Microscale Modeling of Frozen Particle Fluid Systems with a Bonded-Particle Model Method
Abstract
1. Introduction
- Flexibility in agglomerate generation, in which all particles and bonds can have their unique material or geometrical properties;
- Capability in mimicking the breakage behavior of agglomerate, such as the crack initiation, propagation, failure plane, etc.;
- Diversity in functional model usage, with numerous choices of rheological models in the particle-particle, particle-wall relationship, and solid bond models.
1.1. Ice Rheology
1.2. Rheology of Frozen Soil
2. Materials and Methods
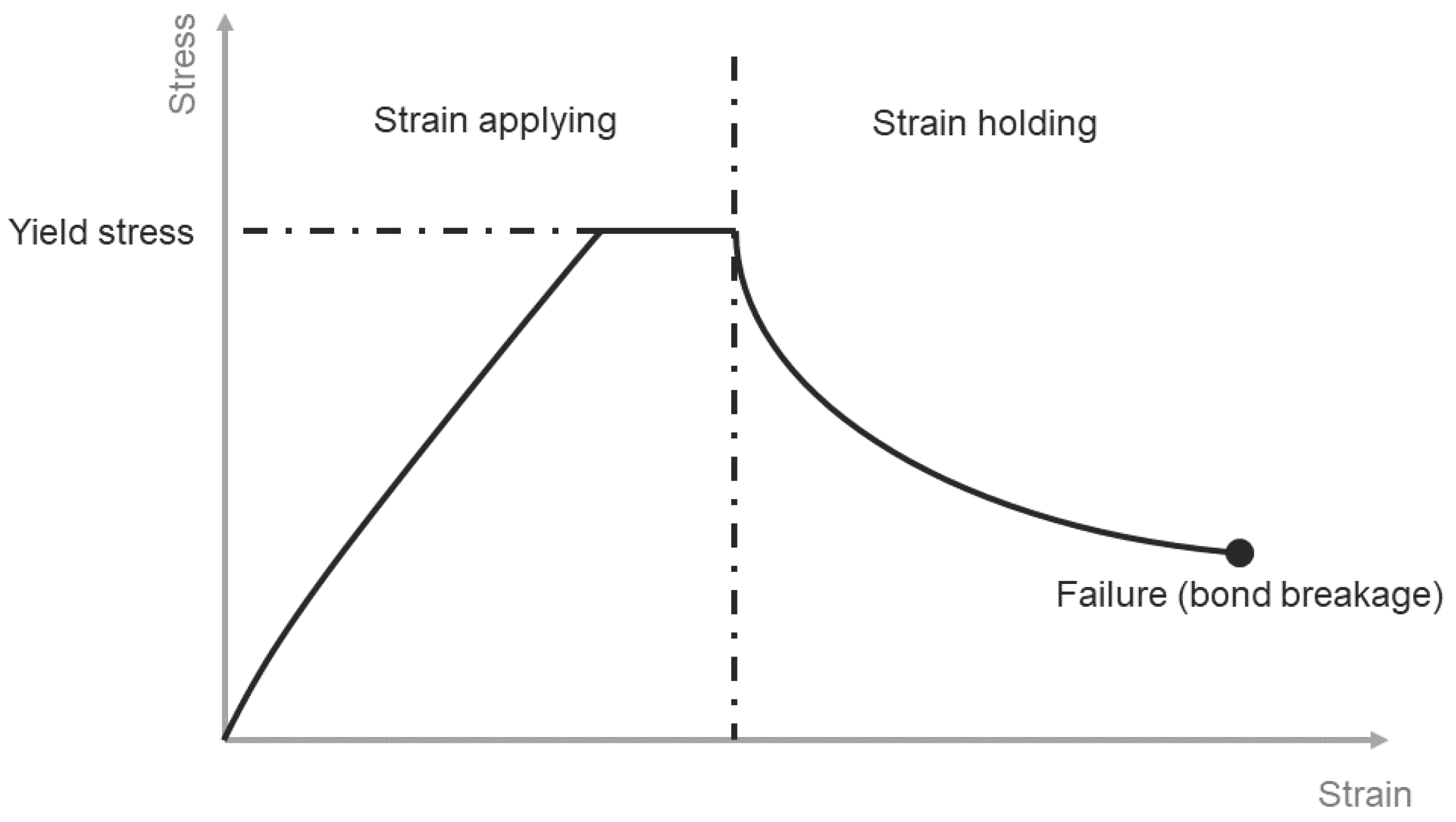
2.1. Uniaxial Compression Test
2.2. Specimen Preparation
2.3. Investigated Parameter Space
2.4. Ice Creep Behavior
2.5. Fracture Patterns of Frozen PFS
2.6. Mechanical Behavior of Frozen PFS
2.7. Bonded-Particle Model Approach
2.8. Solid Bond Model Considering Creep Behavior
3. Result and Discussion
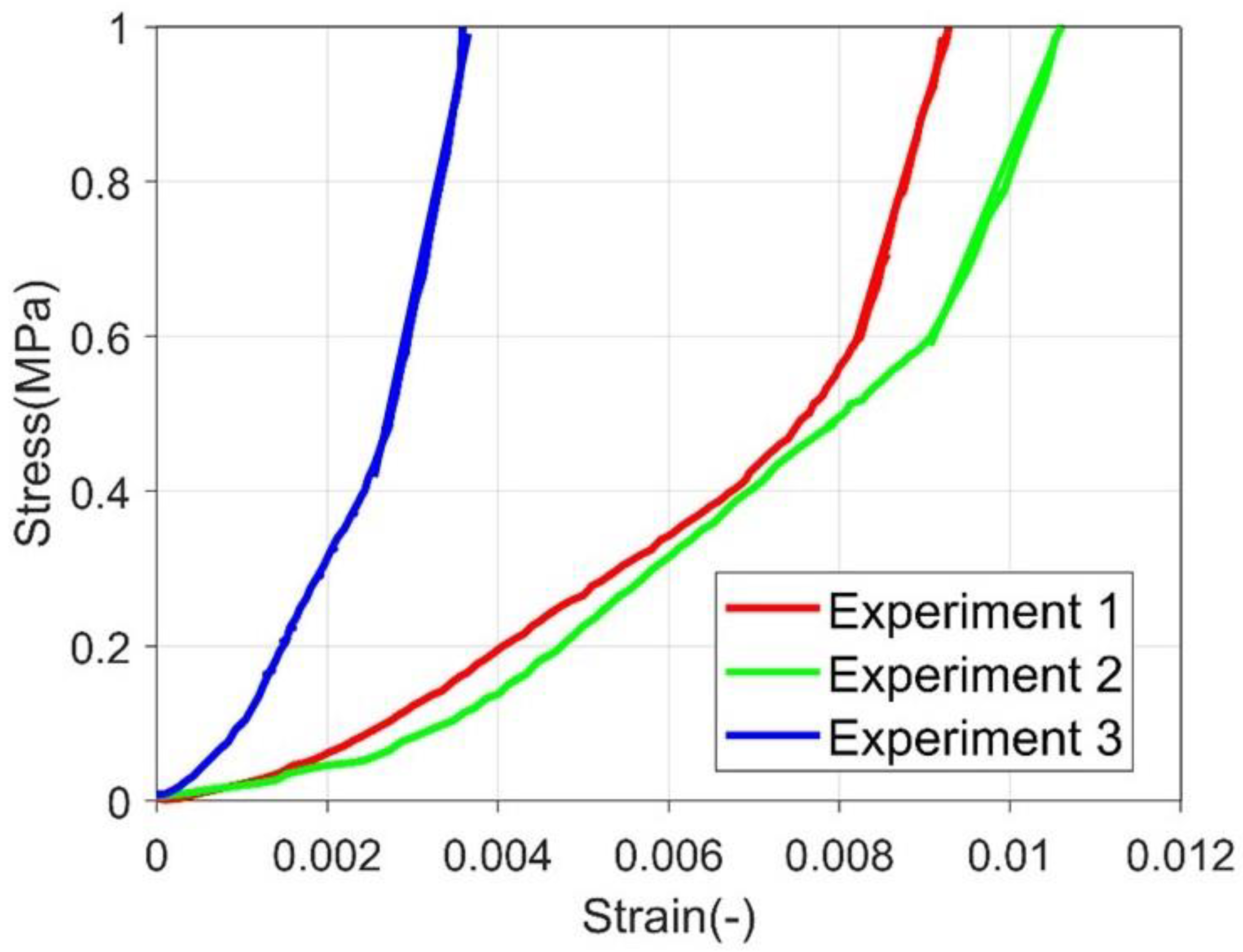
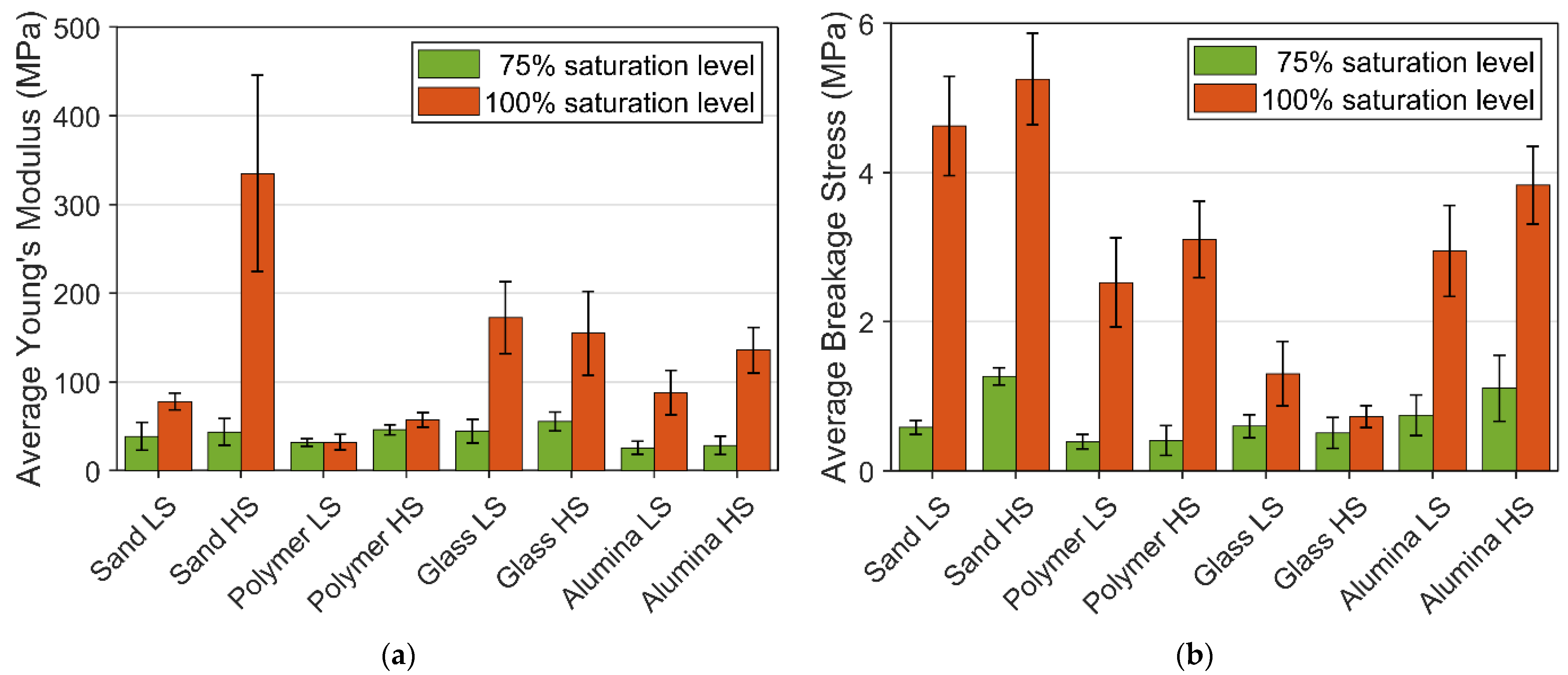
3.1. Experimental Result
3.2. Simulation Setup
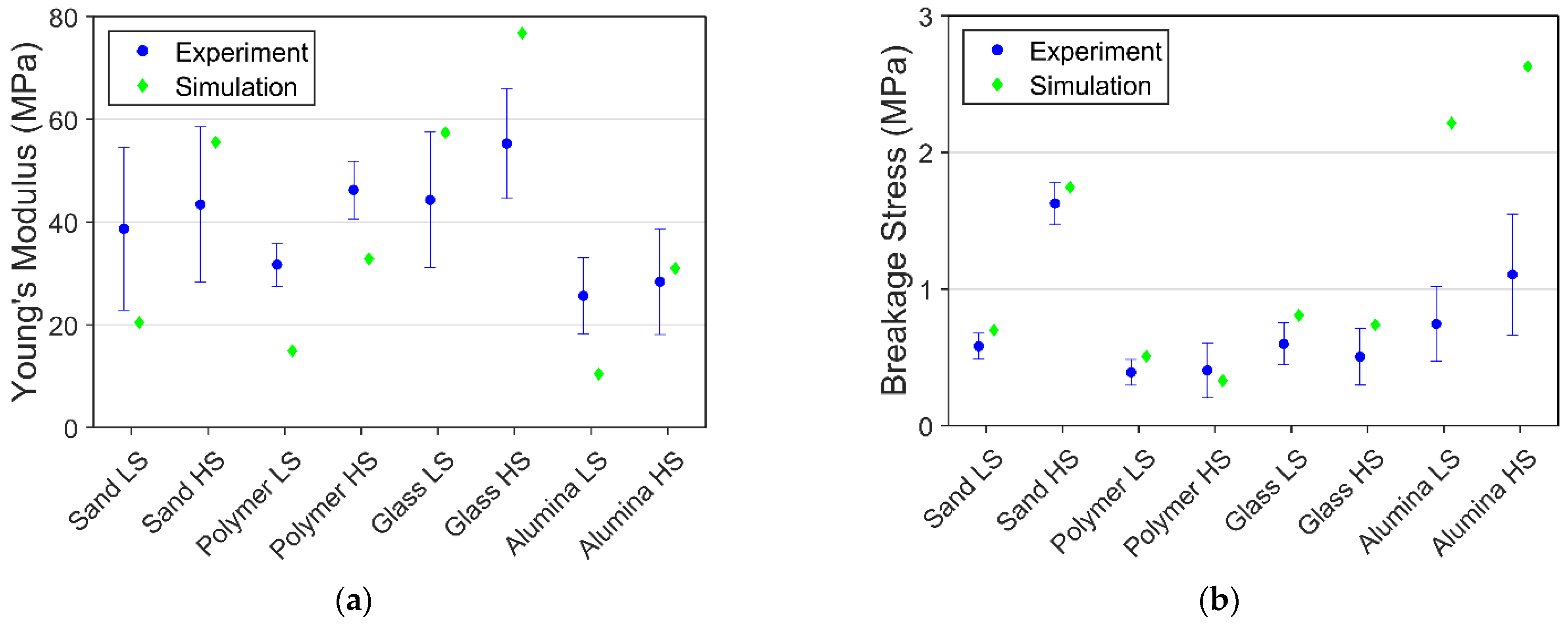
3.3. Comparison of Simulation and Experimental Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arenson, L.U.; Springman, S.M.; Sego, D.C. The Rheology of Frozen Soils. Appl. Rheol. 2007, 17, 12147–1. [Google Scholar] [CrossRef]
- Goodman, D.J.; Frost, H.J.; Ashby, M.F. The plasticity of polycrystalline ice. Philos. Mag. A 1981, 43, 665–695. [Google Scholar] [CrossRef]
