Numerical Study of Joule Heating Effects on Microfluidics Device Reliability in Electrode Based Devices
Abstract
1. Introduction
2. Materials and Methods
2.1. Theory of DEP
2.2. Numerical Modeling for Joule Heating
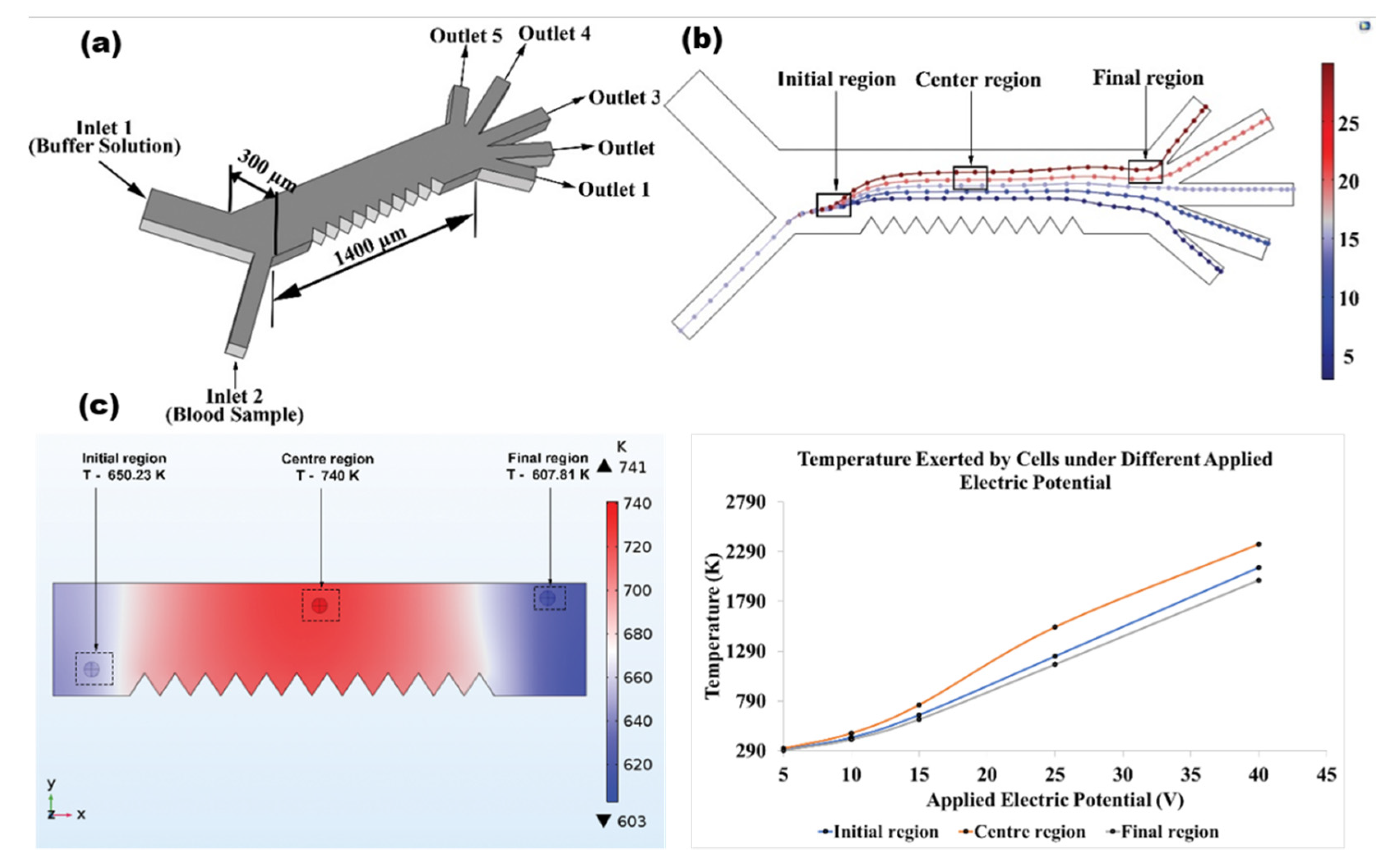
2.3. Device Design and Simulation Studies
- (i)
- Electric current interface is used to generate non-uniform electric field within the microchannel.
