Growth of ZnO Nanorods on ITO Film for Piezoelectric Nanogenerators
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
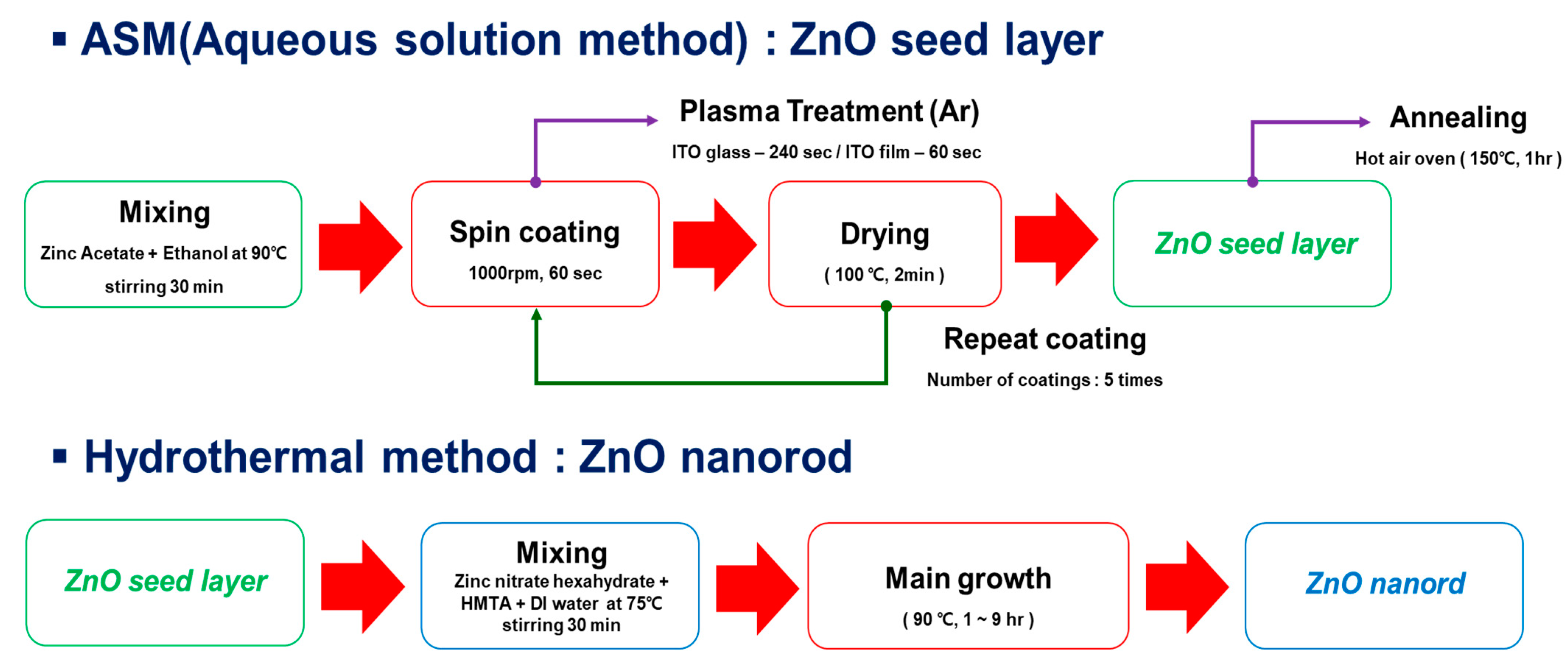
2.2.1. Preparation of the ZnO Seed Layer
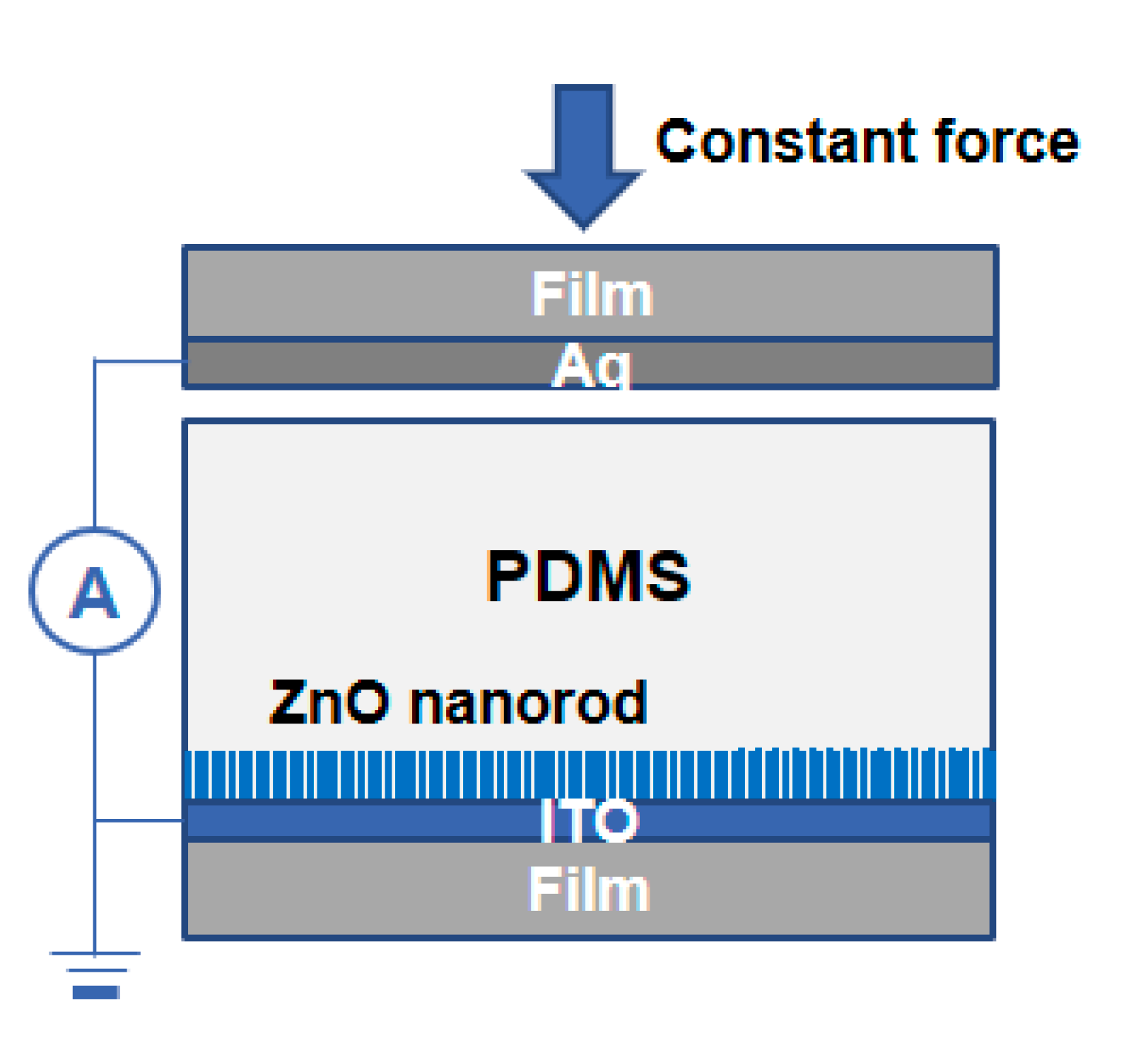
2.2.2. Growth of ZNRs
2.3. Characterization
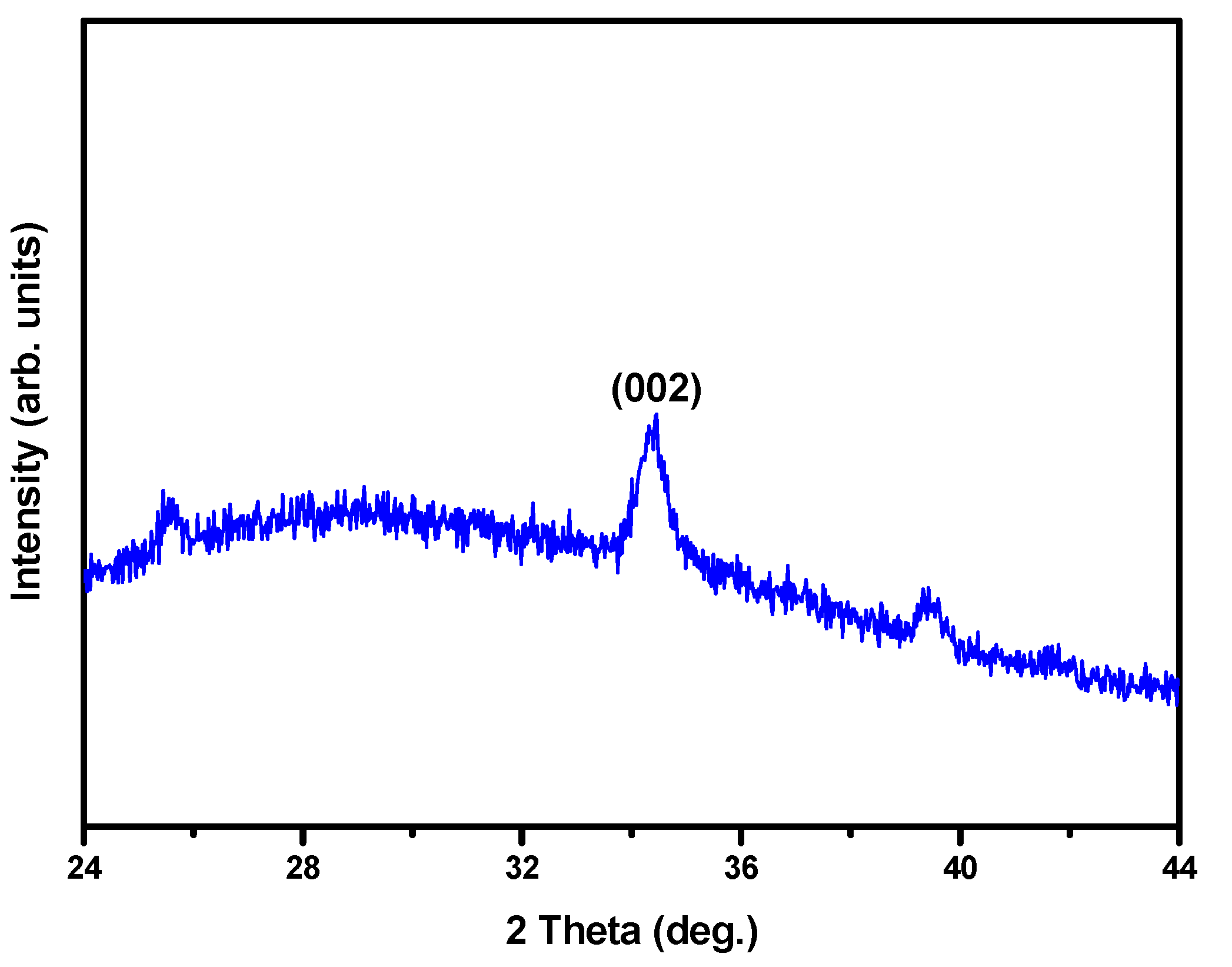
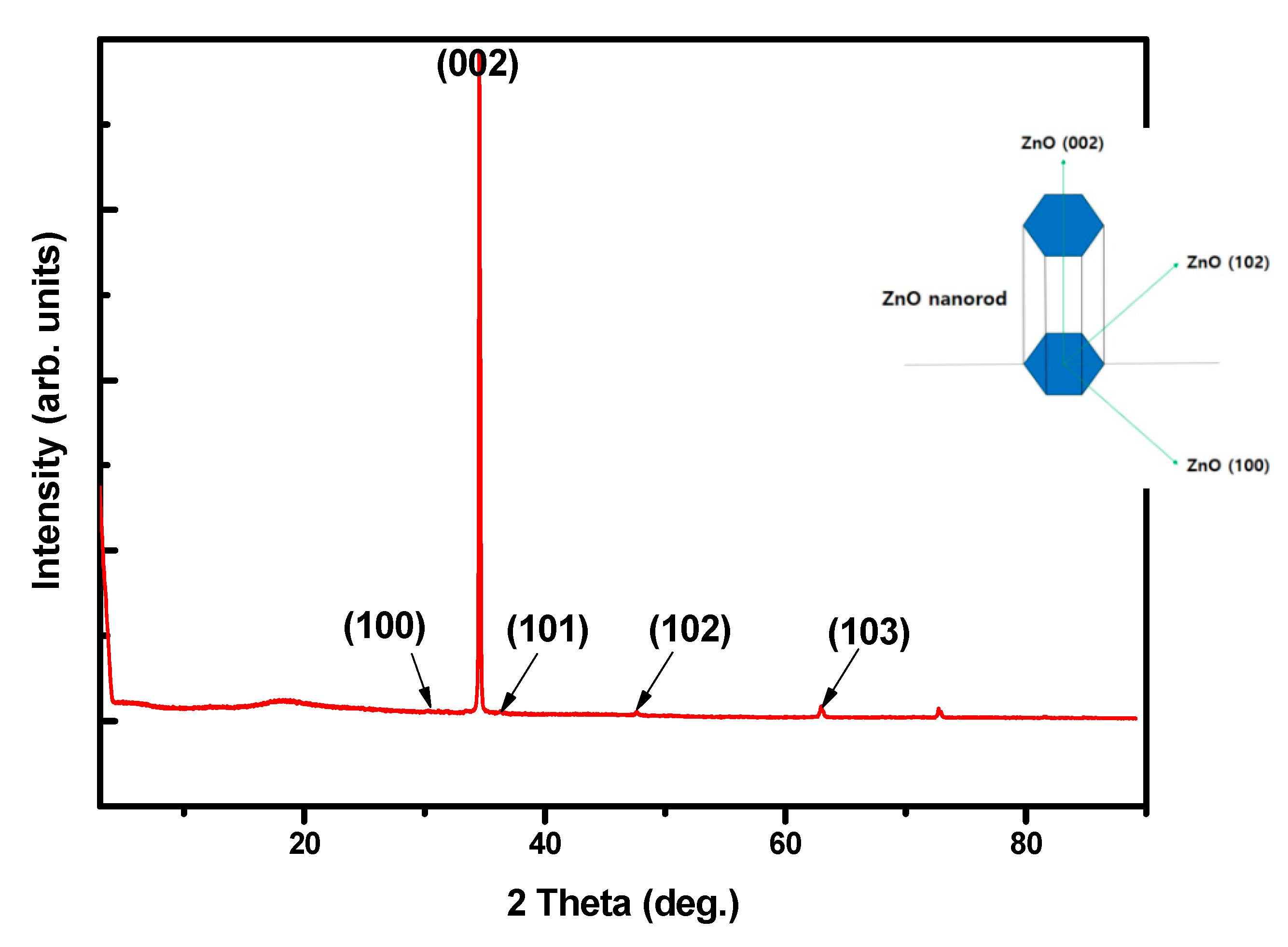
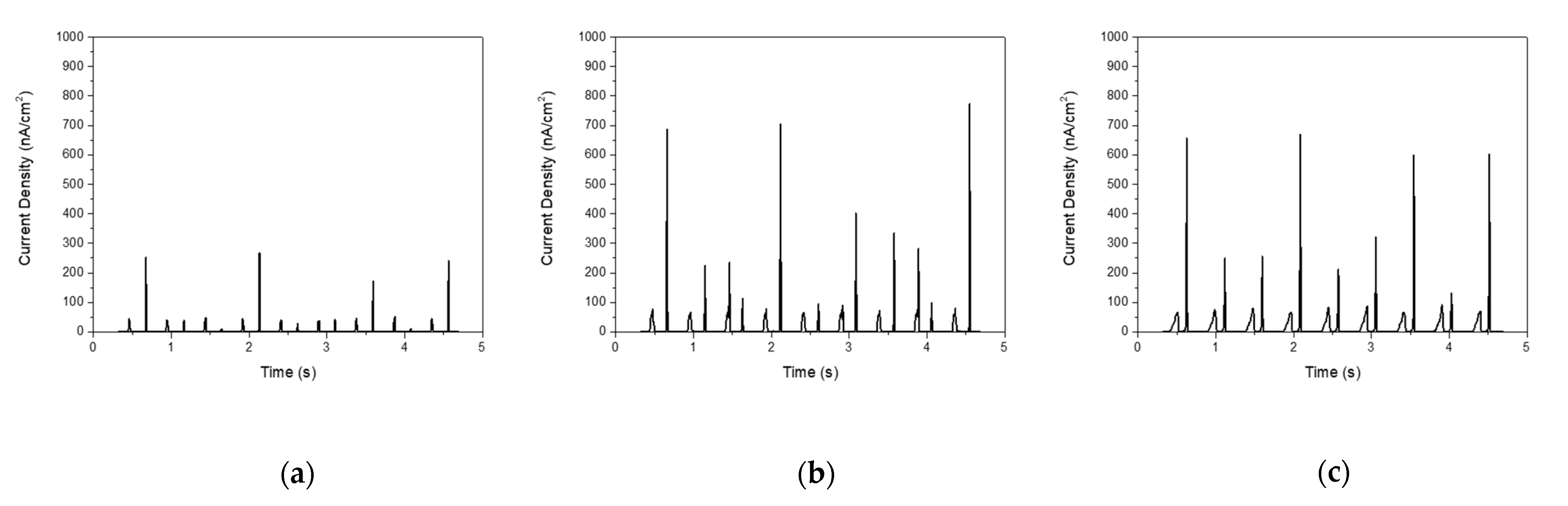
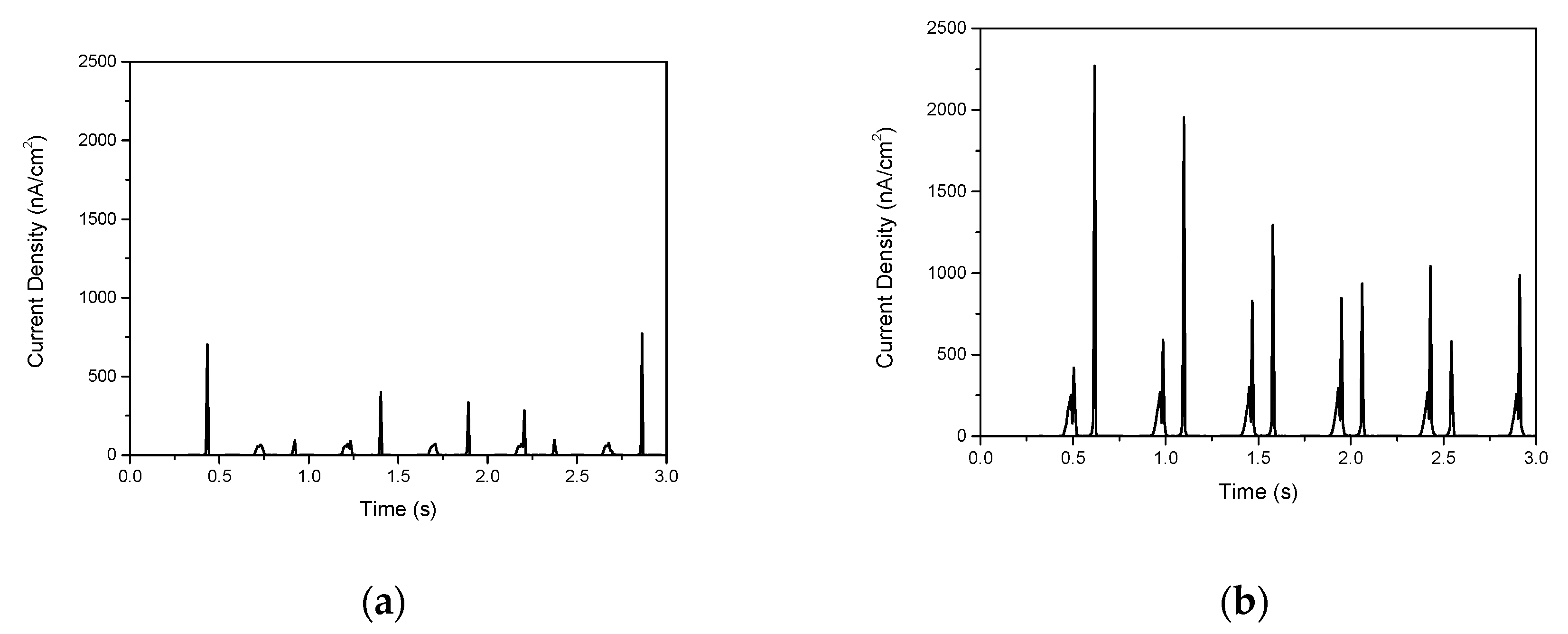
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Priya, S.; Song, H.C.; Zhou, Y.; Varghese, R.; Chopra, A.; Kim, S.G.; Kanno, I.; Wu, L.; Ha, D.S.; Ryu, J.; et al. A Review on Piezoelectric Energy Harvesting: Materials, Methods, and Circuits. Energy Harvest. Syst. 2017, 4, 3–39. [Google Scholar] [CrossRef]
- Park, K.I.; Xu, S.; Liu, Y.; Hwang, G.T.; Kang, S.-J.L.; Wang, Z.L.; Lee, K.J. Piezoelectric BaTiO3 Thin Film Nanogenerator on Plastic Substrates. Nano Lett. 2010, 10, 4939–4943. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Song, J.; Liu, J.; Wang, Z.L. Direct-Current Nanogenerator Driven by Ultrasonic Waves. Science 2007, 316, 102–105. [Google Scholar] [CrossRef]
