Applications of Phyto-Nanotechnology for the Treatment of Neurodegenerative Disorders
Abstract
1. Introduction
2. Materials and Methods
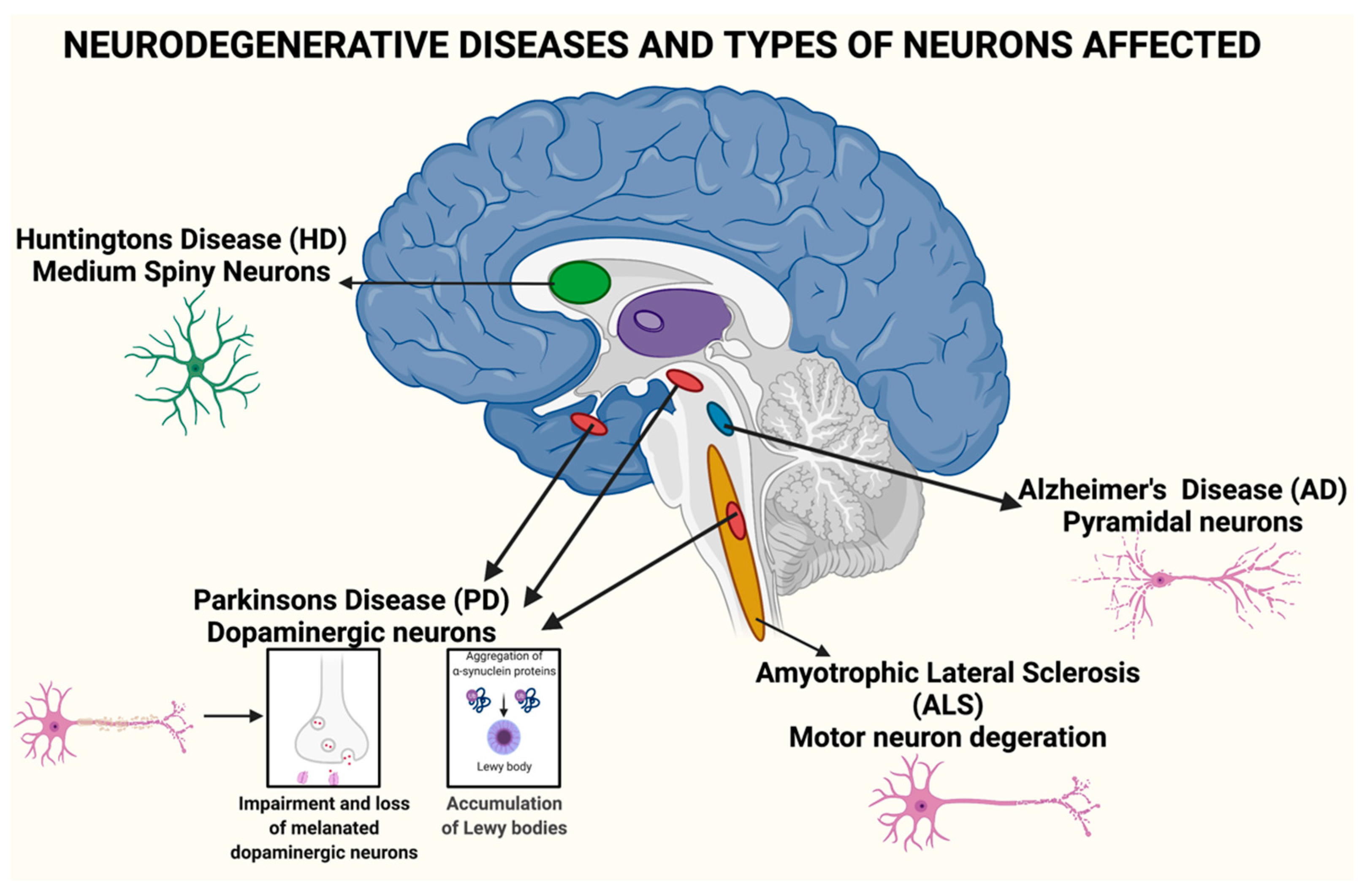
3. Real Problem of Neurodegenerative Disease
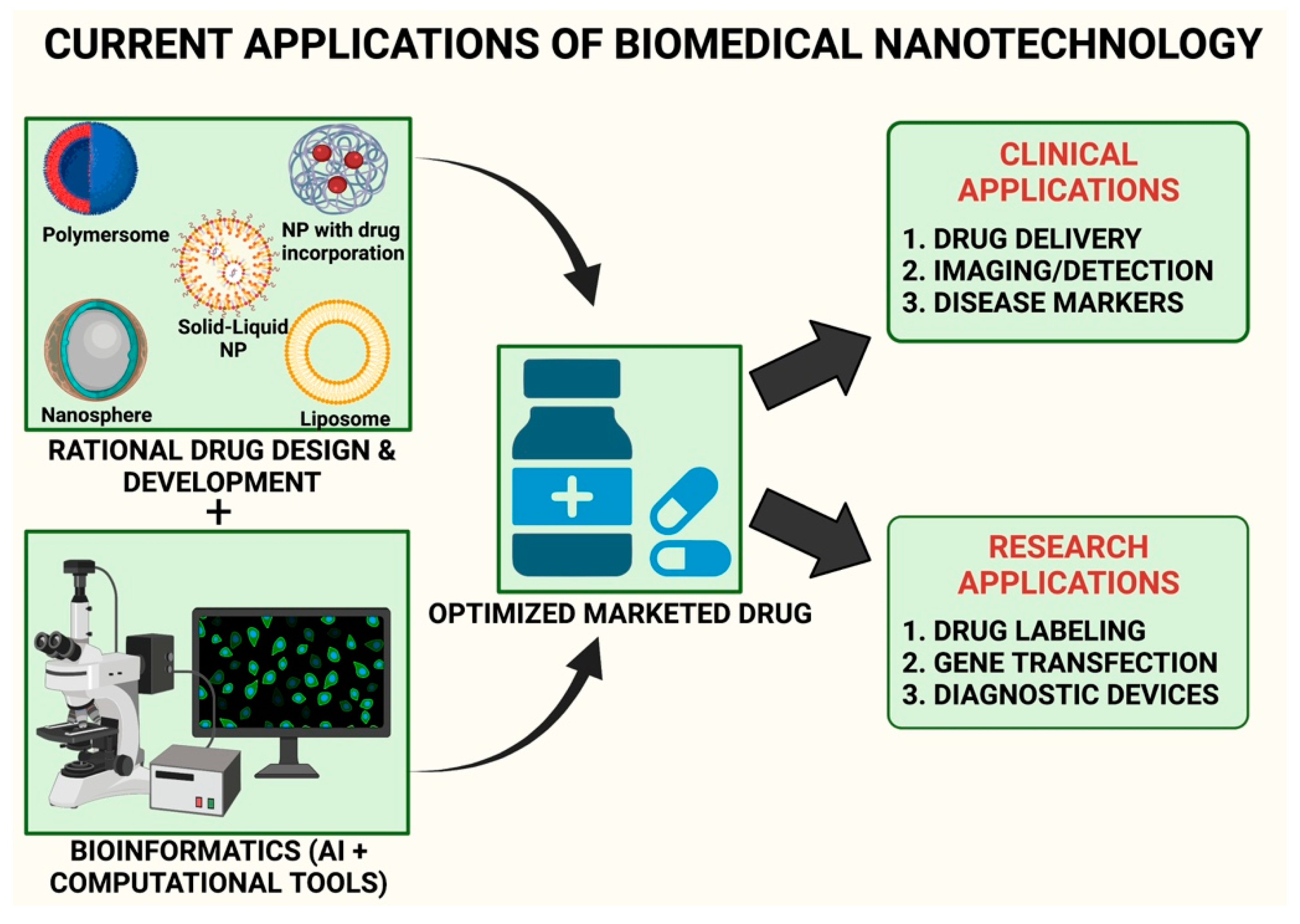
4. Role of Nanotechnology in Neurodegenerative Disorders
4.1. Techniques for Preparation of Nanoparticles and Nanocapsules
4.1.1. Nanoprecipitation
4.1.2. Emulsification-Diffusion Method
4.1.3. Double Emulsion Technique
4.2. Green Method of Synthesis of Nanoparticles
5. Role of Phyto-Nanomedicine on Neurodegenerative Diseases Treatment
5.1. Types of Phyto-Medicines Available for Treatment
- ▪
- Acorus calamus
- ▪
- Allium sativum
- ▪
- Bacopa monnieri
- ▪
- Centella asiatica
- ▪
- Curcuma longa
- ▪
- Celastrus paniculatus Wild
- ▪
- Coriandrum sativum L
- ▪
- Galanthus nivalis
- ▪
- Ginkgo biloba
- ▪
- Glycyrrhiza glabra
- ▪
- Hypericum perforatum
- ▪
- Lycopodium serratum
- ▪
- Melissa officinalis
- ▪
- Ocimum sanctum
- ▪
- Panax Ginseng
- ▪
- Rosmarinus officinalis
- ▪
- Salvia officinalis
- ▪
- Terminalia chebula
- ▪
- Tinospora cordifolia
- ▪
- Withania somnifera
- ▪
- Zizyphus Jujube
5.2. Conventional Approach
6. Recent Trends of Phyto-Neuro Medicine
6.1. Protein-Based Nanoparticles
6.2. Polymeric Nanoparticles
6.2.1. Liposomes-Based Drug Delivery Systems
6.2.2. Green Synthesized Nanoparticles
7. Nanotechnology-Induced Bioinformatics for Early Diagnosis
8. Limitations of Nanotechnology-Based Approaches for ND
9. Future Nano Therapeutics for ND
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BBB | Blood brain barrier |
| CNS | Central nervous system |
| ND | Neurological and neurodegenerative diseases |
| NP | Nanoparticle |
| AD | Alzheimer’s disease |
| PD | Parkinson’s disease |
| ALS | Amyotrophic horizontal sclerosis |
| JNK | c-Jun N-terminal kinase |
| AChE | Acetylcholinesterase |
| IC50 | Half-maximal inhibitory concentration |
| SAC | S-allyl cysteine |
| MPP | Phenyl pyridinium |
| SOD | Superoxide dismutase |
| 6-OHDA | 6-hydroxydopamine |
| CAT | Chloramphenicol acetyltransferase |
| GPx | Gluthathione peroxidase |
| GPR | Glutathione reductase |
| ATP | Adenosine triphosphate |
| iNOS | Inducible nitric oxide synthase |
| COX-2: | Cyclo-oxygenase 2 |
| MDA | Malondialdehyde |
| GSH | Glutathione |
| CPPME | Celastrus paniculate wild whole plant methanolic extract |
| LPO | Lipid peroxidation |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| GABAA | Gamma- amino butyric acid |
| MAO | Monoamine oxidases |
| DNA | Deoxyribo nucleic acid |
| mRNA | Messenger ribonucleic acid |
| 5-HT | 5-hydroxytrptamine/serotonin |
| DA | Dopamine |
| ROS | Reactive oxygen species |
| VMAT | Vesicular monoamine transporter |
| NMDA | N-methyl-D-aspartic acid |
| COMT | Catechol ortho-methyl transferase |
| CSF | Cerebrospinal fluid |
| PET | Positron emission tomography |
| SPECT | Single photon emission computed tomography |
| NIH | National Institute of Health |
| NSF | National Science Foundation |
| USFDA | United States Food and Drug Administration |
| STAIR | Stroke Therapy Academic Industry Roundtable |
| FITBIR | Federal Interagency Traumatic Brain Injury Research |
References
- Bensadoun, J.-C.; Almeida, L.P.d.; Fine, E.G.; Tseng, J.L.; Déglon, N.; Aebischer, P. Comparative study of GDNF delivery systems for the CNS: Polymer rods, encapsulated cells, and lentiviral vectors. J. Control. Release 2003, 87, 107–115. [Google Scholar] [CrossRef]
- Kishima, H.; Poyot, T.; Bloch, J.; Dauguet, J.; Conde, F.; Dolle, F.; Hinnen, F.; Pralong, W.; Palfi, S.; Deglon, N.; et al. Encapsulated GDNF-producing C2C12 cells for Parkinson’s disease: A pre-clinical study in chronic MPTP-treated baboons. Neurobiol. Dis. 2004, 16, 428–439. [Google Scholar] [CrossRef]
- McArthur, J.C. HIV dementia: An evolving disease. J. Neuroimmunol. 2004, 157, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Menei, P.; Montero-Menei, C.; Venier, M.C.; Benoit, J.P. Drug delivery into the brain using poly(lactide-co-glycolide) microspheres. Expert Opin. Drug Deliv. 2005, 2, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Flachenecker, P. Epidemiology of neuroimmunological diseases. J. Neurol. 2006, 253 (Suppl. 5), V2–V8. [Google Scholar] [CrossRef]
- Kabanov, A.V.; Gendelman, H.E. Nanomedicine in the diagnosis and therapy of neurodegenerative disorders. Prog. Polym. Sci. 2007, 32, 1054–1082. [Google Scholar] [CrossRef]
- Ferri, C.P.; Prince, M.; Brayne, C.; Brodaty, H.; Fratiglioni, L.; Ganguli, M.; Hall, K.; Hasegawa, K.; Hendrie, H.; Huang, Y.; et al. Global prevalence of dementia: A Delphi consensus study. Lancet 2005, 366, 2112–2117. [Google Scholar] [CrossRef]
- Popovic, N.; Brundin, P. Therapeutic potential of controlled drug delivery systems in neurodegenerative diseases. Int. J. Pharm. 2006, 314, 120–126. [Google Scholar] [CrossRef]
- Singh, N.; Pillay, V.; Choonara, Y.E. Advances in the treatment of Parkinson’s disease. Prog. Neurobiol. 2007, 81, 29–44. [Google Scholar] [CrossRef]
- Tysnes, O.B.; Storstein, A. Epidemiology of Parkinson’s disease. J. Neura. Transm. (Vienna) 2017, 124, 901–905. [Google Scholar] [CrossRef]
