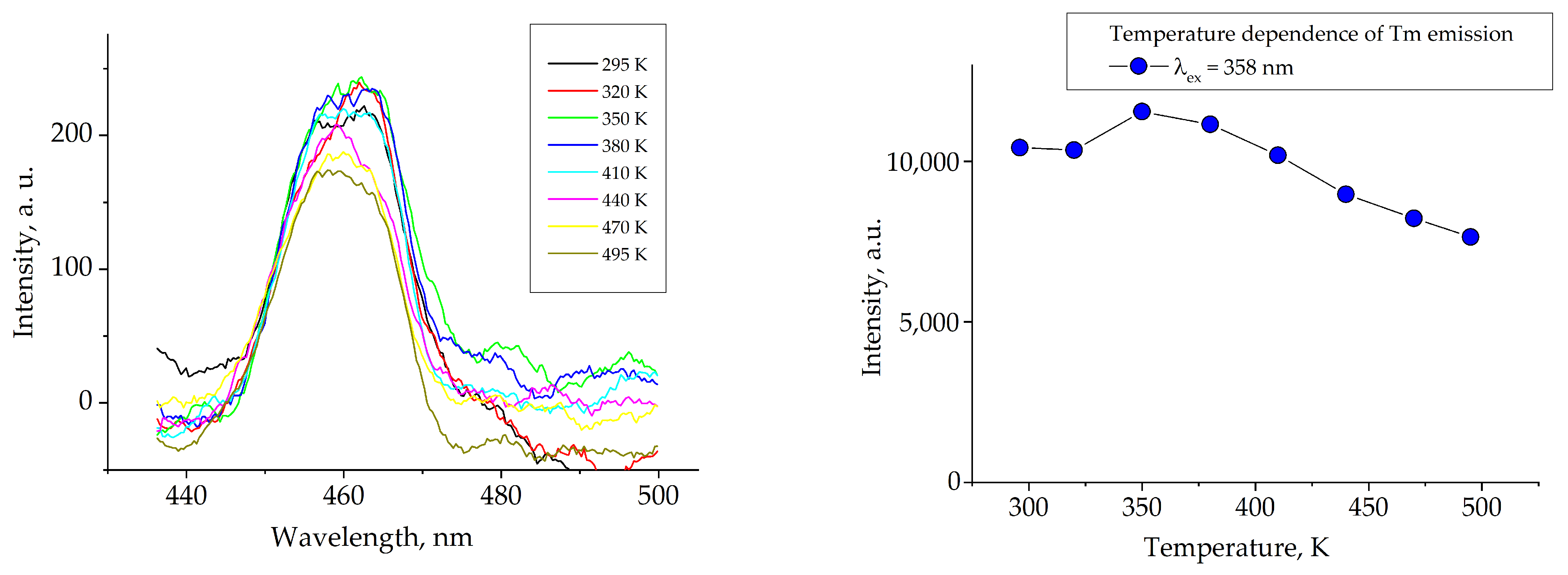
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leonyuk, N.I.; Maltsev, V.V.; Volkova, E.A. Crystal chemistry of high-temperature borates. Molecules 2020, 25, 2450. [Google Scholar] [CrossRef]
- Hölsä, J.; Leskelä, M. Fluorescence spectrum, energy level scheme and crystal field analysis of europium(+III) doped lanthanum magnesium borate LaMgB5O10:Eu3+. Mol. Phys. 1985, 54, 657–667. [Google Scholar] [CrossRef]
- Mitina, D.D.; Maltsev, V.V.; Deineko, D.V.; Volkova, E.A.; Koporulina, E.V.; Kuzmin, N.N.; Kosorukov, V.L.; Jiliaeva, A.I. Luminescent properties of lanthanum-magnesium pentaborate crystals doped by Tb3+ and Eu3+ ions. J. Inorg. Chem. 2023, 9, 117821. [Google Scholar]
- Saubat, B.; Vlasse, M.; Fouassier, C. Synthesis and structural study of the new rare earth magnesium borates LnMgB5O10 (Ln = La, …, Er). J. Solid State Chem. 1980, 34, 271–277. [Google Scholar] [CrossRef]
- Fouassier, C.; Saubat, B.; Hagenmuller, P. Self-quenching of Eu3+ and Tb3+ luminescencein LaMgB5O10: A host structure allowing essentially one-dimensional interactions. J. Lumin. 1981, 23, 405–412. [Google Scholar] [CrossRef]
- Saubat, B.; Fouassier, C.; Hagenmuller, P. Luminescent efficiency of Eu3+ and Tb3+ in LaMgB5O10—Type borates under excitation from 100 to 400 nm. Mater. Res. Bull. 1981, 16, 193–198. [Google Scholar] [CrossRef]
- Knitel, M.J.; Dorenbos, P.; van Eijk, C.W.E.; Plasteig, B.; Viana, B.; Kahn-Harari, A.; Vivien, D. Photoluminescence, and scintillation/thermoluminescence yields of several Ce3+ and Eu2+ activated borates. Nucl. Instrum. Methods Phys. Res. A 2000, 443, 364–374. [Google Scholar] [CrossRef]
- Lin, C.K.; Yu, M.; Pang, M.L.; Lin, J. Photoluminescent properties of sol–gel derived (La,Gd)MgB5O10:Ce3+/Tb3+ nanocrystalline thin films. Opt. Mater. 2006, 28, 913–918. [Google Scholar] [CrossRef]
- El Jouhari, N.; Parent, C.; Le Flem, G. Photoluminescence of Ce3+, Tb3+, and Mn2+ in Glasses of Base Composition LaMgB5O10. J. Solid State Chem. 1996, 123, 398–407. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, H.; Sun, S.; Yuan, F.; Zhang, L.; Lin, Z.; Zhang, G.; Wang, G. Growth, thermal, spectral and laser properties of Nd:LaMgB5O10 crystal—A new promising laser material. J. Alloys Compd. 2015, 646, 1083–1088. [Google Scholar] [CrossRef]
- Sun, S.; Wei, Q.; Li, B.; Shi, X.; Yuan, F.; Lou, F.; Zhang, L.; Lin, Z.; Zhong, D.; Huang, Y.; et al. The YMgB5O10 crystal preparation and attractive multi-wavelength emission characteristics of doping Nd3+ ions. J. Mater. Chem. C 2021, 9, 1945. [Google Scholar] [CrossRef]
