Optical, Structural, and Dielectric Properties of Composites Based on Thermoplastic Polymers of the Polyolefin and Polyurethane Type and BaTiO3 Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. The Preparation of TPU:TPO Films in the Absence and Presence of BaTiO3 NPs
2.3. Microscopy Analysis
2.4. X-ray Diffraction Analysis
2.5. Fourier Transform Infrared (FTIR) Spectroscopic Analysis
2.6. FT-Raman Spectroscopic Analysis
2.7. Photoluminescence Analysis
2.8. Dielectric Properties
2.9. Differential Scanning Calorimetry (DSC) Analysis
3. Results and Discussions
3.1. Morphological and Structural Properties of the Composites Based on the TPU:TPO Blends and BaTiO3 NPs
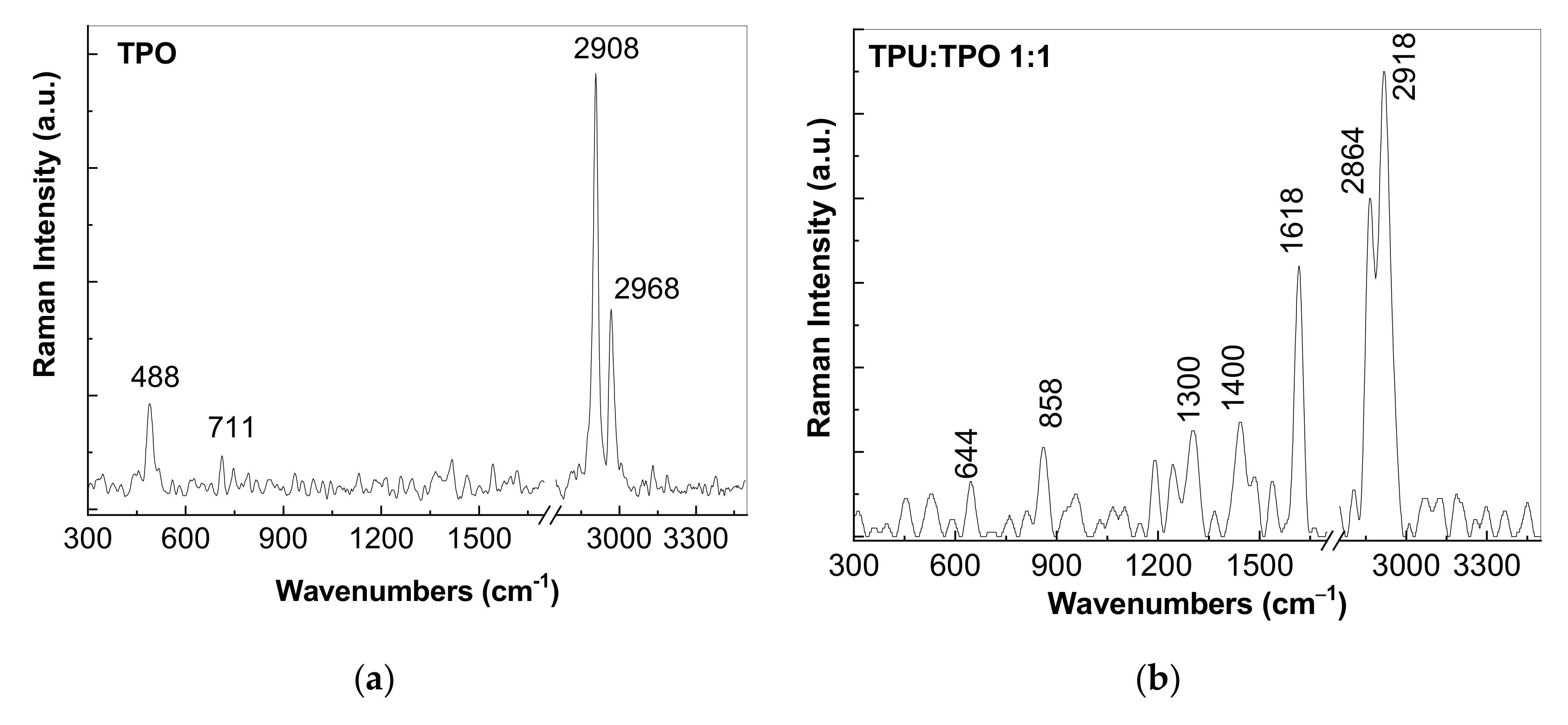
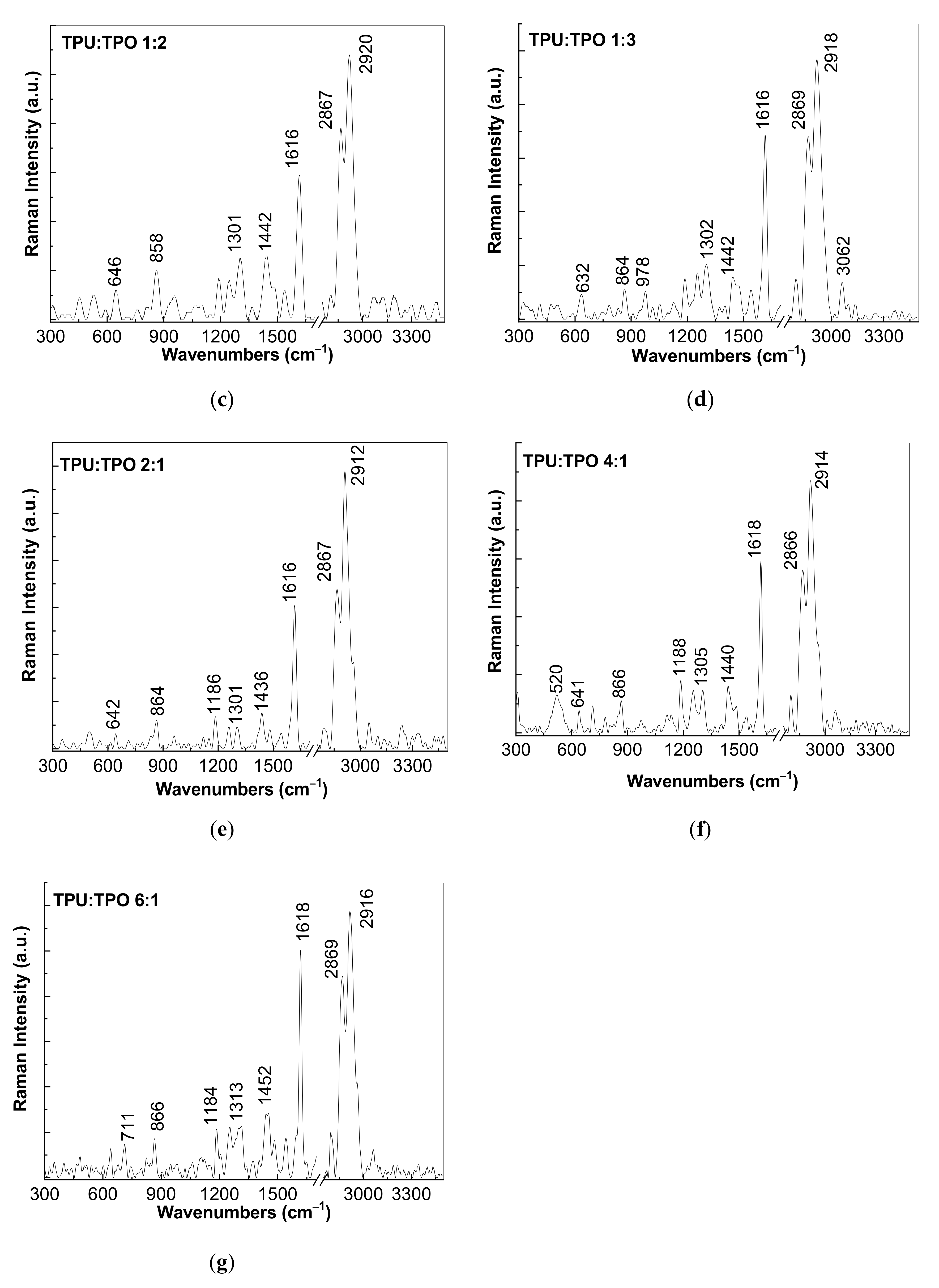
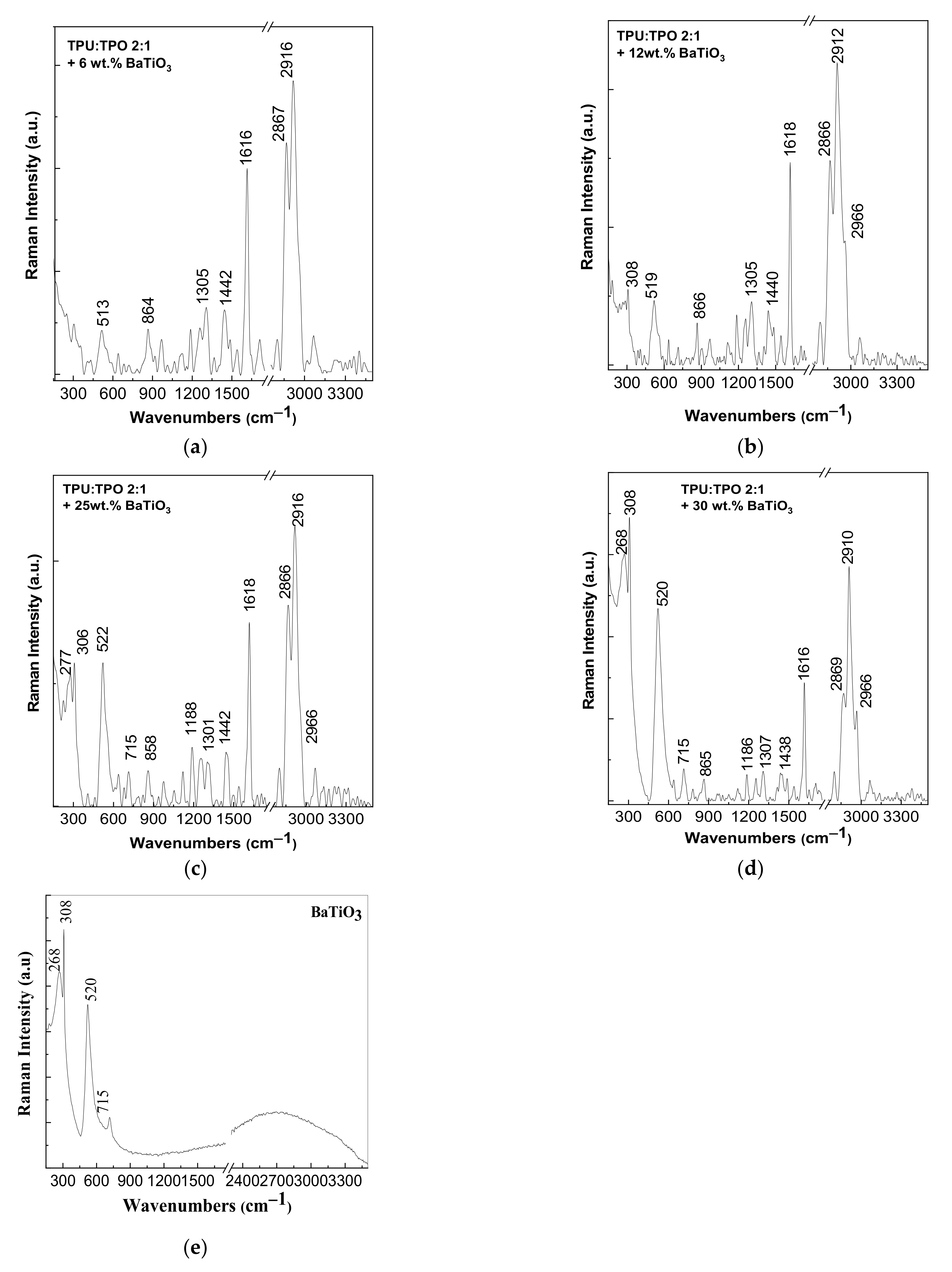
3.2. Vibrational Properties of the Composites Based on the TPU:TPO Blends and BaTiO3 NPs
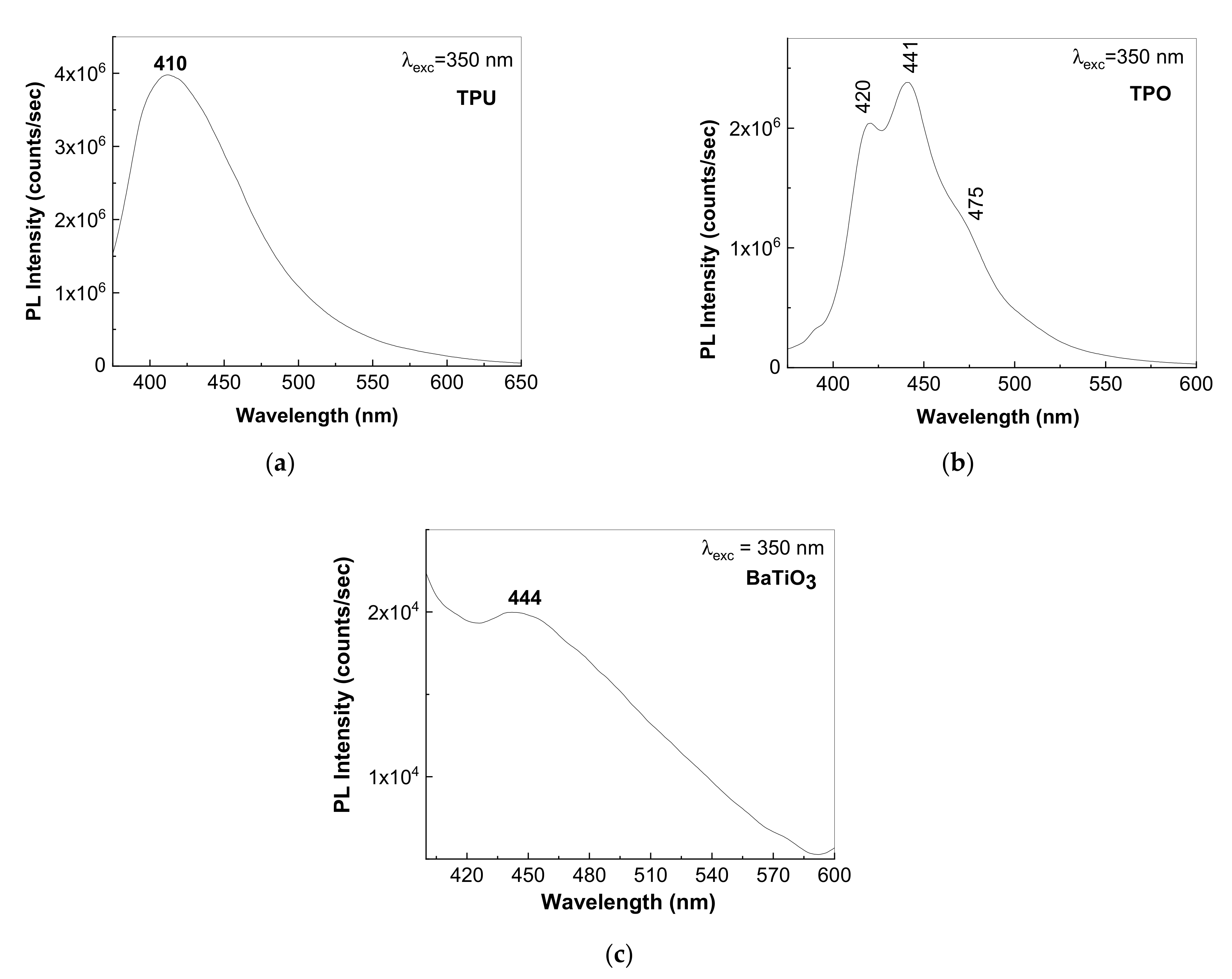
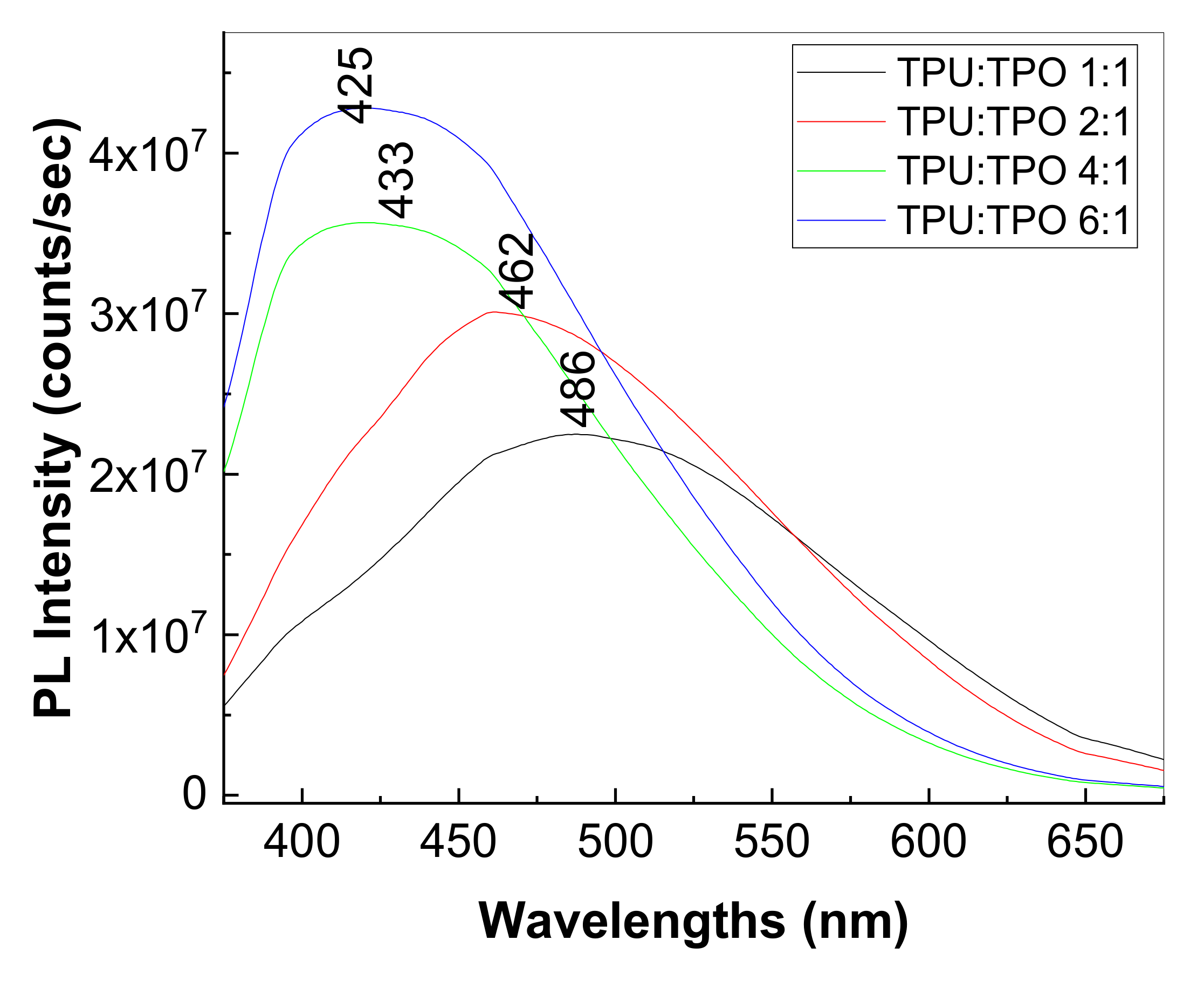
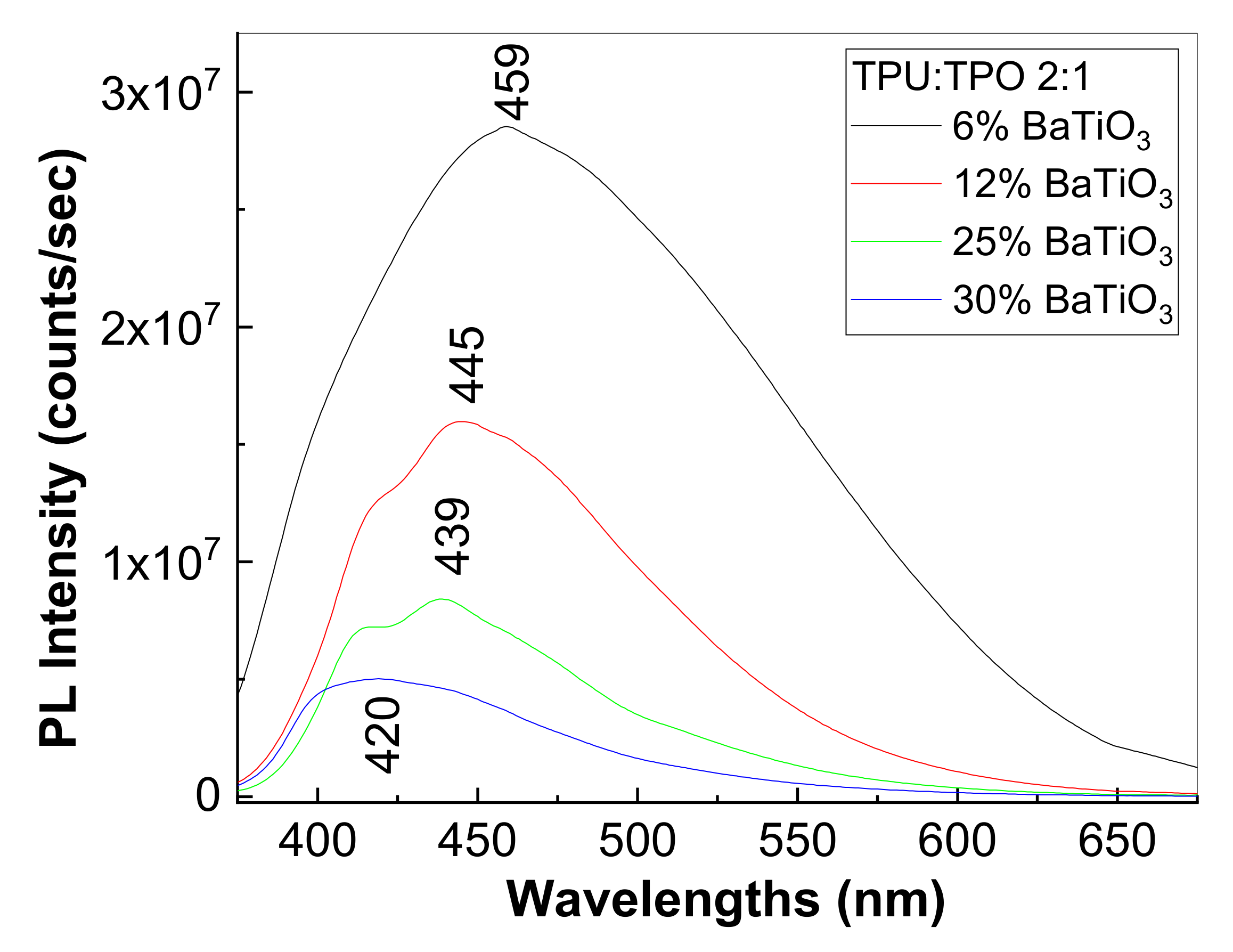
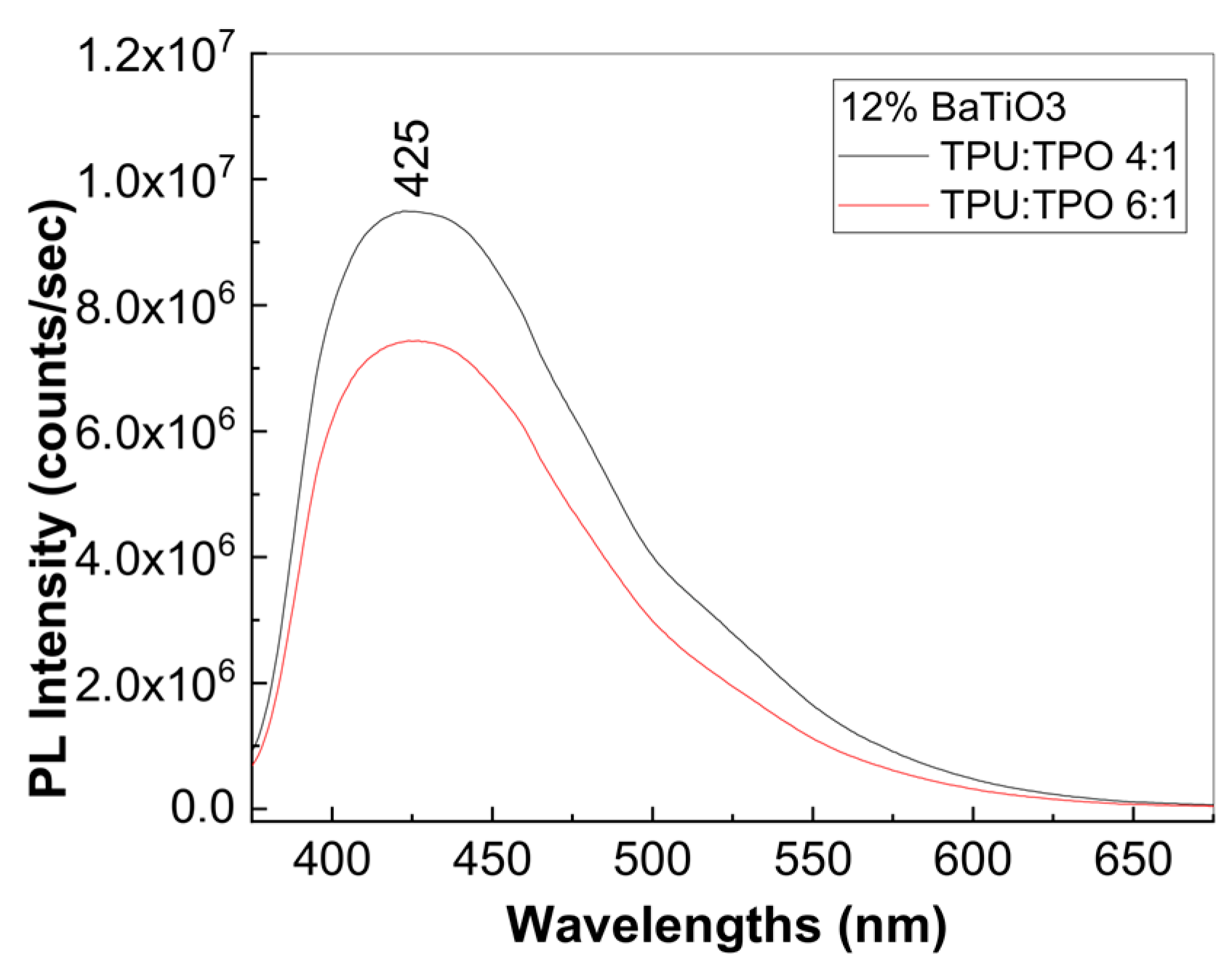
3.3. Photoluminescence Properties of the Composites Based on the TPU:TPO Blends and BaTiO3 NPs
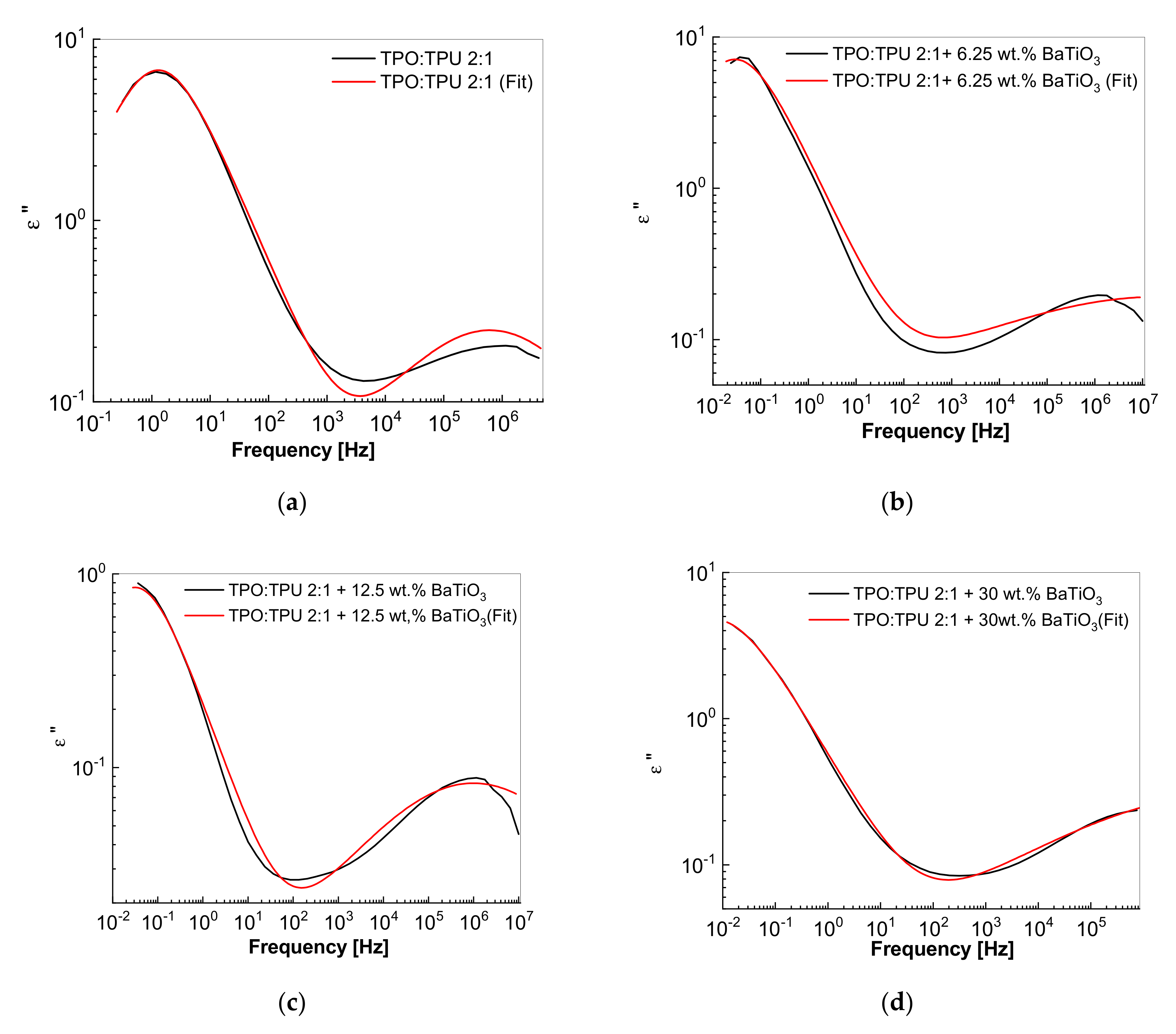
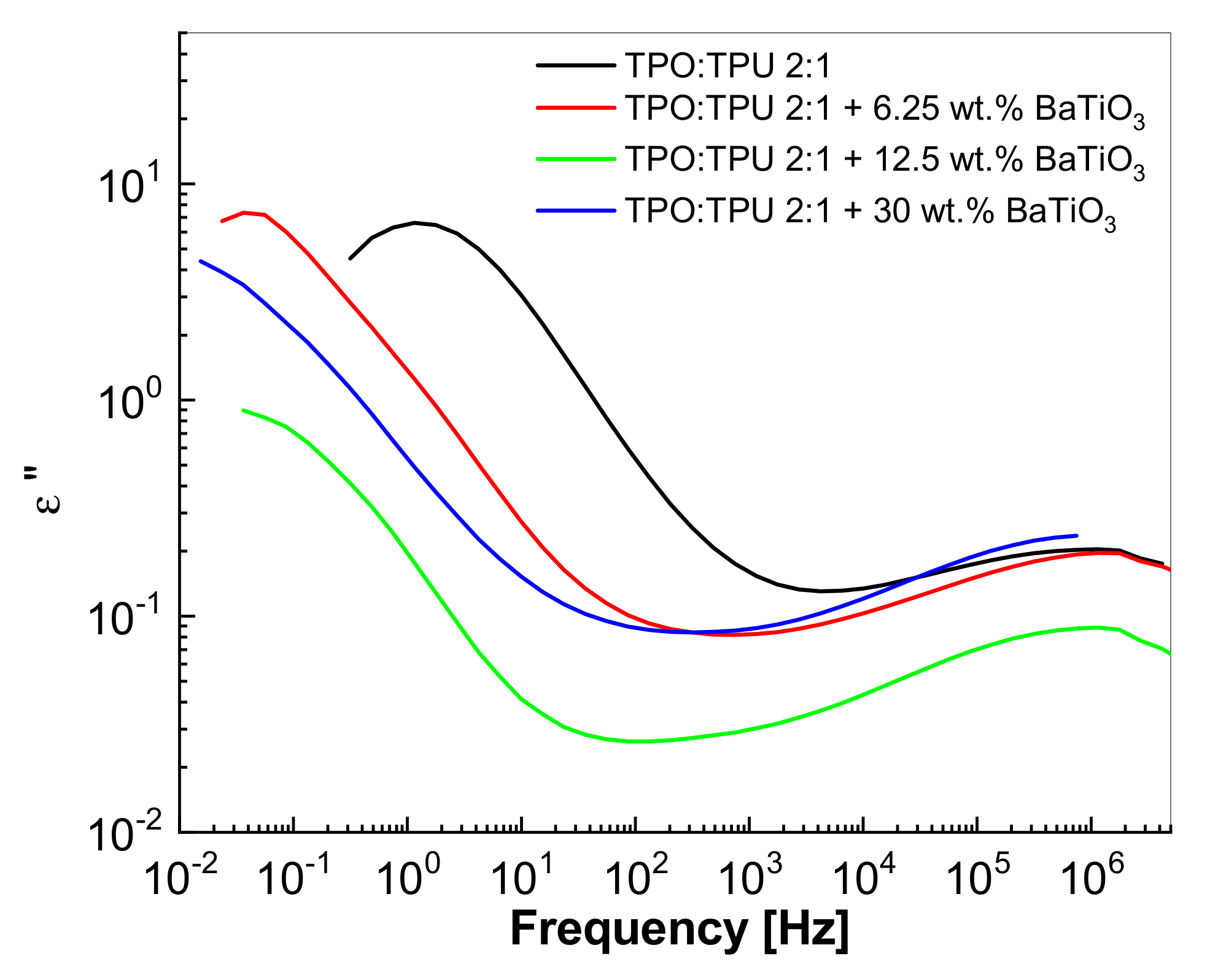
3.4. Dielectric Properties of the Composites Based on the TPU:TPO Blends and BaTiO3 NPs
3.5. Differential Scanning Calorimetry (DSC) Study of the Composites Based on the TPU:TPO Blends and BaTiO3 NPs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, T.; Ye, L.; Liu, Y.; Nie, F. Synthesis and properties of polyester-based TPUs prepared by solution polymerisation. Plast. Rubber Compos. 2008, 37, 331–340. [Google Scholar] [CrossRef]
- Akindoyo, J.O.; Beg, M.D.H.; Ghazali, S.; Islam, M.R.; Nitthiyah, J.; Yuvaraj, A.R. Polyurethane types, synthesis and applications—A review. RSC Adv. 2016, 6, 114453–114482. [Google Scholar] [CrossRef]
- Georgopoulou, A.; Sebastian, T.; Clemens, F. Thermoplastic elastomer composite filaments for strain sensing applications extruded with a fused deposition modelling 3D printer. Flex. Print. Electron. 2020, 5, 035002. [Google Scholar] [CrossRef]