- Freed, C.R.; Greene, P.E.; Breeze, R.E.; Tsai, W.Y.; DuMouchel, W.; Kao, R.; Dillon, S.; Winfield, H.; Culver, S.; Trojanowski, J.Q.; et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N. Engl. J. Med. 2001, 344, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Pollak, P.; Fraix, V.; Krack, P.; Moro, E.; Mendes, A.; Chabardes, S.; Koudsie, A.; Benabid, A.-L. Treatment results: Parkinson’s disease. Mov. Disord. 2002, 17, S75–S83. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.G. Late-onset neurodegenerative diseases—The role of protein insolubility. J. Anat. 2000, 196 Pt 4, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Dickson, D.W. Misfolded, protease-resistant proteins in animal models and human neurodegenerative disease. J. Clin. Investig. 2002, 110, 1403–1405. [Google Scholar] [CrossRef]
- Woodruff, B.K.; Graff-Radford, N.R.; Ferman, T.J.; Dickson, D.W.; DeLucia, M.W.; Crook, J.E.; Arvanitakis, Z.; Brassler, S.; Waters, C.; Barker, W.; et al. Family history of dementia is a risk factor for Lewy body disease. Neurology 2006, 66, 1949–1950. [Google Scholar] [CrossRef]
- Walia, V.; Kaushik, D.; Mittal, V.; Kumar, K.; Verma, R.; Parashar, J.; Akter, R.; Rahman, M.H.; Bhatia, S.; Al-Harrasi, A.; et al. Delineation of Neuroprotective Effects and Possible Benefits of AntioxidantsTherapy for the Treatment of Alzheimer’s Diseases by Targeting Mitochondrial-Derived Reactive Oxygen Species: Bench to Bedside. Mol. Neurobiol. 2021. [Google Scholar] [CrossRef]
- Nischwitz, V.; Berthele, A.; Michalke, B. Speciation analysis of selected metals and determination of their total contents in paired serum and cerebrospinal fluid samples: An approach to investigate the permeability of the human blood-cerebrospinal fluid-barrier. Anal. Chim. Act. 2008, 627, 258–269. [Google Scholar] [CrossRef]
- Gendelman, H.E.; Ding, S.; Gong, N.; Liu, J.; Ramirez, S.H.; Persidsky, Y.; Mosley, R.L.; Wang, T.; Volsky, D.J.; Xiong, H. Monocyte chemotactic protein-1 regulates voltage-gated K+ channels and macrophage transmigration. J. Neuroimmune Pharm. 2009, 4, 47–59. [Google Scholar] [CrossRef]
- Modi, G.; Pillay, V.; Choonara, Y.E.; Ndesendo, V.M.; du Toit, L.C.; Naidoo, D. Nanotechnological applications for the treatment of neurodegenerative disorders. Prog. Neurobiol. 2009, 88, 272–285. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Moher, D. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J. Clin. Epidemiol. 2021, 134, 103–112. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. J. Clin. Epidemiol. 2021, 134, 178–189. [Google Scholar] [CrossRef]
- Kim, J.; Mook-Jung, I. Special issue on neurodegenerative diseases and their therapeutic approaches. Exp. Mol. Med. 2015, 47, e146. [Google Scholar] [CrossRef]
- Ballabh, P.; Braun, A.; Nedergaard, M. The blood-brain barrier: An overview: Structure, regulation, and clinical implications. Neurobiol. Dis. 2004, 16, 1–13. [Google Scholar] [CrossRef]
- Abbott, N.J.; Patabendige, A.A.; Dolman, D.E.; Yusof, S.R.; Begley, D.J. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef]
- Silva, G.A. Neuroscience nanotechnology: Progress, opportunities and challenges. Nat. Rev. Neurosci. 2006, 7, 65–74. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Nanomedicine: Current status and future prospects. FASEB J. 2005, 19, 311–330. [Google Scholar] [CrossRef]
- Goldsmith, M.; Abramovitz, L.; Peer, D. Precision nanomedicine in neurodegenerative diseases. ACS Nano 2014, 8, 1958–1965. [Google Scholar] [CrossRef] [PubMed]
- Illum, L. Nasal drug delivery—Possibilities, problems and solutions. J. Control. Release 2003, 87, 187–198. [Google Scholar] [CrossRef]
- Pardridge, W.M. CSF, blood-brain barrier, and brain drug delivery. Expert Opin. Drug Deliv. 2016, 13, 963–975. [Google Scholar] [CrossRef] [PubMed]
- McDannold, N.; Arvanitis, C.D.; Vykhodtseva, N.; Livingstone, M.S. Temporary disruption of the blood-brain barrier by use of ultrasound and microbubbles: Safety and efficacy evaluation in rhesus macaques. Cancer Res. 2012, 72, 3652–3663. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, A.; Liu, M.; Ojha, T.; Storm, G.; Kiessling, F.; Lammers, T. Ultrasound-mediated drug delivery to the brain: Principles, progress and prospects. Drug Discov. Today Technol. 2016, 20, 41–48. [Google Scholar] [CrossRef]
- Cheng, Z.; Zhang, J.; Liu, H.; Li, Y.; Zhao, Y.; Yang, E. Central nervous system penetration for small molecule therapeutic agents does not increase in multiple sclerosis- and Alzheimer’s disease-related animal models despite reported blood-brain barrier disruption. Drug Metab. Dispos. 2010, 38, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Roy Chowdhury, S.; Mondal, S.; Muthuraj, B.; Balaji, S.N.; Trivedi, V.; Krishnan Iyer, P. Remarkably Efficient Blood-Brain Barrier Crossing Polyfluorene-Chitosan Nanoparticle Selectively Tweaks Amyloid Oligomer in Cerebrospinal Fluid and Abeta1-40. ACS Omega 2018, 3, 8059–8066. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Damian, G. Rotational Correlation Times of 3-Carbamoyl-2,2,5,5-Tetramethyl-3-Pyrrolin-1-Yloxy Spin Label with Respect to Heme and Nonheme Proteins. Biomacromolecules 2003, 4, 1630–1635. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, A.C.; Kumar, A.; Peng, Z.; Khan, A.; Junaid, M.; Ali, A.; Bharadwaj, S.; Wei, D.-Q. Evaluation and validation of synergistic effects of amyloid-beta inhibitor–gold nanoparticles complex on Alzheimer’s disease using deep neural network approach. J. Mater. Res. 2019, 34, 1845–1853. [Google Scholar] [CrossRef]
