Corrosion Products Formed on MgZr Alloy Embedded in Geopolymer Used as Conditioning Matrix for Nuclear Waste—A Proposition of Interconnected Processes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Characterization
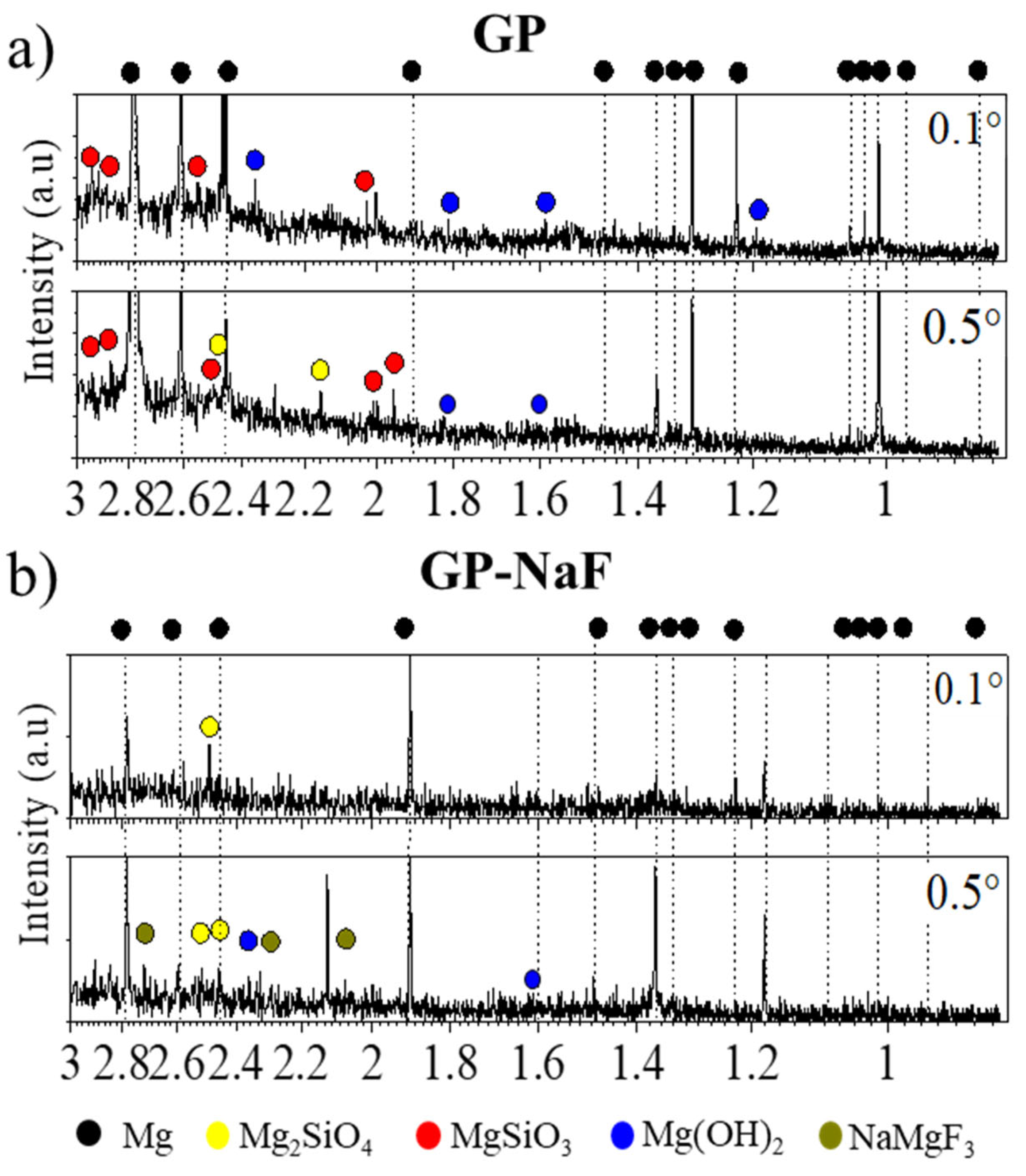
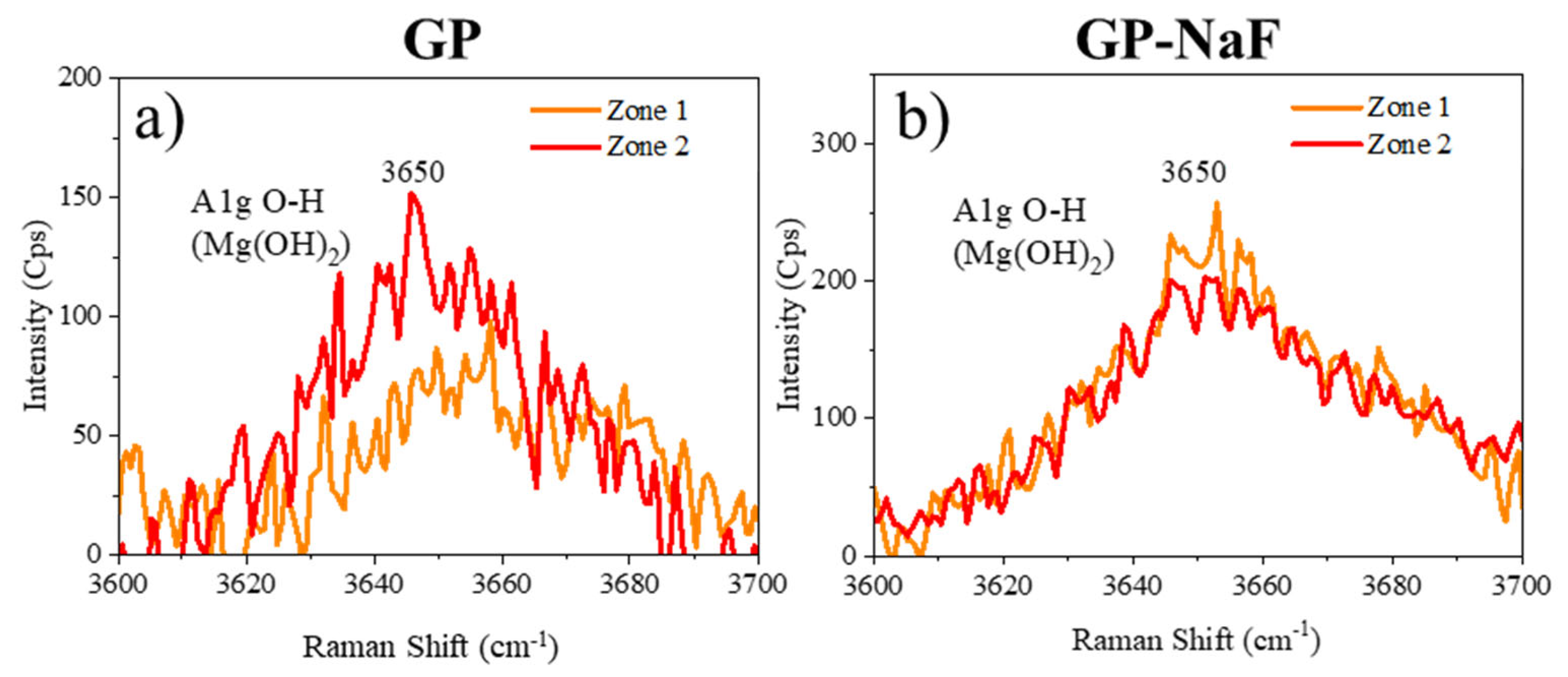
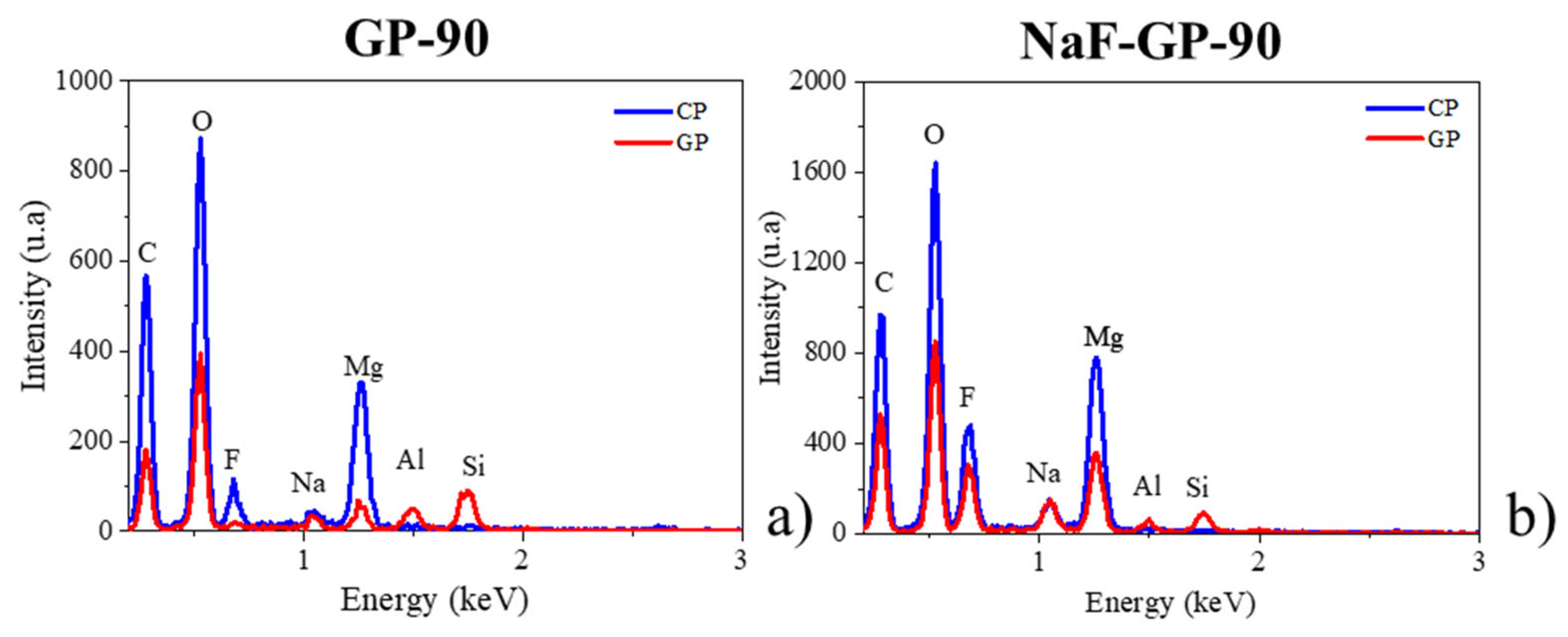
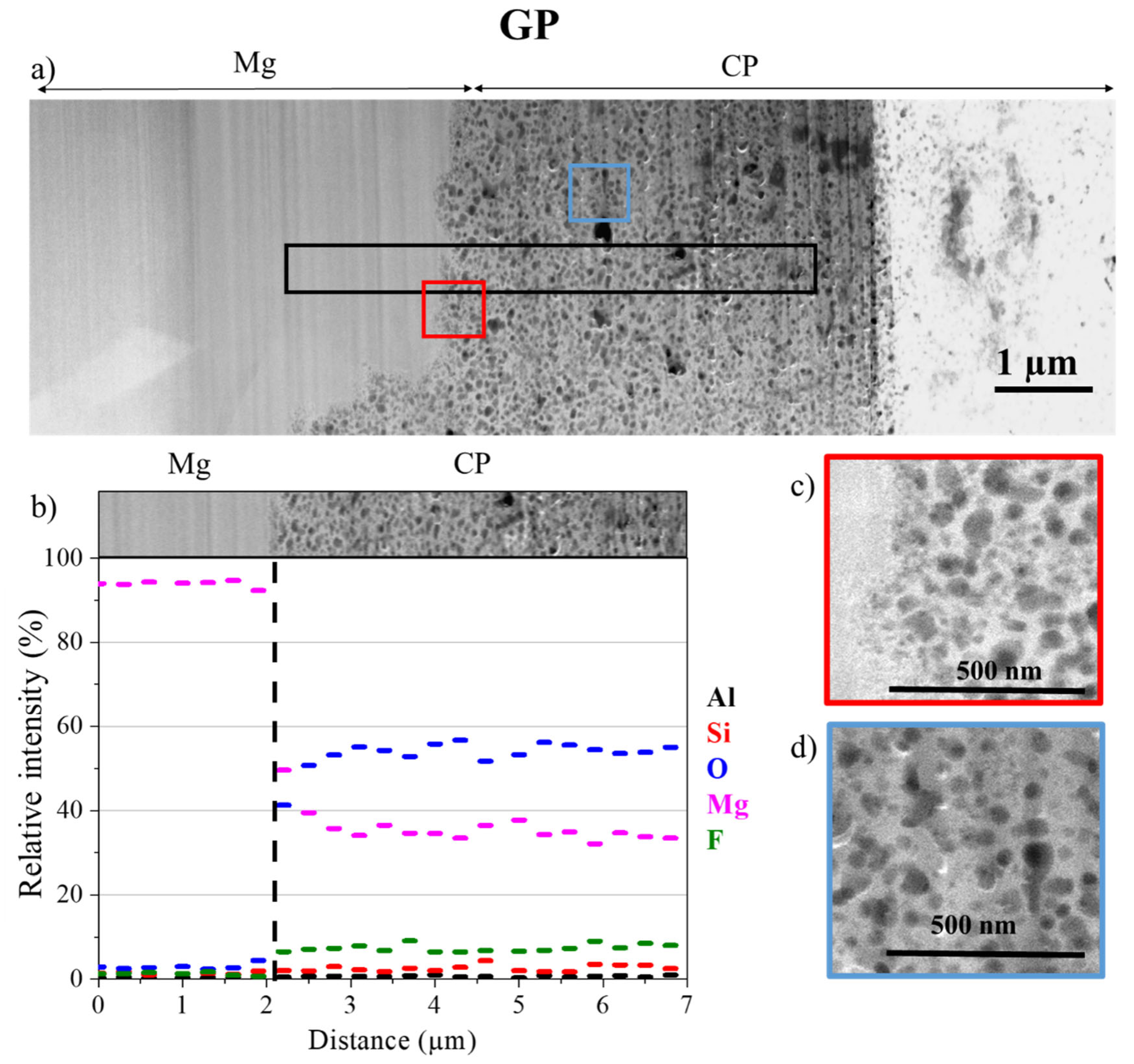
3. Results
3.1. Nature of CP
3.1.1. In Situ GI-XRD
3.1.2. Raman Microspectroscopy
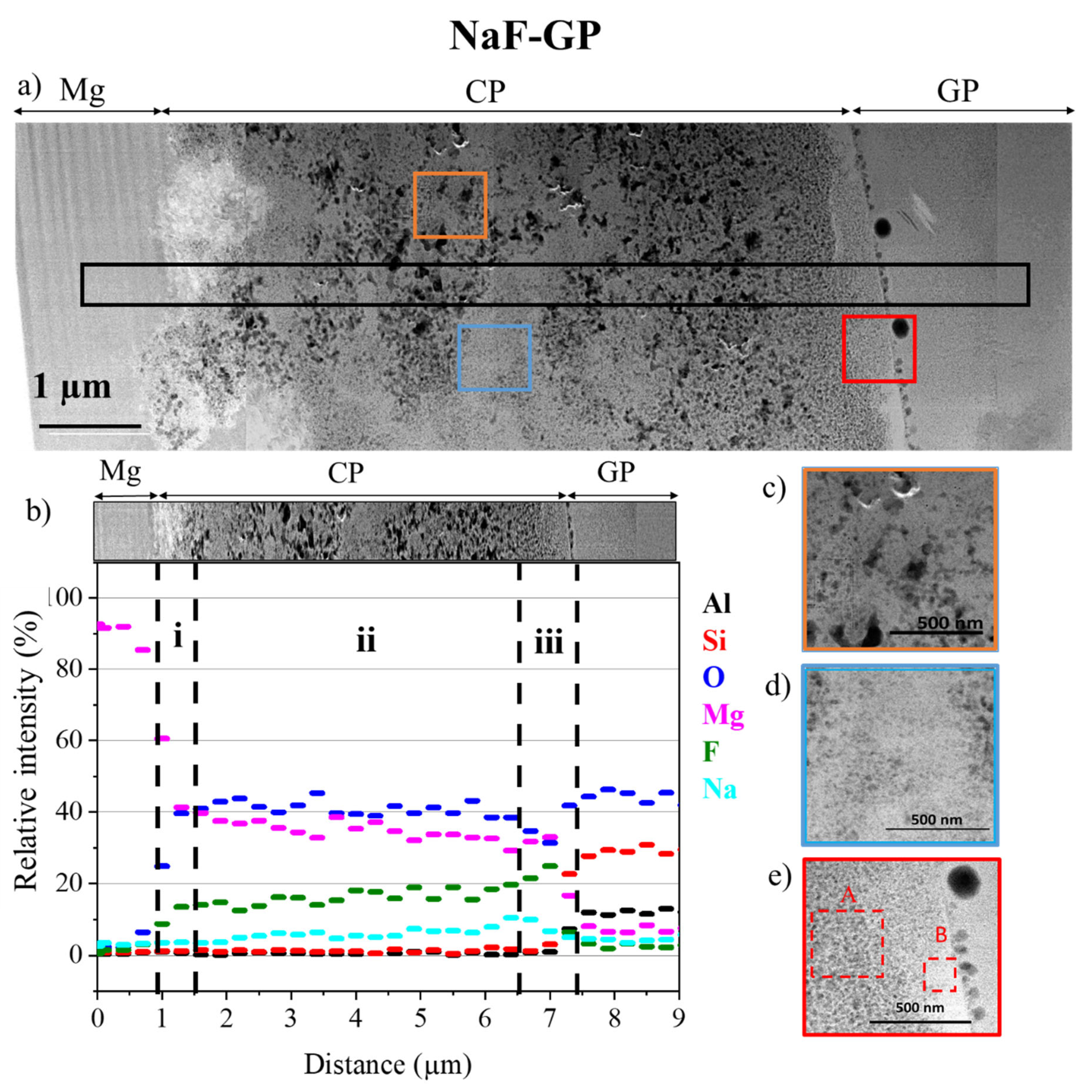
3.2. Morphology and Composition of the Interfaces
3.3. Characterisation at the Nanoscale
Morphology and Composition of CP
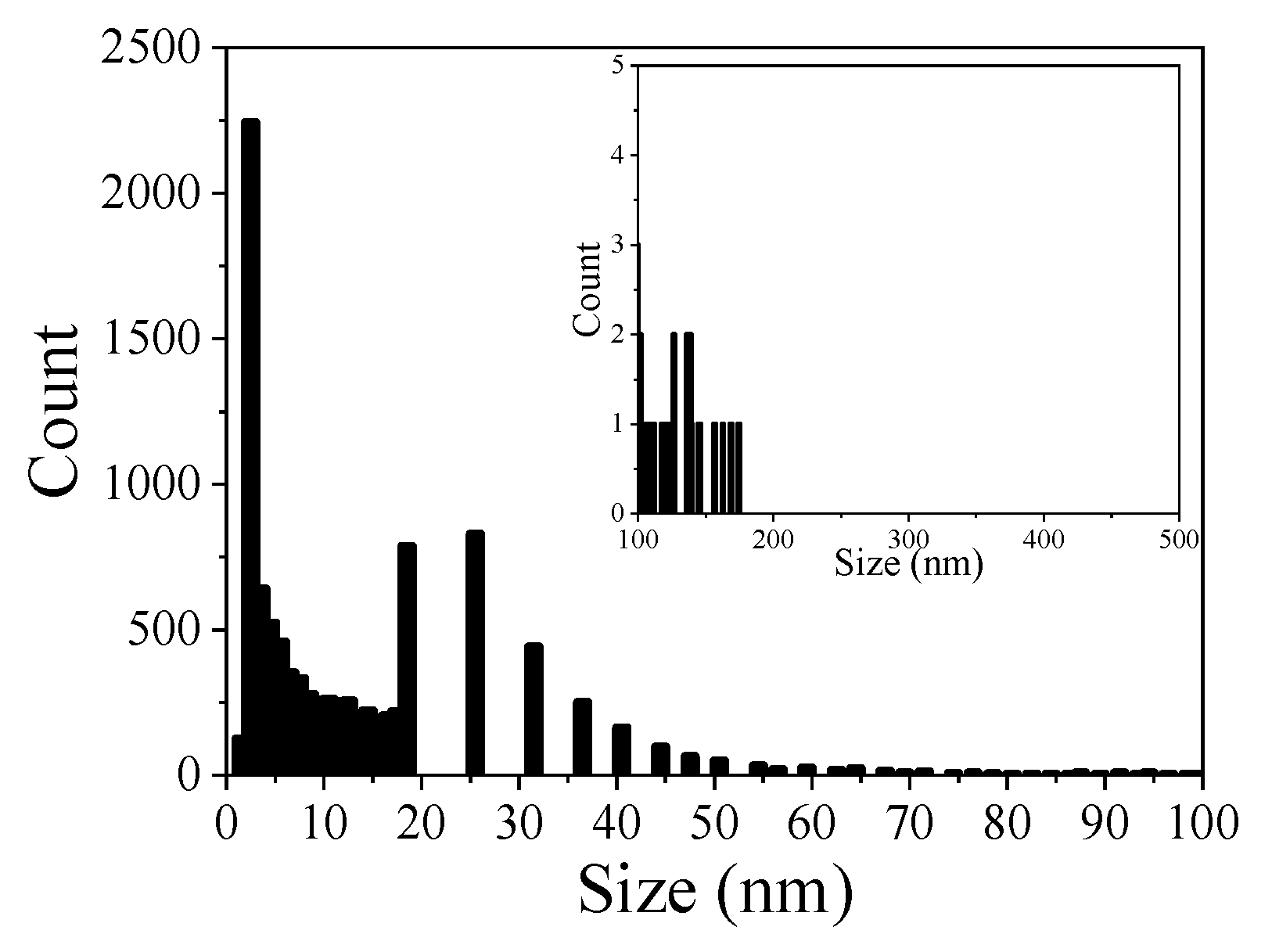
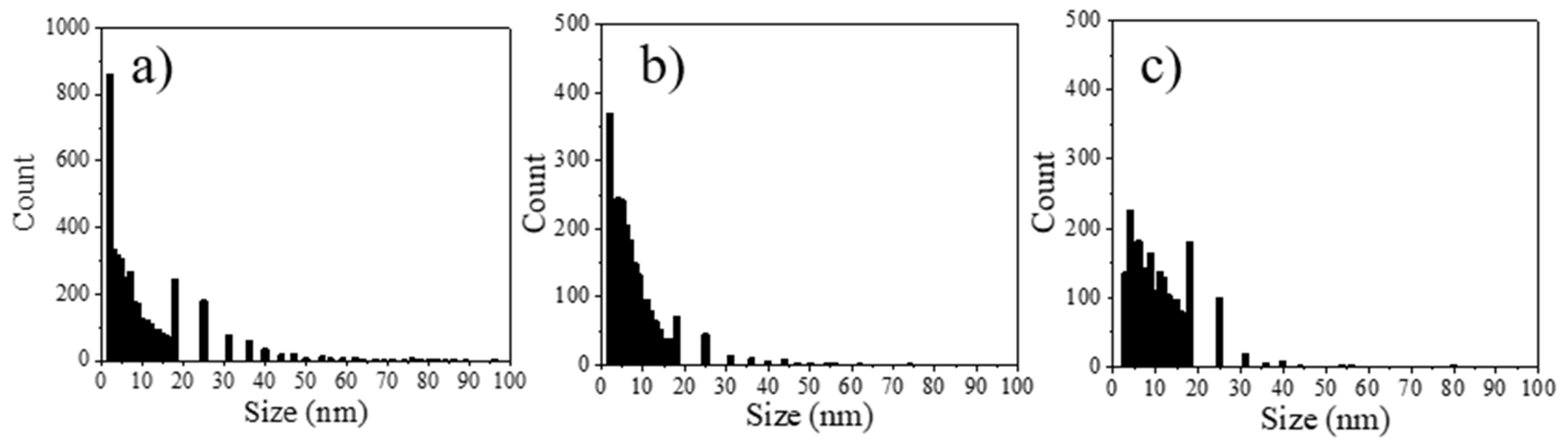
3.4. Porosity of CP Layer
4. Discussion
4.1. General Description of the Samples
4.2. Origin of the Porous Texture
4.3. Relation between the Porous Texture and the Protective Properties of the CP Layer
4.4. Comparison with the CP Layers Formed in Poral Solution Extracted from Geopolymer
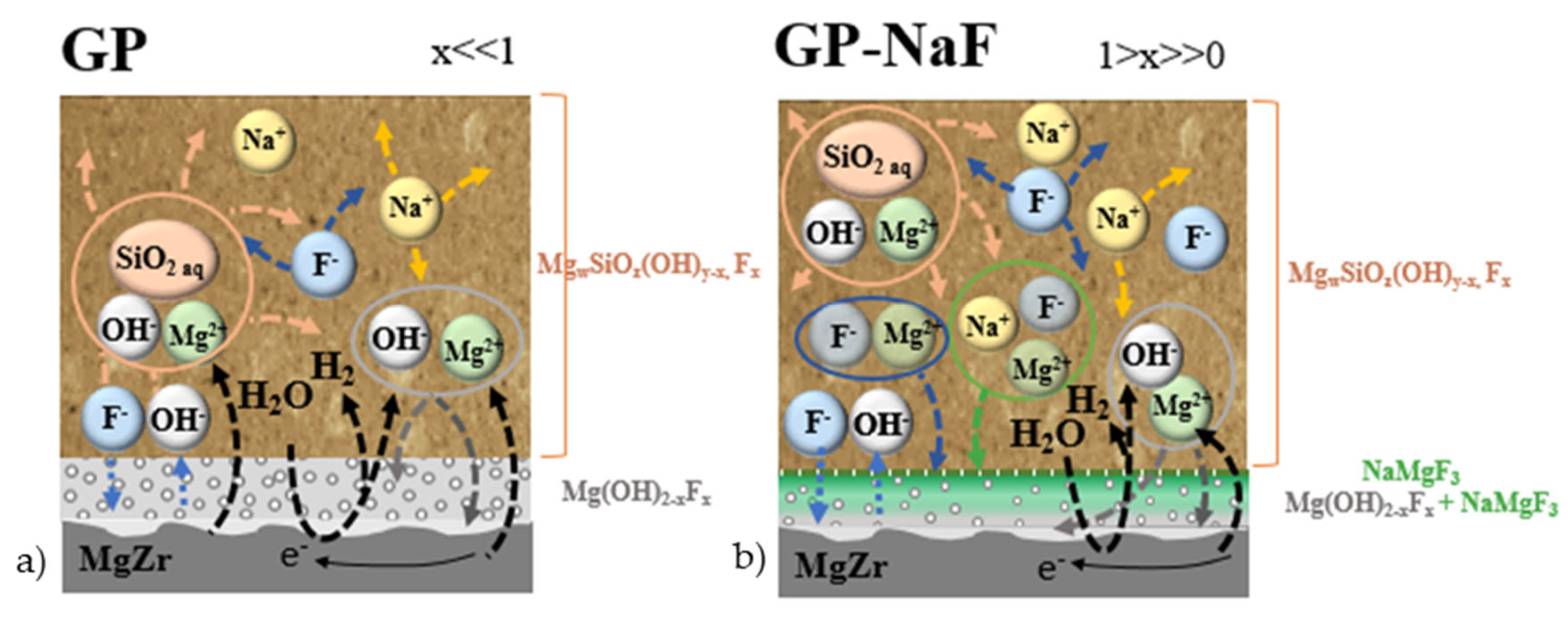
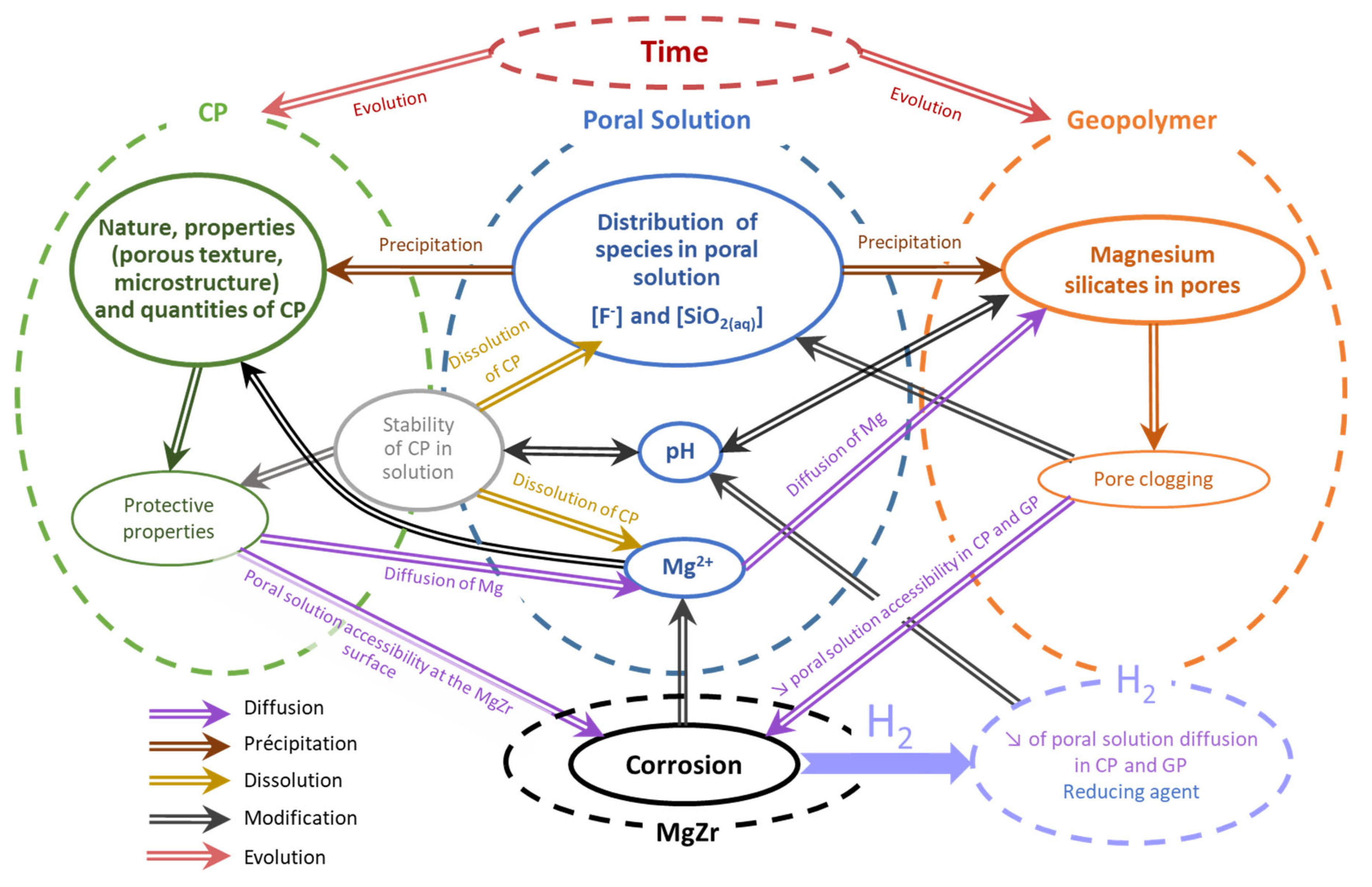
4.5. Proposition of Interconnected Processes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Caillat, R.; Hérenguel, J.; Salesse, M.; Stohr, J. Raisons du choix de l’alliage Mg-Zr pour le gainage des elements combustibles. J. Nucl. Mater. 1963, 8, 1–2. [Google Scholar] [CrossRef]