- Huang, Y.; Lou, F.; Sun, S.; Yuan, F.; Zhang, L.; Lin, Z.; You, Z. Spectroscopy and laser performance of Yb3+:GdMgB5O10 crystal. J. Lumin. 2017, 188, 7–11. [Google Scholar] [CrossRef]
- Maltsev, V.V.; Mitina, D.D.; Belokoneva, E.L.; Volkova, E.A.; Koporulina, E.V.; Jiliaeva, A.I. Synthesis and flux-growth of rare-earth magnesium pentaborate crystals RMgB5O10 (R = Y, Gd, La, Tm and Yb). J. Cryst. Growth 2022, 587, 126628. [Google Scholar] [CrossRef]
- Leonyuk, N.I.; Leonyuk, L.I. Growth and characterization of RM3(BO3)4 crystals. Progr. Cryst. Growth Charact. 1995, 31, 179–278. [Google Scholar] [CrossRef]
- Rigaku Oxford Diffraction. CrysAlis Pro Software System; Version 1.171.64.119a; Rigaku Corporation: Oxford, UK, 2018. [Google Scholar]
- Giannozzi, P.; Baroni, S.; Bonini, N.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Chiarotti, G.L.; Cococcioni, M.; Dabo, I.; et al. QUANTUM ESPRESSO: A modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 2009, 21, 395502. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Lin, J.S.; Qteish, A.; Payne, M.C.; Heine, V. Optimized and transferable nonlocal separableab initiopseudopotentials. Phys. Rev. B 1993, 47, 4174–4180. [Google Scholar] [CrossRef]
- Petricek, V.; Dusek, M.; Palatinus, L. Crystallographic Computing System JANA2006: General features. Z. Kristallogr. 2014, 229, 345–352. [Google Scholar] [CrossRef]
- Zhang, J.; Tao, X.; Cai, G.; Jin, Z. Phase relation, structure, and properties of borate MgYB5O10 in MgO–Y2O3–B2O3 system. Powder Diffr. 2017, 32, 97–106. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, W.; Sun, S.; Yuan, F.; Zhang, L.; Zhao, W.; Wang, G.; Lin, Z. Growth, structure, spectral and laser properties of Yb3+:LaMgB5O10– a new laser material. CrystEngComm 2015, 17, 7392–7397. [Google Scholar] [CrossRef]
- Palatinus, L. Ab initio determination of incommensurately modulated structures by charge-flipping in super space. Acta Cryst. A 2004, 60, 604–610. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Gorbachenya, K.N.; Yasukevich, A.S.; Lazarchuk, A.I.; Kisel, V.E.; Kuleshov, N.V.; Volkova, E.A.; Maltsev, V.V.; Koporulina, E.V.; Yapaskurt, V.O.; Kuzmin, N.N.; et al. Growth and Spectroscopy of Yb:YMgB5O10 Crystal. Crystals 2022, 12, 986. [Google Scholar] [CrossRef]
- Yao, L.; Chen, G.; Yang, T.; Yuan, C.; Zhou, C. Energy transfer, optical and luminescent properties in Tm3+/Tb3+/Sm3+ tri-doped borate glasses. J. Mater. Sci. Mater. Electron. 2016, 28, 553–558. [Google Scholar] [CrossRef]
- Kuwik, M.; Kowalska, K.; Pisarska, J.; Pisarski, W.A. Spectroscopic Properties of Pr3+, Tm3+, and Ho3+ in Germanate-Based Glass Systems Modified by TiO2. Materials 2023, 16, 61. [Google Scholar] [CrossRef]