- Choi, C.H.; Alabi, C.A.; Webster, P.; Davis, M.E. Mechanism of active targeting in solid tumors with transferrin-containing gold nanoparticles. Proc. Natl. Acad. Sci. USA 2010, 107, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.D.; Lee, J.; Ramachandran, S.; Eliceiri, B.P.; Shubayev, V.I.; Lal, R.; Jin, S. Magnetic targeting of nanoparticles across the intact blood-brain barrier. J. Control. Release 2012, 164, 49–57. [Google Scholar] [CrossRef]
- Smith, M.W.; Gumbleton, M. Endocytosis at the blood-brain barrier: From basic understanding to drug delivery strategies. J. Drug Target. 2006, 14, 191–214. [Google Scholar] [CrossRef]
- Fessi, H.; Puisieux, F.; Devissaguet, J.P.; Ammoury, N.; Benita, S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int. J. Pharm. 1989, 55, R1–R4. [Google Scholar] [CrossRef]
- Mora-Huertas, C.E.; Fessi, H.; Elaissari, A. Influence of process and formulation parameters on the formation of submicron particles by solvent displacement and emulsification-diffusion methods critical comparison. Adv. Colloid. Interface Sci. 2011, 163, 90–122. [Google Scholar] [CrossRef]
- Cavalu, S.; Bisboaca, S.; Mates, I.M.; Pasca, P.M.; Laslo, V.; Costea, T.; Fritea, L.; Vicas, S. Novel Formulation Based on Chitosan-Arabic Gum Nanoparticles Entrapping Propolis Extract Production, physico-chemical and structural characterization. Rev. Chim. 2018, 69, 3756–3760. [Google Scholar] [CrossRef]
- Erdogar, N.; Akkin, S.; Bilensoy, E. Nanocapsules for Drug Delivery: An Updated Review of the Last Decade. Recent Pat. Drug Deliv. Formul. 2018, 12, 252–266. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T.; Takeuchi, H.; Hino, T.; Kunou, N.; Kawashima, Y. Preparations of biodegradable nanospheres of water-soluble and insoluble drugs with D,L-lactide/glycolide copolymer by a novel spontaneous emulsification solvent diffusion method, and the drug release behavior. J. Control. Release 1993, 25, 89–98. [Google Scholar] [CrossRef]
- Murakami, H.; Kobayashi, M.; Takeuchi, H.; Kawashima, Y. Preparation of poly(dl-lactide-co-glycolide) nanoparticles by modified spontaneous emulsification solvent diffusion method. Int. J. Pharm. 1999, 187, 143–152. [Google Scholar] [CrossRef]
- Quintanar-Guerrero, D.; Allemann, E.; Fessi, H.; Doelker, E. Pseudolatex preparation using a novel emulsion-diffusion process involving direct displacement of partially water-miscible solvents by distillation. Int. J. Pharm. 1999, 188, 155–164. [Google Scholar] [CrossRef]
- Yusuf, M.; Khan, M.; Khan, R.A.; Ahmed, B. Preparation, characterization, in vivo and biochemical evaluation of brain targeted Piperine solid lipid nanoparticles in an experimentally induced Alzheimer’s disease model. J. Drug Target. 2013, 21, 300–311. [Google Scholar] [CrossRef] [PubMed]
- McCall, R.L.; Sirianni, R.W. PLGA nanoparticles formed by single- or double-emulsion with vitamin E-TPGS. J. Vis. Exp. 2013, 82, 51015. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, C. Tuning the Size of Poly(lactic-co-glycolic Acid) (PLGA) Nanoparticles Fabricated by Nanoprecipitation. Biotechnol. J. 2018, 13, 1700203. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Prokisch, J.; Laslo, V.; Vicas, S. Preparation, structural characterisation and release study of novel hybrid microspheres entrapping nanoselenium, produced by green synthesis. IET Nanobiotechnol. 2017, 11, 426–432. [Google Scholar] [CrossRef]
- Tarhini, M.; Greige-Gerges, H.; Elaissari, A. Protein-based nanoparticles: From preparation to encapsulation of active molecules. Int. J. Pharm. 2017, 522, 172–197. [Google Scholar] [CrossRef]
- Zhang, D.; Ma, X.L.; Gu, Y.; Huang, H.; Zhang, G.W. Green Synthesis of Metallic Nanoparticles and Their Potential Applications to Treat Cancer. Front. Chem. 2020, 8, 799. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Antoniac, I.V.; Mohan, A.; Bodog, F.; Doicin, C.; Mates, I.; Ulmeanu, M.; Murzac, R.; Semenescu, A. Nanoparticles and Nanostructured Surface Fabrication for Innovative Cranial and Maxillofacial Surgery. Materials 2020, 13, 5391. [Google Scholar] [CrossRef]
- Velmurugan, B.K.; Rathinasamy, B.; Lohanathan, B.P.; Thiyagarajan, V.; Weng, C.F. Neuroprotective Role of Phytochemicals. Molecules 2018, 23, 2485. [Google Scholar] [CrossRef]
- Xu, J.; Wold, E.A.; Ding, Y.; Shen, Q.; Zhou, J. Therapeutic Potential of Oridonin and Its Analogs: From Anticancer and Antiinflammation to Neuroprotection. Molecules 2018, 23, 474. [Google Scholar] [CrossRef]
- Hajialyani, M.; Hosein Farzaei, M.; Echeverria, J.; Nabavi, S.M.; Uriarte, E.; Sobarzo-Sanchez, E. Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules 2019, 24, 648. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, N.; Ahmad, R.; Khang, G. Inorganic Nanotheranostics: Strategy development and applications. Drug Deliv. Nanosyst. Biomed. Appl. 2018, 377–419. [Google Scholar] [CrossRef]
- Bar-Zeev, M.; Livney, Y.D.; Assaraf, Y.G. Targeted nanomedicine for cancer therapeutics: Towards precision medicine overcoming drug resistance. Drug Resist. Updat. 2017, 31, 15–30. [Google Scholar] [CrossRef]
- Lausanne, E.P.F.D. On the Way to Nanotheranostics: Diagnosing and Treating Diseases Simultaneously. Available online: www.sciencedaily.com/releases/2019/07/190708131203.htm (accessed on 15 May 2021).