- Rollo, G.; Ronca, A.; Cerruti, P.; Gan, X.; Fei, G.; Xia, H.; Gorokhov, G.; Bychanok, D.; Kuzhir, P.; Lavorgna, M.; et al. On the Synergistic Effect of Multi-Walled Carbon Nanotubes and Graphene Nanoplatelets to Enhance the Functional Properties of SLS 3D-Printed Elastomeric Structures. Polymers 2020, 12, 1841. [Google Scholar] [CrossRef] [PubMed]
- Reifler, F.A.; Sánchez, F.A.; Clemens, F.J.; Varga, K.; Hufenus, R. Flexible ceramic-reinforced polyurethane composite coatings on synthetic fibres: Process of continuous liquid film coating and its influence on the coating thickness. Compos. Sci. Technol. 2010, 70, 1207–1213. [Google Scholar] [CrossRef]
- Lavorgna, M.; Cerruti, P.; Casula, G.; Iannace, S.; Harper, J.F. Curing characteristics and mechanical properties of carbon fiber-interlayered fabric composites based on a polyurethane matrix. Adv. Polym. Technol. 2007, 26, 132–145. [Google Scholar] [CrossRef]
- Fernández-D’Arlas, B.; Khan, U.; Rueda, L.; Coleman, J.N.; Mondragon, I.; Corcuera, M.A.; Eceiza, A. Influence of hard segment content and nature on polyurethane/multiwalled carbon nanotube composites. Compos. Sci. Technol. 2010, 71, 1030–1038. [Google Scholar] [CrossRef]
- Hu, S.; Shou, T.; Guo, M.; Wang, R.; Wang, J.; Tian, H.; Qin, X.; Zhao, X.; Zhang, L. Fabrication of New Thermoplastic Polyurethane Elastomers with High Heat Resistance for 3D Printing Derived from 3,3-Dimethyl-4,4′-diphenyl Diisocyanate. Ind. Eng. Chem. Res. 2020, 59, 10476–10482. [Google Scholar] [CrossRef]
- Phua, S.L.; Yang, L.; Toh, C.L.; Huang, S.; Tsakadze, Z.; Lau, S.K.; Mai, Y.-W.; Lu, X. Reinforcement of Polyether Polyurethane with Dopamine-Modified Clay: The Role of Interfacial Hydrogen Bonding. ACS Appl. Mater. Interfaces 2012, 4, 4571–4578. [Google Scholar] [CrossRef]
- Zo, H.J.; Joo, S.H.; Kim, T.; Seo, P.S.; Kim, J.H.; Park, J.S. Enhanced mechanical and thermal properties of carbon fiber composites with polyamide and thermoplastic polyurethane blends. Fibers Polym. 2014, 15, 1071–1077. [Google Scholar] [CrossRef]
- Guo, Y.; Yan, L.; Zheng, Z.; Chen, L.; Ma, M.; Luo, R.; Bian, J.; Lin, H.; Chen, D. TPU/PLA nanocomposites with improved mechanical and shape memory properties fabricated via phase morphology control and incorporation of multi-walled carbon nanotubes nanofillers. Polym. Eng. Sci. 2020, 60, 1118–1128. [Google Scholar] [CrossRef]