- Benavent, V.; Frizon, F.; Poulesquen, A. Effect of composition and aging on the porous structure of metakaolin-based geopolymers. J. Appl. Cryst. 2016, 49, 2116–2128. [Google Scholar] [CrossRef]
- Duxson, P.; Fernández-Jiménez, A.; Provis, J.L.; Lukey, G.C.; Palomo, A.; Van Deventer, J.S.J. Geopolymer technology: The current state of the art. J. Mater. Sci. 2007, 42, 2917–2933. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymer Chemistry and Applications; Institut Géopolymère: Saint-Quentin, France, 2011; ISBN 9782954453118. [Google Scholar]
- Perera, D.S.; Blackford, M.G.; Vance, E.R.; Hanna, J.V.; Finnie, K.S.; Nicholson, C.L. Geopolymers for the Immobilization of Radioactive Waste. MRS Proc. 2004, 824, CC8.35. [Google Scholar] [CrossRef]
- Jantzen, C.M.; Lee, W.E.; Ojovan, M.I. Radioactive waste (RAW) conditioning, immobilization, and encapsulation processes and technologies: Overview and advances. In Radioactive Waste Management and Contaminated Site Clean-Up: Processes, Technologies and International Experience; Savannah River Site (S.C.): Aiken, SC, USA, 2013; pp. 171–272. ISBN 9780857094353. [Google Scholar]
- Van Jaarsveld, J.G.S.; Van Deventer, J.S.J.; Lorenzen, L. The potential use of geopolymeric materials to immobilise toxic metals: Part I. Theory and applications. Miner. Eng. 1997, 10, 659–669. [Google Scholar] [CrossRef]