- (ii)
- (Bio-heat transfer interface in simulation is used to generate the thermal distribution on the blood cells with boundaries set to room temperature of 290 K. Laminar flow interface is used to enable fluid flow at desired velocity from both inlets (inlet 1 and inlet 2).
- (iii)
- A time-dependent solver is used to solve electric field and thermal distribution that provides understanding of the results through joule heating simultaneously.
3. Results and Discussions
3.1. Contribution of Electrode Shapes in Producing Optimum Joule Heating
3.2. Choice of Electrode Materials Affects Reliablity of the Device
3.3. Change in Buffer Conductivity Affects Survival Time of the Cells
4. Conclusions
- The triangular electrode is found to be effective in generating low thermal gradient at the center region, while maintaining microfluidic device reliable for separation, even at applied potential varied between 10 V and 40 V.
- Among different electrode materials considered, we found that copper generates low thermal gradient compared to other materials. However, copper electrodes degrades and destroys the channel as opposed to gold and platinum, and hinders its application for multiple runs.
- The device material fabricated using glass substrate has potential to generate least amount of joule heating, 313 K, compared to other substrate materials (PDMS, PMMA, polyimide) thereby increasing throughput of the separation system.
- With buffer conductivity of 10 [mS/m] and applied potential of 5 V, the survival time of cells inside microchannel is found to be 1.45 s. This duration is sufficient enough to selectively deflect the cells to target outlets. However, survival time was found to be decreasing with increasing buffer conductivity (upto 55 [mS/m]) making the device less reliable.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yousuff, C.M.; Ho, E.T.W.; Hussain K., I.; Hamid, N.H.B. Microfluidic Platform for Cell Isolation and Manipulation Based on Cell Properties. Micromachines 2017, 8, 15. [Google Scholar] [CrossRef]
- Rahman, N.A.; Ibrahim, F.; Yafouz, B. Dielectrophoresis for Biomedical Sciences Applications: A Review. Sensors 2017, 17, 449. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Wu, H. Joule Heating and Chip Materials. In Encyclopedia of Microfluidics and Nanofluidics; Springer: Manhattan, NY, USA, 2014; pp. 1–15. [Google Scholar]
- Rashed, M.Z.; Green, N.G.; Williams, S.J. Scaling law analysis of electrohydrodynamics and dielectrophoresis for isomotive dielectrophoresis microfluidic devices. Electrophoresis 2020, 41, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.Y.; Yan, D.G.; Yang, C.; Gong, H.Q.; Chai, C.J.; Lam, Y.C. Joule heating and its effects on electroosmotic flow in microfluidic channels. J. Phys. Conf. Ser. 2006, 34, 925–930. [Google Scholar] [CrossRef]
- Punjiya, M.; Nejad, H.R.; Mathews, J.; Levin, M.; Sonkusale, S. A flow through device for simultaneous dielectrophoretic cell trapping and AC electroporation. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kunti, G.; Dhar, J.; Bhattacharya, A.; Chakraborty, S. Joule heating-induced particle manipulation on a microfluidic chip. Biomicrofluidics 2019, 13, 014113. [Google Scholar] [CrossRef]
- Velve-Casquillas, G.; Le Berre, M.; Piel, M.; Tran, P.T. Microfluidic tools for cell biological research. Nano Today 2010, 5, 28–47. [Google Scholar] [CrossRef]
- Valero, A.; Braschler, T.; Demierre, N.; Renaud, P. A miniaturized continuous dielectrophoretic cell sorter and its applications. Biomicrofluidics 2010, 4, 022807. [Google Scholar] [CrossRef]