- Qi, S.; Fu, J.; Xie, Y.; Li, Y.; Gan, R.; Yu, M. Versatile magnetorheological plastomer with 3D printability, switchable mechanics, shape memory, and self-healing capacity. Compos. Sci. Technol. 2019, 183, 107817. [Google Scholar] [CrossRef]
- Di, Y.; Kang, M.; Zhao, Y.; Yan, S.; Wang, X. Morphology and mechanical properties of blends of thermoplastic polyurethane and polyolefins. J. Appl. Polym. Sci. 2006, 99, 875–883. [Google Scholar] [CrossRef]
- Jeong, B.Y.; Choi, M.J.; Lee, W.-K.; Chun, J.H.; Cheon, J.M.; Ha, C.-S. Preliminary studies of polyurethane adhesive for thermoplastic polyolefins(TPOs) using polyolefin polyol. Mol. Cryst. Liq. Cryst. 2018, 660, 115–120. [Google Scholar] [CrossRef]
- Burnett, D.J.; Thielmann, F.; Ryntz, R.A. Correlating thermodynamic and mechanical adhesion phenomena for thermoplastic polyolefins. J. Coat. Technol. Res. 2007, 4, 211–215. [Google Scholar] [CrossRef]
- Deng, K.; Felorzabihi, N.; Winnik, M.A.; Jiang, Z.; Yin, Z.; Liu, Y.; Yaneff, P.V.; Ryntz, R.A. Influence of position and composition on adhesion to injection-molded TPO plaques as model automotive parts. Polymer 2009, 50, 5084–5093. [Google Scholar] [CrossRef]
- Kim, T.K.; Kim, B.K.; Lee, S.Y.; Cho, Y.L.; Kim, M.S.; Jeong, H.M. Thermoplastic polyurethane elastomer/thermoplastic polyolefin elastomer blends compatibilized with a polyolefinic segment in TPU. Macromol. Res. 2010, 18, 177–184. [Google Scholar] [CrossRef]
- Wang, J.S.; Chen, X.D.; Zhang, M.Q.; Rong, M.Z. Polyurethane/Polyolefin Blends: Morphology, Compatibilization and Mechanical Properties. Polym. Polym. Compos. 2006, 14, 1–11. [Google Scholar] [CrossRef]
- Lin, T.A.; Lou, C.-W.; Lin, J.-H. The Effects of Thermoplastic Polyurethane on the Structure and Mechanical Properties of Modified Polypropylene Blends. Appl. Sci. 2017, 7, 1254. [Google Scholar] [CrossRef]
- Fan, B.H.; Zha, J.W.; Wang, D.R.; Zhao, J.; Zhang, X.F.; Dang, Z.M. Preparation and dielectric behaviors of thermoplastic and thermosetting polymer nanocomposite films containing BaTiO3 nanoparticles with different diameters. Compos. Sci. Technol. 2013, 80, 66–72. [Google Scholar] [CrossRef]