- Borrego-Pérez, J.A.; González, F.; Meza-Avendaño, C.A.; De Los Santos, I.M.; López-Juárez, R.; Hernández, I.; Alonso-Guzman, E.M.; Martinez-Molina, W.; Chavez-Garcia, H.L. Structural, optical and photoluminescence properties of TiO2 and TiO2: Tm3+ nanopowders. Optik 2021, 227, 166083. [Google Scholar] [CrossRef]
- Guerbous, L.; Derbal, M.; Chaminade, J.P. Photoluminescence and energy transfer of Tm3+ doped LiIn (WO4)2 blue phosphors. J. Lumin. 2010, 130, 2469–2475. [Google Scholar] [CrossRef]
- Kozhan, T.M.; Kuznetsova, V.V.; Pershukevich, P.P.; Sergeev, I.I.; Khomenko, V.S.; Chernyavskii, V.A. Concentration dependence of the quantum yield of luminescence of Tm3+ ions in gadolinium oxychloride. J. Appl. Spectrosc. 2004, 71, 829–836. [Google Scholar] [CrossRef]
- Makhov, V.N.; Uvarova, T.V.; Kirm, M.; Vielhauer, S. Thermal quenching of luminescence of BaY2F8 crystals activated with Er3+ and Tm3+ ions. Bull. Lebedev Phys. Inst. 2016, 43, 348–351. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Xu, S.; Yu, H.; Zhang, J.; Zhang, X.; Cao, Y.; Cheng, L.; Sun, J.; Chen, B. Temperature-dependent luminescence properties of Dy3+, Tm3+ single-/co-doped YNbO4 phosphors. Optik 2021, 238, 166524. [Google Scholar] [CrossRef]

| Chemical Formula | TmMgB5O10 |
|---|---|
| sp. gr., Z | P21/n, 4 |
| a, b, c, Å | 8.476(1) 7.577(1) 9.368(1) |
| β, ° | 94.035(3) |
| V, Å | 600.1(1) |
| D, g/cm3 | 4.508 |
| Radiation; λ, Å | MoKα; 0.71069 |
| μ, mm−1 | 14.946 |
| T, K | 295 |
| Diffractometer | Xcalibur |
| Scan type | ω |
| Absorption coefficient; Tmin/Tmax | By cut; 0.125/1.000 |
| θmax, ° | 30.61 |
| Limitation of h, k, l | –11 ≤ h ≤ 12; –10 ≤ k ≤ 10; –12 ≤ l ≤ 13 |
| The number of reflections: measured/independent (N1)/I > 3σ(I) (N2) | 8445/1601/1303 |
| Rint, % | 19.2 |
| The method of refinement | OLS by F2 |
| The number of refined parameters | 71 |
| Consideration of extinction, k | Type 2 |
| R1/Rw2 on N1 | 0.0543/0.1045 |
| R1/Rw2 on N2 | 0.0638/0.1056 |
| S | 2.42 |
| Δρmin/Δρmax | –5.54/5.50 |
| Atom | x/a | y/b | z/c | Ueq, Å2 |
|---|---|---|---|---|
| Tm1 | 0.3152(1) | 0.6865(1) | 0.2582(1) | 0.0065(1) |
| Mg1 | 0.596(1) | 0.406(1) | 0.130(1) | 0.005(1) |
| B1 | 0.335(1) | 0.898(1) | 0.491(1) | 0.006(1) |
| B2 | 0.436(1) | 0.186(1) | −0.089(1) | 0.006(1) |
| B3 | −0.084(1) | 0.573(1) | 0.254(1) | 0.008(1) |
| B4 | 0.517(1) | 0.676(1) | 0.602(1) | 0.003(1) |
| B5 | 0.224(1) | −0.034(1) | −0.056(1) | 0.006(1) |
| O1 | 0.250(1) | 0.945(1) | 0.370(1) | 0.006(1) |
| O2 | 0.320(1) | 0.124(1) | −0.011(1) | 0.007(1) |
| O3 | 0.812(1) | 0.532(1) | 0.121(1) | 0.006(1) |