- Kausar, A.; Taherian, R. Effect of External Fields on Electrical Conductivity of Polymer-Based Composites; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Song, S.H.; Kwak, B.S.; Park, J.S.; Kim, W.; Jung, H.I.L. Novel application of Joule heating to maintain biocompatible temperatures in a fully integrated electromagnetic cell sorting system. Sens. Actuators A Phys. 2009, 151, 64–70. [Google Scholar] [CrossRef]
- Pearce, T.M.; Wilson, J.A.; Oakesm, S.G.; Chiu, S.Y.; Williams, J.C. Integrated microelectrode array and microfluidics for temperature clamp of sensory neurons in culture. Lab Chip 2005, 5, 97–101. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Zhao, S.; Zhou, Y.; He, W.; Zhang, Y.; Deng, W. The analysis of viability for mammalian cells treated at different temperatures and its application in cell shipment. PLoS ONE 2017, 12, 1–16. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Z.; Cui, X.; Jiang, L.; Zhi, Y.; Ding, X.; Nie, Z.; Zhou, P.; Cui, D. Microfluidic Device Directly Fabricated on Screen-Printed Electrodes for Ultrasensitive Electrochemical Sensing of PSA. Nanoscale Res. Lett. 2019, 14, 1–6. [Google Scholar] [CrossRef]
- Chen, L.; Liu, X.; Zheng, X.; Zhang, X.; Yang, J.; Tian, T.; Liao, Y. Dielectrophoretic Separation of Particles Using Microfluidic Chip with Composite Three-Dimensional Electrode. Micromachines 2020, 11, 700. [Google Scholar] [CrossRef]
- Vlassakis, J.; Herr, A.E. Joule Heating-Induced Dispersion in Open Microfluidic Electrophoretic Cytometry. Anal. Chem. 2017, 89, 12787–12796. [Google Scholar] [CrossRef] [PubMed]
- Pethig, R. Dielectrophoresis: Status of the theory, technology, and applications. Biomicrofluidics 2010, 4. [Google Scholar] [CrossRef] [PubMed]
- Del Rosal, B.; Sun, C.; Loufakis, D.N.; Lu, C.; Jaque, D. Thermal loading in flow-through electroporation microfluidic devices. Lab Chip 2013, 13, 3119–3127. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Adekanmbi, E.O.; Srivastava, S.K. Dielectrophoretic applications for disease diagnostics using lab-on-a-chip platforms. Lab Chip 2016, 16, 2148–2167. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, D.R.; Sah, R.L.; Bhatia, S.N. Geometric and Material Determinants of Patterning Efficiency by Dielectrophoresis. Biophys. J. 2004, 87, 2131–2147. [Google Scholar] [CrossRef][Green Version]
- Sarno, B.; Heineck, D.; Heller, M.J.; Ibsen, S. Dielectrophoresis: Developments and applications from 2010 to 2020. Electrophoresis. 2020, 1–54. [Google Scholar] [CrossRef] [PubMed]
- Ansar, B.I.M.Z.; Tirth, V.; Yousuff, C.M.; Shukla, N.K.; Islam, S.; Irshad, K.; Aarif, K.O.M. Simulation Guided Microfluidic Design for Multitarget Separation Using Dielectrophoretic Principle. BioChip J. 2020, 14, 390–404. [Google Scholar] [CrossRef]
- Ansar, M.Z.; Caffiyar, M.Y.; Kashif, A.M. Microfluidic device for Multitarget separation using DEP techniques and its applications in clinical research. In 2020 6th International Conference on Bio Signals, Images, Instrumentation, ICBSII 2020; Institute of Electrical and Electronics Engineers Inc.: New York, NY, USA, 2020. [Google Scholar]
- Erickson, D.; Sinton, D.; Li, D. Joule heating and heat transfer in poly(dimethylsiloxane) microfluidic systems. Lab Chip 2003, 3, 141–149. [Google Scholar] [CrossRef]
- Chein, R.; Yang, Y.C.; Lin, Y. Estimation of Joule heating effect on temperature and pressure distribution in electrokinetic-driven microchannel flows. Electrophoresis 2006, 27, 640–649. [Google Scholar] [CrossRef]
- Nedelcu, O.T. A thermal study on joule-heating induced effects in dielectrophoretic microfilters. Rom. J. Inf. Sci. Technol. 2011, 14, 309–323. [Google Scholar]
- Zhu, Y.; Nahavandi, S.; Bui, A.; Petkovic-Duran, K. Joule heating in polymer microfluidic chip. In BioMEMS Nanotechnol, II; Springer: Manhattan, NY, USA, 2005; p. 603612. [Google Scholar]