- Lu, X.; Qu, H.; Skorobogatiy, M. Piezoelectric Micro- and Nanostructured Fibers Fabricated from Thermoplastic Nanocomposites Using a Fiber Drawing Technique: Comparative Study and Potential Applications. ACS Nano 2017, 11, 2103–2114. [Google Scholar] [CrossRef] [PubMed]
- Christ, J.F.; Aliheidari, N.; Ameli, A.; Pötschke, P. 3D printed highly elastic strain sensors of multiwalled carbon nanotube/thermoplastic polyurethane nanocomposites. Mater. Des. 2017, 131, 394–401. [Google Scholar] [CrossRef]
- Han, H.; Voisin, C.; Guillemet-Fritsch, S.; Dufour, P.; Tenailleau, C.; Turner, C.; Nino, J.C. Origin of colossal permittivity in BaTiO3 via broadband dielectric spectroscopy. J. Appl. Phys. 2013, 113, 024102. [Google Scholar] [CrossRef]
- Poudel, A.; Walsh, P.; Kennedy, J.; Thomas, K.; Lyons, J.G.; Coffey, A. Thermal, mechanical, dielectric, and morphological study of dielectric filler–based thermoplastic nanocomposites for electromechanical applications. J. Thermoplast. Compos. Mater. 2019, 32, 178–204. [Google Scholar] [CrossRef]
- Sanchez, F.A.; Redondo, M.; González-Benito, J. Influence of BaTiO3 submicrometric particles on the structure, morphology, and crystallization behavior of poly(vinylidene fluoride). J. Appl. Polym. Sci. 2015, 132, 41497–41507. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, X.; Ma, Z.; Zhang, C.; Ai, J.; Chen, P.; Yan, C.; Su, B.; Shi, Y. A Material Combination Concept to Realize 4D Printed Products with Newly Emerging Property/Functionality. Adv. Sci. 2020, 7, 1903208. [Google Scholar] [CrossRef]
- Drobny, J.G. Polyolefin-based thermoplastic elastomers. In Handbook of Thermoplastic Elastomers; Drobny, J.G., Ed.; Elsevier: Norwich, UK; New York, NY, USA, 2014; pp. 191–199. [Google Scholar] [CrossRef]
- Finnigan, B.; Martin, D.; Halley, P.; Truss, R.; Campbell, K. Morphology and properties of thermoplastic polyurethane composites incorporating hydrophobic layered silicates. J. Appl. Polym. Sci. 2005, 97, 300–309. [Google Scholar] [CrossRef]