- Bandelow, B.; Michaelis, S.; Wedekind, D. Treatment of anxiety disorders. Dialogues. Clin. Neurosci. 2017, 19, 93–107. [Google Scholar]
- Powell, C.R.; Dillon, K.M.; Matson, J.B. A review of hydrogen sulfide (H2S) donors: Chemistry and potential therapeutic applications. Biochem. Pharmacol. 2018, 149, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Shang, A.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Tang, G.Y.; Corke, H.; Mavumengwana, V.; Li, H.B. Bioactive Compounds and Biological Functions of Garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef]
- Chen, X.; Pan, W. The treatment strategies for neurodegenerative diseases by integrative medicine. Integr. Med. Int. 2014, 1, 223–225. [Google Scholar] [CrossRef]
- Roy, S.; Awasthi, H. Herbal medicines as neuroprotective agent: A mechanistic approach. Int. J. Pharm. Pharm. Sci. 2017, 9, 1–7. [Google Scholar] [CrossRef]
- Chopra, H.; Dey, P.S.; Das, D.; Bhattacharya, T.; Shah, M.; Mubin, S.; Maishu, S.P.; Akter, R.; Rahman, M.H.; Karthika, C.; et al. Curcumin Nanoparticles as Promising Therapeutic Agents for Drug Targets. Molecules 2021, 26, 4998. [Google Scholar] [CrossRef] [PubMed]
- Soares, G.A.B.e.; Bhattacharya, T.; Chakrabarti, T.; Tagde, P.; Cavalu, S. Exploring Pharmacological Mechanisms of Essential Oils on the Central Nervous System. Plants 2021, 11, 21. [Google Scholar] [CrossRef]
- Jakka, A. A Study on Nootropic Activity of Celastrus Paniculata Willd Whole Plant Methanolic Extract in Rats. Asian J. Pharm. Clin. Res. 2016, 9, 336–341. [Google Scholar]
- Amadi, C.N.; Offor, S.J.; Frazzoli, C.; Orisakwe, O.E. Natural antidotes and management of metal toxicity. Environ. Sci. Pollut. Res. Int. 2019, 26, 18032–18052. [Google Scholar] [CrossRef]
- Ashraf, A.; Zafar, S.; Zahid, K.; Salahuddin Shah, M.; Al-Ghanim, K.A.; Al-Misned, F.; Mahboob, S. Synthesis, characterization, and antibacterial potential of silver nanoparticles synthesized from Coriandrum sativum L. J. Infect. Public Health 2019, 12, 275–281. [Google Scholar] [CrossRef]
- Owoeye, O.; Obazie, F.I.; Atiba, F.A.; Malomo, A.O. Comparative Neuroprotective Effect of Celosia argentea Linn. and Vitamin E on Mercury-induced Oxidative and Histological Parameters of Rat Brain. Niger. J. Physiol. Sci. 2019, 34, 167–175. [Google Scholar]
- Yang, S.; Chang, M.C. Chronic Pain: Structural and Functional Changes in Brain Structures and Associated Negative Affective States. Int. J. Mol. Sci. 2019, 20, 3130. [Google Scholar] [CrossRef]
- Yuan, Q.; Wang, C.W.; Shi, J.; Lin, Z.X. Effects of Ginkgo biloba on dementia: An overview of systematic reviews. J. Ethnopharmacol. 2017, 195, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Abdolmaleki, A.; Akram, M.; Saeed, M.M.; Asadi, A.; Kajkolah, M. Herbal medicine as neuroprotective potential agent in human and animal models: A historical overview. J. Pharm. Care 2020, 8, 75–82. [Google Scholar] [CrossRef]
- Bridi, H.; Meirelles, G.C.; von Poser, G.L. Structural diversity and biological activities of phloroglucinol derivatives from Hypericum species. Phytochemistry 2018, 155, 203–232. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Kettle, C.; Morton, D.W. A molecular approach in drug development for Alzheimer’s disease. Biomed. Pharm. 2018, 106, 553–565. [Google Scholar] [CrossRef]
- Eudes, J.F.; Silveria, D.; Soares, A.C.; Carneiro, F.P.; de Assis, M.S.; Leite, F.B.; Niraldo, P.; Greice, M.S.; Mônica, V.d.S.; Stéphanie, M.; et al. Effects of lemon balm (Melissa officinalis) on behavioral deficits and memory impairment of rats surviving sepsis. J. Med. Plants Res. 2017, 11, 153–160. [Google Scholar] [CrossRef]
- Kusindarta, D.L.; Wihadmadyatami, H.; Jadi, A.R.; Karnati, S.; Lochnit, G.; Hening, P.; Haryanto, A.; Auriva, M.B.; Purwaningrum, M. Ethanolic extract Ocimum sanctum. Enhances cognitive ability from young adulthood to middle aged mediated by increasing choline acetyl transferase activity in rat model. Res. Vet. Sci. 2018, 118, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Antonescu Mintas, A.I.; Miere Groza, F.; Fritea, L.; Ganea, M.; Zdrinca, M.; Dobjanschi, L.; Antonescu, A.; Vicas, S.I.; Bodog, F.; Sindhu, R.K.; et al. Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants 2021, 10, 1390. [Google Scholar] [CrossRef]
- Ahuja, A.; Kim, J.H.; Kim, J.H.; Yi, Y.S.; Cho, J.Y. Functional role of ginseng-derived compounds in cancer. J. Ginseng Res. 2018, 42, 248–254. [Google Scholar] [CrossRef]
- Nieto, G.; Ros, G.; Castillo, J. Antioxidant and Antimicrobial Properties of Rosemary (Rosmarinus officinalis, L.): A Review. Medicines 2018, 5, 98. [Google Scholar] [CrossRef]
- Lopresti, A.L. Salvia (Sage): A Review of its Potential Cognitive-Enhancing and Protective Effects. Drugs R D 2017, 17, 53–64. [Google Scholar] [CrossRef]
- Lee, D.Y.; Kim, H.W.; Yang, H.; Sung, S.H. Hydrolyzable tannins from the fruits of Terminalia chebula Retz and their alpha-glucosidase inhibitory activities. Phytochemistry 2017, 137, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.; Nayak, P.; Prusty, S.K.; Sahu, P.K. Phytochemistry and pharmacology of Tinospora cordifolia: A review. Syst. Rev. Pharm. 2018, 9, 70–78. [Google Scholar] [CrossRef]
- RajaSankar, S.; Manivasagam, T.; Sankar, V.; Prakash, S.; Muthusamy, R.; Krishnamurti, A.; Surendran, S. Withania somnifera root extract improves catecholamines and physiological abnormalities seen in a Parkinson’s disease model mouse. J. Ethnopharmacol. 2009, 125, 369–373. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Olaniran, A.O.; Okoh, A.I. Therapeutic Potentials of Microalgae in the Treatment of Alzheimer’s Disease. Molecules 2017, 22, 480. [Google Scholar] [CrossRef]
- Stone, N.L.; Murphy, A.J.; England, T.J.; O’Sullivan, S.E. A systematic review of minor phytocannabinoids with promising neuroprotective potential. Br. J. Pharmacol. 2020, 177, 4330–4352. [Google Scholar] [CrossRef]
- Wyant, K.J.; Ridder, A.J.; Dayalu, P. Huntington’s Disease-Update on Treatments. Curr. Neurol. Neurosci. Rep. 2017, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Hussain, G.; Rasul, A.; Anwar, H.; Aziz, N.; Razzaq, A.; Wei, W.; Ali, M.; Li, J.; Li, X. Role of Plant Derived Alkaloids and Their Mechanism in Neurodegenerative Disorders. Int. J. Biol. Sci. 2018, 14, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chaudhary, R.K.; Singh, R.; Singh, S.P.; Wang, S.Y.; Hoe, Z.Y.; Pan, C.T.; Shiue, Y.L.; Wei, D.Q.; Kaushik, A.C.; et al. Nanotheranostic Applications for Detection and Targeting Neurodegenerative Diseases. Front. Neurosci. 2020, 14, 305. [Google Scholar] [CrossRef]
- Khalil, A.T.; Ovais, M.; Ullah, I.; Ali, M.; Shinwari, Z.K.; Khamlich, S.; Maaza, M. Sageretia thea (Osbeck.) mediated synthesis of zinc oxide nanoparticles and its biological applications. Nanomedicine 2017, 12, 1767–1789. [Google Scholar] [CrossRef] [PubMed]