- Brans, T.; Strubbe, F.; Schreuer, C.; Vandewiele, S.; Neyts, K.; Beunis, F. Joule heating monitoring in a microfluidic channel by observing the Brownian motion of an optically trapped microsphere. Electrophoresis 2015, 36, 2102–2109. [Google Scholar] [CrossRef]
- Wang, X.-B.; Yang, J.; Huang, Y.; Vykoukal, J.; Becker, F.F.; Gascoyne, P.R.C. Cell Separation by Dielectrophoretic Field-flow-fractionation. Anal. Chem. 2000, 72, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.S.W.; Mitchell, P.D.; Oreffo, R.O.C.; Morgan, H.; Green, N.G. Image-based sorting and negative dielectrophoresis for high purity cell and particle separation. Electrophoresis 2019, 40, 2718–2727. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.A. Dielectrophoresis of proteins: Experimental data and evolving theory. Anal. Bioanal. Chem. 2020, 412, 3801–3811. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-H.; Chang, W.-H. Effect of Electrode Shape on Impedance of Single HeLa Cell: A COMSOL Simulation. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Dewhirst, M.; Viglianti, B.L.; Lora-Michiels, M.; Hoopes, P.J.; Hanson, M.A. Thermal dose requirement for tissue effect: Experimental and clinical findings. In Thermal Treatment of Tissue: Energy Delivery and Assessment II; SPIE: Bellingham, WA, USA, 2003; p. 37. [Google Scholar]
- Yousuff, C.M.; Hamid, N.H.B.; Hussain, K.I.; Ho, E.T.W. Numerical modelling and simulation of dielectrophoretic based WBC sorting using sidewall electrodes. In Proceedings of the 6th International Conference on Intelligent and Advanced Systems (ICIAS), Kuala Lumpur, Malaysia, 15–17 August 2016; IEEE: Piscataway, NJ, USA, 2017; pp. 1–5. [Google Scholar]
- Zhang, H.; Chang, H.; Neuzil, P. DEP-on-a-chip: Dielectrophoresis applied to microfluidic platforms. Micromachines 2019, 10, 423. [Google Scholar] [CrossRef]
- Jaeger, M.S.; Mueller, T.; Schnelle, T. Thermometry in dielectrophoresis chips for contact-free cell handling. J. Phys. D Appl. Phys. 2007, 40, 95–105. [Google Scholar] [CrossRef]
- Jiang, T.; Ren, Y.; Liu, W.; Tang, D.; Tao, Y.; Xue, R.; Jiang, H. Dielectrophoretic separation with a floating-electrode array embedded in microfabricated fluidic networks. Phys. Fluids 2018, 30, 112003. [Google Scholar] [CrossRef]
- Chen, H.; Cao, B.; Sun, B.; Cao, Y.; Yang, K.; Lin, Y.S.; Chen, H. Highly-sensitive capture of circulating tumor cells using micro-ellipse filters. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Constantin, C.P.; Aflori, M.; Damian, R.F.; Rusu, R.D. Biocompatibility of Polyimides: A Mini-Review. Materials 2019, 12, 3166. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yousuff, C.M.; Tirth, V.; Zackria Ansar Babu Irshad, M.; Irshad, K.; Algahtani, A.; Islam, S. Numerical Study of Joule Heating Effects on Microfluidics Device Reliability in Electrode Based Devices. Materials 2021, 14, 5819. https://doi.org/10.3390/ma14195819
Yousuff CM, Tirth V, Zackria Ansar Babu Irshad M, Irshad K, Algahtani A, Islam S. Numerical Study of Joule Heating Effects on Microfluidics Device Reliability in Electrode Based Devices. Materials. 2021; 14(19):5819. https://doi.org/10.3390/ma14195819
Chicago/Turabian StyleYousuff, Caffiyar Mohammed, Vineet Tirth, Mohamed Zackria Ansar Babu Irshad, Kashif Irshad, Ali Algahtani, and Saiful Islam. 2021. "Numerical Study of Joule Heating Effects on Microfluidics Device Reliability in Electrode Based Devices" Materials 14, no. 19: 5819. https://doi.org/10.3390/ma14195819
APA StyleYousuff, C. M., Tirth, V., Zackria Ansar Babu Irshad, M., Irshad, K., Algahtani, A., & Islam, S. (2021). Numerical Study of Joule Heating Effects on Microfluidics Device Reliability in Electrode Based Devices. Materials, 14(19), 5819. https://doi.org/10.3390/ma14195819