- Baştürk, E.; Madakbaş, S.; Kahraman, M.V. Improved Thermal Stability and Wettability Behavior of Thermoplastic Polyurethane/Barium Metaborate Composites. Mater. Res. 2016, 19, 434–439. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Narumi, K.; Naramoto, H.; Wu, Z.P.; Yamamoto, S.; Miyashita, A.; Tamada, M. A crystalline hydrogenated carbon film obtained by plasma enhanced chemical vapor deposition. J. Appl. Phys. 1999, 86, 1317–1321. [Google Scholar] [CrossRef]
- Archie, L.; Robbins, L.; Mills, K.R. Compositions Comprising an Olefin Polymer, Carbon Black and Dithiobisben Zothazole and Process for Curing Same. Patent No. 770582, 1962. [Google Scholar]
- Morris, H.R.; Turner, J.F.; Munro, B.; Ryntz, R.A.; Treado, P.J. Chemical Imaging of Thermoplastic Olefin (TPO) Surface Architecture. Langmuir 1999, 15, 2961–2972. [Google Scholar] [CrossRef]
- Parnell, S.; Min, K.; Cakmak, M. Kinetic studies of polyurethane polymerization with Raman spectroscopy. Polymer 2003, 44, 5137–5144. [Google Scholar] [CrossRef]
- Strenkoski, M.; Lodarczyk, D.; Piszczyk, L.; Strankowska, J. Polyurethane Nanocomposites Containing Reduced Graphene Oxide, FTIR, Raman, and XRD Studies. J. Spectrosc. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Venkateswaran, U.D.; Naik, V.M.; Naik, R.R. High-pressure Raman studies of polycrystalline BaTiO3. Phys. Rev. B 1998, 58, 14256–14260. [Google Scholar] [CrossRef]
- Pennington, B.D.; Ryntz, R.A.; Urban, M.W. Stratification in thermoplastic olefins (TPO); photoacoustic FT-IR depth profiling studies. Polymer 1999, 40, 4795–4803. [Google Scholar] [CrossRef]
- Gopanna, A.; Mandapati, R.N.; Thomas, S.P.; Rajan, K.P.; Chavali, M. Fourier transform infrared spectroscopy (FTIR), Raman spectroscopy and wide-angle X-ray scattering (WAXS) of polypropylene (PP)/cyclic olefin copolymer (COC) blends for qualitative and quantitative analysis. Polym. Bull. 2019, 76, 4259–4274. [Google Scholar] [CrossRef]
- Ferry, A.; Jacobsson, P.; Van Heumen, J.; Stevens, J. Raman, infra-red and d.s.c. studies of lithium coordination in a thermoplastic polyurethane. Polymer 1996, 37, 737–744. [Google Scholar] [CrossRef]