- Ovais, M.; Zia, N.; Ahmad, I.; Khalil, A.T.; Raza, A.; Ayaz, M.; Sadiq, A.; Ullah, F.; Shinwari, Z.K. Phyto-Therapeutic and Nanomedicinal Approaches to Cure Alzheimer’s Disease: Present Status and Future Opportunities. Front. Aging Neurosci 2018, 10, 284. [Google Scholar] [CrossRef]
- Ovais, M.; Nadhman, A.; Khalil, A.T.; Raza, A.; Khuda, F.; Sohail, M.F.; Islam, N.U.; Sarwar, H.S.; Shahnaz, G.; Ahmad, I.; et al. Biosynthesized colloidal silver and gold nanoparticles as emerging leishmanicidal agents: An insight. Nanomedicine 2017, 12, 2807–2819. [Google Scholar] [CrossRef] [PubMed]
- Gaude, T.T.; Soares, G.A.B.e.; Priolkar, R.N.S.; Biradar, B.; Mamledesai, S. Synthesis of 4-hydroxy-1-(phenyl/methyl)-3-[3-(substituted amino)-2-nitropropanoyl] quinolin-2(1H)-ones as an antimicrobial andantitubercular agents. Indian J. Heterocycl. Chem. 2017, 27, 223–228. [Google Scholar]
- Emmanuel, R.; Saravanan, M.; Ovais, M.; Padmavathy, S.; Shinwari, Z.K.; Prakash, P. Antimicrobial efficacy of drug blended biosynthesized colloidal gold nanoparticles from Justicia glauca against oral pathogens: A nanoantibiotic approach. Microb. Pathog. 2017, 113, 295–302. [Google Scholar] [CrossRef]
- Suganthy, N.; Sri Ramkumar, V.; Pugazhendhi, A.; Benelli, G.; Archunan, G. Biogenic synthesis of gold nanoparticles from Terminalia arjuna bark extract: Assessment of safety aspects and neuroprotective potential via antioxidant, anticholinesterase, and antiamyloidogenic effects. Environ. Sci. Pollut. Res. Int. 2018, 25, 10418–10433. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.H.; Manczak, M.; Yin, X.; Grady, M.C.; Mitchell, A.; Tonk, S.; Kuruva, C.S.; Bhatti, J.S.; Kandimalla, R.; Vijayan, M.; et al. Protective Effects of Indian Spice Curcumin against Amyloid-beta in Alzheimer’s Disease. J. Alzheimers. Dis. 2018, 61, 843–866. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, M.; Khan, M.; Alrobaian, M.M.; Alghamdi, S.A.; Warsi, M.H.; Sultana, S.; Khan, R.A. Brain targeted Polysorbate-80 coated PLGA thymoquinone nanoparticles for the treatment of Alzheimer’s disease, with biomechanistic insights. J. Drug Deliv. Sci. Technol. 2021, 61, 102214. [Google Scholar] [CrossRef]
- Xu, R.; Wang, J.; Xu, J.; Song, X.; Huang, H.; Feng, Y.; Fu, C. Rhynchophylline Loaded-mPEG-PLGA Nanoparticles Coated with Tween-80 for Preliminary Study in Alzheimer’s Disease. Int. J. Nanomed. 2020, 15, 1149–1160. [Google Scholar] [CrossRef]
- Jacob, J.; Haponiuk, J.T.; Thomas, S.; Gopi, S. Biopolymer based nanomaterials in drug delivery systems: A Review. Mater. Today Chem. 2018, 9, 43–55. [Google Scholar] [CrossRef]
- Numata, K.; Kaplan, D.L. Silk-based delivery systems of bioactive molecules. Adv. Drug Deliv. Rev. 2010, 62, 1497–1508. [Google Scholar] [CrossRef]
- Chopra, H.; Kumar, S.; Singh, I. Biopolymer-based Scaffolds for Tissue Engineering Applications. Curr. Drug Targets 2021, 22, 282–295. [Google Scholar] [CrossRef]
- Langer, K.; Balthasar, S.; Vogel, V.; Dinauer, N.; Briesen, H.v.; Schubert, D. Optimization of the preparation process for human serum albumin (HSA) nanoparticles. Int. J. Pharm. 2003, 257, 169–180. [Google Scholar] [CrossRef]
- Jahanban-Esfahlan, A.; Dastmalchi, S.; Davaran, S. A simple improved desolvation method for the rapid preparation of albumin nanoparticles. Int. J. Biol. Macromol. 2016, 91, 703–709. [Google Scholar] [CrossRef]
- Kratz, F. Albumin as a drug carrier: Design of prodrugs, drug conjugates and nanoparticles. J. Control. Release 2008, 132, 171–183. [Google Scholar] [CrossRef]
- Sathya, S.; Shanmuganathan, B.; Devi, K.P. Deciphering the anti-apoptotic potential of alpha-bisabolol loaded solid lipid nanoparticles against Abeta induced neurotoxicity in Neuro-2a cells. Colloids Surf. B Biointerfaces 2020, 190, 110948. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. Quercetin lipid nanoparticles functionalized with transferrin for Alzheimer’s disease. Eur. J. Pharm. Sci. 2020, 148, 105314. [Google Scholar] [CrossRef]
- Giacomeli, R.; Izoton, J.C.; Dos Santos, R.B.; Boeira, S.P.; Jesse, C.R.; Haas, S.E. Neuroprotective effects of curcumin lipid-core nanocapsules in a model Alzheimer’s disease induced by beta-amyloid 1-42 peptide in aged female mice. Brain Res. 2019, 1721, 146325. [Google Scholar] [CrossRef]
- Mathew, A.; Fukuda, T.; Nagaoka, Y.; Hasumura, T.; Morimoto, H.; Yoshida, Y.; Maekawa, T.; Venugopal, K.; Kumar, D.S. Curcumin loaded-PLGA nanoparticles conjugated with Tet-1 peptide for potential use in Alzheimer’s disease. PLoS ONE 2012, 7, e32616. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. RVG29-Functionalized Lipid Nanoparticles for Quercetin Brain Delivery and Alzheimer’s Disease. Pharm. Res. 2020, 37, 139. [Google Scholar] [CrossRef]
- Cano, A.; Ettcheto, M.; Chang, J.H.; Barroso, E.; Espina, M.; Kuhne, B.A.; Barenys, M.; Auladell, C.; Folch, J.; Souto, E.B.; et al. Dual-drug loaded nanoparticles of Epigallocatechin-3-gallate (EGCG)/Ascorbic acid enhance therapeutic efficacy of EGCG in a APPswe/PS1dE9 Alzheimer’s disease mice model. J. Control. Release 2019, 301, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xiong, S.; Liu, P.; Liu, W.; Wang, Q.; Liu, Y.; Tan, H.; Chen, X.; Shi, X.; Wang, Q.; et al. Polymeric Nanoparticles-Based Brain Delivery with Improved Therapeutic Efficacy of Ginkgolide B in Parkinson’s Disease. Int. J. Nanomed. 2020, 15, 10453–10467. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Wang, N.; Zeng, Z.; Huang, J.; Xiang, Z.; Guan, Y.-Q. Neuroprotective effect of chitosan nanoparticle gene delivery system grafted with acteoside (ACT) in Parkinson’s disease models. J. Mater. Sci. Technol. 2020, 43, 197–207. [Google Scholar] [CrossRef]
- Tan, J.P.K.; Voo, Z.X.; Lim, S.; Venkataraman, S.; Ng, K.M.; Gao, S.; Hedrick, J.L.; Yang, Y.Y. Effective encapsulation of apomorphine into biodegradable polymeric nanoparticles through a reversible chemical bond for delivery across the blood-brain barrier. Nanomedicine 2019, 17, 236–245. [Google Scholar] [CrossRef]
- Chopra, H.; Kumar, S.; Singh, I. Strategies and Therapies for Wound Healing: A Review. Curr. Drug Targets 2021, 23, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Han, Y.; Li, M.; Nie, C. Curcumin Promotes Proliferation of Adult Neural Stem Cells and the Birth of Neurons in Alzheimer’s Disease Mice via Notch Signaling Pathway. Cell. Reprogram 2019, 21, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Singh-Moon, R.P.; Ellis, J.A.; Chaudhuri, D.B.; Wang, M.; Reif, R.; Bruce, J.N.; Bigio, I.J.; Straubinger, R.M. Cerebral hypoperfusion-assisted intra-arterial deposition of liposomes in normal and glioma-bearing rats. Neurosurgery 2015, 76, 92–100. [Google Scholar] [CrossRef]