- Gold, L.W. The process of failure of columnar-grained ice. Philos. Mag. 1972, 26, 311–328. [Google Scholar] [CrossRef]
- Jellinek, H.H.G.; Brill, R. Viscoelastic Properties of Ice. J. Appl. Phys. 1956, 27, 1198–1209. [Google Scholar] [CrossRef]
- Glen, J.W. The creep of polycrystalline ice. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1955, 228, 519–538. [Google Scholar] [CrossRef]
- Mellor, M.; Testa, R. Effect of Temperature on the Creep of Ice. J. Glaciol. 1969, 8, 131–145. [Google Scholar] [CrossRef]
- Hassan, M.F.; Lee, H.P.; Lim, S.P. The variation of ice adhesion strength with substrate surface roughness. Meas. Sci. Technol. 2010, 21, 075701. [Google Scholar] [CrossRef]
- Nath, S.; Ahmadi, S.F.; Boreyko, J.B. How ice bridges the gap. Soft Matter 2019, 16, 1156–1161. [Google Scholar] [CrossRef]
- Kellner, L.; Stender, M.; Polach, R.U.F.V.B.U.; Herrnring, H.; Ehlers, S.; Hoffmann, N.; Høyland, K.V. Establishing a common database of ice experiments and using machine learning to understand and predict ice behavior. Cold Reg. Sci. Technol. 2019, 162, 56–73. [Google Scholar] [CrossRef]
- Pernas-Sánchez, J.; Pedroche, D.; Varas, D.; López-Puente, J.; Zaera, R. Numerical modeling of ice behavior under high velocity impacts. Int. J. Solids Struct. 2012, 49, 1919–1927. [Google Scholar] [CrossRef]
- Wang, C.; Hu, X.; Tian, T.; Guo, C.; Wang, C. Numerical simulation of ice loads on a ship in broken ice fields using an elastic ice model. Int. J. Nav. Arch. Ocean Eng. 2020, 12, 414–427. [Google Scholar] [CrossRef]