- Requena, S.; Lacoul, S.; Strzhemechny, Y.M. Luminescent Properties of Surface Functionalized BaTiO3 Embedded in Poly(methyl methacrylate). Materials 2014, 7, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Pattamaprom, C.; Wu, C.-H.; Chen, P.-H.; Huang, Y.-L.; Ranganathan, P.; Rwei, S.-P.; Chuan, F.-S. Solvent-Free One-Shot Synthesis of Thermoplastic Polyurethane Based on Bio-Poly(1,3-propylene succinate) Glycol with Temperature-Sensitive Shape Memory Behavior. ACS Omega 2020, 5, 4058–4066. [Google Scholar] [CrossRef] [PubMed]
- Şen, F.; Baştürk, E.; Karadoğan, B.; Madakbaş, S.; Kahraman, M.V. Effect of Barium Titanate on the Thermal, Morphology, Surface, and Mechanical Properties of the Thermoplastic Polyurethane/Barium Titanate Composites. Polym. Plast. Technol. Eng. 2016, 55, 1325–1331. [Google Scholar] [CrossRef]
- Nejad, S.S.; Babaie, A.; Bagheri, M.; Rezaei, M.; Abbasi, F.; Shomali, A. Effects of graphene quantum dot (GQD) on photoluminescence, mechanical, thermal and shape memory properties of thermoplastic polyurethane nanocomposites. Polym. Adv. Technol. 2020, 31, 2279–2289. [Google Scholar] [CrossRef]
- Ning, L.; De-Ning, W.; Sheng-Kang, Y. Crystallinity and hydrogen bonding of hard segments in segmented poly(urethane urea) copolymers. Polymer 1996, 37, 3577–3583. [Google Scholar] [CrossRef]
- Song, J.; Batra, A.; Rego, J.M.; Macosko, C.W. Polyethylene/polyurethane blends for improved paint adhesion. Prog. Org. Coat. 2011, 72, 492–497. [Google Scholar] [CrossRef]
- Mattia, J.; Painter, P. A Comparison of hydrogen bonding and order in a polyurethane and poly(urethane−urea) and their blends with poly(ethylene glycol). Macromolecules 2007, 40, 1546–1554. [Google Scholar] [CrossRef]
- Allen, N.S.; McKellar, J.F. Photochemical reactions in an MDI-based elastomeric polyurethane. J. Appl. Polym. Sci. 1976, 20, 1441–1447. [Google Scholar] [CrossRef]
- Arai, T.; Hosobuchi, M.; Fuse, N.; Takeda, K.; Ohki, Y. Optical Characterization and Computational Chemical Evaluation of Electronic Localized States in Polyolefin. Electr. Eng. Jpn. 2014, 188, 1–8. [Google Scholar] [CrossRef]
- Ito, T.; Fuse, N.; Ohki, Y. Effects of Additives, Photodegradation, and Water-tree Degradation on the Photoluminescence in Polyethylene and Polypropylene. IEEJ Trans. Fundam. Mater. 2004, 124, 624–630. [Google Scholar] [CrossRef]