- Lin, Y.F.; Song, J.; Ding, Y.; Lu, S.Y.; Wang, Z.L. Piezoelectric nanogenerator using CdS nanowires. Appl. Phys. Lett. 2008, 92, 022105. [Google Scholar] [CrossRef]
- Saryanto, H.; Sebayang, D. The simple fabrication of nanorods mass production for the dye-sensitized solar cell. MATEC Web Conf. 2017, 101, 03006. [Google Scholar]
- Huang, Q.; Fang, L.; Chen, X.; Saleem, M. Effect of polyethyleneimine on the growth of ZnO nanorod arrays and their application in dye-sensitized solar cells. J. Alloy. Compd. 2011, 509, 9456–9459. [Google Scholar] [CrossRef]
- Thakur, P.; Kool, A.; Hoque, N.A.; Bagchi, B.; Khatun, F.; Biswas, P.; Brahma, D.; Roy, S.; Banerjee, S.; Das, S. Superior performances of in situ synthesized ZnO/PVDF thin film based self-poled piezoelectric nanogenerator and self-charged photo-power bank with high durability. Nano Energy 2018, 44, 456–467. [Google Scholar] [CrossRef]
- Al-Ruqeishi, M.S.; Mohiuddin, T.; Al-Habsi, B.; Al-Ruqeishi, F.; Al-Fahdi, A.; Al-Khusaibi, A. Piezoelectric nanogenerator based on ZnO nanorods. Arab. J. Chem. 2019, 12, 5173–5179. [Google Scholar] [CrossRef]
- Lu, M.P.; Song, J.; Lu, M.Y.; Chen, M.T.; Gao, Y.; Chen, L.J.; Wang, Z.L. Piezoelectric Nanogenerator Using p-Type ZnO Nanowire Arrays. Nano Lett. 2009, 9, 1223–1227. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, J.; Song, J.; Liu, J.; Xu, N.; Wang, Z.L. Piezoelectric Field Effect Transistor and Nanoforce Sensor Based on a Single ZnO Nanowire. Nano Lett. 2006, 6, 2768–2772. [Google Scholar] [CrossRef]
- Choi, D.; Choi, M.Y.; Choi, W.M.; Shin, H.J.; Park, H.K.; Seo, J.S.; Park, J.; Yoon, S.M.; Chae, S.J.; Lee, Y.H.; et al. Fully Rollable Transparent Nanogenerators Based on Graphene Electrodes. Adv. Mater. 2010, 22, 2187–2192. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fei, P.; Zhou, J.; Tummala, R.; Wang, Z.L. Toward high output-power nanogenerator. Appl. Phys. Lett. 2008, 92, 173105. [Google Scholar] [CrossRef]