- Long, X.; Liu, S.; Ji, S. Breaking characteristics of ice cover and dynamic ice load on upward–downward conical structure based on DEM simulations. Comput. Part. Mech. 2020, 8, 297–313. [Google Scholar] [CrossRef]
- Yershov, E.D. General Geocryology; Cambridge University Press: Cambridge, UK, 2004. [Google Scholar] [CrossRef]
- Harris, J.S. Ground Freezing in Practice; Thomas Telford Limited: London, UK, 1995. [Google Scholar]
- Wang, Y.; Chen, B.; Nie, C. Numerical Simulation of Nonlinear Fracture Failure Process of Frozen Soil. In Proceedings of the 2009 International Joint Conference on Computational Sciences and Optimization, Sanya, China, 24–26 April 2009; Volume 1, pp. 183–186. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, L.; Wu, L.; Yu, H. Numerical simulation of crack growth in brittle matrix of particle reinforced composites using the xfem technique. Acta Mech. Solida Sin. 2012, 25, 9–21. [Google Scholar] [CrossRef]
- Nishimura, S.; Gens, A.; Olivella, S.; Jardine, R.J. THM-coupled finite element analysis of frozen soil: Formulation and application. Géotechnique 2009, 59, 159–171. [Google Scholar] [CrossRef]
- Cuccurullo, A.; Gallipoli, D. DEM Simulation of Frozen Granular Soils with High Ice Content. National Conference of the Researchers of Geotechnical Engineering; Springer: Cham, Switzerland, 2020. [Google Scholar]
- An, L.; Ling, X.; Geng, Y.; Li, Q.; Zhang, F. DEM Investigation of Particle-Scale Mechanical Properties of Frozen Soil Based on the Nonlinear Microcontact Model Incorporating Rolling Resistance. Math. Probl. Eng. 2018, 2018, 2685709. [Google Scholar] [CrossRef]