- Rastogi, M.; Kushwaha, H.S.; Vaish, R. Highly efficient visible light mediated azo dye degradation through barium titanate decorated reduced graphene oxide sheets. Electron. Mater. Lett. 2016, 12, 281–289. [Google Scholar] [CrossRef]
- Orhan, E.; Varela, J.A.; Zenatti, A.; Gurgel, M.F.C.; Pontes, F.M.; Leite, E.R.; Longo, E.; Pizani, P.S.; Beltrán, A.; Andrés, J. Room-temperature photoluminescence of BaTiO3: Joint experimental and theoretical study. Phys. Rev. B 2005, 71, 085113. [Google Scholar] [CrossRef]
- Yan, J.-J.; Wang, Z.-K.; Lin, X.-S.; Hong, C.-Y.; Liang, H.-J.; Pan, C.-Y.; You, Y.-Z. Polymerizing Nonfluorescent Monomers without Incorporating and Flurescent Agent Produces Strong Fluerescent Polymers. Adv. Mater. 2012, 24, 5617–5624. [Google Scholar] [CrossRef]
- Havriliak, S.; Negami, S. A complex plane representation of dielectric and mechanical relaxation processes in some polymers. Polymer 1967, 8, 161–210. [Google Scholar] [CrossRef]
- Schönhals, A. Broadband Dielectric Spectroscopy; Kremer, F., Schönhals, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Urban, S.; Gestblom, B.; Kresse, H.; Dabrowski, A. Dielectric relaxations studies of 4-n-alkyloxy-4′-cyanobiphenyls (nOCB, n = 5 divided by 8). Z. Nat. A. 1996, 51, 834–842. [Google Scholar]
- Frunză, Ş.; Schönhals, A.; Frunza, L.; Beica, T.; Zgura, I.; Ganea, P.; Stoenescu, D. Dynamics of cyanophenyl alkylbenzoate molecules in the bulk and in a surface layer adsorbed onto aerosil. Variation of the alkyl chain. Chem. Phys. 2010, 372, 51–60. [Google Scholar] [CrossRef]
- Yang, J.; Deng, X.Y.; Li, J.B.; Cai, Q.; Zhang, H.T.; Wang, L.M.; Su, K.F.; Zhang, G.Q.; Wang, C. Broadband Dielectric Spectroscopy Analysis of Dielectric Properties of Barium Titanate Ceramics. Adv. Mater. Res. 2013, 744, 323–328. [Google Scholar] [CrossRef]
- Mančić, D.; Paunović, V.; Vijatović, M.; Stojanović, B.; Živković, L. Electrical Characterization and Impedance Response of Lanthanum Doped Barium Titanate Ceramics. Sci. Sint. 2008, 40, 283–294. [Google Scholar] [CrossRef]
- Park, J.H.; Yoo, D.H.; Kim, C.S.; Yang, H.S.; Moon, B.K. Synthesis, Structure and Dielectric Properties of BaTiO3 Nanoparticles. J. Korean Phys. Soc. 2006, 49, S680–S683. [Google Scholar]