- Li, Q.; Sun, Z.; Tao, D.; Xu, Y.; Li, P.; Cui, H.; Zhai, J. Immobilization of simulated radionuclide 133Cs+ by fly ash-based geopolymer. J. Hazard. Mater. 2013, 262, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ding, Y.; Lu, X. Immobilization of simulated radionuclide 90Sr by fly ash-slag-metakaolin-based geopolymer. Nucl. Technol. 2017, 198, 64–69. [Google Scholar] [CrossRef]
- Cantarel, V.; Nouaille, F.; Rooses, A.; Lambertin, D.; Poulesquen, A.; Frizon, F. Solidification/stabilisation of liquid oil waste in metakaolin-based geopolymer. J. Nucl. Mater. 2015, 464, 16–19. [Google Scholar] [CrossRef]
- Perrault, G.G. The potential-pH diagram of the magnesium-water system. J. Electroanal. Chem. Interfacial Electrochem. 1974, 51, 107–119. [Google Scholar] [CrossRef]
- Chartier, D.; Muzeau, B.; Stefan, L.; Sanchez-Canet, J.; Monguillon, C. Magnesium alloys and graphite wastes encapsulated in cementitious materials: Reduction of galvanic corrosion using alkali hydroxide activated blast furnace slag. MRS Adv. 2016, 1, 4095–4101. [Google Scholar] [CrossRef]
- El-Taib Heakal, F.; Tantawy, N.S.; Shehata, O.S. Impact of chloride and fluoride additions on surface reactivity and passivity of AM60 magnesium alloy in buffer solution. Corros. Sci. 2012, 64, 153–163. [Google Scholar] [CrossRef]