- Cundall, P.A.; Strack, O.D.L. A discrete numerical model for granular assemblies. Géotechnique 1979, 29, 47–65. [Google Scholar] [CrossRef]
- Dosta, M.; Dale, S.; Antonyuk, S.; Wassgren, C.; Heinrich, S.; Litster, J.D. Numerical and experimental analysis of influence of granule microstructure on its compression breakage. Powder Technol. 2016, 299, 87–97. [Google Scholar] [CrossRef]
- Rybczyński, S.; Dosta, M.; Schaan, G.; Ritter, M.; Schmidt-Döhl, F. Numerical study on the mechanical behavior of ultrahigh performance concrete using a three-phase discrete element model. Struct. Concr. 2020, 23, 548–563. [Google Scholar] [CrossRef]
- Beckmann, B.; Schicktanz, D.-I.K.; Reischl, D.-M.D.; Curbach, D.-I.E.M. DEM simulation of concrete fracture and crack evolution. Struct. Concr. 2012, 13, 213–220. [Google Scholar] [CrossRef]
- Obermayr, M.; Dressler, K.; Vrettos, C.; Eberhard, P. A bonded-particle model for cemented sand. Comput. Geotech. 2012, 49, 299–313. [Google Scholar] [CrossRef]
- Ouyang, Y.; Yang, Q.; Chen, X. Bonded-Particle Model with Nonlinear Elastic Tensile Stiffness for Rock-Like Materials. Appl. Sci. 2017, 7, 686. [Google Scholar] [CrossRef]
- Dosta, M.; Jarolin, K.; Gurikov, P. Modelling of Mechanical Behavior of Biopolymer Alginate Aerogels Using the Bonded-Particle Model. Molecules 2019, 24, 2543. [Google Scholar] [CrossRef]