- Greene, L.E.; Law, M.; Goldberger, J.; Kim, F.; Johnson, J.C.; Zhang, Y.; Saykally, R.J.; Yang, P. Low-Temperature Wafer-Scale Production of ZnO Nanowire Arrays. Angew. Chem. Int. Ed. 2003, 42, 3031–3034. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Yang, Z.; Navale, S.T.; Han, S.; Liu, X.; Liu, W.; Lu, Y.; Stadler, F.J.; Zhu, D. Ethanol sensing behavior of Pd-nanoparticles decorated ZnO-nanorod based chemiresistive gas sensors. Sens. Actuators B Chem. 2019, 298, 126850. [Google Scholar]
- Kartopu, G.; Turkay, D.; Ozcan, C.; Hadibrata, W.; Aurang, P.; Yerci, S.; Unalan, H.E.; Barrioz, V.; Qu, Y.; Bowen, L.; et al. Photovoltaic performance of CdS/CdTe junctions on ZnO nanorod arrays. Sol. Energy Mater. Sol. Cells 2018, 176, 100–108. [Google Scholar] [CrossRef]
- Wang, X.; Summers, C.J.; Wang, Z.L. Large-Scale Hexagonal-Patterned Growth of Aligned ZnO Nanorods for Nano-optoelectronic and Nanosensor Arrays. Nano Lett. 2004, 4, 423–426. [Google Scholar] [CrossRef]
- Wang, X.; Lu, H.; Liu, W.; Guo, M.; Zhang, M. Electrodeposition of flexible stainless steel mesh supported ZnO nanorod attays with enhanced photocatalytic performance. Ceram. Int. 2017, 43, 6460–6466. [Google Scholar] [CrossRef]