| O4 | 0.682(1) | 0.150(1) | 0.140(1) | 0.006(1) |
| O5 | 0.451(1) | 0.770(1) | 0.475(1) | 0.007(1) |
| O6 | 0.508(1) | 0.350(1) | −0.073(1) | 0.006(1) |
| O7 | 0.490(1) | 0.093(1) | −0.201(1) | 0.008(1) |
| O8 | 0.583(1) | 0.472(1) | 0.351(1) | 0.007(1) |
| O9 | 0.038(1) | 0.705(1) | 0.228(1) | 0.002(1) |
| O10 | 0.677(1) | 0.611(1) | 0.580(1) | 0.007(1) |
| Atoms | Distance, Å | Atoms | Distance, Å |
|---|---|---|---|
| Tm1–O1 | 2.304(7) | B2–O2 | 1.347(13) |
| Tm1–O1 | 2.236(7) | B2–O6 | 1.387(12) |
| Tm1–O2 | 2.747(7) | B2–O7 | 1.363(13) |
| Tm1–O5 | 2.354(7) | <B2–O> | 1.365 |
| Tm1–O6 | 2.389(7) | B3–O3 | 1.512(12) |
| Tm1–O7 | 2.441(7) | B3–O4 | 1.456(13) |
| Tm1–O8 | 2.875(6) | B3–O7 | 1.452(13) |
| Tm1–O9 | 2.355(6) | B3–O9 | 1.467(12) |
| Tm1–O10 | 2.713(7) | <B3–O> | 1.472 |
| Tm1–O10 | 2.497(1) | B4–O5 | 1.462(12) |
| <Tm1–O> | 2.4911 | B4–O8 | 1.492(12) |
| Mg1–O3 | 2.067(7) | B4–O9 | 1.485(12) |
| Mg1–O4 | 2.071(8) | B4–O10 | 1.472(12) |
| Mg1–O6 | 2.040(7) | <B4–O> | 1.478 |
| Mg1–O6 | 2.104(7) | B5–O2 | 1.490(12) |
| Mg1–O8 | 2.137(7) | B5–O4 | 1.455(13 |
| Mg1–O9 | 2.371(7) | B5–O8 | 1.504(11) |
| <Mg1–O> | 2.131 | B5–O10 | 1.483(13) |
| B1–O1 | 1.347(12) | <B5–O> | 1.483 |
| B1–O3 | 1.353(13) | ||
| B1–O5 | 1.398(12) | ||
| <B1–O> | 1.366 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Volkova, E.A.; Maltsev, V.V.; Antipin, A.M.; Deyneko, D.V.; Nikiforov, I.V.; Spassky, D.A.; Marchenko, E.I.; Mitina, D.D.; Kosorukov, V.L.; Yapaskurt, V.O.; et al. The Synthesis, Structure, and Luminescent Properties of TmMgB5O10 Crystals. Materials 2023, 16, 6084. https://doi.org/10.3390/ma16186084
Volkova EA, Maltsev VV, Antipin AM, Deyneko DV, Nikiforov IV, Spassky DA, Marchenko EI, Mitina DD, Kosorukov VL, Yapaskurt VO, et al. The Synthesis, Structure, and Luminescent Properties of TmMgB5O10 Crystals. Materials. 2023; 16(18):6084. https://doi.org/10.3390/ma16186084
Chicago/Turabian StyleVolkova, Elena A., Victor V. Maltsev, Alexander M. Antipin, Dina V. Deyneko, Ivan V. Nikiforov, Dmitry A. Spassky, Ekaterina I. Marchenko, Diana D. Mitina, Vladimir L. Kosorukov, Vasiliy O. Yapaskurt, and et al. 2023. "The Synthesis, Structure, and Luminescent Properties of TmMgB5O10 Crystals" Materials 16, no. 18: 6084. https://doi.org/10.3390/ma16186084
APA StyleVolkova, E. A., Maltsev, V. V., Antipin, A. M., Deyneko, D. V., Nikiforov, I. V., Spassky, D. A., Marchenko, E. I., Mitina, D. D., Kosorukov, V. L., Yapaskurt, V. O., Naprasnikov, D. A., & Koporulina, E. V. (2023). The Synthesis, Structure, and Luminescent Properties of TmMgB5O10 Crystals. Materials, 16(18), 6084. https://doi.org/10.3390/ma16186084