- Dosta, M.; Skorych, V. MUSEN: An open-source framework for GPU-accelerated DEM simulations. SoftwareX 2020, 12, 100618. [Google Scholar] [CrossRef]
- Gold, L.W. On the Elasticity of Ice Plates. Can. J. Civ. Eng. 1988, 15, 1080–1084. [Google Scholar] [CrossRef]
- Petrovic, J.J. Review Mechanical properties of ice and snow. J. Mater. Sci. 2003, 38, 1–6. [Google Scholar] [CrossRef]
- Haynes, F.D. Effect of Temperature on the Strength of Snow-Ice; U.S. Army Cold Regions Research and Engineering Laboratory: Hanover, NH, USA, 1978; Volume 78. [Google Scholar]
- Schulson, E.M. The structure and mechanical behavior of ice. JOM 1999, 51, 21–27. [Google Scholar] [CrossRef]
- Weertman, J. CREEP DEFORMATION OF ICE. Annu. Rev. Earth Planet. Sci. 1983, 11, 215–240. [Google Scholar] [CrossRef]
- Naumenko, K.; Altenbach, H. Modeling of Creep for Structural Analysis; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007. [Google Scholar] [CrossRef]
- Gold, L.W. Process of failure in ice. Can. Geotech. J. 1970, 7, 405–413. [Google Scholar] [CrossRef]
- Arenson, L.U.; Johansen, M.M.; Springman, S.M. Effects of volumetric ice content and strain rate on shear strength under triaxial conditions for frozen soil samples. Permafr. Periglac. Process 2004, 15, 261–271. [Google Scholar] [CrossRef]
- Arenson, L.U.; Almasi, N.; Springman, S.M. Shearing response of ice-rich rock glacier material. In Proceedings of the Eighth International Conference on Permafrost, Zurich, Switzerland, 21–25 July 2003; pp. 39–44. [Google Scholar]