- Noble, G.T.; Stefanick, J.F.; Ashley, J.D.; Kiziltepe, T.; Bilgicer, B. Ligand-targeted liposome design: Challenges and fundamental considerations. Trends Biotechnol. 2014, 32, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Sancini, G.; Gregori, M.; Salvati, E.; Cambianica, I.; Re, F.; Ornaghi, F.; Canovi, M.; Fracasso, C.; Cagnotto, A.; Colombo, M.; et al. Functionalization with TAT-peptide enhances blood-brain barrier crossing in vitro of nanoliposomes carrying a curcumin-derivative to bind amyloid-β peptide. J. Nanomed. Nanotechnol. 2013, 4, 171. [Google Scholar] [CrossRef]
- Mourtas, S.; Lazar, A.N.; Markoutsa, E.; Duyckaerts, C.; Antimisiaris, S.G. Multifunctional nanoliposomes with curcumin-lipid derivative and brain targeting functionality with potential applications for Alzheimer disease. Eur. J. Med. Chem. 2014, 80, 175–183. [Google Scholar] [CrossRef]
- Naik, S.R.; Pilgaonkar, V.W.; Panda, V.S. Evaluation of antioxidant activity of Ginkgo biloba phytosomes in rat brain. Phytother. Res. 2006, 20, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Uchegbu, I.F.; Vyas, S.P. Non-ionic surfactant based vesicles (niosomes) in drug delivery. Int. J. Pharm. 1998, 172, 33–70. [Google Scholar] [CrossRef]
- Jin, Y.; Wen, J.; Garg, S.; Liu, D.; Zhou, Y.; Teng, L.; Zhang, W. Development of a novel niosomal system for oral delivery of Ginkgo biloba extract. Int J Nanomed. 2013, 8, 421–430. [Google Scholar] [CrossRef]
- Poka, L.P.; Mohan, G.K.; Rao, K.V.; Shanker, K. Neuroprotective Effect of Green Synthesized Iron Oxide Nanoparticles Using Aqueous Extract of Convolvulus Pluricaulis Plant in The Management of Alzheimer’s Disease. Phytopathology 2017, 9, 703–709. [Google Scholar] [CrossRef][Green Version]
- Dehvari, M.; Ghahghaei, A. The effect of green synthesis silver nanoparticles (AgNPs) from Pulicaria undulata on the amyloid formation in alpha-lactalbumin and the chaperon action of alpha-casein. Int. J. Biol. Macromol. 2018, 108, 1128–1139. [Google Scholar] [CrossRef]
- Nagamune, T. Biomolecular engineering for nanobio/bionanotechnology. Nano Converg. 2017, 4, 9. [Google Scholar] [CrossRef]
- Kaushik, A. Biomedical Nanotechnology Related Grand Challenges and Perspectives. Front. Nanotechnol. 2019, 1. [Google Scholar] [CrossRef]
- Maojo, V.; Fritts, M.; Martin-Sanchez, F.; De la Iglesia, D.; Cachau, R.E.; Garcia-Remesal, M.; Crespo, J.; Mitchell, J.A.; Anguita, A.; Baker, N.; et al. Nanoinformatics: Developing new computing applications for nanomedicine. Comput. Sci. Eng. 2012, 94, 521–539. [Google Scholar] [CrossRef] [PubMed]
- Heath, J.R. Nanotechnologies for biomedical science and translational medicine. Proc. Natl. Acad. Sci. USA 2015, 112, 14436–14443. [Google Scholar] [CrossRef]
- Zlokovic, B.V. Current treatments and therapeutic targets in Alzheimer’s disease. Adv. Drug Deliv. Rev. 2002, 54, 1533–1537. [Google Scholar] [CrossRef]
- Loera-Valencia, R.; Cedazo-Minguez, A.; Kenigsberg, P.A.; Page, G.; Duarte, A.I.; Giusti, P.; Zusso, M.; Robert, P.; Frisoni, G.B.; Cattaneo, A.; et al. Current and emerging avenues for Alzheimer’s disease drug targets. J. Intern. Med. 2019, 286, 398–437. [Google Scholar] [CrossRef]
- Cummings, J.; Lee, G.; Ritter, A.; Sabbagh, M.; Zhong, K. Alzheimer’s disease drug development pipeline: 2020. Alzheimers Dement. 2020, 6, e12050. [Google Scholar] [CrossRef]
- Huang, L.K.; Chao, S.P.; Hu, C.J. Clinical trials of new drugs for Alzheimer disease. J. Biomed. Sci. 2020, 27, 18. [Google Scholar] [CrossRef]
- Fu, L.M.; Li, J.T. A systematic review of single chinese herbs for Alzheimer’s disease treatment. Evid. Based Complement. Altern. Med. 2011, 2011, 640284. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Greig, N.H. Small molecules as central nervous system therapeutics: Old challenges, new directions, and a philosophic divide. Future Med. Chem. 2019, 11, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Grabrucker, A.M.; Ruozi, B.; Belletti, D.; Pederzoli, F.; Forni, F.; Vandelli, M.A.; Tosi, G. Nanoparticle transport across the blood brain barrier. Tissue Barriers 2016, 4, e1153568. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, J.J.; Cunnane, E.M.; Ross, A.M.; Duskey, J.T.; Tosi, G.; Grabrucker, A.M. Drug delivery across the blood-brain barrier: Recent advances in the use of nanocarriers. Nanomedicine 2020, 15, 205–214. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Ashish, N.; Bhatt, P.; Toga, A.W. Global Data Sharing in Alzheimer Disease Research. Alzheimer. Dis. Assoc. Disord. 2016, 30, 160–168. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Yang, H.; Wu, Z.; Wang, T.; Li, W.; Tang, Y.; Liu, G. In Silico Prediction of Blood-Brain Barrier Permeability of Compounds by Machine Learning and Resampling Methods. ChemMedChem 2018, 13, 2189–2201. [Google Scholar] [CrossRef]
- Belmadani, M.; Jacobson, M.; Holmes, N.; Phan, M.; Nguyen, T.; Pavlidis, P.; Rogic, S. VariCarta: A Comprehensive Database of Harmonized Genomic Variants Found in Autism Spectrum Disorder Sequencing Studies. Autism. Res. 2019, 12, 1728–1736. [Google Scholar] [CrossRef]
- Shaker, B.; Yu, M.S.; Song, J.S.; Ahn, S.; Ryu, J.Y.; Oh, K.S.; Na, D. LightBBB: Computational prediction model of blood-brain-barrier penetration based on LightGBM. Bioinformatics 2021, 37, 1135–1139. [Google Scholar] [CrossRef]
- Wang, C.N.; Wang, Y.J.; Wang, H.; Song, L.; Chen, Y.; Wang, J.L.; Ye, Y.; Jiang, B. The Anti-dementia Effects of Donepezil Involve miR-206-3p in the Hippocampus and Cortex. Biol. Pharm. Bull. 2017, 40, 465–472. [Google Scholar] [CrossRef]
- Huang, W.; Li, Z.; Zhao, L.; Zhao, W. Simvastatin ameliorate memory deficits and inflammation in clinical and mouse model of Alzheimer’s disease via modulating the expression of miR-106b. Biomed. Pharmacother. 2017, 92, 46–57. [Google Scholar] [CrossRef]
- Li, S.H.; Gao, P.; Wang, L.T.; Yan, Y.H.; Xia, Y.; Song, J.; Li, H.Y.; Yang, J.X. Osthole Stimulated Neural Stem Cells Differentiation into Neurons in an Alzheimer’s Disease Cell Model via Upregulation of MicroRNA-9 and Rescued the Functional Impairment of Hippocampal Neurons in APP/PS1 Transgenic Mice. Front. Neurosci. 2017, 11, 340. [Google Scholar] [CrossRef]
- Jang, M.; Choi, S.H.; Choi, J.H.; Oh, J.; Lee, R.M.; Lee, N.E.; Cho, Y.J.; Rhim, H.; Kim, H.C.; Cho, I.H.; et al. Ginseng gintonin attenuates the disruptions of brain microvascular permeability and microvascular endothelium junctional proteins in an APPswe/PSEN-1 double-transgenic mouse model of Alphalzheimer’s disease. Exp. Ther. Med. 2021, 21, 310. [Google Scholar] [CrossRef]