- Song, J.; Baek, S.; Lim, S. Effect of hydrothermal reaction conditions on the optical properties of ZnO nanorods. Phys. B 2008, 403, 1960–1963. [Google Scholar] [CrossRef]
- Polsongkram, D.; Chamninok, P.; Pukird, S.; Chow, L.; Lupan, O.; Chai, G.; Khallaf, H.; Park, S.; Schulte, A. Effect of synthesis conditions on the growth of ZnO nanorods via hydrothermal method. Phys. B 2008, 403, 3713–3717. [Google Scholar] [CrossRef]
- Li, C.; Fang, G.; Li, J.; Ai, L.; Dong, B.; Zhao, X. Effect of Seed Layer on Structural Properties of ZnO Nanorod Arrays Grown by Vapor-Phase Transport. J. Phys. Chem. C 2008, 112, 990–995. [Google Scholar] [CrossRef]
- Yoon, Y.C.; Park, K.S.; Kim, S.D. Effects of low preheating temperature for ZnO seed layer deposited by sol–gel spin coating on the structural properties of hydrothermal ZnO nanorods. Thin Solid Film. 2015, 597, 125–130. [Google Scholar] [CrossRef]
- Wahab, H.A.; Salama, A.A.; El-Saeid, A.A.; Nur, O.; Willander, M.; Battisha, I.K. Optical, structural and morphological studies of (ZnO) nano-rod thin films for biosensor applications using sol gel technique. Results Phys. 2013, 3, 46–51. [Google Scholar] [CrossRef]
- Vaseem, M.; Umar, A.; Kim, S.H.; Hahn, Y.B. Low-Temperature Synthesis of Flower-Shaped CuO Nanostructures by Solution Process: Formation Mechanism and Structural Properties. J. Phys. Chem. C 2008, 112, 5729–5735. [Google Scholar] [CrossRef]
- Lee, Y.M.; Yang, H.W. Optimization of processing parameters on the controlled growth of ZnO nanorod arrays for the performance improvement of solid-state dye-sensitized solar cells. J. Solid State Chem. 2011, 184, 615–623. [Google Scholar] [CrossRef]
- Choi, H.S.; Vaseem, M.; Kim, S.G.; Im, Y.H.; Hahn, Y.B. Growth of high aspect ratio ZnO nanorods by solution process: Effect of polyethyleneimine. J. Solid State Chem. 2012, 189, 25–31. [Google Scholar] [CrossRef]
- Guo, Q.Z.; Yang, L.C.; Wang, R.C.; Liu, C.P. Tunable Work Function of MgxZn1−xO as a Viable Friction Material for a Triboelectric Nanogenerator. ACS Appl. Mater. Interfaces 2019, 11, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Hu, Y.; Xu, C.; Zhang, Y.; Zhang, R.; Wen, X.; Wang, Z.L. Transparent flexible nanogenerator as self-powered sensor for transportation monitoring. Nano Energy 2013, 2, 75–81. [Google Scholar] [CrossRef]
- Opoku, C.; Dahiya, A.S.; Oshman, C.; Cayrel, F.; Poulin-Vittrant, G.; Alquier, D.; Camara, N. Fabrication of ZnO Nanowire Based Piezoelectric Generators and Related Structures. Phys. Procedia 2015, 70, 858–862. [Google Scholar] [CrossRef]
- Kammel, R.S.; Sabry, R.S. Effects of the aspect ratio of ZnO nanorods on the performance of piezoelectric nanogenerators. J. Sci. Adv. Mater. Devices 2019, 4, 420–424. [Google Scholar] [CrossRef]


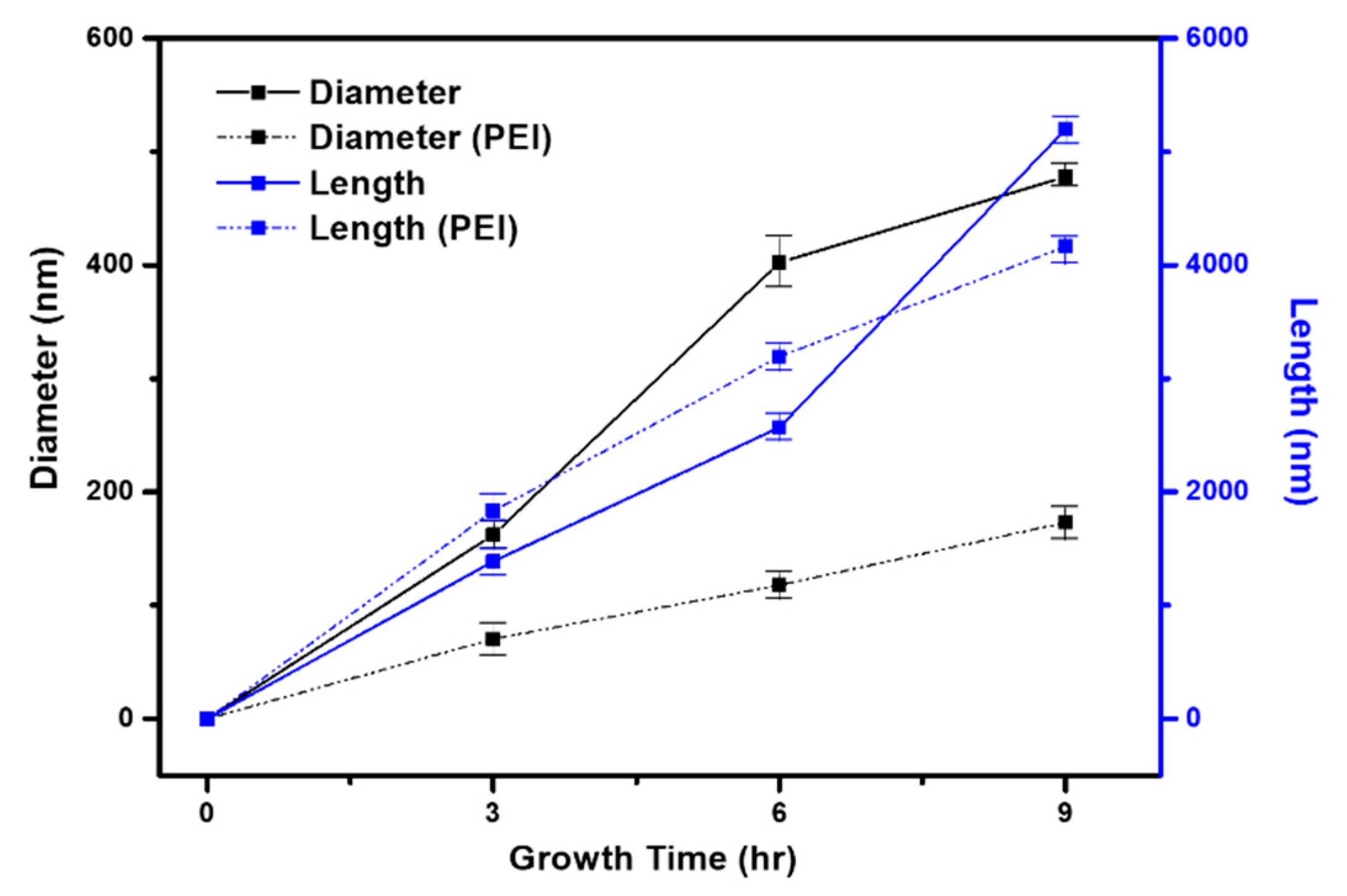

| Growth time (h) | Length (μm) | Diameter (nm) | Aspect Ratio |
|---|---|---|---|
| 3 | 1.389 | 162.2 | 8:1 |
| 3 (w/PEI) | 1.438 | 70.30 | 20:1 |
| 6 | 2.869 | 402.3 | 7:1 |
| 6 (w/PEI) | 3.194 | 142.7 | 22:1 |
| 9 | 5.197 | 478.0 | 11:1 |
| 9 (w/PEI) | 4.164 | 173.0 | 24:1 |
| Structures | Average Work Function (eV) | ΔWork Function with Ag Layer (4.64 eV) |