- Taylor, D.W. Fundamentals of Soil Mechanics; LWW: Philadelphia, PA, USA, 1948. [Google Scholar]
- Arenson, L.U.; Springman, S.M. Triaxial constant stress and constant strain rate tests on ice-rich permafrost samples. Can. Geotech. J. 2005, 42, 412–430. [Google Scholar] [CrossRef]
- Hooke, R.L.; Dahlin, B.B.; Kauper, M.T. Creep of Ice Containing Dispersed Fine Sand. J. Glaciol. 1972, 11, 327–336. [Google Scholar] [CrossRef][Green Version]
- Ting, J.M.; Martin, R.T.; Ladd, C.C. Mechanisms of Strength for Frozen Sand. J. Geotech. Eng. 1983, 109, 1286–1302. [Google Scholar] [CrossRef]
- Arenson, L.U.; Sego, D.C. The effect of salinity on the freezing of coarse-grained sands. Can. Geotech. J. 2006, 43, 325–337. [Google Scholar] [CrossRef]
- Zhao, S.P.; Zhu, Y.L.; He, P. Recent progress in research on the dynamic response of frozen soil. In Proceedings of the Eighth International Conference on Permafrost, Zurich, Switzerland, 21–25 July 2003; pp. 1301–1306. [Google Scholar]
- Anderson, D.M.; Tice, A.R. Predicting Unfrozen Water Contents in Frozen Soils From Surface Area Measurements. Highw. Res. Rec. 1972, 393, 12–18. [Google Scholar]
- Istomin, V.; Chuvilin, E.; Bukhanov, B. Fast estimation of unfrozen water content in frozen soils. Earth’s Cryosphere 2017, 21, 116–120. [Google Scholar] [CrossRef]
- Mellor, M.; Smith, J.S. Creep of Snow and Ice, Cold Regions Research and Engineering Laboratory, Vicksburg, U.S. Research Report. 1966. Available online: https://hdl.handle.net/11681/5879 (accessed on 20 November 2022).