- Shi, R.; Zhang, S.; Cheng, G.; Yang, X.; Zhao, N.; Chen, C. Ginsenoside Rg1 and Acori Graminei Rhizoma Attenuates Neuron Cell Apoptosis by Promoting the Expression of miR-873-5p in Alzheimer’s Disease. Neurochem. Res. 2018, 43, 1529–1538. [Google Scholar] [CrossRef]
- Caggiu, E.; Paulus, K.; Mameli, G.; Arru, G.; Sechi, G.P.; Sechi, L.A. Differential expression of miRNA 155 and miRNA 146a in Parkinson’s disease patients. Eneurologicalsci 2018, 13, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Serafin, A.; Foco, L.; Zanigni, S.; Blankenburg, H.; Picard, A.; Zanon, A.; Giannini, G.; Pichler, I.; Facheris, M.F.; Cortelli, P.; et al. Overexpression of blood microRNAs 103a, 30b, and 29a in L-dopa-treated patients with PD. Neurology 2015, 84, 645–653. [Google Scholar] [CrossRef]
- Schwienbacher, C.; Foco, L.; Picard, A.; Corradi, E.; Serafin, A.; Panzer, J.; Zanigni, S.; Blankenburg, H.; Facheris, M.F.; Giannini, G.; et al. Plasma and White Blood Cells Show Different miRNA Expression Profiles in Parkinson’s Disease. J. Mol. Neurosci. 2017, 62, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Alieva, A.; Filatova, E.V.; Karabanov, A.V.; Illarioshkin, S.N.; Limborska, S.A.; Shadrina, M.I.; Slominsky, P.A. miRNA expression is highly sensitive to a drug therapy in Parkinson’s disease. Parkinsonism. Relat. Disord. 2015, 21, 72–74. [Google Scholar] [CrossRef]
- Waschbisch, A.; Atiya, M.; Linker, R.A.; Potapov, S.; Schwab, S.; Derfuss, T. Glatiramer acetate treatment normalizes deregulated microRNA expression in relapsing remitting multiple sclerosis. PLoS ONE 2011, 6, e24604. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Deshpande, M.; Suhail, H.; Rattan, R.; Giri, S. Targeted Stage-Specific Inflammatory microRNA Profiling in Urine During Disease Progression in Experimental Autoimmune Encephalomyelitis: Markers of Disease Progression and Drug Response. J. Neuroimmune Pharmacol. 2016, 11, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Piket, E.; Khademi, M.; James, T.; Brundin, L.; Olsson, T.; Piehl, F.; Jagodic, M. Circulating miR-150 in CSF is a novel candidate biomarker for multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e219. [Google Scholar] [CrossRef] [PubMed]
- Fattahi, M.; Rezaei, N.; Nematalahi, F.S.; Shaygannejad, V.; Fouladi, S.; Karimi, L.; Fathi, F.; Dehghani, L.; Mirmosayyeb, O.; Eskandari, N. MicroRNA-29b variants and MxA expression change during interferon beta therapy in patients with relapsing-remitting multiple sclerosis. Mult. Scler. Relat. Disord. 2019, 35, 241–245. [Google Scholar] [CrossRef]
- Meira, M.; Sievers, C.; Hoffmann, F.; Derfuss, T.; Kuhle, J.; Kappos, L.; Lindberg, R.L. MiR-126: A novel route for natalizumab action? Mult. Scler. 2014, 20, 1363–1370. [Google Scholar] [CrossRef] [PubMed]
- Sievers, C.; Meira, M.; Hoffmann, F.; Fontoura, P.; Kappos, L.; Lindberg, R.L. Altered microRNA expression in B lymphocytes in multiple sclerosis: Towards a better understanding of treatment effects. Clin. Immunol. 2012, 144, 70–79. [Google Scholar] [CrossRef]
- Ingwersen, J.; Menge, T.; Wingerath, B.; Kaya, D.; Graf, J.; Prozorovski, T.; Keller, A.; Backes, C.; Beier, M.; Scheffler, M.; et al. Natalizumab restores aberrant miRNA expression profile in multiple sclerosis and reveals a critical role for miR-20b. Ann. Clin. Transl. Neurol. 2015, 2, 43–55. [Google Scholar] [CrossRef]
- Michell-Robinson, M.A.; Moore, C.S.; Healy, L.M.; Osso, L.A.; Zorko, N.; Grouza, V.; Touil, H.; Poliquin-Lasnier, L.; Trudelle, A.M.; Giacomini, P.S.; et al. Effects of fumarates on circulating and CNS myeloid cells in multiple sclerosis. Ann. Clin. Transl. Neurol. 2016, 3, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Eftekharian, M.M.; Komaki, A.; Mazdeh, M.; Arsang-Jang, S.; Taheri, M.; Ghafouri-Fard, S. Expression Profile of Selected MicroRNAs in the Peripheral Blood of Multiple Sclerosis Patients: A Multivariate Statistical Analysis with ROC Curve to Find New Biomarkers for Fingolimod. J. Mol. Neurosci. 2019, 68, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Vaknin-Dembinsky, A.; Charbit, H.; Brill, L.; Abramsky, O.; Gur-Wahnon, D.; Ben-Dov, I.Z.; Lavon, I. Circulating microRNAs as biomarkers for rituximab therapy, in neuromyelitis optica (NMO). J. Neuroinflamm. 2016, 13, 179. [Google Scholar] [CrossRef]
- Ebrahimkhani, S.; Beadnall, H.N.; Wang, C.; Suter, C.M.; Barnett, M.H.; Buckland, M.E.; Vafaee, F. Serum Exosome MicroRNAs Predict Multiple Sclerosis Disease Activity after Fingolimod Treatment. Mol. Neurobiol. 2020, 57, 1245–1258. [Google Scholar] [CrossRef]
- Fattahi, M.; Eskandari, N.; Sotoodehnejadnematalahi, F.; Shaygannejad, V.; Kazemi, M. Comparison of The Expression of miR-326 between Interferon beta Responders and Non-Responders in Relapsing-Remitting Multiple Sclerosis. Cell J. 2020, 22, 92–95. [Google Scholar] [CrossRef]
- De Felice, B.; Mondola, P.; Sasso, A.; Orefice, G.; Bresciamorra, V.; Vacca, G.; Biffali, E.; Borra, M.; Pannone, R. Small non-coding RNA signature in multiple sclerosis patients after treatment with interferon-beta. BMC Med. Genom. 2014, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Culla, M.; Irizar, H.; Castillo-Trivino, T.; Saenz-Cuesta, M.; Sepulveda, L.; Lopetegi, I.; Lopez de Munain, A.; Olascoaga, J.; Baranzini, S.E.; Otaegui, D. Blood miRNA expression pattern is a possible risk marker for natalizumab-associated progressive multifocal leukoencephalopathy in multiple sclerosis patients. Mult. Scler. 2014, 20, 1851–1859. [Google Scholar] [CrossRef]
- Fenoglio, C.; De Riz, M.; Pietroboni, A.M.; Calvi, A.; Serpente, M.; Cioffi, S.M.; Arcaro, M.; Oldoni, E.; Scarpini, E.; Galimberti, D. Effect of fingolimod treatment on circulating miR-15b, miR23a and miR-223 levels in patients with multiple sclerosis. J. Neuroimmunol. 2016, 299, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Indrasekara, A.S.D.S.; Wadams, R.C.; Fabris, L. Ligand Exchange on Gold Nanorods: Going Back to the Future. Part. Parcle Syst. Charact. 2014, 31, 819–838. [Google Scholar] [CrossRef]
- de Lau, L.M.; Koudstaal, P.J.; Witteman, J.C.; Hofman, A.; Breteler, M.M. Dietary folate, vitamin B12, and vitamin B6 and the risk of Parkinson disease. Neurology 2006, 67, 315–318. [Google Scholar] [CrossRef]
- Kim, J.W.; Galanzha, E.I.; Zaharoff, D.A.; Griffin, R.J.; Zharov, V.P. Nanotheranostics of circulating tumor cells, infections and other pathological features in vivo. Mol. Pharm. 2013, 10, 813–830. [Google Scholar] [CrossRef]
- Rahman, M.; Akhter, S.; Ahmad, M.Z.; Ahmad, J.; Addo, R.T.; Ahmad, F.J.; Pichon, C. Emerging advances in cancer nanotheranostics with graphene nanocomposites: Opportunities and challenges. Nanomedicine 2015, 10, 2405–2422. [Google Scholar] [CrossRef]