|---|---|---|
| PMDS (40 μm)/ITO/Glass | 4.99 | 0.35 |
| PMDS (80 μm)/ITO/Glass | 5.50 | 0.86 |
| PMDS (100 μm)/ITO/Glass | 5.21 | 0.57 |
| Aspect Ratio | PDMS Thickness (μm) | Current Density (nA/cm2) |
|---|---|---|
| 11:1 | 40 | 360.5 |
| 80 | 773.2 | |
| 100 | 669.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.G.; Kim, E.H.; Kim, S.S. Growth of ZnO Nanorods on ITO Film for Piezoelectric Nanogenerators. Materials 2021, 14, 1461. https://doi.org/10.3390/ma14061461
Kim HG, Kim EH, Kim SS. Growth of ZnO Nanorods on ITO Film for Piezoelectric Nanogenerators. Materials. 2021; 14(6):1461. https://doi.org/10.3390/ma14061461
Chicago/Turabian StyleKim, Hyun Gi, Eun Hye Kim, and Sung Soo Kim. 2021. "Growth of ZnO Nanorods on ITO Film for Piezoelectric Nanogenerators" Materials 14, no. 6: 1461. https://doi.org/10.3390/ma14061461
APA StyleKim, H. G., Kim, E. H., & Kim, S. S. (2021). Growth of ZnO Nanorods on ITO Film for Piezoelectric Nanogenerators. Materials, 14(6), 1461. https://doi.org/10.3390/ma14061461