- Lian, G.; Thornton, C.; Adams, M.J. A Theoretical Study of the Liquid Bridge Forces between Two Rigid Spherical Bodies. J. Colloid Interface Sci. 1993, 161, 138–147. [Google Scholar] [CrossRef]
- Willett, C.D.; Johnson, S.A.; Adams, M.J.; Seville, J.P. Chapter 28 Pendular capillary bridges. Handb. Powder Technol. 2007, 11, 1317–1351. [Google Scholar] [CrossRef]
- Nguyen, H.N.G.; Zhao, C.-F.; Millet, O.; Selvadurai, A. Effects of surface roughness on liquid bridge capillarity and droplet wetting. Powder Technol. 2020, 378, 487–496. [Google Scholar] [CrossRef]
- Mindlin, R.D.; Deresiewicz, H. Elastic Spheres in Contact Under Varying Oblique Forces. J. Appl. Mech. 1953, 20, 327–344. [Google Scholar] [CrossRef]
- Tsuji, Y.; Tanaka, T.; Ishida, T. Lagrangian numerical simulation of plug flow of cohesionless particles in a horizontal pipe. Powder Technol. 1992, 71, 239–250. [Google Scholar] [CrossRef]
- Dosta, M.; Antonyuk, S.; Heinrich, S. Multiscale Simulation of Agglomerate Breakage in Fluidized Beds. Ind. Eng. Chem. Res. 2013, 52, 11275–11281. [Google Scholar] [CrossRef]
- Iwamoto, T.; Murakami, E.; Sawa, T. A finite element simulation on creep behavior in welded joint of chrome-molybdenum steel including interaction between void evolution and dislocation dynamics. Technol. Mech. J. Eng. Mech. 2010, 30, 157–168. [Google Scholar]
- Norton, F.H. The Creep of Steel at High Temperatures; McGraw-Hill B. Company, Incorporated: Columbus, OH, USA, 1929. [Google Scholar]
- Penny, R.K.; Marriott, D.L. Design for Creep; McGraw-Hill: New York, NY, USA; Columbus, OH, USA, 1971. [Google Scholar]
- Dosta, M.; Bistreck, K.; Skorych, V.; Schneider, G.A. Mesh-free micromechanical modeling of inverse opal structures. Int. J. Mech. Sci. 2021, 204, 106577. [Google Scholar] [CrossRef]
- Descantes, Y.; Tricoire, F.; Richard, P. Classical contact detection algorithms for 3D DEM simulations: Drawbacks and solutions. Comput. Geotech. 2019, 114, 103134. [Google Scholar] [CrossRef]
- Leroy, B. Collision between two balls accompanied by deformation: A qualitative approach to Hertz’s theory. Am. J. Phys. 1985, 53, 346–349. [Google Scholar] [CrossRef]
- El Shamy, U.; Zamani, N. Discrete element method simulations of the seismic response of shallow foundations including soil-foundation-structure interaction. Int. J. Numer. Anal. Methods Géoméch. 2011, 36, 1303–1329. [Google Scholar] [CrossRef]
- Wang, X.; Yang, J.; Xiong, W.; Wang, T. Evaluation of DEM and FEM/DEM in Modeling the Fracture Process of Glass Under Hard-Body Impact. Int. Conf. Discret. Elem. Methods 2017, 188, 377–388. [Google Scholar] [CrossRef]
- Nitta, K.-H.; Yamana, M. Poisson’s Ratio and Mechanical Nonlinearity Under Tensile Deformation in Crystalline Polymers; Intec: Rijeka, Croatia, 2012; pp. 113–132. [Google Scholar] [CrossRef]
- Tan, Y.; Yang, D.; Sheng, Y. Study of polycrystalline Al2O3 machining cracks using discrete element method. Int. J. Mach. Tools Manuf. 2008, 48, 975–982. [Google Scholar] [CrossRef]
- Lupo, M.; Sofia, D.; Barletta, D.; Poletto, M. Calibration of DEM simulation of cohesive particles. Chem. Eng. Trans. 2019, 74, 379–384. [Google Scholar] [CrossRef]
- Cao, X.; Li, Z.; Li, H.; Wang, X.; Ma, X. Measurement and Calibration of the Parameters for Discrete Element Method Modeling of Rapeseed. Processes 2021, 9, 605. [Google Scholar] [CrossRef]