- El-Taib Heakal, F.; Fekry, A.M.; Fatayerji, M.Z. Influence of halides on the dissolution and passivation behavior of AZ91D magnesium alloy in aqueous solutions. Electrochim. Acta 2009, 54, 1545–1557. [Google Scholar] [CrossRef]
- Bradford, P.M.; Case, B.; Dearnaley, G.; Turner, J.F.; Woolsey, I.S. Ion beam analysis of corrosion films on a high magnesium alloy (Magnox Al 80). Corros. Sci. 1976, 16, 747–766. [Google Scholar] [CrossRef]
- Gulbrandsen, E.; Taftø, J.; Olsen, A. The passive behaviour of Mg in alkaline fluoride solutions. Electrochemical and electron microscopical investigations. Corros. Sci. 1993, 34, 1423–1440. [Google Scholar] [CrossRef]
- Boubon, R.; Deschanels, X.; Cabié, M.; Rébiscoul, D. Evolution of Corrosion Products Formed during the Corrosion of MgZr Alloy in Poral Solutions Extracted from Na-Geopolymers Used as Conditioning Matrix for Nuclear Waste. Materials 2020, 13, 4958. [Google Scholar] [CrossRef]
- Lambertin, D.; Frizon, F.; Bart, F. Mg-Zr alloy behavior in basic solutions and immobilization in Portland cement and Na-geopolymer with sodium fluoride inhibitor. Surf. Coat. Technol. 2012, 206, 4567–4573. [Google Scholar] [CrossRef]
- Rooses, A.; Steins, P.; Dannoux-Papin, A.; Lambertin, D.; Poulesquen, A.; Frizon, F. Encapsulation of Mg-Zr alloy in metakaolin-based geopolymer. Appl. Clay Sci. 2013, 73, 86–92. [Google Scholar] [CrossRef]