- Li, W.; Luo, R.; Lin, X.; Jadhav, A.D.; Zhang, Z.; Yan, L.; Chan, C.Y.; Chen, X.; He, J.; Chen, C.H.; et al. Remote modulation of neural activities via near-infrared triggered release of biomolecules. Biomaterials 2015, 65, 76–85. [Google Scholar] [CrossRef]
- Xue, Y.; Wu, J.; Sun, J. Four types of inorganic nanoparticles stimulate the inflammatory reaction in brain microglia and damage neurons in vitro. Toxicol. Lett. 2012, 214, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Sunderland, K.S.; Yang, M.; Mao, C. Phage-Enabled Nanomedicine: From Probes to Therapeutics in Precision Medicine. Angew. Chem. Int. Ed. 2017, 56, 1964–1992. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.M.; Ghonaim, G.A.; Gharib, S.M.; Chopra, H.; Farag, A.K.; Hassanin, M.H.; Nagah, A.; Emad-Eldin, M.; Hashem, N.E.; Yahya, G.; et al. Exosomes in Alzheimer’s Disease: From Being Pathological Players to Potential Diagnostics and Therapeutics. Int. J. Mol. Sci. 2021, 22, 794. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.H.L.; Xiang, D.; Tran, T.T.D.; Yin, W.; Zhang, Y.; Kong, L.; Chen, K.; Sun, M.; Li, Y.; Hou, Y.; et al. Exosomes and Nanoengineering: A Match Made for Precision Therapeutics. Adv. Mater. 2020, 32, e1904040. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Ratiu, C.; Ponta, O.; Simon, V.; Rugina, D.; Miclaus, V.; Akin, I.; Goller, G. Improving osseointegration of alumina/zirconia ceramic implants by fluoride surface treatment. Dig. J. Nanomater. Bios. 2014, 9, 797–808. [Google Scholar]
- Kandell, R.M.; Waggoner, L.E.; Kwon, E.J. Nanomedicine for Acute Brain Injuries: Insight from Decades of Cancer Nanomedicine. Mol. Pharm. 2021, 18, 522–538. [Google Scholar] [CrossRef] [PubMed]
| Strategy | Alzheimer’s Disease | Parkinson Disease |
|---|---|---|
| Modulation of neurotransmitters (approved therapies) |
|
|
| Disease modifying therapies (under investigation) |
|
|
| Immunotherapy |
|
|
| Gene-based therapy | Regulation of presenilin expression | Expression of synapsin 3 modulation |
| Other |
|
|
| Material | Name of Active Moiety Entrapped | Size | Outcome of Study | Reference |
|---|---|---|---|---|
| Cholesterol | α-bisabolol | 139.5 nm | NPs drastically decrease free radical generation, lower β-secretase, caspase-3, cholinesterase, and Bax expression, and increase Bcl-2 protein expression. | [104] |
| Cetyl palmitate miglyol-812 | Quercetin | 200 nm | It is non-toxic to hCMEC/D3 cells and penetrates the BBB more so than free drug. NPs also prevent A peptide fibril production. | [105] |
| Caprylic and capric triglycerides, sorbitan monostearate | Curcumin | 247 nm | Curcumin-NPs protect against A42-induced behavioural and neurochemical alterations in AD mice model. | [106] |
| Cholesterol | α-bisabolol | Not reported | Inhibited A aggregation and protected Neuro-2a cells from A-induced neurotoxicity. | [104] |
| PLGA | Curcumin | 150–200 nm | Curcumin encapsulated-PLGA nanoparticles, destroyed amyloid aggregates, exhibited an anti-oxidative property, and are non-cytotoxic. | [107] |
| DSPE-PEG2000-MAL | Quercetin | 200 nm | After 4 h, RVG29-nanoparticles had 1.5 times the permeability across the blood-brain barrier compared to non-functionalized nanoparticles. | [108] |
| PEG, PLGA | Epigallocatechin-3-gallate | 100 nm | GCG and AA NPs resulted in a marked increase in synapses, as judged by synaptophysin (SYP) expression, and reduction of neuroinflammation as well as amyloid β (Aβ) plaque burden and cortical levels of soluble and insoluble Aβ(1–42) peptide. | [109] |
| poly(ethylene glycol)-co-poly(ε-caprolactone) | Ginkgolide B | 91 nm | NPs facilitated the sustained release of GB into the blood, thereby improving its ability to accumulate in the brain and to treat PD. | [110] |
| chitosan poly ethyleneglycol-poly lactic acid | Acteoside | 100 nm | Significantly reversed dopaminergic (DA) neuron loss in the substantia nigra and striatum of sick mice. | [111] |
| Monomethoxy polyethylene glycol | Apomorphine (AMP) | 100 nm | The encapsulation of AMP into the nanoparticles inhibits oxidization. The intranasal administration of the AMP-loaded nanoparticles transports AMP across the BBB. | [112] |
| Drug | miRNA | Source of miRNA | Disease State Used | Reference |
|---|---|---|---|---|
| Donepezil | miRNA-206-3p | Mouse-Hippocampus, cortex | Dementia | [141] |
| Simvastatin | miRNA-106b | SH-SY5Y cells; Mice brain tissue-APP/PS1 | [142] | |
| Osthole | miRNA-9 | overexpressed APP cells | Alzheimer’s Disease (AD) | [143,144] |
| miRNA-107 | Overexpressed APP cells Mice brain tissue-APP/PS1 | [143] | ||
| miRNA-101a-3p | [143] | |||
| AGR-GRg1 | miRNA-873-5p | Mouse Hippocampus | [145] | |
| L-Dopa | miRNA-30b-5p, miRNA-30a-5p | Plasma | PD | [146] |
| miRNA-29a-3p, miRNA-30b-5p, miRNA-103a-3p | Peripheral Blood mononuclear cells (PBMC’s) | [147] | ||
| miRNA-16-2-3p, miRNA-26a-2-3p, miRNA-30a | Peripheral blood | [148] | ||
| miRNA-155 | PBMCs | [149] | ||
| L-Dopa, Amantadine | miRNA-7, miRNA-9-3p, miRNA-9-5p | Peripheral blood | [150] | |
| Interferon-β | miRNA-29 | PBMCs | Multiple Sclerosis | [151] |
| miRNA-145 | Whole blood | [152] | ||
| miRNA-29b-3p | PBMCs | [153] | ||
| miRNA-326 | [154] | |||
| miRNA-26a-5p | [155] | |||
| miRNA-146a | [156] | |||
| Natalizumab | miRNA-150 CSF, | Plasma | [157] | |
| miRNA-126, miRNA-17 | CD4 + T cells | [158,159] | ||
| miRNA-17~92, miRNA-106b~25 | B lymphocytes | [160] | ||
| miRNA-26a, miRNA-155 | PBMCs | [153] | ||
| miRNA-155 | Monocytes | [161] | ||
| Dimethyl fumarate | miRNA-155 | Monocytes | [162] | |
| Fingolimod | miRNA-150 | Plasma | [163] | |
| miRNA-23a | Whole Blood | [164] | ||
| Natalizumab | miRNA-320, miRNA-320b, miRNA-629 | Blood | Progressive multifocal leukoencephalopathy | [163] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhattacharya, T.; Soares, G.A.B.e.; Chopra, H.; Rahman, M.M.; Hasan, Z.; Swain, S.S.; Cavalu, S. Applications of Phyto-Nanotechnology for the Treatment of Neurodegenerative Disorders. Materials 2022, 15, 804. https://doi.org/10.3390/ma15030804
Bhattacharya T, Soares GABe, Chopra H, Rahman MM, Hasan Z, Swain SS, Cavalu S. Applications of Phyto-Nanotechnology for the Treatment of Neurodegenerative Disorders. Materials. 2022; 15(3):804. https://doi.org/10.3390/ma15030804
Chicago/Turabian StyleBhattacharya, Tanima, Giselle Amanda Borges e Soares, Hitesh Chopra, Md. Mominur Rahman, Ziaul Hasan, Shasank S. Swain, and Simona Cavalu. 2022. "Applications of Phyto-Nanotechnology for the Treatment of Neurodegenerative Disorders" Materials 15, no. 3: 804. https://doi.org/10.3390/ma15030804
APA StyleBhattacharya, T., Soares, G. A. B. e., Chopra, H., Rahman, M. M., Hasan, Z., Swain, S. S., & Cavalu, S. (2022). Applications of Phyto-Nanotechnology for the Treatment of Neurodegenerative Disorders. Materials, 15(3), 804. https://doi.org/10.3390/ma15030804