- Daraio, D.; Villoria, J.; Ingram, A.; Alexiadis, A.; Stitt, E.H.; Munnoch, A.L.; Marigo, M. Using Discrete Element method (DEM) simulations to reveal the differences in the γ-Al2O3 to α-Al2O3 mechanically induced phase transformation between a planetary ball mill and an attritor mill. Miner. Eng. 2020, 155, 106374. [Google Scholar] [CrossRef]
- Gu, X.; Zhang, J.; Huang, X. DEM analysis of monotonic and cyclic behaviors of sand based on critical state soil mechanics framework. Comput. Geotech. 2020, 128, 103787. [Google Scholar] [CrossRef]








| Stiffness | Shape | Surface Roughness | Particle Size (mm) | ||||
|---|---|---|---|---|---|---|---|
| Soft | Hard | Spherical | Non-Spherical | Ra | Rz | ||
| Polyethene | X | X | 12.808 | 50.723 | 1.8 | ||
| Glass bead | X | X | 1.767 | 11.462 | 1.65 | ||
| Alpha-alumina | X | X | 49.262 | 187.453 | 1.72 | ||
| Quartz sand | X | X | 13.416 | 49.623 | 0.5 | ||
| Saturation Level | Strain Rate | Smooth Particles (Polymer and Glass) | Rough Particles (Sand and Alpha-Alumina) |
|---|---|---|---|
| 100% | Low | Mostly brittle with failure just after the yield point | Dilatant with slight strain softening or hardening |
| High | Brittle failure | Brittle behavior with failure just after yield or brittle failure | |
| 75% | Low | Brittle failure | Dilatant with vast strain softening |
| High | Brittle failure | Brittle failure |
| Parameter | Polyethene/Glass/ Alpha-Alumina (Spherical) | Sand (Non-Spherical) |
|---|---|---|
| Particle diameter (mm) | 1.8/1.7/1.65 | 0.5 |
| Bond diameter (mm) | 1.0 | 0.3 |
| Particle density (kg/m3) | 960/2500/3960 | 2640 |
| Particle Young’s modulus (GPa) | 0.8/72.3/150 | 72 |
| Particle Poisson’s ratio (-) | 0.36/0.22/0.22 | 0.2 |
| (mm) | 0.7 | 0.2 |
| Numbers of particles (-) | ≈230 | ≈11,200 |
| No. of bonds (-) | ≈1100 | ≈66,000 |
| Porosity (-) | 0.44 | 0.42 |
| Particle-wall sliding friction (-) | 0.45/0.45/0.45 | 0.45 |
| Particle-wall rolling friction (-) | 0.05/0.05/0.05 | 0.5 |
| Particle-particle sliding friction (-) | 0.45/0.4/0.45 | 0.45 |
| Particle-particle rolling friction (-) | 0.05/0.05/0.05 | 0.5 |
| Restitution coefficient (-) | 0.1 | 0.1 |
| Primary Particles | ||||
|---|---|---|---|---|
| Polyethene | Glass | Alpha-Alumina | Natural Sand | |
| Normal and shear strengths | ||||
| 3.5 | 4.2 | 20 | 20 |
| 6 | 2.7 | 20 | 20 |
| Creep parameter A (-) | 0.1 | 0.1 | 0.3 | 0.1 |
| Creep factor m (-) | 0.1 | 0.1 | 0.16 | 0.1 |
| Parameter | Polyethylene/Glass/Alpha-Alumina (Spherical) | Sand (Non-Spherical) |
|---|---|---|
| Bond diameter (mm) | 0.75 | 0.22 |
| (mm) | 0.01 | 0.01 |
| Number of particles | ≈230 | ≈11,200 |
| Number of bonds | ≈550 | ≈34,000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, T.T.; Heinrich, S.; Grabe, J.; Dosta, M. Microscale Modeling of Frozen Particle Fluid Systems with a Bonded-Particle Model Method. Materials 2022, 15, 8505. https://doi.org/10.3390/ma15238505
Chan TT, Heinrich S, Grabe J, Dosta M. Microscale Modeling of Frozen Particle Fluid Systems with a Bonded-Particle Model Method. Materials. 2022; 15(23):8505. https://doi.org/10.3390/ma15238505
Chicago/Turabian StyleChan, Tsz Tung, Stefan Heinrich, Jürgen Grabe, and Maksym Dosta. 2022. "Microscale Modeling of Frozen Particle Fluid Systems with a Bonded-Particle Model Method" Materials 15, no. 23: 8505. https://doi.org/10.3390/ma15238505
APA StyleChan, T. T., Heinrich, S., Grabe, J., & Dosta, M. (2022). Microscale Modeling of Frozen Particle Fluid Systems with a Bonded-Particle Model Method. Materials, 15(23), 8505. https://doi.org/10.3390/ma15238505