- Rooses, A.; Lambertin, D.; Chartier, D.; Frizon, F. Galvanic corrosion of Mg-Zr fuel cladding and steel immobilized in Portland cement and geopolymer at early ages. J. Nucl. Mater. 2013, 435, 137–140. [Google Scholar] [CrossRef]
- Barros, C.F.; Muzeau, B.; L’Hostis, V.; François, R. Impact of fluoride concentration on general corrosion of Mg-Zr alloy in a Na-geopolymer and alkaline solutions. Corros. Sci. 2020, 176, 109009. [Google Scholar] [CrossRef]
- Steins, P.; Poulesquen, A.; Frizon, F.; Diat, O.; Jestin, J.; Causse, J.; Lambertin, D.; Rossignol, S. Effect of aging and alkali activator on the porous structure of a geopolymer. J. Appl. Cryst. 2014, 47, 316–324. [Google Scholar] [CrossRef]
- Cyr, M.; Pouhet, R. Carbonation in the pore solution of metakaolin-based geopolymer. Cem. Concr. Res. 2016, 88, 227–235. [Google Scholar] [CrossRef]
- Thomas, S.; Thomas, R.; Zachariah, A. Microscopy Methods in Nanomaterials Characterization; Elsevier: Amsterdam, Netherlands, 2017; ISBN 9780323461474. [Google Scholar]
- Ricolleau, C.; Nelayah, J.; Oikawa, T.; Kohno, Y.; Braidy, N.; Wang, G.; Hue, F.; Florea, L.; Pierron Bohnes, V.; Alloyeau, D. Performances of an 80–200 kV microscope employing a cold-FEG and an aberration-corrected objective lens. J. Electron. Microsc. 2013, 62, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Dahmen, U.; Kim, M.G.; Searcy, A.W. Microstructural evolution during the decomposition of Mg(OH)2. Ultramicroscopy 1987, 23, 365–370. [Google Scholar] [CrossRef]