- Sareecha, N.; Shah, W.A.; Anisur-Rehman, M.; Mirza, M.L.; Awan, M.S. Electrical investigations of BaTiO3 ceramics with Ba/Ti contents under influence of temperature. Solid State Ion 2017, 303, 16–23. [Google Scholar] [CrossRef]









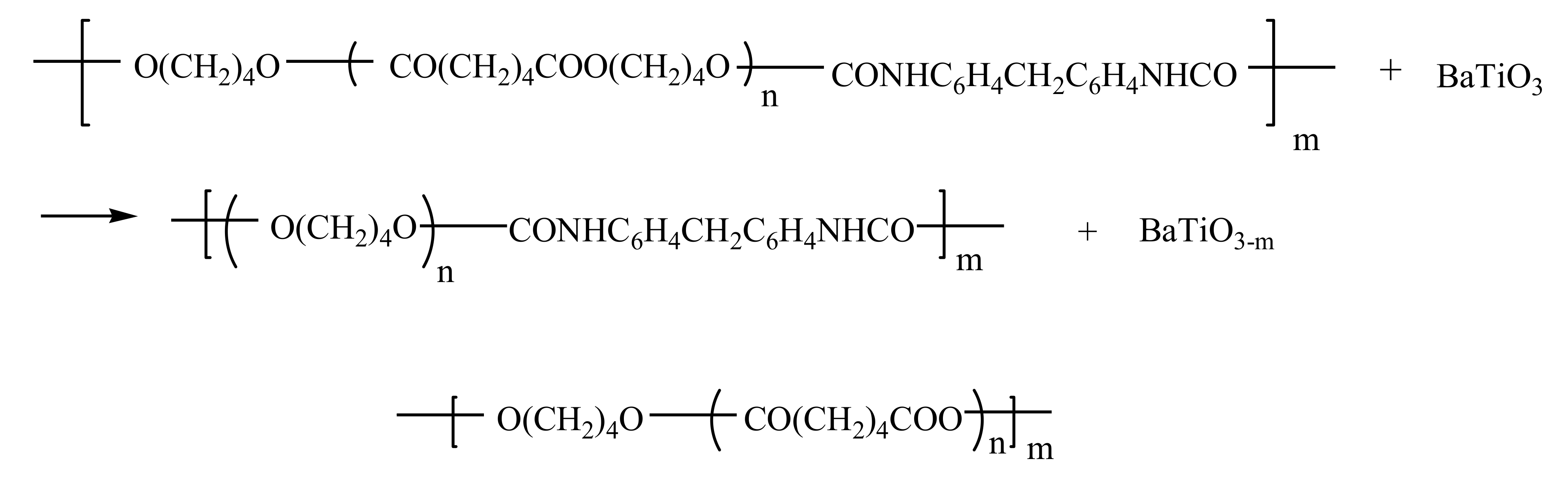

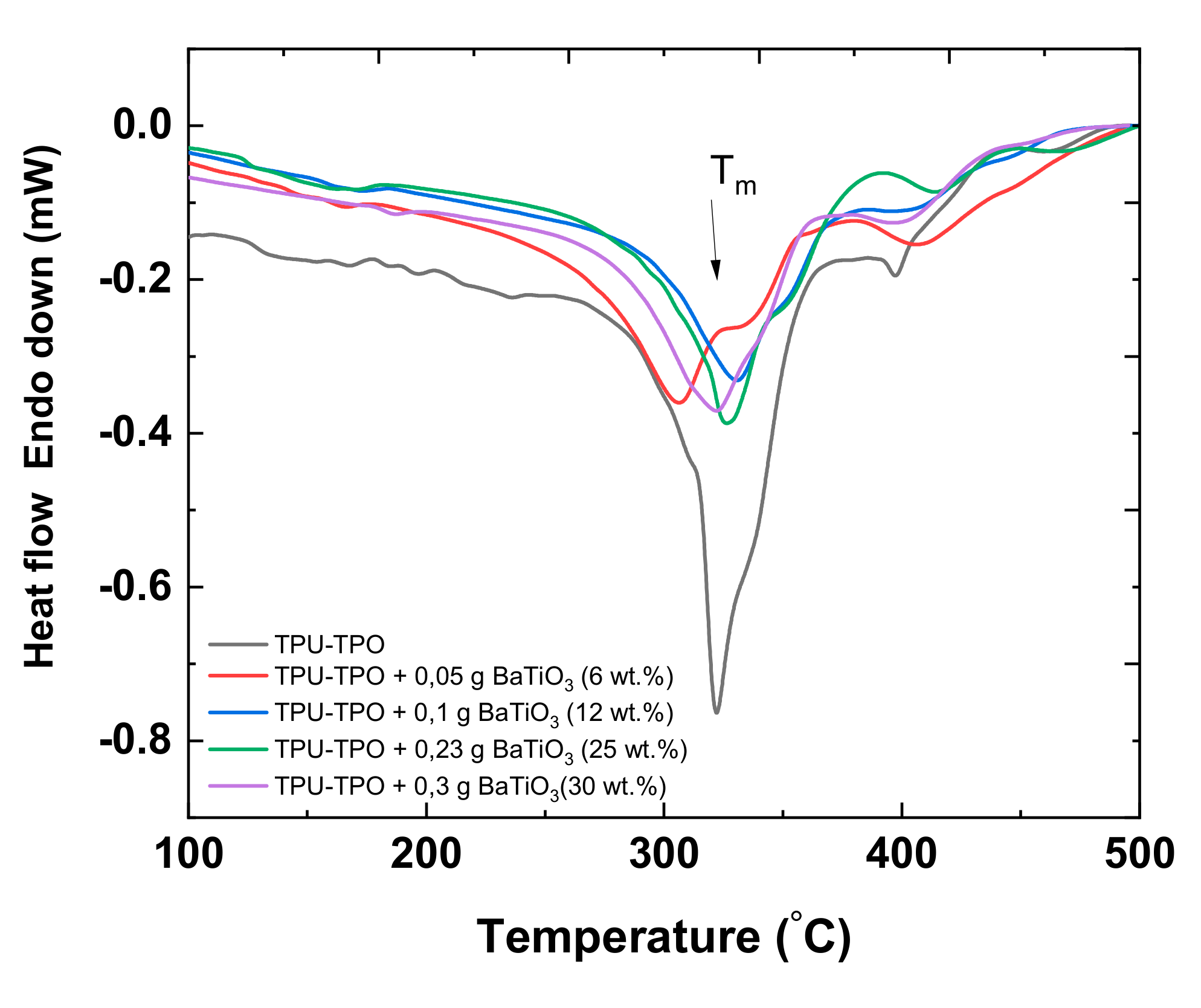
| Sample | Melting Temperature (Max.) [°C] | Enthalpy (ΔH) [J g−1] |
|---|---|---|
| TPU:TPO | 322.1 | 76.37 |
| TPU:TPO + 6.25 wt.% BaTiO3 | 306.5 | 36.20 |
| TPU:TPO + 12 wt.% BaTiO3 | 330.4 | 33.14 |
| TPU:TPO + 25 wt.% BaTiO3 | 325.8 | 38.70 |
| TPU:TPO + 30 wt.% BaTiO3 | 326.8 | 47.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baibarac, M.; Nila, A.; Smaranda, I.; Stroe, M.; Stingescu, L.; Cristea, M.; Cercel, R.C.; Lorinczi, A.; Ganea, P.; Mercioniu, I.; et al. Optical, Structural, and Dielectric Properties of Composites Based on Thermoplastic Polymers of the Polyolefin and Polyurethane Type and BaTiO3 Nanoparticles. Materials 2021, 14, 753. https://doi.org/10.3390/ma14040753
Baibarac M, Nila A, Smaranda I, Stroe M, Stingescu L, Cristea M, Cercel RC, Lorinczi A, Ganea P, Mercioniu I, et al. Optical, Structural, and Dielectric Properties of Composites Based on Thermoplastic Polymers of the Polyolefin and Polyurethane Type and BaTiO3 Nanoparticles. Materials. 2021; 14(4):753. https://doi.org/10.3390/ma14040753
Chicago/Turabian StyleBaibarac, M., A. Nila, I. Smaranda, M. Stroe, L. Stingescu, M. Cristea, R. C. Cercel, A. Lorinczi, P. Ganea, I. Mercioniu, and et al. 2021. "Optical, Structural, and Dielectric Properties of Composites Based on Thermoplastic Polymers of the Polyolefin and Polyurethane Type and BaTiO3 Nanoparticles" Materials 14, no. 4: 753. https://doi.org/10.3390/ma14040753
APA StyleBaibarac, M., Nila, A., Smaranda, I., Stroe, M., Stingescu, L., Cristea, M., Cercel, R. C., Lorinczi, A., Ganea, P., Mercioniu, I., Ciobanu, R., Schreiner, C., Garcia, R. G., & Bartha, C. (2021). Optical, Structural, and Dielectric Properties of Composites Based on Thermoplastic Polymers of the Polyolefin and Polyurethane Type and BaTiO3 Nanoparticles. Materials, 14(4), 753. https://doi.org/10.3390/ma14040753