- Song, G.L.; Unocic, K.A. The anodic surface film and hydrogen evolution on Mg. Corros. Sci. 2015, 98, 758–765. [Google Scholar] [CrossRef]
- Qian, M.; Das, A. Grain refinement of magnesium alloys by zirconium: Formation of equiaxed grains. Scr. Mater. 2006, 54, 881–886. [Google Scholar] [CrossRef]
- Bernard, J.; Caillat, R.; Darras, R. Magnesium and Related Low Alloys; CEA Report: Gif sur Yvette, France, 1959. [Google Scholar]
- Reschka, S.; Gerstein, G.; Dalinger, A.; Herbst, S.; Nürnberger, F.; Zaefferer, S. Visualization and Observation of Morphological Peculiarities of Twin Formation in Mg-Based Samples After Electrically Assisted Forming. Metallogr. Microstruct. Anal. 2019, 8, 806–814. [Google Scholar] [CrossRef]
- Maltseva, A.; Shkirskiy, V.; Lefèvre, G.; Volovitch, P. Effect of pH on Mg(OH) 2 film evolution on corroding Mg by in situ kinetic Raman mapping (KRM). Corros. Sci. 2019, 153, 272–282. [Google Scholar] [CrossRef]
- Taheri, M.; Kish, J.R. Nature of surface film formed on mg exposed to 1 M NaOH. J. Electrochem. Soc. 2013, 160, 36–41. [Google Scholar] [CrossRef]
- Ghali, E.; Dietzel, W.; Kainer, K.-U. General and Localized Corrosion of Magnesium Alloys: A Critical Review. J. Mater. Eng. Perform. 2004, 13, 7–23. [Google Scholar] [CrossRef]
- Brady, M.P.; Rother, G.; Anovitz, L.M.; Littrell, K.C.; Unocic, K.A.; Elsentriecy, H.H.; Song, G.-L.L.; Thomson, J.K.; Gallego, N.C.; Davis, B. Film Breakdown and Nano-Porous Mg(OH) 2 Formation from Corrosion of Magnesium Alloys in Salt Solutions. J. Electrochem. Soc. 2015, 162, C140–C149. [Google Scholar] [CrossRef]
- Steins, P.; Poulesquen, A.; Diat, O.; Frizon, F. Structural Evolution during Geopolymerization from an Early Age to Consolidated Material. Langmuir 2012, 28, 8502–8510. [Google Scholar] [CrossRef]
- Dauphin-Ducharme, P.; Kuss, C.; Rossouw, D.; Payne, N.A.; Danis, L.; Botton, G.A.; Mauzeroll, J. Corrosion Product Formation Monitored Using the Feedback Mode of Scanning Electrochemical Microscopy with Carbon Microelectrodes. J. Electrochem. Soc. 2015, 162, C677–C683. [Google Scholar] [CrossRef]
- Ribbe, P.H. Titanium, fluorine, and hydroxyl in the humite minerals. Am. Mineral. 1979, 64, 1027. [Google Scholar]
- Vermilyea, D.A.; Kirk, C.F. Studies of Inhibition of Magnesium Corrosion. J. Electrochem. Soc. 1969, 116, 1487. [Google Scholar] [CrossRef]
- Sevonkaev, I.; Goia, D.V.; Matijević, E. Formation and structure of cubic particles of sodium magnesium fluoride (neighborite). J. Colloid Interface Sci. 2008, 317, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Baum, M. The Role of Water Properties and Specific Ion Effects on the Evolution of Silica Nanoconfinement. 2018. Available online: http://www.theses.fr (accessed on 11 December 2020).
- Aréna, H.; Rébiscoul, D.; Garcès, E.; Godon, N. Comparative effect of alkaline elements and calcium on alteration of International Simple Glass. NPJ Mater. Degrad. 2019, 3, 1–11. [Google Scholar] [CrossRef]
- Gin, S.; Guittonneau, C.; Godon, N.; Neff, D.; Rebiscoul, D.; Cabí, M.; Mostefaoui, S. Nuclear glass durability: New insight into alteration layer properties. J. Phys. Chem. C 2011, 115, 18696–18706. [Google Scholar] [CrossRef]
- Michelin, A.; Burger, E.; Rebiscoul, D.; Neff, D.; Bruguier, F.; Drouet, E.; Dillmann, P.; Gin, S. Silicate glass alteration enhanced by iron: Origin and long-term implications. Environ. Sci. Technol. 2013, 47, 750–756. [Google Scholar] [CrossRef]
- Burger, E.; Rebiscoul, D.; Bruguier, F.; Jublot, M.; Lartigue, J.E.; Gin, S. Impact of iron on nuclear glass alteration in geological repository conditions: A multiscale approach. Appl. Geochem. 2013, 31, 159–170. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Schott, J.; Castillo, A. Kinetics of brucite dissolution at 25 °C in the presence of organic and inorganic ligands and divalent metals. Geochim. Cosmochim. Acta 2005, 69, 905–918. [Google Scholar] [CrossRef]
- Taheri, M.; Phillips, R.C.; Kish, J.R.; Botton, G.A. Analysis of the surface film formed on Mg by exposure to water using a FIB cross-section and STEM-EDS. Corros. Sci. 2012, 59, 222–228. [Google Scholar] [CrossRef]



| Impurities | Al | As | Co | Cr | Cu | Fe | Mn | Ni | Sb | Zn | Cl |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Quantity (ppm) | <10 | <20 | <10 | 22 | 2 | 9 | 11 | 1 | <10 | 31 | 10 |
| Geopolymer | pH | [Na] mmol.L−1 | [Si] mmol.L−1 | [F] mmol.L−1 | [Cl] mmol.L−1 |
|---|---|---|---|---|---|
| GP | 12.37 | 418.9 ± 6.5 | 46.6 ± 15.8 | 19.0 ± 0.4 | 27.4 ± 0.5 |
| NaF-GP | 12.40 | 1116.5 ± 237.8 | 32.9 ± 9.1 | 1855.9 ± 37.1 | 20.0 ± 0.4 |
| Sample | Zone | Porosity (%) | Mean Pore Size (nm) | Median Pore Size (nm) |
|---|---|---|---|---|
| GP (Figure 6) | CP layer | 38.2 ± 5.9 | 13.6 | 8.5 |
| GP-NaF (Figure 7) | Orange square—image (c) Blue square—image (d) Red square—image (e) zone A zone B | 28.9 ± 0.7 22.5 ± 2.5 21.2 ± 1.8 Not measureable | 10.5 7.9 10.8 Not measurable | 6.5 6.1 9.7 Not measurable |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boubon, R.; Nelayah, J.; Tardif, S.; Deschanels, X.; Rébiscoul, D. Corrosion Products Formed on MgZr Alloy Embedded in Geopolymer Used as Conditioning Matrix for Nuclear Waste—A Proposition of Interconnected Processes. Materials 2021, 14, 2017. https://doi.org/10.3390/ma14082017
Boubon R, Nelayah J, Tardif S, Deschanels X, Rébiscoul D. Corrosion Products Formed on MgZr Alloy Embedded in Geopolymer Used as Conditioning Matrix for Nuclear Waste—A Proposition of Interconnected Processes. Materials. 2021; 14(8):2017. https://doi.org/10.3390/ma14082017
Chicago/Turabian StyleBoubon, Rémi, Jaysen Nelayah, Samuel Tardif, Xavier Deschanels, and Diane Rébiscoul. 2021. "Corrosion Products Formed on MgZr Alloy Embedded in Geopolymer Used as Conditioning Matrix for Nuclear Waste—A Proposition of Interconnected Processes" Materials 14, no. 8: 2017. https://doi.org/10.3390/ma14082017
APA StyleBoubon, R., Nelayah, J., Tardif, S., Deschanels, X., & Rébiscoul, D. (2021). Corrosion Products Formed on MgZr Alloy Embedded in Geopolymer Used as Conditioning Matrix for Nuclear Waste—A Proposition of Interconnected Processes. Materials, 14(8), 2017. https://doi.org/10.3390/ma14082017








